1. Introduction
Legumes are multipurpose crops and consumed either directly as feed or as foodstuff in various processed forms in many countries worldwide [
1]. They are of great interest from a nutritional point of view because of their adequate proportions of starch, proteins, carbohydrates, fibers, fats, vitamins (niacin, riboflavin, thiamine, vitamin B6), and minerals (iron, zinc, calcium, magnesium), and contain many bioactive compounds [
2,
3]. They are considered an excellent source of dietary protein (15.7–31.5%) due to their high biological value, protein bioavailability, and well-balanced amino acid profile [
4,
5,
6]. After cereals, legumes are the most important food and are considered worldwide as a meat alternative because of their low price and sustainability. Despite their high nutritional value, legumes also contain antinutritional components (proteins such as allergens, enzyme inhibitors such as Kunitz-type trypsin inhibitor, Bowman–Birk-type chymotrypsin inhibitor, amylase inhibitor, lectins; phytate such as phytic acid, polyphenols such as tannins, saponins, oligosaccharides such as raffinose, stachyose) that may interfere with nutrient bioavailability. can affect protein digestibility and mineral absorption. Therefore, the nutritional quality of legumes should be evaluated not only based on their nutrient composition but also by considering the presence and activity of antinutritional compounds. In chickpea, antinutritional components such as trypsin inhibitors and lectins are generally present at lower levels than in soybean; however, their occurrence still warrants systematic investigation [
7]. The antinutritional bioactive compounds have diverse physiological effects, which have been involved as plant defense mechanisms against predators, herbivorous insects, extreme environmental conditions, or as a kind of reserve source [
8].
Although many legumes provide valuable proteins, chickpea was selected for this study, as it is one of the major pulse crops and has been used intensively as food in developed countries. Chickpeas are valued for their high protein content, dietary fiber, complex carbohydrates, and bioactive compounds, as well as for their low glycemic index and positive effects on cardiovascular and metabolic health. One of the factors limiting the nutritional quality of many legume proteins is the deficiency of some essential amino acids, particularly sulfur-containing ones; however, this limitation is less pronounced in chickpeas, which provide a more balanced essential amino acid profile [
9]. In addition, chickpeas contribute to soil fertility through biological nitrogen fixation, making them attractive crops from both nutritional and agronomic perspectives.
Chickpea belongs to the family Fabaceae, subfamily
Faboideae [
10]. Chickpea—also known as Bengal gram, Chi-chi beans or Egyptian pea. Based on seed color, shape, size, chickpeas can be divided into two main types: desi and kabuli (
Figure 1).
Desi type chickpeas (desi chana) are small, furrowed seeds, typically dark brown, and mainly cultivated in semiarid climates. In addition to brown, desi types can occur as red, green, or black seeds, each representing distinct varieties [
11]. The black desi chickpea (Ceci neri) is grown in Apulia and Basilicata, southern Italy, and is slightly larger and darker than the regular brown desi type. Kabuli-type chickpeas (white chickpea/garbanzo/garbanzo bean/kabuli chana) are larger, smooth, cream-colored seeds, produced predominantly in temperate climates [
12].
Figure 1.
Different chickpea types of species:
Cicer arietinum L. (Note: Chickpea is mainly classified into two market types, desi (generally light-colored) and kabuli, which differ in seed size, seed coat thickness, and color (e.g., yellow, green, brown, or black), with multiple varieties (cultivars) existing within each type and color group. (self-edited figure based on [
13,
14]).
Figure 1.
Different chickpea types of species:
Cicer arietinum L. (Note: Chickpea is mainly classified into two market types, desi (generally light-colored) and kabuli, which differ in seed size, seed coat thickness, and color (e.g., yellow, green, brown, or black), with multiple varieties (cultivars) existing within each type and color group. (self-edited figure based on [
13,
14]).
Among legumes, chickpea (Cicer arietinum L.) production ranks third after beans, with a mean annual global production exceeding 11.5 million tons. The reported value refers to a mean annual production calculated as an average over multiple years (typically based on data from a 3–10-year period), rather than a single production year.
Land area devoted to chickpea has increased in recent years and reached an estimated 14.56 million hectares worldwide [
15]. Originally, it was a legume of the Middle East, which spread geographically with the help of trading and migration [
16]. Chickpea is a key element in Mediterranean and Middle Eastern cuisines (hummus, falafel). In addition, it is also used in South Asia as a raw material for Dal [
15]. In Indian cuisine, chickpeas are often used in soups, salads, stews, curry, and in chana masala. In the 2020–2021 period, India was the top producer of chickpea with 11,494,250 t, followed by Australia (578,834 t), Turkey (552,500 t), Myanmar (474,662 t), Ethiopia (467,765 t) [
17]. A few countries account for the majority of global chickpea production: India 71.2% (top leader), Pakistan 6.3%, Turkey 5.2%, Australia 4.5%, Ethiopia 3.4%, Myanmar 2.5%, Mexico 2.1%, Iran 2%, Canada and the U.S.A. 1.3–1.5% [
1].
There are more than 81,000 chickpea varieties in gene banks around the world [
18].
In Hungary, chickpea cultivation is currently limited and lacks a long cultivation tradition. Cultivation of chickpea in Hungary was initially used mainly for agricultural purposes, such as animal feed or agronomic functions (e.g., crop rotation practices, soil fertility improvement), and over time its use shifted toward human consumption and gastronomy, where it became an important ingredient in various traditional and modern foods (e.g., hummus, falafel, soups, salads and various main courses). In 2018, the cultivation of chickpea was only 272 hectares in Hungary [
19]. Experimental cultivation began at research institutes such as the Iregszemcse Research Institute in the 1970s, and today it occurs sporadically on small farms and research fields rather than on large commercial scales [
20]. In Hungary, both desi and kabuli types of chickpeas can be cultivated, although their performance depends on local climatic conditions; desi types are more tolerant of drier conditions, while kabuli types require slightly warmer and temperate climates [
20].
Although the widespread cultivation of chickpea has not been successful in Hungary since 1991, currently six varieties are listed in the National List of Varieties. The 6 chickpea varieties are ‘Boglárka’, ‘Bori’, ‘Dora’, ‘Dónia’, ‘Ireg 22’, and ‘Katalin’. The first domestic variety was ‘Dónia’, which was certificated in 1986, while the most recent, ‘Ireg 22’, has been available since January 2024 [
21]. Some Hungarian chickpea varieties, such as ‘Bori’ and ‘Katalin’, have been documented with nutritional profiles with approximately 21–22% protein and 55–56% carbohydrates according to seed distributor descriptions. However, formal scientific studies on the nutritional composition of each individual Hungarian cultivar are limited.
Market demand and cultivation are supposed to continuously grow, even in Hungary. It can be successfully grown in humid conditions, even without irrigation, since it is an extremely drought-tolerant plant, a characteristic that is becoming more and more important as the climate changes. Among the environmental factors, drought stress can considerably affect the germination and the emergence of seedlings. The success of chickpea cultivation depends on the amount and type of precipitation that occurs during the initial growing of seeds. In Hungary, the most favorable regions for chickpea cultivation are located between the Danube and Tisza rivers, the southern Great Plain (e.g., the areas around Békés, Csongrád, and Csanád counties), which are well suited to Hungary’s warmer and drier climate zones, and where the cumulative temperature (sum of daily mean temperatures above the base temperature) during the growing season reaches 1700–2500 °C.
In the case of Hungarian chickpea varieties—like Boglárka, Bori, Dora, Dónia, Ireg, and Katalin—research on these Hungarian chickpea varieties has primarily focused on agronomic traits, such as yield potential, flowering time, disease resistance, and adaptability to local climatic conditions. These studies are mostly in Hungarian agricultural research or breeding programs. Cultivation circumstances of Hungarian breeding chickpea varieties were studied by Bozóki and co-workers under laboratory conditions to evaluate the germination of Hungarian breeding varieties at different temperatures. The results show that germination was highest under alternating temperatures of 30 °C, while higher constant temperatures inhibited seed sprouting, providing key insights for optimizing chickpea cultivation under changing environmental conditions [
22]. As far as nutritional studies are concerned, specific data on the nutritional and antinutritional components of the Hungarian chickpea cultivars Boglárka, Bori, Dora, Dónia, Ireg, and Katalin are still limited, highlighting the need for further investigation to address this research gap. However, due to the increasing popularity of healthy and reform diets in human nutrition, together with the challenges posed by climate change, the demand for the gastronomic utilization and wider introduction of chickpea is steadily increasing. Therefore, the investigation of the nutritional and antinutritional characteristics of these cultivars is both timely and justified. Hungarian chickpea cultivars have been only limitedly studied from a nutritional perspective, and particularly little information is available on their protein-based antinutritional factors.
The present study focuses on the antinutritional protease inhibitors (PIs, such as trypsin and chymotrypsin inhibitors)—naturally concentrated in the seeds and tubers of plants—and lectin protein factors in Hungarian chickpea varieties. In contrast to widely investigated legumes such as soybean or common bean, data on the occurrence and variability of trypsin inhibitors and lectins in chickpea cultivars grown in Hungary are scarce. Therefore, a detailed characterization of these antinutritional proteins is needed to better understand their nutritional quality.
Therefore, the objective of our preliminary study was to analyze the different Hungarian chickpea varieties through three consecutive harvest years in terms of antinutritive protein content based on the determination of their trypsin inhibitor activity and lectin activity. We examined the influence of the different growing domestic locations and the year of cultivation on these parameters and also compared the results with the results of other chickpea varieties originating from other foreign countries. Investigating trypsin inhibitor (TI) content and lectin activity in chickpea cultivars is important because these protein-based antinutritional factors can affect digestibility and nutrient availability in human and animal diets. Knowledge of their levels and variability among cultivars and growing conditions provides critical information for breeding programs aiming to select varieties with improved nutritional quality. Additionally, understanding the activity of these proteins can inform processing strategies (e.g., soaking, cooking, or other treatments) to reduce antinutritional effects, ensuring safer and more nutritious food and feed products. The generated data also contribute to comparative studies with other legumes and may support future functional food or bioactive protein research.
2. Materials and Methods
2.1. Plant Material
Seven varieties of chickpea (
Cicer arietinum L.) seeds involved in Hungarian cultivation were provided by different breeders as (I) Szarvasi Medicago Ltd. (city of Szarvas, Hungary), (II) Agricultural Institute of Iregszemcse (city of Iregszemcse, Hungary), and (III) Fleischmann Rudolf Agricultural Research Institute (city of Kompolt, Hungary) in three subsequent years (Year1, Year2, Year3). Only in the case of some samples was it not possible to provide samples from every year. The experimental sites were located in Szarvas (46.87° N, 20.55° E), Iregszemcse (46.70° N, 18.18° E), and Kompolt (47.49° N, 20.24° E), Hungary, representing different regional climatic conditions of the country’s temperate continental climate. Five varieties of chickpeas originated from foreign countries (IV) were bought at a Hungarian supermarket in Year3.
Table 1 provides information on the parameters associated with the given samples (name of the cultivated variety, place of cultivation, year of cultivation). In the case of commercial samples, the year of purchase was indicated. Chickpea seeds were milled to pass a 0.5 mm sieve using a G-742 grinder (Hauser Magyarország Kft., Budaörs, Hungary) at medium speed (3 × 10 s), with the sample homogenized between each run to ensure uniform particle size. After milling, the powder was stored at 4 °C and used for further analyses.
2.2. Sodium Dodecyl Sulphate Polyacrylamide Electrophoresis (SDS-PAGE)
SDS-PAGE was performed according to Laemmli protocol [
23] following the Instruction Manual of Mini-PROTEAN 3Cell (BioRad, Hercules, CA, USA) using 15% separating gel with 6% stacking gel on the top (0.75 mm thickness, 7 cm width). Separating gel contains (for 2 gels): 4 mL 30% Acrylamide/Bis Solution (29:1), 1.8 mL 1.5 M Tris-HCl (pH 8.8), 10% 50 μL SDS, 2.06 mL distilled water, 6 μL TEMED, 50 μL 10% ammonium-persulphate; while stacking gel contains: 1 mL 30% Acrylamide/Bis Solution (29:1), 10% SDS, 0.66 mL 0.5 M Tris-HCl (6.8), 3.2 mL distilled water, 6 μL TEMED, 50 μL 10% ammonium persulphate.
A total of 2 mg of chickpea flour was extracted by 100 μL 2× Laemmli sample buffer (BioRad 1610737) containing 10% beta-mercaptoethanol and then heated in boiling water for 3 min, and the same amount (6–6 μL) of samples was loaded onto the gel by the use of running buffer (0.025 M Tris, 0.0035 M SDS, 0.193 M glycine).
Separation was performed at 200 V for approx. 65 min at room temperature. After electrophoresis, the separated proteins were fixed on gel with 20% TCA (trichloroacetic acid) for 20 min at room temperature, and then the gel was soaked in distilled water: acetic acid: ethanol/85:5:10 washing liquid (3 × 10 min), with enough liquid to cover the gel well. The staining was performed with 0.2% Coomassie Brilliant Blue R-250 (dissolved in distilled water: acetic acid: ethanol/5:1:5 solution) for 10 min. The background staining was removed by soaking in 10% acetic acid. We replaced the solution with fresh acetic acid every hour and soaked the gel until the dye was completely removed. Image analyses of gels were carried out with BIO-RAD Gel Doc 2000 system (BioRad, Hercules, CA, USA). The SDS-PAGE analyses were repeated three times.
2.3. Separation of Proteins by Native (Non-Denaturing Conditions) PAGE
Native PAGE (in the absence of SDS in gel and running buffer) was performed according to the Laemmli protocol [
23] following the Instruction Manual of Mini-PROTEAN 3Cell (BioRad) using 12% separating gel with 6% stacking gel on the top (0.75 mm thickness, 7 cm width). Separating gel contains (for 2 gels): 3.2 mL 30% Acrylamide/Bis Solution (29:1), 1.5 mL 1.5 M Tris-HCl (pH 8.8), 2.85 mL distilled water, 6 μL TEMED, 50 μL 10% ammonium persulphate, while stacking gel contains: 1 mL 30% Acrylamide/Bis Solution (29:1), 0.66 mL 0.5 M Tris-HCl (pH 6.8), 3.2 mL distilled water, 6 μL TEMED, 50 μL 10% ammonium persulphate.
A total of 3 mg of chickpea flour was extracted in 60 μL sample buffer (as 800 mg saccharose in 4 mL running buffer), vortexed (2 min), then left to stand for 30 min, then further vortexed (2 min), and placed in the deep freezer overnight. The following day, it was centrifuged at 12,000× g for 10 min. A total of 1 µL supernatant of sample extract was loaded onto the gel with the help of running buffer (3.03 g Tris, 14.4 g glycine in 1000 mL distilled water) containing a small amount of bromophenol blue.
Separation was run at 200 V for about 40–55 min at room temperature. After the run, the stacking gel was removed and discarded. The separated proteins were fixed on the resolving gel with 20% TCA for 20 min, then gel was soaked in distilled water: acetic acid: ethanol/85:5:10 washing liquid (3 × 10 min), with enough liquid to cover the gel well. The staining was performed with 0.2% Coomassie Brilliant Blue R-250 staining dye (dissolved in distilled water: acetic acid: ethanol/5:1:5 solution), with enough liquid to cover the gel well. The background staining was removed by soaking in 10% acetic acid, with enough liquid to cover the gel well. We replaced the solution with fresh acetic acid every hour and soaked the gel until the dye was completely removed. Image analyses of gels were carried out with BIO-RAD Gel Doc 2000 system. The native-PAGE analyses were repeated three times. Soybean KTI (Sigma T2327, St. Louis, MO, USA) and soybean BBI (Sigma T9777) standards were used on the gels as control inhibitors.
2.4. Identification of Trypsin and Chymotrypsin Inhibitor Activity in Native-PAGE Gels
Activity of trypsin and chymotrypsin inhibitors was assessed according to Hajós et al. [
24]. The principle behind this method relied on the separation of the chickpea proteins using the native PAGE on 12%/6% separating/stacking gel (see
Section 2.3). Immediately after separation, gel was immersed in 30 mL 0.3 M phosphate buffer (120 mL 0.3 M Na
2HPO
4·2 H
2O solution mixed with 80 mL 0.3 M NaH
2PO
4·2 H
2O solution, pH 7.4) containing 8 mg bovine trypsin (Sigma T4665) or chymotrypsin (Fluka 27270, Seelze, Germany), respectively, for 30 min incubation with shaking at 43 °C. The gels were rinsed in distilled water twice for 2 min to wash out the abundant reagents before incubating in staining solution for 25 min without shaking. For each gel, the negative enzyme staining solution was prepared freshly by dissolving 25 mg of N-acetyl-DL-phenylalanine β-naphthyl ester chromogenic substrate (Sigma A7512) in 10 mL N, N-dimethylformamide, and 42 mg tetrazoitized (zinc chloride complex) o-dianisidine as a dye (Sigma A3502, Fast blue B salt) in 38.5 mL distilled water, which was diluted up to 77 mL with 0.3 M phosphate buffer (pH 7.4), separately. The two solutions were mixed immediately before they were poured to cover the gel. After 20 min, the reaction was stopped with 1% acetic acid.
The gels were rinsed in distilled water and then immersed to fix proteins in 20% TCA for 20 min at room temperature. After that, the gels were soaked in acetic acid: ethanol: water/5:10:85 gel washing liquid for 15 min, with enough liquid to cover the gel well. The background staining was performed with 0.2% Coomassie Brilliant Blue R-250 staining dye dissolved in distilled water: acetic acid: ethanol/5:1:5 (Reanal, Budapest, Hungary), with enough liquid to cover the gel well. The dye was then removed by soaking in 10% acetic acid.
The presence of trypsin or chymotrypsin inhibitors became visible as light bands against a dark pink background. In samples containing trypsin inhibitor, the inhibitor blocked the action of trypsin (or chymotrypsin), thus depleting it. The method is called negative staining because the gel is stained only where the sample does not contain trypsin inhibitor, allowing trypsin to access its substrate in the gel.
Soybean KTI (Sigma T2327) and soybean BBI (Sigma T9777) standards were used on the gels as control inhibitors. Image analyses of gels were carried out with BIO-RAD Gel Doc 2000 system and evaluated by densitometric analysis using BIO-RAD Quantity One 4.3 version. The negative-stained native-PAGE analyses were repeated three times.
2.5. Determination of Trypsin Inhibitor Activity
The total trypsin inhibitor activity (TIA) was assessed according to the ISO 14902:2001 (MSZ EN ISO 14902:2006) protocol (ISO14902, 2001) with a slight modification in trypsin dilution in order to obtain absorbance values within the recommended linear range of the assay specified by the standard, thereby improving accuracy and reproducibility. This adjustment was necessary due to the specific trypsin inhibitor activity of the samples—to ensure absorbance values fell within the optimal and linear measurement range -and does not affect the principle or calculation of the method.
Sample extraction: Chickpea seed flour (m0 = 1 g) was extracted in 50 mL of 0.01 M NaOH (pH 9.5), then allowed to stand overnight at 4 °C. On the following day, it was further diluted with 50 mL of cold distilled water (4 °C) by shaking, and it was allowed to sediment for 15 min (it was not centrifuged). The supernatant from each sample was collected (sample extract) and then diluted (f1) in distilled water (test sample extract) for the experiment, considering achieving approximately 40–50% trypsin inhibitor activity.
The measurement protocol is summarized in
Table 2.
In the case of Abs, As: 1 mL test sample extract was pipetted into test tubes in triplicates and mixed with 5 mL of L-BAPA reagent solution (Nα-Benzoyl-DL-arginine p-nitroanilide hydrochloride as substrate, Sigma B4875; 60 mg/1 mL DMSO made up to 100 mL with Tris-Ca2+ buffer solution (pH 6.2, containing 6.05 g Tris + 735 mg CaCl2, which was diluted up to 1000 mL in distilled water) and 2 mL of distilled water.
In the case of Abr, Ar: no test sample extract was used; tubes consisted of 5 mL BAPA, 3 mL distilled water. According to the next step, only to blank samples (Abr, Abs), 1 mL of 5.3 M acetic acid was further added (not added to Ar and As). All mixtures were then incubated for 10 min at 37 °C. After the 10 min-incubation, the content of tubes was vortexed, then 1 mL trypsin (m1 = 27 mg trypsin from bovine pancreas (Sigma T-4665) was diluted up to 100 mL with 0.001 M HCl, then out of 5 mL was diluted up to 50 mL with Tris-Ca2+ buffer solution) was added to all tubes (Abr, Ar, Abs, As). It was vortexed and incubated at 37 °C for 10 min. In the case of Ar and As tubes, the reaction was stopped by the addition of 1 mL of 5.3 M acetic acid. Then, all mixtures were centrifuged at 3500 rpm for 10 min. The quantity of released p-nitroaniline is measured spectrophotometrically at 410 nm against distilled water, and then TIA was calculated in terms of pure trypsin/g sample as weighed (mg/g).
The inhibition percentage of the sample extract solution: If Ar−Abr is 0.38–0.4, the trypsin works correctly.
Trypsin inhibitor activity (mg inhibited trypsin/g sample):
where
i: inhibition percentage (%);
Ar: absorbance of the solution with trypsin standard (positive control
Abr: absorbance of the blank solution with trypsin standard (as negative control);
As: absorbance of the solution with sample;
Abs: absorbance of the blank with sample;
f1: overall dilution and back-calculation factor (accounts for all dilutions during extraction and assay, allowing the measured absorbance to be converted back to the original sample. For measurements targeting approximately 40% or 50% trypsin inhibition, specific volumes of extract (1.2 mL or 1.5 mL, respectively) are used to achieve these inhibition levels. f1 corrects for both the sample dilution and the fraction of extract tested, giving inhibitor activity per 100 g of sample.
at 40% inhibition: f1 = 100 × 100/1.2
at 50% inhibition: f1 = 100 × 100/1.5
f2: conversation factor 0.0003.
m0: mass of the sample (g); which is 1 g in this case
m1: mass of trypsin (mg); which is 27 mg in this case
The total trypsin inhibitor activity was expressed as mg of inhibited trypsin per g of sample, reflecting the functional inhibition of trypsin by the chickpea proteins under standardized assay conditions. Results were evaluated by statistical analyses (see
Section 2.7).
2.6. Haemagglutinin Activity [25]
Rat blood samples were collected into tubes containing EDTA (Rextra Orvosi Műszer Kereskedelmi Kft., Budapest, Hungary) (7.2 mg), and after collection were diluted tenfold with saline (9 g sodium chloride/L). Rat erythrocytes were pre-treated with pronase (0.2 mg/mL diluted erythrocytes for 30 min, 25 °C). Chickpea flours (20 mg/mL) were extracted in PBS (pH 7.4) buffer for 16 h at 1 °C. After centrifugation (20,000 rpm for 10 min), the clear supernatant fractions were tested for haemagglutinin activity. The supernatant fractions (0.1 mL) were serially twofold diluted and mixed with an equal volume of diluted erythrocytes (final volume 0.2 mL). The results were read from the microtiter plate after 30 min in room temperature.
Haemagglutinin activity is the smallest amount of lectin that causes visible agglutination in a standard red blood cell suspension under given conditions. Results were given in mg chickpea flour/mL, which was the minimal concentration that still showed activity. The collection of rat blood for the hemagglutination assay met all the requirements of the Government Office of Pest County (Registration No. 35/2015) and National Scientific Ethical Committee on Animal Experimentation (Permit No.: KA-2897)
2.7. Statistical Analysis
Statistical analysis was performed with IBM SPSS Version 27.0 (IBM, Chicago, IL, USA) statistical software package. The effect of genotype and cultivation years on the TIA values was evaluated with a t-test (in case when data were available from only two years) or one-way analysis of variance (ANOVA) (data from all three years were available). After ANOVA, Tukey’s Honest Significant Difference (HSD) test was used to determine which group of means differ significantly.
3. Results and Discussion
3.1. SDS-PAGE Results of the Domestic and Foreign Chickpea Varieties
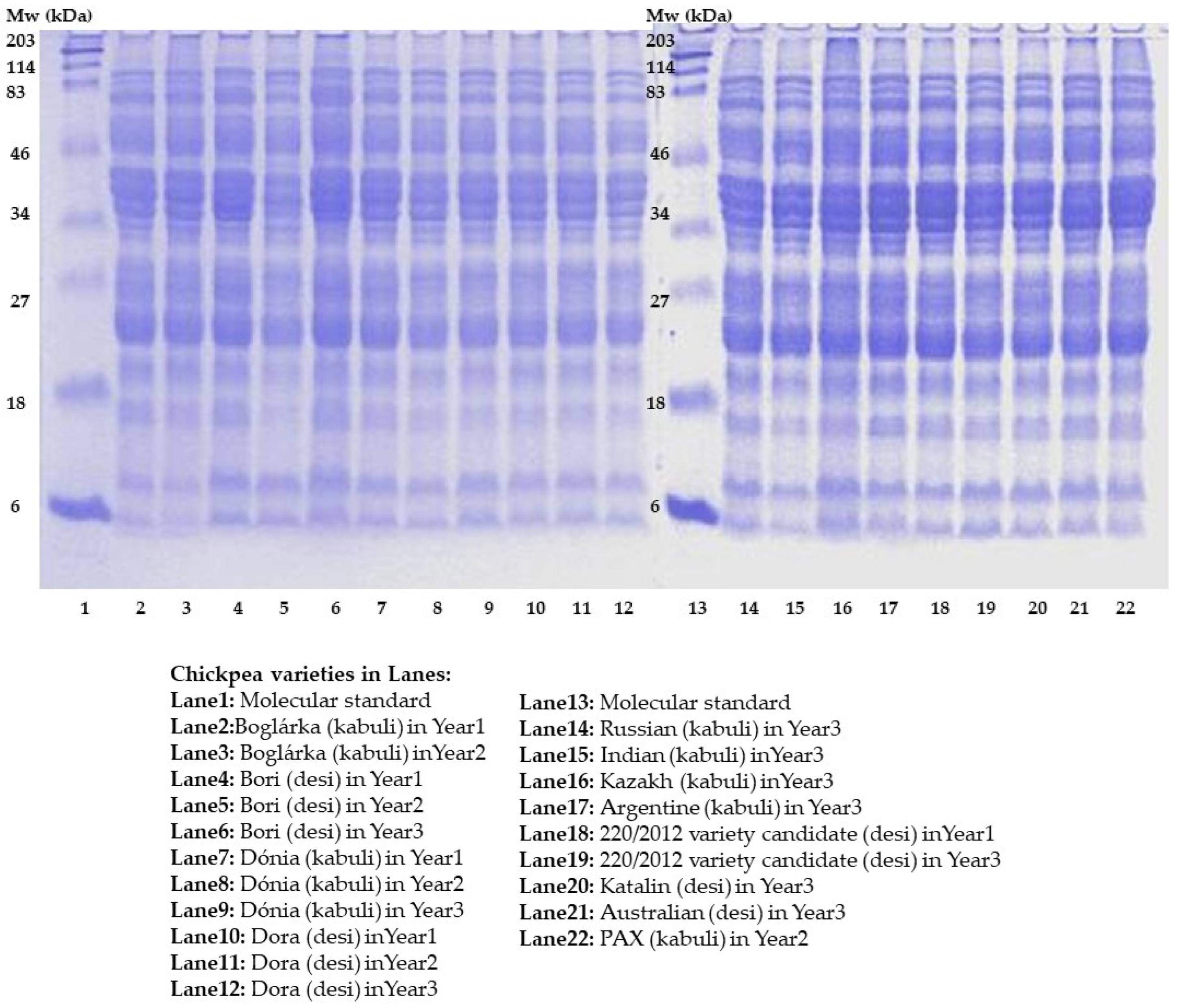
The protein distribution of some available Hungarian chickpea varieties was monitored by SDS-PAGE, and their protein patterns were compared with those of other chickpeas originating from foreign countries (
Figure 2). SDS–PAGE results are provided to confirm the overall protein composition of the samples.
The SDS–PAGE profiles of chickpea samples showed complex protein patterns with multiple bands distributed over a wide molecular weight range (approximately 6–203 kDa). The overall banding patterns were similar among the samples, indicating comparable protein compositions. Major protein fractions were observed predominantly in the 20–50 kDa range, which is characteristic of chickpea storage proteins. No pronounced qualitative differences were detected between the lanes, although minor variations in band intensity suggest differences in relative protein abundance among samples.
3.2. Identification of Trypsin Inhibitors on Native Gel in Domestic and Foreign Chickpea Varieties
Varieties often differ not in new proteins, but in proportions and forms. While SDS-PAGE shows a very similar pattern, native PAGE gives different band numbers, band positions, and intensity between species, as the ratio of isoforms varies, the tendency to aggregate differs, and post-translational modifications may occur. These features enable a more sensitive comparison of biologically relevant differences between samples than denaturing SDS–PAGE.
Native PAGE is an appropriate method, especially for the analysis of trypsin inhibitors, because it preserves the native structure and conformation, and biological activity of the proteins. Trypsin inhibitors require an intact three-dimensional structure, including disulfide bonds and a functional reactive site, to bind and inhibit trypsin. In SDS-PAGE, SDS denatures proteins and disrupts their native conformation, and as a result, trypsin inhibitors lose their inhibitory activity. In native PAGE, proteins remain folded and biologically active, so trypsin inhibitors retain their ability to inhibit trypsin. Inhibitors may exist as monomers, dimers, or higher-order oligomers, and under native conditions, these are preserved.
In
Figure 3A, it can be seen that proteins in the same bandwidth as KTI are visible in the chickpea samples (refer to the KTI-type inhibitors), but the majority of the proteins are concentrated in the higher range. The lack of major differences may reflect the dominance of conserved and stable storage proteins, which constitute most chickpea seed proteins.
According to
Figure 3B, we can establish that Kunitz-type inhibitor bands from chickpea did not co-migrate with the soybean KTI reference, indicating structural and/or charge-related differences between the inhibitors from the two legumes. The occurrence of multiple inhibitory zones in the zymogram in the case of chickpea samples suggests the presence of several active forms of the trypsin inhibitor, likely representing charge variants, isoforms, oligomeric assemblies, or stable enzyme–inhibitor complexes preserved under native electrophoretic conditions [
26].
Previous studies by other researchers on trypsin inhibitors in chickpea showed the existence of six to eight iso-inhibitors [
27]. In the coming years, genotypic variation in chickpea protease inhibitor levels was also reported by [
28,
29,
30]. Saini and co-workers (1992) with a range of Desi and Kabuli chickpea genotypes, developed and grown in Australia, examined their ability to inhibit bovine, porcine, and human trypsin/chymotrypsin, and compared their iso-inhibitor patterns [
31]. After staining for trypsin inhibitors, seven iso-inhibitors were detected in all of the samples. The isoelectric points (pI) of chickpea inhibitors CPI-1 to CPI-7 (denoted according to increasing pI) were 4.9, 6.8, 7.1, 7.5, 8.4, 8.6, and 9.3, respectively. Inhibitors CPI-2, CPI-5, and CPI-6 were most pronounced, and CPI-4 was least pronounced. Differences in intensity among samples were mainly due to differing inhibitor activities of individual samples.
Seven isoforms of protease inhibitor from chickpea were also identified, and their differential accumulation during seed development was described [
32]. The appearance and disappearance of TI bands during seed development indicate their specific post-translational modification and/or the presence of isolated gene groups. Wild Cicer species (close relatives of cultivated chickpea) show even greater diversity in trypsin inhibitor banding patterns, supporting the idea that multiple isoforms exist and vary genetically [
32]. Later, a study on eight chickpea cultivars examined trypsin inhibitor (TI) and gut protease inhibitor activities and reported electrophoretic patterns showing variation in TI bands during seed development. The authors observed that different TI bands appear and vary in intensity, reflecting multiple inhibitor forms detectable by electrophoresis. They noted that TI accumulation changes over developmental time and that different cultivars show distinct electrophoretic TI banding patterns, indicating the presence of multiple TI forms [
33]. Guillamón and co-workers (2009) analyzed 4 Spanish chickpea varieties on zymogram gel and detected 3–4 isoforms [
34]. Similar studies were made by Bale [
35] on 8 chickpea genotypes, and 6 isoforms (Ti1–Ti6) were identified [
35]. Ti1–Ti3 could be observed in all eight chickpea genotypes investigated, with significant variations in their band intensities. Regarding Ti4–Ti6, significant variation was observed among the genotypes studied. Ti6 was present only in one chickpea genotype [
35].
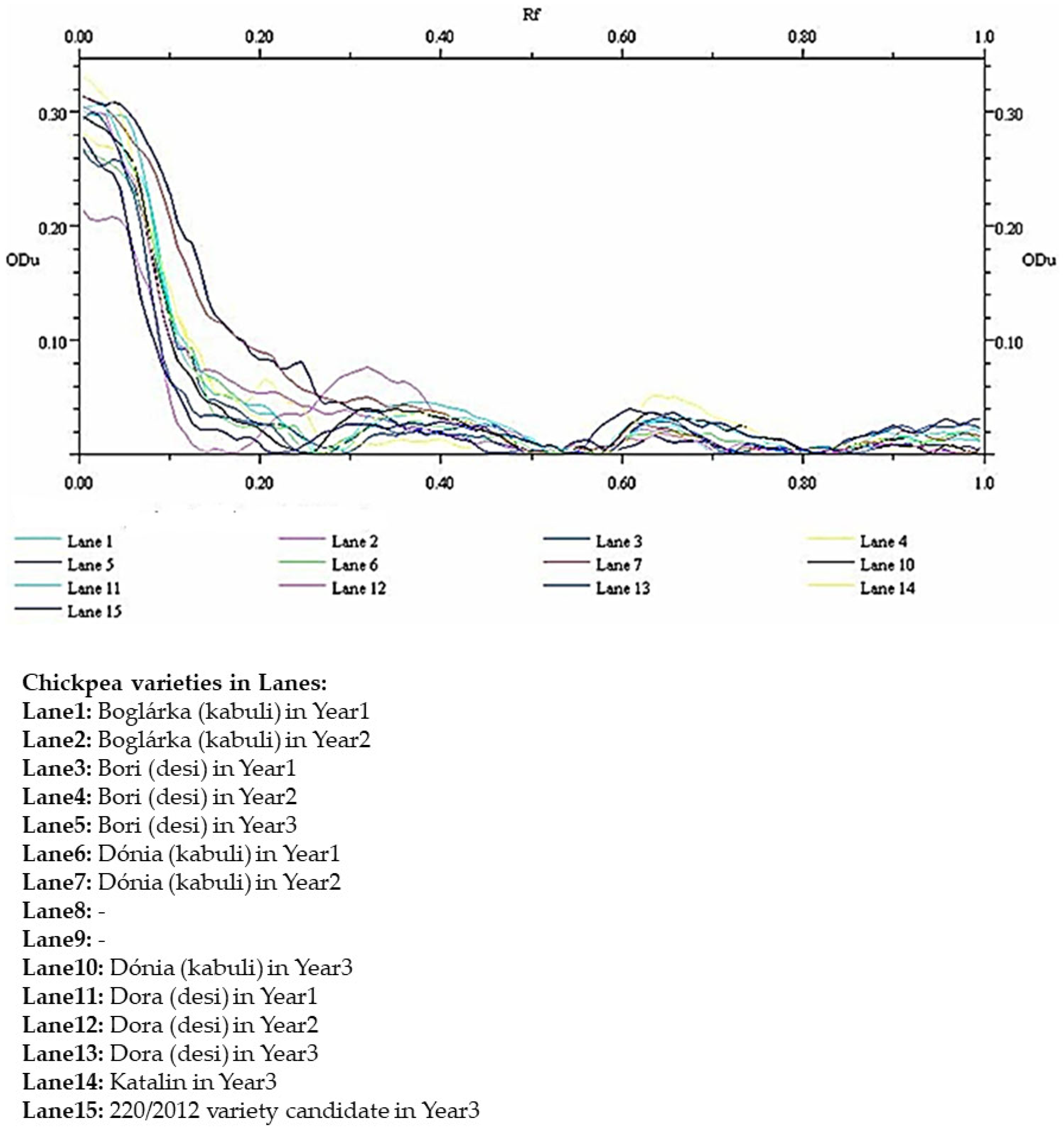
According to our results, two main intensive bands and a few thinner, less intense ones (2–4) showed activity against trypsin. The KTI-type protein bands that showed activity were evaluated by densitometry, and relative quantities were calculated based on integrated optical density (relative front, peak ODU, trace ODU × mm, relative Qty) values within each lane, providing a semi-quantitative comparison of protein abundance. There were only very slight differences between samples; there was no significant difference in terms of the impact of the growing season or the type of variety. To verify these results, the densitograms obtained on Hungarian chickpea varieties are presented (
Figure 4). While small variations in densitometric profiles were observed among the samples, the overall patterns were highly similar (protein bands from different chickpea samples largely overlapped), and these differences were not indicative of significant changes.
3.3. Measurement of Total Trypsin Inhibitor and Lectin Activities in Domestic and Foreign Chickpea Varieties
While on gels, the presence of the Kunitz-type trypsin inhibitors was introduced, the total amount of trypsin inhibitors could be measured spectrophotometrically on the principle of the quantity of released p-nitroaniline, from which the trypsin inhibitor activity (TIA) was calculated in terms of pure mg trypsin/g sample as weighed (mg/g). This method measures the amount of two types of trypsin inhibitors, including KTI-types and BBI-types trypsin inhibitors.
Table 3 summarizes the results of the total trypsin inhibitor activity (TIA) of all analyzed chickpea varieties.
The value of the TIAs in chickpeas are lower than, for example, that of soybean cultivars [
36]. Differences in TIA between Hungarian cultivars were observed, suggesting the possibility of breeding for low TIA. Overall, considering the 3 subsequent years: TIA < 2 mg/g in the case of Boglárka, Bori, and 220/2012 variety candidate; 1.9 < TIA< 2.3 mg/g in the case of Dora and PAX, and the highest TIA 2.77–4.07 mg/g in the case of Dónia and Katalin. The differences between the values of the individual varieties in different years were not significant (
p = 0.05). The type of chickpeas, based on kabuli or desi type, did not cause differences in the TIA results. However, Singh and Jambunathan (1981) analyzed different chickpea cultivars, and they found higher TIU mg-1 sample values in desi (12.7) than in kabuli (10.3) [
28].
Commercial samples (Kazakh, Argentine, Russian, Australian) indicated similar trypsin inhibitor activity in the range 2.41–2.82 mg/g, while the sample originating from India had lower TIA (0.49 ± 0.13 mg/g).
Considering the Hungarian and foreign samples, they were very similar, except in the Indian sample, where the lowest TIA value was detected, and the Katalin sample, where the highest value was measured. Based on the values, three groups were statistically identified with different levels of TIA activity.
Comparing all foreign samples (average: 2.20, ±0.95) with the average of all Hungarian samples (regardless of year) (2.14 ± 0.97), according to the result of the t-test (p = 0.88), there was no difference between the two groups.
A previous study on 25 kabuli-type chickpea cultivars grown in New Zealand reported trypsin inhibitor activity ranging from 2.3 to 4.0 mg inhibited trypsin per g dry matter, with a mean of 2.7 mg/g, which aligns closely with the activity levels observed in our Hungarian and foreign chickpea cultivars (TIA ~0.49–4.07 mg inhibited trypsin/g sample [
37].
While not always reported in exactly the same units, many legume studies do use trypsin inhibitor units per mg sample (TIU/mg). Published data on legumes indicate that trypsin inhibitor activity varies widely among species, with soybean showing high values (~43–84 TIU/mg), common bean intermediate (~21–25 TIU/mg), and chickpea generally lower (~15–19 TIU/mg) on a dry flour basis. These literature values are consistent with the relatively low TIA levels observed in our Hungarian chickpea samples [
34].
Hemagglutination activity of different chickpea varieties was summarized in
Table 4.
The hemagglutination assay [
25] is an easy method to obtain semi-quantitative data on the sugar binding and specificity of lectin. An active lectin agglutinates erythrocytes by recognizing a carbohydrate on the cell surface and forming a cross-linked network in suspension. The agglutination rate is also known to be dependent on the concentration of the lectin and the density of the cell suspension used [
38]. Hemagglutination activity depends on the method and time of heat treatment and the legume plant species [
39].
Through the measurement, we searched for the minimal concentration of the lectin where agglutination could still be detected. The lower the lectin concentration that cause agglutination, the higher the lectin activity is in the given chickpea variety. In chickpea samples, low hemagglutination activity was measured, and in some cases, samples had no lectin activity.
In the case of the Hungarian samples, in Year1, 220/2012 variety candidate (desi) had the highest lectin activity, as at 1.25 mg/mL concentration, it still caused agglutination.
The formation of agglutination requires a higher concentration of lectins, so the lectin activity decreases in the following order: Dónia (2.5 mg/mL), Boglárka (5 mg/mL), and then Dora (10 mg/mL). Bori did not show any lectin activity, neither in Year1, nor in Year2.
Less activity was measured in Year2 in the case of Boglárka (No Activity), but in the case of Dónia (0.63 mg/mL), Dora (5 mg/mL), the lectin activity was higher compared with the results in Year1. Among the commercial foreign chickpea samples, Indian and Russian varieties showed higher lectin activities (1.25 and 2.5 mg/mL) than the Hungarian varieties, while the Australian chickpea had no lectin activity.
Based on the literature, chickpea has lower lectin content than other often consumed pulses like lentil and pea. According to Roy et al. (2010), chickpea lectins have no HA in red blood cells from rats, rabbits, monkeys, or humans (A, B, O types), but agglutinated red blood cells from cows, although not at toxic levels [
40]. Some researchers reported lectin activity of chickpea, but it was below toxic levels [
41,
42].
Comparing lectin activity between legumes, it is around in soybean 692.8 Hu/mg dry flour, in common bean 87.7–88.6 Hu/mg dry flour, in lentil 10.9–11.1 Hu/mg dry flour, in pea 5.5–5.7 Hu/mg dry flour, in chickpea 2.01–2.74 HU/mg dry flour, measured with hemagglutination test where Hu means hemagglutination units (unit of activity required for erythrocyte agglutination) [
43,
44]. Our results on chickpea gave 1–2.5 (only in one case 5) HU/mg flour (converted form mg/mL), closely similar as mentioned by other authors.
Wild chickpea seeds contain lectins that show strong hemagglutination activity with human (A, B, AB, O) and trypsin-treated rabbit erythrocytes, with titers ranging from 8 to 4096 hemagglutination units (HAU). The highest specific lectin activity was found in
Cicer reticulatum ILWC-292, where lectins were stable for 4–6 h and selectively inhibited by complex sugars like desialylated fetuin, suggesting a defensive role in plants and potential use in breeding programs [
45]. A purified chickpea lectin (CAA-II) consists of four identical 30 kDa subunits forming a 120 kDa native protein that agglutinates both untreated and trypsin-treated rabbit erythrocytes as well as human erythrocytes. Hemagglutination inhibition assays showed this lectin has high specificity for N-acetyl-D-galactosamine, distinguishing it from previously reported chickpea lectins [
46]. Broader studies on plant-based foods confirm that Fabaceae family members, including chickpeas, exhibit the highest lectin activity measured by hemagglutination assays using rabbit erythrocytes, and processing methods like soaking and boiling reduce active lectins [
47]. These findings highlight the diversity, carbohydrate specificity, and biological significance of chickpea lectins as assessed by hemagglutination assays.