1. Introduction
The blast furnace hearth can be regarded as a large reaction vessel. Under reducing atmosphere conditions, complex physical and chemical reactions occur, accompanied by heat transfer among slag, molten iron, coke, and refractory materials. These processes collectively determine both the production efficiency of the iron-smelting process and the service life of the equipment. Among studies on melts, research on the physical properties of blast furnace slag, melt structure, and viscosity prediction models is relatively abundant. However, due to the low viscosity of molten iron (1–10 mPa·s) and the challenges associated with high-temperature experiments, it has received comparatively less attention from researchers. But, importantly, in the field of blast furnace ironmaking, many researchers [
1,
2,
3] have suggested that the flow behavior of molten iron has a very important effect on the production efficiency and is also an important factor affecting the service life of blast furnace. Thus, it can be seen that the research which can provide guidance for regulating the viscosity of hot metals should be carried out as soon as possible.
There are many methods for measuring the viscosity of metals and alloy melts, for example, methods using a capillary, an oscillating cup, a falling body, an oscillating plate, a rotating cylinder [
4], a levitated drop, a draining vessel [
5], etc. In particular, the oscillating cup method is well suited for measuring low viscosities with high precision, and it allows the relatively easy fabrication of the crucible. Due to these advantages, the oscillating cup method was selected in this study to verify the calculated results, providing a reliable validation.
The development of semi-empirical viscosity prediction model for metals and alloy melts is faster. From two basic theories of free volume and activation energy, Andrade and Eyring proposed the quasi-crystal hypothesis and the rate theory, respectively. Nowadays, their models have been recognized, inherited, and developed by many researchers [
6]. Most mathematical models incorporate the thermodynamic parameters of the system or the physicochemical properties of the pure components and can achieve a high degree of consistency with experimental results. However, they have some difficulties in predicting the viscosity of blast furnace molten iron, which is mainly because of the nonmetallic alloy composition of the hot metal.
There are some viscosity models that have been proven to accurately predict the viscosity of most pure metals or alloy melts, but they are not applicable to blast furnace molten iron, such as Moelwyn-Hughes [
7], Iida-Ueda-Morita [
8], Kozlov-Romanov-Petrov [
9], etc. The foremost cause of this is that, in the case of calculating the alloy viscosity at a certain temperature by the above models, it is necessary to take the viscosity of the pure components at the corresponding temperature as the input parameter. They are not available to predict the viscosity for liquid iron containing C, S, P, and other components at a high temperature. Consequently, five representative semi-empirical viscosity models were selected for the systematic evaluation conducted in this study. To further quantify the relationship between chemical composition and viscosity of blast furnace hot metal, the available experimental viscosity data of iron-based melts were analyzed and employed to introduce composition-dependent viscosity action coefficients.
2. Experimental
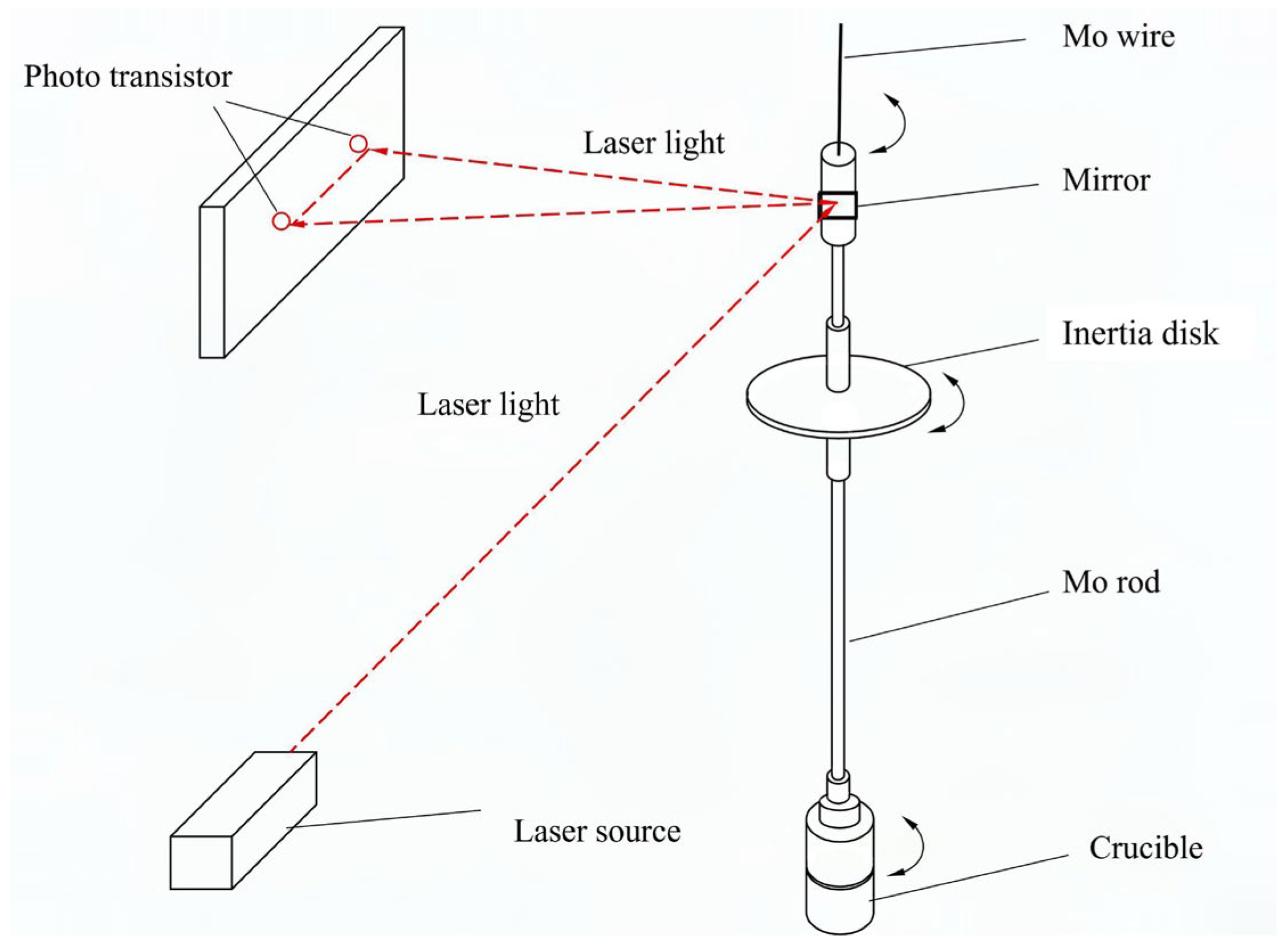
The viscosity of melt sample was determined by the oscillating cup method [
10]. The whole viscometer used in this work is shown schematically in
Figure 1, which consists of a suspension system, an oscillation detection system, a heating system, and a magnetic field system. The suspension and detection systems inside the apparatus are shown in
Figure 2.
The blast furnace iron samples of viscosity measurement experiment were all taken from a steel mill. Before the viscosity measurement, the samples were pre-melted in a high-temperature tubular furnace into a 24 × 55 Φ cylindrical size. In order to eliminate the influence of oxidized impurities in the viscosity measurement, in the process of pre-melting, argon gas is injected into the chamber, and then the surface of every cylindrical sample needs to be polished and cleaned. Chemical analysis was used to obtain the composition information of experimental molten iron, as shown below in
Table 1.
The viscosity was measured during the cooling process, under high-purity argon gas conditions. The highest temperature used in this measurement was 1450 °C, with each measurement temperature point held for 20~40 min. At least five sets of reliable data are measured at each temperature point, and the average value is obtained. The viscosity was determined by using Shvidkovskiy’s equation [
11], in which the height of melt
H, the density of melt
, the moment of inertia of suspension system
I, the logarithmic decrement
, the period of oscillation
T (subscript of 0 represents the parameter of the empty crucible), the correction factor
W (a, b, and c are instrument constants), and the mass of melt
m are required for calculating the viscosity
as follows.
The parameters determined experimentally from oscillation are
and
T; thus, it is important how they are determined [
12]. Before the actual calculation, the logarithmic decrement
and period of oscillation
T0 of the empty crucible must be measured.
Figure 3 shows the effects of temperature of torsion wire on the logarithmic decrement and the period of oscillation for an empty crucible.
3. Component Action Coefficient Calculation
Many factors affect the viscosity of molten iron in a blast furnace, including temperature, the composition of molten iron, and the presence of solid-phase particles. Each factor influences viscosity through a different mechanism, leading to varying degrees of viscosity changes as the content of these components varies. Therefore, it is particularly necessary to clarify the quantitative relationship between component content and the change in molten iron viscosity.
The Fe-C binary system is one of the most basic systems in alloys. Many scientists [
13,
14,
15,
16,
17] who study the physical properties of the melt have measured its viscosity at different temperatures and concentrations. However, due to the high temperature of the experimental conditions as well as the different determination methods and viscosity calculation equations selected by various researchers, the measurement results are mostly not consistent, as shown in
Figure 4.
According to the literature values extracted above, when carbon content is greater than 4 wt%, there are mainly two opposing results about the correlations between carbon content and the molten iron viscosity.
The results of David Belashchenko [
15], Vostryankov [
13], Lucas [
13] and Deng et al. [
17] showed that the viscosity increased with the increase in carbon content when the concentration was greater than 4 wt%. Conversely, Teng [
14], Turovskii [
13], Barfield [
13], Vatolin et al. [
13] found that the viscosity of molten iron decreased with the increase in carbon content in that range. It can also be observed from the literature that the viscosity data for Fe–C melts with carbon contents higher than 4 wt%, which corresponds to the typical composition range of blast furnace hot metal, are relatively scarce. Moreover, the available experimental results in this concentration range exhibit two opposite trends regarding the effect of carbon on viscosity. To avoid arbitrary selection and to ensure methodological completeness, both influence modes were incorporated into the model construction, and the more reasonable relationship was subsequently identified through a systematic comparison with the experimental measurements of industrial molten iron. The viscosity values of Lucas and Vatolin are both complete ([C%] > 4.0 wt%) and selected to be the representatives of the two kinds of relations. Then, the data from them were selected to fit. With the carbon content of 4.5 wt% (
) as the benchmark, the final results are as follows:
Deng [
17] has measured the viscosity of binary and ternary systems of blast furnace molten iron (Fe-xC, Fe-4.5 wt%C-X) and made trend analysis. The measured results were sorted out to obtain the viscosity change curve under different temperature and composition conditions, as shown below in
Figure 5.
The thick black line in
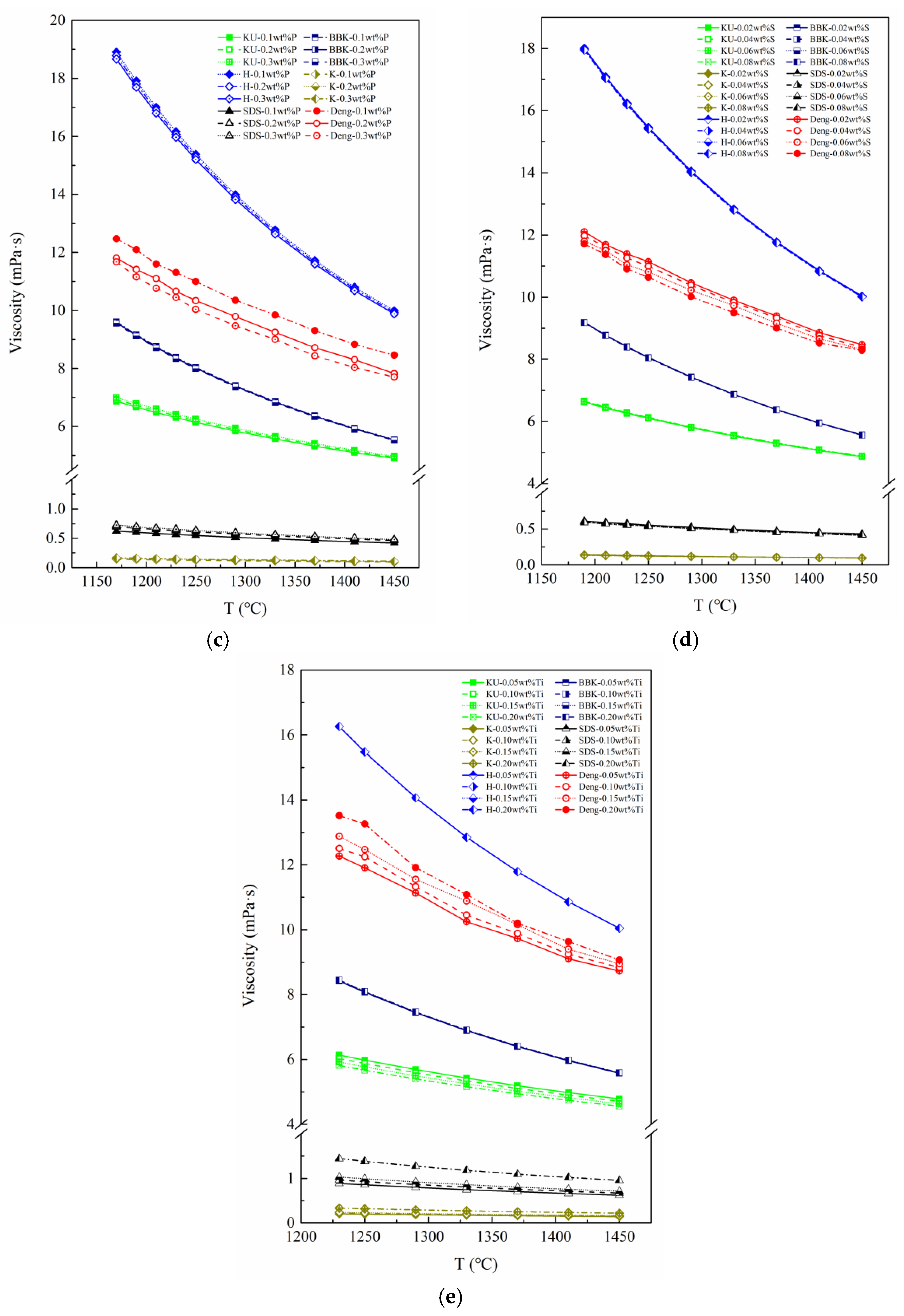
Figure 5 is the binary system with 4.5 wt%C, which is in the middle of all curve distributions. Apparently, after adding the third element, the position of the curve is changed. The curves of Si and Ti shifted upwards, which means that the viscosity of molten iron increased. Similarly, it can be inferred that the components Mn, P and S can improve the fluidity of molten iron. Of all the curves, the Fe-4.5 wt%C-0.2 wt%Ti ternary system has the highest viscosity of molten iron, while the system of 0.5 wt%Mn has the lowest one. Specifically, Si and Ti tend to enhance short-range ordering and increase melt structural complexity, which raises resistance to viscous flow. In contrast, Mn, P, and S act to weaken Fe–Fe bonding or promote depolymerization-like effects in the metallic melt, thereby enhancing fluidity.
On the other hand, after the addition of a third component, the shape of each curve did not change significantly, which is completely different from the effect observed when the carbon content (black curves) in the melt was varied. Therefore, it can be considered that the carbon content of molten iron has the ability to dominate the temperature dependence of viscosity of binary molten iron, and the content of the third elements could dominate the liquidity of molten iron.
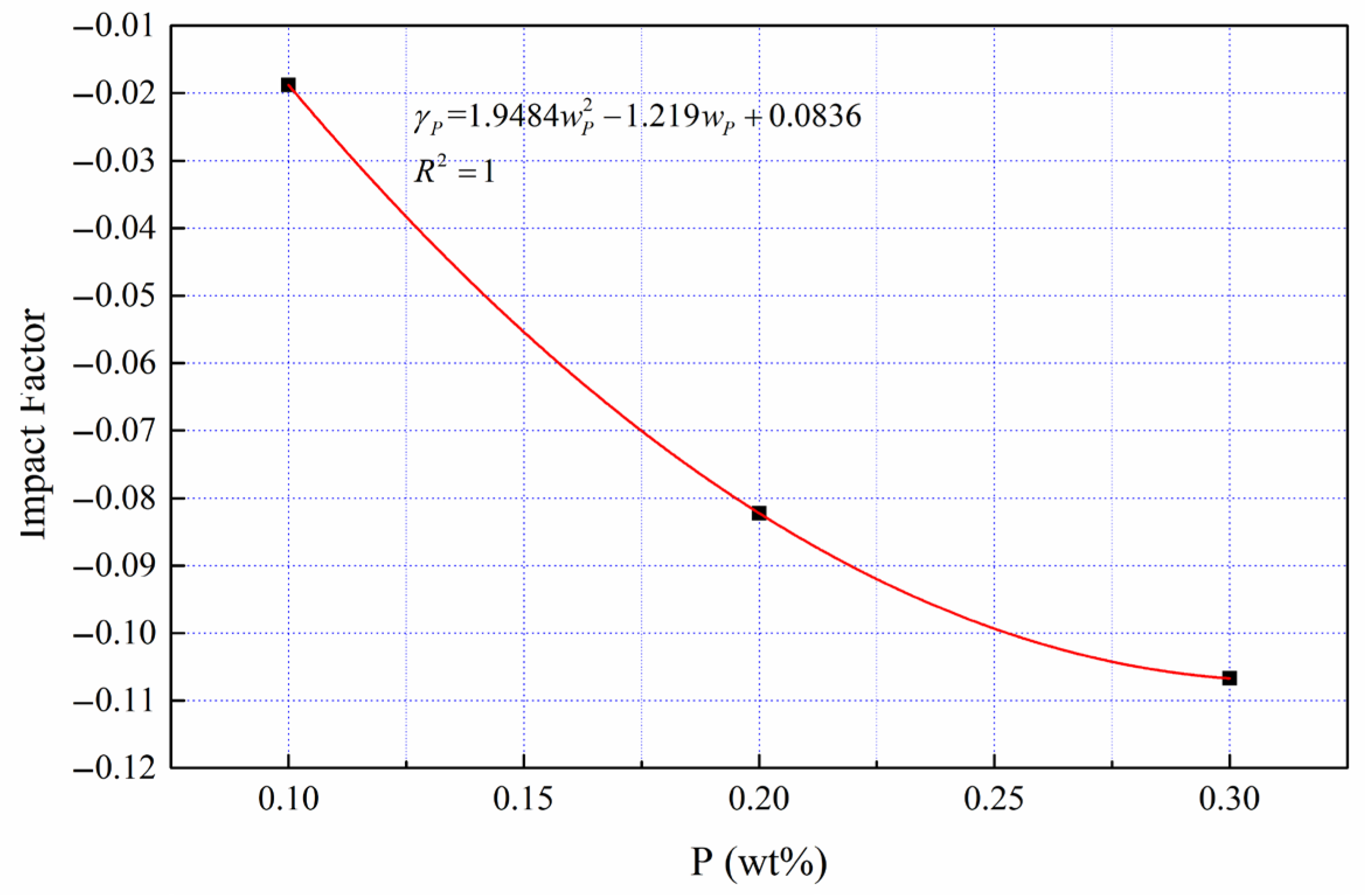
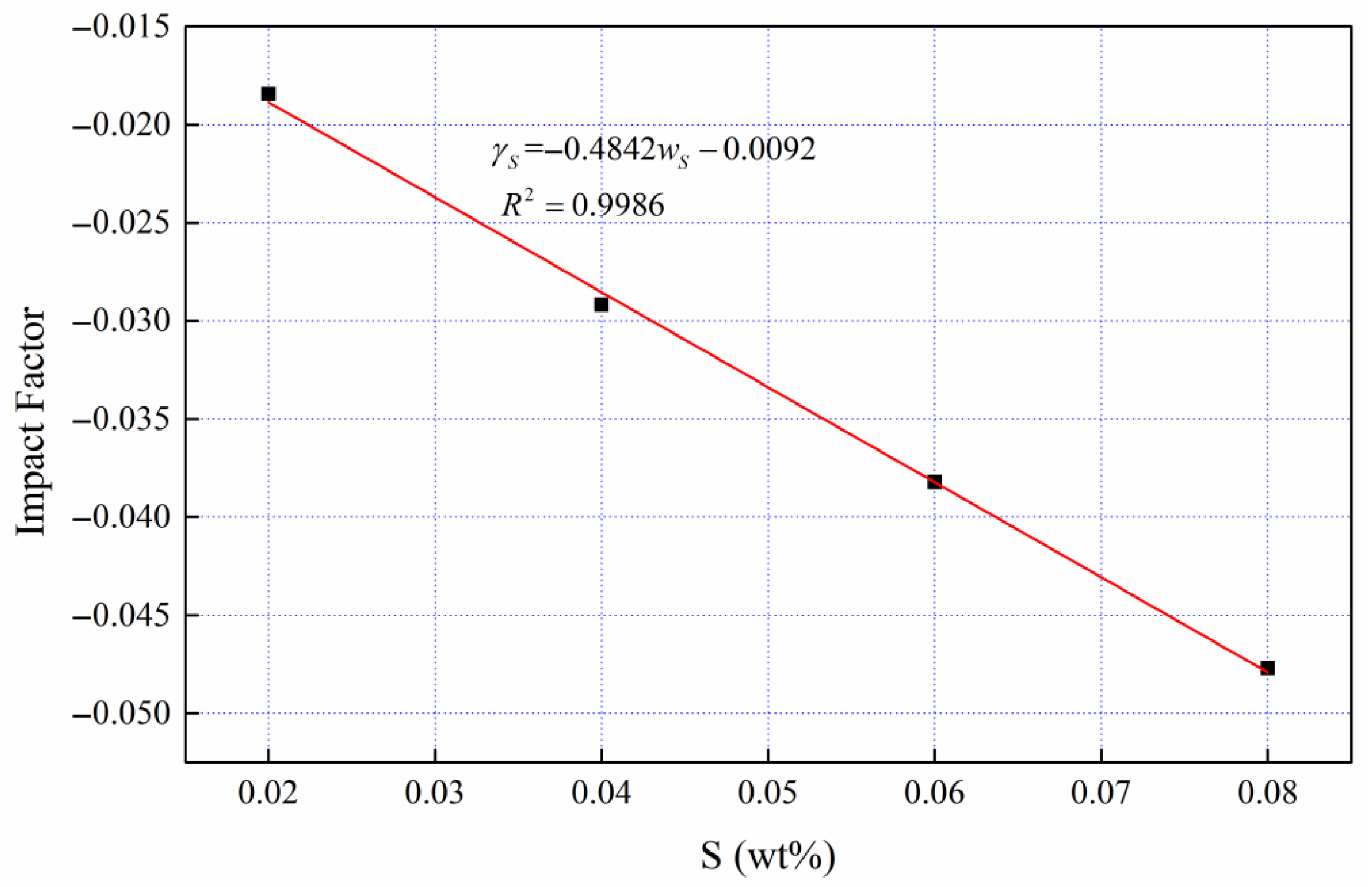
The benchmark viscosity (Fe-4.5 wt %C binary melt) was determined, and the independent viscosity action coefficients of the third elements (Si, Mn, P, S and Ti) was quantified with Equation (7). Then, the average viscosity action coefficient curves of the third elements are shown in
Figure 6,
Figure 7,
Figure 8,
Figure 9 and
Figure 10.
By summarizing Equation (5)–(12), the two total equations of viscosity action coefficient of internal molten iron components can be obtained as follows:
4. Model Selection and Modification
The concentration dependences of molten iron viscosity were calculated using five equations, which are listed below. The following common notations are used: —the dynamic viscosity of melt (); M—the molar mass of melt (); T—the absolute temperature (K); V—the molar volume of melt (); Tm—the melting point (K); x—the mole fraction of melt components; ∆VE—the integral excess volume of mixing (); ∆H—the integral enthalpy of mixing (J/mol); R—the universal gas constant (8.3144 J mol−1 K−1); ρ—the density of melt (kg/m3); h—Planck constant (6.626 × 10−34 J·s); NA—Avogadro number (6.02214 × 1023 mol−1); —Gibbs viscous flow activation energy (J/mol); and i—components i.
Kaptay unified equation (KU) [
18]:
where
A and
B are both the semi-empirical parameters. Kaptay tested 101 measured points of the 15 selected liquid pure metals to find the valid values of the two semi-empirical parameters (
A = (1.80 ± 0.39) × 10
−8 (JK
−1 mol
−1/3)
1/2 and
B = 2.34 ± 0.20 [
18]).
Budai–Benkö–Kaptay model (BBK) [
19]:
where
A and
B are the semi-empirical parameters as in Equation (15), and
q is a semi-empirical parameter determined to be
q ≈ 25.4 ± 2 [
20]. ∆
VE is the excess molar volume upon mixing, which can be neglected for simplicity when the data is not available.
Kaptay equation (K) [
22]:
where α is the parameter (α = 0.155 ± 0.015).
Seetharaman-Du Sichen equation (SDS) [
23]:
Noteworthily, the Kaptay unified equation model was originally constructed for pure metals and has been in good agreement with the previous experimental values. And, in this study, KU is a modification of the original model where
Mi,
Vi and
Tm,i are replaced by the integral system physical properties. In the calculation process, all thermodynamic parameters required for calculation in KU, BBK and H models were calculated by the Factsage 6.4 software. As the original equations in the literature [
22,
23] are not applicable to a molten iron system, the viscous flow activation energies in the K and SDS models were obtained directly by performing an Arrhenius linear fit of the experimental data.
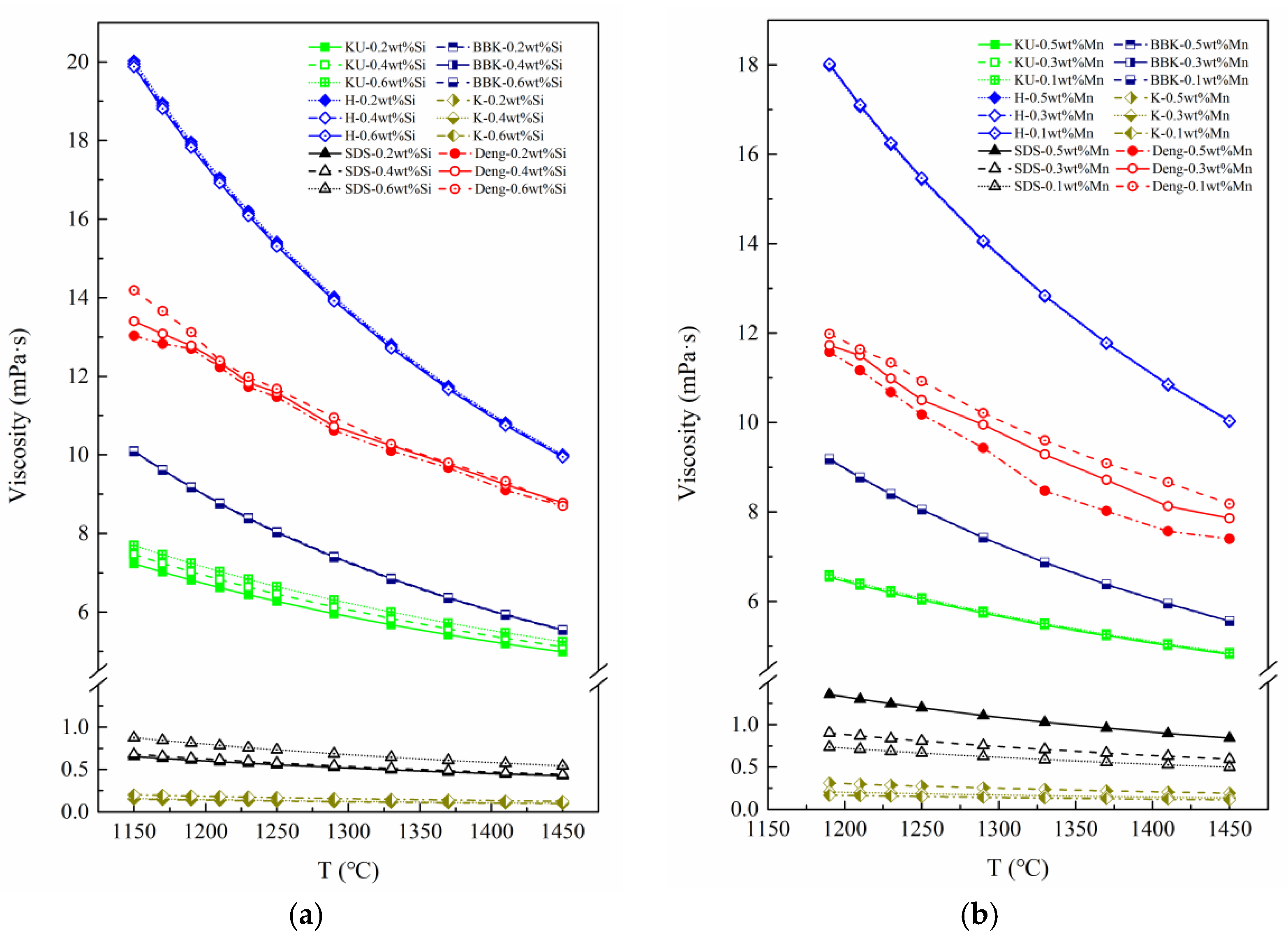
It is seen from
Figure 11 that the models (H, KU, BBK) that are based on the Andrade quasi-crystal theory [
24] agree well with the experimental data from the point of view of curve behavior, and the BBK model is the closest. In terms of numerical value comparison, the values calculated by the H model are larger than the measured values reported in the literature, while the values calculated by the BBK and KU models are slightly lower. The viscosity models based on the Eyring rate equation have good performance in predicting the melt of other alloys, but when predicting the viscosity of blast furnace molten iron, the SDS and K models differ greatly from the measured results in terms of both viscosity values and curve behavior.
Therefore, it can be considered that the three Andrade derivative equations [
6] are selected as the basic equations, which can be applied to the viscosity prediction of molten iron in a blast furnace with a greater precision.
The component action coefficients deduced in Equations (13) and (14) were introduced into the KU, BBK, and H semi-empirical viscosity equations; then, the following alternative molten iron viscosity prediction models were obtained:
H:
where the subscript I means that the equation introduced a positive action coefficient of carbon, and the subscript D means that the equation introduced a negative one.
5. Comparison of Experimental Results
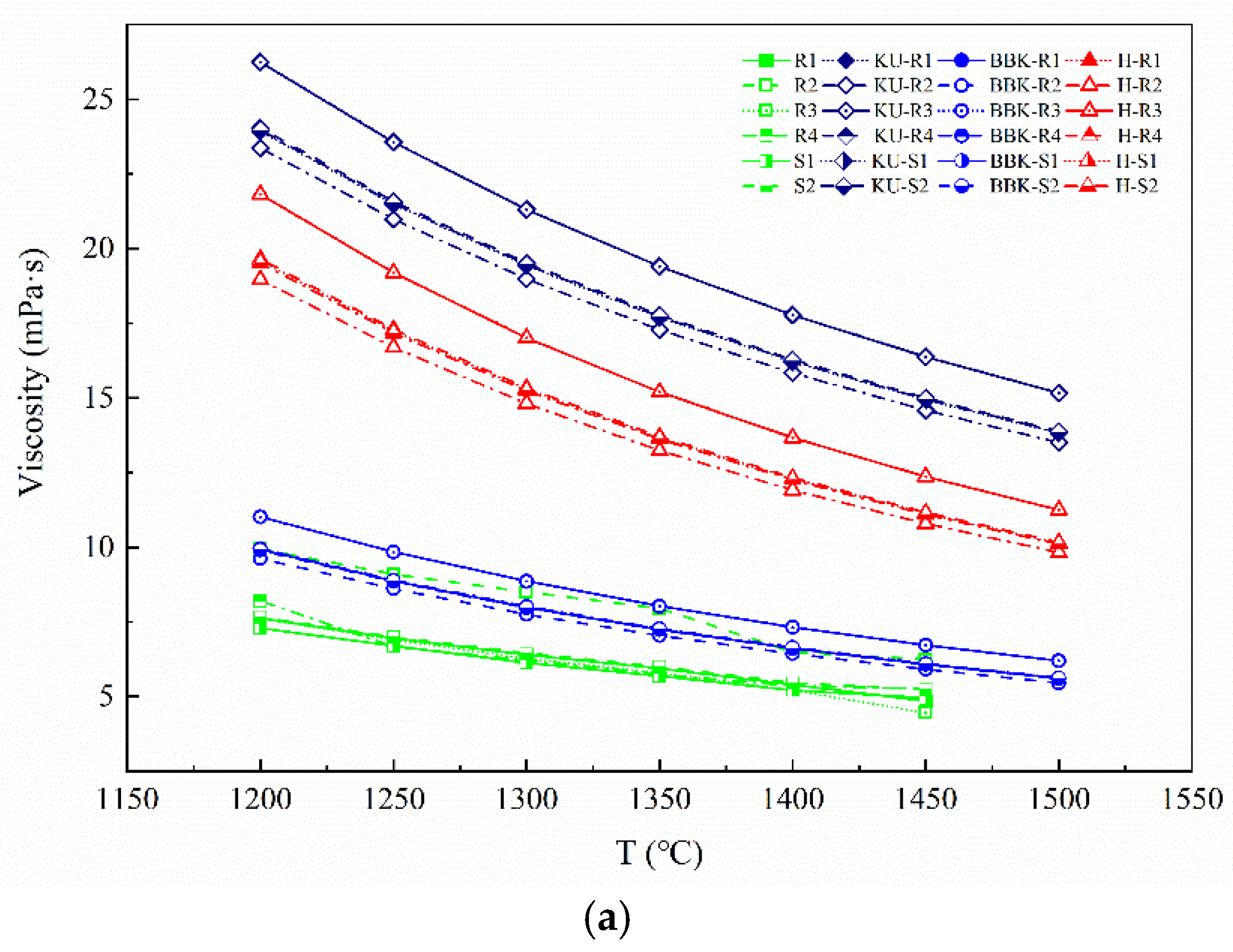
As shown in
Figure 12, the viscosities of actual molten iron samples decrease with the increase in temperature, and the temperature dependences of the viscosity for six samples are almost the same. Apparently, the viscosity value of R2 sample is greater than that of the other five groups of samples at various temperatures. As can be seen from
Figure 13, under high-temperature conditions, the isothermal viscosity curve relationship between the six groups of experimental molten iron did not change significantly.
The theoretical physicochemical properties and thermodynamic parameters of the industrial molten iron were calculated by Factsage 6.4 and introduced into the above three groups of improved models (20) to (25); then, the viscosity prediction results of the industrial molten iron were calculated.
As shown in
Figure 14a, the predicted values of industrial molten iron viscosity of the group I models are all larger than those of the experiment. Only the predicted results of BBK(I) are relatively close to the actual measured values with similar curve characteristics to those of the experiment. In contrast to the results from group I, the results of group D have an obvious better agreement with the results of the experiment in terms of both values and curve behavior. The predicted and measured isothermal viscosities of industrial molten iron at 1450 °C are plotted in
Figure 14c. It can be seen that the predicted results of the KU and H models of group I are approximately 7~11 mPa·s higher than the measured values, and they have the opposite viscosity curve characteristics. However, the curve characteristics demonstrated by the models of group D show good consistency with the measured results, especially for KU, which has three predictions almost identical to those of the experiment.
6. Discussion
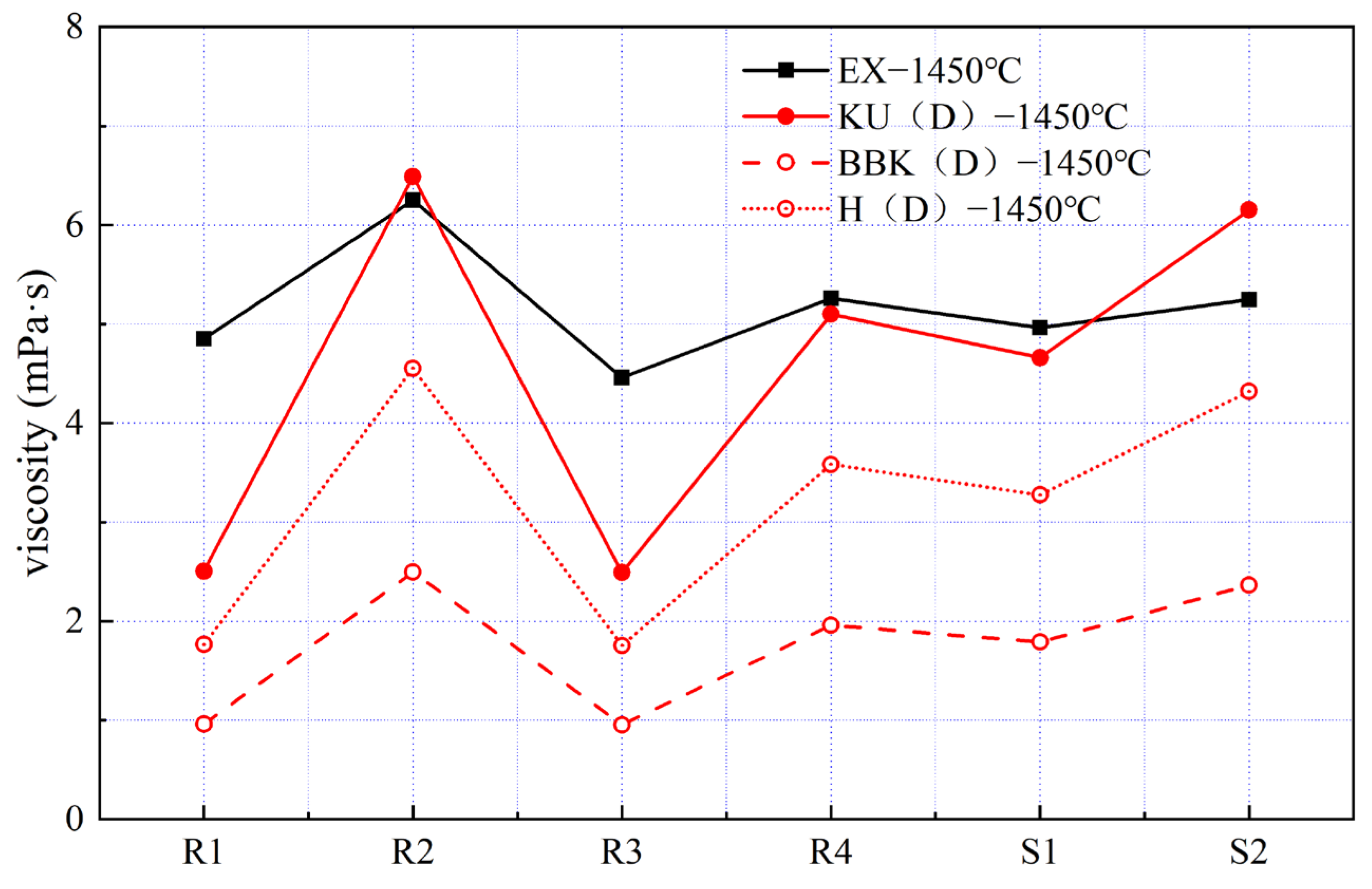
Based on the viscosity isotherms shown in
Figure 15, the performance of the three D-series models can be compared in terms of both numerical values and curve behavior. The measured and predicted isothermal viscosity values are presented in
Table 2, while the relative deviations between the predicted values from the three models and the experimental results were calculated using the variance formula, as shown in
Table 3.
Although the viscosity of the three samples (R2, R4 and S1) calculated by the KU (D) model was within 0.4 mPa·s of the measured values, the calculated viscosity of the other three samples was quite different from the actual measured values. It is worth mentioning that, although the viscosity values predicted by the H (D) model are lower than the experimentally measured values, the relative deviation of its curve characteristics is the smallest, indicating the superior stability of the predicted results. This improved stability can be attributed to the modified Hirai model being derived from the Andrade quasi-crystal theory, which provides a more robust description of the temperature dependence of viscous flow in metallic melts. And the viscosity curve predicted by the BBK (D) model is the most inconsistent with the experimental results, which may be because of the disregard of excess molar volume in the calculation.
However, to date, there is no quantitative definition of molten iron fluidity in blast furnace production. Therefore, understanding the fluidity behavior of molten iron is of relatively greater importance. The H (D) model, which introduces the component action coefficient, can not only predict the viscosity variation law of molten iron at different temperatures, but also successfully and stably predict the viscosity variational trend of molten iron in the case of composition content changes. In terms of current production demand, the H (D) model is selected as the best semi-empirical model for predicting the viscosity of molten iron in a blast furnace.
In recent studies on metallic melt viscosity, increasing attention has been paid to thermodynamic models and data-driven approaches to improve the prediction accuracy for complex alloy systems. Compared with these methods, the present work extends the classical semi-empirical viscosity models by introducing composition-dependent action coefficients, thereby retaining physical interpretability while enhancing the applicability to multi-component industrial molten iron.
7. Conclusions
To represent the viscosity of blast furnace molten iron as a function of the composition and temperature, five equations were utilized, and the viscosities of multi-component industrial molten iron were measured. The conclusions drawn are as follows:
- (1)
In the referred literature of the binary Fe-C viscosity, when [C%] > 4 wt%, the effects of components on viscosity can be divided into two main types. The carbon content of molten iron is the main factor that dominates the relationship between viscosity and temperature. The effect of the third components (Si, Mn, P, S, and Ti) is primarily reflected in the viscosity values rather than in the temperature dependence of viscosity.
- (2)
The models (H, KU, BBK) of group D based on the Andrade quasi-crystal theory are more suitable for predicting the viscosity of molten iron in a blast furnace.
- (3)
In terms of current production demand, the H (D) model can successfully and stably predict the viscosity variational behavior in the case of composition and temperature changes of the molten iron, which is the best semi-empirical model for predicting the viscosity of molten iron in a blast furnace.
Author Contributions
S.G.: conceived the research idea and designed the methodology and drafted the initial version of the manuscript. X.G.: performed data collection and contributed to data analysis and assisted in manuscript writing. X.Z.: performed statistical analysis and prepared figures and tables and contributed to manuscript revision. C.J.: provided academic supervision, guided research design, and contributed to the interpretation of results. Z.Z.: offered overall project guidance and reviewed and critically revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The funding was obtained from China Postdoctoral Science Foundation (Grant No. 2025M773597).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
Authors Shanchao Gao, Xu Geng and Zhenghong Zhao were employed by the Ansteel Group. Author Xiaobo Zhang was employed by Bayuquan Branch of Angang Steel Co., Ltd. Author Chuntang Jia was employed by Angang Steel Company Ltd. The company had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Jia, M. How to pay attention to and solve the problem of iron-carbon interface in blast furnace hearth. In 2012 National Blast Furnace Longevity and High Wind Temperature Technology Symposium Proceedings; The Chinese Society for Metals: Beijing, China, 2012. [Google Scholar]
- Jiao, K.; Zuo, H.; Xing, X.; Zhao, Y. Study on material phase and formation mechanism of viscous layer in blast furnace hearth. J. Northeast. Univ. Nat. Sci. 2014, 35, 987–991. [Google Scholar]
- Zhang, Y.; Zhang, Z.; Mao, X.; Du, H. Discussion on the phenomenon of water stick to tank in blast furnace containing ferrochrome. Baosteel Technol. 2015, 50–54. [Google Scholar]
- Guo, H. Viscous Behavior of Co-Based Alloy Melts. Master’s Thesis, Harbin Institute of Technology, Harbin, China, 2008. [Google Scholar]
- Brooks, R.; Dinsdale, A.; Quested, P. The measurement of viscosity of alloys—A review of methods, data and models. Meas. Sci. Technol. 2005, 16, 354. [Google Scholar] [CrossRef]
- Gao, S.; Jiao, K.; Zhang, J. Review of viscosity prediction models of liquid pure metals and alloys. Philos. Mag. 2019, 99, 853–868. [Google Scholar] [CrossRef]
- Moelwyn-Hughes, E.A.; Göhr, H.; Jaenicke, W.; Sabel, H.-D. Physikalische Chemie; Georg Thieme Verlag: Stuttgart, Germany, 1970. [Google Scholar]
- Morita, Z.; Iida, T.; Ueda, M. The Excess Viscosity of Liquid Binary Alloys. Liq. Met. 1977, 1976, 600–606. [Google Scholar]
- Kozlov, L.Y.; Romanov, L.; Petrov, N. Izv. vysch. uch. zav, Chernaya. Metallurgiya 1983, 3, 7–11. [Google Scholar]
- Wang, C. Development of Oscillating Viscometer for Magnetically Confined High Temperature Melt. Master’s Thesis, Shandong University, Shandong, China, 2017. [Google Scholar]
- Shvidkovskiy, Y.G. Certain Problems Related to the Viscosity of Fused Metals; National Aeronautics and Space Administration: Washington, DC, USA, 1962. [Google Scholar]
- Sato, Y.; Sugisawa, K.; Aoki, D.; Yamamura, T. Viscosities of Fe–Ni, Fe–Co and Ni–Co binary melts. Meas. Sci. Technol. 2005, 16, 363. [Google Scholar] [CrossRef]
- Kawai, Y.; Shiraishi, Y. Handbook of Physico-Chemical Properties at High Temperatures; The Iron and Steel Institute of Japan: Tokyo, Japan, 1988. [Google Scholar]
- Teng, X.-Y.; Wang, B.; Wang, Z.-M.; Geng, H.-R. Fluidity of Molten Binary Fe-xC Alloy. Foundry Technol. 2007, 28, 628. [Google Scholar]
- Ostrovski, O.; Belashchenko, D.K. Thermophysical Properties and Structure of Liquid Fe–C Alloys. High Temp. High Press. 2013, 42, 137–149. [Google Scholar]
- Takeda, O.; Ouchi, N.; Sato, Y. Viscosity of Molten Fe–B Alloy. ISIJ Int. 2015, 55, 500–503. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, J.; Jiao, K. Viscosity measurement and prediction model of molten iron. Ironmak. Steelmak. 2018, 45, 773–777. [Google Scholar] [CrossRef]
- Kaptay, G. A unified equation for the viscosity of pure liquid metals. Z. Für Met. 2005, 96, 24–31. [Google Scholar] [CrossRef]
- Budai, I.; Benkő, M.Z.; Kaptay, G. Comparison of different theoretical models to experimental data on viscosity of binary liquid alloys. Mater. Sci. Forum 2007, 537–538, 489–496. [Google Scholar] [CrossRef]
- Kaptay, G. Modelling interfacial energies in metallic systems. Mater. Sci. Forum 2005, 473–474, 1–10. [Google Scholar] [CrossRef]
- Hirai, M. Estimation of viscosities of liquid alloys. ISIJ Int. 1993, 33, 251–258. [Google Scholar] [CrossRef]
- Kaptay, G. A new equation to estimate the concentration dependence of the viscosity of liquid metallic alloys from the heat of mixing data. In Proceedings of the MicroCAD 2003 International Conference, Miskolc, Hungary, 6–7 March 2003; p. 23. [Google Scholar]
- Seetharaman, S.; Sichen, D. Estimation of the viscosities of binary metallic melts using Gibbs energies of mixing. Metall. Mater. Trans. B 1994, 25, 589–595. [Google Scholar] [CrossRef]
- Andrade, E.D.C. LVIII. A theory of the viscosity of liquids.—Part II. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1934, 17, 698–732. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |