Fortification of the Bioactive and Sensory Profile of Dark Cup Chocolate Formulated with Three Percentages of Cocoa Liquor (Forastero Variety)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Material
2.1.1. Materials
2.1.2. Samples
2.2. Processing to Obtain Dark Cup Chocolate
2.3. Conditioning of Dark Chocolate Samples for Cups
2.4. Antioxidant Capacity
2.4.1. ABTS Assay
2.4.2. DPPH Assay
2.4.3. FRAP Assay
2.5. Total Phenol Content (TPC)
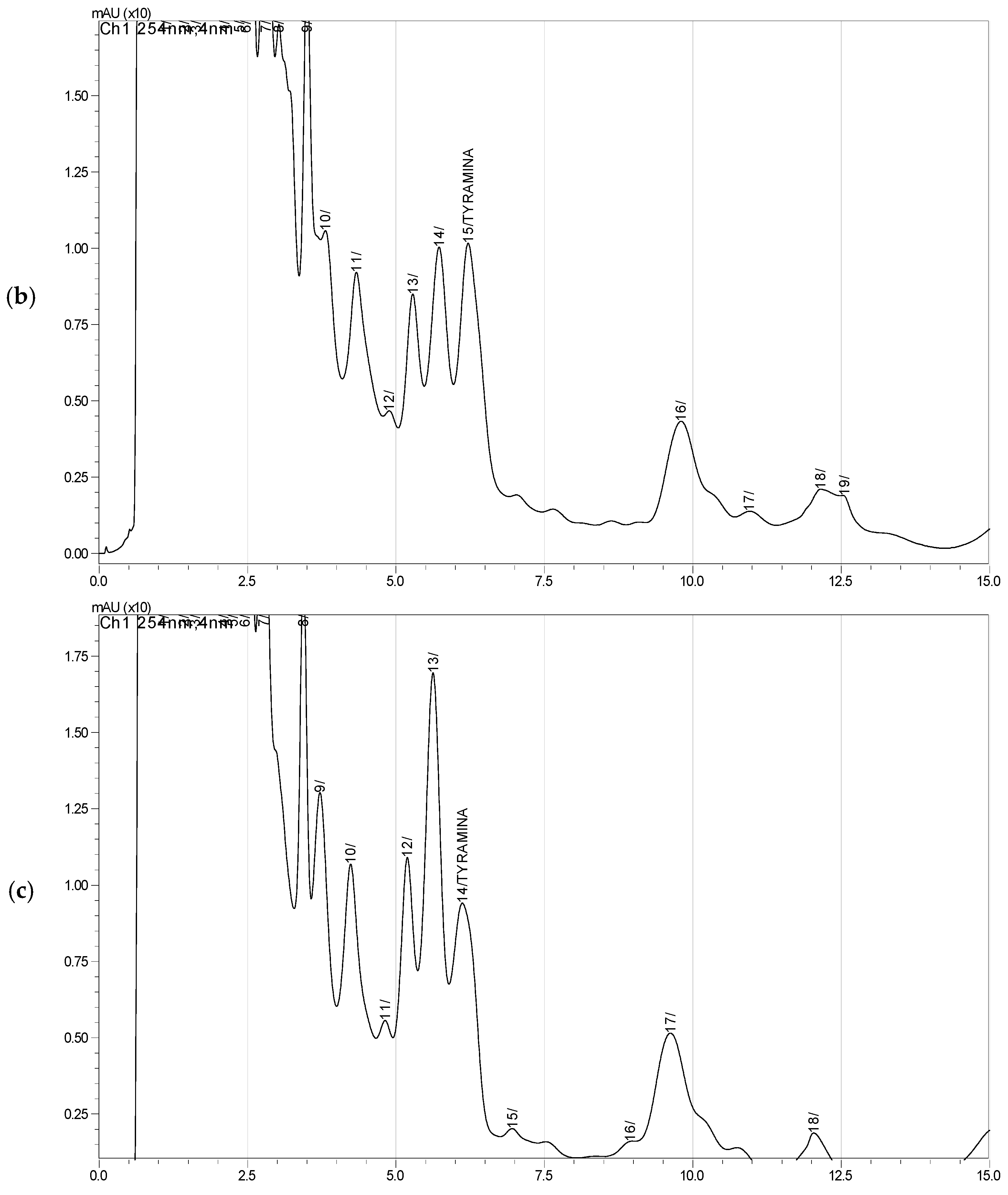
2.6. Tyramine Quantification
2.7. Sensory Profile
2.8. Statistical Analysis
3. Results and Discussion
3.1. Bioactive Profile in Dark Cup Chocolate with Three Percentages of Cocoa Liquor
3.2. Sensory Profile in Dark Cup Chocolate with Three Percentages of Cocoa Liquor
3.3. Correlation Between Bioactive and Sensory Properties in Dark Cup Chocolate in Three Percentages of Cocoa Liquor
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Delgado-Ospina, J.; Di Mattia, C.D.; Paparella, A.; Mastrocola, D.; Martuscelli, M.; Chaves-Lopez, C. Effect of Fermentation, Drying and Roasting on Biogenic Amines and Other Biocompounds in Colombian Criollo Cocoa Beans and Shells. Foods 2020, 9, 520. [Google Scholar] [CrossRef]
- Gil, M.; Uribe, D.; Gallego, V.; Bedoya, C.; Arango-Varela, S. Traceability of Polyphenols in Cocoa during the Postharvest and Industrialization Processes and Their Biological Antioxidant Potential. Heliyon 2021, 7, e07738. [Google Scholar] [CrossRef] [PubMed]
- Anoraga, S.B.; Shamsudin, R.; Hamzah, M.H.; Sharif, S.; Saputro, A.D. Cocoa By-Products: A Comprehensive Review on Potential Uses, Waste Management, and Emerging Green Technologies for Cocoa Pod Husk Utilization. Heliyon 2024, 10, e35537. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, M.B.; Alencar, J.C.G.; Paulino, B.N.; Nascimento, J.C.N.; Ferreira, T.R.; Batista, A.S.; Nascimento, M.M.; Soares, S.E.; Mesquita, P.R.R. Functional and technological potential of by-products from the cocoa (Theobroma cacao L.) production chain. Food Chem. 2025, 493, 145718. [Google Scholar] [CrossRef]
- Tuenter, E.; Sakavitsi, M.E.; Rivera-Mondragón, A.; Hermans, N.; Foubert, K.; Halabalaki, M.; Pieters, L. Ruby Chocolate: A Study of Its Phytochemical Composition and Quantitative Comparison with Dark, Milk and White Chocolate. Food Chem. 2021, 343, 128446. [Google Scholar] [CrossRef]
- Cherniienko, A.; Pawełczyk, A.; Zaprutko, L. Antimicrobial and Odour Qualities of Alkylpyrazines Occurring in Chocolate and Cocoa Products. Appl. Sci. 2022, 12, 11361. [Google Scholar] [CrossRef]
- Paparella, A.; Schirone, M.; López, C.C. The Health Impact of Cocoa from Cultivation to the Formation of Biogenic Amines: An Updated Review. Foods 2025, 14, 255. [Google Scholar] [CrossRef]
- Castro-Alayo, E.M.; Idrogo-Vásquez, G.; Siche, R.; Cardenas-Toro, F.P. Formation of Aromatic Compounds Precursors during Fermentation of Criollo and Forastero Cocoa. Heliyon 2019, 5, e01157. [Google Scholar] [CrossRef]
- Erazo Solórzano, C.Y.; Bravo Franco, K.J.; Tuárez García, D.A.; Fernández Escobar, Á.O.; Torres Navarrete, Y.; Vera, J. Efecto de la fermentación de cacao (Theobroma cacao L.), variedad nacional y trinitario, en cajas de maderas no convencionales sobre la calidad física y sensorial del licor de cacao. Rev. Investig. Talent. 2021, 8, 42–55. [Google Scholar]
- Duana-Ávila, D.; Hernández-Gracía, T.J.; Martínez-Muñoz, E.; García-Velázquez, M.d.R.; Román-Gutiérrez, A.D. Study of the Mexican Cocoa Market: An Analysis of Its Competitiveness (2010–2021). Agronomy 2023, 13, 378. [Google Scholar] [CrossRef]
- Kongor, J.E.; Owusu, M.; Oduro-Yeboah, C. Cocoa Production in the 2020s: Challenges and Solutions. CABI Agric. Biosci. 2024, 5, 102. [Google Scholar] [CrossRef]
- Wahyuni, N.L.; Sunarharum, W.B.; Muhammad, D.R.A.; Saputro, A.D. Formation and Development of Flavour of Cocoa (Theobroma cacao L.) Cultivar Criollo and Forastero: A Review. IOP Conf. Ser. Earth Environ. Sci. 2021, 733, 012078. [Google Scholar] [CrossRef]
- Putri, D.N.; De Steur, H.; Juvinal, J.G.; Gellynck, X.; Schouteten, J.J. Sensory Attributes of Fine Flavor Cocoa Beans and Chocolate: A Systematic Literature Review. J. Food Sci. 2024, 89, 1917–1943. [Google Scholar] [CrossRef] [PubMed]
- do Carmo Brito, B.d.N.; Campos Chisté, R.; da Silva Pena, R.; Abreu Gloria, M.B.; Santos Lopes, A. Bioactive Amines and Phenolic Compounds in Cocoa Beans Are Affected by Fermentation. Food Chem. 2017, 228, 484–490. [Google Scholar] [CrossRef]
- Silveira, P.T.D.S.; Glória, M.B.A.; Tonin, I.P.; Martins, M.O.P.; Efraim, P. Varietal Influence on the Formation of Bioactive Amines during the Processing of Fermented Cocoa with Different Pulp Contents. Foods 2023, 12, 495. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ) Scientific Opinion on Risk Based Control of Biogenic Amine Formation in Fermented Foods. EFSA J. 2011, 9, 2393–2486. [CrossRef]
- Ruiz-Capillas, C.; Herrero, A.M. Impact of Biogenic Amines on Food Quality and Safety. Foods 2019, 8, 62. [Google Scholar] [CrossRef]
- McCabe-Sellers, B.J.; Staggs, C.G.; Bogle, M.L. Tyramine in Foods and Monoamine Oxidase Inhibitor Drugs: A Crossroad Where Medicine, Nutrition, Pharmacy, and Food Industry Converge. J. Food Compos. Anal. 2006, 19, S58–S65. [Google Scholar] [CrossRef]
- Singh, P.K.; Khedkar, R.D.; Chandra, S. Chocolate: An Overview of Functional Potential and Recent Trends in Fortification. Braz. J. Food Technol. 2024, 27, e2023118. [Google Scholar] [CrossRef]
- Sun, P.; Xia, B.; Ni, Z.-J.; Wang, Y.; Elam, E.; Thakur, K.; Ma, Y.-L.; Wei, Z.-J. Characterization of Functional Chocolate Formulated Using Oleogels Derived from β-Sitosterol with γ-Oryzanol/Lecithin/Stearic Acid. Food Chem. 2021, 360, 130017. [Google Scholar] [CrossRef] [PubMed]
- Żyżelewicz, D.; Budryn, G.; Oracz, J.; Antolak, H.; Kręgiel, D.; Kaczmarska, M. The Effect on Bioactive Components and Characteristics of Chocolate by Functionalization with Raw Cocoa Beans. Food Res. Int. 2018, 113, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Dala-Paula, B.M.; Starling, M.d.F.V.; Gloria, M.B.A. Vegetables Consumed in Brazilian Cuisine as Sources of Bioactive Amines. Food Biosci. 2021, 40, 100856. [Google Scholar] [CrossRef]
- Deus, V.L.; Bispo, E.S.; Franca, A.S.; Gloria, M.B.A. Influence of Cocoa Clones on the Quality and Functional Properties of Chocolate – Nitrogenous Compounds. LWT 2020, 134, 110202. [Google Scholar] [CrossRef]
- Deus, V.L.; Bispo, E.S.; Franca, A.S.; Gloria, M.B.A. Understanding Amino Acids and Bioactive Amines Changes during On-Farm Cocoa Fermentation. J. Food Compos. Anal. 2021, 97, 103776. [Google Scholar] [CrossRef]
- Faccinetto-Beltrán, P.; Gómez-Fernández, A.R.; Santacruz, A.; Jacobo-Velázquez, D.A. Chocolate as Carrier to Deliver Bioactive Ingredients: Current Advances and Future Perspectives. Foods 2021, 10, 2065. [Google Scholar] [CrossRef]
- Ostrowska-Ligęza, E.; Górska, A.; Wirkowska-Wojdyła, M.; Bryś, J.; Dolatowska-Żebrowska, K.; Shamilowa, M.; Ratusz, K. Thermogravimetric Characterization of Dark and Milk Chocolates at Different Processing Stages. J. Therm. Anal. Calorim. 2018, 134, 623–631. [Google Scholar] [CrossRef]
- Eisenberg, T.; Abdellatif, M.; Schroeder, S.; Primessnig, U.; Stekovic, S.; Pendl, T.; Harger, A.; Schipke, J.; Zimmermann, A.; Schmidt, A.; et al. Cardioprotection and Lifespan Extension by the Natural Polyamine Spermidine. Nat. Med. 2016, 22, 1428–1438. [Google Scholar] [CrossRef]
- Liang, Y.; Piao, C.; Beuschel, C.B.; Toppe, D.; Kollipara, L.; Bogdanow, B.; Maglione, M.; Lützkendorf, J.; See, J.C.K.; Huang, S.; et al. eIF5A Hypusination, Boosted by Dietary Spermidine, Protects from Premature Brain Aging and Mitochondrial Dysfunction. Cell Rep. 2021, 35, 108941. [Google Scholar] [CrossRef]
- Muñoz-Esparza, N.C.; Latorre-Moratalla, M.L.; Comas-Basté, O.; Toro-Funes, N.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Polyamines in Food. Front. Nutr. 2019, 6, 108. [Google Scholar] [CrossRef]
- Sadowska-Rociek, A. Monitoring of Monochloropropanediol Esters and Glycidyl Esters in Different Types of Chocolates Using the Modified Sample Preparation Approach. LWT 2022, 165, 113692. [Google Scholar] [CrossRef]
- Zugravu, C.; Otelea, M.R. Dark Chocolate: To Eat or Not to Eat? A Review. J. AOAC Int. 2019, 102, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, C.; Gökmen, V. Neuroactive Compounds in Foods: Occurrence, Mechanism and Potential Health Effects. Food Res. Int. 2020, 128, 108744. [Google Scholar] [CrossRef]
- Delgado-Ospina, J.; Acquaticci, L.; Molina-Hernandez, J.B.; Rantsiou, K.; Martuscelli, M.; Kamgang-Nzekoue, A.F.; Vittori, S.; Paparella, A.; Chaves-López, C. Exploring the Capability of Yeasts Isolated from Colombian Fermented Cocoa Beans to Form and Degrade Biogenic Amines in a Lab-Scale Model System for Cocoa Fermentation. Microorganisms 2021, 9, 28. [Google Scholar] [CrossRef]
- Restuccia, D.; Spizzirri, U.G.; Puoci, F.; Picci, N. Determination of Biogenic Amine Profiles in Conventional and Organic Cocoa-Based Products. Food Addit. Contam. Part A 2015, 32, 1156–1163. [Google Scholar] [CrossRef]
- Restuccia, D.; Spizzirri, U.G.; Luca, M.D.; Parisi, O.I.; Picci, N. Biogenic Amines as Quality Marker in Organic and Fair-Trade Cocoa-Based Products. Sustainability 2016, 8, 856. [Google Scholar] [CrossRef]
- Spizzirri, U.G.; Ieri, F.; Campo, M.; Paolino, D.; Restuccia, D.; Romani, A. Biogenic Amines, Phenolic, and Aroma-Related Compounds of Unroasted and Roasted Cocoa Beans with Different Origin. Foods 2019, 8, 306. [Google Scholar] [CrossRef] [PubMed]
- Britto, A.d.A.; Lins da Cruz, M.; Antonia de Souza Oliveira, F.; Soares, S.E.; Druzian, J.I.; Radomille de Santana, L.R.; Oliveira de Souza, C.; da Silva Bispo, E. Influence of Under-Fermented Cocoa Mass in Chocolate Production: Sensory Acceptance and Volatile Profile Characterization during the Processing. LWT 2021, 149, 112048. [Google Scholar] [CrossRef]
- Gültekin-Özgüven, M.; Berktaş, İ.; Özçelik, B. Influence of Processing Conditions on Procyanidin Profiles and Antioxidant Capacity of Chocolates: Optimization of Dark Chocolate Manufacturing by Response Surface Methodology. LWT 2016, 66, 252–259. [Google Scholar] [CrossRef]
- López, Y.G.; Vásquez Vega, J.T.; Rosillo, F.F.; Castro Alayo, E.M.; Cueva Ríos, M.A.; Huatangari, L.Q.; Mendoza, M.M. Predicting the Shelf Life of Cup Chocolate Using the Arrhenius Model Based on Peroxide Value. Math. Model. Eng. Probl. 2024, 11, 517. Available online: https://openurl.ebsco.com/contentitem/doi:10.18280%2Fmmep.110224?sid=ebsco:plink:crawler&id=ebsco:doi:10.18280%2Fmmep.110224 (accessed on 27 August 2025). [CrossRef]
- Mursu, J.; Voutilainen, S.; Nurmi, T.; Rissanen, T.H.; Virtanen, J.K.; Kaikkonen, J.; Nyyssönen, K.; Salonen, J.T. Dark Chocolate Consumption Increases HDL Cholesterol Concentration and Chocolate Fatty Acids May Inhibit Lipid Peroxidation in Healthy Humans. Free Radic. Biol. Med. 2004, 37, 1351–1359. [Google Scholar] [CrossRef]
- Torres-Moreno, M.; Torrescasana, E.; Salas-Salvadó, J.; Blanch, C. Nutritional Composition and Fatty Acids Profile in Cocoa Beans and Chocolates with Different Geographical Origin and Processing Conditions. Food Chem. 2015, 166, 125–132. [Google Scholar] [CrossRef]
- Galleano, M.; Oteiza, P.I.; Fraga, C.G. Cocoa, Chocolate, and Cardiovascular Disease. J. Cardiovasc. Pharmacol. 2009, 54, 483. [Google Scholar] [CrossRef]
- Dala-Paula, B.M.; Custódio, F.B.; Gloria, M.B. Health Concerns Associated with Biogenic Amines in Food and Interaction with Amine Oxidase Drugs. Curr. Opin. Food Sci. 2023, 54, 101090s. [Google Scholar] [CrossRef]
- Edinoff, A.N.; Swinford, C.R.; Odisho, A.S.; Burroughs, C.R.; Stark, C.W.; Raslan, W.A.; Cornett, E.M.; Kaye, A.M.; Kaye, A.D. Clinically Relevant Drug Interactions with Monoamine Oxidase Inhibitors. Health Psychol. Res. 2022, 10, 39576. [Google Scholar] [CrossRef] [PubMed]
- Canlı, M.; Çelik, E.E.; Kocadağlı, T.; Kanmaz, E.Ö.; Gökmen, V. Formation of Bioactive Tyrosine Derivatives during Sprouting and Fermenting of Selected Whole Grains. J. Agric. Food Chem. 2021, 69, 12517–12526. [Google Scholar] [CrossRef]
- Lan, Q.; Pei, H.; Deng, L.; Zhang, Y.; Yang, L.; He, W.; Ma, Y.; Li, J.; Yang, Y. Research Progress of Tyramine Formation and Control Methods in Fermented Meat Products. Sci. Technol. Food Ind. 2024, 45, 380–388. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, X.; Guo, M.; Liu, J.; Kang, M. The Key Tyrosine Decarboxylase Gene and Its Negative Transcription Factor for GX-50 Biosynthesis in Sichuan Pepper (Zanthoxylum Armatum). Ind. Crops Prod. 2024, 210, 118128. [Google Scholar] [CrossRef]
- CXS 87-1981; Standard for Chocolate and Chocolate Products. Codex Alimentarius Commission: Rome, Italy, 1981.
- Fernández-Romero, E.; Chavez-Quintana, S.G.; Siche, R.; Castro-Alayo, E.M.; Cardenas-Toro, F.P. The Kinetics of Total Phenolic Content and Monomeric Flavan-3-Ols during the Roasting Process of Criollo Cocoa. Antioxidants 2020, 9, 146. [Google Scholar] [CrossRef]
- Summa, C.; Raposo, F.C.; McCourt, J.; Scalzo, R.L.; Wagner, K.-H.; Elmadfa, I.; Anklam, E. Effect of Roasting on the Radical Scavenging Activity of Cocoa Beans. Eur. Food Res. Technol. 2006, 222, 368–375. [Google Scholar] [CrossRef]
- Zhou, S.; Seo, S.; Alli, I.; Chang, Y.-W. Interactions of Caseins with Phenolic Acids Found in Chocolate. Food Res. Int. 2015, 74, 177–184. [Google Scholar] [CrossRef]
- de Souza, V.R.; Pereira, P.A.P.; da Silva, T.L.T.; de Oliveira Lima, L.C.; Pio, R.; Queiroz, F. Determination of the Bioactive Compounds, Antioxidant Activity and Chemical Composition of Brazilian Blackberry, Red Raspberry, Strawberry, Blueberry and Sweet Cherry Fruits. Food Chem. 2014, 156, 362–368. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Ramos-Escudero, F.; Casimiro-Gonzales, S.; Fernández-Prior, Á.; Cancino Chávez, K.; Gómez-Mendoza, J.; de la Fuente-Carmelino, L.; Muñoz, A.M. Colour, Fatty Acids, Bioactive Compounds, and Total Antioxidant Capacity in Commercial Cocoa Beans (Theobroma cacao L.). LWT 2021, 147, 111629. [Google Scholar] [CrossRef]
- Melo, T.S.; Pires, T.C.; Engelmann, J.V.P.; Monteiro, A.L.O.; Maciel, L.F.; Bispo, E.d.S. Evaluation of the Content of Bioactive Compounds in Cocoa Beans during the Fermentation Process. J. Food Sci. Technol. 2021, 58, 1947–1957. [Google Scholar] [CrossRef] [PubMed]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant Activity of Dietary Polyphenols As Determined by a Modified Ferric Reducing/Antioxidant Power Assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In Methods in Enzymology; Oxidants and Antioxidants Part A; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178.s. [Google Scholar]
- Llerena, W.; Samaniego, I.; Vallejo, C.; Arreaga, A.; Zhunio, B.; Coronel, Z.; Quiroz, J.; Angós, I.; Carrillo, W. Profile of Bioactive Components of Cocoa (Theobroma cacao L.) By-Products from Ecuador and Evaluation of Their Antioxidant Activity. Foods 2023, 12, 2583. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.; Sun, Y.N.; Liu, L.; Yao, F.Y.; Zhang, Y.; Guo, F.Y.; Zhao, W.J.; Liu, J.L.; Zhang, N. Determination of Biogenic Amines in Different Parts of Lycium Barbarum L. by HPLC with Precolumn Dansylation. Molecules 2021, 26, 1046. [Google Scholar] [CrossRef]
- Chiacchierini, E.; Restuccia, D.; Vinci, G. Evaluation of Two Different Extraction Methods for Chromatographic Determination of Bioactive Amines in Tomato Products. Talanta 2006, 69, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Murcia-Artunduaga, K.; Gasca-Torres, L.; Castañeda, M.d.R. Evaluación físico-sensorial de granos de cacao (Theobroma cacao L.), región sur del Huila (Colombia). Inf. Técnico 2022, 86, 194–204. [Google Scholar] [CrossRef]
- Jaćimović, S.; Popović-Djordjević, J.; Sarić, B.; Krstić, A.; Mickovski-Stefanović, V.; Pantelić, N.Đ. Antioxidant Activity and Multi-Elemental Analysis of Dark Chocolate. Foods 2022, 11, 1445. [Google Scholar] [CrossRef]
- Caponio, G.R.; Lorusso, M.P.; Sorrenti, G.T.; Marcotrigiano, V.; Difonzo, G.; De Angelis, E.; Guagnano, R.; Ciaula, A.D.; Diella, G.; Logrieco, A.F.; et al. Chemical Characterization, Gastrointestinal Motility and Sensory Evaluation of Dark Chocolate: A Nutraceutical Boosting Consumers’ Health. Nutrients 2020, 12, 939. [Google Scholar] [CrossRef]
- Bibi Sadeer, N.; Montesano, D.; Albrizio, S.; Zengin, G.; Mahomoodally, M.F. The Versatility of Antioxidant Assays in Food Science and Safety—Chemistry, Applications, Strengths, and Limitations. Antioxidants 2020, 9, 709. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef] [PubMed]
- Abeyrathne, E.D.N.S.; Nam, K.; Ahn, D.U. Analytical Methods for Lipid Oxidation and Antioxidant Capacity in Food Systems. Antioxidants 2021, 10, 1587. [Google Scholar] [CrossRef]
- Miller, K.B.; Stuart, D.A.; Smith, N.L.; Lee, C.Y.; McHale, N.L.; Flanagan, J.A.; Ou, B.; Hurst, W.J. Antioxidant Activity and Polyphenol and Procyanidin Contents of Selected Commercially Available Cocoa-Containing and Chocolate Products in the United States. J. Agric. Food Chem. 2006, 54, 4062–4068. [Google Scholar] [CrossRef] [PubMed]
- Todorovic, V.; Redovnikovic, I.R.; Todorovic, Z.; Jankovic, G.; Dodevska, M.; Sobajic, S. Polyphenols, Methylxanthines, and Antioxidant Capacity of Chocolates Produced in Serbia. J. Food Compos. Anal. 2015, 41, 137–143. [Google Scholar] [CrossRef]
- Santos, G.; Dala-Paula, B.M.; Bispo, E.S.; Gloria, M.B.A. Bioaccessibility of Bioactive Amines in Dark Chocolates Made with Different Proportions of Under-Fermented and Fermented Cocoa Beans. Food Chem. 2023, 404, 134725. [Google Scholar] [CrossRef]
- Dala-Paula, B.M.; Deus, V.L.; Tavano, O.L.; Glória, M.B.A. Bioactive Amines in 70% Cocoa Dark Chocolate: What You Eat and What You Get. Food Chem. 2021, 343, 128397. [Google Scholar] [CrossRef]
- Carpéné, C.; Viana, P.; Fontaine, J.; Laurell, H.; Grolleau, J.-L. Multiple Direct Effects of the Dietary Protoalkaloid N-Methyltyramine in Human Adipocytes. Nutrients 2022, 14, 3118. [Google Scholar] [CrossRef]
- Melfi, F.; Carradori, S.; Angeli, A.; D’Agostino, I. Nature as a Source and Inspiration for Human Monoamine Oxidase B (hMAO-B) Inhibition: A Review of the Recent Advances in Chemical Modification of Natural Compounds. Expert Opin. Drug Discov. 2023, 18, 851–879. [Google Scholar] [CrossRef]
- Oracz, J.; Nebesny, E. Influence of Roasting Conditions on the Biogenic Amine Content in Cocoa Beans of Different Theobroma cacao Cultivars. Food Res. Int. 2014, 55, 1–10. [Google Scholar] [CrossRef]
- Lang, S.; Liu, L.; Li, Z.; Liu, S.; Liang, J.; Lu, L.; Wang, L. Untargeted Metabolomics Reveals Phenolic Compound Dynamics during Mung Bean Fermentation. Food Chem. X 2025, 31, 103189. [Google Scholar] [CrossRef] [PubMed]
- Aprotosoaie, A.C.; Luca, S.V.; Miron, A. Flavor Chemistry of Cocoa and Cocoa Products-An Overview: Flavor Chemistry of Cocoa and Cocoa Products—An Overview. Compr. Rev. Food Sci. Food Saf. 2016, 15, 73–91. [Google Scholar] [CrossRef]
- Giacometti, J.; Jolić, S.M.; Josić, D. Cocoa Processing and Impact on Composition. In Processing and Impact on Active Components in Food; Elsevier: Amsterdam, The Netherlands, 2015; pp. 605–612. ISBN 978-0-12-404699-3. [Google Scholar]
- Kongor, J.E.; Hinneh, M.; De Walle, D.V.; Afoakwa, E.O.; Boeckx, P.; Dewettinck, K. Factors Influencing Quality Variation in Cocoa (Theobroma cacao) Bean Flavour Profile — A Review. Food Res. Int. 2016, 82, 44–52. [Google Scholar] [CrossRef]
- Thamke, I.; Dürrschmid, K.; Rohm, H. Sensory Description of Dark Chocolates by Consumers. LWT 2009, 42, 534–539. [Google Scholar] [CrossRef]
- Sari, I.A.; Murti, R.H.M.; Misnawi, M.; Putra, E.T.S.P.; Susilo, A.W.S. Sensory Profiles of Cocoa Genotypes in Indonesia. Biodivers. J. Biol. Divers. 2022, 23, 648–654. [Google Scholar] [CrossRef]
- Oberrauter, L.-M.; Januszewska, R.; Schlich, P.; Majchrzak, D. Sensory Evaluation of Dark Origin and Non-Origin Chocolates Applying Temporal Dominance of Sensations (TDS). Food Res. Int. 2018, 111, 39–49. [Google Scholar] [CrossRef] [PubMed]
- McClure, A.P.; Hopfer, H.; Grün, I.U. Optimizing Consumer Acceptability of 100% Chocolate through Roasting Treatments and Effects on Bitterness and Other Important Sensory Characteristics. Curr. Res. Food Sci. 2022, 5, 167–174. [Google Scholar] [CrossRef] [PubMed]






| Input | Specification | 100% | 70% | 40% | |||
|---|---|---|---|---|---|---|---|
| % | Kg | % | Kg | % | Kg | ||
| Cocoa liquor | ≥35% | 100 | 2.60 | 70 | 1.820 | 40 | 1.040 |
| Cocoa butter | ≥18% CDE | -- | -- | 8 | 0.208 | 3 | 0.078 |
| Starch | <8% | -- | -- | 7 | 0.182 | 7 | 0.182 |
| Dry lean cocoa extract * | ≥14% CDE | -- | -- | 15 | 0.390 | 50 | 1.300 |
| Total | 2.60 | 2.60 | 2.60 | ||||
| Descriptors | Operational Definition |
|---|---|
| Smell/Aroma | Intensity of aromas perceived through direct sniffing |
| Cocoa Flavor | Intensity of typical processed cocoa character |
| Bitterness | Bitter sensation on tongue (back, sides) |
| Sweetness | Global perception of sweet notes from any source |
| Acidity | Sour sensation (lateral salivation) |
| Astringency | Dryness/roughness sensation on oral mucosa |
| Floral Flavor | Floral aromas (white flowers, jasmine, rose) |
| Spicy Flavor | Spice aromas (cinnamon, nutmeg, clove) |
| Fresh fruit Flavor | Fresh fruit (citrus, berries) or dried fruit (raisin, fig) |
| Nutty Flavor | Nutty aromas (hazelnut, almond, walnut) |
| Defect | Off notes (vinegary, ammoniacal, moldy, rancid) |
| Aftertaste | Duration and intensity of post-swallow sensations |
| Dried Fruit Flavor | Sweet, concentrated aromas reminiscent of dehydrated fruits (raisins, dates, figs, prunes) |
| Overall Impression | Global quality and acceptability assessment |
| Sample | ABTS (µmol TE/g) | DPPH (µmol TE/g) | FRAP (µmol Fe2+/g) | TPC (mg GAE/g) |
|---|---|---|---|---|
| CHO–40% | 96.46 c ± 2.17 | 64.35 b ± 2.27 | 61.89 c ± 2.46 | 19.00 b ± 0.23 |
| CHO–70% | 176.37 b ± 3.71 | 202.06 ab ± 3.80 | 126.78 b ± 2.46 | 19.80 ab ± 0.16 |
| CHO–100% | 273.36 a ± 4.02 | 278.85 a ± 2.72 | 213.07 a ± 5.13 | 27.32 a ± 0.07 |
| Sample | Retention Time | Area | Concentration (mg/L) | Concentration (mg/kg) |
|---|---|---|---|---|
| CHO–40% | 5.82 ± 0.02 | 9980 ± 862 | 0.37 b ± 0.03 | 1.85 b ± 0.03 |
| CHO–70% | 6.16 ± 0.05 | 184,366 ± 5323 | 6.83 ab ± 0.20 | 34.15 ab ± 0.20 |
| CHO–100% | 6.12 ± 0.00 | 226,179 ± 15,870 | 8.38 a ± 0.59 | 41.90 a ± 0.59 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cabrejos-Barrios, E.M.; Fernandez-Rosillo, F.; León-Roque, N.; Cabrejos-Barrios, A.S.; Medina-Mendoza, M.; Castro-Alayo, E.M.; Balcázar-Zumaeta, C.R. Fortification of the Bioactive and Sensory Profile of Dark Cup Chocolate Formulated with Three Percentages of Cocoa Liquor (Forastero Variety). Processes 2026, 14, 697. https://doi.org/10.3390/pr14040697
Cabrejos-Barrios EM, Fernandez-Rosillo F, León-Roque N, Cabrejos-Barrios AS, Medina-Mendoza M, Castro-Alayo EM, Balcázar-Zumaeta CR. Fortification of the Bioactive and Sensory Profile of Dark Cup Chocolate Formulated with Three Percentages of Cocoa Liquor (Forastero Variety). Processes. 2026; 14(4):697. https://doi.org/10.3390/pr14040697
Chicago/Turabian StyleCabrejos-Barrios, Eliana Milagros, Frank Fernandez-Rosillo, Noemí León-Roque, Aleida Soledad Cabrejos-Barrios, Marleni Medina-Mendoza, Efraín M. Castro-Alayo, and César R. Balcázar-Zumaeta. 2026. "Fortification of the Bioactive and Sensory Profile of Dark Cup Chocolate Formulated with Three Percentages of Cocoa Liquor (Forastero Variety)" Processes 14, no. 4: 697. https://doi.org/10.3390/pr14040697
APA StyleCabrejos-Barrios, E. M., Fernandez-Rosillo, F., León-Roque, N., Cabrejos-Barrios, A. S., Medina-Mendoza, M., Castro-Alayo, E. M., & Balcázar-Zumaeta, C. R. (2026). Fortification of the Bioactive and Sensory Profile of Dark Cup Chocolate Formulated with Three Percentages of Cocoa Liquor (Forastero Variety). Processes, 14(4), 697. https://doi.org/10.3390/pr14040697







