Screening and Evaluation of Anti-Salt Surfactant/Polymer System for Enhanced Oil Recovery in a Low-Permeability Reservoir in Changqing Oilfield, China
Abstract
1. Introduction
2. Experiments
2.1. Experimental Reagents and Instruments
2.1.1. Experimental Equipment
2.1.2. Experimental Materials and Reagents
2.2. Experimental Process and Methods
2.2.1. Determination of Polymer Viscosity
2.2.2. Determination of Polymer Diafiltration
2.2.3. Determination of Interfacial Tension
2.2.4. Determination of Surfactant Emulsification
2.2.5. Determination of FR and Recovery Ratio
2.2.6. Determination of Flow Rate in Parallel Core Displacement Experiment
3. Results and Discussion
3.1. Polymer Performance Evaluation
3.1.1. Salt-Tolerant Polymer Screening
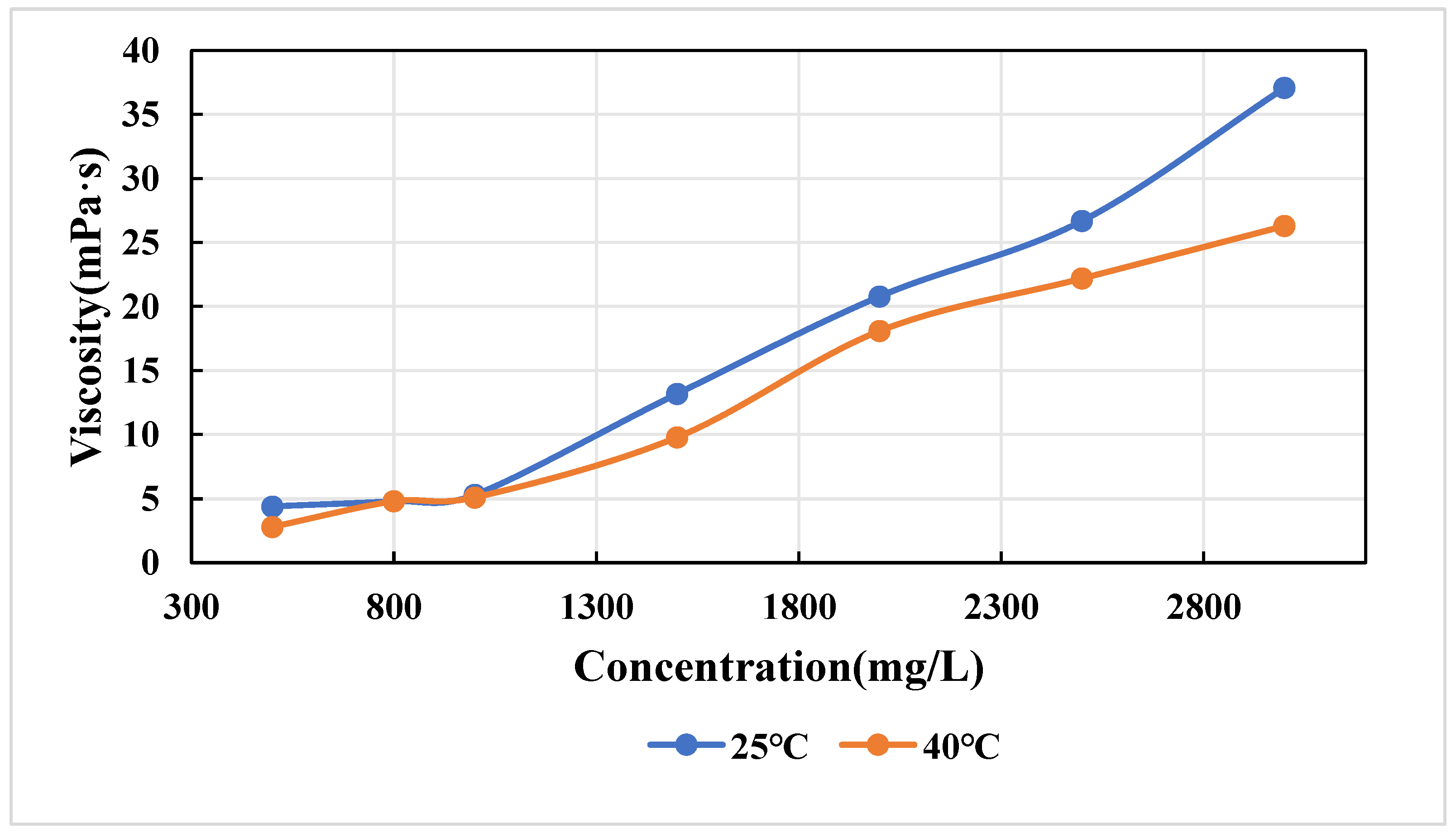
3.1.2. Determination Results of Polymer Viscosity
3.1.3. Polymer Diafiltration Determination Results
3.1.4. Evaluation of Polymer Displacement Effect
3.2. Surfactant Performance Evaluation
3.2.1. Salt-Tolerant Surfactant Screening
3.2.2. Salt-Tolerance Evaluation
3.2.3. Emulsifying Evaluation of Surfactant
3.2.4. Evaluation of Surfactant Displacement Effect
3.3. Performance Evaluation of SP System
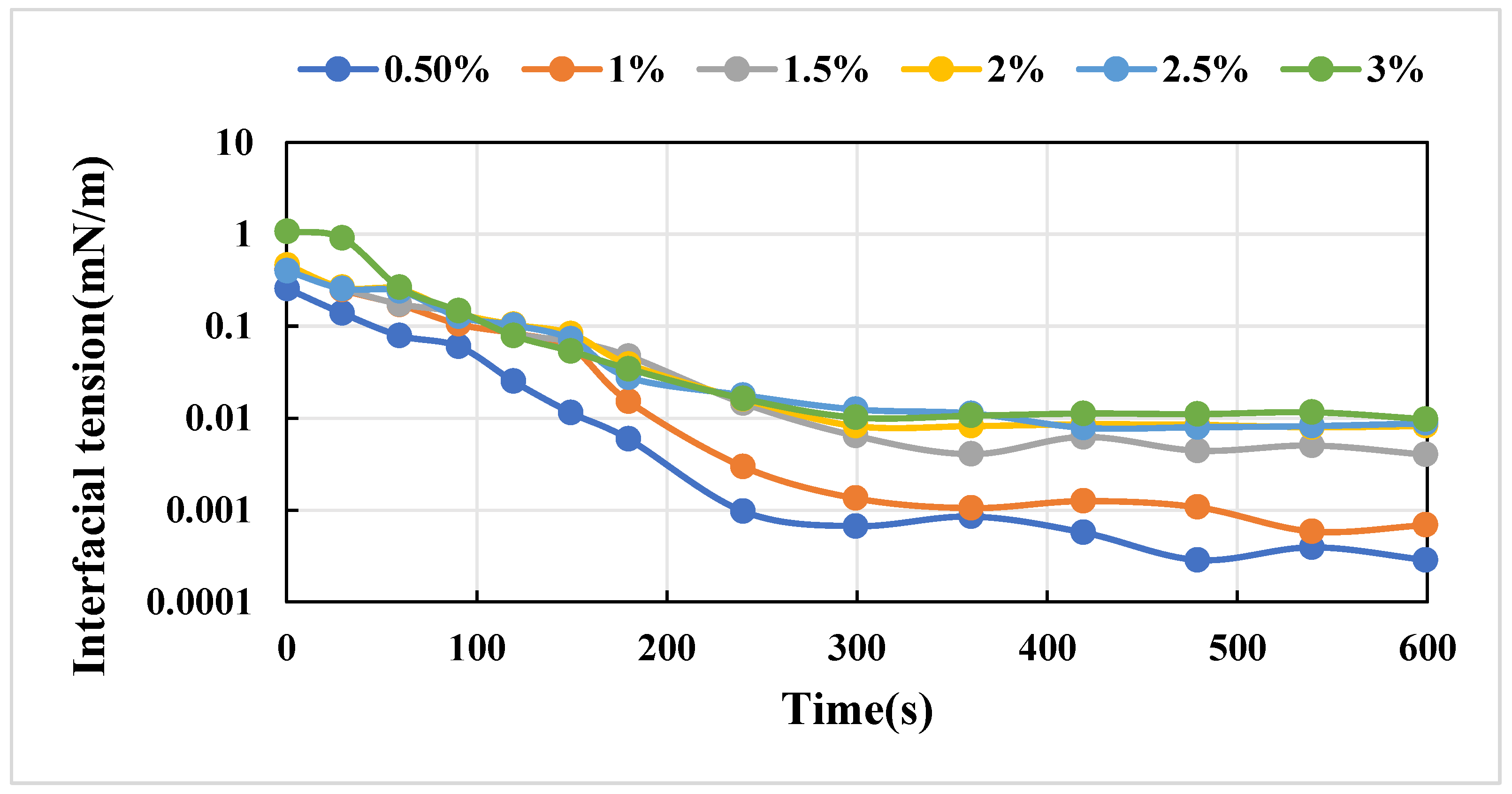
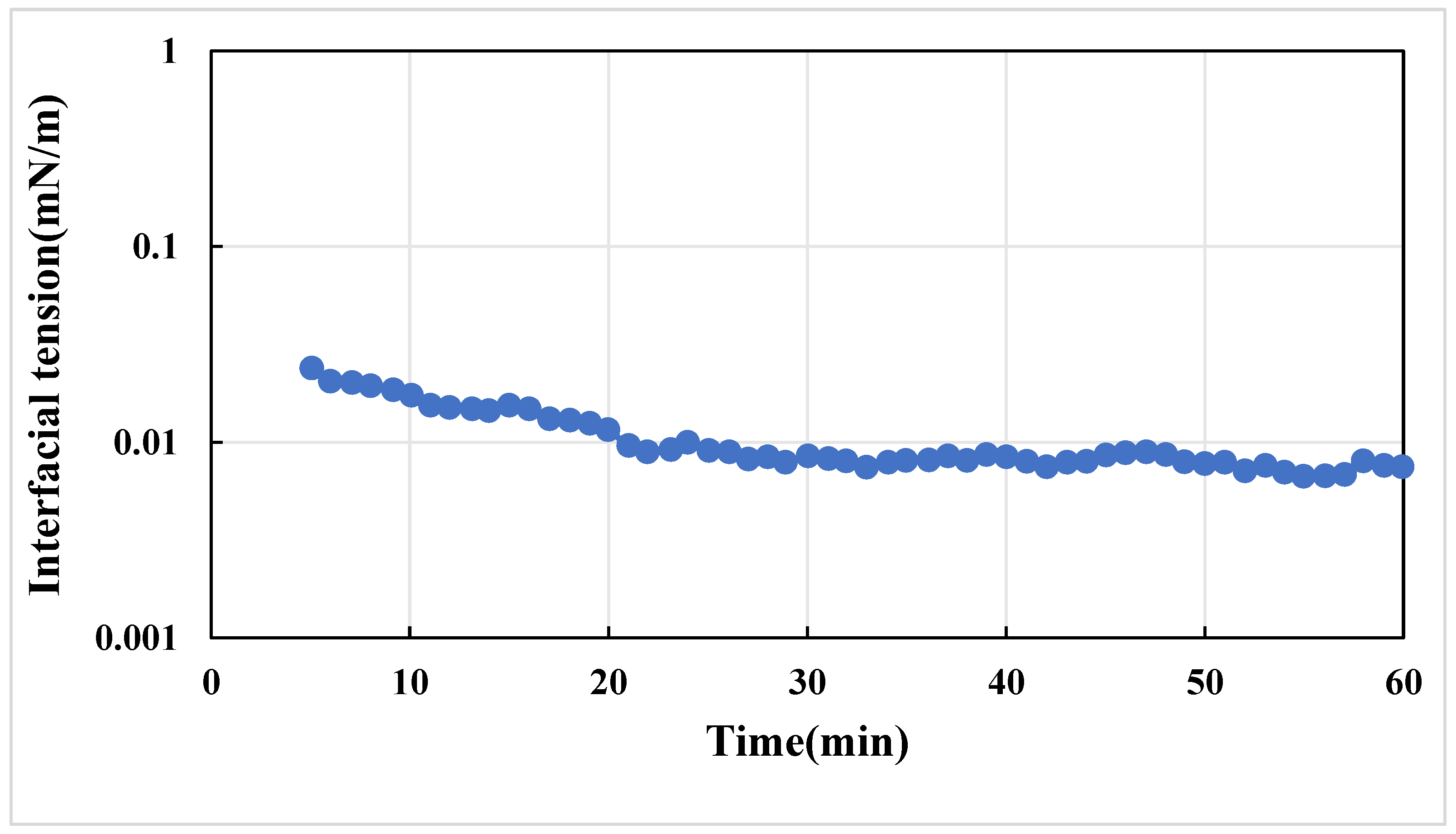
3.3.1. Viscosity and Interfacial Tension of the Composite Systems
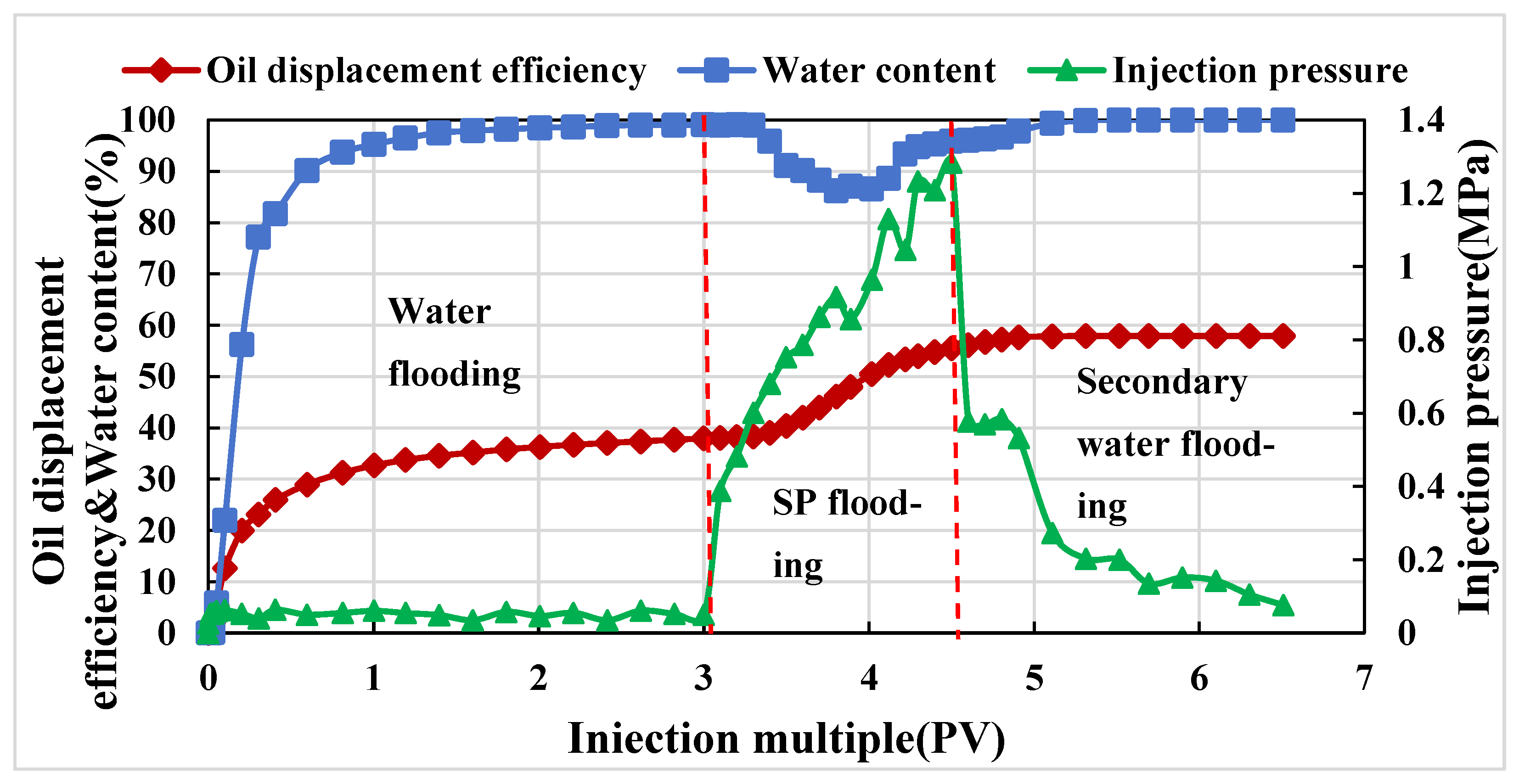
3.3.2. Evaluation of Composite Systems Displacement Effect
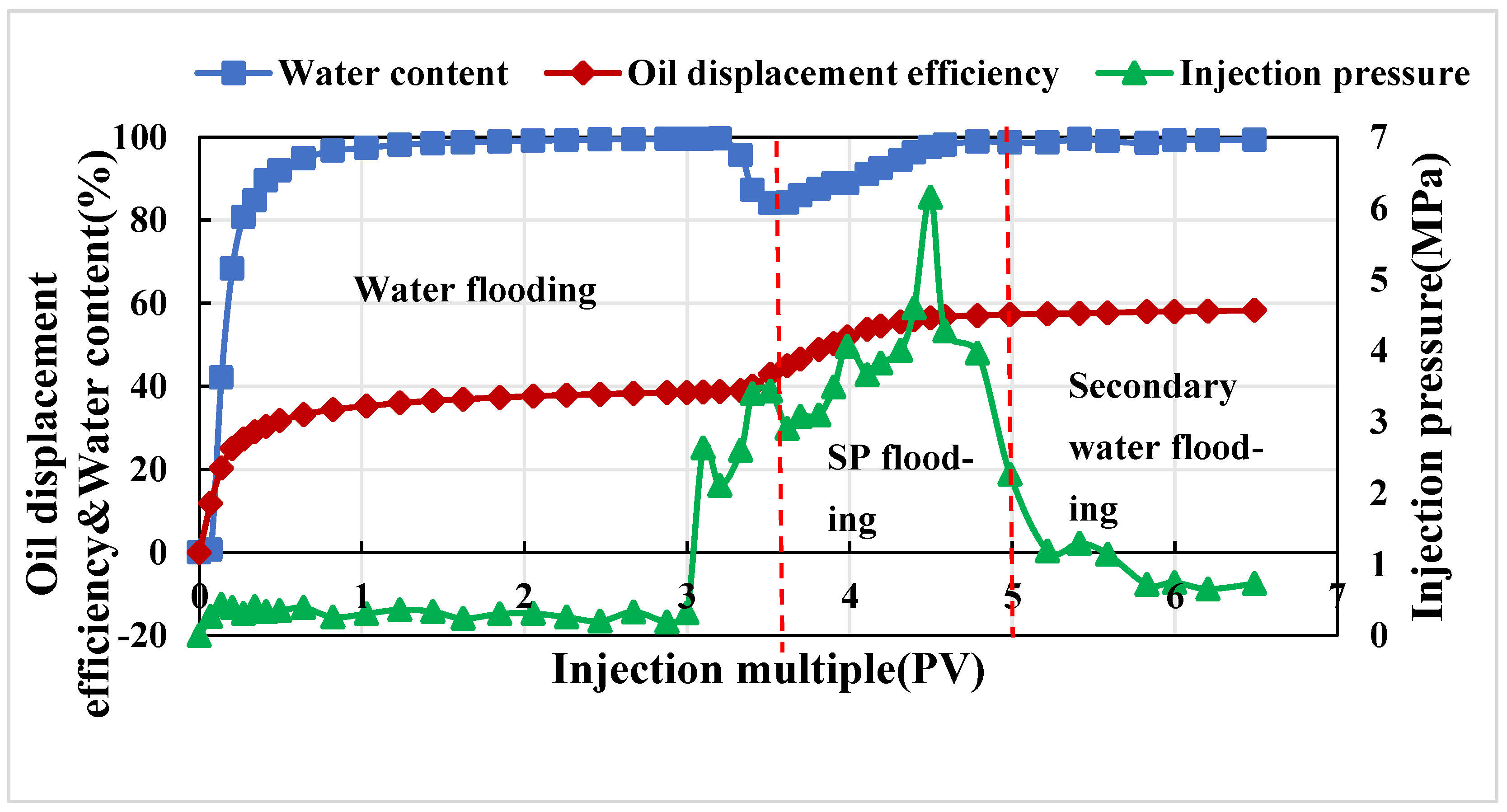
3.3.3. Parallel Core Displacement Experiment Shunt Flow Experimental Evaluation
3.4. Economic Analysis of SP System
4. Conclusions
- (1)
- The viscosity of the polymer was determined according to the formation conditions, and acrylic acid/polyacrylamide/2-acryloylamino-2-methyl-1-propanesulfonic acid was selected. The polymer had good filtration characteristics. The viscosity remained stable at the formation temperature, which could increase the oil displacement efficiency by 14.5%.
- (2)
- The interfacial tension of various surfactants was scanned, and the surfactant system with A:B of 7:3 was selected. It has good adsorption resistance, and the interfacial tension can reach 0.0009 mN/m. It can improve the oil displacement efficiency by more than 10% on the basis of water flooding, and the effect of improving oil recovery is obvious.
- (3)
- The polymer–surfactant binary system was constructed. The surfactant promotes the partial association of the polymer and improves the oil displacement capacity of the system, which can improve the oil displacement efficiency by 19.5% in the natural core displacement experiment. In the parallel core experiment, the recovery rate can be improved by 19.96%. The results of fractional flow show that the swept volume of the low-permeability reservoir increases significantly.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Song, Y.; Li, Z.; Jiang, Z.; Luo, Q.; Liu, D.; Gao, Z. Progress and development trend of unconventional oil and gas geological research. Pet. Explor. Dev. 2017, 44, 675–685. [Google Scholar] [CrossRef]
- Wang, H.J.; Ma, F.; Tong, X.G.; Liu, Z.; Zhang, X.; Wu, Z.; Li, D.; Wang, B.; Xie, Y.; Yang, L. Assessment of global unconventional oil and gas resources. Pet. Explor. Dev. 2016, 43, 925–940. [Google Scholar] [CrossRef]
- Das, A.; Nguyen, N.; Nguyen, Q.P. Low tension gas flooding for secondary oil recovery in low-permeability, high-salinity reservoirs. Fuel 2020, 264, 116601. [Google Scholar] [CrossRef]
- Leitner, W. Reactions in supercritical carbon dioxide (sc CO2). In Modern Solvents in Organic Synthesis; Springer: Berlin/Heidelberg, Germany, 1999; pp. 107–132. [Google Scholar]
- Yang, Z.; Li, M.; Peng, B.; Lin, M.; Dong, Z. Dispersion property of CO2 in oil. 1. Volume expansion of CO2+ alkane at near critical and supercritical condition of CO2. J. Chem. Eng. Data 2012, 57, 882–889. [Google Scholar] [CrossRef]
- Li, X.; Ross, D.A.; Trusler, J.P.M.; Maitland, G.C.; Boek, E.S. Molecular dynamics simulations of CO2 and brine interfacial tension at high temperatures and pressures. J. Phys. Chem. B 2013, 117, 5647–5652. [Google Scholar] [CrossRef]
- Ping, G.; Miao, L. A study on the miscible conditions of CO2 injection in low-permeability sandstone reservoirs. Oil Gas Geol 2007, 28, 687–692. [Google Scholar]
- Wang, R.; Zhang, Y.; Lyu, C.; Lun, Z.; Cui, M.; Lang, D. Displacement characteristics of CO2 flooding in extra-high water-cut reservoirs. Energy Geosci. 2024, 5, 100115. [Google Scholar] [CrossRef]
- Ahmad, W.; Vakili-Nezhaad, G.; Al-Bemani, A.S.; Al-Wahaibi, Y. Experimental determination of minimum miscibility pressure. Procedia Eng. 2016, 148, 1191–1198. [Google Scholar] [CrossRef]
- Wang, Y.J.; Song, X.M.; Tian, C.B.; Shi, C.; Li, J.; Hui, G.; Hou, J.; Gao, C.; Wang, X.; Liu, P. Dynamic fractures are an emerging new development geological attribute in water-flooding development of ultra-low permeability reservoirs. Pet. Explor. Dev. 2015, 42, 247–253. [Google Scholar] [CrossRef]
- Veatch, J.R.; McMurray, M.A.; Nelson, Z.W.; Gottschling, D.E. Mitochondrial dysfunction leads to nuclear genome instability via an iron-sulfur cluster defect. Cell 2009, 137, 1247–1258. [Google Scholar] [CrossRef]
- Wang, Y.; Shang, Q.; Zhou, L.; Jiao, Z. Utilizing macroscopic areal permeability heterogeneity to enhance the effect of CO2 flooding in tight sandstone reservoirs in the Ordos Basin. J. Pet. Sci. Eng. 2021, 196, 107633. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, M.; You, Q.; Fan, H.; Li, W.; Liu, Y.; Fang, J.; Zhao, G.; Jin, Z.; Dai, C. Smart mobility control agent for enhanced oil recovery during CO2 flooding in ultra-low permeability reservoirs. Fuel 2019, 241, 442–450. [Google Scholar] [CrossRef]
- Wei, J.; Zhou, J.; Li, J.; Zhou, X.; Dong, W.; Cheng, Z. Experimental study on oil recovery mechanism of CO2 associated enhancing oil recovery methods in low permeability reservoirs. J. Pet. Sci. Eng. 2021, 197, 108047. [Google Scholar] [CrossRef]
- Xiao, P.; Yang, Z.; Wang, X.; Xiao, H.; Wang, X. Experimental investigation on CO2 injection in the Daqing extra/ultra-low permeability reservoir. J. Pet. Sci. Eng. 2017, 149, 765–771. [Google Scholar] [CrossRef]
- Shen, H.; Yang, Z.; Li, X.; Peng, Y.; Lin, M.; Zhang, J.; Dong, Z. CO2-responsive agent for restraining gas channeling during CO2 flooding in low permeability reservoirs. Fuel 2021, 292, 120306. [Google Scholar] [CrossRef]
- Zhang, J.; Seyyedi, M.; Clennell, M.B. Molecular dynamics simulation of transport and structural properties of CO2–alkanes. Energy Fuels 2021, 35, 6700–6710. [Google Scholar] [CrossRef]
- Rellegadla, S.; Prajapat, G.; Agrawal, A. Polymers for enhanced oil recovery: Fundamentals and selection criteria. Appl. Microbiol. Biotechnol. 2017, 101, 4387–4402. [Google Scholar] [CrossRef] [PubMed]
- Wever, D.A.Z.; Picchioni, F.; Broekhuis, A.A. Polymers for enhanced oil recovery: A paradigm for structure–property relationship in aqueous solution. Prog. Polym. Sci. 2011, 36, 1558–1628. [Google Scholar] [CrossRef]
- Wu, G.; Yu, L.; Jiang, X. Synthesis and properties of an acrylamide-based polymer for enhanced oil recovery: A preliminary study. Adv. Polym. Technol. 2018, 37, 2763–2773. [Google Scholar] [CrossRef]
- Khakpour, H.; Abdollahi, M. Rheological properties of acrylamide/butyl acrylate/2-acrylamido-2-methyl-1-propane sulfonic acid co-and terpolymers synthesized by heterogeneous and micellar methods. Polym. Bull. 2017, 74, 5145–5161. [Google Scholar] [CrossRef]
- Evani, S. Water-Dispersible Hydrophobic Thickening Agent. U.S. Patent 4432881, 20 February 1984. [Google Scholar]
- Mohan, K. Alkaline surfactant flooding for tight carbonate reservoirs. In Proceedings of the SPE Annual Technical Conference and Exhibition, New Orleans, LA, USA, 4–7 October 2009; SPE: Richardson, TX, USA, 2009. SPE-129516-STU. [Google Scholar]
- Hincapie, R.E.; Rock, A.; Wegner, J.; Ganzer, L. Oil mobilization by viscoelastic flow instabilities effects during polymer EOR: A pore-scale visualization approach. In Proceedings of the SPE Latin America and Caribbean Petroleum Engineering Conference, Buenos Aires, Argentina, 17–19 May 2017; SPE: Richardson, TX, USA, 2017. D011S002R002. [Google Scholar]
- Foster, W.R. A low-tension waterflooding process. J. Pet. Technol. 1973, 25, 205–210. [Google Scholar] [CrossRef]
- Ning, J.; Wei, B.; Mao, R.; Wang, Y.; Shang, J.; Sun, L. Pore-level obscrvations of an alkali-induced mild O/W emulsionflooding for economic enhanced oil recovery. Energy Fuels 2018, 32, 10595–10604. [Google Scholar] [CrossRef]
- Dong, M.; Ma, S.; Liu, Q. Enhanced heavy oil recovery through interfacial instability: A study ofchemical flooding for Brintnell heavy oil. Fuel 2009, 88, 1049–1056. [Google Scholar] [CrossRef]
- An, Y.X.; Yao, X.T.; Zhong, J.P.; Pang, S.; Xie, H. Enhancement of oil recovery by surfactant-polymer synergy flooding: A review. Polym. Polym. Compos. 2022, 30, 09673911221145834. [Google Scholar] [CrossRef]
- Holley, S.M.; Cayias, J.L. Design, operation, and evaluation of a surfactant/polymer field pilot test. SPE Reserv. Eng. 1992, 7, 9–14. [Google Scholar] [CrossRef]
- Avwioroko, J.; Taiwo, O.; Mohammed, I.; Dala, J.; Olafuyi, O. A Laboratory Study of ASP Flooding on Mixed Wettability for Heavy Oil Recovery Using Gum Arabic as a Polymer. In Proceedings of the SPE Nigeria Annual International Conference and Exhibition, Lagos, Nigeria, 5–7 August 2014. [Google Scholar]
- Liu, J.; Zhang, K.; Huang, L.; Li, H. Research on Factors Influencing Weak Alkali Surfactant Polymer Flooding System. Chem. Technol. Fuels Oils 2021, 57, 724–731. [Google Scholar] [CrossRef]
- Zaitoun, A.; Kohler, N. The Role of Adsorption in Polymer Propagation Through Reservoir Rocks. In Proceedings of the SPE International Symposium on Oilfield Chemistry, San Antonio, TX, USA, 4–6 February 1987; Society of Petroleum Engineers: Richardson, TX, USA, 1987. [Google Scholar]
- Zhang, J.; Ge, D.; Wang, X.; Wang, W.; Cui, D.; Yuan, G.; Wang, K.; Zhang, W. Influence of Surfactant and Weak-Alkali Concentrations on the Stability of O/W Emulsion in an Alkali-Surfactant–Polymer Compound System. ACS Omega 2021, 6, 5001–5008. [Google Scholar] [CrossRef] [PubMed]
- Sorbie, K.S. Polymer-Improved Oil Recovery; Springer Science & Business Media: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Chan, A.F.; Parmley, J.P. Gravel sizing criteria for sand control and productivity optimization. II―Evaluation of the long-termed stability. In Proceedings of the SPE Formation Damage Control Symposium, Lafayette, Louisiana, 26–27 February 1992; pp. 37–54. [Google Scholar] [CrossRef]
- Wei, J.; Chen, Y.; Zhou, X.; Wang, L.; Fu, P.; Yakushev, V.; Khaidina, M.; Zhang, D.; Shi, X.; Zhou, R. Experimental studies of surfactant-polymer flooding: An application case investigation. Int. J. Hydrogen Energy 2022, 47, 32876–32892. [Google Scholar] [CrossRef]
- Al-Murayri, M.T.; Hassan, A.A.; AlAbdullah, M.B.; Abdulrahim, A.M.; Marlière, C.; Hocine, S.; Tabary, R.; Suzanne, G.P. Surfactant/polymer flooding: Chemical-formulation design and evaluation for Raudhatain lower Burgan Reservoir, Kuwait. SPE Reserv. Eval. Eng. 2019, 22, 923–940. [Google Scholar] [CrossRef]
- Sidorovskaya, E.A.; Adakhovskij, D.S.; Tret, N.Y.; Panicheva, L.P.; Volkova, S.S.; Turnaeva, E.A. Integrated laboratory studies when optimizing surfactant-polymer formulations for oil deposits in Western Siberia. Нефть и газ = Oil Gas Stud. 2020, 107–118. [Google Scholar] [CrossRef]
- Le Van, S.; Chon, B.H. Numerical studies on the effects of various complicated barrier configurations on sweep efficiency in surfactant/polymer flooding. J. Ind. Eng. Chem. 2016, 38, 200–210. [Google Scholar] [CrossRef]
- Suzanne, G.; Soltani, A.; Charonnat, S.; Delamaide, E. Use of numerical simulation enhanced by machine learning techniques to optimize chemical EOR application. In Proceedings of the Abu Dhabi International Petroleum Exhibition and Conference, Abu Dhabi, United Arab Emirates, 31 October–3 November 2022; SPE: Richardson, TX, USA, 2022. D021S059R003. [Google Scholar]
- Yuan, L.; Wu, Y.; Fan, Q.; Li, P.; Liang, J.; Wang, Z.; Li, R.; Shi, L. Spatial distribution, composition, and source analysis of petroleum pollutants in soil from the Changqing Oilfield, Northwest China. Mar. Pollut. Bull. 2022, 185, 114338. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Liu, X.; Huang, D.; Lan, Y.; Wang, S. Natural gas exploration and development in Changqing Oilfield and its prospect in the 13th Five-Year Plan. Nat. Gas Ind. B 2016, 3, 291–304. [Google Scholar] [CrossRef]
- Cui, C.; Zhou, Z.; He, Z. Enhance oil recovery in low permeability reservoirs: Optimization and evaluation of ultra-high molecular weight HPAM/phenolic weak gel system. J. Pet. Sci. Eng. 2020, 195, 107908. [Google Scholar] [CrossRef]
- Xue, D.; Wu, L.; Gao, R.; Cao, Y.; Zhao, C.; Liu, Q.; Li, Y.; Tang, Y.; Slaný, M.; Chen, G. Synthesis and performance study of the fracturing thickener P (AM/AA/AMPS/NVP/DMAAC-16) for rapid fluid preparation. J. Mol. Liq. 2025, 435, 128113. [Google Scholar] [CrossRef]
- Ding, X.; Zhang, G.; Wang, X.; Xin, K.; Wang, F.; Zhou, T.; Wang, X.; Zhang, Z. Preparation of thickened P (AA-AMPS) copolymers by inverse emulsion polymerization and evaluation of fracturing and oil flooding performance. J. Mol. Liq. 2024, 415, 126400. [Google Scholar] [CrossRef]
- Lin, Z.; Lu, X.; Zhang, B.; Liu, W.; Ding, B.; Chang, Y.; Rui, Z.; Zeng, F.; Zhang, S. Feasibility study of CO2-based cyclic solvent injection and polymer flooding alternation process to enhance heavy oil recovery. J. CO2 Util. 2025, 102, 103276. [Google Scholar] [CrossRef]
- Farhadinia, M.A.; Mohanty, K.K. Heavy oil recovery using hot polymer flooding: Simulation studies on mechanisms and injection patterns. Fuel 2026, 405, 136435. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, F.; Tan, B.; Wang, Y.; Liu, P.; Kim, T.; Guo, L.; Han, X.; Liu, R. Enhanced particle removal ability of two representative nonionic surfactants: A reasonable interpretation based on DFT and coarse-grained molecular dynamics methods. J. Mol. Liq. 2024, 413, 125984. [Google Scholar] [CrossRef]
- Zhao, H.; Kang, W.; Yang, H.; Huang, Z.; Zhou, B.; Sarsenbekuly, B. Emulsification and stabilization mechanism of crude oil emulsion by surfactant synergistic amphiphilic polymer system. Colloids Surf. A Physicochem. Eng. Asp. 2021, 609, 125726. [Google Scholar] [CrossRef]
- Varel, F.T.; Dai, C.; Shaikh, A.; Li, J.; Sun, N.; Yang, N.; Zhao, G. Chromatography and oil displacement mechanism of a dispersed particle gel strengthened Alkali/Surfactant/Polymer combination flooding system for enhanced oil recovery. Colloids Surf. A Physicochem. Eng. Asp. 2021, 610, 125642. [Google Scholar] [CrossRef]





| Number | Polymer |
|---|---|
| 1 | Xanthan gum |
| 2 | Polyacrylamide |
| 3 | Partially hydrolyzed polyacrylamide |
| 4 | Anionic Polyacrylamide |
| 5 | 2-Acryloylamino-2-methyl-1-propanesulfonic acid/Acrylonitrile-butadine-styrene/Chromium |
| 6 | Diallyl dimethyl ammonium chloride/Acrylamide/2-Acryloylamino-2-methyl-1-propanesulfonic acid |
| 7 | Star-shape cationic polyacrylamide |
| 8 | Acrylamide/Organotitanium polymer |
| 9 | N,N-diethylprop-2-enamide/Polyacrylamide/2-Acryloylamino-2-methyl-1-propanesulfonic acid(AAND-1) |
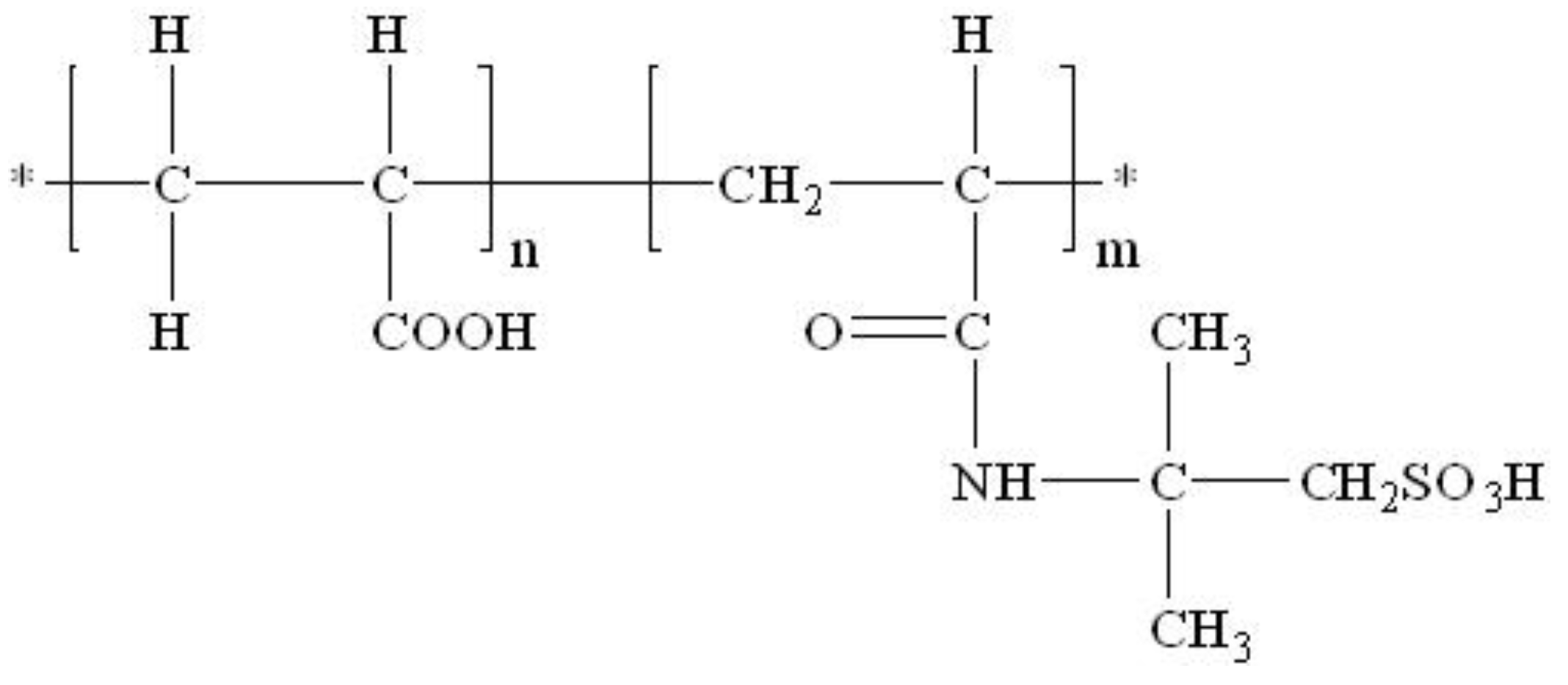
| 10 | Acrylic acid/Polyacrylamide/2-Acryloylamino-2-methyl-1-propanesulfonic acid |
| Number | Surfactant |
|---|---|
| A | Sulfonated Betaine |
| B | Amides, coco |
| C | Sodium Laurylsulfate |
| D | Coconut oil acid diethanolamine |
| E | Sulfonic acids |
| F | Lauroylamide Propylbetaine |
| G | Lauryl betaine |
| H | AmyI gemini quaternary ammonium salt |
| I | Cocamidopropyl Betaine |
| J | Dodecyl dimethyl betaine |
| K | Dodecyl benzenesulphonic acid |
| L | Benzenesulfonic acid |
| Core Number | Permeability (mD) | Pore Volume (cm3) | Saturated Oil Volume (cm3) | Porosity (%) | Oil Saturation (%) |
|---|---|---|---|---|---|
| 1 | 29.2 | 5.59 | 3.65 | 22.8 | 65.3 |
| 2 | 29.4 | 5.52 | 3.54 | 22.5 | 64.2 |
| 3 | 24.1 | 5.52 | 3.54 | 22.5 | 64.2 |
| 4 | 30.7 | 5.54 | 3.58 | 22.6 | 64.6 |
| 5 | 20.11 | 5.56 | 3.6 | 22.7 | 64.8 |
| 6 | 37.38 | 5.74 | 3.8 | 23.4 | 66.2 |
| 7 | 35.9 | 5.66 | 3.72 | 23.1 | 65.7 |
| 8 | 140.4 | 4.61 | 2.55 | 18.8 | 55.3 |
| Polymer | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Molecular weight(W) | 900 | 900 | 1000 | 900 | 900 |
| Viscosity(mPa·s) | 17.4 | 16.4 | 19.2 | 18.8 | 16.1 |
| Polymer | 6 | 7 | 8 | 9 | 10 |
| Molecular weight(W) | 1000 | 900 | 1000 | 900 | 900 |
| Viscosity(mPa·s) | 18.0 | 18.8 | 17.2 | 18.7 | 21.4 |
| Core Number | Permeability (mD) | Viscosity (mPa·s) | Viscosity Retention Rate (%) | |
|---|---|---|---|---|
| Injection Liquid | Produced Liquid | |||
| 1 | 29.2 | 26.7 | 25.7 | 96.3 |
| 2 | 29.4 | 21.3 | 20.9 | 98.1 |
| Water Sample | Viscosity Before Adsorption (mPa·s) | Viscosity After Adsorption (mPa·s) | Core Viscosity Retention Rate After Adsorption (%) |
|---|---|---|---|
| Produced water | 34 | 32.1 | 94.40% |
| Polymer | 10 (Acrylic Acid/Polyacrylamide/2-Acryloylamino-2-methyl-1-propanesulfonic Acid) | |
|---|---|---|
| Pore size of microporous filter membrane (μm) | 3 | 10 |
| Filtration factor (FR) | 1.02 | 1.02 |
| Retention rate of filter shear viscosity (%) | 81.3 | 82.1 |
| Surfactant | A | B | C | D | E | F |
|---|---|---|---|---|---|---|
| σ, mN/m | 0.0081 | 0.0058 | 0.056 | 0.042 | 0.0525 | 0.302 |
| Surfactant | G | H | I | J | K | L |
| σ, mN/m | 0.628 | 0.177 | 0.292 | 0.167 | 0.019 | 0.0226 |
| A:B | 10:0 | 9:1 | 8:2 | 7:3 | 6:4 | 5:5 | 4:6 | 3:7 | 2:8 | 1:9 | 0:10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| σ, mN/m | 0.0081 | 0.0042 | 0.0017 | 0.0009 | 0.0013 | 0.0028 | 0.0033 | 0.0036 | 0.0043 | 0.0051 | 0.0058 |
| Experiment Number | Emulsifiability (%) | Emulsion Stability (%) | Emulsification Comprehensive Index (%) |
|---|---|---|---|
| 1 | 85.60 | 78.63 | 82.04 |
| 2 | 87.30 | 79.21 | 83.16 |
| 3 | 89.70 | 79.66 | 84.53 |
| Average value | 87.53 | 79.17 | 83.24 |
| Interfacial Tension (mN/m) | Viscosity (mPa·s) | ||
|---|---|---|---|
| Surfactant | Surfactant + Polymer | Surfactant | Surfactant + Polymer |
| 0.0042 | 0.007 | 23.9 | 25.3 |
| Polymer Prices ($) | Surfactant Price ($) | |
|---|---|---|
| Old chemical agents | 1930.5/ton | 1859/ton |
| New chemical agents | 1510.08/ton | 2059.2/ton |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Shangguan, Y.; Qu, X.; Yuan, G.; Xiong, W.; Tang, K.; Tian, Q.; Liu, L.; Guan, H.; Wang, Q.; Kang, X.; et al. Screening and Evaluation of Anti-Salt Surfactant/Polymer System for Enhanced Oil Recovery in a Low-Permeability Reservoir in Changqing Oilfield, China. Processes 2026, 14, 408. https://doi.org/10.3390/pr14030408
Shangguan Y, Qu X, Yuan G, Xiong W, Tang K, Tian Q, Liu L, Guan H, Wang Q, Kang X, et al. Screening and Evaluation of Anti-Salt Surfactant/Polymer System for Enhanced Oil Recovery in a Low-Permeability Reservoir in Changqing Oilfield, China. Processes. 2026; 14(3):408. https://doi.org/10.3390/pr14030408
Chicago/Turabian StyleShangguan, Yangnan, Xuefeng Qu, Guowei Yuan, Weiliang Xiong, Kang Tang, Qianqian Tian, Lei Liu, Hua Guan, Qi Wang, Xingmei Kang, and et al. 2026. "Screening and Evaluation of Anti-Salt Surfactant/Polymer System for Enhanced Oil Recovery in a Low-Permeability Reservoir in Changqing Oilfield, China" Processes 14, no. 3: 408. https://doi.org/10.3390/pr14030408
APA StyleShangguan, Y., Qu, X., Yuan, G., Xiong, W., Tang, K., Tian, Q., Liu, L., Guan, H., Wang, Q., Kang, X., Cheng, L., & Hao, H. (2026). Screening and Evaluation of Anti-Salt Surfactant/Polymer System for Enhanced Oil Recovery in a Low-Permeability Reservoir in Changqing Oilfield, China. Processes, 14(3), 408. https://doi.org/10.3390/pr14030408





