Cr(III) Adsorption on Green Mesoporous Silica: Effect of Amine Functionalization and pH
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Procedure
2.1.1. Starting Materials
2.1.2. Green Mesoporous Silica (GMS) Synthesis Using Sodium Silicate
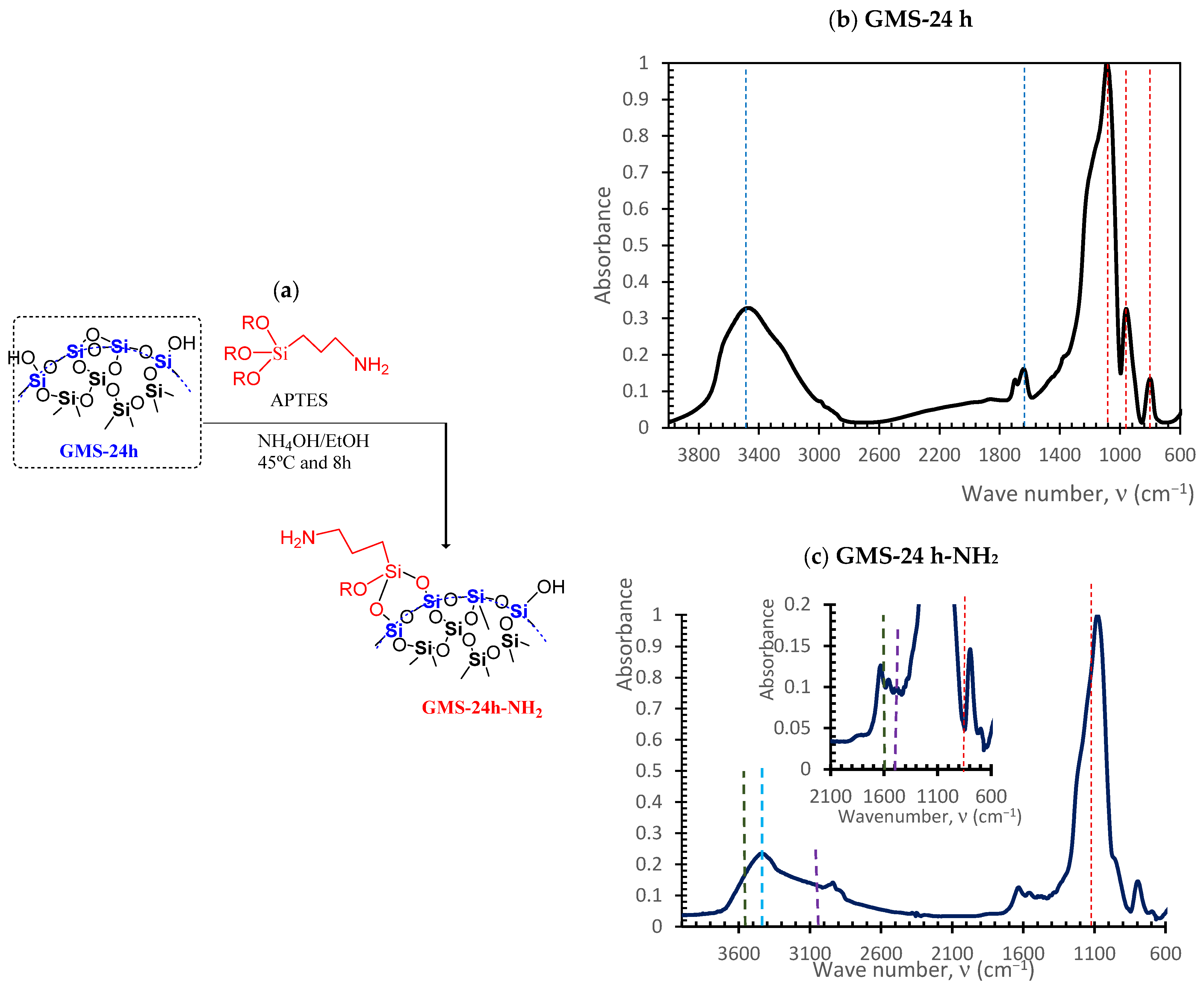
2.1.3. Functionalization of GMS with Amine Groups
2.1.4. Characterization for GMS and GMS-Amino
2.1.5. Cr(III) Adsorption Studies
3. Results
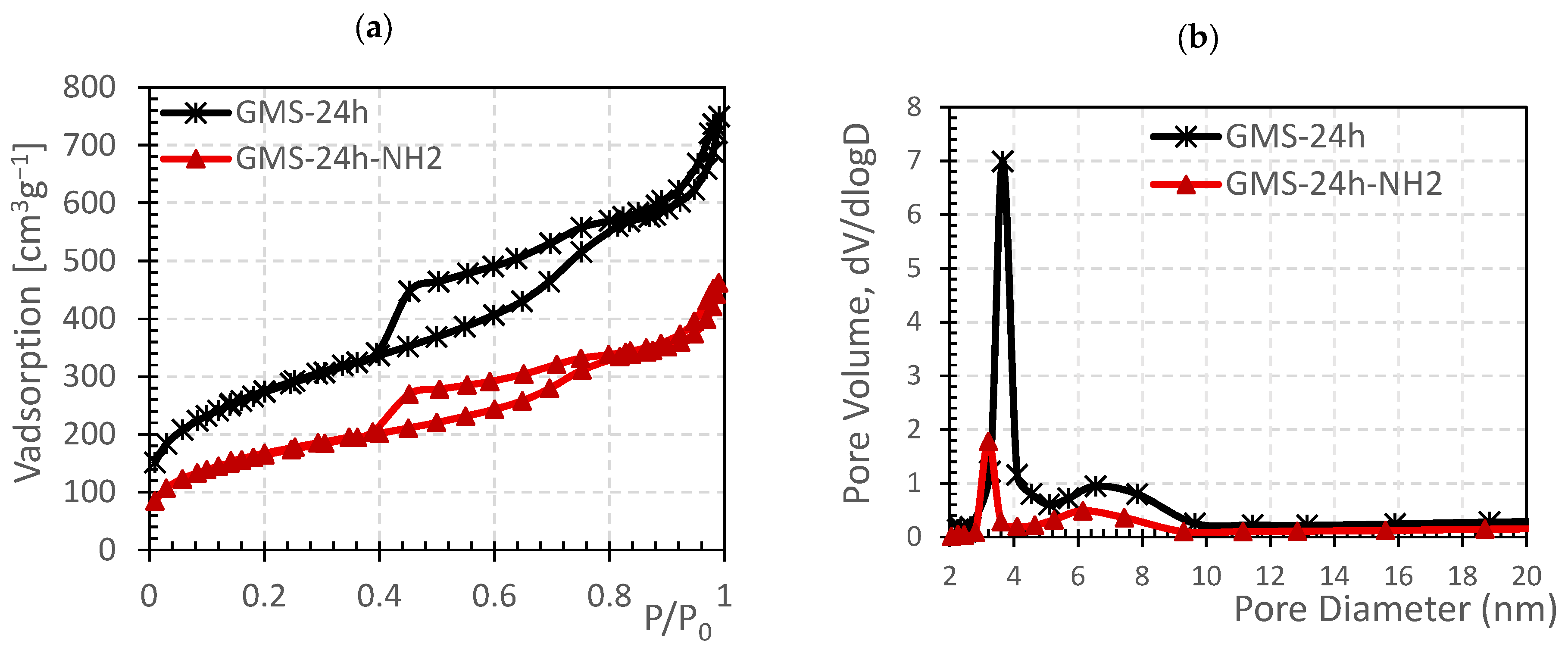
3.1. Effect of Aging on Textural Properties of GMS
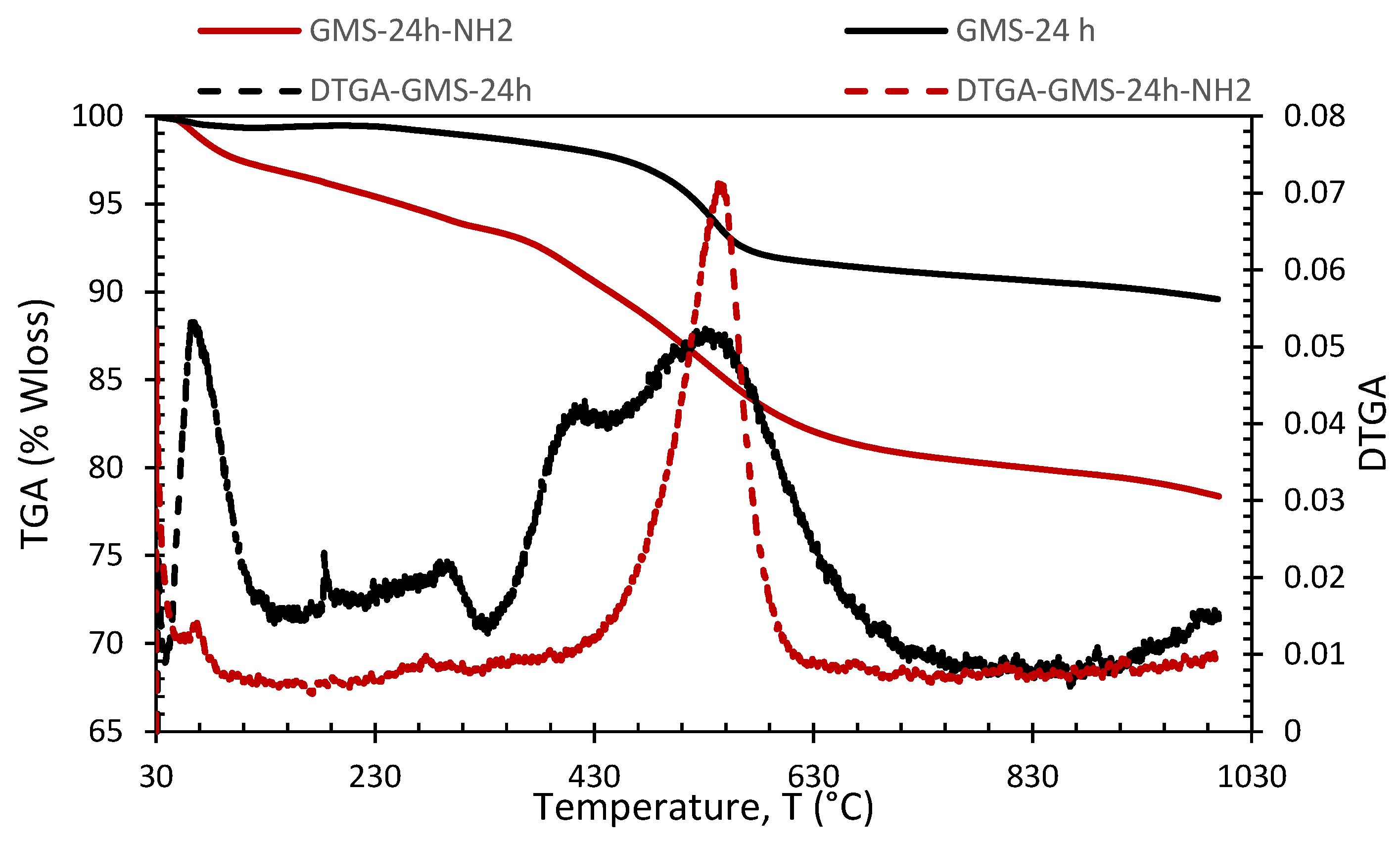
3.2. GMS Modification with Amine Groups
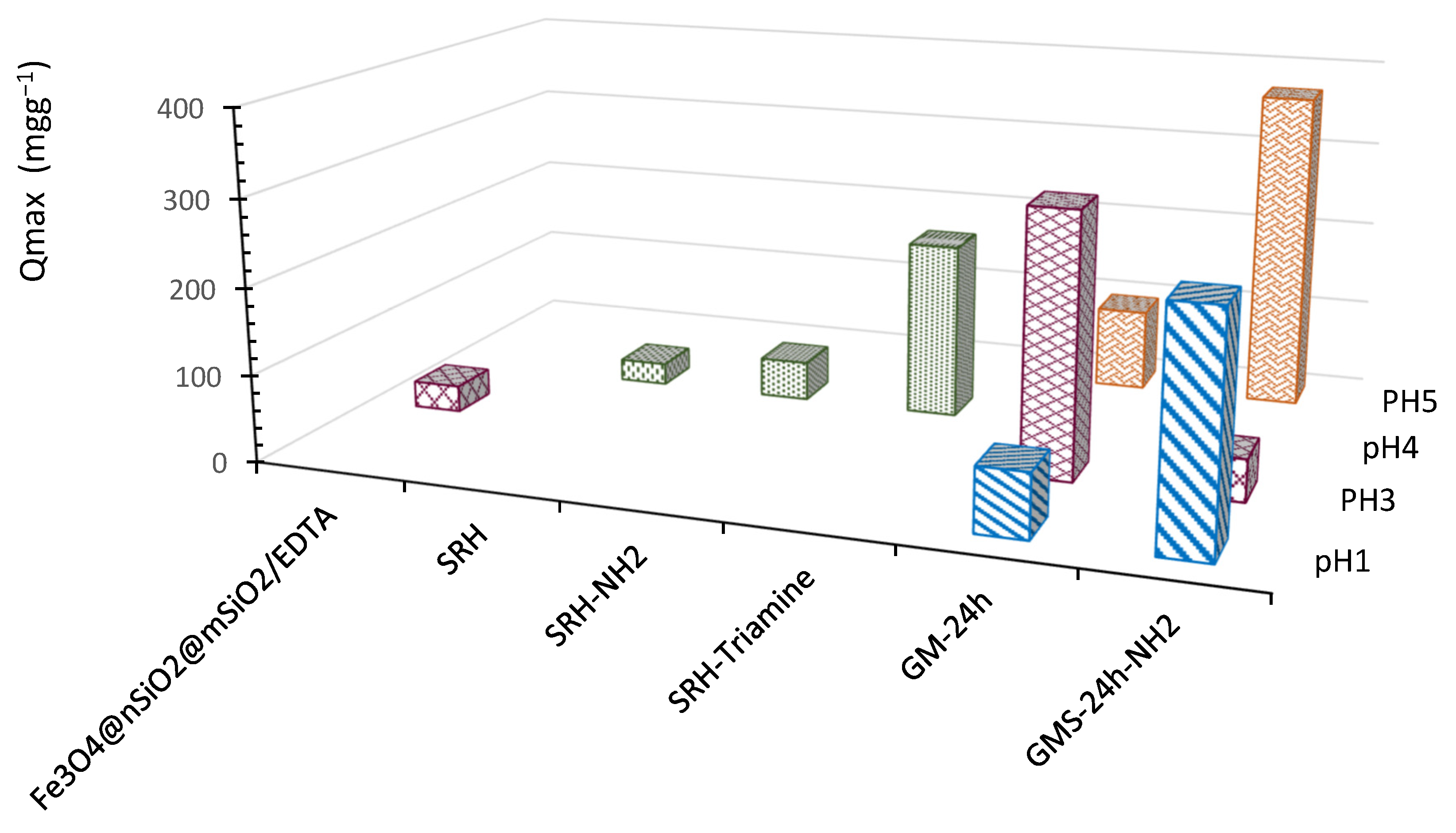
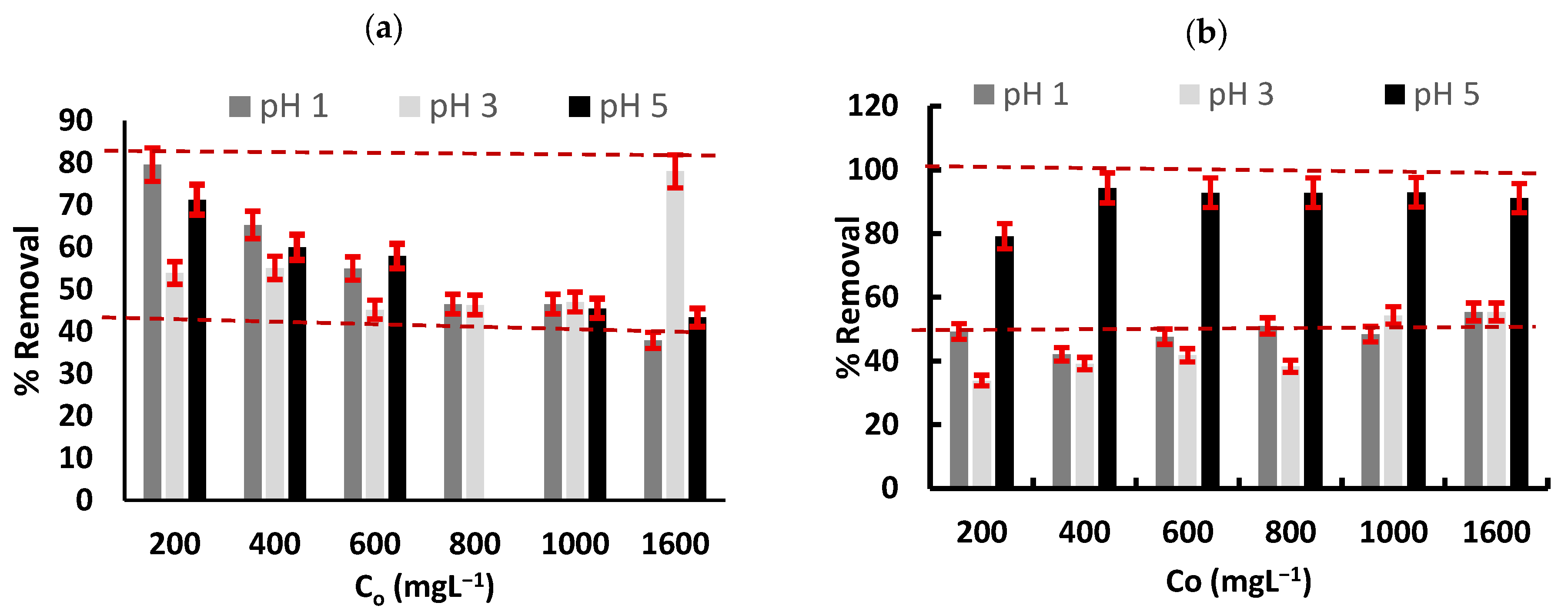
3.3. Cr(3+) Removal Using GMS-24 h and GMS-24 h-NH2
3.4. Kinetic Studies of Cr3+ Adsorption in Stock Solutions
4. Conclusions
- The GMS-24 h material exhibited a high chromium removal capacity at pH 3, with a maximum adsorption of 303 mg·g−1. In contrast, the amine-functionalized GMS-24 h–NH2 showed its highest adsorption capacity at pH 5, reaching 370 mg·g−1.
- Compared to other green mesoporous silica materials, such as SRH and SRH-Triamine, the two modified GMS samples demonstrated an increase of approximately 44% in adsorption capacity.
- Adsorption kinetics evaluated using the pseudo-second-order (PSO) model showed excellent agreement with the experimental data, with the systems tending rapidly toward equilibrium.
- The Weber diffusion model revealed four kinetic slopes for GMS-24 h and five for GMS-24 h–NH2, suggesting a similar adsorption mechanism for both materials. The additional slope observed in GMS-24 h–NH2 is attributed to the chemical coordination step between Cr(III) and surface amine groups (N → Cr).
- No significant changes in the adsorption mechanism were observed with respect to pH. For GMS-24 h, the highest adsorption at pH 3 is associated with ion exchange or electrostatic interaction as the dominant step. At pH 1 and 5, analyte diffusion toward the surface was the predominant mechanism.
- For GMS-24 h–NH2, the diffusion of Cr(III) to the surface was identified as the dominant step across all studied pH conditions.
- GMS-24 h-NH2, due to its excellent adsorption capacity for Cr3+, can be effectively applied for removing chromium from wastewater originating from the tannery industry in León, Guanajuato, Mexico.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shruthi, S.; Hemavathy, R.V. Myco-remediation of chromium heavy metal from industrial wastewater: A review. Toxicol. Rep. 2024, 13, 101740. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Kamaraj, N.; Selvaraj, R.; Nanoth, R. Emerging trends and future Outlook on chromium removal in the lab, pilot scale and industrial waster system: An update review exploring 10 years of research. Environ. Monit. Assess. 2025, 197, 547. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Huang, J.; Xu, Q.; Liu, R.; Yang, F.; Xie, H. Synergistic oxygen vacancies and surface hydroxyl groups in NFM@γ-Al2O3 for enhanced catalytic ozonation of refractory organics and phosphorus in swine wastewater. Sep. Purif. Technol. 2025, 378, 134510. [Google Scholar] [CrossRef]
- Liu, T.; Yin, M.; Du, W.; Zhang, S.; Wen, Z.; Yu, L.; Sun, J.; Zhang, Q.; Sun, M.; Wang, X.; et al. Metal ion-induced structural reconstruction in a porphyrin MOF for ultrasensitive detection of Cr3+ and Al3+. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2026, 344, 126624. [Google Scholar] [CrossRef] [PubMed]
- Niu, K.; Zhang, X.; Zhu, P.; Liu, J.; Liu, H.; Wang, L.; Wang, J.; Zhang, J.; Liu, Q.; Ma, S.; et al. Facile synthesis of polyethyleneimine-reinforced PHTA resin for efficient Cr(VI) removal. Sep. Purif. Technol. 2025, 377, 134343. [Google Scholar] [CrossRef]
- Hosen, M.S.; Sahen, M.S.; Ahmed, H.; Reza, M.S.; Bhowmik, P.; Mim, F.; Islam, M.B.; Naim, M.A.H.K.; Rahman, M.M.; Rahman, M.M. Tannery shaving dust-based charcoal blended adsorbent for efficient heavy metal remediation: An experimental and machine learning approach. Case Stud. Chem. Environ. Eng. 2025, 12, 101256. [Google Scholar] [CrossRef]
- Quiroz, C.E.A.; Ruiz, Y.G.; Rios, J.F.U.; Escobar, E.G.L.; Rodrigues, M.A.S. Electrochemical separation of chromium/collagen from wet blue in a single step: Recycling of tannery waste to promote a circular economy. Results Eng. 2025, 26, 104828. [Google Scholar] [CrossRef]
- Santis, A.; Arbeláez, O.; Mendivelso, K.; Riaño, M.; Prieto, V.; Velásquez, P. Sustainable innovation: Removal of chromium (VI) from wastewater from plastic chromium plating industries using rice husk as photocatalyst. Results Eng. 2025, 26, 104756. [Google Scholar] [CrossRef]
- Kandasamy, S.; Meenachi, S.; Ohm Prakash, K.; Siraj Kumar, K.; Thamaraikannan, V. Removal of chromium from tannery wastewater by rice husk ash nanosilica. Appl. Res. 2023, 2, e202200093. [Google Scholar] [CrossRef]
- Rabago-Velasquez, Z.; Patiño-Saldivar, L.; Ardila A, A.N.; Talavera-Lopez, A.; Salazar-Hernández, M.; Hernández-Soto, R.; Hernández, J.A. Removal of Cr(III) from tannery wastewater using Citrus aurantium (grapefruit peel) as biosorbent. Desalination Water Treat. 2023, 283, 36–49. [Google Scholar] [CrossRef]
- Munagapati, V.S.; Wen, H.-Y.; Gutha, Y.; Wen, J.-C.; Venkateswarlu, S.; Yarramuthi, V.; Gollakota, A.R.K.; Shu, C.-M.; Lohith, E.A.; Jyothi, N.V.V. Advances in biocompatible chitosan composites for chromium, arsenic, and radionuclide remediation: Experimental and computational insights. Coord. Chem. Rev. 2026, 547, 217116. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Mohamed, A.A. The use of chitosan-based composites for environmental remediation: A review. Int. J. Biol. Macromol. 2023, 242, 124787. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Y.; Nie, Y.; Sun, D.; Wu, D.; Ban, L.; Zhang, H.; Yang, S.; Chen, J.; Du, H.; et al. Recent advances and perspectives in functional chitosan-based composites for environmental remediation, energy, and biomedical applications. Prog. Mater. Sci. 2025, 152, 101460. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, S.; Liu, R.; Jiang, H.; Jing, S.; Zhuang, H. Smart-responsive chitosan-based materials for precise degradation control and environmental remediation: A review. Int. J. Biol. Macromol. 2025, 319, 144982. [Google Scholar] [CrossRef] [PubMed]
- Rathore, B.S.; Perumal, P.; Singh, G.P.; Rathore, R.; Rawal, M.K.; Jadoun, S.; Chauhan, N.P.S. Polyaniline, Chitosan and Metal Oxide based Nanocomposites: Synthesis, Characterization, and Their Water Remediation Applications. J. Mol. Struct. 2025, 1337, 142157. [Google Scholar] [CrossRef]
- Purrostam, S.; Rahimi-Ahar, Z.; Babapoor, A.; Nematollahzadeh, A.; Salahshoori, I.; Seyfaee, A. Melamine functionalized mesoporous silica SBA-15 for separation of chromium (VI) from wastewater. Mater. Chem. Phys. 2023, 307, 128240. [Google Scholar] [CrossRef]
- Sun, X.D.; Feng, D.; Xu, J.; Gao, X.; Yan, K.; Tian, Z. Mesoporous silica modified zwitterionic poly(ionic liquids) with enhanced absorption for Cr3+ and dye as a tanning agent. Microporous Mesoporous Mater. 2025, 396, 113742. [Google Scholar] [CrossRef]
- Zhang, S.; Xu, X.; Yue, Q.; Wang, W.; Gao, B. Improving the removal of chromium by polymer epichlorohydrin-dimethylamine functionalized mesoporous silica. Desalination Water Treat. 2019, 169, 294–304. [Google Scholar] [CrossRef]
- Kobylinska, N. Single-stage separation and quantitation of anionic and cationic forms of transition metals from environmental water using organo-functionalized ordered mesoporous silica. Chem. Eng. J. Adv. 2025, 22, 100741. [Google Scholar] [CrossRef]
- Zhu, H.; Ding, K.; You, Y.; Sheng, G.; Dong, H.; Liu, H. In-situ fabrication of exceptionally efficient traps namely nanoscale zero valent iron wrapped on mesoporous silica microspheres with different size in effectively purifying and scavenging Re(VII) and Cr(VI) from water matrices. J. Environ. Chem. Eng. 2025, 13, 116367. [Google Scholar] [CrossRef]
- Priya, A.K.; Yogeshwaran, V.; Rajendran, S.; Hoang, T.K.A.; Soto-Moscoso, M.; Ghfar, A.A.; Bathula, C. Investigation of mechanism of heavy metals (Cr6+, Pb2+ & Zn2+) adsorption from aqueous medium using rice husk ash: Kinetic and thermodynamic approach. Chemosphere 2022, 286, 131796. [Google Scholar] [CrossRef]
- Gutiérrez-Valtierra, M.; Salazar-Hernández, C.; Mendoza-Miranda, J.M.; Elorza-Rodríguez, E.; Puy-Alquiza, M.J.; Caudillo-González, M.; Salazar-Hernández, M.M. Cr(III) removal from tannery effluents using silica obtained from rice husk and modified silica. Desalination Water Treat. 2019, 158, 152–163. [Google Scholar] [CrossRef]
- Ijaz, S.; Kausar, A.; Iqbal, M.; El Messaoudi, N.; Miyah, Y.; Knani, S.; Graba, B. Advances in extraction of silica from rice husk and its modification for friendly environmental wastewater treatment via adsorption technology. J. Water Process Eng. 2025, 71, 107187. [Google Scholar] [CrossRef]
- Khemis, I.B.; Aouaini, F.; Knani, S.; Al-Mugren, K.S.; Lamine, A.B. Microscopic and macroscopic analysis of hexavalent chromium adsorption on polypyrrole-polyaniline@rice husk ash adsorbent using statistical physics modeling. Heliyon 2024, 10, e37061. [Google Scholar] [CrossRef] [PubMed]
- Amirhandeh, S.Z.H.; Salem, A.; Salem, S. Sono-chemical extraction of silica from rice husk for uptake of chromium species from tannery wastewater: Effect of aging time on porous structure. Mater. Lett. 2022, 327, 132933. [Google Scholar] [CrossRef]
- Khan, M.; Din, I.; Aziz, F.; Qureshi, I.U.; Zahid, M.; Mustafa, G.; Sher, A.; Hakim, S. Chromium adsorption from water using mesoporous magnetic iron oxide-aluminum silicate adsorbent: An investigation of adsorption isotherms and kinetics. Curr. Res. Green Sustain. Chem. 2023, 7, 100368. [Google Scholar] [CrossRef]
- Tsegaye, G.; Kiflie, Z.; Mekonnen, T.H.; Jida, M. Amine-functionalized magnetic bio-nanocomposite for fluoride and chromium removal in water. Results Chem. 2025, 18, 102685. [Google Scholar] [CrossRef]
- Wei, Y.; He, L.; Luo, Z.; Zhou, X.; Zhang, Z.; Luo, Y.; Liu, Q. Removal of hexavalent chromium from aqueous solution using low-cost magnetic microspheres derived from alkali-activated iron-rich copper slag. J. Environ. Chem. Eng. 2023, 11, 110436. [Google Scholar] [CrossRef]
- Zhang, X.; Liang, P.; Pan, Y.; Wang, G. Fabrication of hydrophilic defective MOF-801 thin-film nanocomposite membranes via interfacial polymerization for efficient chromium removal from water. J. Environ. Manag. 2025, 384, 125561. [Google Scholar] [CrossRef]
- Mubarak, A.S.; Salih, S.S.; Kadhom, M.; Ghosh, T.K. Removal of heavy metals from contaminated water using Metal-Organic Frameworks (MOFs): A review on techniques and applications. Mater. Sci. Eng. B 2025, 315, 118105. [Google Scholar] [CrossRef]
- Khosroshahi, N.; Bakhtian, M.; Safarifard, V. Mechanochemical synthesis of ferrite/MOF nanocomposite: Efficient photocatalyst for the removal of meropenem and hexavalent chromium from water. J. Photochem. Photobiol. A Chem. 2022, 431, 114033. [Google Scholar] [CrossRef]
- Tokalıoğlu, Ş.; Moghaddam, S.T.H.; Demir, S. A zirconium metal–organic framework functionalized with a S/N containing carboxylic acid (MOF-808(Zr)-Tz) as an efficient sorbent for the ultrafast and selective dispersive solid phase micro extraction of chromium, silver, and rhodium in water samples. Talanta 2024, 274, 126094. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Amitani, N.; Yokoyama, T.; Ueda, A.; Kusakabe, M.; Unami, S.; Odashima, Y. Synthesis of mesoporous silica from geothermal water. Sci. Rep. 2021, 11, 23811. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Lee, J.H.; Chun, J. Facile approach for the synthesis of spherical mesoporous silica nanoparticles from sodium silicate. Mater. Lett. 2021, 283, 128765. [Google Scholar] [CrossRef]
- Hossain, K.-Z.; Sayari, A. Synthesis of onion-like mesoporous silica from sodium silicate in the presence of α,ω-diamine surfactant. Microporous Mesoporous Mater. 2008, 114, 387–394. [Google Scholar] [CrossRef]
- Ewlad-Ahmed, A.M.; Morris, M.; Holmes, J.; Patwardhan, S.V.; Gibson, L.T. Green Nanosilicas for Monoaromatic Hydrocarbons Removal from Air. Silicon 2022, 14, 1447–1454. [Google Scholar] [CrossRef]
- Salazar-Hernández, M.; Salazar-Hernández, C.; Rodríguez, E.E.; Miranda, J.M.M.; de Jesús Puy-Alquiza, M.; Miranda-Aviles, R.; Rodríguez, C.R. Using of Green Silica Amine-Fe3O4 modified from recovery Ag(I) on aqueous system. Silicon 2024, 16, 1509–1524. [Google Scholar] [CrossRef]
- Rimmelin-Maury, P.; Moutin, T.; Quéguiner, B. A new method for nanomolar determination of silicic acid in seawater. Anal. Chim. Acta 2007, 587, 281–286. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Ceram. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Barton, T.J.; Bull, L.M.; Klemperer, W.G.; Loy, D.A.; McEnaney, B.; Misono, P.A.; Pez, G.; Scherer, G.W.; Vartuli, J.C.; Yaghi, O.M. Tailored Porous Materials. Chem. Mater. 1999, 11, 2633–2656. [Google Scholar] [CrossRef]
- Tella, J.O.; Ajanaku, K.O.; Adekoya, J.A.; Banerjee, R.; Patra, C.R.; Pavuluri, S.; Sreedhar, B. Physicochemical and textural properties of amino-functionalised mesoporous silica nanomaterials from different silica sources. Results Chem. 2024, 7, 101505. [Google Scholar] [CrossRef]
- Launer, P.J. Infrared analysis of organosilicon compounds: Spectra structure correlations. In Silicon Compounds Silanes & Silicones; Arkles, B., Ed.; Gelest Inc.: Morrisville, PA, USA, 2013. [Google Scholar]
- Salazar-Hernández, C.; Salazar-Hernández, M.; Mendoza-Miranda, J.M.; León-Reyes, M.R.; Rocha-Ramírez, A.H. Mesoporous silica-based drug delivery systems obtained from sodium silicate as an economical synthesis. In Trends and Challenges in Multidisciplinary Research for Global Sustainable Development; Hernández Ponce, A.M., Marcos Escobar, K., Canales Hernández, L.D., Zea Ortiz, M., Sánchez Alonso, R.E., Eds.; ICASAT 2023; Lecture Notes in Networks and Systems; Springer: Cham, Switzerland, 2024; Volume 965. [Google Scholar] [CrossRef]
- Hu, Q.; Lan, R.; He, L.; Liu, H.; Pei, X. A critical review of adsorption isotherm models for aqueous contaminants: Curve characteristics, site energy distribution and common controversies. J. Environ. Manag. 2023, 329, 117104. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the use and interpretation of adsorption isotherm models: A review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef]
- Alafnan, S.; Awotunde, A.; Glatz, G.; Adjei, S.; Alrumaih, I.; Gowida, A. Langmuir adsorption isotherm in unconventional resources: Applicability and limitations. J. Pet. Sci. Eng. 2021, 207, 109172. [Google Scholar] [CrossRef]
- Foo, K.F.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Wang, J.; Chen, Y.; Sun, T.; Saleem, A.; Wang, C. Enhanced removal of Cr(III)-EDTA chelates from high-salinity water by ternary complex formation on DETA functionalized magnetic carbon-based adsorbents. Ecotoxicol. Environ. Saf. 2021, 209, 111858. [Google Scholar] [CrossRef]
- Jean-Pierre, S. On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef]
- Wu, F.-C.; Tseng, R.-L.; Huang, S.-C.; Juang, R.-S. Characteristics of pseudo-second-order kinetic model for liquid-phase adsorption: A mini-review. Chem. Eng. J. 2009, 151, 1–9. [Google Scholar] [CrossRef]
- Largitte, L.; Pasquier, R. A review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem. Eng. Res. Des. 2016, 109, 495–504. [Google Scholar] [CrossRef]









| [Si(OH)4]; (mg L−1) | ABET [m2 g−1] | Average Pore Volume [cm3 g−1] | Pore Diameter (BJH) [nm] | |
|---|---|---|---|---|
| GMS-0 h | 626.1 ± 15 | 444.5 ± 4.9 | 0.341 ± 0.02 | 3.9; broad between 5 and 20 |
| GMS-6 h | 265.9 ± 12 | 491.4 ± 4.6 | 0.416 ± 0.015 | 3.9 |
| GMS-24 h | 97.7 ± 11 | 496.5 ± 2.6 | 0.552 ± 0.015 | 3.9 |
| GMS-48 h | 52.9 ± 8 | 466 ± 4.2 | 0.497 ± 0.023 | 3.9 and 7.8 |
| SiO2-HCl * | – | 13.27 | 0.052 | – |
| ABET [m2 g−1] | Average Pore Volume [cm3 g−1] | Pore Diameter (BJH) [nm] | |
|---|---|---|---|
| GMS-24 h | 496.5 ± 2.6 | 0.552 ± 0.015 | 3.9 |
| GMS-24 h-NH2 | 171.1 ± 4.02 | 0.368 ± 0.012 | 3.2 |
| Parameter | pH 1 | pH 3 | pH 5 | ||||
|---|---|---|---|---|---|---|---|
| GMS-24 h | GMS-24 h-NH2 | GMS-24 h | GMS-24 h-NH2 | GMS-24 h | GMS-24 h-NH2 | ||
| Langmuir | Q0 [mg g−1] | 72.99 | 263.16 | 303.03 | 48.31 | 95.24 | 370.37 |
| KL [L mg−1] | 0.0042 | 0.00034 | 0.00039 | 0.00097 | 0.0024 | 0.0045 | |
| R2 | 0.9632 | 0.9568 | 0.8783 | 0.8675 | 0.9099 | 0.9052 | |
| Δq(%) | 0.1389 | 0.1832 | 0.313 | 0.1967 | 0.00012 | 0.09134 | |
| G [KJ mol−1] | 3.59 | 9.79 | 9.46 | 7.23 | 5.04 | 3.42 | |
| Freundlich | KF[(mg g−1)/(mg L−1)]1/n | 4.21 | 0.042 | 3.843 | 0.00197 | 1.823 | 3.653 |
| 1/n | 0.385 | 1.14 | 0.832 | 1.63 | 0.527 | 0.744 | |
| R2 | 0.9929 | 0.956 | 0.9666 | 0.9907 | 0.9842 | 0.9945 | |
| Δq(%) | 0.0718 | 0.3552 | 0.2199 | 0.1068 | 0.1269 | 0.0561 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Salazar-Hernández, C.; Salazar-Hernández, M.; Elorza-Rodríguez, E.; Mendoza-Miranda, J.M.; Miranda-Avilés, R.; León-Reyes, M.d.R.; Sánchez, C.D.M.; Arroyo, M.A.C.; Rodríguez-Dahmlow, J.E. Cr(III) Adsorption on Green Mesoporous Silica: Effect of Amine Functionalization and pH. Processes 2026, 14, 358. https://doi.org/10.3390/pr14020358
Salazar-Hernández C, Salazar-Hernández M, Elorza-Rodríguez E, Mendoza-Miranda JM, Miranda-Avilés R, León-Reyes MdR, Sánchez CDM, Arroyo MAC, Rodríguez-Dahmlow JE. Cr(III) Adsorption on Green Mesoporous Silica: Effect of Amine Functionalization and pH. Processes. 2026; 14(2):358. https://doi.org/10.3390/pr14020358
Chicago/Turabian StyleSalazar-Hernández, Carmen, Mercedes Salazar-Hernández, Enrique Elorza-Rodríguez, Juan Manuel Mendoza-Miranda, Raúl Miranda-Avilés, María de Rosario León-Reyes, Cristina Daniela Moncada Sánchez, Mario Alberto Corona Arroyo, and Jesús E. Rodríguez-Dahmlow. 2026. "Cr(III) Adsorption on Green Mesoporous Silica: Effect of Amine Functionalization and pH" Processes 14, no. 2: 358. https://doi.org/10.3390/pr14020358
APA StyleSalazar-Hernández, C., Salazar-Hernández, M., Elorza-Rodríguez, E., Mendoza-Miranda, J. M., Miranda-Avilés, R., León-Reyes, M. d. R., Sánchez, C. D. M., Arroyo, M. A. C., & Rodríguez-Dahmlow, J. E. (2026). Cr(III) Adsorption on Green Mesoporous Silica: Effect of Amine Functionalization and pH. Processes, 14(2), 358. https://doi.org/10.3390/pr14020358