Silicon Carbide-Silicon Nitride Materials: Part 2—Corrosion Resistance and Oxidation in Al Reduction Cells and at Lab Testing
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Corrosion and Oxidation of Si3N4-SiC Materials in Industrial Al Reduction Cells
3.2. Corrosion and Oxidation of Si3N4-SiC Materials at Laboratory Corrosion Testing
4. Discussion
4.1. On Corrosion and Oxidation of Si3N4-SiC Materials in Al Reduction Cells and Lab Testing
4.2. On Priority of Corrosion Resistance of Silicon Carbide over Silicon Nitride and of α-Silicon Nitride over β-Silicon Nitride
5. Conclusions
- More probably the reactions of silicon carbide and silicon nitride with liquid cryolite take place after the stage of oxidation. In the gas phase the greater part of the oxidation reactions of silicon carbide and silicon nitride proceed with positive volume transformation (decreasing the porosity of material).
- In the current research the results of chemical and phase analyses of Si3N4-SiC materials after lab corrosion testing with molten cryolite differed according to the different zones (above the level of molten cryolite, in the zone of the level of the melt and below the level of the melt of cryolite).
- There is no direct indication of the influence of silicon nitride modifications on the bulk volume loss in Si3N4-SiC materials, yet, according to SEM and XRD, α-Si3N4 in the zone of reaction (maximal exposure) dissolves a little bit quicker than β-Si3N4 and silicon nitride disappears in the course of the chemical interactions a little bit more quickly than silicon carbide.
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Karadimas, G.; Salonitis, K. Ceramic Matrix Composites for Aero Engine Applications—A Review. Appl. Sci. 2023, 13, 3017. [Google Scholar] [CrossRef]
- Subha, S.; Benny, N.; Singh, D. Thermo-mechanical behavior of SiC-based composites for gas turbine engines. Mater. Today Proc. 2022, 68, 2301–2309. [Google Scholar] [CrossRef]
- Hou, X.; Wang, E.; Li, B.; Chen, J.; Chou, K.-C. Corrosion behavior of porous silicon nitride ceramics in different atmospheres. Ceram. Int. 2017, 43, 4344–4352. [Google Scholar] [CrossRef]
- Long, M.; Li, Y.; Qin, H.; Xue, W.; Jiang, P.; Sun, J.; Kumar, R.V. Mechanism of active and passive oxidation of reaction-bonded Si3N4-SiC refractories. Ceram. Int. 2017, 43, 10720–10725. [Google Scholar] [CrossRef]
- Tatami, J.; Uda, M.; Takahashi, T.; Yahagi, T.; Iijima, M.; Matsui, K.; Ohji, T.; Nakano, H. Microscopic mechanical properties of silicon nitride ceramics corroded in sulfuric acid solution. J. Eur. Ceram. Soc. 2024, 44, 5415–5421. [Google Scholar]
- Sørlie, M.; Øye, H. Cathodes in Aluminium Electrolysis, 3rd ed.; Aluminium-Verlag: Dusseldorf, Germany, 2010; 662p. [Google Scholar]
- Yurkov, A. Refractories for Aluminium: Electrolysis and the Cast House, 2nd ed.; Springer International Publishing AG: Berlin/Heidelberg, Germany, 2017; 276p, ISBN 978-3-319-53588-3. [Google Scholar]
- Skybakmoen, E.; Gudbransen, H.; Stoen, L.T. Chemical Resistance of Sidelining materials based on SiC and Carbon in Cryolitic melts—A laboratory study. Light Met. 1999, 128, 215–222. [Google Scholar]
- Skybakmoen, E.; Stoen, L.; Kvello, J.H.; Darrel, O. Quality evaluation in Nitride bonded Silicon Carbide Sidelining Materials. Light Met. 2005, 134, 773–778. [Google Scholar]
- Laucournet, R.; Laurent, V.; Lombard, D. Chemical resistance of sidelining refractory based on Si3N4 bonded SiC. Light Met. 2008, 2008, 961–966. [Google Scholar]
- Zhao, J.; Zhang, Z.; Wang, W.; Liu, G. Test method for Resistance of SiC material to Cryolite. Light Met. 2006, 135, 663–666. [Google Scholar]
- Gao, B.L.; Wang, Z.W.; Qiu, Z.X. Corrosion Tests and Electrical Resistivity Measurement of SiC-Si3N4 Refractory Materials. Light Met. 2004, 133, 419–424. [Google Scholar]
- Cao, C.; Gao, B.; Wang, Z.; Hu, X.; Qui, Z. A new test method for evaluating Si3N4-SiC bricks corrosion resistance to aluminium electrolyte and oxygen. Light Met. 2006, 135, 659–661. [Google Scholar]
- Proshkin, A.V.; Pingin, V.V.; Polyakov, P.V.; Kalinovskaya, T.G.; Pogodaev, A.M.; Isaeva, L.A. Study of the state and dynamics of Wear of the side lining in the cathode of Aluminium Cells. J. Sib. Fed. Univ. Eng. Technol. 2013, 3, 276–284. [Google Scholar]
- Yurkov, A.L. Silicon Carbide–Silicon Nitride Refractory Materials: Part 1 Materials Science and Processing. Processes 2023, 11, 2134. [Google Scholar] [CrossRef]
- Metson, J.; McIntoch, G.; Etzion, R. Materials science constraints on the development of Aluminium Reduction Cells, Advanced Materials Development and Performance (AMDP2011). Int. J. Mod. Phys. Conf. Ser. 2012, 6, 25–30. [Google Scholar] [CrossRef]
- Gábrišová, Z.; Švec, P.; Brusilová, A. Microstructure and Selected Properties of Si3N4 + SiC Composite. Manuf. Technol. 2020, 20, 293–299. [Google Scholar] [CrossRef]
- Kong, J.H.; Ma, H.J.; Jung, W.K.; Hong, J.; Jun, K.; Kim, D.K. Self-reinforced and high-thermal conductivity silicon nitride by tailoring α-β phase ratio with pressureless multi-step sintering. Ceram. Int. 2021, 47, 13057–13064. [Google Scholar]
- Schoenhahl, J.; Jorge, E.; Marguin, O.; Kubiak, S.; Temme, P. Optimization of Si3N4 bonded SiC refractories for aluminium reduction cells. Light Met. 2001, 130, 251–255. [Google Scholar]
- Jorge, E.; Marguin, O. Si3N4 Bonded SiC Refractories for Higher Aluminium Cell Performance. Aluminium Times, September 2004; pp. 47–50. [Google Scholar]
- Jorge, E.; Marguin, O.; Temme, P. The usage of N-SiC refractories for the increasing of productivity of aluminium reduction cells. Alum. Sib. 2003, 9, 203–208. [Google Scholar]
- Etzion, R.; Metson, J.B. Factors Affecting Corrosion Resistance of Silicon Nitride Bonded Silicon Carbide Refractory Blocks. J. Am. Ceram. Soc. 2012, 95, 410–415. [Google Scholar] [CrossRef]
- Paulek, R. SiC in Electrolysis Pots: An Update. Light Met. 2006, 135, 655–658. [Google Scholar]
- Kehren, J.T.; Steffen, T.; Hauke, M.; Linden, C.; Dannert, C.; Krause, O. The influence of firing parameters on formation of nitride phazez in nitride bonded silicon carbide. In Proceedings of the UNITECR, Frankfurt, Germany, 27–29 September 2023. [Google Scholar]
- Wang, Z.; Skybakmoen, E.; Grande, T. Spent Si3N4 Bonded Sidelining Materials in Aluminium Electrolysis Cells. Light Met. 2009, 353–358. [Google Scholar]
- Skybakmoen, E.; Grande, T.; Wang, Z. The influence of microstructure of Si3N4-SiC side-lining materials on chemical /oxidation resistance behavior tasted in laboratory scale. In Proceedings of the 11th Australasian Aluminium Smelting Technology Conference, Dubai, United Arab Emirates, 6–11 December 2014; Welch, B., Scillos-Kazakos, M., Eds.; UNSW: Sydney, Australia, 2014. ISBN 978-0-7334-3518-8. [Google Scholar]
- Skybakmoen, E. Quality Evaluation of Nitride bonded SiC Sidelining Materials. Historical Trends 1997-2022 Including Results and Development of Test Methods. Light Met. 2022, 921–928. [Google Scholar] [CrossRef]
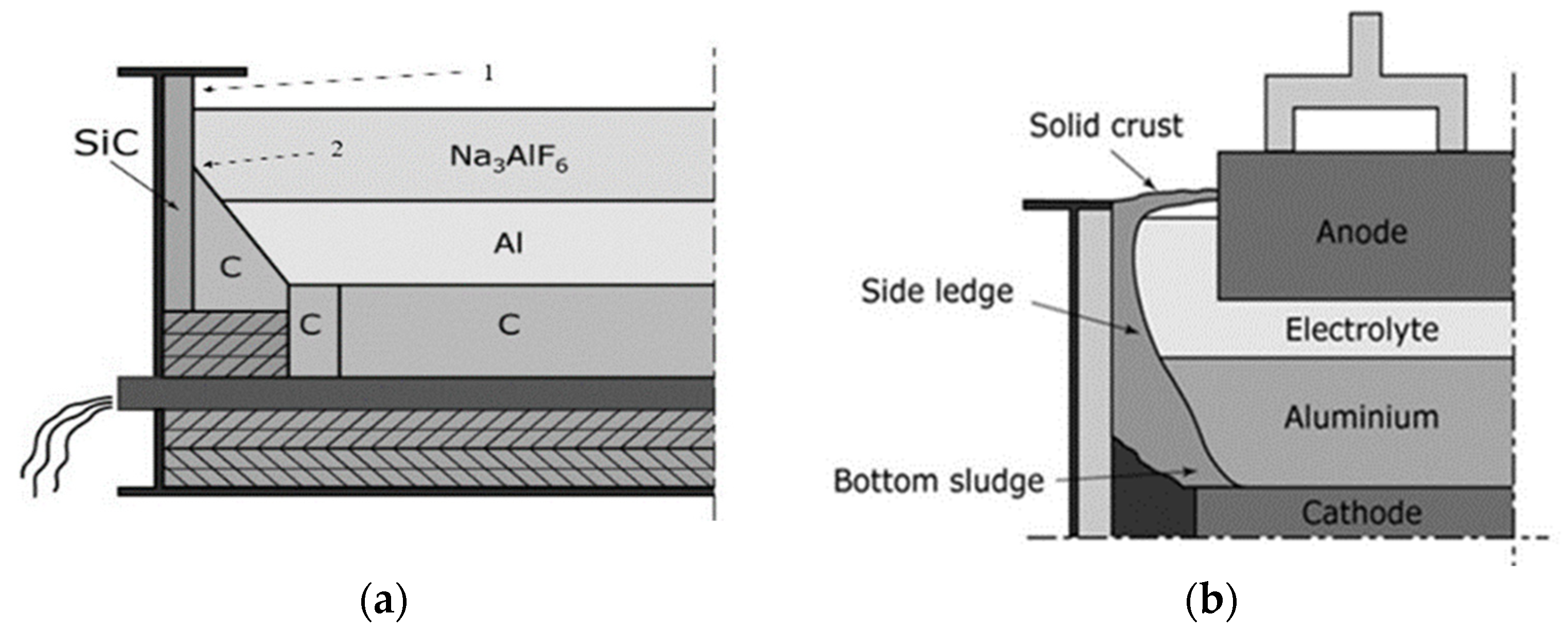




| Test | Electrolysis | Pre Oxidation | Atmosphere | Movement of Specimen Rods |
|---|---|---|---|---|
| SINTEF Skybakmoen [8,9] | yes | no | Air. Some vapors (mainly NaAIF4) and CO2/CO from the burning anode. | No |
| Lacournet [10] | no | yes | Air. Some vapors (mainly NaAIF4) | No |
| LIRR [11,12,13] | yes | no | Some vapors (mainly NaAIF4). CO2 flow | Yes. Rotation of rods |
| RUSAL Proshkin [14] | no | no | Some vapors (mainly NaAIF4). Air | Yes. Specimen rods are dipped in the cryolite and taken out |
| No | Apparent Density (Initial), g/cm3 | Apparent Density (After 180 Days), g/cm3 | Open Porosity (Initial), % | Open Porosity (After 180 Days), % |
|---|---|---|---|---|
| 1 | 2.68 | 2.75 | 15.8 | 10.4 |
| 2 | 2.68 | 2.77 | 15.6 | 7.5 |
| Composition | Service Time, Months | Comments | |||||
|---|---|---|---|---|---|---|---|
| SiC wt.% | Si3N4, wt.% | SiO2, wt.% | Si, wt.% | Oxides, Including Na2SiO3, wt.% | |||
| 1 [11] | 50.7 | 16.62 | 11.1 | - | 21.58 | 46 | - |
| 2 | 73.1 | 15.4 | 7.3 | - | 2.2 | 39 | Upper part |
| 3 | 68.1 | 18.2 | 7.2 | - | 6.5 | 39 | Lower part |
| 4 | 73.7 | 23.3 | 1.65 | 0.34 | 0.98 | 36 | Upper part |
| 5 | 71 | 25.3 | 2.3 | 0.3 | 1.1 | 36 | Lower part |
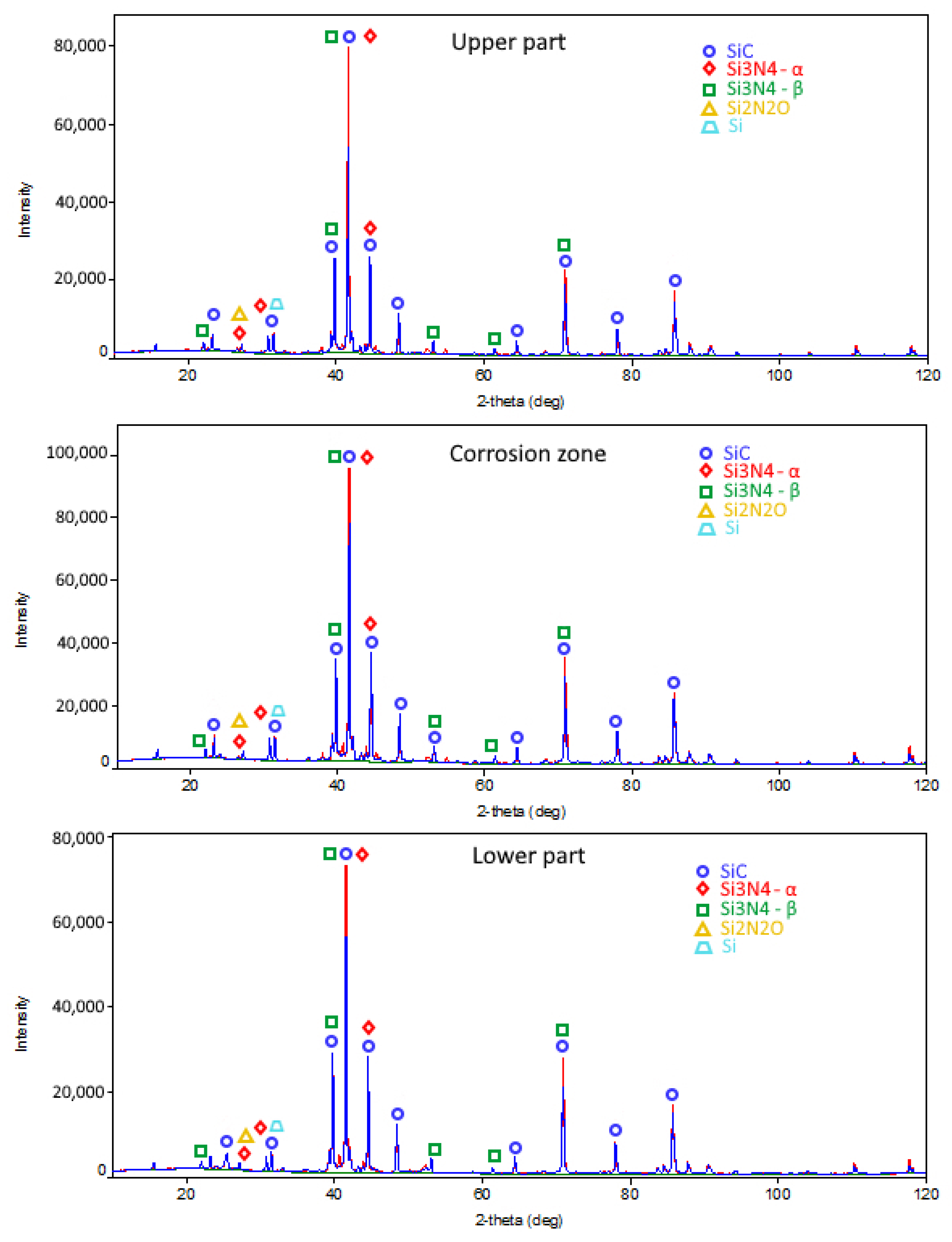
| SiC, wt.% | β-Si3N4, wt.% | α-Si3N4, wt.% | α/β | Σ Si3N4, wt.% | Si3N4/SiC | Si2ON2, wt.% | Si, wt.% | SiO2, wt.% | |
|---|---|---|---|---|---|---|---|---|---|
| before corrosion test | 81.6 | 5 | 10.1 | 2.02 | 15.1 | 0.185 | 3 | 0.3 | - |
| after corrosion test | |||||||||
| upper part | 73.3 | 6.7 | 10.5 | 1.57 | 17.2 | 0.23 | 4.5 | 5 | - |
| corrosion zone | 81.6 | 9 | 3.1 | 0.34 | 12.1 | 0.148 | 6.3 | - | - |
| lower part, dipped in cryolite | 76.3 | 6.7 | 11.1 | 1.66 | 17.8 | 0.23 | 3.5 | - | 2.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yurkov, A. Silicon Carbide-Silicon Nitride Materials: Part 2—Corrosion Resistance and Oxidation in Al Reduction Cells and at Lab Testing. Processes 2026, 14, 202. https://doi.org/10.3390/pr14020202
Yurkov A. Silicon Carbide-Silicon Nitride Materials: Part 2—Corrosion Resistance and Oxidation in Al Reduction Cells and at Lab Testing. Processes. 2026; 14(2):202. https://doi.org/10.3390/pr14020202
Chicago/Turabian StyleYurkov, Andrey. 2026. "Silicon Carbide-Silicon Nitride Materials: Part 2—Corrosion Resistance and Oxidation in Al Reduction Cells and at Lab Testing" Processes 14, no. 2: 202. https://doi.org/10.3390/pr14020202
APA StyleYurkov, A. (2026). Silicon Carbide-Silicon Nitride Materials: Part 2—Corrosion Resistance and Oxidation in Al Reduction Cells and at Lab Testing. Processes, 14(2), 202. https://doi.org/10.3390/pr14020202





