To complement calorimetry and determine the formation of ROS at 425 kHz, the sonoreactor using a set of well-established dosimeters that detect various aspects of cavitation chemistry was profiled. Potassium iodide measured the local oxidizing environment by forming I3− from I−. This process is initiated by •OH/HO2• and promoted by in situ H2O2. The strong absorbance of KI at 351 nm enables high sensitivity to interfacial radical fluxes. Bulk H2O2 accumulation integrated ROS production in the liquid phase, primarily the recombination of •OH. This provides a volumetric marker that scales with acoustic energy input. The Fricke system provided a quantitative standardized measure of hydroxyl radical yield under acidic conditions via Fe2+ conversion to Fe3+ with a defined response factor. Finally, 4-NP acted as an organic probe that was rapidly consumed by electrophilic •OH attack. This links dosimetric signals to pollutant-like reactivity. Together, KI, H2O2, the Fricke system, and 4-NP triangulated ROS generation across bubble–liquid regions and chemistries. This approach showed a relation between volumetric acoustic power density and specific oxidative pathways and enabled systematic evaluation of process variables.
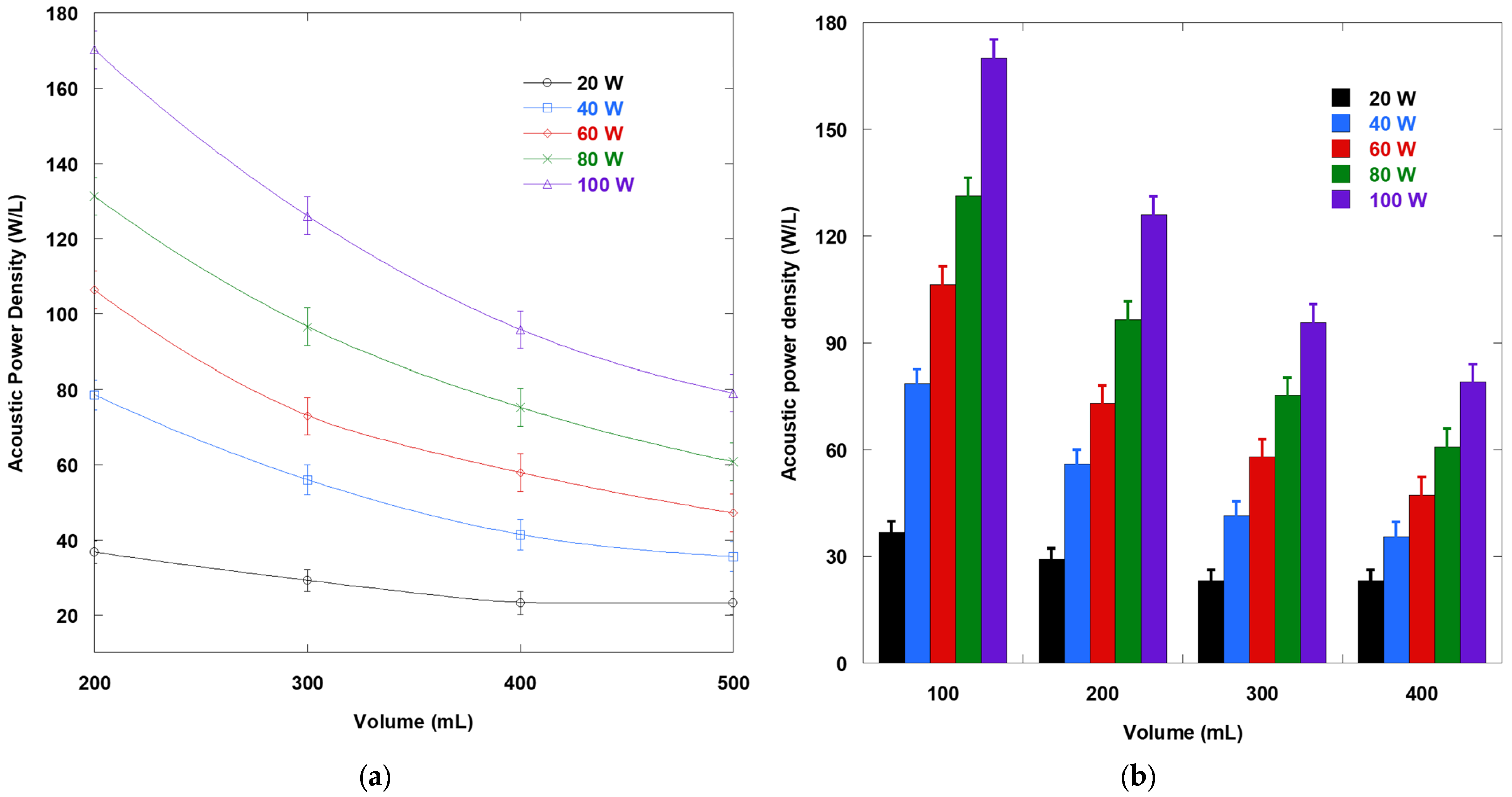
A working volume of 200 mL was selected because calorimetry demonstrated that, at a fixed electrical set power, the delivered acoustic power was essentially volume independent, so the volumetric acoustic power density decreased as the fill increased. Under typical conditions, this corresponded to nearly twice the power density at 200 mL compared with 500 mL, representing the strongest energy density achievable without hardware modification. From a reactor physics standpoint, at 425 kHz, a smaller working volume shortened the liquid height, reduced attenuation, and better aligned the bulk with antinode regions. At the same time, 200 mL avoided the drawbacks of very small fills, such as rapid heating, evaporation, and field distortion, while still delivering the high power density required to resolve kinetics over extended runs.
3.2.1. KI Dosimeter
To identify the primary ROS at 425 kHz ultrasonication, the formation of triiodide in 0.1 M KI in the absence and presence of AHM was examined. In KI solution, the
•OH radical oxidized iodide through a short chain (I
•/I
2•−) to form I
2, which rapidly converted to I
3−. The I
3− concentration was measured and found that its linear increase over time corresponded to the zero-order
•OH production rate during steady ultrasonication. In the presence of AHM, H
2O
2 formed from
•OH recombination was catalytically converted with I
− to I
2 (H
2O
2 + 2I
− → I
2 + 2OH
−). Thus, the measured I
3− slope reflects the total oxidant flux (
•OH + H
2O
2). Subtracting the KI-only slope yields the H
2O
2 formation rate. These procedures are commonly used to distinguish between radical and molecular oxidants produced by cavitation [
13].
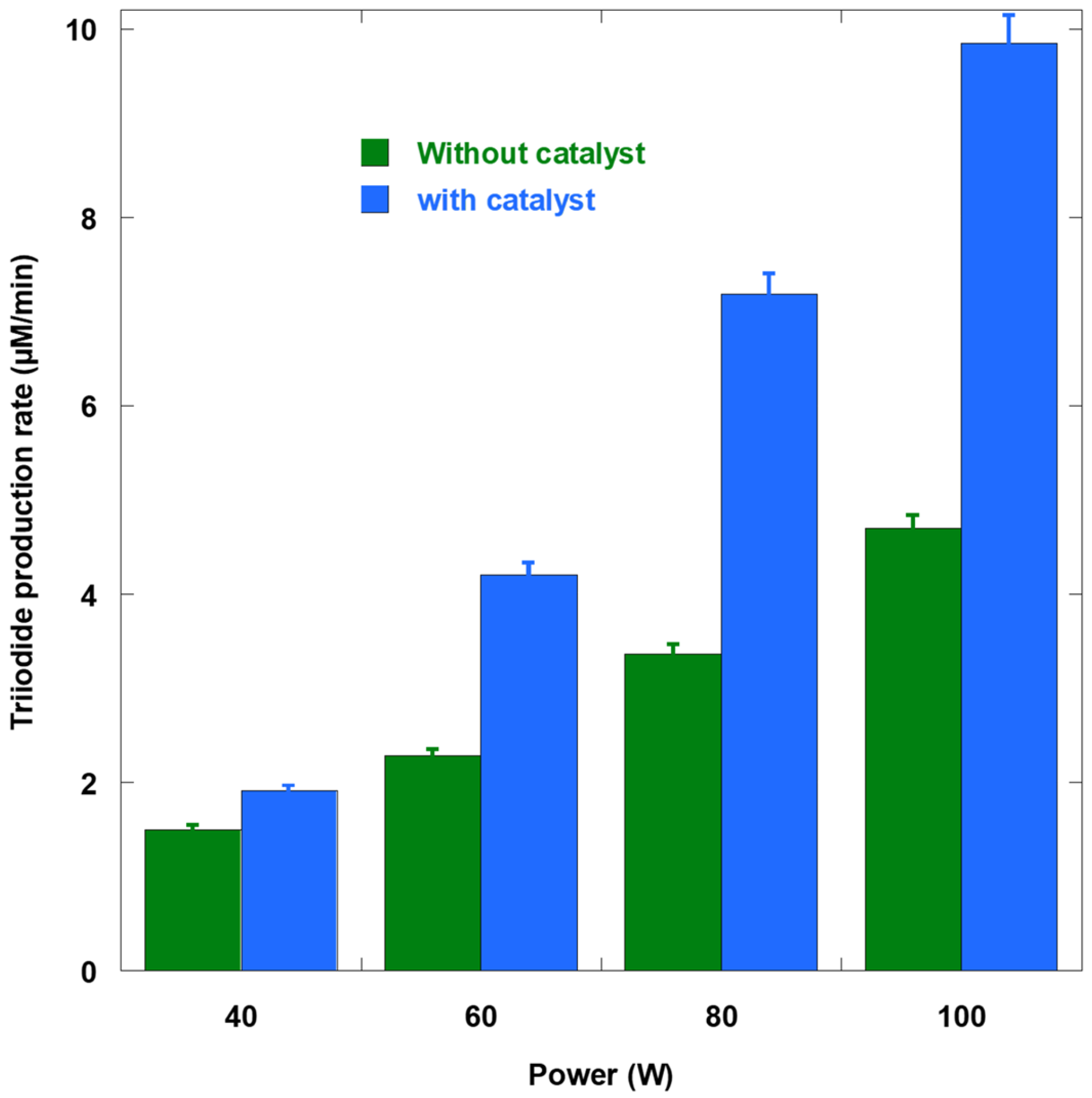
Figure 2 showed that the formation rate of I
3− from KI dosimetry increased monotonically with input power, regardless of the presence of AHM. When the KI-only slopes were interpreted as
•OH-equivalent production rates, the
•OH flux increased nearly linearly with input power. Specifically, it increases by a factor of 3.13 from 40 to 100 W (from 1.50 to 4.70 µM/min). This trend aligns with calorimetric power density scaling, wherein greater acoustic energy delivered per unit volume results in more violent collapses and higher
•OH production rates. As power increased, so did the fraction of the oxidant captured as H
2O
2 (21%, 46%, 53%, and 52% at 40, 60, 80, and 100 W, respectively). This suggested that, at higher acoustic intensities, a greater percentage of nascent
•OH species combined before reacting with solutes. This outcome is expected because the bubble number density and coalescence increase, thereby enhancing recombination pathways (
•OH +
•OH → H
2O
2). H
2O
2 is a stable molecular product that primarily forms through radical recombination in and around collapsing bubbles. The yield of H
2O
2 increases with ultrasonication power and acoustic path length.
In the presence of AHM, the enhancement increased from 1.3-fold at 40 W to 1.8-fold at 60 W to 2.1-fold at 80–100 W. This showed that H2O2 became a more significant sink for •OH at higher power settings. AHM mechanistically accelerates iodometric capture of H2O2 (H2O2 + 2I− → I2 + 2OH−), enabling real-time observation of the growing H2O2 pool. Without AHM, KI cannot practically detect H2O2 within the measurement timescale.
The production rate of •OH established the upper limit of pollutant transformation via direct radical attack, scaling with calorimetric acoustic power density. The H2O2 share indicates the proportion of the radical inventory diverted to non-radical oxidant pools. This information is crucial when coupling ultrasound with a process for which H2O2 is a useful co-reagent rather than a radical sink. Along with calorimetric maps, the results of the KI dosimeters (with and without AHM) provide a quantitative link between energy input, ROS speciation, and expected reactivity. These results will be used to interpret the behavior of the other dosimeters and the kinetics of SSY oxidation. KI (with and without AHM) remains a reliable method for determining the contributions of •OH and H2O2 in high-frequency reactors.
3.2.2. H2O2 Formation
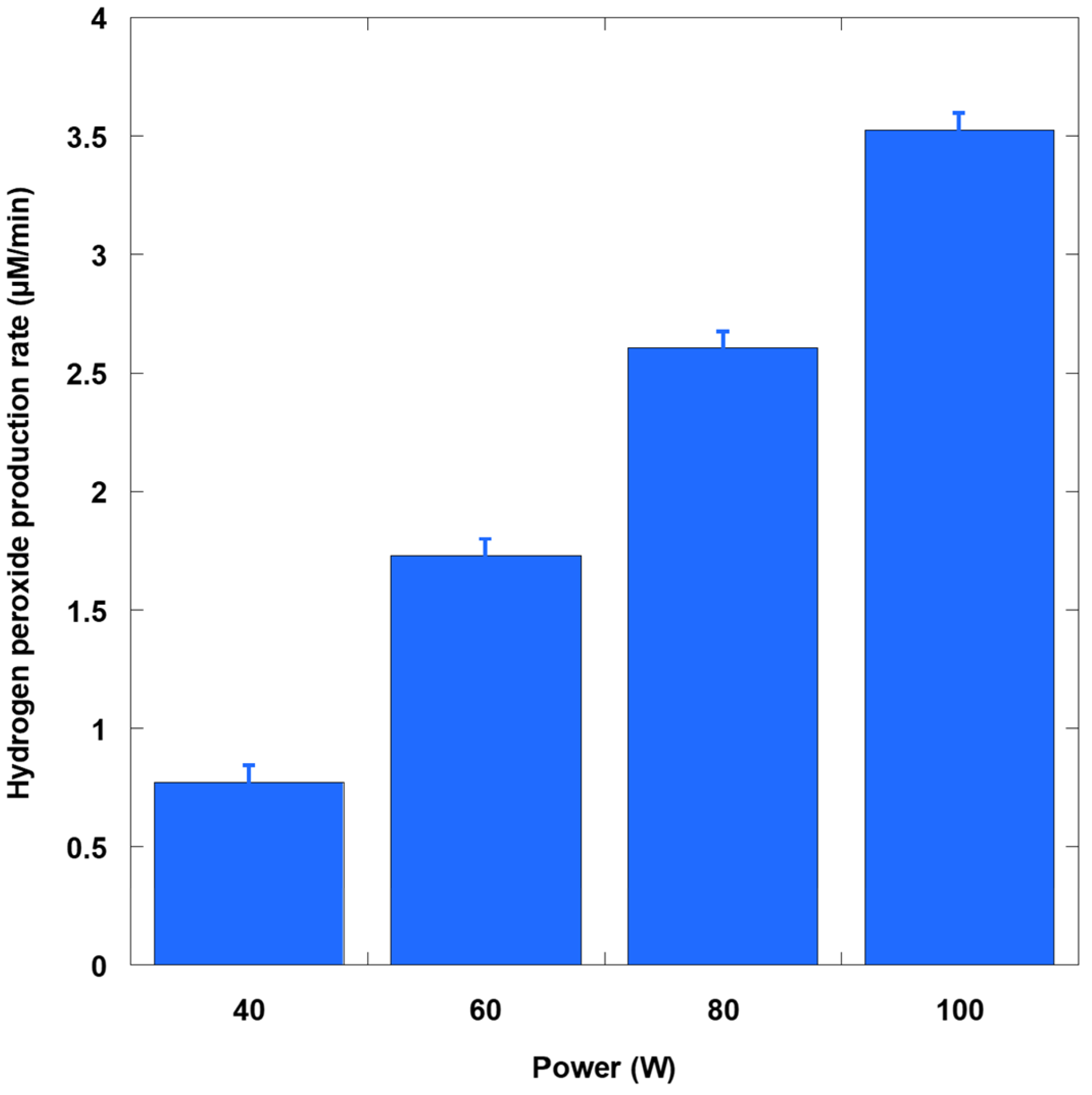
The production rate of H
2O
2 increased monotonically with input power (40 to 100 W), ranging from approximately 0.75 to 3.5 µM/min (
Figure 3). Since hydrogen peroxide in pure water primarily arose from hydroxyl radical self-recombination and, to a lesser extent, from hydroxyl radical pathways formed from hydrogen/oxygen reactions, its accumulation indicated net ROS generation under acoustic cavitation. Thus, increasing the delivered acoustic energy raises the number of active bubbles and their collapse intensity, thereby boosting primary water sonolysis (H
2O →
•OH +
•H) and subsequent ROS coupling to H
2O
2.
As expected, the scaling is not perfectly linear because the measured H2O2 rate reflected the balance between production and concurrent losses. At higher powers, ultrasound attenuation, bubble–bubble interactions, and local heating can reduce collapse severity (shielding), while H2O2 itself can be consumed by further sonolysis and radical reactions (e.g., H2O2 + •OH → HO2• + H2O), moderating net accumulation. Thus, the sublinear gain at the highest settings likely reflects physical (acoustic) and chemical (secondary ROS) limitations superimposed on the primary power effect.
Notably, the power-response profile aligned with earlier calorimetric mapping results. Increasing acoustic power density increases H2O2 productivity. This finding further establishes calorimetry as a meaningful normalization method for comparing reactor fills and geometries. Since H2O2 integrates ROS generated across hot spots and interfacial regions, these maps provide a useful baseline for interpreting the behavior of more selective dosimeters, such as KI, Fricke, and 4-NP. Ultimately, these maps clarify the trends in SSY abatement reported below.
To reconcile iodometric and hydrogen peroxide dosimeters, KI assays performed with and without AHM were compared to independent bulk H2O2 measurements, thereby quantifying how ROS are formed and evolve in the 425 kHz sonoreactor. In neat KI, I3− originates primarily from the Weissler reaction, in which interfacial •OH oxidizes I−. AHM addition generates catalytic species that rapidly oxidize I− using freshly formed H2O2 (H2O2 + 2 I− + 2 H+ → I2 + 2 H2O; I2 + I− → I3−). Because this catalytic pathway is both fast and selective, the increment in I3− production (ΔKI = with − without AHM) serves as a proxy for the instantaneous H2O2 formation rate near the bubble–liquid interface, before peroxide undergoes back-decomposition or diffusion losses.
A minimal kinetic framework rationalizes the divergence between KI without AHM, KI with AHM, and bulk H
2O
2 at high power. Let r
•OH denote the volumetric formation rate of
•OH radicals near the interface. Loss of
•OH radicals occurs by recombination, with second-order rate constant k
2, and by first-order scavenging, with effective rate constant k
S, which represents reactions with solutes and the liquid matrix. Under quasi-steady-state conditions:
The instantaneous interfacial H
2O
2 formation rate is then
Two limiting regimes follow:
When the •OH radical concentration scales linearly with r•OH, and the H2O2 formation rate scales with . In this regime, most oxidative capacity remains with short-lived radicals.
When the •OH radical concentration scales as , and the H2O2 formation rate approaches r•OH, oxidative capacity is transferred largely through molecular oxidants.
Representative aqueous constants support this shift. The reaction •OH + •OH → H2O2 has k2 = 5.5 × 109 M−1·s−1 at 25 °C. The reaction H2O2 + •OH → HO2• + H2O has k = 2.7 × 107 M−1·s−1, providing a significant back reaction at elevated radical levels. Hydroperoxyl self-reaction, HO2• + HO2• → H2O2 + O2, proceeds with k = 8 × 105 M−1·s−1. The acid–base pair HO2•/O2•− has pKa = 4.8, so conversion to superoxide increases with pH and alters secondary sinks.
These values explain the growing KI + AHM increments and the sublinear bulk H2O2 gains at the highest powers by showing that bimolecular termination accelerates strongly as r•OH increases and that secondary loss of H2O2 becomes non-negligible at elevated radical concentrations.
KI dosimetry without AHM gave triiodide formation rates of approximately 1.50, 2.28, 3.36, and 4.70 µM/min at 40, 60, 80, and 100 W, respectively. With AHM, the rates increased to about 1.91, 4.20, 7.18, and 9.85 µM/min, corresponding to boosts of nearly 27–110% as power increased. The AHM-induced increments (ΔKI) of nearly 0.41, 1.92, 3.82, and 5.15 µM/min thus reflect a growing peroxide contribution at higher acoustic powers. Independent bulk H2O2 accumulation assays yielded approximately 0.77, 1.73, 2.61, and 3.53 µM/min over the same power range. At powers of 80−100 W, these bulk rates fell below the corresponding ΔKI values, indicating that net peroxide destruction (via sonolysis inside/near bubbles and secondary radical reactions) increasingly offsets formation as the ultrasound field intensifies. This divergence is consistent with the dual role of H2O2 as both a product of •OH recombination and a substrate for further sonochemical degradation.
Because two hydroxyl radicals recombine to form one H2O2, the observed bulk accumulation provides a lower-bound recombination sink of 1.54, 3.46, 5.21, and 7.05 µM/min at 40–100 W. Adding these values to the KI without AHM rates (•OH captured by I−) yields minimum total •OH generation estimates of approximately 3.04, 5.74, 8.57, and 11.75 µM/min, respectively. Expressed as fractions, the share of •OH lost to recombination rises from about 51% at 40 W to 60% at 60−100 W, showing that stronger fields not only increase absolute radical production but also favor radical–radical coupling. This trend aligns with the expected behavior of high-frequency cavitation clouds, where higher power expands the active bubble population and shortens radical diffusion paths, thereby promoting bimolecular •OH loss to H2O2.
As an ensemble, the observations reveal that the two dosimetric approaches bracket the actual ROS dynamics. KI without AHM emphasizes interfacial •OH capture, KI with AHM approximates the combined flux of •OH and freshly formed H2O2 near bubbles, and bulk H2O2 accumulation reports the net peroxide that survives mixing and secondary chemistry in the sonoreactor volume. The internal consistency of these trends, and their systematic dependence on acoustic power, confirmed that power primarily scaled •OH generation while shifting a larger fraction toward H2O2 via recombination. The KI/AHM trap is thus valuable for capturing all oxidizing equivalents produced at the cavity interface, and using both KI and H2O2 readouts provides complementary bounds on ROS budgets that can be directly related to calorimetric acoustic power density for sonoreactor optimization and scale-up.
3.2.3. Fricke Dosimeter
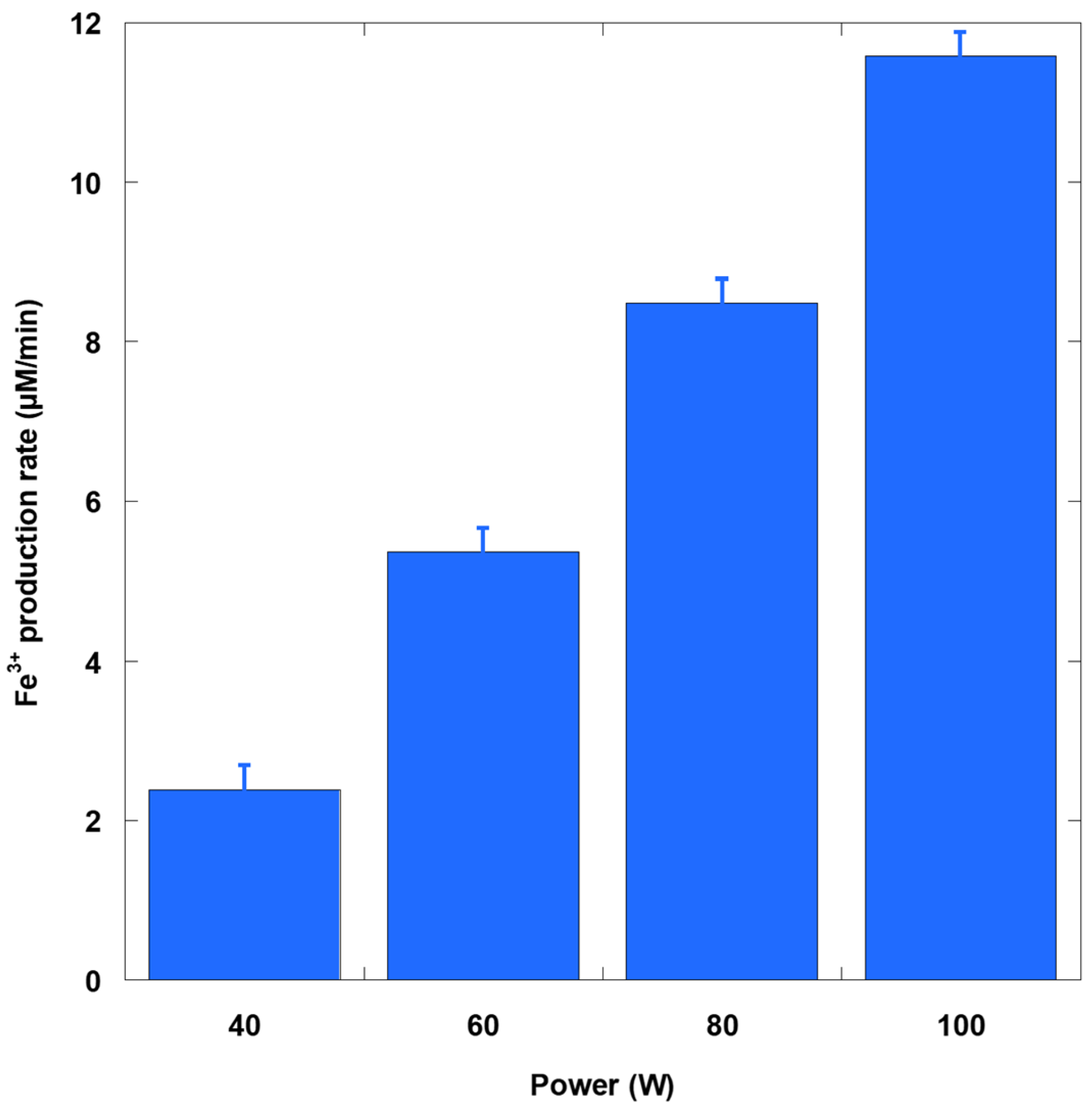
Figure 4 showed a monotonic increase in oxidizing capacity with acoustic power, as quantified by the Fricke dosimeter. As the input power rose from 40 to 100 W, the Fe
3+ formation rate increased from 2.39 to 11.58 μM/min. This scaling is expected, since stronger driving enhances both cavitation number density and collapse intensity, thereby increasing radical generation in the bubble–liquid interfacial zone and the surrounding liquid. In acidic Fricke solution, the primary oxidant is
•OH, with a smaller contribution from HO
2•/H
2O
2 via Fenton-type secondary chemistry. Each
•OH (and HO
2• under these conditions) oxidizes one Fe
2+ to Fe
3+, making the Fe
3+ formation rate a reliable indicator of the total oxidizing radical flux in the bulk liquid. Two features of the response are noteworthy. First, the rate–power curve becomes slightly superlinear at higher powers, consistent with the growth of nonlinear cavitation activity at elevated acoustic power densities. Second, absolute Fe
3+ formation rates are substantially higher than measured H
2O
2 production rates (
Figure 3), consistent with the Fricke system integrating contributions from both primary
•OH and secondary oxidants such as HO
2•/H
2O
2 in strongly acidic media.
The KI (Weissler) dosimeter corroborates the power dependence but also reveals chemistry-specific differences. The triiodide formation rate rises from 1.50 to 4.70 µM/min without AHM and from 1.91 to 9.85 with AHM as the power increases from 40 to 100 W. This enhancement stems from the well-established Mo(VI)-catalyzed oxidation of I− by H2O2 in the Weissler reaction. H2O2, formed by the recombination of •OH, enters a catalytic cycle that swiftly converts I− to I3−. This enables the KI assay to detect direct •OH attacks on I− and the H2O2 pathway. Consequently, the KI+AHM system reports a larger fraction of the overall ROS budget than the uncatalyzed KI system, and its rates approach those of the Fricke system.
Bulk H2O2 accumulation increased with power, rising from 0.77 to 3.52 µM/min over a range of 40 to 100 W. Because H2O2 is predominantly formed by radical recombination (2•OH → H2O2), the lower-bound •OH production rates are approximately twice the H2O2 rates (1.54, 3.46, 5.21, and 7.05 µM/min). These values remain lower than the Fricke Fe3+ and KI+AHM rates at the same powers. This outcome is consistent with the fact that H2O2 reflects only the recombination branch of •OH chemistry, is subject to back reactions and loss processes, and undergoes additional Fe2+ oxidation pathways in the Fricke solution. The qualitative ordering observed across all powers (Fricke ≥ KI+AHM > KI without AHM > H2O2) is therefore mechanistically coherent. Fricke dosimeter integrates •OH and secondary oxidants in an acidic environment, KI with AHM directly senses •OH and the H2O2 formed from •OH. Uncatalyzed KI primarily detects interfacial •OH, and H2O2 alone reflects only the recombination sink of •OH.
Synthesizing these findings, the three dosimeters provide a consistent picture of ROS generation at 425 kHz. Increasing power elevates acoustic power density, amplifying •OH formation and H2O2 yields downstream. The AHM-promoted KI response confirms that a significant portion of the oxidative capacity originates from the H2O2 produced by •OH recombination. Meanwhile, Fricke rates demonstrate that the total oxidizing equivalents accessible to the bulk liquid increase due to the efficient conversion of •OH and peroxy species into Fe3+ under acidic conditions.