Influence of Dielectric Barrier Discharge Plasma Treatment on Corn Starch Properties
Abstract
1. Introduction
2. Material and Methods
2.1. Materials
2.2. Plasma Treatment
2.3. Starch Characterization
2.3.1. Amylose
2.3.2. Solubility and Water Absorption Index (WAI)
2.3.3. Turbidity
2.3.4. Chemical Groups and Molecular Structure
2.3.5. Morphology
2.4. Starch Flowability
2.5. Statistical Analysis
3. Results and Discussion
3.1. Amylose
3.2. Solubility and Water Absorbance Index
3.3. Turbidity
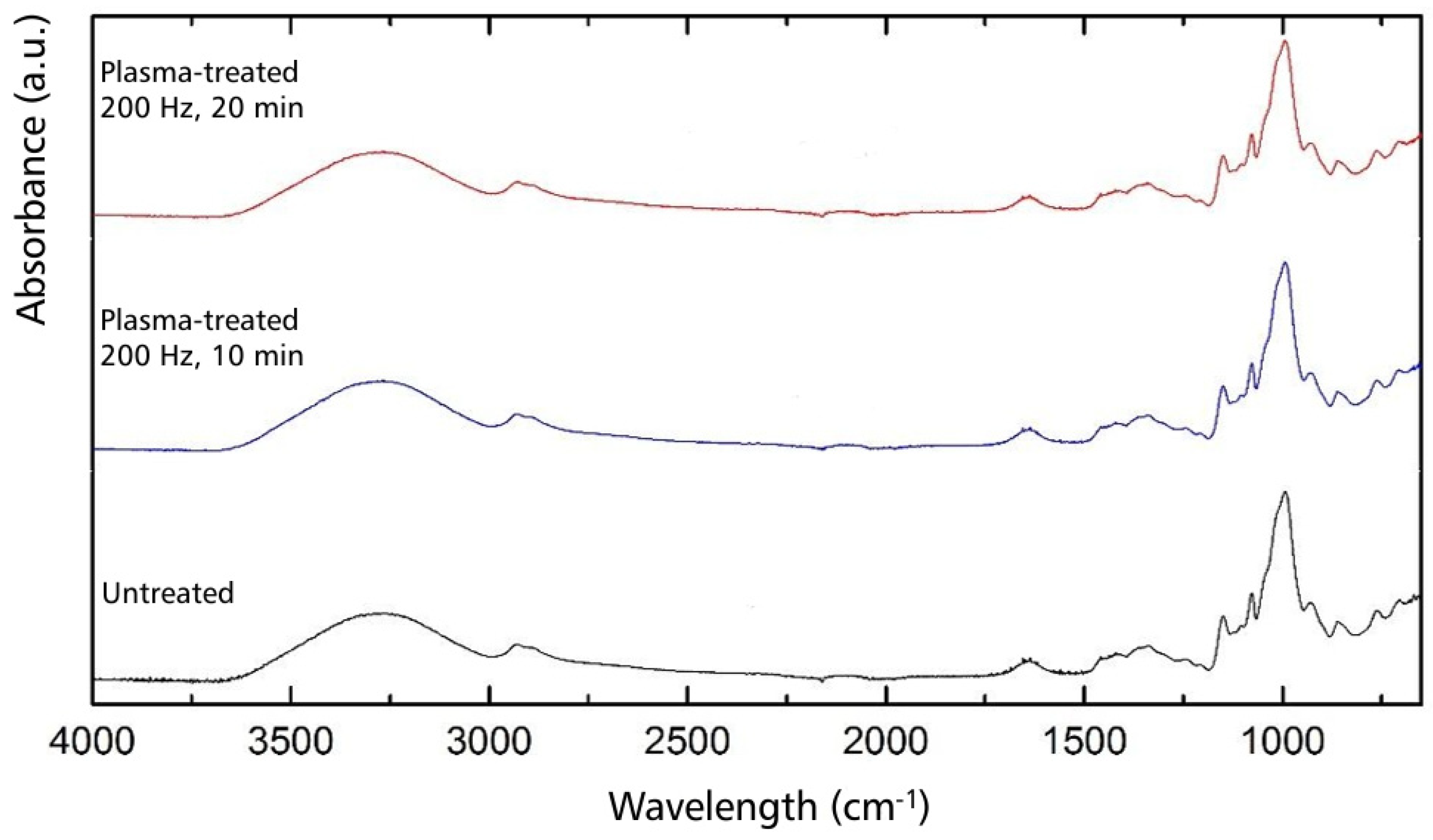
3.4. Molecular Structure
3.5. Surface Morphology
3.6. Powder Flowability
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, X.; Fang, J.; Cheng, L.; Gu, Z.; Hong, Y. Interaction of starch and non-starch polysaccharides in raw potato flour and their effects on thickening stability. Int. J. Biol. Macromol. 2023, 242, 124702. [Google Scholar] [CrossRef]
- Li, X.; Hu, B.; Ma, R.; Zhang, X.; Sun, C.; Zhao, Y.; Fang, Y. Core-shell starch as a platform for reducing starch digestion and saturated fat intake. Biomaterials 2023, 299, 122144. [Google Scholar] [CrossRef]
- Sun, X.; Saleh, A.S.; Sun, Z.; Ge, X.; Shen, H.; Zhang, Q.; Yu, X.; Yuan, L.; Li, W. Modification of multi-scale structure, physicochemical properties, and digestibility of rice starch via microwave and cold plasma treatments. LWT 2022, 153, 112483. [Google Scholar] [CrossRef]
- Apostolidis, E.; Stergiou, A.; Kioupis, D.; Sadeghpour, A.; Paximada, P.; Kakali, G.; Mandala, I. Production of nanoparticles from resistant starch via a simple three-step physical treatment. Food Hydrocoll. 2023, 137, 108412. [Google Scholar] [CrossRef]
- Luo, W.; Li, B.; Zhang, Y.; Tan, L.; Hu, C.; Huang, C.; Chen, Z.; Huang, L. Unveiling the retrogradation mechanism of a novel high amylose content starch-Pouteria campechiana seed. Food Chem. X 2023, 18, 100637. [Google Scholar] [CrossRef] [PubMed]
- Purwitasari, L.; Wulanjati, M.P.; Pranoto, Y.; Witasari, L.D. Characterization of porous starch from edible canna (Canna edulis Kerr.) produced by enzymatic hydrolysis using thermostable α-amylase. Food Chem. Adv. 2023, 2, 100152. [Google Scholar] [CrossRef]
- Luo, X.-E.; Wang, R.-Y.; Wang, J.-H.; Li, Y.; Luo, H.-N.; Zeng, X.-A.; Woo, M.-W.; Han, Z. Combining pulsed electric field and cross-linking to enhance the structural and physicochemical properties of corn porous starch. Food Chem. 2023, 418, 135971. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wu, Z.; Meng, Y.; Wang, Z.; He, X.; Gu, J.; Zhang, Y.; Wang, L.; Qin, X. Cationic etherification modification of corn starch and its sizing property. Text. Res. J. 2023, 004051752311638. [Google Scholar] [CrossRef]
- Dong, F.; Gao, W.; Liu, P.; Kang, X.; Yu, B.; Cui, B. Digestibility, structural and physicochemical properties of microcrystalline butyrylated pea starch with different degree of substitution. Carbohydr. Polym. 2023, 314, 120927. [Google Scholar] [CrossRef]
- Zhu, F. Plasma modification of starch. Food Chem. 2017, 232, 476–486. [Google Scholar] [CrossRef]
- Jang, H.J.; Jung, E.Y.; Parsons, T.; Tae, H.-S.; Park, C.-S. A Review of Plasma Synthesis Methods for Polymer Films and Nanoparticles under Atmospheric Pressure Conditions. Polymers 2021, 13, 2267. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Gou, Q.; Yang, L.; Yu, Q.-L.; Han, L. Dielectric barrier discharge plasma: A green method to change structure of potato starch and improve physicochemical properties of potato starch films. Food Chem. 2022, 370, 130992. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.-J.; Liu, C.-J.; Eliasson, B. Modification of starch by glow discharge plasma. Carbohydr. Polym. 2004, 55, 23–26. [Google Scholar] [CrossRef]
- Goiana, M.L.; de Brito, E.S.; Filho, E.G.A.; Miguel, E.D.C.; Fernandes, F.A.N.; de Azeredo, H.M.C.; Rosa, M.D.F. Corn starch based films treated by dielectric barrier discharge plasma. Int. J. Biol. Macromol. 2021, 183, 2009–2016. [Google Scholar] [CrossRef]
- Bie, P.; Pu, H.; Zhang, B.; Su, J.; Chen, L.; Li, X. Structural characteristics and rheological properties of plasma-treated starch. Innov. Food Sci. Emerg. Technol. 2016, 34, 196–204. [Google Scholar] [CrossRef]
- Hernandez-Perez, P.; Flores-Silva, P.C.; Velazquez, G.; Morales-Sanchez, E.; Rodríguez-Fernández, O.; Hernández-Hernández, E.; Mendez-Montealvo, G.; Sifuentes-Nieves, I. Rheological performance of film-forming solutions made from plasma-modified starches with different amylose/amylopectin content. Carbohydr. Polym. 2020, 255, 117349. [Google Scholar] [CrossRef] [PubMed]
- Okyere, A.Y.; Boakye, P.G.; Bertoft, E.; Annor, G.A. Temperature of plasma-activated water and its effect on the thermal and chemical surface properties of cereal and tuber starches. Curr. Res. Food Sci. 2022, 5, 1668–1675. [Google Scholar] [CrossRef]
- Lii, C.Y.; Liao, C.D.; Stobinski, L.; Tomasik, P. Behaviour of granular starches in low-pressure glow plasma. Carbohydr. Polym. 2002, 49, 499–507. [Google Scholar] [CrossRef]
- Carvalho, A.P.M.G.; Barros, D.R.; da Silva, L.S.; Sanches, E.A.; Pinto, C.D.C.; de Souza, S.M.; Clerici, M.T.P.S.; Rodrigues, S.; Fernandes, F.A.N.; Campelo, P.H. Dielectric barrier atmospheric cold plasma applied to the modification of Ariá (Goeppertia allouia) starch: Effect of plasma generation voltage. Int. J. Biol. Macromol. 2021, 182, 1618–1627. [Google Scholar] [CrossRef]
- Shen, H.; Ge, X.; Zhang, Q.; Zhang, X.; Lu, Y.; Jiang, H.; Zhang, G.; Li, W. Dielectric barrier discharge plasma improved the fine structure, physicochemical properties and digestibility of α-amylase enzymatic wheat starch. Innov. Food Sci. Emerg. Technol. 2022, 78, 102991. [Google Scholar] [CrossRef]
- Chaiwat, W.; Wongsagonsup, R.; Tangpanichyanon, N.; Jariyaporn, T.; Deeyai, P.; Suphantharika, M.; Fuongfuchat, A.; Nisoa, M.; Dangtip, S. Argon Plasma Treatment of Tapioca Starch Using a Semi-continuous Downer Reactor. Food Bioprocess Technol. 2016, 9, 1125–1134. [Google Scholar] [CrossRef]
- Fernandes, F.A.; Santos, V.O.; Rodrigues, S. Effects of glow plasma technology on some bioactive compounds of acerola juice. Food Res. Int. 2018, 115, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Marenco-Orozco, G.A.; Rosa, M.F.; Fernandes, F.A.N. Effects of multiple-step cold plasma processing on banana (Musa sapientum) starch-based films. Packag. Technol. Sci. 2022, 35, 589–601. [Google Scholar] [CrossRef]
- Hu, J.; Cheng, F.; Lin, Y.; Zhao, K.; Zhu, P. Dissolution of starch in urea/NaOH aqueous solutions. J. Appl. Polym. Sci. 2016, 133, 43390. [Google Scholar] [CrossRef]
- Fang, C.; Huang, J.; Pu, H.; Yang, Q.; Chen, Z.; Zhu, Z. Cold-water solubility, oil-adsorption and enzymolysis properties of amorphous granular starches. Food Hydrocoll. 2021, 117, 106669. [Google Scholar] [CrossRef]
- Chen, G.; Chen, Y.; Jin, N.; Li, J.; Dong, S.; Li, S.; Zhang, Z. Zein films with porous polylactic acid coatings via cold plasma pre-treatment. Ind. Crop. Prod. 2020, 150, 112382. [Google Scholar] [CrossRef]
- Warren, F.J.; Gidley, M.J.; Flanagan, B.M. Infrared spectroscopy as a tool to characterise starch ordered structure—A joint FTIR–ATR, NMR, XRD and DSC study. Carbohydr. Polym. 2016, 139, 35–42. [Google Scholar] [CrossRef]
- Lima, A.C.d.S.; Afonso, M.R.A.; Rodrigues, S.; de Aquino, A.C. Flowability of spray-dried sapodilla pulp powder. J. Food Process. Eng. 2022, 45, e14092. [Google Scholar] [CrossRef]
- Filho, E.G.A.; Cullen, P.J.; Frias, J.M.; Bourke, P.; Tiwari, B.K.; Brito, E.S.; Rodrigues, S.; Fernandes, F.A. Evaluation of plasma, high-pressure and ultrasound processing on the stability of fructooligosaccharides. Int. J. Food Sci. Technol. 2016, 51, 2034–2040. [Google Scholar] [CrossRef]
- Almeida, F.D.L.; Gomes, W.F.; Cavalcante, R.S.; Tiwari, B.K.; Cullen, P.J.; Frias, J.M.; Bourke, P.; Fernandes, F.A.; Rodrigues, S. Fructooligosaccharides integrity after atmospheric cold plasma and high-pressure processing of a functional orange juice. Food Res. Int. 2017, 102, 282–290. [Google Scholar] [CrossRef]
- DeNardin, C.C.; da Silva, L.P. Estrutura dos grânulos de amido e sua relação com propriedades físico-químicas. Ciência Rural 2009, 39, 945–954. [Google Scholar] [CrossRef]
- Thirumdas, R.; Trimukhe, A.; Deshmukh, R.; Annapure, U. Functional and rheological properties of cold plasma treated rice starch. Carbohydr. Polym. 2017, 157, 1723–1731. [Google Scholar] [CrossRef]
- Yan, S.; Chen, G.; Hou, Y.; Chen, Y. Improved solubility of banana starch by dielectric barrier discharge plasma treatment. Int. J. Food Sci. Technol. 2020, 55, 641–648. [Google Scholar] [CrossRef]
- Kizil, R.; Irudayaraj, J.; Seetharaman, K. Characterization of Irradiated Starches by Using FT-Raman and FTIR Spectroscopy. J. Agric. Food Chem. 2002, 50, 3912–3918. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Guo, Y.; Zhao, J.; Zhao, J.; Shen, H.; Yan, W. Dielectric barrier discharge cold plasma combined with cross-linking: An innovative way to modify the multi-scale structure and physicochemical properties of corn starch. Int. J. Biol. Macromol. 2022, 215, 465–476. [Google Scholar] [CrossRef]
- Wang, J.; Guo, K.; Fan, X.; Feng, G.; Wei, C. Physicochemical Properties of C-Type Starch from Root Tuber of Apios fortunei in Comparison with Maize, Potato, and Pea Starches. Molecules 2018, 23, 2132. [Google Scholar] [CrossRef]
- Gao, S.; Liu, H.; Sun, L.; Cao, J.; Yang, J.; Lu, M.; Wang, M. Rheological, thermal and in vitro digestibility properties on complex of plasma modified Tartary buckwheat starches with quercetin. Food Hydrocoll. 2020, 110, 106209. [Google Scholar] [CrossRef]
- Shen, H.; Ge, X.; Zhang, B.; Su, C.; Zhang, Q.; Jiang, H.; Zhang, G.; Yuan, L.; Yu, X.; Li, W. Preparing potato starch nanocrystals assisted by dielectric barrier discharge plasma and its multiscale structure, physicochemical and rheological properties. Food Chem. 2022, 372, 131240. [Google Scholar] [CrossRef]

| Plasma Generation Frequency | Processing Time | Amylose Content | Amylopectin Content |
|---|---|---|---|
| (Hz) | (min) | (%) | (%) |
| 0 | 0 | 24.2 ± 0.3 a | 75.8 ± 0.3 a |
| 100 | 10 | 21.6 ± 0.4 b | 78.4 ± 0.4 b |
| 100 | 20 | 23.7 ± 0.9 ac | 76.3 ± 0.9 c |
| 200 | 10 | 22.1 ± 1.2 c | 77.9 ± 1.2 b |
| 200 | 20 | 20.8 ± 0.9 bc | 79.2 ± 0.9 d |
| 300 | 10 | 22.4 ± 0.4 c | 77.6 ± 0.4 b |
| 300 | 20 | 23.7 ± 0.4 ac | 76.3 ± 0.4 c |
| Excitation Frequency (Hz) | Processing Time (min) | Solubility (%) | Water Absorption Index (%) | Turbidity |
|---|---|---|---|---|
| 0 (Control) | 90.6 ± 0.63 a | 1.97 ± 0.05 a | 1.73 ± 0.03 a | |
| 100 | 10 | 83.8 ± 0.60 b | 1.98 ± 0.11 a | 1.73 ± 0.01 a |
| 100 | 20 | 83.7 ± 0.84 b | 1.97 ± 0.04 a | 1.78 ± 0.02 ab |
| 200 | 10 | 82.6 ± 1.38 ab | 2.07 ± 0.47 ab | 1.77 ± 0.02 ab |
| 200 | 20 | 84.0 ± 1.17 b | 2.07 ± 0.16 ab | 1.78 ± 0.02 ab |
| 300 | 10 | 83.3 ± 1.10 ab | 2.10 ± 0.10 b | 1.77 ± 0.02 ab |
| 300 | 20 | 82.7 ± 0.79 ab | 2.18 ± 0.08 b | 1.81 ± 0.02 b |
| Freq. (Hz) | Time (min) | Absorbance (cm−1) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2900 | 1337 | 1150 | 1102 | 1078 | 1045 | 1022 | 995 | 962 | 762 | 703 | ||
| Control | 0 | 0.085 | 0.084 | 0.166 | 0.140 | 0.232 | 0.280 | 0.402 | 0.514 | 0.242 | 0.180 | 0.209 |
| 100 | 10 | 0.086 | 0.084 | 0.166 | 0.141 | 0.230 | 0.271 | 0.396 | 0.500 | 0.239 | 0.179 | 0.206 |
| 100 | 20 | 0.087 | 0.086 | 0.170 | 0.144 | 0.236 | 0.284 | 0.407 | 0.519 | 0.246 | 0.184 | 0.212 |
| 200 | 10 | 0.098 | 0.097 | 0.191 | 0.161 | 0.265 | 0.318 | 0.459 | 0.594 | 0.279 | 0.208 | 0.240 |
| 200 | 20 | 0.091 | 0.089 | 0.176 | 0.149 | 0.244 | 0.293 | 0.422 | 0.537 | 0.257 | 0.190 | 0.217 |
| 300 | 10 | 0.089 | 0.087 | 0.172 | 0.144 | 0.238 | 0.286 | 0.411 | 0.525 | 0.246 | 0.188 | 0.216 |
| 300 | 20 | 0.085 | 0.083 | 0.167 | 0.141 | 0.233 | 0.282 | 0.405 | 0.517 | 0.244 | 0.180 | 0.207 |
| Frequency (Hz) | Time (min) | 1045/1022 cm−1 | 995/1022 cm−1 |
|---|---|---|---|
| Control | -- | 0.697 | 1.279 |
| 100 | 10 | 0.699 | 1.263 |
| 100 | 20 | 0.698 | 1.275 |
| 200 | 10 | 0.693 | 1.294 |
| 200 | 20 | 0.694 | 1.273 |
| 300 | 10 | 0.696 | 1.277 |
| 300 | 20 | 0.696 | 1.277 |
| Control | Plasma-Treated | ||
|---|---|---|---|
| δ1 (kPa) | δc (kPa) | δ1 (kPa) | δc (kPa) |
| 1.212 | 0.496 | 1.249 | 0.571 |
| 2.576 | 0.778 | 2.555 | 0.817 |
| 4.875 | 1.019 | 4.922 | 1.046 |
| 9.158 | 1.473 | 10.723 | 1.031 |
| 23.624 | 2.366 | 24.785 | 3.743 |
| Untreated | Plasma-Treated | |
|---|---|---|
| Bulk density (kg/m3) | 421.0 | 409.1 |
| Tapped density (kg/m3) | 1198.4 | 1125.1 |
| Compressibility index (%) | 43.73 | 42.99 |
| Hausner ratio | 1.78 | 1.75 |
| Maximum Cohesion (kPa) | 0.689 | 1.112 |
| Minimum wall friction angle (°) | 18.6 | 19.9 |
| Maximum wall friction angle (°) | 23.7 | 26.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goiana, M.L.; Fernandes, F.A.N. Influence of Dielectric Barrier Discharge Plasma Treatment on Corn Starch Properties. Processes 2023, 11, 1966. https://doi.org/10.3390/pr11071966
Goiana ML, Fernandes FAN. Influence of Dielectric Barrier Discharge Plasma Treatment on Corn Starch Properties. Processes. 2023; 11(7):1966. https://doi.org/10.3390/pr11071966
Chicago/Turabian StyleGoiana, Mayara L., and Fabiano A. N. Fernandes. 2023. "Influence of Dielectric Barrier Discharge Plasma Treatment on Corn Starch Properties" Processes 11, no. 7: 1966. https://doi.org/10.3390/pr11071966
APA StyleGoiana, M. L., & Fernandes, F. A. N. (2023). Influence of Dielectric Barrier Discharge Plasma Treatment on Corn Starch Properties. Processes, 11(7), 1966. https://doi.org/10.3390/pr11071966








