Effect of Different Concentrations of NiMnGa Micro/Nanoparticles on the Kinetics of Natural Gas Hydration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Experimental Apparatus
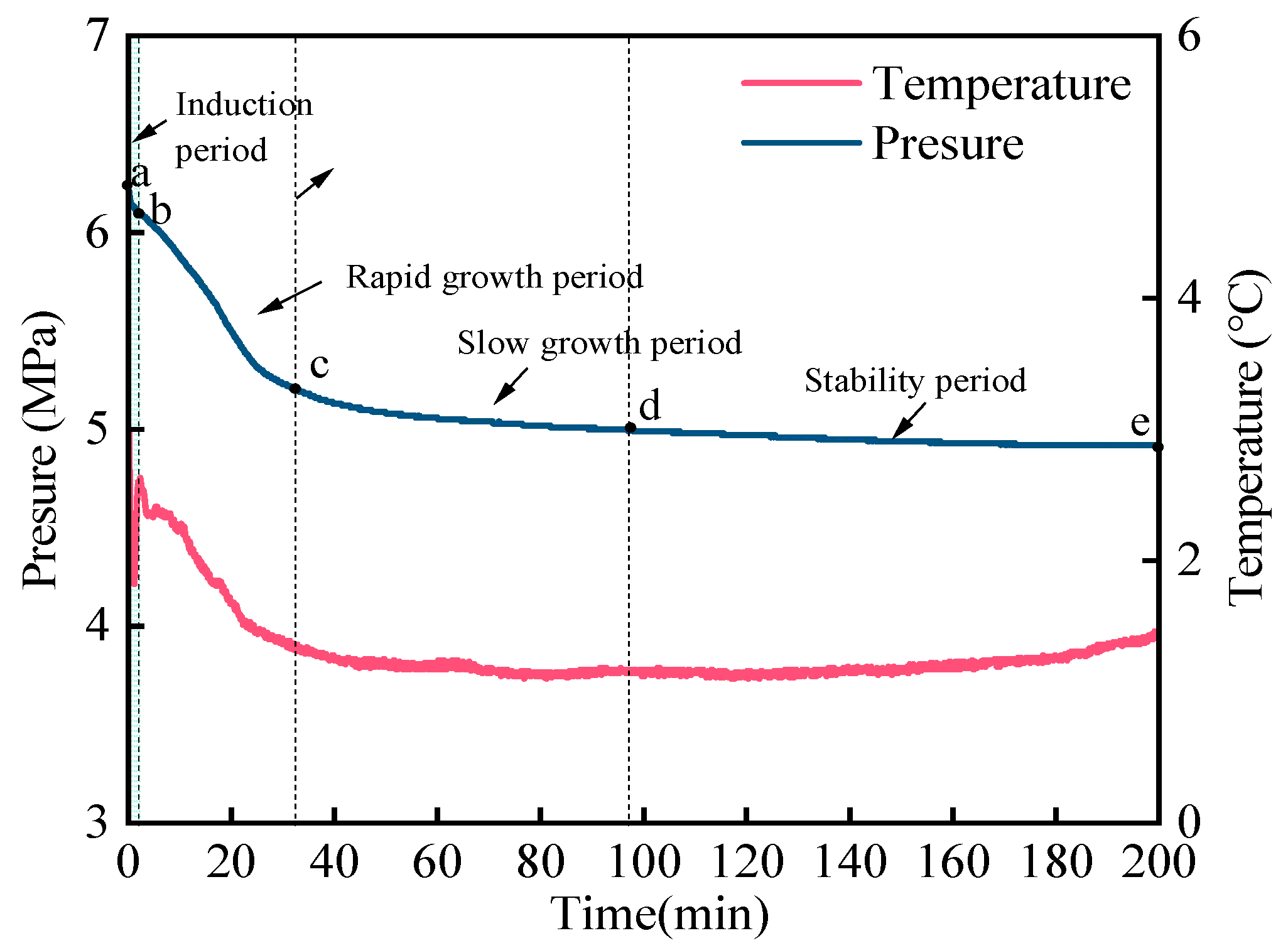
2.3. Experimental Procedure
2.4. Determination of Kinetic Parameters
3. Results
4. Discussion
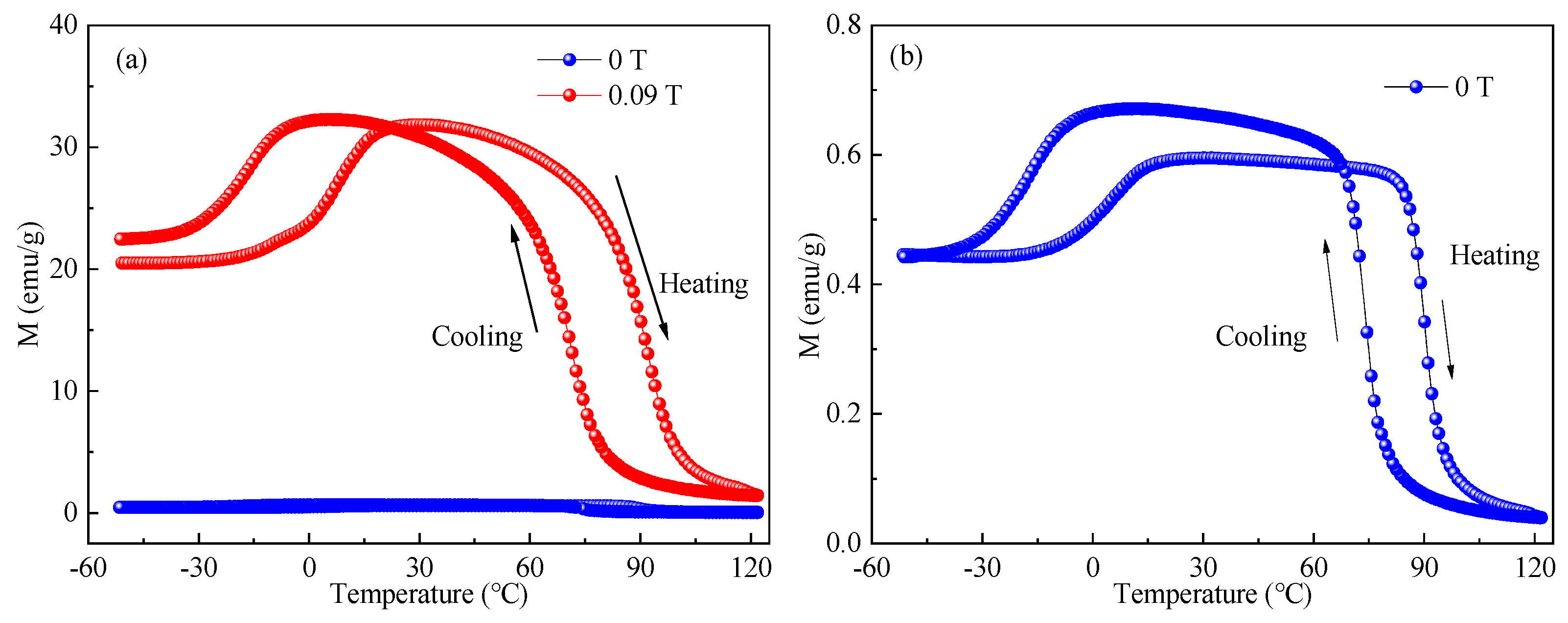
4.1. The Magnetic Properties of NiMnGa Alloy Particles
4.2. The Effect of NiMnGa Particle Concentration
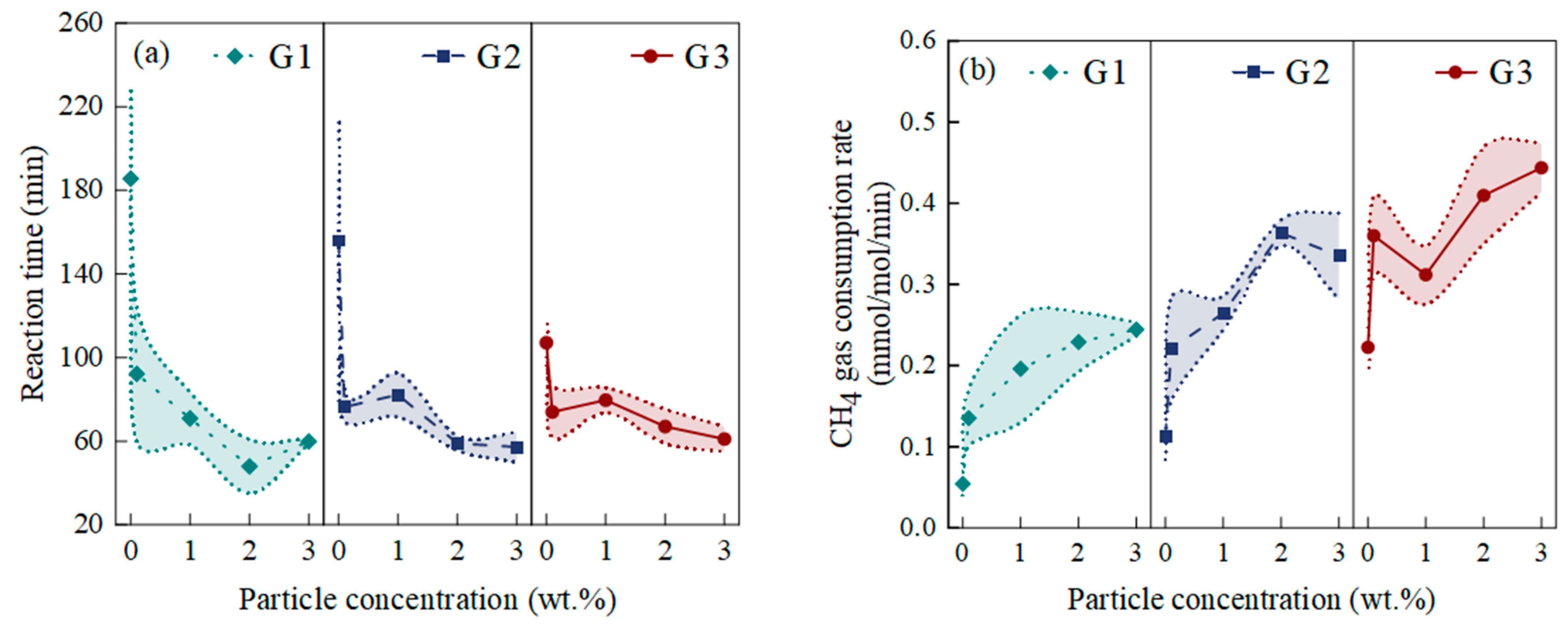
4.2.1. The Induction Time
4.2.2. The Gas Consumption
4.2.3. The Gas Consumption Rate
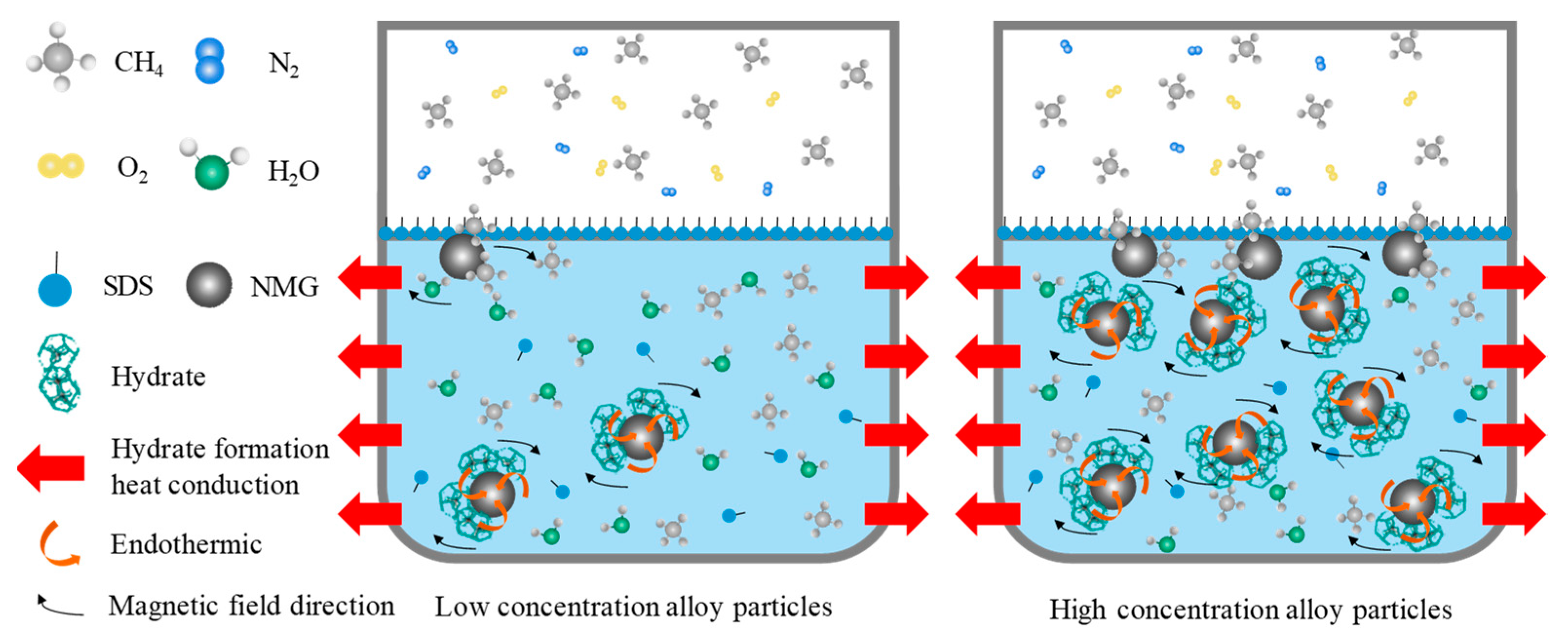
4.3. Analysis of Gas Hydrate Enhancement by NiMnGa Micro/Nanofluids
4.3.1. The Effect of NiMnGa Particle Concentration on the Kinetics of the Gas Hydration Reaction
4.3.2. The Effect of External Magnetic Field on the Kinetics of Gas Hydration Reaction in NiMnGa Micro/Nanofluids
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sloan, E.D., Jr. Fundamental principles and applications of natural gas hydrates. Nature 2003, 426, 353–359. [Google Scholar] [CrossRef]
- Nguyen, N.N.; Nguyen, A.V. Recent insights into the anomalous dual nature (both promotion and Inhibition) of chemical additives on gas hydrate formation. Chem. Eng. J. 2023, 475, 146362. [Google Scholar] [CrossRef]
- Chen, G.J.; Sun, C.Y.; Ma, Q.L. Gas Hydrate Science and Technology; Chemical Industry Press: Beijing, China, 2007; pp. 1–30. [Google Scholar]
- Lee, Y.; Seo, D.; Lee, S.; Park, Y. Unlocking enhanced gas storage capacity in tuned methane hydrates: Exploring eutectic compositions and water-to-hydrate conversion. Chem. Eng. J. 2023, 475, 146381. [Google Scholar] [CrossRef]
- Abedi-Farizhendi, S.; Iranshahi, M.; Mohammadi, A.; Manteghian, M.; Mohammadi, A.H. Kinetic study of methane hydrate formation in the presence of carbon nanostructures. Pet. Sci. 2019, 16, 657–668. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, R.D.; Xu, H.J.; Tian, C.S. Recent Advances on experimental study of nucleation process of methane hydrate (Invited). Acta Photonica Sin. 2021, 50, 82–96. [Google Scholar]
- Seo, D.; Lee, S.; Moon, S.; Lee, Y.; Park, Y. Investigating two synthetic routes for gas hydrate formation to control the trapping of methane from natural gas. Chem. Eng. J. 2023, 467, 143512. [Google Scholar] [CrossRef]
- Fan, S.S.; Yu, C.; Lang, X.M.; Wang, Y.H.; Chen, J.B. Micro-Nano-Scale studies on occurrence and gas production and storage technology of marine gas hydrates. Earth Sci. 2018, 43, 1542–1548. [Google Scholar]
- Lang, X.M.; Fan, S.S.; Wang, Y.H.; Li, G.; Yu, C.; Wang, S.L. Opportunities for energy and chemical engineering through clathrate hydrates. Chem. Ind. Eng. Prog. 2021, 40, 4703–4710. [Google Scholar]
- Huang, L.; Tan, J.; Fu, H.; Liu, J.; Chen, X.; Liao, X.; Wang, X.; Wang, C. The non-plane initiation and propagation mechanism of multiple hydraulic fractures in tight reservoirs considering stress shadow effects. Eng. Fract. Mech. 2023, 292, 109570. [Google Scholar] [CrossRef]
- Dontsov, E.V. Analysis of a constant height hydraulic fracture driven by a power-law fluid. Rock Mech. Bull. 2022, 1, 100003. [Google Scholar] [CrossRef]
- Lang, X.M.; Yao, L.M.; Fan, S.S.; Li, G.; Wang, Y.H. Numerical simulation of methane hydrate formation and heat transfer in porous materials. CIESC J. 2022, 73, 3851–3860. [Google Scholar]
- Mirzakimov, U.Z.; Farhadian, A.; Semenov, M.E.; Pavelyev, R.S.; Heydari, A.; Chirkova, Y.F.; Valiullin, L.R. Enhanced methane storage capacity in clathrate hydrate induced by novel biosurfactants: Kinetics, stability, in vivo, and biodegradation investigations. J. Energy Storage 2023, 73, 108802. [Google Scholar] [CrossRef]
- Yan, S.; Huang, J.Y.; Rao, Y.C.; Wang, S.L.; Jia, R.; Liu, B.; Chen, F. Effects of graphene oxide and SDS on CO2 hydrate formation characteristics. J. Chang. Univ. 2020, 32, 68–73. [Google Scholar]
- Sadeh, E.; Farhadian, A.; Mohammadi, A.; Maddah, M.; Pourfath, M.; Yang, M. Energy-efficient storage of methane and carbon dioxide capture in the form of clathrate hydrates using a novel non-foaming surfactant: An experimental and computational investigation. Energy Convers. Manag. 2023, 293, 117475. [Google Scholar] [CrossRef]
- Qin, Y.; Bao, R.X.; Shang, L.Y.; Zhou, L.; Liu, Z.M. Growth and occurrence characteristics of methane hydrate in a complex system of silica sand and sodium dodecyl sulfate. Chem. Eng. Sci. 2021, 249, 117349. [Google Scholar] [CrossRef]
- Yang, L.; Li, C.; Pei, J.; Wang, X.; Liu, N.; Xie, Y.; Cui, G.; Liu, D. Enhanced clathrate hydrate phase change with open-cell copper foam for efficient methane storage. Chem. Eng. J. 2022, 440, 135912. [Google Scholar] [CrossRef]
- Khandelwal, H.; Fahed, Q.M.; Zheng, J.J.; Venkataraman, P.; Barckholtz, T.A.; Mhadeshwar, A.B.; Linga, P. Effect of L-Tryptophan in promoting the kinetics of carbon dioxide hydrate formation. Energy Fuels 2021, 35, 649–658. [Google Scholar] [CrossRef]
- Liu, X.J.; Ren, J.J.; Chen, D.Y.; Yin, Z.Y. Comparison of SDS and L-Methionine in promoting CO2 hydrate kinetics: Implication for hydrate-based CO2 storage. Chem. Eng. J. 2022, 438, 135504. [Google Scholar] [CrossRef]
- Yi, J.; Zhong, D.L.; Yan, J.; Lu, Y.Y. Impacts of the surfactant sulfonated lignin on hydrate based CO2 capture from a CO2/CH4 gas mixture. Energy 2019, 171, 61–68. [Google Scholar] [CrossRef]
- Kastanidis, P.; Tsimpanogiannis, I.N.; Romanos, G.E.; Stubos, A.K.; Economou, I.G. Recent advances in experimental measurements of mixed-gas three-phase hydrate equilibria for gas mixture separation and energy-related applications. J. Chem. Eng. Data 2019, 64, 4991–5016. [Google Scholar] [CrossRef]
- Fang, G.H.; Sun, P.B.; Yu, M.H.; Zhang, W.T.; Tang, X. Research progress and application about paraffin-nanoparticle composite phase-change materials. Mod. Chem. Ind. 2022, 42, 68–71. [Google Scholar]
- Dai, M.L.; Sun, Z.G.; Li, J.; Li, C.M.; Huang, H.F. Progress on promotion technology for gas storage in hydrates. Chem. Ind. Eng. Prog. 2020, 39, 3975–3986. [Google Scholar]
- Liu, G.Q.; Wang, F.; Luo, S.J.; Xu, D.Y.; Guo, R.B. Enhanced methane hydrate formation with SDS-coated Fe3O4 nanoparticles as promoters. J. Mol. Liq. 2016, 230, 315–321. [Google Scholar] [CrossRef]
- Ghozatloo, A.; Hosseini, M.; Shariaty-Niassar, M. Improvement and enhancement of natural gas hydrate formation process by Hummers’ graphen. Gas Sci. Eng. 2015, 27, 1229–1233. [Google Scholar] [CrossRef]
- Park, S.-S.; Lee, S.-B.; Kim, N.-J. Effect of multi-walled carbon nanotubes on methane hydrate formation. J. Ind. Eng. Chem. 2010, 16, 551–555. [Google Scholar] [CrossRef]
- Arjang, S.; Manteghian, M.; Mohammadi, A. Effect of synthesized silver nanoparticles in promoting methane hydrate formation at 4.7 MPa and 5.7 MPa. Chem. Eng. Res. Des. 2013, 91, 1050–1054. [Google Scholar] [CrossRef]
- Rahmati-Abkenar, M.; Manteghlan, M.; Pahlavazadeh, H. Experimental and theoretical investigation of methane hydrate induction time in the presence of triangular silver nanoparticles. Chem. Eng. Res. Des. 2017, 120, 325–332. [Google Scholar] [CrossRef]
- Rahmati-Abkenar, M.; Manteghlan, M.; Pahlavazadeh, H. Nucleation of ethane hydrate in water containing silver nanoparticles. Mater. Des. 2017, 126, 190–196. [Google Scholar] [CrossRef]
- Aliabadi, M.; Rasoolzadeh, A.; Esmaeilzadeh, F.; Alamdari, A. Experimental study of using CuO nanoparticles as a methane hydrate promoter. Gas Sci. Eng. 2015, 27, 1518. [Google Scholar] [CrossRef]
- Abdi-Khanghah, M.; Adelizadeh, M.; Naserzadeh, Z.; Barati, H. Methane hydrate formation in the presence of ZnO nanoparticle and SDS: Application to transportation and storage. J. Nat. Gas Sci. Eng. 2018, 54, 120. [Google Scholar] [CrossRef]
- Kakati, H.; Mandal, A.; Laik, S. Promoting effect of Al2O3/ZnO-based nanofluids stabilized by SDS surfactant on CH4+C2H6+C3H8 hydrate formation. J. Ind. Eng. Chem. 2016, 35, 357–368. [Google Scholar] [CrossRef]
- Wang, F.; Song, Y.M.; Liu, G.Q.; Guo, G.; Luo, S.J.; Guo, R.B. Rapid methane hydrate formation promoted by Ag & SDS-coated nanospheres for energy storage. Appl. Energy 2018, 213, 227–234. [Google Scholar]
- Chen, C.; Yuan, H.Y.; Wang, X.M.; Lin, Y.; He, Y.; Wang, F. Recyclable and high-efficiency methane hydrate formation promoter based on SDS-coated superparamagnetic nano-Fe3O4. Chem. Eng. J. 2022, 437 Pt 1, 135365. [Google Scholar] [CrossRef]
- Yan, H.C.; Song, X.F.; Xin, F. Storage capacity and duration of methane hydration in a slurry of solid n-Tetradecane. Energy Fuels 2014, 29, 130–136. [Google Scholar] [CrossRef]
- Song, X.F.; XIN, F.; YAN, H.C. Intensification and kinetics of methane hydrate formation under heat removal by phase change of n-tetradecane. AIChE J. 2015, 61, 3441–3450. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, K.; Liang, D.; Fan, S. Effects of magnetic fields on HCFC-141b refrigerant gas hydrate formation. Sci. China Ser. B Chem. 2003, 46, 407–415. [Google Scholar] [CrossRef]
- Sun, S.; Li, Y.; Gu, L.; Yang, Z.; Zhao, J. Experimental study on carbon dioxide hydrate formation in the presence of static magnetic field. J. Chem. Thermodyn. 2022, 170, 106764. [Google Scholar] [CrossRef]
- Yu, L.; Wang, H.Q.; Lu, W.; Sun, H. A lumped parameter model of the longitudinal NiMnGa transducer based on piezomagnetic equations. J. Acoust. Soc. Am. 2022, 152, 1416–1424. [Google Scholar]
- Fan, J.L.; Ai, Y.L.; Ou, Y.S.; Zhu, J.W. Research Progress of Chemical Composition Dependence on the Thermal and Mechanical-Magnetic Properties of NiMnGa-Based Alloys. Rare Metal Mat. Eng. 2021, 50, 4185–4192. [Google Scholar]
- Wu, Q.; Lin, N.Y.; Li, L.; Chen, F.; Xv, X.F.; Zhang, B.Y.; Wu, Q.; Liu, C.H. Synergistic enhancement of coalbed methane hydration process by magnetic field and NiMnGa micro-nano fluid. Front. Energy Res. 2022, 10, 974647. [Google Scholar] [CrossRef]
- Tian, B.; Chen, F.; Liu, Y.; Zheng, Y.F. Structural transition and atomic ordering of Ni49.8Mn28.5Ga21.7 ferromagnetic shape memory alloy powders prepared by ball milling. Mater. Lett. 2008, 62, 2851–2854. [Google Scholar] [CrossRef]
- Li, X.S.; Xu, C.G.; Chen, Z.Y.; Wu, H.J. Tetra-n-butyl ammonium bromide semi-clathrate hydrate process for post-combustion capture of carbon dioxide in the presence of dodecyl trimethyl ammonium chloride. Energy 2010, 35, 3902–3908. [Google Scholar] [CrossRef]
- Tian, B.; Chen, F.; Liu, Y.; Zhang, Y.F. Effect of ball milling and post-annealing on magnetic properties of Ni49.8Mn28.5Ga21.7 alloy powders. Intermetallics 2008, 16, 1279–1284. [Google Scholar] [CrossRef]
- Tian, B.; Chen, F.; Tong, Y.X. Phase transition of Ni–Mn–Ga alloy powders prepared by vibration ball milling. J. Alloys Compd. 2011, 509, 4563–4568. [Google Scholar] [CrossRef]
- Xu, X.F. Research on Phase Transformation and Magnetic Properties of Nicomnin Magnetic Refrigeration Alloy. Master’s Thesis, Harbin Engineering University, Harbin, China, 2020. [Google Scholar]
- Tian, B.; Chen, F.; Tong, Y.X. Bending properties of epoxy resin matrix composites filled with Ni-Mn-Ga ferromagnetic shape memory alloy powders. Mater. Lett. 2009, 63, 1729–1732. [Google Scholar] [CrossRef]
- Wu, Q.; Li, Z.; Chen, F.; Lin, N.Y.; Wang, X.Y.; Zhang, B.Y.; Li, L. Ferromagnetic shape memory alloy Ni52.5Mn22.5Ga25: Phase transformation at high pressure and effects on natural gas hydration. Mater. Lett. 2023, 343, 134371. [Google Scholar] [CrossRef]
- Pons, J.; Chernenko, V.; Santamarta, R.; Cesari, E. Crystal structure of martensitic phases in Ni-Mn-Ga shape memory alloys. Acta Mater. 2000, 48, 3027–3038. [Google Scholar] [CrossRef]



| Particle Conc. (wt.%) | Exp. No. | Induction Time (min) | Gas Consumption (mmol/mol) | Hydration Reaction Time (min) | Gas Consumption Rate (mmol/mol/min) |
|---|---|---|---|---|---|
| 0 | G1-1 | 113.9 | 9.7 | 185.6 | 0.054 |
| G2-1 | 48.3 | 16.5 | 155.9 | 0.113 | |
| G3-1 | 42.9 | 23.7 | 107.1 | 0.222 | |
| 0.1 | G1-2 | 3.4 | 11.9 | 92.2 | 0.135 |
| G2-2 | 8.9 | 16.6 | 76.4 | 0.221 | |
| G3-2 | 5.8 | 26.2 | 74.0 | 0.360 | |
| 1 | G1-3 | 4.0 | 13.2 | 70.9 | 0.196 |
| G2-3 | 2.4 | 21.6 | 82.1 | 0.264 | |
| G3-3 | 1.4 | 25.0 | 79.6 | 0.312 | |
| 2 | G1-4 | 5.3 | 10.8 | 48.0 | 0.229 |
| G2-4 | 2.6 | 21.4 | 59.0 | 0.363 | |
| G3-4 | 3.0 | 27.1 | 67.0 | 0.410 | |
| 3 | G1-5 | 1.9 | 14.6 | 59.9 | 0.244 |
| G2-5 | 1.3 | 19.0 | 57.2 | 0.335 | |
| G3-5 | 0.9 | 27.0 | 61.1 | 0.444 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Wu, Q.; Li, Z.; Meng, H.; Elhefnawey, M.; Wang, X.; Wu, Q.; Li, L.; Zhang, B. Effect of Different Concentrations of NiMnGa Micro/Nanoparticles on the Kinetics of Natural Gas Hydration. Processes 2023, 11, 3149. https://doi.org/10.3390/pr11113149
Zhao Z, Wu Q, Li Z, Meng H, Elhefnawey M, Wang X, Wu Q, Li L, Zhang B. Effect of Different Concentrations of NiMnGa Micro/Nanoparticles on the Kinetics of Natural Gas Hydration. Processes. 2023; 11(11):3149. https://doi.org/10.3390/pr11113149
Chicago/Turabian StyleZhao, Zhiwei, Qiong Wu, Zhen Li, Huiyuan Meng, Maged Elhefnawey, Xinyan Wang, Qiang Wu, Li Li, and Baoyong Zhang. 2023. "Effect of Different Concentrations of NiMnGa Micro/Nanoparticles on the Kinetics of Natural Gas Hydration" Processes 11, no. 11: 3149. https://doi.org/10.3390/pr11113149
APA StyleZhao, Z., Wu, Q., Li, Z., Meng, H., Elhefnawey, M., Wang, X., Wu, Q., Li, L., & Zhang, B. (2023). Effect of Different Concentrations of NiMnGa Micro/Nanoparticles on the Kinetics of Natural Gas Hydration. Processes, 11(11), 3149. https://doi.org/10.3390/pr11113149











