Abstract
It is well known that cheese yield and quality are affected by animal genetics, milk quality (chemical, physical, and microbiological), production technology, and the type of rennet and dairy cultures used in production. Major differences in the same type of cheese (i.e., hard cheese) are caused by the rennet and dairy cultures, which affect the ripening process. This review aims to explore current technological advancements in animal genetics, methods for the isolation and production of rennet and dairy cultures, along with possible applications of microencapsulation in rennet and dairy culture production, as well as the challenge posed to current dairy technologies by the preservation of biodiversity. Based on the reviewed scientific literature, it can be concluded that innovative approaches and the described techniques can significantly improve cheese production.
1. Introduction
Cheese production is a process dating back several thousands of years, and cheese, which is found in every part of the world, has found its place on the top of the foodstuffs chart thanks to its nutritional value and rich diversity. The earliest indication of a cheese-making process is in cave paintings around 5000 BC, representing the oldest technological application of enzymes [1]. This application has made possible the intentional conversion of milk into cheese, making it safer and longer-lasting.
Since its beginning, the technology of cheese production has changed, thanks to technological and scientific advancements in its materials and production process. Currently, cheese production is very high-level, thanks to modern materials and incorporated technology. Cheese quality is affected by the genotypic and phenotypic characteristics of animals, the chemical and microbiological properties of milk, and production technology [2,3,4]. An important factor in the diversity of milk from the same breed comes from dairy cultures and the type of rennet used in cheese production, because they affect chemical processes during production and ripening.
Currently, cheese quality is still affected by the same factors, but due to globalization and industrial advancements, originality and dissimilarities within certain regions have been lost. Large-scale cheese production is mainly based on the usage of pasteurized milk and cheesemakers are supplied by a few worldwide producers of dairy cultures and rennet. When considering the same type of cheese production (i.e., hard cheese), this means that differences between producers are based solely on the quality of milk and the selection of rennet and dairy cultures.
The characteristics of milk affecting cheese quality are regulated by animal genetics, which creates the possibility of using quantitative genetics to preserve biodiversity and indigenous traits. Raw milk and cheeses from raw, unpasteurized milk present rich sources of beneficial microbes, such as lactic acid bacteria (LAB) with probiotic properties [5]. The isolation of indigenous LAB cultures and their use in cheese production can result in the preservation of biodiversity and better diversification between cheese producers globally and regionally.
Microencapsulation is seen as a novel approach to the preservation of biodiversity and to delivering important ingredients into cheese, although it should be borne in mind that the successful encapsulation of payload relevant to cheese production, such as microorganisms, enzymes, peptides, aromatic compounds, chemical agents (Ca2+), and even essential oils, can be extremely challenging. Furthermore, combinations of more than one active ingredient can make the process of encapsulation even more complex [6,7,8,9].
Considering that little research on this topic has been undertaken, this paper aims to investigate current progress in DNA characterization and the use of quantitative genetics for improving desired traits in dairy animals, rennet production, and analysis, the isolation and production of lactic acid bacteria, and the possible applications of microencapsulation in the development of new, innovative, and sustainable technologies in cheese production, with an emphasis on indigenous forms and the preservation of biodiversity.
2. The Impact of Livestock Genetics on Milk as an Important Component in Cheese Production
The production of cheese is a complex process, whose quality and uniqueness depend on a variety of factors. The composition and characteristics of raw milk, which are largely determined by the genetics of the animals involved in its production, are certainly among the important factors that contribute to the uniqueness of cheese as a final product [10]. Therefore, the genetics of an individual or a specific population (breed) is one of the crucial elements for the successful production of indigenous dairy products, which are often commercially associated with the Protected Designation of Origin (PDO). At the same time, the influence of animal genetics on the quality and distinctiveness of cheese fits well with the concept of sustainable animal husbandry and the protection of the diversity of farm animals and dairy products.
2.1. Polygenic Inheritance
It was recognized very early that milk secretion is influenced by heredity [11]. Later, it was clearly defined that the composition and characteristics of raw milk are measured on a continuous scale and, like most economically relevant livestock production traits (phenotypes), are inherited as quantitative or complex genetic traits (for more detailed explanations, see [12]). For a long time, the inheritance of quantitative traits was successfully modeled by an infinitesimal model, in which an infinite number of loci, each with an infinitesimal effect (polygenic component), and the influence of the environment are responsible for measured (phenotypic) variations [13,14,15]. Therefore, in many cases, the inheritance of raw milk quantity and composition has been quantified by parameters such as heritability, more specifically heritability in the narrow sense (h2), i.e., the proportion of phenotypic variation explained by additive gene effects [12]. Milk, milk fat yield, and later, protein and lactose yield, had the longest tradition of measurement and were recognized as the most important factors in milk production for a century [16]. It is therefore not surprising that the quantitative inheritance of these traits is among the best-studied, while a large number of studies provide estimates of heritability and genetic correlations. In Table 1, we present a representative overview of the estimated h2 values for the most important traits related to milk production in cattle.
It is important to point out that the traditional traits of milk production (milk yield, fat yield, and protein yield) have been successfully incorporated into breeding programs because they fulfill the desirable characteristics of selection programs. According to Shook [17], successful breeding programs rely on preferred traits that should meet the following selection criteria: (1) they must have relatively high genetic variability and heritability, (2) they must have economic value that increases the profitability of production, (3) they must be positively correlated with other traits used in the breeding program, and, finally, (4) they must be clearly defined and measurable at low cost. Shortening the generation interval increases the annual genetic progress (response).

Table 1.
Heritability estimates for milk production traits in cattle.
Table 1.
Heritability estimates for milk production traits in cattle.
| Species | Trait | Heritability | Reference |
|---|---|---|---|
| Cattle | Milk yield | 0.34 | [18] |
| 0.09 | [19] | ||
| 0.10 | [20] | ||
| 0.07 | [21] | ||
| Milk fat (%) | 0.43 | [18] | |
| 0.11 | [20] | ||
| 0.39 | [19] | ||
| Protein (%) | 0.34 | [18] | |
| 0.29 | [20] | ||
| 0.30 | [19] | ||
| Casein (%) | 0.34 | [21] | |
| 0.35 | [20] | ||
| 0.35 | [19] | ||
Therefore, all these traits should be measured at an early life stage. Furthermore, the reliable estimation of heritability and genetic correlations requires a large sample size, which is sometimes difficult to ensure, even in experimental research analyses.
Some other traits, such as saturated and unsaturated fatty acid content, mineral content (Ca, P, Mg, K, Se, and Zn), milk protein fraction content (casein and serum proteins), and milk coagulation properties (RCT—rennet coagulation time, min; k20—the time needed for the curd to obtain firmness of 20 mm, min; a30—curd firmness at the end of analysis, mm) have been of interest to researchers for a long time [22,23,24,25,26,27], but their implementation in the selection process has been difficult because their routine measurements were expensive and time-consuming, which prevented their implementation in breeding programs. The estimates of heritability for some important traits (variables) related to cheese production and manufacture in cattle, sheep, and goats are presented in Table 2, while a more detailed description of the majority of genetic factors that have a major influence on coagulation traits in cattle is provided by Bittante et al. [27].

Table 2.
Heritability estimates for the milk production traits in cattle.
However, Etzion et al. [29] used mid-infrared spectrometry (MIR) to predict milk protein content. Their publication seems to have stimulated a large number of studies on the use of the same methodology for different milk constituents. For example, Soyeurt et al. [30] showed that fatty acid content can be efficiently and inexpensively predicted from MIR signals, soon followed by the use of MIR to estimate the coagulation properties of milk [31,32,33], the mineral content (i.e., Ca and P) [34], and the content of milk protein fractions [35,36]. More detailed information on the use of MIR in the estimation of different variables (chemical components) from milk is described by Bittante and Cecchinato [37] and De Marchi et al. [38]. In this way, MIR phenotyping has enabled the estimation of genetic parameters for many chemical components associated with cheese production.
As might be noted from Table 2, the heritability estimates for almost all cheese production traits are within the range that would allow sufficiently large and profitable selection progress.
2.2. Candidate Genes
The remarkable improvements of domestic animals achieved by artificial selection in the 20th century were based exclusively on the infinitesimal model [39,40], while the idea that genes with large effects or candidate genes could influence continuously varying traits has been challenged sporadically [41,42,43,44]. Thus, the main objective of a series of studies conducted from 1970 to the end of the 20th century was to find significant genotype–phenotype associations that could be functionally explained in so-called candidate gene association studies (CGAS). In the absence of efficient molecular genetic methods and possibilities to determine the genotype of each functional gene on a large scale (population), the major ruminant milk proteins (in ruminants, αs1-CN, β-CN, αs2-CN, and γ-CN represent four caseins, which together with β -LA and β -LG are responsible for coding 95% of the proteins in milk) were among the best-studied candidate genes, since their polymorphism was discovered quite early [45,46]. They were quite polymorphic [47,48,49,50], their genotyping (“phenotyping” as it was based on electrophoretic migration of proteins) was simple and inexpensive [51], while their causal relationship with milk production and technological traits was biologically expected. A large number of significant associations in milk production traits and milk protein polymorphisms were found in different breeds of cattle [52,53,54,55,56,57], sheep [58,59,60,61,62], and goats [62].
We should mention that four casein genes are tightly linked in a physical map in the order CSNAS1–CSNB–CSNS2–CSN3 within a 250 kb region on chromosome 6 in cattle [63,64], goats [63], and sheep [65], so it is impossible to completely separate the effects of each casein gene. For example, Meier et al. [64] analyzed whole-genome sequences in 14 cattle breeds (1821 animals) and found 37 different haplotypes. Haplotype B-A2-A-A (CSNAS1–CSNB–CSNS2–CSN3) was predominant in Angus (0.64), Charolais (0.33), Flechvieh (0.42), Gelbvieh (0.49), Holstein (0.53), Herford (0.36), Limousin (0.42), and Simmental (0.45) cattle breeds, while other haplotypes were predominant in Brown Swiss (B–A2–A–B = 0.50), Danish Red (B–A1–A–A = 0.44), DNS (B–A1–A–A = 0.72), Jersey (C–A2–A–B = 0.51), Normande (C–A2–A–B = 0.28), and Montebéliarde (B–A2–D–B = 0.36).
Therefore, it is quite reasonable to estimate the effects of segregating casein haplotypes (a combination of alleles at different loci on the same chromosome of an organism that are inherited together from one parent), as has been achieved with cattle [66,67], sheep [61], and goats [68,69]. For more details on the association of milk protein polymorphism with milk production and/or technological traits in cattle, sheep, and goats, see Buchberger and Dovč [70], Martin et al. [71], Caroli et al. [49], Bittante et al. [27], and Selvaggi et al. [50].
Using various sources of information, Ogorevc et al. [72] identified a large number of other potential candidate genes, some of which were identified in association studies that were likely to be associated with milk production traits. The most promising candidate genes are the following: ATP-binding cassette sub-family G—ABCG2 [73,74], Diacylglycerol acyltransferase—DGAT1 [75,76,77,78], growth hormone receptor—GRH [77], leptin—LEP [77,78,79], peroxisome proliferator-activated receptor-γ coactivator-1—PPRGC1 [80,81], prolactin—PRL [82,83,84], signal transducer and activator of transcription 5A—STAT5A [85], and insulin-like growth factor 1 -IGF-1 [86]. For more information on candidate genes with major effects on milk production, see Ibeagha-Awemu et al. [87] and Strucken et al. [88].
2.3. Genome-Wise Analyses and Genomic Selection
Recent developments in molecular genetics have enabled the genotyping of a large number of markers distributed across the genome, which has greatly improved our understanding of variation in complex traits [89,90,91]. Thus, based on new evidence from genome-wide association (GWA) studies, we have revised the concept of infinitesimal inheritance to a mixed inheritance model, in which variability in complex traits is caused by a polygenic component (many genes with small effects), as well as the existence of a set of candidate genes with medium-to-large effects that rarely explain more than 5% of phenotypic variation in humans [92,93,94].
In human populations, genes with moderate-to-large effects occur at low frequencies as rare or “private” mutations [95], whereas the occurrence of mutations with moderate-to-large effects at moderate or high frequencies has been documented more in domestic animal populations [95,96,97]. This difference is thought to be the result of artificial selection pressure and specific breeding goals. For good examples of candidate genes with strong effects on complex traits that segregate at moderate-to-high frequencies, see Kemper and Goddard [95] and Curik et al. [97].
Overall, the invention of the concept of genomic selection [98] and the availability of high-throughput genotyping data at low cost have revolutionized animal breeding in the last two decades. As a result, numerous GWA studies have been conducted on milk production [99,100,101,102,103,104,105] and technological traits [106,107] in cattle, sheep, and goat populations, pointing to a number of influential causative genes (mutations) and/or statistically associated markers. While some of these analyses have confirmed the results of the association studies, we have also been able to identify genes that have illuminated our biological understanding of milk as a raw material for good cheese production.
In general, genomic selection significantly accelerates annual genetic gain by shortening the generational interval and allowing the more efficient and rapid achievement of breeding goals [108,109,110,111].
2.4. Future Perspectives and Sustainable Development
Over the past two decades, our current technological knowledge of breeding for more specific milk composition and coagulation characteristics has improved considerably. This is primarily due to developments in and the availability of large-scale high-throughput genotyping [109,110,112], the phenotyping of new traits—previously measured at very high cost [113,114], the utilization of genomics in animal breeding [110,115], and our ability to analyze very large data sets [113,116]. Therefore, efficient breeding for more specialized dairy products is a very realistic option. At the same time, we are more aware of the potential for the adaptation of some landscape breeds to harsh environmental conditions and the potential negative consequences that could result from current climate changes [117,118]. However, on a broader scale, we are still not bold enough to radically shift our breeding objectives focused on milk quantity and basic milk composition (fat and protein) to more specialized milk composition and dairy products (cheese). Fortunately, the new options available to us are recent and offer many opportunities and challenges to make more sustainable choices in the future.
3. Isolation and Techniques of Rennet Production
3.1. Nomenclature and Function of Rennet
Peptidases are a large and important group of enzymes, many of which have been applied in technological processes involving food, beverages, feed, medicines, detergents, the production of chemicals, leather, paper, and textile processing. The active milk-clotting enzymes in all preparations that are successful in cheesemaking are aspartic proteinases (EC 3.4.23), which are so named because aspartic (Asp) residues are ligands of the activated water molecule within their active sites, which mediates nucleophilic attacks on scissile peptide bonds [119]. In the great majority of known aspartic peptidases, two Asp residues act together to bind and activate catalytic water molecules but in some, the residues of other amino acids replace the second Asp. One notable characteristic of aspartic peptidases is that all the enzymes described so far are endopeptidases. Endopeptidases cleave peptide bonds in the inner parts of their polypeptide chains, away from the N- and the C-terminus.
Peptidases are distinguished by the functional group in their active site: (A) for aspartate, (C) for cysteine, (G) for glutamic, (M) for metallo, (P) for mixed, (S) for serine, (T) for threonine, (N) for asparagine lyase, and (U) for as-yet unclassified peptidases of unknown catalytic type. Based on statistically significant similarities in their primary structures, peptidases are classified into families (approximately 268), which are identified by a letter representing their catalytic type. Further, some families may contain subfamilies. Some families are divided into subfamilies based on evidence of ancient divergence within the family (e.g., S1A, S1B). Families are then grouped into about 62 clans according to similarities in their 3D structures. Some clans are divided into subclans based on evidence of ancient divergences within the clan. The name of the clan consists of two letters. The first letter represents the catalytic type and the second letter is assigned sequentially as each clan is identified. This method of classification is used in the MEROPS database [120] for peptidases and the proteins that inhibit them [121,122].
In addition to this classification, depending on the functional group in the active peptidase site, peptidases are additionally classified according to the specificity of certain amino acids (“sequence specificity”), which form sensitive peptide bonds. For example, aspartate endopeptidases from the pepsin family hydrolyze peptide bonds with large hydrophobic amino acids in P1 or P1′ [121,123,124].
Aspartic peptidases are assigned to clans AA, AC, AD, AE, and AF [121]. They are a group of peptidases in the pepsin family (A1), with the same catalytic mechanism, and usually function in acidic solutions, which is why they are called acid peptidases. Aspartic peptidases have a long history and are found in animals, fungi, plants, protozoa, bacteria, and viruses. The feature of action in specific acidic environments limits their functions in living organisms and they are less abundant than other groups of peptidases, but because of its great physiological and commercial importance, this class of peptidases is unique [125]. Most aspartic proteases show maximal activity at low pH (pH 3 to 4) and have isoelectric points in the range of pH 3.0 to 4.5. Their molecular masses are between 30 to 45 kDa [126].
According to its classification in the MEROPS databanks, chymosin (EC 3.4.23.4) is a member of clan AA, family (pepsin) A1, subfamily A1A peptidase within the third class of hydrolases [121,122,127,128]. Deschamps performed the first isolation of this enzyme in 1840 and suggested the name chymosin (Gr. chyme, “gastric juice”) [126]. In 1890, Lea and Dickinson [129] suggested the name rennin (derived from rennet), but due to confusion with the related proteolytic enzyme, renin, from the kidneys, in 1970, Foltman suggested a return to the first name, chymosin, which was accepted by the International Union of Biochemistry and Molecular Biology (IUBMB) [130]. In 1872, Olof Hammarsten showed that rennet (chymosin) was synthesized and stored in an inactive form and activated by contact with stomach acid; this was the first thorough study on the effects of rennet on casein [131].
According to the MEROPS databanks, pepsin A is a member of clan AA, family (pepsin) A1, subfamily A1A peptidase within the third class of hydrolases [127,132]. Pepsin A (EC 3.4.23.1), known simply as “pepsin”, was discovered and recognized, in the 18th century, as the first enzyme that starts the digestion of food proteins in the stomach. Pepsin was originally named by Schwann in 1825. Pepsin is the predominant gastric peptidase in the fundus of adult mammals [121,131,132] and is exceptionally stable and active in acidic conditions. It is an endopeptidase characterized by lower specific activity, high general proteolytic activity, and a high pH dependency. Pepsin B (EC 3.4.23.2) is a minor proteinase found in porcine stomachs [1].
Plant aspartic peptidases are members of clans AA and AD. In clan AA, they are distributed among the families A1, A3, A11, and A12. The majority of plant aspartic peptidases, together with pepsin-like enzymes, belong to the A1 family [133].
Rennet (a mixture of chymosin and pepsin) is produced in the fourth stomach (the abomasum) of suckling ruminants (calves, lambs, kids/caprine, etc.). Chymosin is a neonatal peptidase that has a postnatal uptake of immunoglobulins [1] and can be found in fetuses from as early as the sixth month of gestation, with the biggest increase between the ninth month and the third or fourth day post-partum [134]. The reason for the secretion of chymosin in the stomach of newborn ruminants is the coagulation of milk to increase the nutritional value during retention in the intestine, allowing young animals to utilize more nutrients. Both peptidases (chymosin and pepsin) are secreted in their inactive form as zymogens (prochymosin and pepsinogen) into the channel with a direct connection to the lumen of the stomach, and they are activated by the low pH in the abomasum to chymosin and pepsin by the removal of the N-terminal pro-segment. The pro-segment is responsible for the stability of the inactive forms of zymogens and prevents the binding of the substrate into the active site. Chymosin is a polypeptide built from 323 amino acids, with a molecular weight of 35.6 kDa [119,135]. Generally, chymosin has low proteolytic activity but high milk-clotting activity. Calf chymosin has a narrow substrate specificity and cleaves the specific single-peptide bond in κ-casein between Phe 105 and Met 106, converting it into an insoluble form of curd with calcium para-κ-casein [136]. According to Harboe et al. [1], chymosin has been found to have high activity against milk of its own species. Calf chymosin is found in three allelic forms: A, B, and C. Chymosin B is the most abundant. The main difference between the A and B variants is that variant A has an aspartic acid (Asp) in position 244 and variant B has a glycine (Gly) in the same position. Variant A has 50% more proteolytic activity than the B form [126]. Variant C is genetically distinct and is a product of a different allele [137]. Thanks to its use in the cheese industry, calf chymosin is well investigated and characterized at enzymatic and molecular levels [133].
3.2. Types of Rennets and Most Commonly Used Coagulants in Cheese Production
The most important enzyme preparation in cheese production is rennet. It is a mixture of chymosin and pepsin, which is responsible for milk coagulation, but also proteolytic processes, during cheese ripening. Traditionally, rennet was produced by extracting the gastric juices of suckling mammals (calves, lambs, or kids) to obtain an animal rennet. Until the 19th century, the cheese was produced directly on farms and producers used homemade extracts from dried abomasa to coagulate the milk. Starting in the 1850s, cooperative dairies were established, which increased the demand for the production of larger amounts of rennet [126]. The industrial production of calf rennet (chymosin) dates back to 1874, in Denmark, when Christian Hansen started with the extraction of chymosin from calves’ abomasa for cheese production. Rennet was the first industrially produced enzyme preparation sold with standardized enzyme activity. The worldwide increase in cheese production during the 20th century led to a rennet shortage because the demand began to exceed the available supply around the 1950s and 1960s [138]. This problem was solved with the introduction of new types of milk-clotting agent, such as animal rennets (e.g., bovine pepsin) [139], microbial rennets (Rhizomucor miehei, Rhizomucor pussillus, Parasitica coagulans) [140], fermentation-produced chymosin (FPC) produced from Kluyveromyces marxianus var. lactis and Escherichia coli K-12 [141] or plant origins (Calotropis procera, Cynara cardunculus) [142,143].
Calf chymosin is one of the most frequently studied animal enzymes, and it was one of the first mammalian enzymes to have been successfully cloned, in the 19th century [144,145]. Because of its commercial importance, the gene for chymosin is the most commonly used and has been cloned and expressed at a higher level in various microbial hosts, such as: (1) bacteria Escherichia coli (Pfizer) [145,146,147], (2) mold Aspergillus niger (C. Hansen) [1], and (3) yeast Kluyveromyces marxianus var. lactis (Gist-brocades) [1,148]. Bovine chymosin is also successfully cloned in other microbial hosts, such as A. nidulans [149] and Pichia pastoris [150,151,152,153]. There is a significant volume of scientific literature about the production and biochemical properties of recombinant chymosin from other mammalian species: (1) camel [133,154,155,156,157], (2) lamb/ovine [158], (3) kid/caprine [155,159,160,161], (4) buffalo [155,162,163], (5) yak [164,165], and (6) alpaca [166].
According to Jacob et al. [167], of all coagulants or rennet substitutes, recombinant Bos Taurus chymosin is by far the most prominent genetically engineered clotting enzyme. The downside regarding FPC is its production through the fermentation of genetically modified organisms, which may be the reason why consumers prefer different products. Microbial rennets, on the other hand, are not products of GMO; they are easily produced in unlimited quantities, and their characteristics can be improved using biotechnology tools [168]. Since they are not produced from animal tissue (i.e., calf, lamb, kid) there is no possibility of transferring bovine spongiform encephalopathy (BSE) or scrapie [167]. Microbial coagulants, however, show higher proteolytic activity compared to rennet or FPC, which can cause lower cheese yield during production and the development of bitterness during the ripening process [167,168]. Plant or vegetable coagulants are rarely used for commercial production [133]. Ben Amira et al. [169] state that the use of vegetable coagulants is very limited for large-scale production because of their negative effect on cheese yield and the development of bitterness. Garcia et al. [170] reported that dry matter content, hardness, gumminess, and chewiness showed significant differences in cheeses made with vegetable coagulants and calf rennet, with higher values being obtained in cheeses made with vegetable coagulants. Manuelian et al. [171], in their study on mozzarella made from buffalo milk, reported slower coagulation after the addition of vegetable coagulants than with rennet. They concluded that longer coagulation times could be overcome by adding more coagulant, but further studies are needed to assess the sensory properties and textural characteristics of the product. Peptidases extracted from the cardoon flowers of various species of the genus Cynara have been used to make artisanal sheep cheeses in Portugal and Spain, especially Serra da Estrela cheese [143].
The original rennet preparation, commonly called animal rennet, is an extract from ruminant abomasum. According to this definition, it is agreed that the name “rennet” should be used for enzyme preparations from ruminant stomachs, and that other milk-clotting enzymes should be named “coagulants”. Rennets and coagulants are mostly categorized according to their source: (1) animal rennet and coagulants, (2) microbial coagulants, (3) fermentation-produced chymosin, and (4) vegetable coagulants. Chymosin produced by a genetically modified organism, as in, A. niger and K. lactis, is named fermentation-produced chymosin (FPC) [1,133,172]. According to the literature review, the known fermentation-produced chymosins (FPCs) from mammals are recombinant bovine chymosin [155], ovine prochymosin [158], caprine prochymosin [159,173,174], water buffalo chymosin [162], and camel chymosin [154].
Rennet, obtained from the gastric juices of suckling mammals (lambs, calves, kids) is produced mostly in liquid, solid, and paste form. Rennets in the form of pastes are traditionally used in countries in the Mediterranean region at the artisanal level and contain variable amounts of lipolytic activity with chymosin and pepsin [119,175,176]. Lamb and kid rennets are used in Greece for the production of Feta and Ketalotyri cheese from sheep and goats’ milk [175].
Due to its high content of chymosin, calf rennet is regarded as the ideal enzyme product for cheesemaking [1]. In the abomasum and extracts from its tissues, the proportion varies between chymosin and pepsin. The ratio between these two enzymes depends on the age and the feeding regime of the animal, as shown in Table 3 [126,177]. The most desirable proportion of chymosin to pepsin in preparation for cheese-making in regular calf rennets is 80:20 [119,126,177]. Generally, extracts from younger and milk-fed animals are higher in chymosin, while extracts from adult animals are higher in pepsin. In the world production of rennet, animals are slaughtered at different ages, and extracts feature different kinds of enzyme mixture.

Table 3.
The ratio between chymosin and pepsin in the bovine abomasal mucosa, depending on the age and feeding regime.
Although calf chymosin is the industrial gold standard for cheese manufacturing, chymosin from other animal sources (e.g., lamb, kid, buffalo, and camel) has also been studied as an alternative [119,133]. Several products of lamb/ovine and kid/caprine origin exist, but they are best suited to clotting milk of their species [1,178]. The most commonly used alternative to calf rennet is rennet of adult bovine origin. The most common commercially available recombinant chymosins are Chy-max, obtained by fermentation in E. coli K-12; Chymogen and ChymoStar, produced by Aspergillus niger var awamori; and Maxiren, generated in K. marxianus var lactis. A proteinase from R. miehei has been cloned and expressed in A. oryzae by Novo Nordisk A/S (Denmark) [1].
3.3. Rennet Production Methods
Procedures for industrial rennet production are very scarce and little is known about them. Although the procedures used by producers vary, the production of animal rennet involves the following commonly used steps: (1) production (abomasa preparation, extraction, activation), (2) the purification and determination of the chymosin: pepsin ratio, (3) standardization, and (4) quality control of the enzyme product (chemical and microbiological). In the past, animal rennet was produced from dried or fresh abomasa; however, currently, it is mainly produced from frozen abomasa [1]. The cleaned enzyme-containing tissues are preserved by salting, freezing, or drying in the slaughterhouse. The abomasa may be washed, dried, and salted before extraction. They are cut into small pieces and minced; the enzymes are then extracted, usually with a 5–10% NaCl and acid solution with or without preservatives, to produce single-strength rennet. The single-strength rennet is then concentrated using ultrafiltration to produce double- or triple-strength rennet [179]. Foltmann [180] used 1/3 M Al2(SO4) for the simultaneous clarification and activation of raw extract followed by the addition of 1 M Na2HPO4 to adjust pH to final 5.6. After the removal of tissue by filtration, NaCl is usually adjusted to 20% [126,181]. Anifantakis and Green [175] experimented by changing solutions four times on the same tissue in four days and reported that most enzymes are extracted in the first 24 h, and the rennet is most stable at a pH value of 5.5. The use of ultrasound resulted in an increase in activity and a yield of chymosin with a significantly shorter extraction time (80 min) [182,183,184]. The crude extract is then activated by lowering the pH to around 2.0 for a certain amount of time. Subsequently, the suspension is often clarified by another centrifugation, filtration, or purified by ion-exchange chromatography to obtain a product enriched with chymosin. The final product is then standardized for strength and enzyme composition (chymosin to pepsin), and subjected to quality control according to specifications. Commercial rennets are found on the market in liquid, powder, or tablet form.
3.4. Determination of Rennet Strength
The most important parameters of rennets are strength (enzyme activity), enzyme composition, identity, and purity. There are many methods to measure the strength (enzyme activity) of rennets, several of which have been inspired by Soxhlet and Berridge. In 1877, Von Soxhlet defined Soxhlet’s units (SU) as the volume of raw milk, which one volume of enzyme preparation can clot in 40 min at 35 °C. The strength is expressed as the clotting ratio, e.g., 1:10,000 (1 mL of rennet can clot 10,000 mL of raw milk in 40 min at 35 °C). The SU is easy for cheesemakers to understand, but it depends on both the pH and the milk quality, which causes variations because no reference standards are used; therefore, SU should be considered only as an indicative measurement of milk-clotting activity [1,185]. In 1952, an English scientist, N. J. Berridge [186], introduced standardized milk powder reconstituted in 0.1 M CaCl2 instead of raw milk. The Berridge, or Rennin unit (RU), was created and was widely used until the 1990s by the British Standard Institution (BSI). One RU is defined as the activity that can clot 10 mL of standardized milk in 100 s at 30 °C. The major drawbacks of RU units are that the pH value of the standardized substrate is 6.3, which is lower than in any milk used in cheesemaking (i.e., pH 6.4–6.6), and the very high calcium content of Berridge milk, which gives unreliable results because of calcium ions’ effects on cheese curd.
In general, bovine rennets contain both the main milk-clotting enzymes, chymosin and pepsin. Each of these two enzymes has its physicochemical characteristics, which influence the milk-clotting activity and cheese-making properties. The major difference between these two milk-clotting enzymes is in the stronger pH dependence and general proteolytic activity of the pepsin. Thus, for both economic and processing reasons, it is very important to determine the total milk-clotting activity of a certain rennet type in comparison with an internationally recognized reference standard of known composition and milk-clotting activity. The current standard method for bovine rennets and measures of milk-clotting activity is IDF Standard 157:2007/ISO 11815, developed in 1997 by the International Dairy Federation (IDF), known as the “relative milk-clotting activity test” or REMCAT [1,185,187]. The IDF has developed methods for the determination of the milk-clotting activity of microbial enzymes [188] and ovine and caprine rennets [189]. The strength of the enzyme activity i.e., the milk-clotting activity of rennets, is expressed in international milk-clotting units per gram or milliliter (IMCU/g or IMCU/mL). The Berridge clotting-time method is used in all the IDF Standards for the determination of the total milk-clotting activity as the indicator of milk flocculation appearance [185]. The internationally recognized reference standards for the known composition and milk-clotting activity for calf rennet reference standard powder (containing >98% chymosin and <2% bovine pepsin) and adult bovine rennet reference standard powder (containing <2% chymosin and >98% bovine pepsin) were produced by the Association of Manufacturers of Animal-Derived Food Enzymes (AMAFE), appointed by the IDF. Their milk-clotting activity was adjusted to be the same on a standardized milk substrate (0.05 g CaCl2 per 100 mL) at pH 6.5 and 32 °C; it is set at 1000 IMCU/g [126,190].
4. Isolation, Determination, and Application of Indigenous Dairy Lactic Acid Bacteria
4.1. Indigenous Lactic Acid Bacteria (LAB) of Raw Milk and Cheese
The composition of raw milk microbiota is very complex and related to different intrinsic and extrinsic factors, such as udder health, general farm management, milking procedures, hygiene conditions, milk storage on farms, etc. From the public health point of view, the microbiological safety of milk should be always discussed through a tight connection with animal health and environmental hygiene. Our recent findings showed that raw milk from vending machines may be contaminated, although rarely, with foodborne pathogens, such as L. monocytogenes [191,192], or multi-resistant Staphylococcus aureus [193].
The LAB number in raw milk is very variable and likely related to the phase of the milk chain where and when it is collected. For instance, the LAB population in raw cows’ milk at farm level range from 2.0 to 3.6 log CFU/mL [194], while at retail level (vending machines) it may exceed 5 log CFU/mL [192] due to the specific conditions of milk cooling that dictate the growth of psychotropic bacteria during storage, including LAB population. Concerning cheese production, LAB population counts are related to cheese type and storage conditions [195]. Therefore, in soft cheeses, LAB increases from 4 to 7 log CFU/g within a few days at cold storage, while during the ripening and storage of hard cheeses for 1 year, a reduction from 7 to 2 log CFU/g occurs [195].
Within the LAB group, enterococci are considered highly controversial bacteria, and usually referred to as being at the limits of food safety [196]. Enterococci are not necessarily indicators of faecal contamination, as they are ubiquitous bacteria. For example, multi-resistant enterococci strains were isolated from raw milk collected from the udders of healthy cows, which had never been treated with antibiotics but were kept with treated animals [197]. Currently, enterococci are considered beneficial microbes with probiotic properties, while, on the other hand, they are potentially virulent bacteria, producers of biogenic amines, or carriers of resistance genes [198]. Therefore, enterococci are bacteria with a dual role in dairy production, and their most frequently isolated species are E. faecalis and E. faecium [199].
Enterococci colonize raw materials, including milk, through the digestive system or contaminated environments, where they can survive and multiply during the fermentation process [200]. As fermentative bacteria, enterococci can be crucial for the development of specific sensorial properties in cheese; however, their resistance to high temperatures, salt, and low pH may lead to their excessive growth and cause spoilage [201].
Enterococci also demonstrate strong antimicrobial (antilisterial) activity by producing enterocins [202]. Many dairy-related enterococcal strains are characterized and intended for experimental applications as functional cultures in fermented food production [203]. Raw-milk-originated Enterococcus faecalis EF-101 was recently characterized as a novel bacteriocin-producer with inhibitory properties against the majority of food-related Listeria strains in vitro [204].
4.2. Determination of Lactic Acid Bacteria in Milk and Cheese
The determination of milk microbiota is often very complex and the reported results are very diverse, depending strongly on the methodology used [205]. Different methods that are used for its identification and detection can be culture-dependent and culture-independent, as shown in Table 4.

Table 4.
The methods for the detection and analysis of microbiota.
Traditional microbiological methods of isolation and identification are costly and time-consuming, with uncertain results for bacterial identification at the species level due to high biochemical (enzymatic) variability, especially in lactic acid bacteria, including enterococci [194]. The results of several studies dealing with LAB in raw milk and cheeses in Croatia are summarized in Table 5. The diversity of non-starter LAB (NSLAB) in autochthonous dairy products from Western Balkan Countries has been recently reviewed by Terzić-Vidojević et al. [209].

Table 5.
Results of determination of indigenous microbiota lactic acid bacteria (LAB) of different types of milk and cheese in Croatia.
4.3. Selection of Indigenous LAB for Dairy Cultures
In general, food safety and quality depend on many specific factors, including favorable or harmful microbial properties. The upgrading of cheese safety and quality may be achieved by applying competitive starter cultures or their metabolites, which are capable of preventing cheese spoilage and extending shelf-life through microbial interactions, reducing or eliminating potential hazards (i.e., pathogens, spoilers, biogenic amine producers, mycotoxins producers, resistant bacteria), and exerting beneficial effects on consumers’ health through their probiotic properties [217,218]. LAB strains with probiotic properties have been isolated from different dairy-related niches, including cows’ or ewes’ raw-milk cheeses, and whey. Hence, many studies are focused on implementing probiotic strains in fermented milk beverages or cheeses to produce novel products with enhanced health benefits [5].
The most adequate strains for starter cultures are usually selected from autochthonous microbiota, since they are well adapted to the food environment and the specific manufacturing process [214]. The selection procedure for potential starters should involve all potentially “risky” criteria, such as toxigenicity, acquired transmissible antimicrobial resistance, or technologically unacceptable pathways [219]. The general criteria for the selection of starter cultures include safety, technological properties, and economic aspects [214]. Some of the specific criteria for their selection include: (1) the quick and adequate production of lactic acid, the production of L (+)—lactic acid; (2) rapid growth at different temperatures, salt concentrations, and pH; (3) homofermentative metabolism; (4) catalase activity and the hydrolysis of hydrogen peroxide; (5) proteolytic and lipolytic enzyme activity; (6) tolerance or synergism towards other microbial components of starter culture; (7) production of antimicrobial compounds; (8) antagonism towards pathogenic and technologically undesirable microorganisms; (9) lack of antimicrobial resistance, biogenic amines, and mucus production; and finally (10) probiotic properties (tolerance to low pH, tolerance to bile salts, adhesion to human intestinal cells) [214,220,221]. The sensory appearance of traditional cheeses can be achieved and preserved by using selected autochthonous strains as starter cultures in thermally treated milk. Another possible technique in cheese-making is the introduction of starter cultures in the form of whey collected the day before the cheese is made. In addition to quality characteristics, autochthonous microbiota in the form of protective cultures, usually added as adjunct cultures, can affect the hygienic and safety characteristics of cheese. Protective cultures do not alter the sensory properties of cheese, but, rather, the cheese safety by suppressing the pathogenic microbiota. In this sense, their antimicrobial properties in cheese production are directed to toxigenic molds, amine-producing microbiota, or foodborne pathogens, such as Listeria monocytogenes [203,215,222].
4.4. Application of Microencapsulated Bacteria Strains in Cheese Production
Despite their wide application in the pharmaceutical industry, microencapsulated products are still a rarity in the food industry. It is a well-established fact that the viability of probiotic strains in food products is often restricted by many factors. For this purpose, some alternative methods were established to protect microorganisms against undesirable environmental conditions. One of the most promising techniques for bacterial protection is microencapsulation [223]. Gomes da Cruz et al. [224] suggest it as a possible solution to losses in viability due to salting or extended storage.
Microencapsulation is defined as a process of coating small particles of solids, liquids, or gaseous components, with a protective coating material [225], thus providing numerous benefits to encapsulated materials. One of its main advantages is the controlled release mechanism in which the active ingredients are released at controlled rates over prolonged periods [226]. This improves the stability of starter and/or adjunct cultures with a reduction in ripening time. There are many extensive reviews on the microencapsulation techniques used in the food industry, mostly based on the different encapsulating bioactive ingredients and methods that are used. Spray-coating and gel-particle technologies are most often used for the microencapsulation of probiotics [227]. Different types of encapsulating material have been used to trap probiotic bacteria, but the most effective is alginate [228].
Goderska et al. [229] demonstrated that Lactobacillus rhamnosus microencapsulated in alginate matrix kept their viability up to 48 h, unlike free cells that were inactivated completely under the same conditions. Similarly, Abd-Elhamid [230] proved that microencapsulation significantly increased the survival of Bifidobacterium adolescentis in Kariesh cheese during cold storage. However, by contrast, Godward and Kailasapathy [231] claimed that the microencapsulation of probiotic cells in Feta cheese caused a higher microbial loss, probably by inhibiting the disposal of cell metabolites that may have accumulated inside the encapsulated capsule. In addition to the increased microbial viability, microencapsulation can also affect the chemical and physical properties of foods containing microencapsulated bacteria. In a survey conducted by Ozer et al. [232], the authors noted that the development of proteolysis was more pronounced in cheeses containing probiotic bacteria in the encapsulated form.
Despite the aforementioned facts, microencapsulation technology is yet to become an everyday tool in the development of functional and sustainable food, which can only be achieved by comprehensive research.
5. Microencapsulation
Encapsulation in Cheese Production
The encapsulation process involves the entrapment of active substances into another substance wall material that produces particles on various scales [233,234,235]. In general, there are two main forms and structures (morphology) of encapsulation system: matrix type (spheres) and core-shell type (capsules) (Figure 1) [236]. The substance that is encapsulated is usually labeled as the core, fill, active, internal, or payload phase, while the wall material used for the encapsulation is labeled as coating membrane, shell, capsule, carrier material, external phase, or matrix [233,237,238].
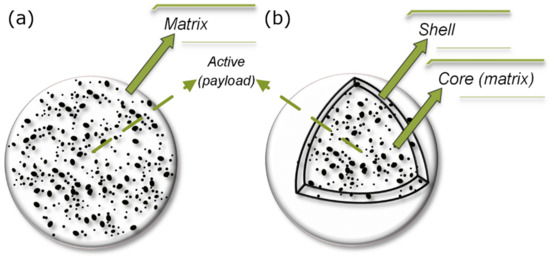
Figure 1.
Schematic representation of main forms for active substance encapsulation: (a) matrix type (sphere) and (b) core-shell type (capsule).
Encapsulation technology can significantly contribute to the cheese production process, and it can protect encapsulated ingredients within an effective barrier against environmental factors such as oxygen, light, free radicals, etc. and enable their controlled delivery [239,240,241]. Due to the increasing popularity of the use of natural materials, encapsulation in biodegradable polymers is becoming versatile. Biopolymers are relatively easy to obtain from natural sources and they may also be prepared with the use of microorganisms or synthesized with precision and with predetermined properties [242]. Numerous techniques can be used for encapsulation in biopolymeric matrices, but the ionic gelation method is often utilized in production, mainly because it uses mild conditions throughout the encapsulation process [243,244,245]. Encapsulation in biopolymeric microparticles is continuously improving and mostly advancing in the direction of the improvement of physicochemical, functional, and release properties while keeping in mind cost-effectiveness and the use of environmentally friendly and “green” material throughout the process [246].
The encapsulation process depends on the type of biopolymers used, since they vary in their composition and physicochemical properties. To achieve the successful encapsulation of active ingredients, the proper choice and understanding of biopolymer structure are necessary [247]. The encapsulation process and its effectiveness generally depend on the material that is to be encapsulated (payload), the encapsulation matrix material, and the encapsulation method/process. The proper selection of the encapsulation conditions may result in the desirable functionality and properties of the systems encapsulating the core [248].
Before proceeding with the encapsulation process, some issues that may arise should be addressed. The development of microparticles with more than one active ingredient starts with the determination of the conditions of preparation. It is important to observe the influence of each component used in the process of encapsulation. For example, the nature of microorganisms is to interact with their environment; thus, it is important to determine molecular interactions with other components before the encapsulation process [247]. Encapsulation in biopolymeric matrices is suitable for the formulation of particles loaded with more than one ingredient, such as lactic acid bacteria, enzymes, and chemical agents (relevant to the cheese production process, i.e., Ca2+) [249,250]. What makes this challenging is the simultaneous encapsulation of more than one ingredient. These types of particle can be created so that the inner core contains bacterial culture, while the outer shell contains enzymes. Depending on the fundamental desirability, the active ingredient in the outer shell releases faster, while the core material releases at a significantly slower speed. Remarkably, research concerning the development and implementation of these types of microparticle in the cheese production process is non-existent. The proper optimization of particles simultaneously loaded with multiple ingredients may result in a product that can be easily implemented in cheese production, making the entire process simpler from the technological point of view.
Throughout cheese production, there are processes, such as cheese ripening, that are not fully controllable. This is not only time-consuming but also a very complex process that involves the enzymatic breakdown of the protein, carbohydrate, and lipid contents of curd [251,252]. The use of encapsulation technology enables the acceleration of cheese ripening via encapsulated flavourzyme [253]. This is important because the conventional addition of the “free” enzyme is generally not recommended due to the negative influence on the flavor and texture [254,255,256]. Furthermore, the conveniently encapsulated enzyme can be physically separated from the curd mixture during cheese-making and may be released into the matrix during ripening [257]. This is only one example that shows how encapsulation technology can be implemented in the process of cheese production. In addition to accelerating cheese ripening, encapsulation technology is mainly used to fortify cheese with bioactive compounds or to increase the shelf-life of cheese products [258]. In Table 6, we present recent examples of the employment of the encapsulation method that may aid in the process of cheese production, increasing its shelf-life and adding value.
It is well known that, if present in an appropriate amount of food, probiotics can withstand hostile conditions in the gastrointestinal tract, adhere to cells, and promote positive effects [259]. The sustainability of probiotic microorganisms in any food depends on not only the storage of food but on the food matrix itself, as well as its components (fat, protein, moisture content). However, more importantly, probiotic bacteria must be stable during food processing and storage conditions. However, microorganisms, such as lactic acid bacteria, show low survivability in food products, as well as during food production, while their encapsulation leads to their preservation [260]. For example, Afzaal et al. [261] encapsulated probiotic bacteria, which resulted in a significantly higher survival rate with improved stability and protection during cheese production. Furthermore, Ningtyas et al. [262] encapsulated probiotics with the addition of β-glucan and phytosterol emulsion, which maintained viable cell count throughout the storage of cream cheese. The encapsulation of probiotic bacteria can be useful for the cheese production process and, when considering its survival and protection, there is the possibility of including natural additives. This may further improve bacterial survivability and even enhance its abilities. With this in mind, the proper selection of natural encapsulation material, with an economically viable and mild production process and the selection of compatible co-encapsulants, is of crucial importance. When considering the implementation of “unconventional” technologies in conventional cheese production processes, encapsulation in biopolymeric microparticles offers numerous benefits. However, some sensory issues may still arise. For example, microparticles are generally in the 1–1000 µm size range [263], and this may influence consumers’ appreciation of the product. When considering the application of microparticles into real food products, i.e., cheese, in addition to the enhancement of encapsulated components’ properties, protection, and sustained/controlled release, microparticles may offer benefits in terms of sensory characteristics. According to some reports, consumers showed a preference for products with particle sizes in the range of 250–500 μm, and this was associated with attractive qualities such as crispiness and granularity, which were not perceived in lower size ranges [264,265,266,267]. Furthermore, there are some reports in which sizes from 620–980 μm had no negative influence on the sensory acceptability of the real food product [268]. With this in mind, in addition to the numerous benefits that may be achieved via the microencapsulation of ingredients relevant to cheese production, additional desirable sensory characteristics can be obtained for cheese products.
Modern encapsulation science moves forward because it takes into account materials that can add desired functionality to particles, making them more than just carriers. Even though encapsulation science has helped to produce highly sophisticated systems, the gold standard for the encapsulation of microorganisms remains calcium alginate particles. Simultaneous encapsulation (more than one “active” ingredient) enhances its efficiency and may even have a synergistic effect. It is expected that soon, microencapsulated formulations with multiple ingredients will not only have a smooth introduction into the conventional production of cheese, but into food production in general [247,249].

Table 6.
Most recent examples of the encapsulation of various active agents (functional components) to aid in cheese production and increase its bioactive value and shelf-life.
Table 6.
Most recent examples of the encapsulation of various active agents (functional components) to aid in cheese production and increase its bioactive value and shelf-life.
| Encapsulated Agents | Fabrication Method/Carrier | Particle Size | Results/Outcomes | Reference |
|---|---|---|---|---|
| Bifidobacterium bifidum ATTC-29521 |
| n.s. * |
| [261] |
| nisin from Lactococcus lactis (n.s. *) |
| 120 nm |
| [269] |
| Lactobacillus rhamnosus (n.s. *) |
| n.s. * |
| [262] |
| mandarin peel extract |
| 250–450 nm |
| [270] |
| Lactobacillus paracasei LAFTI® L26 |
| n.s. * |
| [271] |
| oregano essential oil (Origanum vulgare L.) |
| n.s. * |
| [272] |
| Lactobacillus acidophilus Chr. Hansen A/S (Denmark) |
| 60–500 μm |
| [260] |
| aromatic compounds- 3-methylbutanal (from Carnobacterium maltaromaticum LMA 28) |
| 1.69 ± 0.15 mm |
| [273] |
| Lactobacillus rhamnosus 6134 |
| n.s. * |
| [274] |
| flavourzyme® |
| 180 nm |
| [253] |
| catechin and epigallocatechin gallate |
| 133 nm |
| [275] |
n.s. *—not specified.
6. Conclusions and Prospects
Recent developments in and the availability of the genotyping and phenotyping of desired traits combined with advancements in the statistical analysis of large data sets offer a good foundation for specialized dairy products studies (i.e., cheese). A new approach with the use of genetics in the production of rennet and dairy cultures should be used to challenge conventional production.
The production of animal rennets has been thoroughly researched in the 20th century. Because of the constant growth in cheese production and the shortage of this type of rennet, producers have turned to other coagulants, which can be produced in desired quantities. According to the literature, rennet has the best influence on the milk of the same species, but only calf rennet and bovine substitutes are produced. The production and use of rennet produced for a specific type of cheese or region are currently possible, and we suggest that more studies on this topic are needed.
The isolation and determination of indigenous lactic acid bacteria for use in dairy cultures are possible, and their use in a given cheese or region should maximize desirable characteristics.
Numerous benefits may be achieved with the microencapsulation of ingredients relevant to cheese production, including additional desirable sensory characteristics. Modern encapsulation techniques can be used to add the desired functionality to the product. Despite high technological advancements, the gold standard for the encapsulation of microorganisms remains calcium alginate particles. It is expected that microencapsulated formulations with multiple ingredients (simultaneous encapsulation) might have a synergistic effect, along with the enhancement of efficiency.
Technological and scientific advancements in animal science, chemistry, microbiology, and dairy science have enabled the presentation of the described topics in a single review to stimulate the application of sustainable and innovative technologies in cheese production. New options that are currently available should be used to develop sustainable production systems, with an emphasis on the preservation of biodiversity and the introduction of new technologies into conventional cheese production. This approach could enable producers to create new dairy products, emphasize differences between regions, or highlight the different ways in which they produce the same product with the preservation of biodiversity on a broader scale.
Author Contributions
Genetics, V.C.-C. and I.C.; isolation and techniques of rennet production, F.O., N.A., J.Š. and N.M.; isolation, determination, and application of indigenous dairy lactic acid bacteria, S.K., M.K. and N.Z.; microencapsulation, S.J. and M.V. All authors have read and agreed to the published version of the manuscript.
Funding
The funding was provided by the European Union (EU), Operational Program Competitiveness and Cohesion 2014–2020. Investment in science and innovations—first call, KK.01.1.1.04.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The research was performed as part of the project: “Potential of microencapsulation in cheese production”, KK.01.1.1.04.0058. This research was also supported by the project from Croatian Science Foundation: ANAGRAMS-IP-2018-01-8708.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Harboe, M.K.; Broe, M.L.; Qvist, K.B. The Production, Action and Application of Rennet and Coagulants. In Technology of Cheesemaking, 2nd ed.; Law, B.A., Tamime, A.Y., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 98–129. [Google Scholar]
- Fox, P.F.; Guinee, T.P.; Cogan, T.M.; McSweeney, P.L.H. Fundamentals of Cheese Science, 3rd ed.; Springer US: Boston, MA, USA, 2017. [Google Scholar] [CrossRef]
- Cipolat-Gotet, C.; Cecchinato, A.; Drake, M.A.; Marangon, A.; Martin, B.; Bittante, G. From cow to cheese: Novel phenotypes related to the sensory profile of model cheeses from individual cows. J. Dairy Sci. 2018, 101, 5865–5877. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, M.; Cecchinato, A.; Biasioli, F.; Gasperi, F.; Martin, B.; Bittante, G. From cow to cheese: Genetic parameters of the flavour fingerprint of cheese investigated by direct-injection mass spectrometry (PTR-ToF-MS). Genet. Sel. Evol. 2016, 48, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.S.; Ray, R.C.; Zdolec, N. Lactobacillus plantarum with Functional Properties: An Approach to Increase Safety and Shelf-Life of Fermented Foods. Biomed Res. Int. 2018, 2018, 9361614. [Google Scholar] [CrossRef] [PubMed]
- Vinceković, M.; Jurić, S.; Đermić, E.; Topolovec-Pintarić, S. Kinetics and Mechanisms of Chemical and Biological Agents Release from Biopolymeric Microcapsules. J. Agric. Food Chem. 2017, 65, 9608–9617. [Google Scholar] [CrossRef] [PubMed]
- Jurić, S.; Đermić, E.; Topolovec-Pintarić, S.; Bedek, M.; Vinceković, M. Physicochemical properties and release characteristics of calcium alginate microspheres loaded with Trichoderma viride spores. J. Integr. Agric. 2019, 18, 2534–2548. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Jambrak, A.R.; Vinceković, M. Tailoring alginate/chitosan microparticles loaded with chemical and biological agents for agricultural application and production of value-added foods. Appl. Sci. 2021, 11, 4061. [Google Scholar] [CrossRef]
- Jurić, S.; Šegota, S.; Vinceković, M. Influence of surface morphology and structure of alginate microparticles on the bioactive agents release behavior. Carbohydr. Polym. 2019, 218, 234–242. [Google Scholar] [CrossRef]
- Priyashantha, H.; Lundh, Å. Graduate Student Literature Review: Current understanding of the influence of on-farm factors on bovine raw milk and its suitability for cheesemaking. J. Dairy Sci. 2021, 104, 12173–12183. [Google Scholar] [CrossRef]
- Gowen, J.W. Genetics of Breeding Better Dairy Stock. J. Dairy Sci. 1926, 9, 153–170. [Google Scholar] [CrossRef]
- Falconer, D.S. Introduction to Quantitative Genetics, 4th ed.; Prentice Hall: Harlow, UK, 1996. [Google Scholar]
- Fisher, R.A. The correlation between relatives on the supposition of Mendelian inheritance. Trans. R. Soc. Edin-Burgh 1918, 52, 399–433. [Google Scholar] [CrossRef]
- Bulmer, M.G. The Effect of Selection on Genetic Variability. Am. Nat. 1971, 105, 201–211. [Google Scholar] [CrossRef]
- Bulmer, M.G. The Mathematical Theory of Quantitative Genetics; Oxford University Press: Oxford, UK, 1980. [Google Scholar]
- Miglior, F.; Fleming, A.; Malchiodi, F.; Brito, L.F.; Martin, P.; Baes, C.F. A 100-Year Review: Identification and genetic selection of economically important traits in dairy cattle. J. Dairy Sci. 2017, 100, 10251–10271. [Google Scholar] [CrossRef] [PubMed]
- Shook, G.E. Selection for Disease Resistance. J. Dairy Sci. 1989, 72, 1349–1362. [Google Scholar] [CrossRef]
- Ikonen, T.; Ahlfors, K.; Kempe, R.; Ojala, M.; Ruottinen, O. Genetic Parameters for the Milk Coagulation Properties and Prevalence of Noncoagulating Milk in Finnish Dairy Cows. J. Dairy Sci. 1999, 82, 205–214. [Google Scholar] [CrossRef]
- Cassandro, M.; Comin, A.; Ojala, M.; Dal Zotto, R.; De Marchi, M.; Gallo, L.; Carnier, P.; Bittante, G. Genetic Parameters of Milk Coagulation Properties and Their Relationships with Milk Yield and Quality Traits in Italian Holstein Cows. J. Dairy Sci. 2008, 91, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Cecchinato, A.; Penasa, M.; De Marchi, M.; Cipolat-Gotet, C.; Bazzoli, I.; Cologna, N.; Bittante, G. Methods for the assessment of milk coagulation properties: A genetic analysis. J. Dairy Sci. 2011, 89, 705. [Google Scholar]
- Penasa, M.; Cassandro, M.; Pretto, D.; De Marchi, M.; Comin, A.; Chessa, S.; Dal Zotto, R.; Bittante, G. Short communication: Influence of composite casein genotypes on additive genetic variation of milk production traits and coagulation properties in Holstein-Friesian cows. J. Dairy Sci. 2010, 93, 3346–3349. [Google Scholar] [CrossRef]
- Edwards, R.A.; King, J.W.B.; Yousef, I.M. A note on the genetic variation in the fatty acid composition of cow milk. Anim. Sci. 1973, 16, 307–310. [Google Scholar] [CrossRef]
- Karijord, Ø.; Standal, N.; Syrstad, O. Sources of variation in composition of milk fat. Zeitschrift für Tierzüchtung und Züchtungsbiologie 1982, 99, 81–93. [Google Scholar] [CrossRef]
- Lindström, U.B.; Antila, V.; Syväjärvi, J. A Note on Some Genetic and Non-Genetic Factors Affecting Clotting Time of Ayrshire Milk. Acta Agric. Scand. 1984, 34, 349–355. [Google Scholar] [CrossRef]
- Hayes, J.F.; Ng-Kwai-Hang, K.F.; Moxley, J.E. Heritability of Milk Casein and Genetic and Phenotypic Correlations with Production Traits. J. Dairy Sci. 1984, 67, 841–846. [Google Scholar] [CrossRef]
- van Hulzen, K.J.E.; Sprong, R.C.; van der Meer, R.; van Arendonk, J.A.M. Genetic and nongenetic variation in concentration of selenium, calcium, potassium, zinc, magnesium, and phosphorus in milk of Dutch Holstein-Friesian cows. J. Dairy Sci. 2009, 92, 5754–5759. [Google Scholar] [CrossRef] [PubMed]
- Bittante, G.; Penasa, M.; Cecchinato, A. Invited review: Genetics and modeling of milk coagulation properties. J. Dairy Sci. 2012, 95, 6843–6870. [Google Scholar] [CrossRef] [PubMed]
- Bonfatti, V.; Cecchinato, A.; Gallo, L.; Blasco, A.; Carnier, P. Genetic analysis of detailed milk protein composition and coagulation properties in Simmental cattle. J. Dairy Sci. 2011, 94, 5183–5193. [Google Scholar] [CrossRef] [PubMed]
- Etzion, Y.; Linker, R.; Cogan, U.; Shmulevich, I. Determination of Protein Concentration in Raw Milk by Mid-Infrared Fourier Transform Infrared/Attenuated Total Reflectance Spectroscopy. J. Dairy Sci. 2004, 87, 2779–2788. [Google Scholar] [CrossRef]
- Soyeurt, H.; Dardenne, P.; Dehareng, F.; Lognay, G.; Veselko, D.; Marlier, M.; Bertozzi, C.; Mayeres, P.; Gengler, N. Estimating Fatty Acid Content in Cow Milk Using Mid-Infrared Spectrometry. J. Dairy Sci. 2006, 89, 3690–3695. [Google Scholar] [CrossRef]
- Dal Zotto, R.; De Marchi, M.; Cecchinato, A.; Penasa, M.; Cassandro, M.; Carnier, P.; Gallo, L.; Bittante, G. Reproducibility and repeatability of measures of milk coagulation properties and predictive ability of mid-infrared reflectance spectroscopy. J. Dairy Sci. 2008, 91, 4103–4112. [Google Scholar] [CrossRef]
- De Marchi, M.; Fagan, C.C.; O’Donnell, C.P.; Cecchinato, A.; Dal Zotto, R.; Cassandro, M.; Penasa, M.; Bittante, G. Prediction of coagulation properties, titratable acidity, and pH of bovine milk using mid-infrared spectroscopy. J. Dairy Sci. 2009, 92, 423–432. [Google Scholar] [CrossRef]
- Cecchinato, A.; De Marchi, M.; Gallo, L.; Bittante, G.; Carnier, P. Mid-infrared spectroscopy predictions as indicator traits in breeding programs for enhanced coagulation properties of milk. J. Dairy Sci. 2009, 92, 5304–5313. [Google Scholar] [CrossRef]
- Soyeurt, H.; Bruwier, D.; Romnee, J.M.; Gengler, N.; Bertozzi, C.; Veselko, D.; Dardenne, P.; Bovenhuis, H.; Verstege, A.J. M Potential estimation of major mineral contents in cow milk using mid-infrared spectrometry. J. Dairy Sci. 2009, 92, 2444–2454. [Google Scholar] [CrossRef]
- De Marchi, M.; Bonfatti, V.; Cecchinato, A.; Di Martino, G.; Carnier, P. Prediction of protein composition of individual cow milk using mid-infrared spectroscopy. Ital. J. Anim. Sci. 2009, 8, 399–401. [Google Scholar] [CrossRef]
- Rutten, M.J.M.; Bovenhuis, H.; Heck, J.M.L.; van Arendonk, J.A.M. Predicting bovine milk protein composition based on Fourier transform infrared spectra. J. Dairy Sci. 2011, 94, 5683–5690. [Google Scholar] [CrossRef] [PubMed]
- Bittante, G.; Cecchinato, A. Genetic analysis of the Fourier-transform infrared spectra of bovine milk with emphasis on individual wavelengths related to specific chemical bonds. J. Dairy Sci. 2013, 96, 5991–6006. [Google Scholar] [CrossRef] [PubMed]
- De Marchi, M.; Toffanin, V.; Cassandro, M.; Penasa, M. Invited review: Mid-infrared spectroscopy as phenotyping tool for milk traits. J. Dairy Sci. 2014, 97, 1171–1186. [Google Scholar] [CrossRef]
- Hill, W.G. Applications of Population Genetics to Animal Breeding, from Wright, Fisher and Lush to Genomic Prediction. Genetics 2014, 196, 1–16. [Google Scholar] [CrossRef]
- Gianola, D.; Rosa, G.J.M. One Hundred Years of Statistical Developments in Animal Breeding. Annu. Rev. Anim. Biosci. 2015, 3, 19–56. [Google Scholar] [CrossRef]
- Smith, C. Improvement of metric traits through specific genetic loci. Anim. Sci. 1967, 9, 349–358. [Google Scholar] [CrossRef]
- Brum, E.W.; Rausch, W.H.; Hines, H.C.; Ludwick, T.M. Association Between Milk and Blood Polymorphism Types and Lactation Traits of Holstein Cattle. J. Dairy Sci. 1968, 51, 1031–1038. [Google Scholar] [CrossRef]
- Soller, M.; Genizi, A. The Efficiency of Experimental Designs for the Detection of Linkage between a Marker Locus and a Locus Affecting a Quantitative Trait in Segregating Populations. Biometrics 1978, 34, 47. [Google Scholar] [CrossRef]
- Rothschild, M.; Jacobson, C.; Vaske, D.; Tuggle, C.; Wang, L.; Short, T.; Eckardt, G.; Sasaki, S.; Vincent, A.; McLaren, D.; et al. The estrogen receptor locus is associated with a major gene influencing litter size in pigs. Proc. Natl. Acad. Sci. USA 1996, 93, 201–205. [Google Scholar] [CrossRef]
- Aschaffenburg, R.; Drewry, J. Occurrence of Different Beta-Lactoglobulins in Cow’s Milk. Nature 1955, 176, 218–219. [Google Scholar] [CrossRef] [PubMed]
- Aschaffenburg, R. Inherited Casein Variants in Cow’s Milk. Nature 1961, 192, 431–432. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Ollivier-Bousquet, M.; Grosclaude, F. Genetic polymorphism of caseins: A tool to investigate casein micelle organization. Int. Dairy J. 1999, 9, 163–171. [Google Scholar] [CrossRef]
- Amigo, L.; Recio, I.; Ramos, M. Genetic polymorphism of ovine milk proteins: Its influence on technological properties of milk—A review. Int. Dairy J. 2000, 10, 135–149. [Google Scholar] [CrossRef]
- Caroli, A.M.; Chessa, S.; Erhardt, G.J. Invited review: Milk protein polymorphisms in cattle: Effect on animal breeding and human nutrition. J. Dairy Sci. 2009, 92, 5335–5352. [Google Scholar] [CrossRef]
- Selvaggi, M.; Laudadio, V.; Dario, C.; Tufarelli, V. Investigating the genetic polymorphism of sheep milk proteins: A useful tool for dairy production. J. Sci. Food Agric. 2014, 94, 3090–3099. [Google Scholar] [CrossRef]
- Bovenhuis, H.; Verstege, A.J.M. Improved method for phenotyping milk protein variants by isoelectric focusing using PhastSystem. Neth. Milk Dairy J. 1989, 43, 447–451. [Google Scholar]
- Bovenhuis, H.; Van Arendonk, J.A.M.; Korver, S. Associations Between Milk Protein Polymorphisms and Milk Production Traits. J. Dairy Sci. 1992, 75, 2549–2559. [Google Scholar] [CrossRef]
- Jakob, E.; Puhan, Z. Technological properties of milk as influenced by genetic polymorphism of milk proteins—A review. Int. Dairy J. 1992, 2, 157–178. [Google Scholar] [CrossRef]
- Ng-Kwai-Hang, K.F.; Hayes, J.F.; Moxley, J.E.; Monardes, H.G. Variation in Milk Protein Concentrations Associated with Genetic Polymorphism and Environmental Factors. J. Dairy Sci. 1987, 70, 563–570. [Google Scholar] [CrossRef]
- Ng-Kwai-Hang, K.F.; Kim, S. Different amounts of β-lactoglobulin A and B in milk from heterozygous AB cows. Int. Dairy J. 1996, 6, 689–695. [Google Scholar] [CrossRef]
- Barillet, F.; Arranz, J.-J.; Carta, A. Mapping quantitative trait loci for milk production and genetic polymorphisms of milk proteins in dairy sheep. Genet. Sel. Evol. 2005, 37, S109. [Google Scholar] [CrossRef] [PubMed]
- Cecchinato, A.; Chessa, S.; Ribeca, C.; Cipolat-Gotet, C.; Bobbo, T.; Casellas, J.; Bittante, G. Genetic variation and effects of candidate-gene polymorphisms on coagulation properties, curd firmness modeling and acidity in milk from Brown Swiss cows. Animal 2015, 9, 1104–1112. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ramos, A.M.; Matos, C.A.P.; Russo-Almeida, P.A.; Bettencourt, C.M.V.; Matos, J.; Martins, A.; Pinheiro, C.; Rangel-Figueiredo, T. Candidate genes for milk production traits in Portuguese dairy sheep. Small Rumin. Res. 2009, 82, 117–121. [Google Scholar] [CrossRef]
- Corral, J.M.; Padilla, J.A.; Izquierdo, M. Associations between milk protein genetic polymorphisms and milk production traits in Merino sheep breed. Livest. Sci. 2010, 129, 73–79. [Google Scholar] [CrossRef]
- Giambra, I.J.; Brandt, H.; Erhardt, G. Milk protein variants are highly associated with milk performance traits in East Friesian Dairy and Lacaune sheep. Small Rumin. Res. 2014, 121, 382–394. [Google Scholar] [CrossRef]
- Noce, A.; Pazzola, M.; Dettori, M.L.; Amills, M.; Castelló, A.; Cecchinato, A.; Bittante, G.; Vacca, G.M. Variations at regulatory regions of the milk protein genes are associated with milk traits and coagulation properties in the Sarda sheep. Anim. Genet. 2016, 47, 717–726. [Google Scholar] [CrossRef]
- Clark, S.; Sherbon, J.W. Genetic variants of alphas1-CN in goat milk: Breed distribution and associations with milk composition and coagulation properties. Small Rumin. Res. 2000, 38, 135–143. [Google Scholar] [CrossRef]
- Hayes, H.; Petit, E.; Bouniol, C.; Popescu, P. Localization of the α-S2-casein gene (CASAS2) to the homoeologous cattle, sheep, and goat chromosomes 4 by in situ hybridization. Cytogenet. Genome Res. 1993, 64, 281–285. [Google Scholar] [CrossRef]
- Meier, S.; Korkuć, P.; Arends, D.; Brockmann, G.A. DNA Sequence Variants and Protein Haplotypes of Casein Genes in German Black Pied Cattle (DSN). Front. Genet. 2019, 10, 1129. [Google Scholar] [CrossRef]
- Echard, G.; Broad, T.E.; Hill, D.; Pearce, P. Present status of the ovine gene map (Ovis aries); comparison with the bovine map (Bos taurus). Mamm. Genome 1994, 5, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Ikonen, T.; Bovenhuis, H.; Ojala, M.; Ruottinen, O.; Georges, M. Associations Between Casein Haplotypes and First Lactation Milk Production Traits in Finnish Ayrshire Cows. J. Dairy Sci. 2001, 84, 507–514. [Google Scholar] [CrossRef]
- Boettcher, P.J.; Caroli, A.; Stella, A.; Chessa, S.; Budelli, E.; Canavesi, F.; Ghiroldi, S.; Pagnacco, G. Effects of Casein Haplotypes on Milk Production Traits in Italian Holstein and Brown Swiss Cattle. J. Dairy Sci. 2004, 87, 4311–4317. [Google Scholar] [CrossRef]
- Hayes, B.; Hagesæther, N.; Ådnøy, T.; Pellerud, G.; Berg, P.R.; Lien, S. Effects on Production Traits of Haplotypes Among Casein Genes in Norwegian Goats and Evidence for a Site of Preferential Recombination. Genetics 2006, 174, 455–464. [Google Scholar] [CrossRef]
- Pizarro Inostroza, M.G.; Navas González, F.J.; Landi, V.; León Jurado, J.M.; Delgado Bermejo, J.V.; Álvarez, J.F.; Martínez Martínez, M.D.A. Bayesian Analysis of the Association between Casein Complex Haplotype Variants and Milk Yield, Composition, and Curve Shape Parameters in Murciano-Granadina Goats. Animals 2020, 10, 1845. [Google Scholar] [CrossRef]
- Buchberger, J.; Dovč, P. Lactoprotein Genetic Variants in Cattle and Cheese Making Ability. Food Technol. Biotechnol. 2000, 38, 91–98. [Google Scholar]
- Martin, P.; Szymanowska, M.; Zwierzchowski, L.; Leroux, C. The impact of genetic polymorphisms on the protein composition of ruminant milks. Reprod. Nutr. Dev. 2002, 42, 433–459. [Google Scholar] [CrossRef]
- Ogorevc, J.; Kunej, T.; Razpet, A.; Dovc, P. Database of cattle candidate genes and genetic markers for milk production and mastitis. Anim. Genet. 2009, 40, 832–851. [Google Scholar] [CrossRef]
- Cohen-Zinder, M.; Seroussi, E.; Larkin, D.M.; Loor, J.J.; Everts-van der Wind, A.; Lee, J.H.; Drackley, J.K.; Band, M.R.; Hernandez, A.G.; Shani, M.; et al. Identification of a missense mutation in the bovine ABCG2 gene with a major effect on the QTL on chromosome 6 affecting milk yield and composition in Holstein cattle. Genome Res. 2005, 15, 936–944. [Google Scholar] [CrossRef]
- Ron, M.; Cohen-Zinder, M.; Peter, C.; Weller, J.I.; Erhardt, G. Short Communication: A Polymorphism in ABCG2 in Bos indicus and Bos taurus Cattle Breeds. J. Dairy Sci. 2006, 89, 4921–4923. [Google Scholar] [CrossRef]
- Grisart, B.; Coppieters, W.; Farnir, F.; Karim, L.; Ford, C.; Berzi, P.; Cambisano, N.; Mni, M.; Reid, S.; Simon, P.; et al. Positional Candidate Cloning of a QTL in Dairy Cattle: Identification of a Missense Mutation in the Bovine DGAT1 Gene with Major Effect on Milk Yield and Composition. Genome Res. 2002, 12, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Spelman, R.J.; Ford, C.A.; McElhinney, P.; Gregory, G.C.; Snell, R.G. Characterization of the DGAT1 Gene in the New Zealand Dairy Population. J. Dairy Sci. 2002, 85, 3514–3517. [Google Scholar] [CrossRef]
- Banos, G.; Woolliams, J.A.; Woodward, B.W.; Forbes, A.B.; Coffey, M.P. Impact of Single Nucleotide Polymorphisms in Leptin, Leptin Receptor, Growth Hormone Receptor, and Diacylglycerol Acyltransferase (DGAT1) Gene Loci on Milk Production, Feed, and Body Energy Traits of UK Dairy Cows. J. Dairy Sci. 2008, 91, 3190–3200. [Google Scholar] [CrossRef]
- Glantz, M.; Lindmark Månsson, H.; Stålhammar, H.; Paulsson, M. Effect of polymorphisms in the leptin, leptin receptor, and acyl-coenzyme A:diacylglycerol acyltransferase 1 (DGAT1) genes and genetic polymorphism of milk proteins on cheese characteristics. J. Dairy Sci. 2011, 94, 3295–3304. [Google Scholar] [CrossRef] [PubMed]
- Liefers, S.C.; Veerkamp, R.F.; te Pas, M.F.W.; Delavaud, C.; Chilliard, Y.; Platje, M.; Lende, T. Leptin promoter mutations affect leptin levels and performance traits in dairy cows1. Anim. Genet. 2005, 36, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Weikard, R.; Kühn, C.; Goldammer, T.; Freyer, G.; Schwerin, M. The bovine PPARGC1A gene: Molecular characterization and association of an SNP with variation of milk fat synthesis. Physiol. Genomics 2005, 21, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Khatib, H.; Zaitoun, I.; Wiebelhaus-Finger, J.; Chang, Y.M.; Rosa, G.J.M. The Association of Bovine PPARGC1A and OPN Genes with Milk Composition in Two Independent Holstein Cattle Populations. J. Dairy Sci. 2007, 90, 2966–2970. [Google Scholar] [CrossRef]
- Bole-Feysot, C.; Goffin, V.; Edery, M.; Binart, N.; Kelly, P.A. Prolactin (PRL) and Its Receptor: Actions, Signal Transduction Pathways and Phenotypes Observed in PRL Receptor Knockout Mice. Endocr. Rev. 1998, 19, 225–268. [Google Scholar] [CrossRef]
- Brym, P.; Kamiński, S.; Wójcik, E. Nucleotide sequence polymorphism within exon 4 of the bovine prolactin gene and its associations with milk performance traits. J. Appl. Genet. 2005, 46, 179–185. [Google Scholar]
- He, F.; Sun, D.; Yu, Y.; Wang, Y.; Zhang, Y. Association between SNPs within prolactin gene and milk performance traits in Holstein Dairy Cattle. Asian-Australasian J. Anim. Sci. 2006, 19, 1384–1389. [Google Scholar] [CrossRef]
- Khatib, H.; Monson, R.L.; Schutzkus, V.; Kohl, D.M.; Rosa, G.J.M.; Rutledge, J.J. Mutations in the STAT5A Gene Are Associated with Embryonic Survival and Milk Composition in Cattle. J. Dairy Sci. 2008, 91, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Mullen, M.P.; Berry, D.P.; Howard, D.J.; Diskin, M.G.; Lynch, C.O.; Giblin, L.; Kenny, D.A.; Magee, D.A.; Meade, K.G.; Waters, S.M. Single Nucleotide Polymorphisms in the Insulin-Like Growth Factor 1 (IGF-1) Gene are Associated with Performance in Holstein-Friesian Dairy Cattle. Front. Genet. 2011, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Ibeagha-Awemu, E.M.; Kgwatalala, P.; Zhao, X. A critical analysis of production-associated DNA polymorphisms in the genes of cattle, goat, sheep, and pig. Mamm. Genome 2008, 19, 591–617. [Google Scholar] [CrossRef] [PubMed]
- Strucken, E.M.; Laurenson, Y.C.S.M.; Brockmann, G.A. Go with the flow-biology and genetics of the lactation cycle. Front. Genet. 2015, 6, 118. [Google Scholar] [CrossRef]
- Frazer, K.A.; Murray, S.S.; Schork, N.J.; Topol, E.J. Human genetic variation and its contribution to complex traits. Nat. Rev. Genet. 2009, 10, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Mackay, T.F.C.; Stone, E.A.; Ayroles, J.F. The genetics of quantitative traits: Challenges and prospects. Nat. Rev. Genet. 2009, 10, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Goddard, M.E.; Hayes, B.J. Mapping genes for complex traits in domestic animals and their use in breeding programmes. Nat. Rev. Genet. 2009, 10, 381–391. [Google Scholar] [CrossRef]
- Flint, J.; Mackay, T.F.C. Genetic architecture of quantitative traits in mice, flies, and humans. Genome Res. 2009, 19, 723–733. [Google Scholar] [CrossRef]
- Yang, J.; Benyamin, B.; McEvoy, B.P.; Gordon, S.; Henders, A.K.; Nyholt, D.R.; Madden, P.A.; Heath, A.C.; Martin, N.G.; Montgomery, G.W.; et al. Common SNPs explain a large proportion of the heritability for human height. Nat. Genet. 2010, 42, 565–569. [Google Scholar] [CrossRef]
- Visscher, P.M.; Brown, M.A.; McCarthy, M.I.; Yang, J. Five Years of GWAS Discovery. Am. J. Hum. Genet. 2012, 90, 7–24. [Google Scholar] [CrossRef]
- Kemper, K.E.; Goddard, M.E. Understanding and predicting complex traits: Knowledge from cattle. Hum. Mol. Genet. 2012, 21, R45–R51. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.B.; VanRaden, P.M.; O’Connell, J.R.; Van Tassell, C.P.; Sonstegard, T.S.; Schnabel, R.D.; Taylor, J.F.; Wiggans, G.R. Distribution and location of genetic effects for dairy traits. J. Dairy Sci. 2009, 92, 2931–2946. [Google Scholar] [CrossRef] [PubMed]
- Curik, I.; Druml, T.; Seltenhammer, M.; Sundström, E.; Pielberg, G.R.; Andersson, L.; Sölkner, J. Complex Inheritance of Melanoma and Pigmentation of Coat and Skin in Grey Horses. PLoS Genet. 2013, 9, e1003248. [Google Scholar] [CrossRef] [PubMed]
- Meuwissen, T.H.E.; Hayes, B.J.; Goddard, M.E. Prediction of Total Genetic Value Using Genome-Wide Dense Marker Maps. Genetics 2001, 157, 1819–1829. [Google Scholar] [CrossRef] [PubMed]
- Mai, M.D.; Sahana, G.; Christiansen, F.B.; Guldbrandtsen, B. A genome-wide association study for milk production traits in Danish Jersey cattle using a 50K single nucleotide polymorphism chip. J. Anim. Sci. 2010, 88, 3522–3528. [Google Scholar] [CrossRef] [PubMed]
- Schopen, G.C.B.; Visker, M.H.P.W.; Koks, P.D.; Mullaart, E.; van Arendonk, J.A.M.; Bovenhuis, H. Whole-genome association study for milk protein composition in dairy cattle. J. Dairy Sci. 2011, 94, 3148–3158. [Google Scholar] [CrossRef] [PubMed]
- Mucha, S.; Mrode, R.; Coffey, M.; Kizilaslan, M.; Desire, S.; Conington, J. Genome-wide association study of conformation and milk yield in mixed-breed dairy goats. J. Dairy Sci. 2018, 101, 2213–2225. [Google Scholar] [CrossRef]
- Jiang, J.; Ma, L.; Prakapenka, D.; VanRaden, P.M.; Cole, J.B.; Da, Y. A Large-Scale Genome-Wide Association Study in U.S. Holstein Cattle. Front. Genet. 2019, 10, 412. [Google Scholar] [CrossRef]
- Marina, H.; Gutiérrez-Gil, B.; Esteban-Blanco, C.; Suárez-Vega, A.; Pelayo, R.; Arranz, J.J. Analysis of whole genome resequencing datasets from a worldwide sample of sheep breeds to identify potential causal mutations influencing milk composition traits. Animals 2020, 10, 1542. [Google Scholar] [CrossRef]
- Scholtens, M.; Jiang, A.; Smith, A.; Littlejohn, M.; Lehnert, K.; Snell, R.; Lopez-Villalobos, N.; Garrick, D.; Blair, H. Genome-wide association studies of lactation yields of milk, fat, protein and somatic cell score in New Zealand dairy goats. J. Anim. Sci. Biotechnol. 2020, 11, 55. [Google Scholar] [CrossRef]
- Pryce, J.E.; Bolormaa, S.; Chamberlain, A.J.; Bowman, P.J.; Savin, K.; Goddard, M.E.; Hayes, B.J. A validated genome-wide association study in 2 dairy cattle breeds for milk production and fertility traits using variable length haplotypes. J. Dairy Sci. 2010, 93, 3331–3345. [Google Scholar] [CrossRef] [PubMed]
- Dadousis, C.; Biffani, S.; Cipolat-Gotet, C.; Nicolazzi, E.L.; Rosa, G.J.M.; Gianola, D.; Rossoni, A.; Santus, E.; Bittante, G.; Cecchinato, A. Genome-wide association of coagulation properties, curd firmness modeling, protein percentage, and acidity in milk from Brown Swiss cows. J. Dairy Sci. 2016, 99, 3654–3666. [Google Scholar] [CrossRef] [PubMed]
- Marina, H.; Pelayo, R.; Suárez-Vega, A.; Gutiérrez-Gil, B.; Esteban-Blanco, C.; Arranz, J.J. Genome-wide association studies (GWAS) and post-GWAS analyses for technological traits in Assaf and Churra dairy breeds. J. Dairy Sci. 2021, 104, 11850–11866. [Google Scholar] [CrossRef]
- Hayes, B.J.; Bowman, P.J.; Chamberlain, A.J.; Goddard, M.E. Invited review: Genomic selection in dairy cattle: Progress and challenges. J. Dairy Sci. 2009, 92, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Hickey, J.M.; Chiurugwi, T.; Mackay, I.; Powell, W. Genomic prediction unifies animal and plant breeding programs to form platforms for biological discovery. Nat. Genet. 2017, 49, 1297–1303. [Google Scholar] [CrossRef]
- Wiggans, G.R.; Cole, J.B.; Hubbard, S.M.; Sonstegard, T.S. Genomic Selection in Dairy Cattle: The USDA Experience. Annu. Rev. Anim. Biosci. 2017, 5, 309–327. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.B.; VanRaden, P.M. Symposium review: Possibilities in an age of genomics: The future of selection indices. J. Dairy Sci. 2018, 101, 3686–3701. [Google Scholar] [CrossRef]
- Van Eenennaam, A.L.; Weigel, K.A.; Young, A.E.; Cleveland, M.A.; Dekkers, J.C.M. Applied Animal Genomics: Results from the Field. Annu. Rev. Anim. Biosci. 2014, 2, 105–139. [Google Scholar] [CrossRef]
- Koltes, J.E.; Cole, J.B.; Clemmens, R.; Dilger, R.N.; Kramer, L.M.; Lunney, J.K.; McCue, M.E.; McKay, S.D.; Mateescu, R.G.; Murdoch, B.M.; et al. A Vision for Development and Utilization of High-Throughput Phenotyping and Big Data Analytics in Livestock. Front. Genet. 2019, 10, 1197. [Google Scholar] [CrossRef]
- Mota, L.F.M.; Pegolo, S.; Baba, T.; Peñagaricano, F.; Morota, G.; Bittante, G.; Cecchinato, A. Evaluating the performance of machine learning methods and variable selection methods for predicting difficult-to-measure traits in Holstein dairy cattle using milk infrared spectral data. J. Dairy Sci. 2021, 104, 8107–8121. [Google Scholar] [CrossRef]
- Georges, M.; Charlier, C.; Hayes, B. Harnessing genomic information for livestock improvement. Nat. Rev. Genet. 2019, 20, 135–156. [Google Scholar] [CrossRef] [PubMed]
- Rexroad, C.; Vallet, J.; Matukumalli, L.K.; Reecy, J.; Bickhart, D.; Blackburn, H.; Boggess, M.; Cheng, H.; Clutter, A.; Cockett, N.; et al. Genome to Phenome: Improving Animal Health, Production, and Well-Being—A New USDA Blueprint for Animal Genome Research. Front. Genet. 2019, 10, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- Brito, L.F.; Bedere, N.; Douhard, F.; Oliveira, H.R.; Arnal, M.; Peñagaricano, F.; Schinckel, A.P.; Baes, C.F.; Miglior, F. Review: Genetic selection of high-yielding dairy cattle toward sustainable farming systems in a rapidly changing world. Animal 2021, 15, 100292. [Google Scholar] [CrossRef]
- Moran, D.; Blair, K.J. Review: Sustainable livestock systems: Anticipating demand-side challenges. Animal 2021, 15, 100288. [Google Scholar] [CrossRef] [PubMed]
- Yegin, S.; Dekker, P. Progress in the field of aspartic proteinases in cheese manufacturing: Structures, functions, catalytic mechanism, inhibition, and engineering. Dairy Sci. Technol. 2013, 93, 565–594. [Google Scholar] [CrossRef]
- MEROPS Database. Available online: https://www.ebi.ac.uk/merops/ (accessed on 13 January 2022).
- Rawlings, N.D.; Barrett, A.J. Peptidases. In eLS; Wiley: Hoboken, NJ, USA, 2014. [Google Scholar] [CrossRef]
- Rawlings, N.D.; Barrett, A.J.; Thomas, P.D.; Huang, X.; Bateman, A.; Finn, R.D. The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res. 2018, 46, D624–D632. [Google Scholar] [CrossRef] [PubMed]
- Schechter, I.; Berger, A. On the size of the active site in proteases. I. Papain. Biochem. Biophys. Res. Commun. 1967, 27, 157–162. [Google Scholar] [CrossRef]
- Schechter, I. Mapping of the Active Site of Proteases in the 1960s and Rational Design of Inhibitors/Drugs in the 1990s. Curr. Protein Pept. Sci. 2005, 6, 501–512. [Google Scholar] [CrossRef]
- Tang, J. Aspartic Proteases: Structure, Function, and Inhibition. In Aspartic Acid Proteases as Therapeutic Targets; Wiley: Hoboken, NJ, USA, 2011; Volume 45, pp. 23–41. [Google Scholar] [CrossRef]
- Andrén, A. Cheese: Rennets and Coagulants. In Encyclopedia of Dairy Sciences, 2nd ed.; Academic Press: San Diego, CA, USA, 2011; pp. 574–578. [Google Scholar] [CrossRef]
- Barrett, A.J.; Rawlings, N.D.; Salvesen, G.; Fred Woessner, J. Introduction. In Handbook of Proteolytic Enzymes; Elsevier: Amsterdam, The Netherlands, 2013; pp. li–liv. [Google Scholar] [CrossRef]
- Bela Szecsi, P.; Harboe, M. Chymosin. In Handbook of Proteolytic Enzymes; Elsevier: Amsterdam, The Netherlands, 2013; pp. 37–42. [Google Scholar] [CrossRef]
- Lea, A.S.; Dickinson, W.L. Notes on the mode of action of Rennin and Fibrin-ferment. J. Physiol. 1890, 11, 307–311. [Google Scholar] [CrossRef]
- Foltmann, B.; Szecsi, P.B. Chymosin. In Handbook of Proteolytic Enzymes, 2nd ed.; Barrett, A.J., Rawlings, N.D., Woessner, J.F., Eds.; Academic Press: London, UK, 2004; pp. 29–32. [Google Scholar] [CrossRef]
- Szecsi, P.B. The aspartic proteases. Scand. J. Clin. Lab. Invest. 1992, 52, 5–22. [Google Scholar] [CrossRef]
- Tang, J. Chapter 3—Pepsin A. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, N.D., Salvesen, G., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 27–35. [Google Scholar] [CrossRef]
- Uniacke-Lowe, T.; Fox, P.F. Chapter 4—Chymosin, Pepsins and Other Aspartyl Proteinases: Structures, Functions, Catalytic Mechanism and Milk-Clotting Properties. In Cheese, 4th ed.; McSweeney, P.L.H., Fox, P.F., Cotter, P.D., Everett, D.W., Eds.; Academic Press: San Diego, CA, USA, 2017; pp. 69–113. [Google Scholar] [CrossRef]
- Pang, S.H. Rennin Content of Abomasa from Bovine Fetuses. 1969. All Graduate Theses and Dissertations, 5269. Available online: https://digitalcommons.usu.edu/etd/5269 (accessed on 20 January 2022).
- Davies, D.R. The Structure and Function of the Aspartic Proteinases. Annu. Rev. Biophys. Biophys. Chem. 1990, 19, 189–215. [Google Scholar] [CrossRef] [PubMed]
- Visser, S.; van Rooijen, P.J.; Schattenkerk, C.; Kerling, K.E.T. Peptide substrates for chymosin (Rennin). Kinetic studies with peptides of different chain lenght including parts of the sequence 101–112 of bovine κ-casein. Biochim. Biophys. Acta Enzymol. 1976, 438, 265–272. [Google Scholar] [CrossRef]
- Rampilli, M.; Larsen, R.; Harboe, M. Natural heterogeneity of chymosin and pepsin in extracts of bovine stomachs. Int. Dairy J. 2005, 15, 1130–1137. [Google Scholar] [CrossRef]
- Jaros, D.; Rohm, H. Rennets: Applied Aspects, 4th ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Kumar, A.; Grover, S.; Sharma, J.; Batish, V.K. Chymosin and other milk coagulants: Sources and biotechnological interventions. Crit. Rev. Biotechnol. 2010, 30, 243–258. [Google Scholar] [CrossRef]
- Andren, A. Rennets and coagulants. In Encyclopedia of Dairy Sciences, 1st ed.; Roginski, H., Fuquay, W.J., Fox, P.F., Eds.; Academic Press: London, UK, 2003; pp. 283–286. [Google Scholar]
- Teuber, M. Production of chymosin (EC 3.4.23.4) by microorganisms and its use for cheesemaking. Bull. Int. Dairy Fed. 1990, 251, 3–15. [Google Scholar]
- Aworh, O.C.; Muller, H.G. Cheese-making properties of vegetable rennet from sodom apple (Calotropis procera). Food Chem. 1987, 26, 71–79. [Google Scholar] [CrossRef]
- Sousa, M.J.; Malcata, F.X. Advances in the role of a plant coagulant (Cynara cardunculus) in vitro and during ripening of cheeses from several milk species. Lait 2002, 82, 151–170. [Google Scholar] [CrossRef]
- Uchiyama, H.; Uozumi, T.; Beppu, T.; Arima, K. Purification of Prorennin mRNA and Its Translation in Vitro. Agric. Biol. Chem. 1980, 44, 1373–1381. [Google Scholar] [CrossRef]
- Foltmann, B. General and Molecular Aspects of Rennets. In Cheese: Chemistry, Physics and Microbiology; Springer US: Boston, MA, USA, 1993; pp. 37–68. [Google Scholar] [CrossRef]
- Emtage, J.S.; Angal, S.; Doel, M.T.; Harris, T.J.; Jenkins, B.; Lilley, G.; Lowe, P.A. Synthesis of calf prochymosin (prorennin) in Escherichia coli. Proc. Natl. Acad. Sci. USA 1983, 80, 3671–3675. [Google Scholar] [CrossRef]
- Kumar, A.; Sharma, J.; Mohanty, A.K.; Grover, S.; Batish, V.K. Purification and characterization of milk clotting enzyme from goat (Capra hircus). Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2006, 145, 108–113. [Google Scholar] [CrossRef]
- Starovoitova, V.V.; Velichko, T.I.; Baratova, L.A.; Filippova, I.Y.; Lavrenova, G.I. A comparative study of functional properties of calf chymosin and its recombinant forms. Biochemistry 2006, 71, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Cullen, D.; Gray, G.L.; Wilson, L.J.; Hayenga, K.J.; Lamsa, M.H.; Rey, M.W.; Norton, S.; Berka, R.M. Controlled Expression and Secretion of Bovine Chymosin in Aspergillus Nidulans. Nat. Biotechnol. 1987, 5, 369–376. [Google Scholar] [CrossRef]
- Noseda, D.G.; Recúpero, M.N.; Blasco, M.; Ortiz, G.E.; Galvagno, M.A. Cloning, expression and optimized production in a bioreactor of bovine chymosin B in Pichia (Komagataella) pastoris under AOX1 promoter. Protein Expr. Purif. 2013, 92, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Noseda, D.G.; Recúpero, M.; Blasco, M.; Bozzo, J.; Galvagno, M.Á. Production in stirred-tank bioreactor of recombinant bovine chymosin B by a high-level expression transformant clone of Pichia pastoris. Protein Expr. Purif. 2016, 123, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Espinoza-Molina, J.A.; Acosta-Muñiz, C.H.; Sepulveda, D.R.; Zamudio-Flores, P.B.; Rios-Velasco, C. Codon Optimization of the “Bos Taurus Chymosin” Gene for the Production of Recombinant Chymosin in Pichia pastoris. Mol. Biotechnol. 2016, 58, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Belenkaya, S.V.; Balabova, D.V.; Belov, A.N.; Koval, A.D.; Shcherbakov, D.N.; Elchaninov, V.V. Basic Biochemical Properties of Recombinant Chymosins (Review). Appl. Biochem. Microbiol. 2020, 56, 363–372. [Google Scholar] [CrossRef]
- Kappeler, S.R.; van den Brink, H.J.M.; Rahbek-Nielsen, H.; Farah, Z.; Puhan, Z.; Hansen, E.B.; Johansen, E. Characterization of recombinant camel chymosin reveals superior properties for the coagulation of bovine and camel milk. Biochem. Biophys. Res. Commun. 2006, 342, 647–654. [Google Scholar] [CrossRef]
- Vallejo, J.A.; Ageitos, J.M.; Poza, M.; Villa, T.G. Short communication: A comparative analysis of recombinant chymosins. J. Dairy Sci. 2012, 95, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Langholm, J.J.; Mølgaard, A.; Navarro Poulsen, J.C.; Harboe, M.K.; Simonsen, J.B.; Lorentzen, A.M.; Hjernø, K.; van den Brink, J.M.; Qvist, K.B.; Larsen, S. Camel and bovine chymosin: The relationship between their structures and cheese-making properties. Acta Crystallogr. Sect. D Biol. Crystallogr. 2013, 69, 901–913. [Google Scholar] [CrossRef]
- Wang, N.; Wang, K.Y.; Li, G.; Guo, W.; Liu, D. Expression and characterization of camel chymosin in Pichia pastoris. Protein Expr. Purif. 2015, 111, 75–81. [Google Scholar] [CrossRef]
- Rogelj, I.; Perko, B.; Francky, A.; Penca, V.; Pungerčar, J. Recombinant Lamb Chymosin as an Alternative Coagulating Enzyme in Cheese Production. J. Dairy Sci. 2001, 84, 1020–1026. [Google Scholar] [CrossRef]
- Vega-Hernández, M.C.; Gómez-Coello, A.; Villar, J.; Claverie-Martıín, F. Molecular cloning and expression in yeast of caprine prochymosin. J. Biotechnol. 2004, 114, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.; Kumar, A.; Yadav, A.K.; Chandola Saklani, A.; Grover, S.; Batish, V.K. Functional expression of recombinant goat chymosin in Pichia pastoris bioreactor cultures: A commercially viable alternate. LWT Food Sci. Technol. 2016, 69, 217–224. [Google Scholar] [CrossRef]
- Liu, W.G.; Wang, Y.P.; Zhang, Z.J.; Wang, M.; Lv, Q.X.; Liu, H.W.; Meng, L.C.; Lu, M. Generation and characterization of caprine chymosin in corn seed. Protein Expr. Purif. 2017, 135, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, J.A.; Ageitos, J.M.; Poza, M.; Villa, T.G. Cloning and Expression of Buffalo Active Chymosin in Pichia pastoris. J. Agric. Food Chem. 2008, 56, 10606–10610. [Google Scholar] [CrossRef]
- Tyagi, A.; Kumar, A.; Mohanty, A.K.; Kaushik, J.K.; Grover, S.; Batish, V.K. Expression of buffalo chymosin in Pichia pastoris for application in mozzarella cheese. LWT 2017, 84, 733–739. [Google Scholar] [CrossRef]
- Luo, F.; Jiang, W.H.; Yang, Y.X.; Li, J.; Jiang, M.F. Cloning and Expression of Yak Active Chymosin in Pichia pastoris. Asian-Australas. J. Anim. Sci. 2016, 29, 1363–1370. [Google Scholar] [CrossRef]
- Ersöz, F.; İnan, M. Large-scale production of yak (Bos grunniens) chymosin A in Pichia pastoris. Protein Expr. Purif. 2019, 154, 126–133. [Google Scholar] [CrossRef]
- Belenkaya, S.V.; Rudometov, A.P.; Shcherbakov, D.N.; Balabova, D.V.; Kriger, A.V.; Belov, A.N.; Koval, A.D.; Elchaninov, V.V. Biochemical Properties of Recombinant Chymosin in Alpaca (Vicugna pacos L.). Appl. Biochem. Microbiol. 2018, 54, 569–576. [Google Scholar] [CrossRef]
- Jacob, M.; Jaros, D.; Rohm, H. Recent advances in milk clotting enzymes. Int. J. Dairy Technol. 2011, 64, 14–33. [Google Scholar] [CrossRef]
- Moschopoulou, E. Microbial milk coagulants. In Microbrobial Enzyme Technology in Food Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 199–213. [Google Scholar] [CrossRef]
- Ben Amira, A.; Besbes, S.; Attia, H.; Blecker, C. Milk-clotting properties of plant rennets and their enzymatic, rheological, and sensory role in cheese making: A review. Int. J. Food Prop. 2017, 20, S76–S93. [Google Scholar] [CrossRef]
- García, V.; Rovira, S.; Teruel, R.; Boutoial, K.; Rodriguez, R.; Roa, I.; López, M.B. Effect of vegetable coagulant, microbial coagulant and calf rennet on physicochemical, proteolysis, sensory and texture profiles of fresh goats cheese. Dairy Sci. Technol. 2012, 92, 691–707. [Google Scholar] [CrossRef]
- Manuelian, C.L.; Boselli, C.; Vigolo, V.; Giangolini, G.; De Marchi, M. Effects of animal versus vegetal rennet on milk coagulation traits in Mediterranean buffalo bulk milk. J. Dairy Sci. 2020, 103, 4958–4964. [Google Scholar] [CrossRef]
- Andren, A. Milk-clotting activity of various rennets and coagulants: Background and information regarding IDF standards. Int. Dairy Fed. 1998, 332, 9–14. [Google Scholar]
- Kumar, A.; Sharma, J.; Grover, S.; Kumar Mohanty, A.; Kumar Batish, V. Molecular Cloning and Expression of Goat (Capra hircus) Prochymosin in E.coli. Food Biotechnol. 2007, 21, 57–69. [Google Scholar] [CrossRef]
- Yang, B.; Wang, X.; Luo, J.; Tang, G.; Li, W.; Zhang, L. Cloning and sequence analyses of preprochymosin cDNA of Xinong Saanen Goat. Anim. Husb. Vet. Med. 2007, 4, 2–7. [Google Scholar]
- Anifantakis, E.; Green, M.L. Preparation and properties of rennets from lamb’s and kid’s abomasa. J. Dairy Res. 1980, 47, 221–230. [Google Scholar] [CrossRef]
- Moschopoulou, E. Characteristics of rennet and other enzymes from small ruminants used in cheese production. Small Rumin. Res. 2011, 101, 188–195. [Google Scholar] [CrossRef]
- Andren, A.; Bjorck, L.; Claesson, O. Imunohistochemical Studies on the Development of Prochymosin- and Pepsinogen-Containing Cells in Bovine Abomasal Mucosa. J. Physiol. 1982, 327, 247–254. [Google Scholar] [CrossRef]
- Foltmann, B. Chymosin: A short review on foetal and neonatal gastric proteases. Scand. J. Clin. Lab. Invest. Suppl. 1992, 210, 65–79. [Google Scholar] [CrossRef]
- Ward, O.P. Proteases. In Comprehensive Biotechnology, 3rd ed.; Moo-Young, M., Ed.; Elsevier: Amsterdam, Netherlands, 2019; pp. 604–615. [Google Scholar] [CrossRef]
- Foltmann, B. Studies on Rennin II. On the Crystallisation, Stability and Proteolytic Activity of Rennin. Acta Chem. Scand. 1959, 13, 1927–1935. [Google Scholar] [CrossRef][Green Version]
- Andrén, A. Milk-Clotting Enzymes. In Agents of Change: Enzymes in Milk and Dairy Products; Kelly, A.L., Larsen, L.B., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 349–362. [Google Scholar] [CrossRef]
- Kim, S.M.; Zayas, J.F. Influence of Ultrasound on the Properties of Chymosin and the Ultrastructure of Abomasum During Chymosin Extraction. J. Food Process. Preserv. 1991, 15, 89–100. [Google Scholar] [CrossRef]
- Kim, S.M.; Zayas, J.F. Processing Parameters of Chymosin Extraction by Ultrasound. J. Food Sci. 1989, 54, 700–703. [Google Scholar] [CrossRef]
- Zhang, F.X.; Wang, B.N. Optimization of processing parameters for the ultrasonic extraction of goat kid rennet. Int. J. Dairy Technol. 2007, 60, 286–291. [Google Scholar] [CrossRef]
- Tabayehnejad, N.; Castillo, M.; Payne, F.A. Comparison of total milk-clotting activity measurement precision using the Berridge clotting time method and a proposed optical method. J. Food Eng. 2012, 108, 549–556. [Google Scholar] [CrossRef]
- Berridge, N.J. Some observations on the determination of the activity of rennet. Analyst 1952, 77, 57–62. [Google Scholar] [CrossRef]
- Standard 157:2007; Milk: Determination of Total Milk-Clotting Activity of Bovine Rennets. International Dairy Federation: Geneva, Switzerland; Brussels, Belgium, 2007.
- Standard 176:2002; Milk and Milk Products: Microbial Coagulants: Determination of Total Milk-Clotting Activity. International Dairy Federation: Geneva, Switzerland; Brussels, Belgium, 2002.
- Standard 199:2006; Milk and Milk Products: Ovine and Caprine Rennets: Determination of Total Milk-Clotting Activity. International Dairy Federation: Geneva, Switzerland; Brussels, Belgium, 2006.
- Standard 110:2012; Milk and Milk Products—Calf Rennet and Adult Bovine Rennet—Determination by Chromatography of Chymosin and Bovine Pepsin Contents. International Dairy Federation: Geneva, Switzerland; Brussels, Belgium, 2012.
- Zdolec, N.; Jankuloski, D.; Kiš, M.; Hengl, B.; Mikulec, N. Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia. Beverages 2019, 5, 46. [Google Scholar] [CrossRef]
- Mikulec, N.; Špoljarić, J.; Zamberlin, Š.; Krga, M.; Radeljević, B.; Plavljanić, D.; Horvat Kesić, I.; Zdolec, N.; Dobranić, V.; Antunac, N. The investigation of suitability of raw milk consumption from vending machines in Croatia. J. Cent. Eur. Agric. 2019, 20, 1076–1088. [Google Scholar] [CrossRef]
- Kiš, M.; Kolačko, I.; Zdolec, N. Unprocessed milk as a source of multidrug-resistant Staphylococcus aureus strains. Acta Vet. Brno 2021, 90, 357–363. [Google Scholar] [CrossRef]
- Dobranić, V.; Kazazić, S.; Filipović, I.; Mikulec, N.; Zdolec, N. Composition of raw cow’s milk microbiota and identification of enterococci by MALDI-TOF MS—Short communication. Vet. Arh. 2016, 86, 581–590. [Google Scholar]
- Vrdoljak, J.; Dobranić, V.; Filipović, I.; Zdolec, N. Microbiological Quality of Soft, Semi-Hard and Hard Cheeses During the Shelf-Life. Maced. Vet. Rev. 2016, 39, 59–64. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Holzapfel, W.H.; Stiles, M.E. Enterococci at the crossroads of food safety? Int. J. Food Microbiol. 1999, 47, 1–24. [Google Scholar] [CrossRef]
- Zdolec, N.; Dobranić, V.; Butković, I.; Koturić, A.; Filipović, I.; Medvid, V. Osjetljivost na antimikrobne tvari bakterija izdvojenih iz mlijeka zdravih i liječenih vimena krava. Vet. Arh. 2016, 86, 163–172. [Google Scholar]
- Zdolec, N. Technological Interventions in Fermented Meat Production: The Commercial Perspective. In Innovations in Technologies for Fermented Food and Beverage Industries; Springer International Publishing: Cham, Switzerland, 2018; pp. 175–188. [Google Scholar] [CrossRef]
- Čanžek Majhenič, A. Enterococci: Yin—yang microbes. Mljekarstvo 2006, 56, 5–20. [Google Scholar]
- Hugas, M. Functionalty of enterococci in meat products. Int. J. Food Microbiol. 2003, 88, 223–233. [Google Scholar] [CrossRef]
- Stiles, M. Safety aspects of enterococci from food point of view. In Proceedings of the Symposium on Enterococci in Foods-Functional and Safety aspects, Berlin, Germany, 30–31 May 2002. [Google Scholar]
- Kos, B.; Šušković, J.; Beganović, J.; Gjuračić, K.; Frece, J.; Iannaccone, C.; Canganella, F. Characterization of the three selected probiotic strains for the application in food industry. World J. Microbiol. Biotechnol. 2008, 24, 699–707. [Google Scholar] [CrossRef]
- Leboš Pavunc, A.; Kos, B.; Beganović, J.; Uroić, K.; Bučan, D.; Šušković, J. Antibiotic susceptibility and antimicrobial activity of autochthonous starter cultures as safety parameters for fresh cheese production. Mljekarstvo 2013, 63, 185–194. [Google Scholar]
- Vukušić, N.; Zdolec, N. Utjecaj bakteriocina enterokoka na odabrane uzročnike bolesti prenosivih hranom. Vet. stanica 2020, 51, 139–143. [Google Scholar] [CrossRef]
- Oikonomou, G.; Addis, M.F.; Chassard, C.; Nader-Macias, M.E.F.; Grant, I.; Delbès, C.; Bogni, C.I.; Le Loir, Y.; Even, S. Milk Microbiota: What Are We Exactly Talking About? Front. Microbiol. 2020, 11, 60. [Google Scholar] [CrossRef]
- Klaubauf, S.; Segers, F.J.J. Research Tools and Methods for the Analysis of Microbiota in Dairy Products. In Microbial Cultures and Enzymes in Dairy Technology; Budak, Ş.Ö., Akal, H.C., Eds.; IGI Global: Herhshey, PA, USA, 2018; pp. 23–53. [Google Scholar] [CrossRef]
- Coppola, S.; Blaiotta, G.; Ercolini, D. Dairy Products. In Molecular Techniques in the Microbial Ecology of Fermented Foods; Springer New York: New York, NY, USA, 2007; pp. 31–90. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Barba-Orellana, S.; Barba, F.J.; Quilez, F.; Roohinejad, S.; Koubaa, M. New Trends in Molecular Techniques to Identify Microorganisms in Dairy Products. In Molecular Techniques in Food Biology, 1st ed.; El Sheikha, A.F., Levin, R., Xu, J., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2018; pp. 309–322. [Google Scholar] [CrossRef]
- Terzić-Vidojević, A.; Veljović, K.; Tolinački, M.; Živković, M.; Lukić, J.; Lozo, J.; Fira, Đ.; Jovčić, B.; Strahinić, I.; Begović, J.; et al. Diversity of non-starter lactic acid bacteria in autochthonous dairy products from Western Balkan Countries—Technological and probiotic properties. Food Res. Int. 2020, 136, 109494. [Google Scholar] [CrossRef]
- Mrkonjic Fuka, M.; Zgomba Maksimovic, A.; Tanuwidjaja, I.; Hulak, N.; Schloter, M. Characterization of Enterococcal Community Isolated from an Artisan Istrian Raw Milk Cheese: Biotechnological and Safety Aspects. Food Technol. Biotechnol. 2017, 55, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Skelin, A.; Mrkonjic Fuka, M.; Majhenič, A.Č.; Redžepović, S.; Samaržija, D.; Matijašić, B.B. Phenotypic and genotypic characterization of indigenous Lactobacillus community from traditional Istrian Ewe’s cheese. Food Technol. Biotechnol. 2012, 50, 362–370. [Google Scholar]
- Mrkonjic Fuka, M.; Wallisch, S.; Engel, M.; Welzl, G.; Havranek, J.; Schloter, M. Dynamics of Bacterial Communities during the Ripening Process of Different Croatian Cheese Types Derived from Raw Ewe’s Milk Cheeses. PLoS ONE 2013, 8, e80734. [Google Scholar] [CrossRef]
- Golić, N.; Čadež, N.; Terzić-Vidojević, A.; Šuranská, H.; Beganović, J.; Lozo, J.; Kos, B.; Šušković, J.; Raspor, P.; Topisirović, L. Evaluation of lactic acid bacteria and yeast diversity in traditional white pickled and fresh soft cheeses from the mountain regions of Serbia and lowland regions of Croatia. Int. J. Food Microbiol. 2013, 166, 294–300. [Google Scholar] [CrossRef]
- Frece, J.; Markov, K. Autochtonous starter cultures. In Fermented Meat Products: Health Aspects, 1st ed.; Zdolec, N., Ed.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2016; pp. 270–293. [Google Scholar]
- Zdolec, N.; Filipović, I.; Cvrtila Fleck, Ž.; Marić, A.; Jankuloski, D.; Kozačinski, L.; Njari, B. Antimicrobial susceptibility of lactic acid bacteria isolated from fermented sausages and raw cheese. Vet. Arh. 2011, 81, 133–141. [Google Scholar]
- Zdolec, N.; Lazić, S.; Kozačinski, L.; Hadžiosmanović, M.; Filipović, I. Inhibicijsko djelovanje bakterija mliječne kiseline izoliranih iz svježeg kravljeg sira. Mljekarstvo 2007, 57, 5–13. [Google Scholar]
- Zdolec, N.; Lorenzo, J.M.; Ray, R.C. Use of Microbes for Improving Food Safety and Quality. Biomed Res. Int. 2018, 2018, 3902698. [Google Scholar] [CrossRef]
- Nova, R.J.; Botsaris, G.; Cerda-Leal, F. Probiotics in fermented meat products. In Fermented Meat Products: Health Aspects, 1st ed.; Zdolec, N., Ed.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2016; pp. 294–318. [Google Scholar]
- Zdolec, N.; Dobranić, V.; Horvatić, A.; Vučinić, S. Selection and application of autochthonous functional starter cultures in traditional croatian fermented sausages. Int. Food Res. J. 2013, 20, 1–6. [Google Scholar]
- Šušković, J.; Kos, B.; Goreta, J.; Matošić, S. Role of Lactic Acid Bacteria and Bifidobacteria in Synbiotic Effect. Food Technol. Biotechnol. 2001, 39, 227–235. [Google Scholar]
- Šušković, J.; Kos, B.; Beganović, J.; Pavunc Leboš, A.; Habjanič, K.; Matoć, S. Antimicrobial activity—The most important property of probiotic and starter lactic acid bacteria. Food Technol. Biotechnol. 2010, 48, 296–307. [Google Scholar]
- Zdolec, N.; Bogdanović, T.; Severin, K.; Dobranić, V.; Kazazić, S.; Grbavac, J.; Pleadin, J.; Petričević, S.; Kiš, M. Biogenic Amine Content in Retailed Cheese Varieties Produced with Commercial Bacterial or Mold Cultures. Processes 2022, 10, 10. [Google Scholar] [CrossRef]
- Özer, B.; Kirmaci, H.A.; Şenel, E.; Atamer, M.; Hayaloğlu, A. Improving the viability of Bifidobacterium bifidum BB-12 and Lactobacillus acidophilus LA-5 in white-brined cheese by microencapsulation. Int. Dairy J. 2009, 19, 22–29. [Google Scholar] [CrossRef]
- Gomes da Cruz, A.; Alonso Buriti, F.C.; Batista de Souza, C.H.; Fonseca Faria, J.A.; Isay Saad, S.M. Probiotic cheese: Health benefits, technological and stability aspects. Trends Food Sci. Technol. 2009, 20, 344–354. [Google Scholar] [CrossRef]
- Calvo, P.; Castaño, Á.L.; Hernández, M.T.; González-Gómez, D. Effects of microcapsule constitution on the quality of microencapsulated walnut oil. Eur. J. Lipid Sci. Technol. 2011, 113, 1273–1280. [Google Scholar] [CrossRef]
- Jeyakumari, A. Microencapsulation of Bioactive Food Ingredients and Controlled Release—A Review. MOJ Food Process. Technol. 2016, 2, 59. [Google Scholar] [CrossRef]
- Champagne, C.P.; Fustier, P. Microencapsulation for the improved delivery of bioactive compounds into foods. Curr. Opin. Biotechnol. 2007, 18, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Ortakci, F.; Broadbent, J.R.; McManus, W.R.; McMahon, D.J. Survival of microencapsulated probiotic Lactobacillus paracasei LBC-1e during manufacture of Mozzarella cheese and simulated gastric digestion. J. Dairy Sci. 2012, 95, 6274–6281. [Google Scholar] [CrossRef]
- Goderska, K.; Zybała, M.; Czarnecki, Z. Characterisation Of Microencapsulated Lactobacillus Rhamnosus LR7 Strain. Polish J. Food Nutr. Sci. 2003, 53, 21–24. [Google Scholar]
- Abd-Elhamid, A.M. Production of functional kariesh cheese by microencapsulation of bifidobacterium adolescentis atcc 15704. Adv. J. Food Sci. Technol. 2012, 4, 112–117. [Google Scholar]
- Godward, G.; Kailasapathy, K. Viability and survival of free, encapsulated and co-encapsulated probiotic bacteria in yoghurt. Milchwissenschaft-milk Sci. Int. 2003, 58, 161–164. [Google Scholar]
- Özer, B.; Uzun, Y.S.; Kirmaci, H.A. Effect of Microencapsulation on Viability of Lactobacillus acidophilus LA-5 and Bifidobacterium bifidum BB-12 During Kasar Cheese Ripening. Int. J. Dairy Technol. 2008, 61, 237–244. [Google Scholar] [CrossRef]
- Kavitake, D.; Kandasamy, S.; Devi, P.B.; Shetty, P.H. Recent developments on encapsulation of lactic acid bacteria as potential starter culture in fermented foods—A review. Food Biosci. 2018, 21, 34–44. [Google Scholar] [CrossRef]
- Vlahoviček-Kahlina, K.; Jurić, S.; Marijan, M.; Mutaliyeva, B.; Khalus, S.V.; Prosyanik, A.V.; Vinceković, M. Synthesis, Characterization, and Encapsulation of Novel Plant Growth Regulators (PGRs) in Biopolymer Matrices. Int. J. Mol. Sci. 2021, 22, 1847. [Google Scholar] [CrossRef] [PubMed]
- Lemic, D.; Orešković, M.; Mikac, K.M.; Marijan, M.; Jurić, S.; Vlahoviček-Kahlina, K.; Vinceković, M. Sustainable Pest Management Using Biodegradable Apitoxin-Loaded Calcium-Alginate Microspheres. Sustainability 2021, 13, 6167. [Google Scholar] [CrossRef]
- Vinceković, M.; Viskić, M.; Jurić, S.; Giacometti, J.; Bursać Kovačević, D.; Putnik, P.; Donsì, F.; Barba, F.J.; Režek Jambrak, A. Innovative technologies for encapsulation of Mediterranean plants extracts. Trends Food Sci. Technol. 2017, 69, 1–12. [Google Scholar] [CrossRef]
- Burgain, J.; Gaiani, C.; Linder, M.; Scher, J. Encapsulation of probiotic living cells: From laboratory scale to industrial applications. J. Food Eng. 2011, 104, 467–483. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Encapsulation of polyphenols—A review. Trends Food Sci. Technol. 2010, 21, 510–523. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Siddique, M.A.B.; Fathi, M. Vegetable Oils Rich in Polyunsaturated Fatty Acids: Nanoencapsulation Methods and Stability Enhancement. Food Rev. Int. 2020, 32–69. [Google Scholar] [CrossRef]
- Desai, K.G.H.; Park, H.J. Encapsulation of vitamin C in tripolyphosphate cross-linked chitosan microspheres by spray drying. J. Microencapsul. 2005, 22, 179–192. [Google Scholar] [CrossRef]
- Fathi, M.; Vinceković, M.; Jurić, S.; Viskić, M.; Režek Jambrak, A.; Donsì, F. Food-Grade Colloidal Systems for the Delivery of Essential Oils. Food Rev. Int. 2021, 37, 1–45. [Google Scholar] [CrossRef]
- Chakravarthi, S.S.; Robinson, D.H. Pharmaceutical Manufacturing Handbook; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar] [CrossRef]
- Belščak-Cvitanović, A.; Jurić, S.; Đorđević, V.; Barišić, L.; Komes, D.; Ježek, D.; Bugarski, B.; Nedović, V. Chemometric evaluation of binary mixtures of alginate and polysaccharide biopolymers as carriers for microencapsulation of green tea polyphenols. Int. J. Food Prop. 2017, 20, 1971–1986. [Google Scholar] [CrossRef]
- Vinceković, M.; Jalšenjak, N.; Topolovec-Pintarić, S.; Dermić, E.; Bujan, M.; Jurić, S. Encapsulation of Biological and Chemical Agents for Plant Nutrition and Protection: Chitosan/Alginate Microcapsules Loaded with Copper Cations and Trichoderma viride. J. Agric. Food Chem. 2016, 64, 8073–8083. [Google Scholar] [CrossRef] [PubMed]
- Jurić, S.; Jurić, M.; Król-Kilińska, Ż.; Vlahoviček-Kahlina, K.; Vinceković, M.; Dragović-Uzelac, V.; Donsì, F. Sources, stability, encapsulation and application of natural pigments in foods. Food Rev. Int. 2020, 1–56. [Google Scholar] [CrossRef]
- Lozano-Vazquez, G.; Lobato-Calleros, C.; Escalona-Buendia, H.; Chavez, G.; Alvarez-Ramirez, J.; Vernon-Carter, E.J. Effect of the weight ratio of alginate-modified tapioca starch on the physicochemical properties and release kinetics of chlorogenic acid containing beads. Food Hydrocoll. 2015, 48, 301–311. [Google Scholar] [CrossRef]
- Jurić, S. Bioencapsulation as a Sustainable Delivery of Active Agents For plant Nutrition/Protection and Production of Functional Foods; University of Zagreb: Zagreb, Croatia, 2020. [Google Scholar]
- John, R.P.; Tyagi, R.D.; Brar, S.K.; Surampalli, R.Y.; Prévost, D. Bio-encapsulation of microbial cells for targeted agricultural delivery. Crit. Rev. Biotechnol. 2011, 31, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Mrkonjic Fuka, M.; Zgomba Maksimovic, A.; Hulak, N.; Kos, I.; Marusic Radovcic, N.; Juric, S.; Tanuwidjaja, I.; Karolyi, D.; Vincekovic, M. The survival rate and efficiency of non-encapsulated and encapsulated native starter cultures to improve the quality of artisanal game meat sausages. J. Food Sci. Technol. 2021, 58, 710–719. [Google Scholar] [CrossRef]
- Jurić, S.; Tanuwidjaja, I.; Mrkonjic Fuka, M.; Vlahoviček Kahlina, K.; Marijan, M.; Boras, A.; Udiković Kolić, N.; Vinceković, M. Encapsulation of two fermentation agents, Lactobacillus sakei and calcium ions in microspheres. Colloids Surfaces B Biointerfaces 2021, 197, 1–11. [Google Scholar] [CrossRef]
- Forde, A.; Fitzgerald, G.F. Biotechnological approaches to the understanding and improvement of mature cheese flavour. Curr. Opin. Biotechnol. 2000, 11, 484–489. [Google Scholar] [CrossRef]
- Azarnia, S.; Robert, N.; Lee, B. Biotechnological Methods to Accelerate Cheddar Cheese Ripening. Crit. Rev. Biotechnol. 2006, 26, 121–143. [Google Scholar] [CrossRef]
- Jahadi, M.; Khosravi-Darani, K.; Ehsani, M.R.; Mozafari, M.R.; Saboury, A.A.; Zoghi, A.; Mohammadi, M. Modelling of proteolysis in Iranian brined cheese using proteinase-loaded nanoliposome. Int. J. Dairy Technol. 2016, 69, 57–62. [Google Scholar] [CrossRef]
- Kheadr, E.E.; Vuillemard, J.-C.; El Deeb, S.A. Accelerated Cheddar cheese ripening with encapsulated proteinases. Int. J. Food Sci. Technol. 2000, 35, 483–495. [Google Scholar] [CrossRef]
- Anjani, K.; Kailasapathy, K.; Phillips, M. Microencapsulation of enzymes for potential application in acceleration of cheese ripening. Int. Dairy J. 2007, 17, 79–86. [Google Scholar] [CrossRef]
- Jahadi, M.; Khosravi-Darani, K.; Ehsani, M.R.; Mozafari, M.R.; Saboury, A.A.; Pourhosseini, P.S. The encapsulation of flavourzyme in nanoliposome by heating method. J. Food Sci. Technol. 2015, 52, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Kailasapathy, K.; Lam, S.H. Application of encapsulated enzymes to accelerate cheese ripening. Int. Dairy J. 2005, 15, 929–939. [Google Scholar] [CrossRef]
- Mohammadi, R.; Mahmoudzadeh, M.; Atefi, M.; Khosravi-Darani, K.; Mozafari, M.R. Applications of nanoliposomes in cheese technology. Int. J. Dairy Technol. 2015, 68, 11–23. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Santacruz, S.; Castro, M. Viability of free and encapsulated Lactobacillus acidophilus incorporated to cassava starch edible films and its application to Manaba fresh white cheese. LWT 2018, 93, 570–572. [Google Scholar] [CrossRef]
- Afzaal, M.; Saeed, F.; Ateeq, H.; Ahmed, A.; Ahmad, A.; Tufail, T.; Ismail, Z.; Anjum, F.M. Encapsulation of Bifidobacterium bifidum by internal gelation method to access the viability in cheddar cheese and under simulated gastrointestinal conditions. Food Sci. Nutr. 2020, 8, 2739–2747. [Google Scholar] [CrossRef]
- Ningtyas, D.W.; Bhandari, B.; Bansal, N.; Prakash, S. The viability of probiotic Lactobacillus rhamnosus (non-encapsulated and encapsulated) in functional reduced-fat cream cheese and its textural properties during storage. Food Control 2019, 100, 8–16. [Google Scholar] [CrossRef]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, Microspheres, and Microcapsules for Advanced Drug Delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef]
- Torri, L.; Piochi, M.; Lavelli, V.; Monteleone, E. Descriptive sensory analysis and consumers’ preference for dietary fibre- and polyphenol-enriched tomato purees obtained using winery by-products. LWT Food Sci. Technol. 2015, 62, 294–300. [Google Scholar] [CrossRef]
- Arce, A.; Ustunol, Z. Effect of microencapsulated ferrous sulfate particle size on Cheddar cheese composition and quality. J. Dairy Sci. 2018, 101, 6814–6822. [Google Scholar] [CrossRef] [PubMed]
- Jurić, S.; Ferrari, G.; Velikov, K.P.; Donsì, F. High-pressure homogenization treatment to recover bioactive compounds from tomato peels. J. Food Eng. 2019, 262, 170–180. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Ferrari, G.; Režek Jambrak, A.; Donsì, F. Lycopene-rich cream obtained via high-pressure homogenisation of tomato processing residues in a water–oil mixture. Int. J. Food Sci. Technol. 2021, 56, 4907–4914. [Google Scholar] [CrossRef]
- Narsaiah, K.; Sharma, M.; Sridhar, K.; Dikkala, P. Garlic Oil Nanoemulsions Hybridized in Calcium Alginate Microcapsules for Functional Bread. Agric. Res. 2019, 8, 356–363. [Google Scholar] [CrossRef]
- Soto, K.M.; Hernández-Iturriaga, M.; Loarca-Piña, G.; Luna-Bárcenas, G.; Mendoza, S. Antimicrobial effect of nisin electrospun amaranth: Pullulan nanofibers in apple juice and fresh cheese. Int. J. Food Microbiol. 2019, 295, 25–32. [Google Scholar] [CrossRef]
- El-Messery, T.M.; El-Said, M.M.; Farahat, E.S.A. Production of Functional Processed Cheese Supplemented with Nanoliposomes of Mandarin Peel Extract. Pakistan J. Biol. Sci. 2019, 22, 247–256. [Google Scholar] [CrossRef]
- Moghaddas Kia, E.; Alizadeh, M.; Esmaiili, M. Development and characterization of probiotic UF Feta cheese containing Lactobacillus paracasei microencapsulated by enzyme based gelation method. J. Food Sci. Technol. 2018, 55, 3657–3664. [Google Scholar] [CrossRef]
- Bedoya-Serna, C.M.; Dacanal, G.C.; Fernandes, A.M.; Pinho, S.C. Antifungal activity of nanoemulsions encapsulating oregano (Origanum vulgare) essential oil: In vitro study and application in Minas Padrão cheese. Brazilian J. Microbiol. 2018, 49, 929–935. [Google Scholar] [CrossRef]
- González Ariceaga, C.C.; Afzal, M.I.; Umer, M.; Shah, S.M.U.; Ahmad, H.; Jacquot, M.; Cailliez-Grimal, C. Effect of alginate beads on olfactory sensory perception of paraffin coated cheese. Czech J. Food Sci. 2018, 36, 255–260. [Google Scholar] [CrossRef]
- Liu, L.; Chen, P.; Zhao, W.; Li, X.; Wang, H.; Qu, X. Effect of microencapsulation with the Maillard reaction products of whey proteins and isomaltooligosaccharide on the survival rate of Lactobacillus rhamnosus in white brined cheese. Food Control 2017, 79, 44–49. [Google Scholar] [CrossRef]
- Rashidinejad, A.; Birch, E.J.; Sun-Waterhouse, D.; Everett, D.W. Delivery of green tea catechin and epigallocatechin gallate in liposomes incorporated into low-fat hard cheese. Food Chem. 2014, 156, 176–183. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).