3.1. Experimental Work: Methodology and Data Processing
All experimental tests were performed with half-calcined dolomite (HCD). The preferable dissolution of magnesium salts leads to pores opening and the ions inside the pores being more accessible. Therefore, the particle diameter and surface properties change during the process. A better magnesium salt dissolution causes calcium salts to become more accessible, which makes the Ca
2+/Mg
2+ ratio vary during the process [
30].
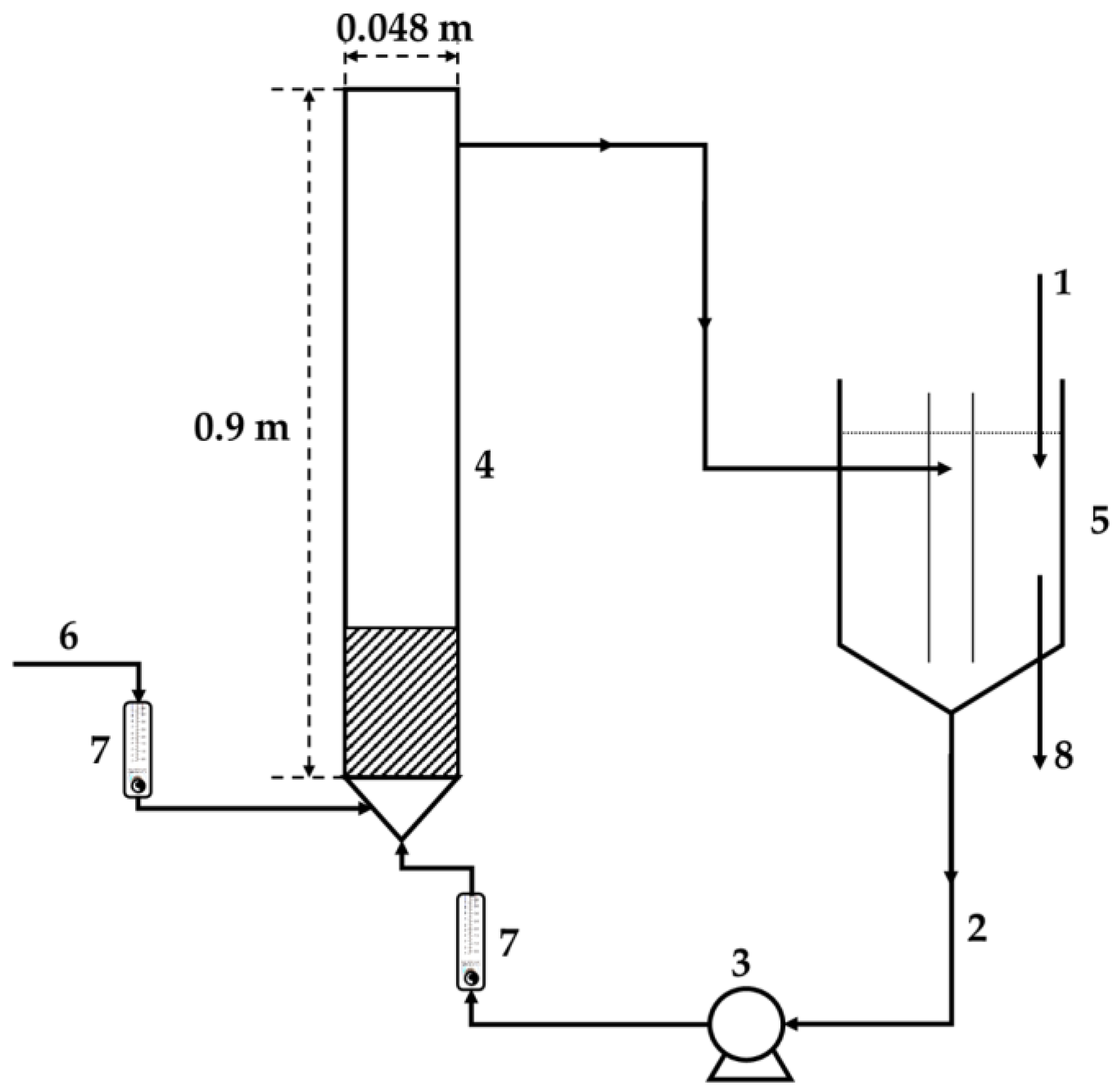
A batch reaction system is not practical for large volumes of treated liquids. A continuous system, either with a fixed bed or fluidized bed, was considered for large volumes of processed water. Preliminary tests indicated that a fixed bed of HCD and a single pass of water through the bed cannot ensure the required enrichment of drinking water by the biogenic elements. Considering the contact intensity of liquid and solids, a system with a fluidized bed and an internal circulation loop was proposed. The treated water was pumped from the water tank through the reactor and back (
Figure 1).
The water tank serves as a reservoir of the solution with the optimal salts concentration for the recarbonization process (the temperature, CO2 absorption, and water circulation flow rate), where fine particles eventually entrained from the fluidized bed can be separated. The overall residence time of water was determined by the circulation flow rate and the number of water cycles between these two pieces of equipment. It was assumed that water from the water tank is mixed with drinking water in the water pipeline in the required proportion.
It is generally acceptable that in a solid–liquid system, the smaller the solid particles (HCD), the faster the process [
13,
23]. However, the conditions for liquid–solid fluidized bed applications include particles of an adequate size, both in terms of the pressure drop in the HCD bed (and the associated cost of liquid pumping), solid material handling (the large fraction of dust in the solid phase), and material loss (entrainment) when fluid flows through the grain material (material loss and higher process costs).
The chemical reaction rate depends on the reactants’ concentration. A concentration of Mg
2+ as a function of time was measured in the system without soft water and a so-called saturation curve was obtained (
Figure 2). As it was mentioned above, Mg
2+ ions are released preferably, and this process affects the HCD particle properties. The second saturation test was done with the same HCD particles and fresh water. The data indicated the significant effect of the HCD particles “age” on the Mg
2+ concentration and thus must be taken into account in the design of the process and equipment for the long-term operation of water recarbonization systems. The relatively high reaction rate decreases with time and the Mg
2+ concentration for fresh HCD particles and used particles drops as well. Apparently, an optimum concentration of around 200 mg Mg
2+/L can be achieved at a reasonable saturation time and it indicates the required Mg
2+ concentration in drinking water in the interval from 12 to 30 mg/L, which can be achieved at the ratio of concentrate and fresh water of 1/10 [
30].
The Mg
2+ concentration obtained over the following 150 h of the experiment (
Figure 3) shows a significant change due to the changes in the main process parameters (the height of the fluidized bed (Lt)). At the time of 31 h, the CO
2 flow was discontinued, and the pH increased while the Mg
2+ release rate decreased. After the CO
2 flow was resumed, the Mg
2+ release rate returned to its original value. Additionally, HCD was introduced into the reactor and the Mg ion concentration rate increased to more than 300 mg/L [
30], as it was expected.
The results indicate a direct effect of the HCD amount in the system on the concentration of Mg2+ and Ca2+. However, the effect of the HCD age on the Mg2+ and Ca2+ concentration is also evident.
The reaction rate, generally, depends on the process conditions, especially the temperature, and on the concentration of products in the reaction mixture. Since the system temperature is defined by the temperature of the treated water and the energy delivered by a circulation pump, it is not easily affected. Therefore, the effect of the product concentration on the rate of the process was investigated at a temperature of approximately 25 °C. The results obtained in the laboratory system with an internal circulation are presented in
Figure 4. A higher increase in the Mg
2+ concentration at twice the HCD amount is a logical result initially caused by a low Mg
2+ concentration in water (below 200 mg/L). At higher concentrations, the Mg
2+ release rate decreases as it follows on from the reaction kinetics.
From the course of the dependencies, the similarity of the specific Mg
2+ release rates at different amounts of HCD is apparent, encouraging a possible process scale-up. Moreover, the Mg
2+ release rate from HCD decreases with the Mg
2+ concentration in the solution, indicating an optimal Mg
2+ concentration interval within which the Mg
2+ release rate is advantageous and a further increase in the solution concentration is not necessary. The optimal concentration interval of Mg
2+ ions is between 100 and 200 mg/L. The water tank volume also depends on its cost and the water flow to consumers. The appropriate water tank volume and design ensure that the water flow rates caused by the reactor solution feed and water from the water tank do not obstruct the particles settling [
30].
3.2. Recarbonization Process at Continuous Conditions
The characteristics of the continuous laboratory scale system selected for further experiments were as follows: a total volume of the system of 43 L, a water circulation rate of 6 L/min, a fluidized bed height of 21 cm, and a soft water inlet of 50 mL/min. The concentration of ions in the water tank was continuously monitored by measuring the solution conductivity, and the pH value and temperature in the system were also recorded.
Usual industrial applications employ filter-based flow devices capable of filtering the entire amount of water. Linear water flow velocities are limited by the filter size (diameter) as well as by the treated water flow rate. The direct introduction of treated water into the water distribution system requires the periodic operation of these devices due to the fluctuating consumption of water, which affects the flow through the filter. This also results in changing water recarbonization conditions. At low flow rates, the flow through the layer of solid filter material is uneven and the formation of channels occurs (lower resistance), which results in a decreased contact between the water and the filter surface. In addition, water remains in the solid material layer for a longer time at low flow rates and a local formation of concentrated solution can be expected, which contains insoluble products of dolomite decomposition and filter material sintering with precipitated calcium carbonate at high pH values. Therefore, the filters used in recarbonization processes require periodic washing and a large space and operation time.
Fluidization is an alternative method to fix bed gas–liquid–solid contact processes; here, solid particles of suitable size fluidize in the reaction column and a good mass transfer is ensured due to the large interface area between the solid and liquid phases. Volumetric reaction rates are significantly higher due to the large interface compared to conventional packed bed reaction systems. Additionally, the abrasion of a solid particle surface, caused by their intense movement, removes a part of the unreacted solid, and their surface is constantly renewed. In case of half-calcined dolomite, as the most suitable material, this phenomenon also leads to a change in the pore size during MgO dissolution and the internal surface of the particles is thus better accessible for water saturated with carbon dioxide. The change of the solid phase structure makes the “old surface” more brittle and easier to remove. The resulting large interfacial area of the fine particles of the solid phase improves the conditions for a mass transfer and increases the recarbonization rate. However, small particles from the layer can be entrained in the liquid and reactions outside of the main fluidized bed volume can occur.
A fluidized bed enables the intensification of some processes at the solid–liquid interface and various branches of the chemical industry, anaerobic and anoxic wastewater treatment processes, as well as drinking water denitrification process, employ this method. However, to the best of our knowledge, the chemical treatment of water or the recarbonization process have not utilized this reaction process. Parameters like the treatment of large flow rates of drinking water and the minimization of the recarbonization process investment costs identified a fluidized bed recarbonization reactor to be the best design option.
The designed process provides a concentrate of biogenic elements which is then mixed into drinking water supplied to consumers in suitable proportions based on the real rate of ion release into water and on the size of the device. The system for ion concentrates preparation is more stable concerning ions concentration and the elements required in drinking water are supplied more evenly [
30].
In the following experiments, Ca2+ and Mg2+ concentration was determined in the water circulation stream of the reactor and the water tank with CO2 fed to the reactor, below the distributor, and the storage tank. Thus, the Mg2+ concentration in the water tank, which is necessary for the tank volume minimization (constructional and static constraints at the installation site), was determined and a sufficient Mg2+ concentration in the water pumped into the water tank was ensured.
The hydrodynamics of fluidized bed reactors can be obtained in laboratory-scale systems if the particle size and pore size distribution of the particles is the same since these can significantly affect fluidization. In the case of drinking water recarbonization, fluidization characteristics are affected also by the CO2 flow rate which also affects the reaction kinetics as the reaction proceeds not only at the liquid–solid interface (dissolved CO2 reacts) but also at the solid–wet gas interface. The quantification of this type of kinetics is very complex and reliable experimental data in real process conditions are required for proper reactor design. To avoid these complications in the reactor design, an extensive and high-quality experimental study at the predicted process conditions is required.
The experimental set up for the process parameters determination consisted of a fluidized bed reactor, water storage tank, CO2 source, circulation pump, piping, and water and gas flowmeters. Soft water was introduced into the storage tank through a controlled rate and the outlet flow rate was equal to the inlet flow rate. Process parameters like the water circulation flow rate, CO2 flow rate, inlet and outlet flow rate of soft water, and concentrate flow rate as well as the temperature in the system and concentrations of biogenic ions were measured and controlled to obtain steady state (or quasi steady state) conditions in the system.
Long-term experiments were developed to verify the assumptions drawn from the laboratory observations. The following parameters were investigated: the mass of HCD which directly affects the bed height, its “average age” in the system, is affected by the rate of HCD addition, the water flow rate in the circulating line, the CO
2 flow rate, and the soft water flow rate to the system. Concentrations of Mg
2+ and Ca
2+ and the fluidized bed height were experimentally determined. The total operating time was approximately 1500 h. The results are presented in
Figure 5 [
30].
Results in
Figure 5 show opposite trends of Mg
2+ concentration and CO
2 flow in the predominant measurement period. Mg
2+ concentration features a certain delay compared to the fluidized bed height, probably due to the reaction time required to release Mg
2+ and Ca
2+ ions from the HCD matrix into treated water. The fluidized bed height values correlate well with the treated water flow rate, especially for longer reaction times. However, the treated drinking water flow rate is by about two orders of magnitude lower than the total water flow rate through the reactor (the internal water recirculation), which determines the fluidized bed hydrodynamics. The time dependence of the process performance on the soft water flow rate into the system helps to understand the effect of the periodic consumption of drinking water in real recarbonization systems.
Despite the stable soft water flow rate (which is relatively high per system volume), the relatively stable Mg concentration decreased with time with the decrease in the CO2 flow rate and the increase in the fluidized bed height due to the HCD addition to the reactor. A sharp increase in the HCD amount in the reactor (bed height of 33 cm) caused a fast concentration increase and then a gradual decrease despite an increased CO2 flow rate. When the soft water flow (the reactor feed) was discontinued, the Mg concentration increased up to 500 mg/L and decreased after the soft water flow was restored, even at higher CO2 flow rates.
Hence, the Mg
2+ concentration in the solution is significantly affected by the HCD amount, soft water flow rate, as well as by the CO
2 flow rate, in a lower extent. It can also be stated that the Mg
2+ concentration in the concentrate can be safely maintained in the concentration interval between 100 and 200 mg/L despite dramatic changes in process parameters [
31].
3.3. Characteristics of HCD Particles
The physical characteristics of the HCD particles change during the process, as it was mentioned above. The preferable release of Mg2+ ions from the HCD particles and the mutual contact of particles in the fluidized bed cause changes not only in particle size but also in internal particle structure. From this point of view, the “age“ of particles in the system is a very important parameter for the sustainable, long-term evolution of biogenic ions from HCD.
Particle size analysis (Mastersizer 2000) indicated a significant change in particle size distribution of fresh and used HCD [
31]. The results showed that the size and shape of the particles changes due to the consumption of the solid material and due to the abrasion of the material in the fluidized bed. Both effects improve the use of semi-calcined dolomite in the recarbonization process.
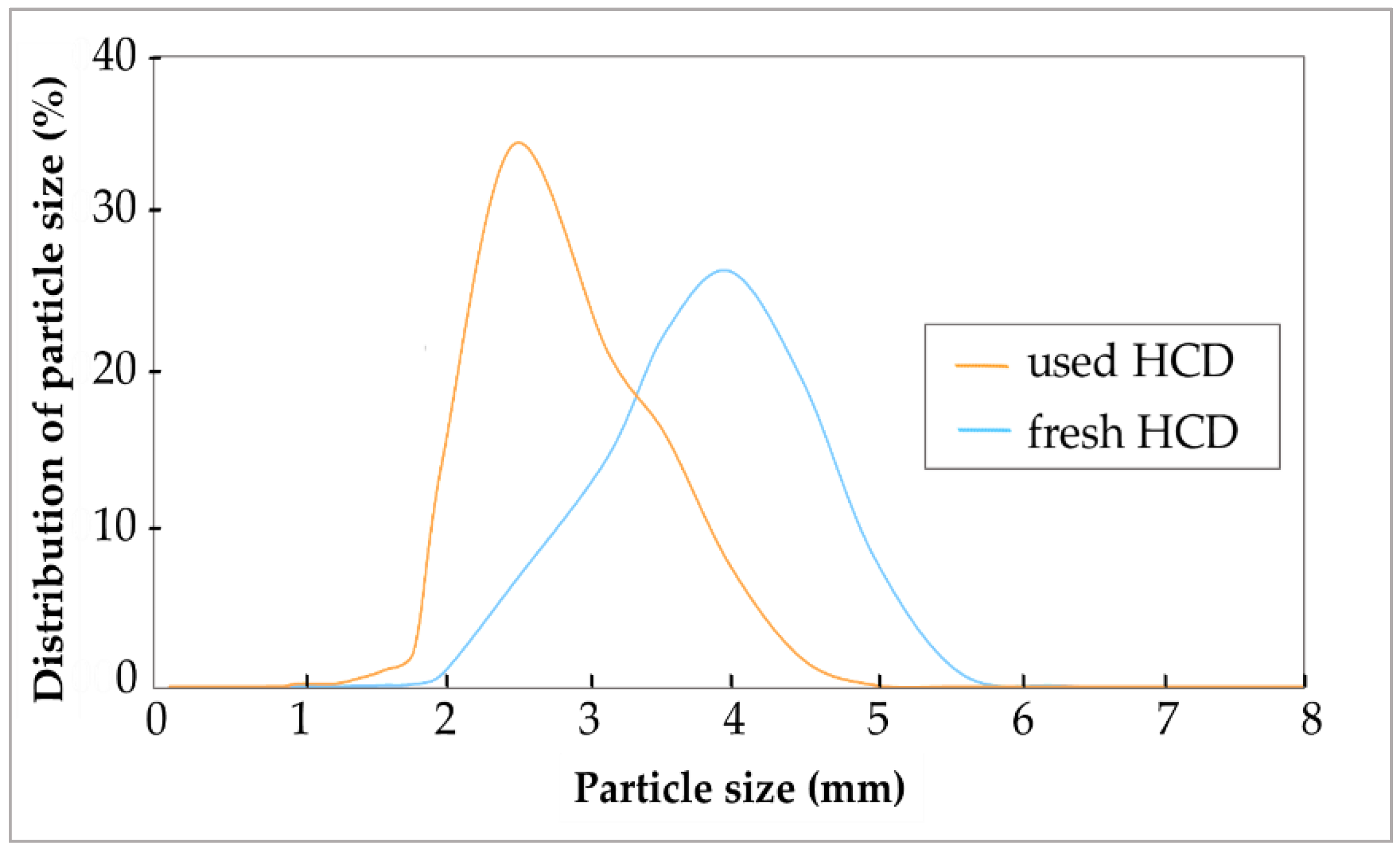
The experimental results presented in
Figure 6 and
Figure 7 indicate a significant effect of the recarbonization process on HCD particle size. Since HCD releases ions from their surface, the average size of the particles decreases and therefore both the cumulative and differential particle size distributions shift towards the smaller diameters. The release of ions from the particles makes them more brittle, therefore the particles change the diameter also by attrition since the particles are in intensive mutual contact in a fluidized bed. The presence of small particles formed due to attrition in the system which improved the water–solid particles contact, which in turn positively affects the effectiveness of the recarbonization process. On the other hand, very small particles can be entrained from the fluidized bed reactor that decreases the utilization of HCD for the process.
Changes in particle shape caused mainly by attrition are presented in
Figure 8. Fresh particles have sharp edges but, due to fluidization and the mutual contact of solids and HCD consumption, the particles lose their sharpness and they are rather rounded. These phenomena affect the fluidization characteristics of the HCD. The presented results indicate that the recarbonization process in a three-phase fluidized bed is very complex and it is difficult to predict the system behavior using the common methods. Therefore, long term experimentation at different process conditions is the only way how to design reliable equipment and sustainable process.
Fresh and used HCD particles internal characteristics were also analyzed using a porosimeter [
31] and the results are presented in
Table 1.
Pore size distribution of the fresh and used HCD is presented in
Figure 9 and
Figure 10. While fresh HCD has a peak on the radius in the interval from 20 to 30 nm, pore sizes of used HCD are more uniform, and they are in the interval from 1 to 100 nm. This clearly indicates that HCD particles subsequently release biogenic elements and change their properties, affecting thus not only the fluidized bed characteristics of HCD particles but also the ratio of released Mg
2+ and Ca
2+ elements. The longer the residence time (“age”) of particles in the bed, the lower the Mg
2+ and the higher Ca
2+ release intensity. These findings must be included in the reactor system design together with the amount and frequency of HCD addition into the reactor [
32].
3.4. Design and Scale-Up of Systems for Drinking Water Recarbonization
A water recarbonization process system was designed for the capacity of 11,000 m3/annum at the local water reservoir volume of 80 m3. The magnesium content in the water from the local well was approximately 5 mg/L and needs to be increased to a value from the interval of 12 to 20 mg/L. The water temperature was approximately 8–10 °C. Based on these data, a system comprising a reactor with a fluidized bed, water tank, and other equipment like a circulation pump, soft water pump, CO2 flow meter, control system, and other accessories was designed.
In the first step, it was necessary to define the amount of Mg2+ necessary and a reasonable HCD amount in the reactor to ensure a biogenic ions concentration of approximately 150–200 mg Mg2+/L in the water tank and around 12–15 mg/L in the water reservoir, considering the local periodic water consumption during a day.
The design and scale-up of the recarbonization system were based on the principles of geometrical similarity, hydrodynamic similarity, the inlet concentration of Mg
2+ ions, rate of production of Mg
2+ ions obtained at laboratory conditions for its concentration between 150 and 200 mg/L, specific CO
2 consumption, and average water consumption in the village. The dimensions of the main equipment were determined for the hydrodynamic conditions corresponding to conditions close to the minimum fluidization velocity for HCD particles of 2–4 mm, as follows [
31,
32]:
Diameter of reactor: 160 mm,
Bottom part length: 300 mm,
Main reactor part: 2400 mm,
Head of the reactor: 1000 mm,
Volume of water tank: 500 L,
Circulation pump flow rate: 3.6 m3/h,
Inlet/outlet water flow rate: 120 L/h.
This type of reactor system can safely process from 20 to 30 kg of HCD, which can guarantee the required enrichment of drinking water at the defined conditions of the real application of the system.
The diagram of the designed system is presented in
Figure 11 [
32].
The reactor system was constructed, installed, and tested at very well-controlled conditions in terms of the ions concentration in the inlet water, temperature in the system, circulation flow rate, and treated water flow rate for different amounts of HCD to determine the rate of ions release at the assumed conditions, robustness of the system in terms of the water enrichment with time, HCD consumption during the process, as well as the minimization of CO2 consumption during the process. All these parameters significantly affected the operational cost of the process as well as the operational time and effort needed. Continuous HCD addition into the system according to its consumption is an ideal process; however, in rural conditions, this approach is expensive, especially regarding the equipment for a small production.
The effects of the HCD amount in the system and the time on the biogenic ions concentration are presented in
Figure 12. The test started with 15 kg of HCD, and a high concentration of Mg
2+ and a low concentration of Ca
2+ ions were observed at the beginning. During the first 100 h when the cumulative amount of HCD increased to ca 20 kg, the ratio of Mg
2+ and Ca
2+ ions changed dramatically, indicating that the “average age of HCD” in the system is an important parameter and the periodic addition of HCD is required to maintain the reasonable enrichment of water by Mg
2+ ions. These results indicate that the data obtained in laboratory scale experiments are reproducible and the “age of HCD” particles is important, especially for the Mg
2+/Ca
2+ ratio determination. The results prove that, after approximately 300 h of operation of the system, the production rate of Mg
2+ and Ca
2+ is relatively stable and a further addition of HCD can shift the production towards Mg
2+ ions.
The inlet flow of soft water into the system increased in the time interval from 288 to 296 h with a similar effect to the drop in ions concentration and change in the Mg2+/Ca2+ concentration ratio.
The course of the ions concentration and their ratio was relatively stable, excluding the time when an insufficient amount of CO2 was temporarily introduced into the system (from 560 to 600 h). The course of the ions concentration and their ratio was relatively stable, excluding the time when an insufficient amount of CO2 was temporarily present in the system (from 560 to 600 h).
The results indicate that the conditions: a circulation rate of ca 3.6 m
3/h, an inlet water flow rate of 120 L/h, and a CO
2 flow rate of ca 180 g/h are acceptable to maintain the Mg
2+ concentration in the required interval if HCD is added into the system regularly. These results also indicate that periodic HCD addition is possible, and it is recommended to add 4 kg of HCD every four days to maintain a reasonable concentration of ions and the Mg
2+/Ca
2+ ratio at local conditions [
31].
Further experiments were done after a modification of the gas distributor to achieve a better gas distribution and a lower specific CO
2 consumption. The effect of the CO
2 flow rate on Mg ions production is presented in
Figure 13 at the same hydrodynamic and thermodynamic conditions and with an unchanged amount of HCD in the system (no addition).
At the above-mentioned conditions, the average Mg
2+ concentration for 350 h was 105 mg Mg
2+/L, the concentration of Ca was 77 mg/L, and the efficiency of the HCD utilization was 48% for both the magnesium and calcium ions [
31].
The water tank was plastic, as well as all of the tubing and piping. A stainless steel membrane circulation pump and glass rotameters were used in all of the experiments. To achieve the required enrichment of drinking water by biogenic ions, it is recommended to use from 25 to 30 kg of HCD in the reactor. Considering the HCD utilization efficiency, to release biogenic ions into water it is required to add of 4 kg of HCD every four days.
Specific consumption of required materials for recommended process conditions and drinking water consumption 11,000 m
3/annum is given in
Table 2.
The system presented in
Figure 11 was installed in a village with approximately 300 inhabitants with a local well and it has been in operation from June 2021, providing water enriched with Mg
2+ and Ca
2+ ions in the required concentrations. Based on this design and the experience obtained from the equipment operation, another recarbonization system with a ten times larger productivity was designed and put in operation in another place [
30,
31,
32].