Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction
2.3. Encapsulation
2.4. Colour
2.5. Water Activity
2.6. Morphological Analysis
2.7. Fourier Transform Infrared Spectroscopy (FTIR)
2.8. Total Phenolic Content
2.9. Total Monomeric Anthocyanins Content
2.10. Antioxidant Capacity
2.11. Thermal Properties
2.12. Storage Stability
2.13. Forced Storage Test
2.14. Statistical Analysis
3. Results
3.1. Colour
3.2. Water Activity
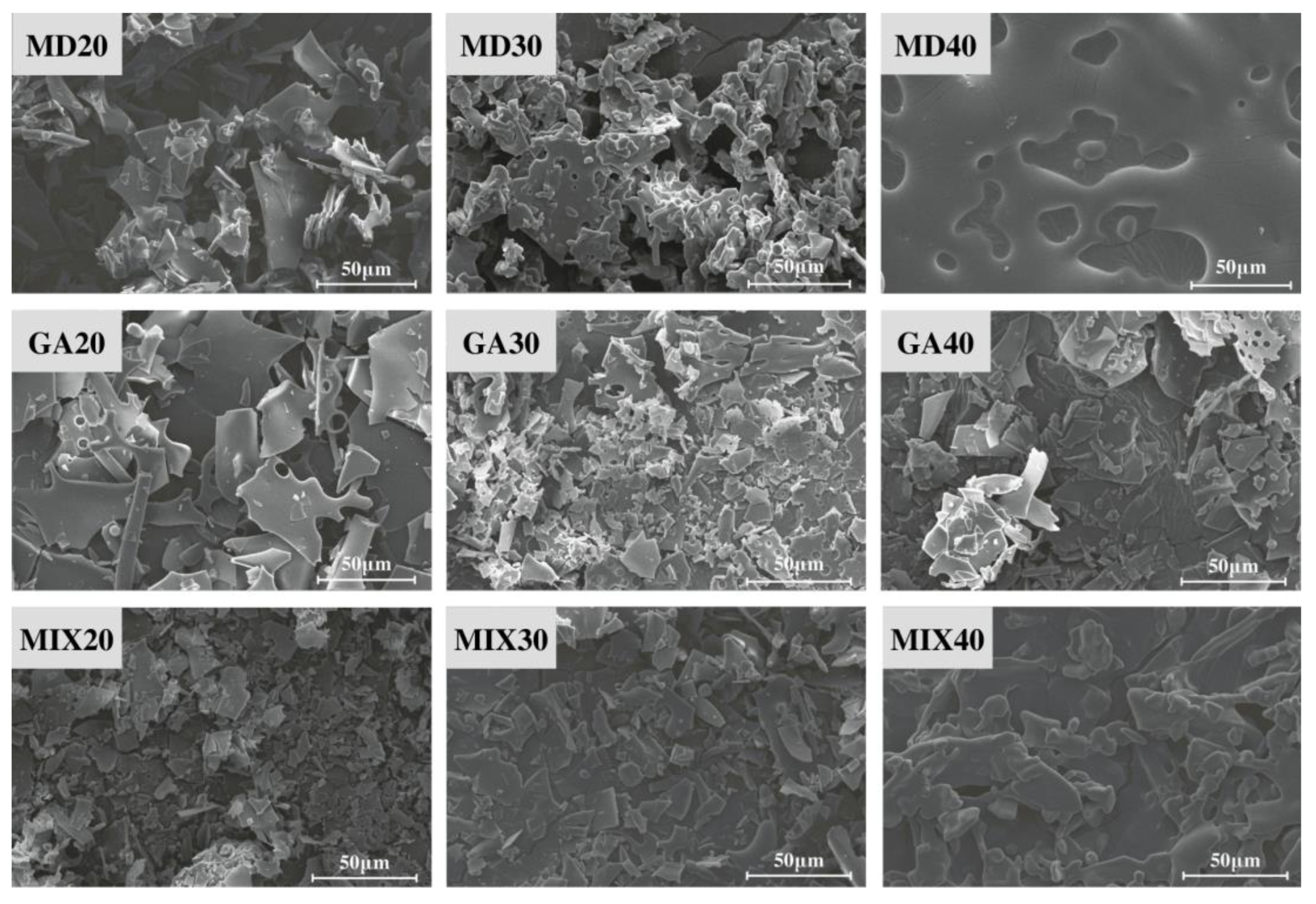
3.3. Morphological Analysis
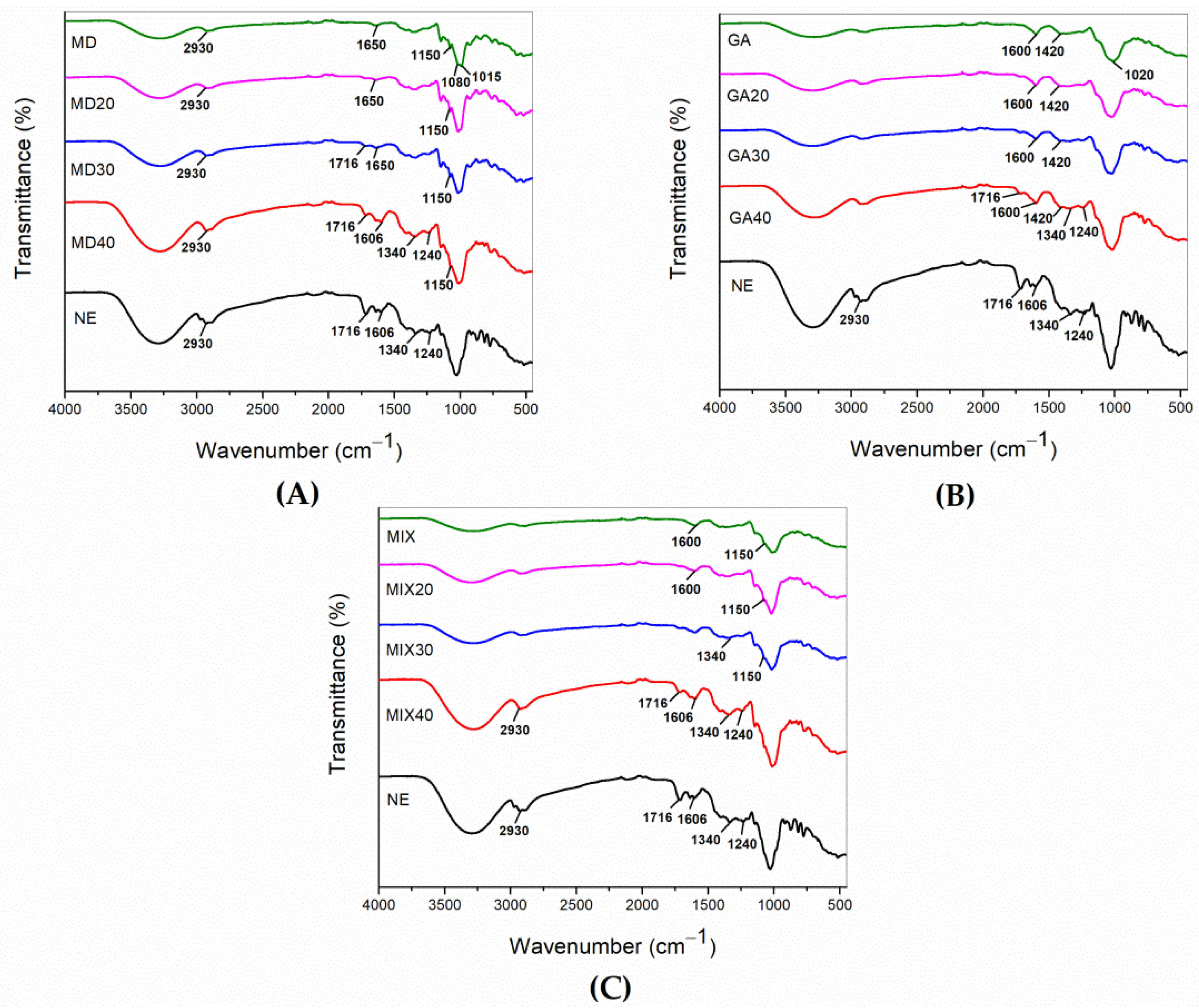
3.4. Fourier Transform Infrared Spectroscopy (FTIR)
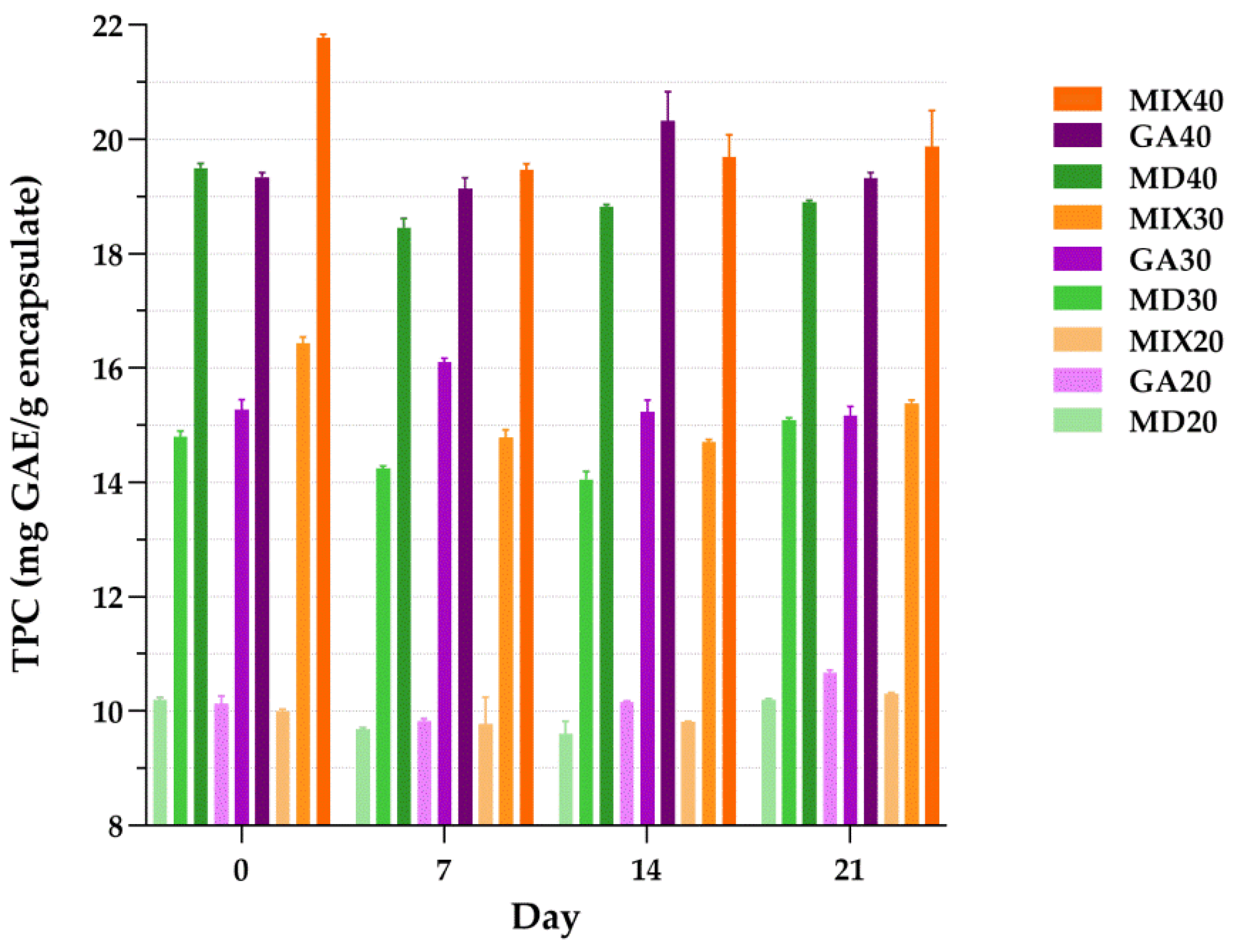
3.5. Total Phenolic Content
3.6. Total Monomeric Anthocyanins Content
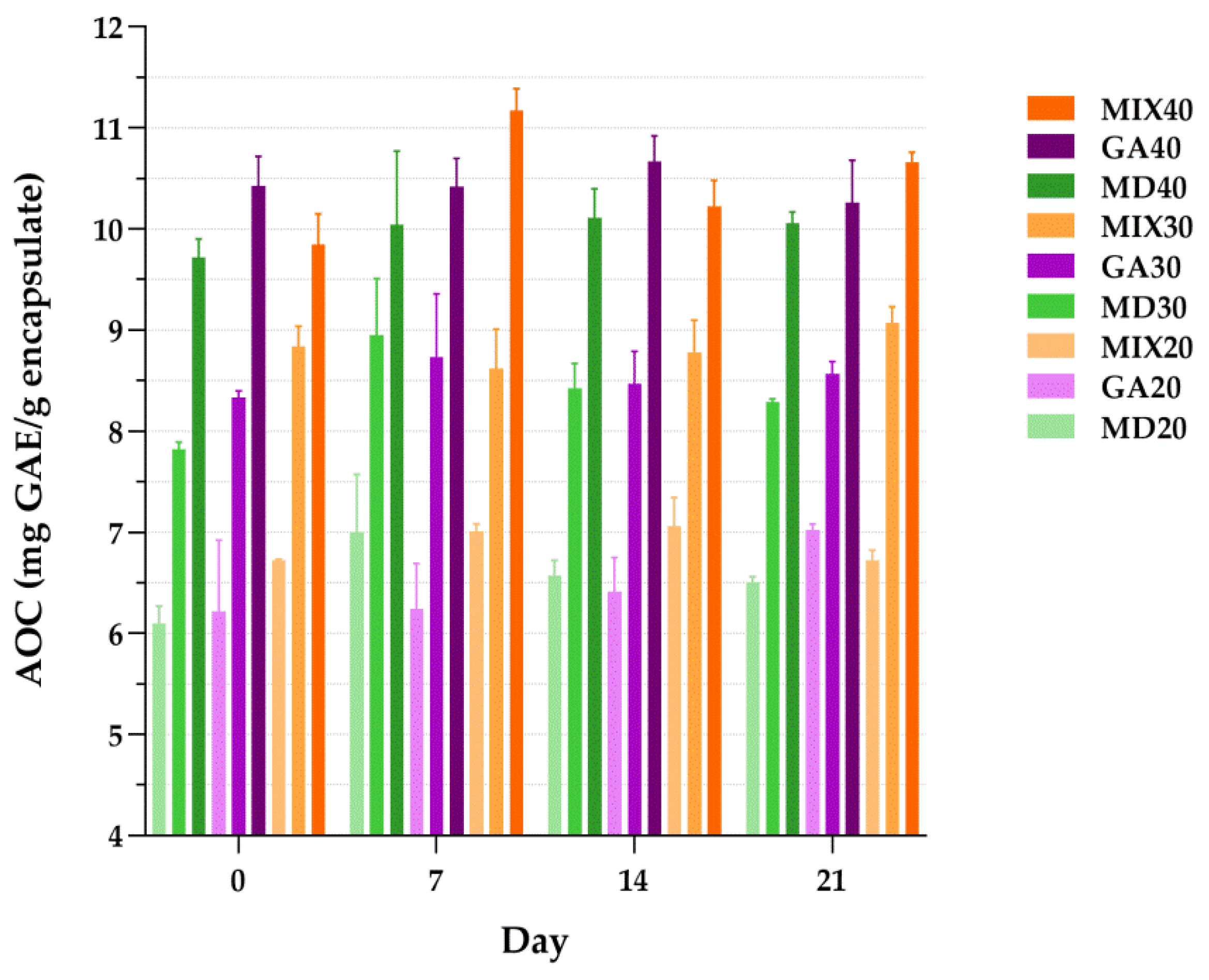
3.7. Antioxidant Capacity
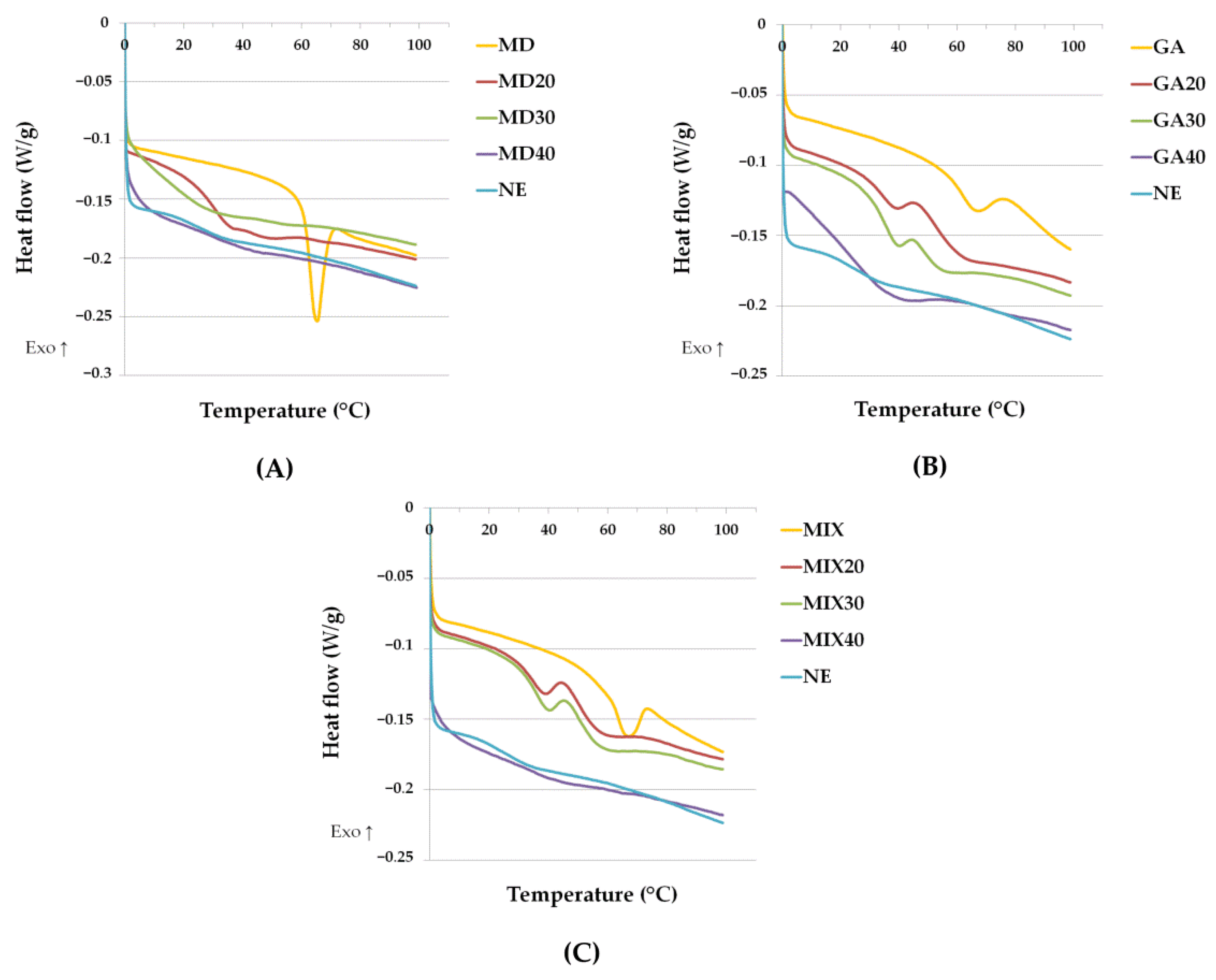
3.8. Thermal Properties
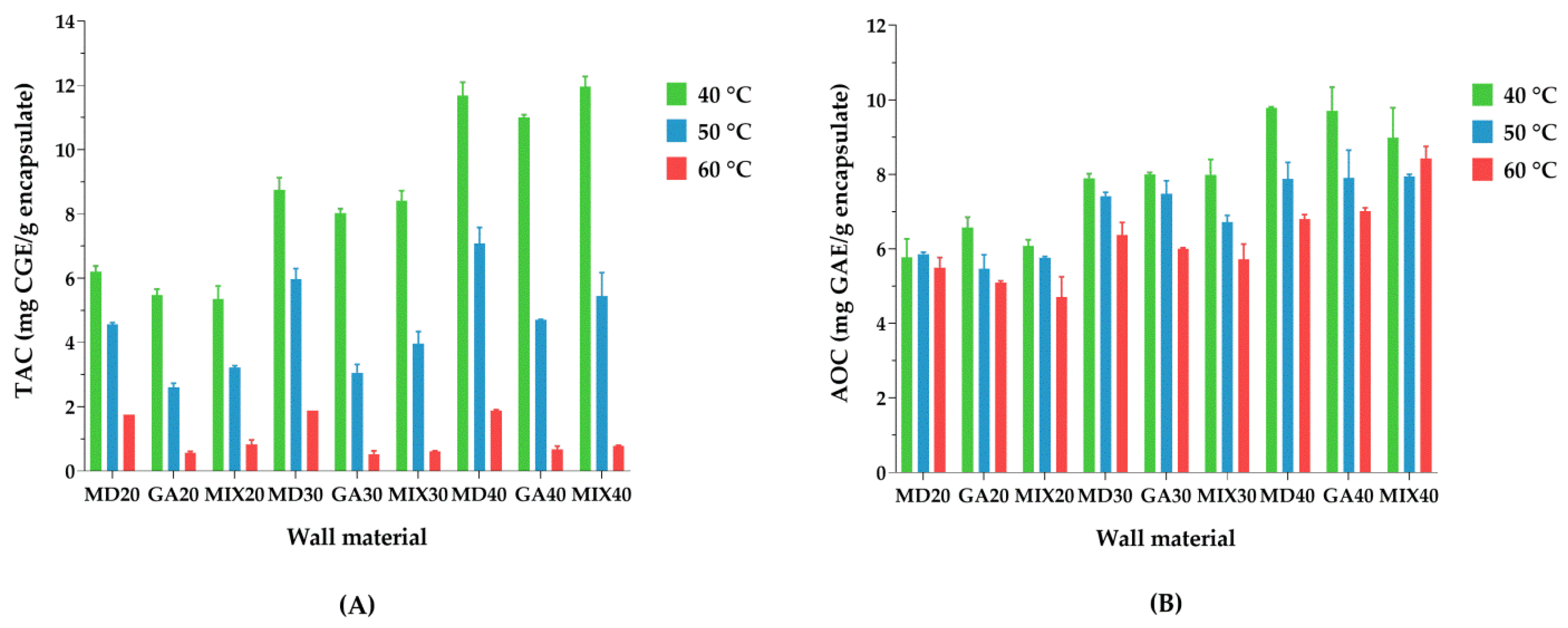
3.9. Forced Storage Test
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Menrad, K. Market and Marketing of Functional Food in Europe. J. Food Eng. 2003, 56, 181–188. [Google Scholar] [CrossRef]
- Daglia, M. Polyphenols as Antimicrobial Agents. Curr. Opin. Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jung, J.; Tomasino, E.; Zhao, Y. Optimization of Solvent and Ultrasound-Assisted Extraction for Different Anthocyanin Rich Fruit and Their Effects on Anthocyanin Compositions. LWT-Food Sci. Technol. 2016, 72, 229–238. [Google Scholar] [CrossRef]
- He, J.; Giusti, M.M. Anthocyanins: Natural Colorants with Health-Promoting Properties. Annu. Rev. Food Sci. Technol. 2010, 1, 163–187. [Google Scholar] [CrossRef]
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C.F.R. Food colorants: Challenges, Opportunities and Current Desires of Agro-Industries to Ensure Consumer Expectations and Regulatory Practices. Trends Food Sci. Technol. 2016, 52, 1–15. [Google Scholar] [CrossRef]
- Masone, D.; Chanforan, C. Study on the Interaction of Artificial and Natural Food Colorants with Human Serum Albumin: A Computational Point of View. Comput. Biol. Chem. 2015, 56, 152–158. [Google Scholar] [CrossRef]
- Padmanabhan, P.; Correa-Betanzo, J.; Paliyath, G. Berries and Related Fruits. In Encyclopedia of Food and Health, 1st ed.; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 364–371. [Google Scholar] [CrossRef]
- Pires, T.C.S.P.; Caleja, C.; Santos-Buelga, C.; Barros, L.; Ferreira, I.C.F.R. Vaccinium myrtillus L. Fruits as a Novel Source of Phenolic Compounds with Health Benefits and Industrial Applications—A Review. Curr. Pharm. Des. 2020, 26, 1917–1928. [Google Scholar] [CrossRef]
- De Mello, V.D.; Lankinen, M.A.; Lindström, J.; Puupponen-Pimiä, R.; Laaksonen, D.E.; Pihlajamäki, J.; Lehtonen, M.; Uusitupa, M.; Tuomilehto, J.; Kolehmainen, M.; et al. Fasting Serum Hippuric Acid is Elevated After Bilberry (Vaccinium myrtillus) Consumption and Associates with Improvement of Fasting Glucose Levels and Insulin Secretion in Persons at High Risk of Developing Type 2 Diabetes. Mol. Nutr. Food Res. 2017, 61, 1700019. [Google Scholar] [CrossRef]
- Erlund, I.; Koli, R.; Alfthan, G.; Marniemi, J.; Puukka, P.; Mustonen, P.; Mattila, P.; Jula, A. Favorable Effects of Berry Consumption on Platelet Function, Blood Pressure, and HDL Cholesterol. Am. J. Clin. Nutr. 2008, 87, 323–331. [Google Scholar] [CrossRef]
- Kolehmainen, M.; Mykkänen, O.; Kirjavainen, P.V.; Leppänen, T.; Moilanen, E.; Adriaens, M.; Laaksonen, D.E.; Hallikainen, M.; Puupponen-Pimiä, R.; Pulkkinen, L.; et al. Bilberries Reduce Low-Grade Inflammation in Individuals with Features of Metabolic Syndrome. Mol. Nutr. Food Res. 2012, 56, 1501–1510. [Google Scholar] [CrossRef]
- Li, J.; Zhao, R.; Jiang, Y.; Xu, Y.; Zhao, H.; Lyu, X.; Wu, T. Bilberry Anthocyanin Improves the Neuroinflammation and Cognitive Disfunctions in APP/PSEN1 Mice via the CD33/TREM2/TYROBP Signaling Pathway in Microglia. Food Funct. 2020, 11, 1572–1584. [Google Scholar] [CrossRef] [PubMed]
- Schantz, M.; Mohn, C.; Baum, M.; Richling, E. Antioxidative Efficiency of an Anthocyanin Rich Bilberry Extract in the Human Colon Tumor Cell Lines Caco-2 and HT-29. J. Berry Res. 2010, 1, 25–33. [Google Scholar] [CrossRef]
- Muhamad, I.I.; Jusoh, Y.M.M.; Nawi, N.M.; Aziz, A.A.; Padzil, A.M.; Lian, H.L. Chapter 15—Advanced Natural Food Colorant Encapsulation Methods: Anthocyanin Plant Pigment. In Natural and Artificial Flavoring Agents and Food Dyes, Handbook of Food Bioengineering, 1st ed.; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; Volume 7, pp. 495–526. [Google Scholar] [CrossRef]
- Nedović, V.; Kalušević, A.; Manojlović, V.; Petrović, T.; Bugarski, B. Encapsulation Systems in the Food Industry. In Advances in Food Process Engineering Research and Applications, 1st ed.; Yanniotis, S., Taoukis, P., Stoforos, N.G., Karathanos, V.T., Eds.; Springer: New York, NY, USA, 2013; pp. 229–253. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Encapsulation of Polyphenols—A Review. Trends Food Sci. Technol. 2010, 21, 510–523. [Google Scholar] [CrossRef]
- Baldelli, A.; Wells, S.; Pratap-Singh, A. Impact of Product Formulation on Spray-Dried Microencapsulated Zinc for Food Fortification. Food Bioproc. Technol. 2021, 14, 2286–2301. [Google Scholar] [CrossRef]
- Baldelli, A.; Liang, D.Y.; Guo, Y.; Pratap-Singh, A. Effect of the Formulation on Mucoadhesive Spray-Dried Microparticles Containing Iron for Food Fortification. Food Hydrocoll. 2023, 134, 107906. [Google Scholar] [CrossRef]
- Rezvankhah, A.; Emam-Djomeh, Z.; Askari, G. Encapsulation and Delivery of Bioactive Compounds Using Spray and Freeze-Drying Techniques: A Review. Dry Technol. 2020, 38, 235–258. [Google Scholar] [CrossRef]
- Arueya, G.L.; Akomolafe, B.O. Stability Studies of Microencapsulated Anthocyanins of Roselle (Hibiscus Sabdariffa L) in Native Starch and Its Potential Application in Jam Production. IOSR J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 112–122. [Google Scholar] [CrossRef]
- Baretto, F.J.F.P.; Clemente, H.A.; Santana, A.L.B.D.; Vasconcelo, M.A.S. Stability of Encapsulated and Non-Encapsulated Anthocyanin in Yogurt Produced with Natural Dye Obtained from Solanum melongena L. Bark. Rev. Bras. Frutic. 2020, 42, e-137. [Google Scholar] [CrossRef]
- Çam, M.; İçyer, N.C.; Erdoğan, F. Pomegranate peel phenolics: Microencapsulation, Storage Stability and Potential Ingredient for Functional Food Development. LWT-Food Sci. Technol. 2014, 55, 117–123. [Google Scholar] [CrossRef]
- Sarabandi, K.; Jafari, S.M.; Mahoonak, A.S.; Mohammadi, A. Application of Gum Arabic and Maltodextrin for Encapsulation of Eggplant Peel Extract as a Natural Antioxidant and Color Source. Int. J. Biol. Macromol. 2019, 140, 59–68. [Google Scholar] [CrossRef]
- Estupiñan-Amaya, M.; Fuenmayor, C.A.; López-Córdoba, A. New Freeze-Dried Andean Blueberry Juice Powders for Potential Application as Functional Food Ingredients: Effect of Maltodextrin on Bioactive and Morphological Features. Molecules 2020, 25, 5635. [Google Scholar] [CrossRef] [PubMed]
- Pudziuvelyte, L.; Marksa, M.; Sosnowska, K.; Winnicka, K.; Morkuniene, R.; Bernatoniene, J. Freeze-Drying Technique for Microencapsulation of Elsholtzia ciliata Ethanolic Extract Using Different Coating Materials. Molecules 2020, 25, 2237. [Google Scholar] [CrossRef] [PubMed]
- Eun, J.B.; Maruf, A.; Das, P.R.; Nam, S.H. A Review of Encapsulation of Carotenoids Using Spray Drying and Freeze Drying. Crit. Rev. Food Sci. Nutr. 2020, 60, 3547–3572. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, V.; Roos, Y.H. Limonene Encapsulation in Freeze-Drying of Gum Arabic–Sucrose–Gelatin Systems. LWT-Food Sci. Technol. 2007, 40, 1381–1391. [Google Scholar] [CrossRef]
- Martins, W.D.S.; de Arajo, J.S.F.; Feitosa, B.F.; Oliveira, J.R.; Kotzebue, L.R.V.; Agostini, D.L.D.S.; de Oliveira, D.L.V.; Mazzetto, S.E.; Cavalcanti, M.T.; da Silva, A.L. Lemongrass (Cymbopogon citratus DC. Stapf) Essential Oil Microparticles: Development, Characterization, and Antioxidant Potential. Food Chem. 2021, 355, 129644. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.; Howes, T.; Bhandari, B.; Troung, V. Effect of Addition of Maltodextrin on Drying Kinetics and Stickiness of Sugar and Acid-Rich Foods during Convective Drying: Experiments and Modelling. J. Food Eng. 2004, 62, 53–68. [Google Scholar] [CrossRef]
- Mosquera, L.H.; Moraga, G.; Martínez-Navarrete, N. Effect of Maltodextrin on the Stability of Freeze-Dried Borojó (Borojoa patinoi Cuatrec.) Powder. J. Food Eng. 2010, 97, 72–78. [Google Scholar] [CrossRef]
- Idham, Z.; Muhamad, I.I.; Sarmidi, M.R. Degradation Kinetics and Color Stability of Spray-Dried Encapsulated Anthocyanins from Hibiscus Sabdariffa L. J. Food Process Eng. 2011, 35, 522–542. [Google Scholar] [CrossRef]
- Wandrey, C.; Bartkowiak, A.; Harding, E.S. Materials for Encapsulation. In Encapsulation Technologies for Food Active Ingredients and Food Processing, 1st ed.; Zuidam, N.J., Nedovic, V.A., Eds.; Springer: New York, NY, USA, 2009; pp. 31–100. [Google Scholar] [CrossRef]
- Može, Š.; Polak, T.; Gašperlin, L.; Koron, D.; Vanzo, A.; Ulrih, N.P.; Abram, V. Phenolics in Slovenian Bilberries (Vaccinium myrtillus L.) and Blueberries (Vaccinium corymbosum L.). J. Agric. Food Chem. 2011, 59, 6998–7004. [Google Scholar] [CrossRef]
- Terpinc, P.; Čeh, B.; Ulrih, N.P.; Abramovič, H. Studies of the Correlation between Antioxidant Properties and the Total Phenolic Content of Different Oil Cake Extracts. Ind. Crops Prod. 2012, 39, 210–217. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of Total Monomeric Anthocyanin Pigment Content of Fruit Juices, Beverages, Natural Colorants, and Wines by the pH Differential Method: Collaborative Study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Abramovič, H.; Grobin, B.; Ulrih, N.P.; Cigić, B. The Methodology Applied in DPPH, ABTS and Folin-Ciocalteau Assays Has a Large Influence on the Determined Antioxidant Potential. Acta Chim. Slov. 2017, 64, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Kalušević, A.; Lević, S.; Čalija, B.; Pantić, M.; Belović, M.; Pavlović, V.; Bugarski, B.; Milić, J.; Žilić, S.; Nedović, V. Microencapsulation of Anthocyanin-Rich Black Soybean Coat Extract by Spray Drying Using Maltodextrin, Gum Arabic and Skimmed Milk Powder. J. Microencapsul. 2017, 34, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, G.I.N.; Sun-Waterhouse, D.; Su, G.; Zhao, H.; Zhao, M. Spray-Drying of Antioxidant-Rich Blueberry Waste Extracts; Interplay between Waste Pretreatments and Spray-Drying Process. Food Bioprocess Technol. 2017, 10, 1074–1092. [Google Scholar] [CrossRef]
- Baeza, R.; Sánchez, V.; Salierno, G.; Molinari, F.; López, P.; Chirife, J. Storage Stability of Anthocyanins in Freeze-Dried Elderberry Pulp using Low Proportions of Encapsulating Agents. Food Sci. Technol. Int. 2020, 27, 135–144. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Anthocyanin Stability and Antioxidant Activity of Spray-Dried Açai (Euterpe oleracea Mart.) Juice Produced with Different Carrier Agents. Food Res. Int. 2010, 43, 907–914. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Indrani, D.; Jena, B.S.; Anandharamakrishnan, C. Freeze Drying Technique for Microencapsulation of Garcinia Fruit Extract and Its Effect on Bread Quality. J. Food Eng. 2013, 117, 513–520. [Google Scholar] [CrossRef]
- Mazuco, R.A.; Cardoso, P.M.M.; Bindaco, É.S.; Scherer, R.; Castilho, R.O.; Faraco, A.A.G.; Ruas, F.G.; Oliveira, J.P.; Guimarães, M.C.C.; de Andrade, T.U.; et al. Maltodextrin and Gum Arabic-Based Microencapsulation Methods for Anthocyanin Preservation in Juçara Palm (Euterpe edulis Martius) Fruit Pulp. Plant Foods Hum. Nutr. 2018, 73, 209–215. [Google Scholar] [CrossRef]
- Šturm, L.; Črnivec, I.G.O.; Istenič, K.; Ota, A.; Megušar, P.; Slukan, A.; Humar, M.; Lević, S.; Nedović, V.; Kopinč, R.; et al. Encapsulation of Non-Dewaxed Propolis by Freeze-Drying and Spray-Drying Using Gum Arabic, Maltodextrin and Inulin as Coating Materials. Food Bioprod. Process. 2019, 116, 196–211. [Google Scholar] [CrossRef]
- He, J.; Rodriguez-Saona, L.E.; Giusti, M.M. Midinfrared Spectroscopy for Juice Authentication—Rapid Differentiation of Commercial Juices. J. Agric. Food Chem. 2007, 55, 4443–4452. [Google Scholar] [CrossRef]
- Pereira, V.A.; de Arruda, I.N.Q.; Stefani, R. Active chitosan/PVA Films with Anthocyanins from Brassica oleraceae (Red Cabbage) as Time–Temperature Indicators for Application in Intelligent Food Packaging. Food Hydrocoll. 2015, 43, 180–188. [Google Scholar] [CrossRef]
- Krishnaiah, D.; Sarbatly, R.; Nithyanandam, R. Microencapsulation of Morinda citrifolia L. Extract by Spray-Drying. Chem. Eng. Res. Des. 2012, 90, 622–632. [Google Scholar] [CrossRef]
- Cai, C.; Ma, R.; Duan, M.; Lu, D. Preparation and Antimicrobial Activity of Thyme Essential Oil Microcapsules Prepared with Gum Arabic. RSC Adv. 2019, 9, 19740–19747. [Google Scholar] [CrossRef] [PubMed]
- Kozarski, M.; Klaus, A.; Nikšić, M.P.; Vrvić, M.M.; Todorović, N.; Jakovljević, D.V.; Griensven, L.J. Antioxidative Activities and Chemical Characterization of Polysaccharide Extracts from the Widely Used Mushrooms Ganoderma applanatum, Ganoderma lucidum, Lentinus edodes and Trametes versicolor. J. Food Compos. Anal. 2012, 26, 144–153. [Google Scholar] [CrossRef]
- Arancibia-Avila, P.; Namiesnik, J.; Toledo, F.; Werner, E.; Martinez-Ayala, A.L.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Gorinstein, S. The Influence of Different Time Durations of Thermal Processing on Berries Quality. Food Control 2012, 26, 587–593. [Google Scholar] [CrossRef]
- Arbeláez, A.F.A.; Pérez, E.D.; López-Alarcón, C.; Cortés, F.B.; Rojano, B.A. Immobilization of Andean Berry (Vaccinium meridionale) Polyphenols on Nanocellulose Isolated from Banana Residues: A Natural Food Additive with Antioxidant Properties. Food Chem. 2019, 294, 503–517. [Google Scholar] [CrossRef]
- Zhang, C.; Khoo, S.L.A.; Swedlund, P.; Ogawa, Y.; Shan, Y.; Quek, S.Y. Fabrication of Spray-Dried Microcapsules Containing Noni Juice Using Blends of Maltodextrin and Gum Acacia: Physicochemical Properties of Powders and Bioaccessibility of Bioactives during In Vitro Digestion. Foods 2020, 9, 1316. [Google Scholar] [CrossRef]
- Casati, C.B.; Baeza, R.; Sánchez, V. Physicochemical Properties and Bioactive Compounds Content in Encapsulated Freeze-Dried Powders Obtained from Blueberry, Elderberry, Blackcurrant and Maqui Berry. J. Berry Res. 2019, 9, 431–447. [Google Scholar] [CrossRef]
- Giovanelli, G.; Buratti, S. Comparison of Polyphenolic Composition and Antioxidant Activity of Wild Italian Blueberries and Some Cultivated Varieties. Food Chem. 2009, 112, 903–908. [Google Scholar] [CrossRef]
- Castro-López, C.; Sánchez-Alejo, E.J.; Saucedo-Pompa, S.; Rojas, R.; Aranda-Ruiz, J.; Martínez-Avila, G.C.G. Fluctuations in Phenolic Content, Ascorbic Acid and Total Carotenoids and Antioxidant Activity of Fruit Beverages During Storage. Heliyon 2016, 2, e00152. [Google Scholar] [CrossRef]
- Piljac-Žegarac, J.; Valek, L.; Martinez, S.; Belščak, A. Fluctuations in the Phenolic Content and Antioxidant Capacity of Dark Fruit Juices in Refrigerated Storage. Food Chem. 2009, 113, 394–400. [Google Scholar] [CrossRef]
- Riihinen, K.; Jaakola, L.; Kärenlampi, S.; Hohtola, A. Organ-specific Specific Distribution of Phenolic Compounds in Bilberry (Vaccinium myrtillus) and ‘Northblue’ Blueberry (Vaccinium corymbosum × V. angustifolium). Food Chem. 2008, 110, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Akhavan Mahdavi, S.; Jafari, S.M.; Assadpour, E.; Ghorbani, M. Storage Stability of Encapsulated Barberry’s Anthocyanin and its Application in Jelly Formulation. J. Food Eng. 2016, 18, 59–66. [Google Scholar] [CrossRef]
- Ersus, S.; Yurdagel, U. Microencapsulation of Anthocyanin Pigments of Black Carrot (Daucuscarota L.) by Spray Drier. J. Food Eng. 2007, 80, 805–812. [Google Scholar] [CrossRef]
- Fischer, U.A.; Carle, R.; Kammerer, D.R. Thermal Stability of Anthocyanins and Colourless Phenolics in Pomegranate (Punica granatum L.) Juices and Model Solutions. Food Chem. 2013, 138, 1800–1809. [Google Scholar] [CrossRef]
- Pinelo, M.; Manzocco, L.; Nuñez, M.J.; Nicoli, M.C. Interaction among Phenols in Food Fortification: Negative Synergism on Antioxidant Capacity. J. Agric. Food Chem. 2004, 52, 1177–1180. [Google Scholar] [CrossRef]
- Mochizuki, T.; Alvino Granados, A.E.; Sogabe, T.; Kawai, K. Effects of Glass Transition, Operating Process, and Crystalline Additives on the Hardness of Thermally Compressed Maltodextrin. Food Eng. Rev. 2020, 13, 215–224. [Google Scholar] [CrossRef]
- Kurozawa, L.E.; Park, K.J.; Hubinger, M.D. Effect of Maltodextrin and Gum Arabic on Water Sorption and Glass Transition Temperature of Spray Dried Chicken Meat Hydrolysate Protein. J. Food Eng. 2009, 91, 287–296. [Google Scholar] [CrossRef]
- Rutz, J.K.; Borges, C.D.; Zambiazi, R.C.; da Rosa, C.G.; da Silva, M.M. Elaboration of Microparticles of Carotenoids from Natural and Synthetic Sources for Applications in Food. Food Chem. 2016, 202, 324–333. [Google Scholar] [CrossRef]
- Jiménez-Aguilar, D.M.; Ortega-Regules, A.E.; Lozada-Ramírez, J.D.; Pérez-Pérez, M.C.I.; Vernon-Carter, E.J.; Welti-Chanes, J. Color and Chemical Stability of Spray-Dried Blueberry Extract Using Mesquite Gum as Wall Material. J. Food Compos. Anal. 2011, 24, 889–894. [Google Scholar] [CrossRef]
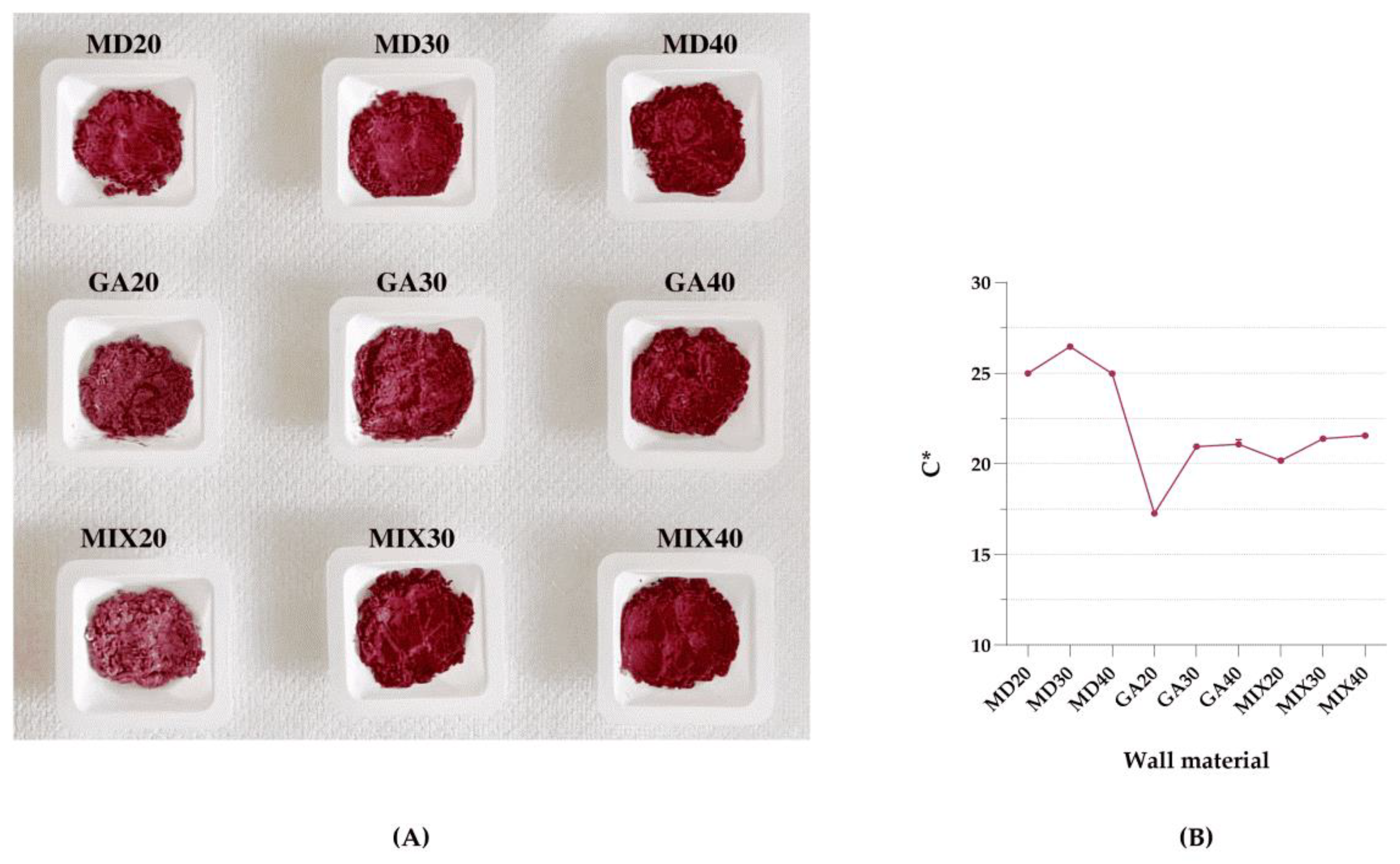

| Wall Material | aw | L* | a* | b* | h° | C* |
|---|---|---|---|---|---|---|
| MD20 | 0.22 ± 0.00 a | 51.01 ± 0.08 a | 23.87 ± 0.07 a | 7.41 ± 0.06 a | 0.30 ± 0.00 a | 24.99 ± 0.09 a |
| MD30 | 0.29 ± 0.00 b | 51.25 ± 0.21 a | 25.24 ± 0.05 b | 7.94 ± 0.02 b | 0.30 ± 0.00 ab | 26.46 ± 0.05 b |
| MD40 | 0.23 ± 0.00 c | 46.44 ± 0.14 b | 23.78 ± 0.01 a | 7.63 ± 0.14 c | 0.31 ± 0.01 b | 24.97 ± 0.03 a |
| GA20 | 0.18 ± 0.00 d | 52.21 ± 0.44 a | 17.11 ± 0.08 c | 2.38 ± 0.05 d | 0.14 ± 0.00 c | 17.27 ± 0.08 c |
| GA30 | 0.28 ± 0.00 e | 51.74 ± 0.37 a | 20.22 ± 0.09 d | 5.46 ± 0.05 e | 0.26 ± 0.00 d | 20.95 ± 0.10 d |
| GA40 | 0.30 ± 0.00 f | 47.11 ± 1.20 b | 20.11 ± 0.24 d | 6.28 ± 0.11 f | 0.30 ± 0.00 a | 21.07 ± 0.26 df |
| MIX20 | 0.26 ± 0.00 g | 59.61 ± 0.23 c | 19.65 ± 0.09 e | 4.54 ± 0.06 g | 0.23 ± 0.00 e | 20.17 ± 0.08 e |
| MIX30 | 0.17 ± 0.00 h | 46.99 ± 0.17 b | 20.31 ± 0.07 d | 6.71 ± 0.01 h | 0.32 ± 0.00 f | 21.39 ± 0.07 fg |
| MIX40 | 0.17 ± 0.00 h | 45.79 ± 0.83 b | 20.28 ± 0.15 d | 7.29 ± 0.03 a | 0.35 ± 0.00 g | 21.55 ± 0.15 g |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Todorović, A.; Šturm, L.; Salević-Jelić, A.; Lević, S.; Osojnik Črnivec, I.G.; Prislan, I.; Skrt, M.; Bjeković, A.; Poklar Ulrih, N.; Nedović, V. Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability. Processes 2022, 10, 1991. https://doi.org/10.3390/pr10101991
Todorović A, Šturm L, Salević-Jelić A, Lević S, Osojnik Črnivec IG, Prislan I, Skrt M, Bjeković A, Poklar Ulrih N, Nedović V. Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability. Processes. 2022; 10(10):1991. https://doi.org/10.3390/pr10101991
Chicago/Turabian StyleTodorović, Ana, Luka Šturm, Ana Salević-Jelić, Steva Lević, Ilja Gasan Osojnik Črnivec, Iztok Prislan, Mihaela Skrt, Ana Bjeković, Nataša Poklar Ulrih, and Viktor Nedović. 2022. "Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability" Processes 10, no. 10: 1991. https://doi.org/10.3390/pr10101991
APA StyleTodorović, A., Šturm, L., Salević-Jelić, A., Lević, S., Osojnik Črnivec, I. G., Prislan, I., Skrt, M., Bjeković, A., Poklar Ulrih, N., & Nedović, V. (2022). Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability. Processes, 10(10), 1991. https://doi.org/10.3390/pr10101991








