The Association between ADHD and Celiac Disease in Children
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
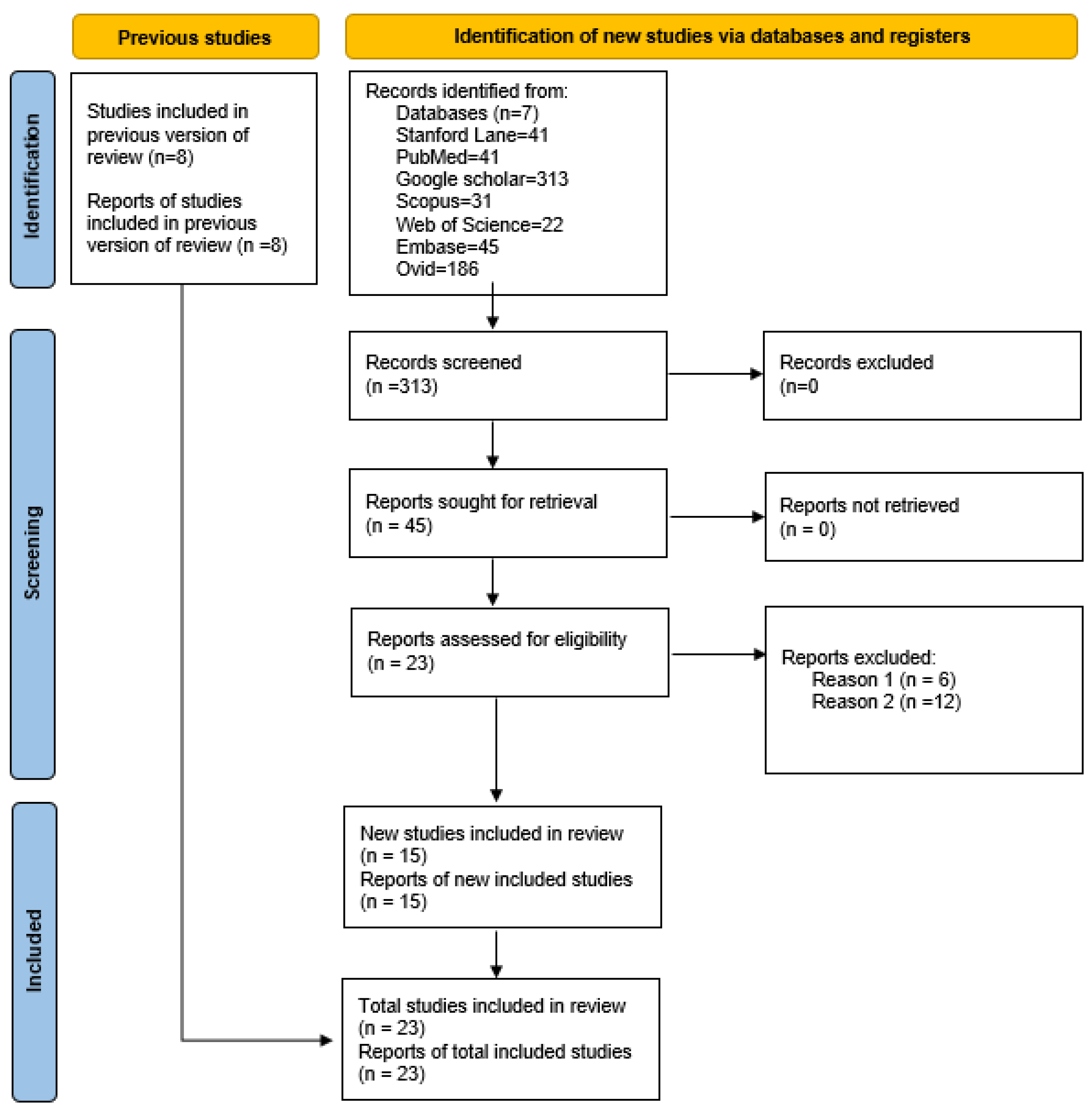
2.2. Screening and Selection of Studies
2.3. Outcome Measures
3. Results
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Concept | Synonym | MeSH | Syntax |
|---|---|---|---|
| Celiac Disease | “Celiac Disease”[TW] “Celiac Sprue”[TW] “Gluten Sensitive Enteropathy”[TW] | “Celiac Disease”[MH] | (“Celiac Disease”[MH] OR “Celiac Disease”[TW] OR “Celiac Sprue”[TW] OR “Gluten Sensitive Enteropathy”[TW]) |
| ADHD | “Attention Deficit Disorder with Hyperactivity”[TW] ADDH[TW] ADHD[TW] “Attention Deficit Disorder”[TW] “Attention Deficit Disorders with Hyperactivity”[TW] “Attention Deficit Hyperactivity Disorder”[TW] “Attention Deficit Hyperactivity Disorders”[TW] “Attention Deficit-Hyperactivity Disorder”[TW] | “Attention Deficit Disorder with Hyperactivity”[MH] | (“Attention Deficit Disorder with Hyperactivity”[MH] OR “Attention Deficit Disorder with Hyperactivity”[TW] OR “ADDH”[TW] OR “ADHD”[TW] OR “Attention Deficit Disorder”[TW] OR “Attention Deficit Disorders with Hyperactivity”[TW] OR “Attention Deficit Hyperactivity Disorder”[TW] OR “Attention Deficit Hyperactivity Disorders”[TW] OR “Attention Deficit-Hyperactivity Disorder”[TW]) |
References
- Singh, P.; Arora, S.; Lal, S.; Strand, T.A.; Makharia, G.K. Risk of Celiac Disease in the First- and Second-Degree Relatives of Patients With Celiac Disease: A Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2015, 110, 1539–1548. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Åström, J.; Sedvall, G. Psychic Disturbances in Adult Coeliac Disease: III. Reduced Central Monoamine Metabolism and Signs of Depression. Scand. J. Gastroenterol. 1982, 17, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Ruggieri, M.; Incorpora, G.; Polizzi, A.; Parano, E.; Spina, M.; Pavone, P. Low Prevalence of Neurologic and Psychiatric Manifestations in Children with Gluten Sensitivity. J. Pediatrics 2008, 152, 244–249.e1. [Google Scholar] [CrossRef] [PubMed]
- Terrone, G.; Parente, I.; Romano, A.; Auricchio, R.; Greco, L.; Del Giudice, E. The Pediatric Symptom Checklist as Screening Tool for Neurological and Psychosocial Problems in a Paediatric Cohort of Patients with Coeliac Disease. Acta Paediatr 2013, 102, e325–e328. [Google Scholar] [CrossRef]
- Verlaet, A.A.J.; Noriega, D.B.; Hermans, N.; Savelkoul, H.F.J. Nutrition, Immunological Mechanisms and Dietary Immunomodulation in ADHD. Eur. Child Adolesc. Psychiatry 2014, 23, 519–529. [Google Scholar] [CrossRef]
- Esparham, A.; Evans, R.; Wagner, L.; Drisko, J. Pediatric Integrative Medicine Approaches to Attention Deficit Hyperactivity Disorder (ADHD). Children 2014, 1, 186–207. [Google Scholar] [CrossRef] [Green Version]
- Addolorato, G.; Giuda, D.D.; Rossi, G.D.; Valenza, V.; Domenicali, M.; Caputo, F.; Gasbarrini, A.; Capristo, E.; Gasbarrini, G. Regional Cerebral Hypoperfusion in Patients with Celiac Disease. Am. J. Med. 2004, 116, 312–317. [Google Scholar] [CrossRef]
- Usai, P.; Serra, A.; Marini, B.; Mariotti, S.; Satta, L.; Boi, M.F.; Spanu, A.; Loi, G.; Piga, M. Frontal Cortical Perfusion Abnormalities Related to Gluten Intake and Associated Autoimmune Disease in Adult Coeliac Disease: 99mTc-ECD Brain SPECT Study. Dig. Liver Dis. 2004, 36, 513–518. [Google Scholar] [CrossRef]
- Norman, L.J.; Carlisi, C.; Lukito, S.; Hart, H.; Mataix-Cols, D.; Radua, J.; Rubia, K. Structural and Functional Brain Abnormalities in Attention-Deficit/Hyperactivity Disorder and Obsessive-Compulsive Disorder: A Comparative Meta-Analysis. JAMA Psychiatry 2016, 73, 815. [Google Scholar] [CrossRef]
- Clappison, E.; Hadjivassiliou, M.; Zis, P. Psychiatric Manifestations of Coeliac Disease, a Systematic Review and Meta-Analysis. Nutrients 2020, 12, 142. [Google Scholar] [CrossRef] [Green Version]
- Kittel-Schneider, S.; Reif, A. Adulte Aufmerksamkeitsdefizit-/Hyperaktivitätsstörung und Komorbidität: Neue Befunde zu epidemiologischen und genetischen Faktoren. Nervenarzt 2020, 91, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Catassi, C. Coeliac Disease in Children. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Haggård, L.; Emilsson, L.; Söderling, J.; Roelstraete, B.; Butwicka, A.; Green, P.H.R.; Ludvigsson, J.F. Psychiatric Disorders in Patients With a Diagnosis of Celiac Disease During Childhood From 1973 to 2016. Clin. Gastroenterol. Hepatol. 2021, 19, 2093–2101.e13. [Google Scholar] [CrossRef] [PubMed]
- Sel, Ç.G.; Aksoy, E.; Aksoy, A.; Yüksel, D.; Özbay, F. Neurological Manifestations of Atypical Celiac Disease in Childhood. Acta Neurol. Belg. 2017, 117, 719–727. [Google Scholar] [CrossRef]
- Pynnönen, P.A.; Isometsä, E.T.; Aronen, E.T.; Verkasalo, M.A.; Savilahti, E.; Aalberg, V.A. Mental Disorders in Adolescents With Celiac Disease. Psychosomatics 2004, 45, 325–335. [Google Scholar] [CrossRef]
- Alonso Canal, L.; Carreras Sáez, I.; Fournier del Castill, M.C.; Asensio Antón, J.; de Prada Vicente, I.; Isasi Zaragoza, C.; Tejada Gallego, J.; Martínez Gómez, M.J. Gluten-Free Diet for the Treatment of ADHD; Pilot Study. OBM Neurobiol. 2019, 3. [Google Scholar] [CrossRef] [Green Version]
- Kedem, S.; Yust-Katz, S.; Carter, D.; Levi, Z.; Kedem, R.; Dickstein, A.; Daher, S.; Katz, L.H. Attention Deficit Hyperactivity Disorder and Gastrointestinal Morbidity in a Large Cohort of Young Adults. WJG 2020, 26, 6626–6637. [Google Scholar] [CrossRef]
- Carlson, C.L.; Mann, M. Sluggish Cognitive Tempo Predicts a Different Pattern of Impairment in the Attention Deficit Hyperactivity Disorder, Predominantly Inattentive Type. J. Clin. Child Adolesc. Psychol. 2002, 31, 123–129. [Google Scholar] [CrossRef]
- Barkley, R.A. Distinguishing Sluggish Cognitive Tempo from Attention-Deficit/Hyperactivity Disorder in Adults. J. Abnorm. Psychol. 2012, 121, 978–990. [Google Scholar] [CrossRef]
- Hartman, C.A.; Willcutt, E.G.; Rhee, S.H.; Pennington, B.F. The Relation between Sluggish Cognitive Tempo and DSM-IV ADHD. J. Abnorm. Child Psychol. 2004, 32, 491–503. [Google Scholar] [CrossRef]
- McBurnett, K.; Pfiffner, L.J.; Frick, P.J. Symptom Properties as a Function of ADHD Type: An Argument for Continued Study of Sluggish Cognitive Tempo. J. Abnorm. Child Psychol. 2001, 29, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Bauermeister, J.J.; Barkley, R.A.; Bauermeister, J.A.; Martínez, J.V.; McBurnett, K. Validity of the Sluggish Cognitive Tempo, Inattention, and Hyperactivity Symptom Dimensions: Neuropsychological and Psychosocial Correlates. J. Abnorm. Child Psychol. 2012, 40, 683–697. [Google Scholar] [CrossRef] [PubMed]
- Garner, A.A.; Marceaux, J.C.; Mrug, S.; Patterson, C.; Hodgens, B. Dimensions and Correlates of Attention Deficit/Hyperactivity Disorder and Sluggish Cognitive Tempo. J. Abnorm. Child Psychol. 2010, 38, 1097–1107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Germone, M.M.; Ariefdjohan, M.; Stahl, M.; Shull, M.; Mehta, P.; Nagle, S.; Tarbell, S.; Liu, E. Family Ties: The Impact of Celiac Disease on Children and Caregivers. Qual. Life Res. 2022. [CrossRef] [PubMed]
- Riglin, L.; Leppert, B.; Dardani, C.; Thapar, A.K.; Rice, F.; O’Donovan, M.C.; Davey Smith, G.; Stergiakouli, E.; Tilling, K.; Thapar, A. ADHD and Depression: Investigating a Causal Explanation. Psychol. Med. 2021, 51, 1890–1897. [Google Scholar] [CrossRef] [Green Version]
- Ertürk, E.; Wouters, S.; Imeraj, L.; Lampo, A. Association of ADHD and Celiac Disease: What Is the Evidence? A Systematic Review of the Literature. J. Atten. Disord. 2020, 24, 1371–1376. [Google Scholar] [CrossRef]
- Lichtwark, I.T.; Newnham, E.D.; Robinson, S.R.; Shepherd, S.J.; Hosking, P.; Gibson, P.R.; Yelland, G.W. Cognitive Impairment in Coeliac Disease Improves on a Gluten-Free Diet and Correlates with Histological and Serological Indices of Disease Severity. Aliment Pharmacol. Ther. 2014, 40, 160–170. [Google Scholar] [CrossRef] [Green Version]
- Lahat, E.; Broide, E.; Leshem, M.; Evans, S.; Scapa, E. Prevalence of Celiac Antibodies in Children with Neurologic Disorders. Pediatric Neurol. 2000, 22, 393–396. [Google Scholar] [CrossRef]
- Alkhayyat, M.; Qapaja, T.; Aggarwal, M.; Almomani, A.; Abureesh, M.; Al-otoom, O.; Zmaili, M.; Mansoor, E.; Abou Saleh, M. Epidemiology and Risk of Psychiatric Disorders among Patients with Celiac Disease: A Population-based National Study. J. Gastroenterol. Hepatol. 2021, 36, 2165–2170. [Google Scholar] [CrossRef]
- Zelnik, N.; Pacht, A.; Obeid, R.; Lerner, A. Range of Neurologic Disorders in Patients With Celiac Disease. Pediatrics 2004, 113, 1672–1676. [Google Scholar] [CrossRef] [Green Version]
- Niederhofer, H.; Pittschieler, K. A Preliminary Investigation of ADHD Symptoms in Persons With Celiac Disease. J. Atten Disord. 2006, 10, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Niederhofer, H. Association of Attention-Deficit/Hyperactivity Disorder and Celiac Disease: A Brief Report. Prim. Care Companion CNS Disord. 2011, 13, PCC.10br01104. [Google Scholar] [CrossRef] [PubMed]
- Dazy, K.M.; Rubenstein, J.H.; Holevinski, L.; Kao, J.Y. Sa1285 The Prevalence of ADHD in Adults and Children Previously Diagnosed With Celiac Disease: A Hospital-Based Study. Gastroenterology 2013, 5 (Suppl. S1), S-251–S-252. [Google Scholar] [CrossRef]
- Diaconu, G.; Burlea, M.; Grigore, I.; Anton, D.T.; Trandafir, L.M. Celiac Disease with Neurologic Manifestations in Children. Rev. Med. Chir. Soc. Med. Nat. Iasi. 2013, 117, 88–94. [Google Scholar] [PubMed]
- Güngör, S.; Celiloğlu, Ö.S.; Özcan, Ö.Ö.; Raif, S.G.; Selimoğlu, M.A. Frequency of Celiac Disease in Attention-Deficit/Hyperactivity Disorder. J. Pediatric Gastroenterol. Nutr. 2013, 56, 211–214. [Google Scholar] [CrossRef]
- Işikay, S.; Kocamaz, H. The Neurological Face Of Celiac Disease. Arq. Gastroenterol. 2015, 52, 167–170. [Google Scholar] [CrossRef]
- Kumperscak, H.G.; Rebec, Z.K.; Sobocan, S.; Fras, V.T.; Dolinsek, J. Prevalence of Celiac Disease Is Not Increased in ADHD Sample. J. Atten Disord. 2020, 24, 1085–1089. [Google Scholar] [CrossRef]
- Butwicka, A.; Lichtenstein, P.; Frisén, L.; Almqvist, C.; Larsson, H.; Ludvigsson, J.F. Celiac Disease Is Associated with Childhood Psychiatric Disorders: A Population-Based Study. J. Pediatrics 2017, 184, 87–93.e1. [Google Scholar] [CrossRef]
- Prinzbach, A.; Moosavinasab, S.; Rust, S.; Boyle, B.; Barnard, J.A.; Huang, Y.; Lin, S. Comorbidities in Childhood Celiac Disease: A Phenome Wide Association Study Using the Electronic Health Record. J. Pediatric Gastroenterol. Nutr. 2018, 67, 488–493. [Google Scholar] [CrossRef]
- Honar, N.; Barkhordarian, M.; Ghanizadeh, A.; Radanfar, R. Association of Celiac Disease with Attention Deficit Hyperactivity Disorder. J. Compr. Ped. 2019, 11. [Google Scholar] [CrossRef] [Green Version]
- Kristensen, V.A.; Valeur, J.; Brackmann, S.; Jahnsen, J.; Brunborg, C.; Tveito, K. Attention Deficit and Hyperactivity Disorder Symptoms Respond to Gluten-Free Diet in Patients with Coeliac Disease. Scand. J. Gastroenterol. 2019, 54, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Daniel, S.; Francis, D.; Messina, C.; Chung, J.; Small-Harary, L.; Fischel, J.E.; Chawla, A. Intellectual and Attentional Characteristics of Children with Celiac Disease after Initiation of a Gluten-Free Diet. Int. J. Celiac Dis. 2020, 8, 129–135. [Google Scholar] [CrossRef]
- Coburn, S.; Rose, M.; Sady, M.; Parker, M.; Suslovic, W.; Weisbrod, V.; Kerzner, B.; Streisand, R.; Kahn, I. Mental Health Disorders and Psychosocial Distress in Pediatric Celiac Disease. J. Pediatric Gastroenterol. Nutr. 2019. [CrossRef] [PubMed]
- Kırsaçlıoğlu, C.; Hijal, G.; Dinc, G.; Guner, S.; Sandra, S.; Uneri, O. Attention-Deficit Hyperactivity Disorder and Gluten Sensitivity in Children. J. PediaRestr. 2021, 8, 326–329. [Google Scholar] [CrossRef]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition Guidelines for the Diagnosis of Coeliac Disease. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 136–160. [Google Scholar] [CrossRef]
- Bosch, A.; Bierens, M.; de Wit, A.G.; Ly, V.; van der Velde, J.; de Boer, H.; van Beek, G.; Appelman, D.; Visser, S.; Bos, L.; et al. A Two Arm Randomized Controlled Trial Comparing the Short and Long Term Effects of an Elimination Diet and a Healthy Diet in Children with ADHD (TRACE Study). Rationale, Study Design and Methods. BMC Psychiatry 2020, 20, 262. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, P.; Haracopos, D.; Knivsberg, A.-M.; Reichelt, K.L.; Parlar, S.; Jacobsen, J.; Seim, A.; Pedersen, L.; Schondel, M.; Shattock, P. The ScanBrit Randomised, Controlled, Single-Blind Study of a Gluten- and Casein-Free Dietary Intervention for Children with Autism Spectrum Disorders. Nutr. Neurosci. 2010, 13, 87–100. [Google Scholar] [CrossRef]
- Lykogeorgou, M.; Karkelis, S.; Papadaki-Papandreou, O.; Nikita, M. PS-253b Gluten Free Diet For Children With Attention Deficit And Hyperactivity Disorder. Arch. Dis. Child. 2014, 99 (Suppl. 2), A204–A205. [Google Scholar] [CrossRef] [Green Version]
- Whiteley, P.; Shattock, P.; Knivsberg, A.-M.; Seim, A.; Reichelt, K.L.; Todd, L.; Carr, K.; Hooper, M. Gluten- and Casein-Free Dietary Intervention for Autism Spectrum Conditions. Front. Hum. Neurosci. 2013, 6. [Google Scholar] [CrossRef] [Green Version]
- Becker, S.P.; Leopold, D.R.; Burns, G.L.; Jarrett, M.A.; Langberg, J.M.; Marshall, S.A.; McBurnett, K.; Waschbusch, D.A.; Willcutt, E.G. The Internal, External, and Diagnostic Validity of Sluggish Cognitive Tempo: A Meta-Analysis and Critical Review. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 163–178. [Google Scholar] [CrossRef] [Green Version]
- Pelsser, L.M.; Frankena, K.; Toorman, J.; Savelkoul, H.F.; Dubois, A.E.; Pereira, R.R.; Haagen, T.A.; Rommelse, N.N.; Buitelaar, J.K. Effects of a Restricted Elimination Diet on the Behaviour of Children with Attention-Deficit Hyperactivity Disorder (INCA Study): A Randomised Controlled Trial. Lancet 2011, 377, 494–503. [Google Scholar] [CrossRef]
- Nardecchia, S.; Auricchio, R.; Discepolo, V.; Troncone, R. Extra-Intestinal Manifestations of Coeliac Disease in Children: Clinical Features and Mechanisms. Front. Pediatr. 2019, 7, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. ACG Clinical Guidelines: Diagnosis and Management of Celiac Disease. Am. J. Gastroenterol. 2013, 108, 656–676. [Google Scholar] [CrossRef] [Green Version]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo Definitions for Coeliac Disease and Related Terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Pallav, K.; Kabbani, T.; Tariq, S.; Vanga, R.; Kelly, C.P.; Leffler, D.A. Clinical Utility of Celiac Disease-Associated HLA Testing. Dig. Dis. Sci. 2014, 59, 2199–2206. [Google Scholar] [CrossRef]
- Tylee, D.S.; Sun, J.; Hess, J.L.; Tahir, M.A.; Sharma, E.; Malik, R.; Worrall, B.B.; Levine, A.J.; Martinson, J.J.; Nejentsev, S.; et al. Genetic Correlations among Psychiatric and Immune-Related Phenotypes Based on Genome-Wide Association Data. Am. J. Med. Genet. 2018, 177, 641–657. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.C.; Nassar, N.; Shand, A.W.; Jones, H.; Guastella, A.J.; Dale, R.C.; Lain, S.J. Association of Maternal Autoimmune Disease With Attention-Deficit/Hyperactivity Disorder in Children. JAMA Pediatr 2021, 175, e205487. [Google Scholar] [CrossRef]
- Nielsen, P.R.; Benros, M.E.; Dalsgaard, S. Associations Between Autoimmune Diseases and Attention-Deficit/Hyperactivity Disorder: A Nationwide Study. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 234–240.e1. [Google Scholar] [CrossRef]
- Akmatov, M.K.; Ermakova, T.; Bätzing, J. Psychiatric and Nonpsychiatric Comorbidities among Children with ADHD: An Exploratory Analysis of Nationwide Claims Data in Germany. J. Atten. Disord. 2021, 25, 874–884. [Google Scholar] [CrossRef]
- Moruzzi, S.; Rijsdijk, F.; Battaglia, M. A Twin Study of the Relationships among Inattention, Hyperactivity/Impulsivity and Sluggish Cognitive Tempo Problems. J. Abnorm Child. Psychol. 2014, 42, 63–75. [Google Scholar] [CrossRef]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- King, J.A.; Jeong, J.; Underwood, F.E.; Quan, J.; Panaccione, N.; Windsor, J.W.; Coward, S.; deBruyn, J.; Ronksley, P.E.; Shaheen, A.-A.; et al. Incidence of Celiac Disease Is Increasing Over Time: A Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2020, 115, 507–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lebwohl, B.; Mehta, R.; Cao, Y.; Green, P.H.R.; Grodstein, F.; Jovani, M.; Lochhead, P.; Okereke, O.I.; Sampson, L.; et al. Long-Term Intake of Gluten and Cognitive Function Among US Women. JAMA Netw. Open 2021, 4, e2113020. [Google Scholar] [CrossRef]
- Beets, M.W.; Weaver, R.G.; Ioannidis, J.P.A.; Geraci, M.; Brazendale, K.; Decker, L.; Okely, A.D.; Lubans, D.; van Sluijs, E.; Jago, R.; et al. Identification and Evaluation of Risk of Generalizability Biases in Pilot versus Efficacy/Effectiveness Trials: A Systematic Review and Meta-Analysis. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Source | Study Design | Inclusion Criteria | No. of Subjects | No. of Controls | Age Range ± Mean (years) | ADHD Criteria | ADHD Method of Diagnosis | Celiac Disease (CeD) Serology/Biopsy/HLA | Association |
|---|---|---|---|---|---|---|---|---|---|
| Lahat, 2000 [28] | OB, CS | ADHD | 39 | 34 | 6–13, 8.6 | DSM-IV | Nil | IgA, IgG, AGA, EMA | No |
| Zelnick, 2004 [30] | OB, CO, CC | CeD/GFD | 111 | 211 | 20.8 ± 8.9 | DSM-IV | Neurological examination | IgA, EMA, biopsy | Yes |
| Pynnonen, 2004 [15] | OB, CO, RET | CeD/GFD | 29 | 29 | 12–17 | DSM-IV | KSADS-PL, CBCL, YSR | EMA, tTGAb, biopsy | No |
| Niederhofer 2006 [31] | OB, COH, RET | CeD | 132 | 0 | 3–57, 19.3 | ICD-10, DSM-IV | Hypescheme | EMA, tTGAb | Yes |
| Ruggieri, 2008 [3] | OB, CO, COH, PRO | CeD | 835 | 0 | 9 mths–17, 7.3 | Nil | Neurological examination | AGA, EMA, HLA, biopsy | No |
| Niederhofer2011 [32] | OB, COH, PRO | ADHD | 67 | 0 | 7–42, 11.4 | ICD-10, DSM-IV | Hypescheme | EMA, tTGAb | Yes |
| Dazy, 2013 [33] | OB, CO, CC | CeD | 281 | 301 | <25 | ICD-9 | Nil | EMA/tTG, biopsy | No |
| Diaconu, 2013 [34] | PRO | CeD/GFD | 48 | 0 | 2–18 | DSM-IV | Psychological examination | Biopsy | No |
| Gungor, 2013 [35] | OB, CO, CC | ADHD | 362 | 390 | 5–15 | DSM-IV TR | KSADS | tTgIgG, tTgIgA, biopsy | No |
| Lichtwark, 2014 [27] | LO, PS | CeD/GFD | 11 | 0 | 22–39, 22 | Nil | SCIT, Trails | Nil | Yes |
| Isikay, 2015 [36] | CS, PRO | CeD | 40 | 297 | <18 | Nil | Neurological examination | tTG, biopsy | Yes |
| Kumpersack, 2006 [37] | PRO | ADHD | 53 | 0 | 13.2 | ICD-10 | Nil | ESPGHAN | No |
| Butwicka, 2017 [38] | COH, POP, PRO | CeD | 10,903 | 12,710 | <18 | ICD-9, ICD-10 | Adult ADHD Self-Report Scale | Nil | Yes |
| Prinzbach, 2018 [39] | COH, RET | CeD | 433 | 4330 | 0–21, 9.53 | ICD-10 | Nil | Nil | No |
| Canal, 2019 [16] | PS | ADHD | 6 | 0 | 9–13 | DSM-5 | CPT-III | ESPGHAN | Yes |
| Honar, 2019 [40] | CS | ADHD | 99 | 0 | 4–18 | DSM-IV | ADHD checklist | tTGAb | Yes |
| Kristenson, 2019 [41] | LO, PRO | CeD | 31 | 60 | >18 | DSM-IV | Adult ADHD Self-Report Scale | Nil | Yes |
| Lee, 2020 [42] | PRO | CeD/GFD | 33 | 0 | 6–18,10.7 | DSM-IV | CPT-III, CBCL | tTIg/IgG | Yes |
| Coburn, 2020 [43] | PRO | CeD/GFD | 73 | 0 | 3–18 | DSM-IV | Mental health diagnosis | Nil | Yes |
| Kedem, 2020 [17] | RET, COH | ADHD | 117 | 0 | 17–35 | ICD-9 | Nil | ESPGHAN | No |
| Kirsacliglu, 2020 [44] | PRO | ADHD | 117 | 0 | 6–18 | DSM-IV | Nil | ESPGHAN | No |
| Lebwohl, 2021 [13] | COH | CeD | 3174 | 13,286 | <18 | ICD-9, ICD-10 | Nil | Nil | Yes |
| Alkhayyat, 2021 [29] | OB, COH, RET | CeD | OR=1.75 | 112,240 | All age ranges | ICD-9 | Nil | Nil | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaur, S. The Association between ADHD and Celiac Disease in Children. Children 2022, 9, 781. https://doi.org/10.3390/children9060781
Gaur S. The Association between ADHD and Celiac Disease in Children. Children. 2022; 9(6):781. https://doi.org/10.3390/children9060781
Chicago/Turabian StyleGaur, Sonia. 2022. "The Association between ADHD and Celiac Disease in Children" Children 9, no. 6: 781. https://doi.org/10.3390/children9060781
APA StyleGaur, S. (2022). The Association between ADHD and Celiac Disease in Children. Children, 9(6), 781. https://doi.org/10.3390/children9060781






