Stress, Inflammation and Metabolic Biomarkers Are Associated with Body Composition Measures in Lean, Overweight, and Obese Children and Adolescents †
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Clinical Evaluation
2.3. Hair Sampling
2.4. Saliva Sampling
2.5. Blood Sampling
2.6. Heart Rate Variability Measurements
2.7. Questionnaires
2.8. Statistical Analyses
3. Results
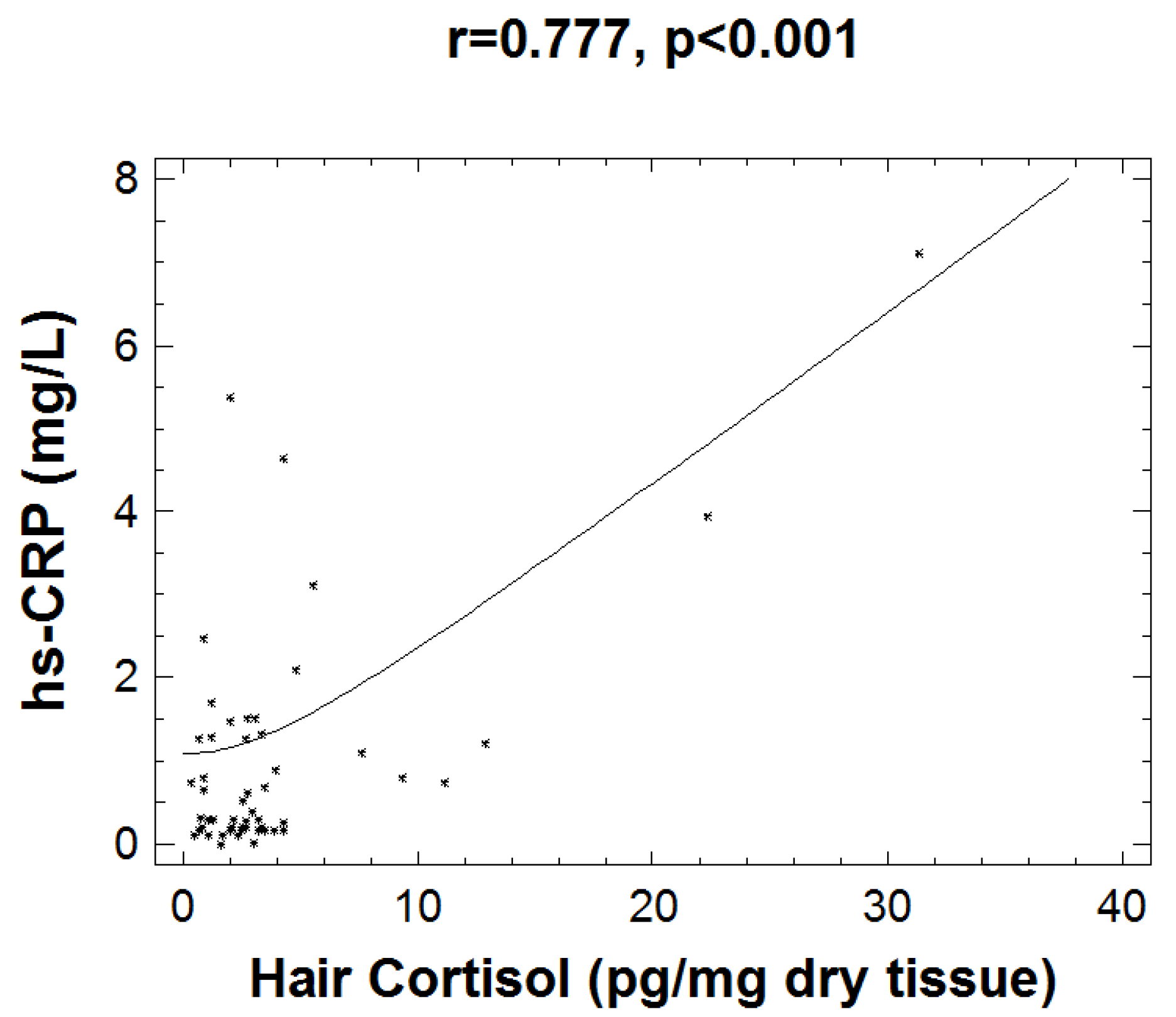
Correlation Analyses
4. Discussion
4.1. Stress, Non-Adaptive “Para-Inflammation” Biomarkers, and Body Composition
4.2. Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Styne, D.M.; Arslanian, S.A.; Connor, E.L.; Farooqi, I.S.; Murad, M.H.; Silverstein, J.H.; Yanovski, J.A. Pediatric Obesity-Assessment, Treatment, and Prevention: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2017, 102, 709–757. [Google Scholar] [CrossRef]
- Charmandari, E.; Kino, T.; Souvatzoglou, E.; Chrousos, G.P. Pediatric stress: Hormonal mediators and human development. Horm. Res. 2003, 59, 161–179. [Google Scholar] [CrossRef] [PubMed]
- Stavrou, S.; Nicolaides, N.C.; Papageorgiou, I.; Papadopoulou, P.; Terzioglou, E.; Chrousos, G.P.; Darviri, C.; Charmandari, E. The effectiveness of a stress-management intervention program in the management of overweight and obesity in childhood and adolescence. J. Mol. Biochem. 2016, 5, 63–70. [Google Scholar] [PubMed]
- Kappes, C.; Stein, R.; Korner, A.; Merkenschlager, A.; Kiess, W. Stress, Stress Reduction and Obesity in Childhood and Adolescence. Horm. Res. Paediatr. 2021, 94, 281–289. [Google Scholar] [CrossRef]
- Bose, M.; Olivan, B.; Laferrere, B. Stress and obesity: The role of the hypothalamic-pituitary-adrenal axis in metabolic disease. Curr. Opin. Endocrinol. Diabetes Obes. 2009, 16, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Giannakakis, G.; Grigoriadis, D.; Giannakaki, K.; Simantiraki, O.; Roniotis, A.; Tsiknakis, M. Review on psychological stress detection using biosignals. In IEEE Transactions on Affective Computing; IEEE: Piscataway, NJ, USA, 2019. [Google Scholar] [CrossRef]
- Jackson, S.E.; Kirschbaum, C.; Steptoe, A. Hair cortisol and adiposity in a population-based sample of 2527 men and women aged 54 to 87 years. Obesity 2017, 25, 539–544. [Google Scholar] [CrossRef]
- Veldhorst, M.A.; Noppe, G.; Jongejan, M.H.; Kok, C.B.; Mekic, S.; Koper, J.W.; van Rossum, E.F.; van den Akker, E.L. Increased scalp hair cortisol concentrations in obese children. J. Clin. Endocrinol. Metab. 2014, 99, 285–290. [Google Scholar] [CrossRef][Green Version]
- Papafotiou, C.; Christaki, E.; van den Akker, E.L.; Wester, V.L.; Apostolakou, F.; Papassotiriou, I.; Chrousos, G.P.; Pervanidou, P. Hair cortisol concentrations exhibit a positive association with salivary cortisol profiles and are increased in obese prepubertal girls. Stress 2017, 20, 217–222. [Google Scholar] [CrossRef]
- Giannakakis, G.; Marias, K.; Tsiknakis, M. A stress recognition system using HRV parameters and machine learning techniques. In Proceedings of the 2019 8th International Conference on Affective Computing and Intelligent Interaction Workshops and Demos (ACIIW), Cambridge, UK, 3–6 September 2019; pp. 269–272. [Google Scholar] [CrossRef]
- Wen, X.; Pekkala, S.; Wang, R.; Wiklund, P.; Feng, G.; Cheng, S.M.; Tan, X.; Liu, Y.; Chen, P.; Eriksson, J.G.; et al. Does systemic low-grade inflammation associate with fat accumulation and distribution? A 7-year follow-up study with peripubertal girls. J. Clin. Endocrinol. Metab. 2014, 99, 1411–1419. [Google Scholar] [CrossRef][Green Version]
- Singer, K.; Lumeng, C.N. The initiation of metabolic inflammation in childhood obesity. J. Clin. Investig. 2017, 127, 65–73. [Google Scholar] [CrossRef]
- Fujita, B.; Strodthoff, D.; Fritzenwanger, M.; Pfeil, A.; Ferrari, M.; Goebel, B.; Figulla, H.R.; Gerdes, N.; Jung, C. Altered red blood cell distribution width in overweight adolescents and its association with markers of inflammation. Pediatric Obes. 2013, 8, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Wiebe, N.; James, M.T.; Naugler, C.; Manns, B.J.; Klarenbach, S.W.; Hemmelgarn, B.R. Red cell distribution width associations with clinical outcomes: A population-based cohort study. PLoS ONE 2019, 14, e0212374. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Srinivasan, S.R.; Xu, J.; Berenson, G.S. Black-white divergence in the relation of white blood cell count to metabolic syndrome in preadolescents, adolescents, and young adults: The Bogalusa Heart Study. Diabetes Care 2010, 33, 2474–2476. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Bixler, E.O.; Papanicolaou, D.A.; Chrousos, G.P. Chronic systemic inflammation in overweight and obese adults. JAMA 2000, 283, 2235–2236. [Google Scholar] [CrossRef] [PubMed]
- Sternberg, E.M.; Chrousos, G.P.; Wilder, R.L.; Gold, P.W. The stress response and the regulation of inflammatory disease. Ann. Intern. Med. 1992, 117, 854–866. [Google Scholar] [CrossRef]
- Cronstein, B.N.; Kimmel, S.C.; Levin, R.I.; Martiniuk, F.; Weissmann, G. A mechanism for the antiinflammatory effects of corticosteroids: The glucocorticoid receptor regulates leukocyte adhesion to endothelial cells and expression of endothelial-leukocyte adhesion molecule 1 and intercellular adhesion molecule 1. Proc. Natl. Acad. Sci. USA 1992, 89, 9991–9995. [Google Scholar] [CrossRef]
- Chrousos, G.P. The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. N. Engl. J. Med. 1995, 332, 1351–1362. [Google Scholar] [CrossRef]
- Chrousos, G.P. Stress and disorders of the stress system. Nat. Rev. Endocrinol. 2009, 5, 374–381. [Google Scholar] [CrossRef]
- Kiecolt-Glaser, J.K.; Derry, H.M.; Fagundes, C.P. Inflammation: Depression fans the flames and feasts on the heat. Am. J. Psychiatry 2015, 172, 1075–1091. [Google Scholar] [CrossRef]
- Ruiz, L.D.; Zuelch, M.L.; Dimitratos, S.M.; Scherr, R.E. Adolescent Obesity: Diet Quality, Psychosocial Health, and Cardiometabolic Risk Factors. Nutrients 2019, 12, 43. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed]
- Chiotis, D.; Krikos, X.; Tsiftis, G.; Hatzisymeaon, M.; Maniati-Christidi, M.; Dacou-Voutetakis, C. Body mass index and prevalence of obesity in subjects of Hellenic origin aged 0-18 years, living in the Athens area. Ann Clin. Pediatr. Univ. Atheniensis 2004, 11, 139–154. [Google Scholar]
- Collins, N. Measuring height and weight. Adv. Ski. Wound Care 2002, 15, 91–92. [Google Scholar] [CrossRef] [PubMed]
- Kelly, O.J.; Gilman, J.C.; Boschiero, D.; Ilich, J.Z. Osteosarcopenic Obesity: Current Knowledge, Revised Identification Criteria and Treatment Principles. Nutrients 2019, 11, 747. [Google Scholar] [CrossRef] [PubMed]
- Wester, V.L.; van Rossum, E.F. Clinical applications of cortisol measurements in hair. Eur. J. Endocrinol. 2015, 173, M1–M10. [Google Scholar] [CrossRef] [PubMed]
- Adam, E.K.; Kumari, M. Assessing salivary cortisol in large-scale, epidemiological research. Psychoneuroendocrinology 2009, 34, 1423–1436. [Google Scholar] [CrossRef]
- Pervanidou, P.; Bastaki, D.; Chouliaras, G.; Papanikolaou, K.; Kanaka-Gantenbein, C.; Chrousos, G. Internalizing and externalizing problems in obese children and adolescents: Associations with daily salivary cortisol concentrations. Hormones 2015, 14, 623–631. [Google Scholar] [CrossRef]
- Teixeira, R.R.; Diaz, M.M.; Santos, T.V.; Bernardes, J.T.; Peixoto, L.G.; Bocanegra, O.L.; Neto, M.B.; Espindola, F.S. Chronic stress induces a hyporeactivity of the autonomic nervous system in response to acute mental stressor and impairs cognitive performance in business executives. PLoS ONE 2015, 10, e0119025. [Google Scholar] [CrossRef]
- Eyre, E.L.; Duncan, M.J.; Birch, S.L.; Fisher, J.P. The influence of age and weight status on cardiac autonomic control in healthy children: A review. Auton. Neurosci. Basic Clin. 2014, 186, 8–21. [Google Scholar] [CrossRef]
- Von Rosenberg, W.; Hoting, M.O.; Mandic, D.P. A physiology based model of heart rate variability. Biomed. Eng. Lett. 2019, 9, 425–434. [Google Scholar] [CrossRef]
- Von Rosenberg, W.; Chanwimalueang, T.; Adjei, T.; Jaffer, U.; Goverdovsky, V.; Mandic, D.P. Resolving Ambiguities in the LF/HF Ratio: LF-HF Scatter Plots for the Categorization of Mental and Physical Stress from HRV. Front. Physiol. 2017, 8, 360. [Google Scholar] [CrossRef] [PubMed]
- Gaudry, E.; Vagg, P.; Spielberger, C.D. Validation of the State-Trait Distinction in Anxiety Research. Multivar. Behav. Res. 1975, 10, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Psychountaki, M.; Zervas, Y.; Karteroliotis, K.; Spielberger, C. Reliability and validity of the Greek version of the STAIC. Eur. J. Psycho.l Assess. 2003, 19, 124–130. [Google Scholar] [CrossRef]
- Giannakopoulos, G.; Kazantzi, M.; Dimitrakaki, C.; Tsiantis, J.; Kolaitis, G.; Tountas, Y. Screening for children’s depression symptoms in Greece: The use of the Children’s Depression Inventory in a nation-wide school-based sample. Eur. Child Adolesc. Psychiatry 2009, 18, 485–492. [Google Scholar] [CrossRef]
- Chen, S.Y.; Feng, Z.; Yi, X. A general introduction to adjustment for multiple comparisons. J. Thorac. Dis. 2017, 9, 1725–1729. [Google Scholar] [CrossRef] [PubMed]
- Cole, T.J.; Bellizzi, M.C.; Flegal, K.M.; Dietz, W.H. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 2000, 320, 1240–1243. [Google Scholar] [CrossRef]
- Koski, M.; Naukkarinen, H. The Relationship between Stress and Severe Obesity: A Case-Control Study. Biomed. Hub 2017, 2, 1–13. [Google Scholar] [CrossRef]
- Baba, R.; Koketsu, M.; Nagashima, M.; Inasaka, H.; Yoshinaga, M.; Yokota, M. Adolescent obesity adversely affects blood pressure and resting heart rate. Circ. J. Off. J. Jpn. Circ. Soc. 2007, 71, 722–726. [Google Scholar] [CrossRef]
- Singh, K.; Shen, B.J. Abdominal obesity and chronic stress interact to predict blunted cardiovascular reactivity. Int. J. Psychophysiol. Off. J. Int. Organ. Psychophysiol. 2013, 90, 73–79. [Google Scholar] [CrossRef]
- Michels, N.; Sioen, I.; Clays, E.; De Buyzere, M.; Ahrens, W.; Huybrechts, I.; Vanaelst, B.; De Henauw, S. Children’s heart rate variability as stress indicator: Association with reported stress and cortisol. Biol. Psychol. 2013, 94, 433–440. [Google Scholar] [CrossRef]
- Soares-Miranda, L.; Alves, A.J.; Vale, S.; Aires, L.; Santos, R.; Oliveira, J.; Mota, J. Central fat influences cardiac autonomic function in obese and overweight girls. Pediatric Cardiol. 2011, 32, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Yamamoto, S.S.; Brosschot, J.F. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int. J. Cardiol. 2010, 141, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.D.; Kouros, C.; Frankel, S.A.; McCauley, E.; Diamond, G.S.; Schloredt, K.A.; Garber, J. Longitudinal relations between stress and depressive symptoms in youth: Coping as a mediator. J. Abnorm. Child Psychol. 2015, 43, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Bates, R.; Salsberry, P.; Ford, J. Measuring Stress in Young Children Using Hair Cortisol: The State of the Science. Biol. Res. Nurs. 2017, 19, 499–510. [Google Scholar] [CrossRef]
- Gonzalez, D.; Jacobsen, D.; Ibar, C.; Pavan, C.; Monti, J.; Fernandez Machulsky, N.; Balbi, A.; Fritzler, A.; Jamardo, J.; Repetto, E.M.; et al. Hair Cortisol Measurement by an Automated Method. Sci. Rep. 2019, 9, 8213. [Google Scholar] [CrossRef]
- Kyrou, I.; Tsigos, C. Stress hormones: Physiological stress and regulation of metabolism. Curr. Opin. Pharmacol. 2009, 9, 787–793. [Google Scholar] [CrossRef]
- Adam, E.K.; Quinn, M.E.; Tavernier, R.; McQuillan, M.T.; Dahlke, K.A.; Gilbert, K.E. Diurnal cortisol slopes and mental and physical health outcomes: A systematic review and meta-analysis. Psychoneuroendocrinology 2017, 83, 25–41. [Google Scholar] [CrossRef]
- Miller, G.E.; Chen, E.; Parker, K.J. Psychological stress in childhood and susceptibility to the chronic diseases of aging: Moving toward a model of behavioral and biological mechanisms. Psychol. Bull. 2011, 137, 959–997. [Google Scholar] [CrossRef]
- Peppa, M.; Stefanaki, C.; Papaefstathiou, A.; Boschiero, D.; Dimitriadis, G.; Chrousos, G.P. Bioimpedance analysis vs. DEXA as a screening tool for osteosarcopenia in lean, overweight and obese Caucasian postmenopausal females. Hormones 2017, 16, 181–193. [Google Scholar] [CrossRef]
- Manios, Y.; Moschonis, G.; Chrousos, G.P.; Lionis, C.; Mougios, V.; Kantilafti, M.; Tzotzola, V.; Skenderi, K.P.; Petridou, A.; Tsalis, G.; et al. The double burden of obesity and iron deficiency on children and adolescents in Greece: The Healthy Growth Study. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2013, 26, 470–478. [Google Scholar] [CrossRef]
- Yanoff, L.B.; Menzie, C.M.; Denkinger, B.; Sebring, N.G.; McHugh, T.; Remaley, A.T.; Yanovski, J.A. Inflammation and iron deficiency in the hypoferremia of obesity. Int. J. Obes. 2007, 31, 1412–1419. [Google Scholar] [CrossRef] [PubMed]
- Tsigos, C.; Stefanaki, C.; Lambrou, G.I.; Boschiero, D.; Chrousos, G.P. Stress and inflammatory biomarkers and symptoms are associated with bioimpedance measures. Eur. J. Clin. Investig. 2015, 45, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Earthman, C.P.; Pichard, C.; Coss-Bu, J.A. Body composition during growth in children: Limitations and perspectives of bioelectrical impedance analysis. Eur. J. Clin. Nutr. 2015, 69, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Stefanaki, C.; Pervanidou, P.; Boschiero, D.; Chrousos, G.P. Chronic stress and body composition disorders: Implications for health and disease. Hormones 2018, 17, 33–43. [Google Scholar] [CrossRef]
- Stefanaki, C.; Peppa, M.; Boschiero, D.; Chrousos, G.P. Healthy overweight/obese youth: Early osteosarcopenic obesity features. Eur. J. Clin. Investig. 2016, 46, 767–778. [Google Scholar] [CrossRef]
- Kim, J.; Im, J.S.; Choi, C.H.; Park, C.H.; Lee, J.I.; Son, K.H.; Choi, Y.H. The Association between Red Blood Cell Distribution Width and Sarcopenia in U.S. Adults. Sci. Rep. 2018, 8, 11484. [Google Scholar] [CrossRef]
- Rosmond, R.; Bjorntorp, P. The hypothalamic-pituitary-adrenal axis activity as a predictor of cardiovascular disease, type 2 diabetes and stroke. J. Intern. Med. 2000, 247, 188–197. [Google Scholar] [CrossRef]
- Christaki, E.; Kokkinos, A.; Costarelli, V.; Alexopoulos, E.C.; Chrousos, G.P.; Darviri, C. Stress management can facilitate weight loss in Greek overweight and obese women: A pilot study. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2013, 26 (Suppl. S1), 132–139. [Google Scholar] [CrossRef]
- Lynch, T.; Azuero, A.; Lochman, J.E.; Park, N.J.; Turner-Henson, A.; Rice, M. The Influence of Psychological Stress, Depressive Symptoms, and Cortisol on Body Mass and Central Adiposity in 10- to-12-Year-Old Children. J. Pediatric Nurs. 2019, 44, 42–49. [Google Scholar] [CrossRef]
- Pervanidou, P.; Chrousos, G.P. Stress and obesity/metabolic syndrome in childhood and adolescence. Int. J. Pediatric Obes. IJPO Off. J. Int. Assoc. Study Obes. 2011, 6 (Suppl. S1), 21–28. [Google Scholar] [CrossRef]
- Pervanidou, P.; Bastaki, D.; Chouliaras, G.; Papanikolaou, K.; Laios, E.; Kanaka-Gantenbein, C.; Chrousos, G.P. Circadian cortisol profiles, anxiety and depressive symptomatology, and body mass index in a clinical population of obese children. Stress 2013, 16, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Craven, J.; Desbrow, B.; Sabapathy, S.; Bellinger, P.; McCartney, D.; Irwin, C. The Effect of Consuming Carbohydrate With and Without Protein on the Rate of Muscle Glycogen Re-synthesis During Short-Term Post-exercise Recovery: A Systematic Review and Meta-analysis. Sports Med.-Open 2021, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Marginean, C.O.; Melit, L.E.; Ghiga, D.V.; Marginean, M.O. Early Inflammatory Status Related to Pediatric Obesity. Front. Pediatrics 2019, 7, 241. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Normal Weight (N = 40) | Overweight/ Obese (N = 81) | p Value |
|---|---|---|---|
| Age | 8.74 ± 2.14 | 9.02 ± 2.28 | 0.522 |
| Sex | 70% Female 30% male | 61.7% Female 38.3% male | 0.375 |
| BMI z-score | −0.19 ± 0.60 | 2.20 ± 1.43 | <0.001 * |
| Tanner stage | 90% pre-pubertal 6.5% mid pubertal 3.5% post pubertal | 80.6% pre-pubertal 13.9% mid pubertal 5.6% post pubertal | 0.279 |
| Waist-to-Hip ratio (WtH) | 0.85 ± 0.53 | 0.91 ± 0.61 | <0.001 * |
| Levels of exercise (hours/per week) | 6.16 ± 4.00 | 5.51 ± 3.29 | 0.359 |
| Family income (EUR) | 2.17 ± 0.66 | 1.89 ± 0.68 | 0.53 |
| Parents’ education (years) | 14.47 ± 2.30 | 14.29 ± 2.83 | 0.778 |
| Screen time (hours/per week) | 12.18 ± 9.6 | 15.6 ± 8.94 | 0.064 |
| STAIC-state scoring | 24.9 ± 4 | 27.96 ± 5.18 | 0.002 * |
| STAIC-trait scoring | 30.23 ± 5.78 | 30.18 ± 6.21 | 0.969 |
| CDI scoring | 5.27 ± 4.66 | 6.01 ± 4.70 | 0.438 |
| Total Body Water (% of body weight) | 60.2 ± 8.38 | 48.47 ± 6.80 | <0.001 * |
| Extracellular Water (ECW) (% of body weight) | 52.58± 4.72 | 46.88 ± 5.05 | <0.001 * |
| Intracellular Water (% of body weight) | 47.43 ± 4.71 | 53.12 ± 5.05 | <0.001 * |
| Fat Free Mass (FFM) (% of body weight) | 89.9 ± 6.18 | 70.72 ± 7.48 | <0.001 * |
| Fat Free Mass (Kg) | 27.58 ± 6.32 | 34.91 ± 10.42 | <0.001 * |
| Fat Mass (FM) (% of body weight) | 10.1 ± 6.18 | 29.28 ± 7.48 | <0.001 * |
| Fat Mass (Kg) | 3.49 ± 3.17 | 15.07 ± 8.45 | <0.001 * |
| Glycogen (% of body weight) | 0.74 ± 0.13 | 0.81 ± 0.11 | <0.001 * |
| Abdominal adipose tissue (% of body weight) | 12.64 ± 7.75 | 36.59 ± 9.38 | <0.001 * |
| Abdominal adipose tissue (Kg) | 4.37 ± 3.97 | 18.84 ± 10.57 | <0.001 * |
| Visceral organs tissue (Kg) | 16.83 ± 4.47 | 15.89 ± 2.85 | 0.046 * |
| Skeletal muscle mass (Kg) | 7.69 ± 2.79 | 11.03 ± 4.35 | <0.001 * |
| Skeletal muscle mass (% of body weight) | 27.59 ± 6.17 | 33.07 ± 5.66 | <0.001 * |
| Body Density | 1.06 ± 0.13 | 1.02 ± 0.15 | <0.001 * |
| Phase angle | 3.02 ± 0.49 | 2.92 ± 0.78 | 0.454 |
| Resting heart rate (RHR) (pulses/min) | 85.98 ± 11.5 | 83.97 ± 10.69 | 0.323 |
| SDNN (ms) | 133.026 ± 216.58 | 109.025 ± 121.78 | 0.441 |
| Scatter area (ms²) | 195.9 × 103 ± 64.6 × 103 | 80.5 × 103 ± 383.1 × 103 | 0.225 |
| LF power | 7.23 ± 1.63 | 7.44 ± 1.11 | 0.410 |
| HF power | 7.64 ± 1.88 | 7.76 ± 1.62 | 0.717 |
| LF/HF ratio | 0.833 ± 0.71 | 0.82 ± 0.66 | 0.920 |
| Dependent Variable | p-Value | FDR Adjusted ai |
|---|---|---|
| AUCg | 0.200 | 0.029 |
| Hair cortisol concentration (HCC) (pg/mg) | 0.917 | 0.046 |
| Serum cortisol (mcg/dL) | 0.697 | 0.033 |
| hsCRP_(mg/L) | 0.028 | 0.013 |
| FMP (%) | <0.001 | 0.004 |
| Insulin (μU/mL) | 0.028 | 0.017 |
| White blood cells count (WBC × 109/L) | 0.008 | 0.009 |
| HCt (%) | 0.719 | 0.038 |
| Red cell distribution width (RDW %) | 0.114 | 0.025 |
| Iron (mcg/dL) | 0.960 | 0.050 |
| Ferritin (ng/mL) | 0.093 | 0.021 |
| Glucose (mg/dL) | 0.823 | 0.042 |
| Normal Weight (N = 40) | Overweight/Obese (N = 81) | p Value | |
|---|---|---|---|
| Hair cortisol concentration (pg/mg) | 2.98 ± 5.41 | 3.16 ± 2.53 | 0.829 |
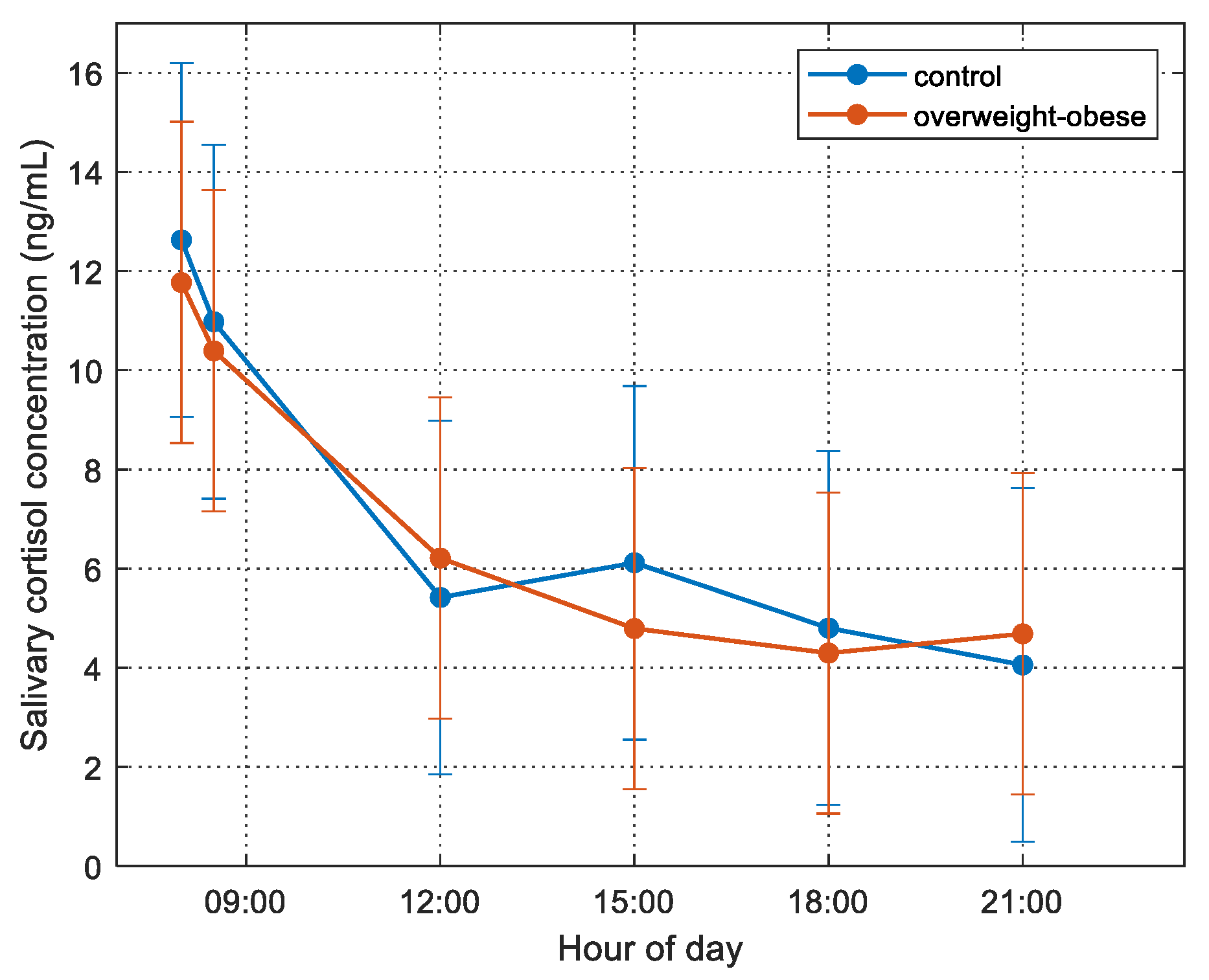
| Morning salivary cortisol (first sample of the day) (ng/mL) | 12.32 ± 5.79 | 11.79 ± 6.41 | 0.674 |
| Red cell distribution width (RDW %) | 13.45 ± 0.85 | 13.87 ± 1.11 | 0.026 * |
| White blood cells count (WBC × 109/L) | 6.72 ± 1.42 | 6.8 ± 1.72 | 0.811 |
| Iron (mcg/dL) | 98.1 ± 26.59 | 81.21 ± 24.85 | 0.002 * |
| Ferritin (ng/mL) | 43.06 ± 22.44 | 49.40 ± 28.46 | 0.242 |
| Serum cortisol (mcg/dL) | 13.48 ± 6.07 | 12.39 ± 5.74 | 0.369 |
| Insulin (μU/mL) | 5.95 ± 2.75 | 11.87 ± 7.78 | <0.001 * |
| Uric acid(mg/dL) | 3.75 ± 0.62 | 4.37 ± 0.94 | <0.001 * |
| Aspartate transaminase (SGOT) (U/L) | 29.00 ± 13.11 | 27.01 ± 13.27 | 0.448 |
| Serum glutamic pyruvic transaminase (SGPT) (U/L) | 17.47 ± 9.17 | 25.19 ± 28.74 | 0.11 |
| Gamma-glutamyl Transferace (γGT)(U/L) | 11.51 ± 3.19 | 14.91 ± 6.05 | 0.002 * |
| Triglycerides (mg/dL) | 52.84 ± 16.76 | 74.68 ± 44.94 | 0.005 * |
| Total Cholesterol (mg/dL) | 166.32 ± 27.62 | 161.90 ± 29.6 | 0.441 |
| Low-density lipoprotein (mg/dL) | 93.73 ± 22.95 | 92.89 ± 25.3 | 0.863 |
| High-density lipoprotein (mg/dL) | 61.97 ± 11.28 | 54.58 ± 12.87 | 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christaki, E.V.; Pervanidou, P.; Papassotiriou, I.; Bastaki, D.; Valavani, E.; Mantzou, A.; Giannakakis, G.; Boschiero, D.; Chrousos, G.P. Stress, Inflammation and Metabolic Biomarkers Are Associated with Body Composition Measures in Lean, Overweight, and Obese Children and Adolescents. Children 2022, 9, 291. https://doi.org/10.3390/children9020291
Christaki EV, Pervanidou P, Papassotiriou I, Bastaki D, Valavani E, Mantzou A, Giannakakis G, Boschiero D, Chrousos GP. Stress, Inflammation and Metabolic Biomarkers Are Associated with Body Composition Measures in Lean, Overweight, and Obese Children and Adolescents. Children. 2022; 9(2):291. https://doi.org/10.3390/children9020291
Chicago/Turabian StyleChristaki, Eirini V., Panagiota Pervanidou, Ioannis Papassotiriou, Despoina Bastaki, Eleni Valavani, Aimilia Mantzou, Giorgos Giannakakis, Dario Boschiero, and George P. Chrousos. 2022. "Stress, Inflammation and Metabolic Biomarkers Are Associated with Body Composition Measures in Lean, Overweight, and Obese Children and Adolescents" Children 9, no. 2: 291. https://doi.org/10.3390/children9020291
APA StyleChristaki, E. V., Pervanidou, P., Papassotiriou, I., Bastaki, D., Valavani, E., Mantzou, A., Giannakakis, G., Boschiero, D., & Chrousos, G. P. (2022). Stress, Inflammation and Metabolic Biomarkers Are Associated with Body Composition Measures in Lean, Overweight, and Obese Children and Adolescents. Children, 9(2), 291. https://doi.org/10.3390/children9020291








