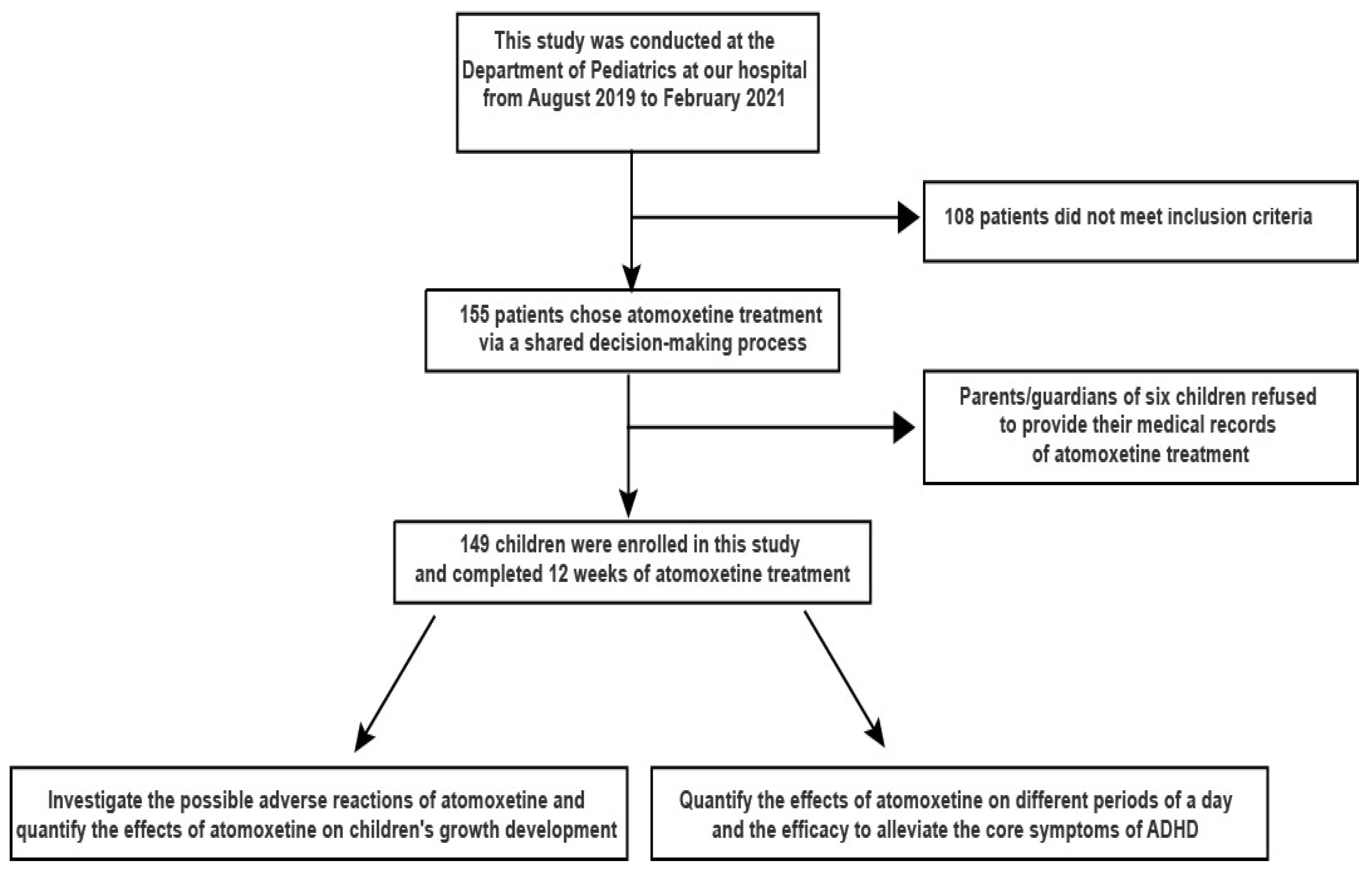
1. Introduction
Attention-deficit/hyperactivity disorder (ADHD) is the most common childhood neurodevelopmental disorder, with a prevalence of approximately 5% worldwide [
1]. It is defined as a persistent pattern of inattention, hyperactivity, and impulsivity that significantly impedes children’s academic and social development [
1,
2,
3]. If left untreated, ADHD may cause long-term morbidity and poor quality of life in adulthood [
2]. Therefore, school-aged children with ADHD must receive essential medical treatment.
Behavioral and psychosocial treatments (BPT) are the recommended nonpharmacological treatments to alleviate core the symptoms of ADHD, and they are the first-line treatments for very young children (<5 years old) [
4,
5]. However, the efficiency of BPT remains questionable, with controversial evidence and conclusions in previous studies [
5]. Pharmacological treatment is still widely used as ADHD treatment in most clinical practices and guidelines [
6,
7,
8]. The current standard pharmacological treatment for primary school children with ADHD involves using stimulants or non-stimulants that regulate catecholamines [
3]. In China, methylphenidate is widely used as a central nervous system stimulant, while atomoxetine is used as a non-stimulant. Atomoxetine was the first non-stimulant medication approved by the United States Food and Drug Administration (FDA) to treat ADHD patients aged over six years [
9]. Methylphenidate is a short-acting stimulant that is well tolerated in the short term. It reduces height and weight in the long term [
10,
11], while the short-term effects of atomoxetine on growth development are not well documented. Long-term atomoxetine treatment may be associated with deficits in height growth [
10]. In addition, some studies have indicated that atomoxetine might have additional side effects beyond norepinephrine reuptake inhibition. Hence, the safety and efficacy of this treatment require further investigation [
11,
12]. Therefore, this study was conducted to investigate the short-term adverse effects of atomoxetine, and its potential impacts on height and weight changes in primary school children.
Studies performed during the last decade indicate that the development of ADHD involves a complex interaction of the neurobiological and neurochemical systems. However, the precise etiology and pathophysiology behind this disease are still not fully understood [
8,
13,
14]. Neurotrophins, which include brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and insulin-like growth factor-1 (IGF-1), are a family of proteins that regulate neural growth, survival, and differentiation [
15,
16]. Recently, animal and clinical studies have indicated that the brain-derived neurotrophic factors involved the pathophysiology of ADHD and could be potential biomarkers [
17,
18]. IGF-1 is one of the more frequently investigated neurotrophins. It is the principal mediator of growth hormone (GH), and plays a critical role in regulating both anabolic and catabolic pathways in skeletal muscle, and in promoting bone formation and growth [
19,
20]. In addition, the IGF-1/IGFBP-3 axis was considered to be a potential biochemical growth maturity indicator, especially in children [
21,
22,
23,
24]. Previous studies applied IGF-1, IGFBP-3, and thyroid function tests as growth parameters to quantify the effects of methylphenidate on children’s growth development [
25,
26,
27]. However, few studies have explored the impact of atomoxetine on children’s growth development according to these three growth parameters. Hence, this study investigated the changes in serum IGF-1, IGFBP-3 levels, thyroid function, total protein (TP), albumin (ALB), and hemoglobin (HGB) to quantify the effects of atomoxetine on children’s growth development in the short term.
Although it is well known that primary school children with ADHD often present with difficulties in the morning prior to school and in the evening, these two periods, and the family interactions that they entail, are often neglected in studies of ADHD [
28,
29]. A novel phase III delayed release and extended release methylphenidate (HLD200) was designed to provide an extended duration of release from the morning prior to school, throughout the day, and into the evening [
28,
30,
31,
32]. Kelsey et al.’s study indicated that a once-daily administration of atomoxetine in the morning provided safe, rapid, and continuous symptom relief throughout the day [
33]. However, information about the effects of atomoxetine on different periods of the day is limited. Questionnaire–Children with Difficulties (QCD) has been widely used in China to evaluate parents’ perceptions of ADHD and patients’ daily behaviors during different times of the day, such as in the morning, during school, after school, in the evening, and at night time [
34,
35]. Therefore, this study first used the QCD to quantify the efficacy of atomoxetine treatment in improving behavioral difficulties at different times.
4. Discussion
The current prospective study investigated the improvement of behavioral difficulties at different times after a 12-week atomoxetine treatment for primary children diagnosed with ADHD aged between 6 and 12 years, and the possible influence on the growth parameters of this treatment. Although behavioral and psychosocial treatments were recommended before pharmacological therapy by the United Kingdom and Germany [
50,
51], effective behavioral and psychosocial treatments are still more expensive, time-consuming, and less available than pharmacological treatment in medically underserved areas [
5]. There are only two drugs, methylphenidate and atomoxetine, approved as the first line to treat ADHD by the China Food and Drug Administration (CFDA,
https://www.nmpa.gov.cn, accessed on 16 October 2021). At the same time, clonidine and bupropion were recommended as the second-line medications [
36]. As a non-stimulant, atomoxetine works by inhibiting the reuptake of norepinephrine, and effectively increases norepinephrine concentration in the synaptic cleft [
1]. To decide upon a first-line pharmacological treatment when choosing between methylphenidate and atomoxetine, the guardians or parents must be fully informed about the adverse reactions at first. This shared decision-making process needs physicians to cooperate with the guardians or parents. In China, the guardians or parents prefer to choose the atomoxetine treatment for the following reasons: (1) fewer adverse reactions on height; (2) they hope the effect of the drug will last throughout the day; (3) the ADHD symptoms are not severe and immediate onset is not needed; (4) concerns about drug abuse [
5,
52,
53]. Short stature and growth deficits remain the most common pediatric concerns, and this is mainly presented with growth hormone (GH) deficiency accompanied by lower serum IGF-1 levels [
54,
55]. However, non-GH deficient short stature disorders, such as idiopathic short stature (ISS) and growth hormone insensitivity (GHI), often present with normal GH when accompanied by lower serum IGF-1 levels [
56,
57]. Hence, the Growth Hormone Research Society (GRS) and the Chinese official guidelines for diagnosing and treating children with short stature recommended IGF-1 and its main binding protein, IGFBP-3, as valuable biomarkers in assessing growth deficits [
58,
59]. Our prospective study indicated that the use of atomoxetine significantly reduced the symptoms and behavioral difficulties experienced by children who have ADHD. Furthermore, the treatment was generally well tolerated, with few adverse events, and was not associated with growth deficits in the short term.
To the best of our knowledge, this is the first report using the QCD to examine the efficiency of atomoxetine for children aged 6–12 years old with ADHD, in terms of reducing the functional difficulties at different times. The QCD was created by Yamashita, and is widely used in Asian countries, such as China and Japan [
60]. The QCD has three key characteristics: the evaluation of life function, the assessment of life function at different periods of the day, and the convenience of use in daily clinical practice [
34,
35]. Compared with the Child Behavior Checklist (CBCL), which is often used in Western countries, QCD has shown its advantages because it includes only 20 practical and easy-to-understand questions, and only a short time is needed to complete the QCD [
61]. The QCD also provides physicians with the necessary information for selecting appropriate drug therapy [
34]. In this study, we focused on the two periods that have often been neglected in previous studies of ADHD. The effect of atomoxetine in the morning prior to school (
p < 0.001, r = 0.66) and in the evening (
p < 0.001, r = 0.73) was assessed by comparing QCD scores at different times. Our QCD results indicated that atomoxetine could significantly reduce the functional difficulties experienced by ADHD children in these two periods. Family interactions were another neglected point in previous studies of ADHD [
29]. Although it is well known that primary school children with ADHD often present with difficulties in the morning prior to school and in the evening [
28], previous studies have indicated that parents find it more challenging to take care of ADHD children in the evening. They have reported difficulties in persuading their children to complete their homework and enjoy family time [
62,
63]. Hence, alleviating the core symptoms of ADHD in the morning prior to school and in the evening may effectively solve this problem and reduce parental stress levels. Minimizing parenting stress and improving family interactions are important because they can reduce the risk of adverse outcomes in the long term [
64,
65,
66]. Consistent with our QCD results, atomoxetine treatment may be a choice in China to alleviate parental stress and to improve family interactions in children with ADHD. This study also compared the SNAP-IV scale and CPRS after 12 weeks of treatment with atomoxetine, which further confirmed that atomoxetine is an effective treatment for primary school children with ADHD. The SNAP-IV results showed a significant improvement in the core symptoms of ADHD, while the CPRS results indicated a significant improvement in controlling ADHD symptoms across two different domains, learning problems (r = 0.81) and hyperactivity (r = 0.86). Consistent with previous studies [
67,
68,
69], our findings indicate that atomoxetine can reduce the core symptoms in primary school children with ADHD.
In this study, the levels of serum growth parameters were closely monitored throughout treatment. These markers have essential roles in promoting children’s growth and development [
70,
71,
72]. In contrast to the BDNF that was lower in newly diagnosed ADHD patients [
73], this study found that the serum levels of IGF-1 and IGFBP-3 were within the normal range in newly diagnosed primary school patients with ADHD. These results indirectly confirmed that the secretion and role of endogenous growth hormones were normal in newly diagnosed primary school patients with ADHD. In addition, there was no difference in the levels of IGF-, IGFBP-3, and thyroid function before and after 12 weeks of atomoxetine treatment. A mice model study also showed that atomoxetine did not reduce the expression of the IGF-1 gene [
74]. Our serum biochemical results suggested that the atomoxetine did not affect the level of serum growth parameters in children. However, whether the treatment of atomoxetine will affect the levels of serum growth parameters in primary school children with ADHD in the long term remains to be further studied and discussed.
Our study reported no severe adverse reactions, such as suicide attempt or suicidal ideation, throughout treatment. The most significant initial adverse reactions were gastrointestinal symptoms, including the loss of appetite or anorexia, constipation, nausea, and vomiting. These symptoms might be caused by temporary disorders of the central norepinephrine system associated with hunger or satiety [
28]. Interestingly, there was no significant difference in the levels of ALB, HGB, and HCT after treatment compared with the baseline. This indicated that, although atomoxetine could cause gastrointestinal adverse reactions, it did not affect the nutritional status of ADHD children. In addition, the height and BMI of children did not lag behind children of the same age without ADHD, and the serum growth parameters were also normal. In our study, despite a significant weight reduction being noted after treatment (
p < 0.05), the effect size was slight (r = 0.09). A survey by Germinario revealed that the height and weight gradually returned to normal after two years of ADHD treatment. This improvement was more evident for atomoxetine when compared with treatment using methylphenidate [
75]. Consistent with our previous results, we speculated that atomoxetine might lead to temporary weight loss due to a loss of appetite. This factor may affect the selection of the short-term pharmacological treatment in China. Based on the guidelines from National Institute for Health and Care Excellence (NICE), temporary weight loss does not require particular caution. If weight loss is of clinical concern, medication should be taken either during or after meals rather than before meals, and additional meals or snacks early in the morning or late in the evening are also suggested [
51].