Effects on Recovery of Pediatric Patients Undergoing Total Intravenous Anesthesia with Propofol versus Ketofol for Short—Lasting Laparoscopic Procedures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Intraoperative Monitoring
2.3. Study Design
2.4. Outcomes of the Study
2.5. Sample Size Calculation
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dagorno, C.; Montalva, L.; Ali, L.; Brustia, R.; Paye-Jouaen, A.; Pio, L.; Bonnard, A. Enhancing recovery after minimally invasive surgery in children: A systematic review of the literature and meta-analysis. J. Pediatr. Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Pogorelić, Z.; Huskić, D.; Čohadžić, T.; Jukić, M.; Šušnjar, T. Learning curve for laparoscopic repair of pediatric inguinal hernia using percutaneous internal ring suturing. Children 2021, 8, 294. [Google Scholar] [CrossRef]
- Pogorelić, Z.; Čohadžić, T.; Jukić, M.; Nevešćanin Biliškov, A. Percutaneous internal ring suturing for the minimal invasive treatment of pediatric inguinal hernia: A 5-year single surgeon experience. Surg. Laparosc. Endosc. Percutan. Tech. 2021, 31, 150–154. [Google Scholar] [CrossRef]
- Pogorelić, Z.; Sopta, M.; Jukić, M.; Nevešćanin, A.; Jurić, I.; Furlan, D. Laparoscopic varicocelectomy using polymeric ligating clips and its effect on semen parameters in pediatric population with symptomatic varicocele: A 5-year single surgeon experience. J. Laparoendosc. Adv. Surg. Tech. A 2017, 27, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Short, S.M.; Aun, C.S. Haemodynamic effects of propofol in children. Anaesthesia 1991, 46, 783–785. [Google Scholar] [CrossRef]
- Kanaya, A. Emergence agitation in children: Risk factors, prevention, and treatment. J. Anesth. 2016, 30, 261–267. [Google Scholar] [CrossRef]
- Biricik, E.; Karacaer, F. Comparasion of TIVA with different combinations of ketamine-propofol mixtures in pediatric patients. J. Anesth. 2018, 32, 104–111. [Google Scholar] [CrossRef]
- Aouad, M.T.; Moussa, A.R.; Dagher, C.M.; Muwakkit, S.A.; Jabbour- Khoury, S.I.; Zbeidy, R.A.; Abboud, M.R.; Kanazi, G.E. Addition of ketamine to propofol for initiation of procedural anesthesia in children reduces propofol consumption and preserves hemodynamic stability. Acta Anaesthesiol. Scand. 2008, 52, 561–565. [Google Scholar] [CrossRef]
- Smischney, N.J.; Beach, M.L.; Loftus, R.W.; Dodds, T.M.; Koff, M.D. Ketamine/propofol admixture (ketofol) is associated with improved hemodynamics as induction agent: A randomized, controlled trial. J. Trauma Acute Care Surg. 2012, 73, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Weatherall, A.; Venclovas, R. Experience with a propofol–ketamine mixture for sedation during pediatric orthopedic surgery. Pediatr. Anesth. 2010, 20, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Trissel, L.A.; Gilbert, D.L.; Martinez, J.F. Compatibility of propofol injectable emulsion with selected drugs during simulated Y-site administration. Am. J. Health Syst. Pharm. 1997, 54, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Andolfatto, G.; Willman, E. A prospective case series of pediatric procedural sedation and analgesia in emergency department using single-syringe ketamine–propofol combination (ketofol). Acad. Emerg. Med. 2010, 17, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, K.; Inomata, S.; Okubo, N.; Watanabe, I. Pretreatment with small-dose ketamine reduces predicted effect-site concentration of propofol required for loss of consciousness and laryngeal mask airway insertion in women. J. Clin. Anesth. 2011, 23, 113–118. [Google Scholar] [CrossRef]
- Camu, F.; Vanlersberghe, C. Pharmacology of systemic analgesics. Best Pract. Res. Clin. Anaesthesiol. 2002, 16, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Coulter, F.L.; Hannam, J.A.; Anderson, B.J. Ketofol simulation for dosing in pediatric anesthesia. Pediatr. Anesth. 2014, 24, 806–812. [Google Scholar] [CrossRef] [Green Version]
- McFarlan, C.S.; Anderson, B.J.; Short, T.G. The use of propofol infusions in pediatric anesthesia: A practical guide. Pediatr. Anesth. 1999, 9, 209–216. [Google Scholar]
- Nevešćanin, A.; Vickov, J.; Elezović Baloević, S.; Pogorelić, Z. Laryngeal mask airway versus tracheal intubation for laparoscopic hernia repair in children: Analysis of respiratory complications. J. Laparoendosc. Adv. Surg. Tech. A 2020, 30, 76–80. [Google Scholar] [CrossRef]
- Aldrete, J.A. The post-anesthesia recovery score revisited. J. Clin. Anesth. 1995, 7, 89–91. [Google Scholar] [CrossRef]
- Chandler, J.R.; Myers, D.; Mehta, D.; Whyte, E.; Groberman, M.K.; Montgomery, C.J.; Ansermino, J.M. Emergence delirium in children: A randomized trial to compare total intravenous anesthesia with propofol and remifentanil to inhalational sevoflurane anesthesia. Pediatr. Anesth. 2013, 23, 309–315. [Google Scholar] [CrossRef]
- Louvet, N.; Rigouzzo, A.; Sabourdin, N.; Constant, I. Bispectral index under propofol anesthesia in children: A comparative randomized study between TIVA and TCI. Pediatr. Anesth. 2016, 26, 899–908. [Google Scholar] [CrossRef]
- Lauder, G.R. Total intravenous anesthesia will supercede inhalational anesthesia in pediatric anesthetic practice. Pediatr. Anesth. 2015, 25, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Hig, C.C., Jr.; McLeskey, C.H.; Nahrwold, M.L.; Roizen, M.F.; Stanley, T.H.; Thisted, R.A.; Walawander, C.A.; White, P.F.; Apfelbaum, J.L.; Grasela, T.H.; et al. Hemodynamic effects of propofol: Data from over 25,000 patients. Anesth. Analg. 1993, 77, 21–29. [Google Scholar]
- Dallimore, D.; Anderson, B.J.; Short, T.G.; Herd, D.W. Ketamine anesthesia in children—Exploring infusion regimens. Pediatr. Anesth. 2008, 18, 708–714. [Google Scholar] [CrossRef]
- Herds, D.; Anderson, B.; Keene, N.A.; Holford, N.H. Investigating the pharmacodynamics of ketamine in children. Pediatr. Anesth. 2008, 18, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Ayatollahi, V.; Vafaiyan, M.; Hatami, M.; Behdad, S. Two different concentrations of ketofol for procedural sedation and analgesia in closed reduction of nasal fracture. J. Craniofac. Surg. 2016, 27, 996–1000. [Google Scholar] [CrossRef] [PubMed]
- Daabis, M.; Elsherbiny, M.; AlOtibi, R. Assessment of different concentrations of ketofol in procedural operation. BJMP 2009, 2, 27–31. [Google Scholar] [CrossRef]
- Coulter, F.L.; Hannam, J.A.; Anderson, B.J. Ketofol dosing simulations for procedural sedation. Pediatr. Emerg. Care 2014, 30, 621–630. [Google Scholar] [CrossRef]
- Bray, R.J. The propofol infusion syndrome in infants and children: Can we predict the risk? Curr. Opin. Anaesthesiol. 2002, 15, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Hans, P.; Dewandre, P.Y.; Brichant, J.F.; Bonhomme, V. Comparative effects of ketamine on bispectral index and spectral entropy of the electroencephalogram under sevoflurane anesthesia. Br. J. Anaesth. 2005, 94, 336–340. [Google Scholar] [CrossRef] [Green Version]
- Vereecke, H.E.; Struys, M.M.; Mortier, E.P. A comparison of bispectral index and ARX derived auditory evoked potential index in measuring the clinical interaction between ketamine and propofol anaesthesia. Anaesthesia 2003, 58, 957–961. [Google Scholar] [CrossRef]



| Group I | Group II | p | |
|---|---|---|---|
| Propofol (n = 100) | Ketofol (n = 100) | ||
| Demographic characteristics of patients | |||
| Age (years) median (IQR) | 5 (3, 5) | 5 (3, 5.5) | 0.144 * |
| Gender, n (%) Male Female | 75 (75) 25 (25) | 77 (77) 23 (23) | 0.740 † |
| Weight (g) median (IQR) | 20 (14.5, 29) | 19.5 (13, 27) | 0.139 * |
| Hemodynamic characteristics of patients, median (IQR) | |||
| Systolic blood pressure (mmHg) | 117 (110, 124) | 119 (108, 123) | 0.530 * |
| Diastolic arterial pressure (mmHg) | 71 (66, 76) | 69 (64, 76) | 0.365 * |
| Heart rate (bpm) | 128 (100, 147) | 126 (103, 146) | 0.235 * |
| Group I | Group II | p | |
|---|---|---|---|
| Propofol (n = 100) | Ketofol (n = 100) | ||
| Sefofluran (for iv. canula), n (%) | 57 (57) | 64 (64) | 0.311 * |
| Length of iv infusion (min) median (IQR) | 24.5 (19, 30) | 23 (16, 30) | 0.371 † |
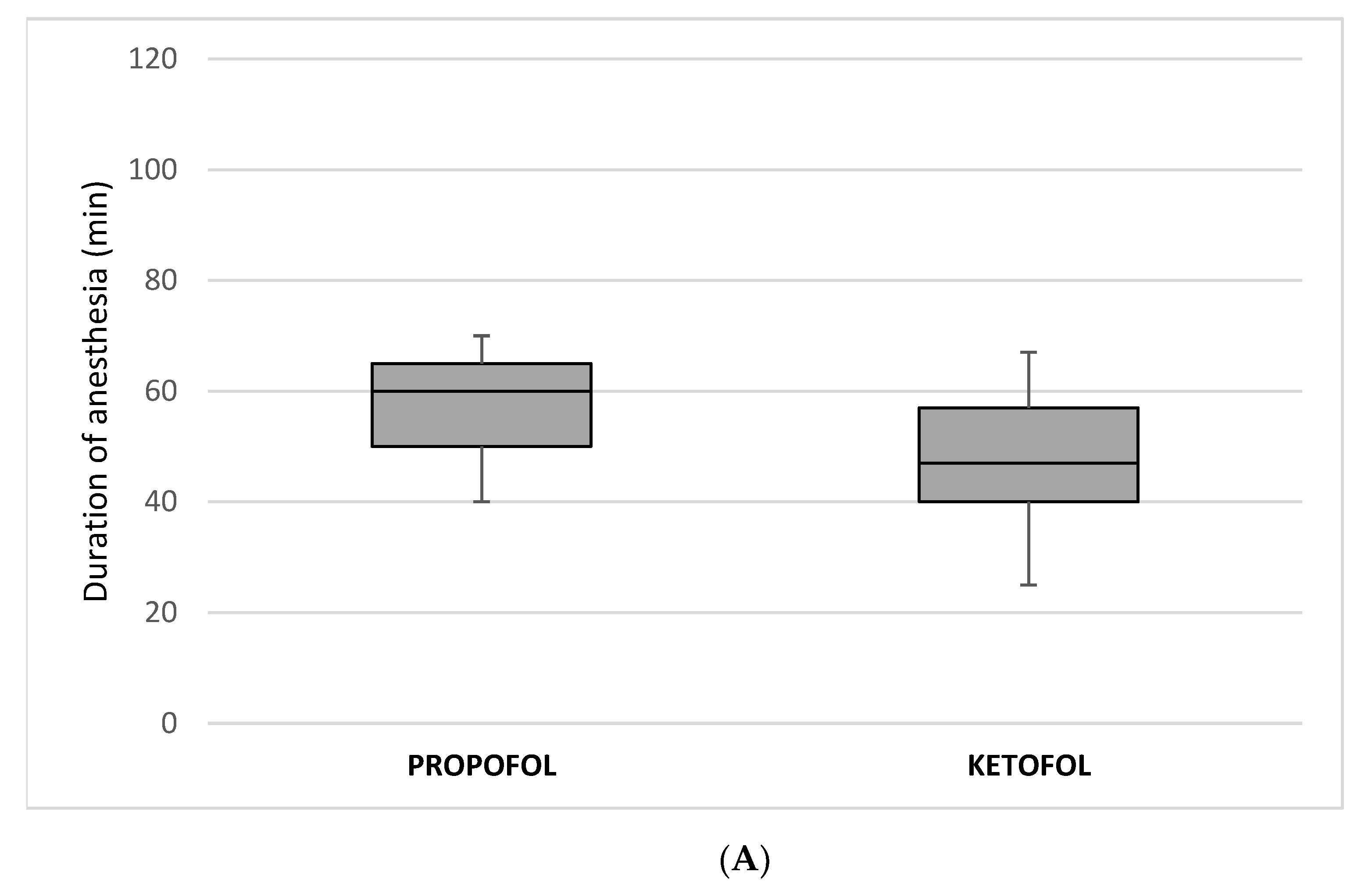
| Duration of anesthesia (min) median (IQR) | 60 (50, 65) | 47 (40, 57) | <0.00001 † |
| Extubation time (s) median (IQR) | 530 (410, 600) | 240 (120, 330) | <0.00001 † |
| Fentanyl (μg/kg) median (IQR) | 4 (3, 5) | 2 (1, 2) | <0.00001 † |
| Fentanyl—TOTAL (μg) median (IQR) | 100 (80, 125) | 50 (40, 60) | <0.00001 † |
| Propofol (mg/kg) median (IQR) | 13 (9, 15) | 5.8 (4, 7.5) | <0.00001 † |
| Propofol—TOTAL (mg) median (IQR) | 260 (200, 350) | 160 (120, 210) | <0.00001 † |
| Group I | Group II | p | |
|---|---|---|---|
| Propofol | Ketofol | ||
| (n = 100) | (n = 100) | ||
| SpO2 PACU—at arrival median (IQR) | 93 (92, 94.5) | 99 (98, 99) | <0.00001 * |
| SpO2 PACU—after 30 min median (IQR) | 98 (97, 99) | 99 (98, 100) | <0.00001 * |
| Length of stay in PACU (min) median (IQR) | 100 (90, 110) | 35 (30, 35) | <0.00001 * |
| Number of patients requiring oxygen in PACU, n (%) | 56 (56%) | 0 (0%) | <0.00001 † |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biliškov, A.N.; Ivančev, B.; Pogorelić, Z. Effects on Recovery of Pediatric Patients Undergoing Total Intravenous Anesthesia with Propofol versus Ketofol for Short—Lasting Laparoscopic Procedures. Children 2021, 8, 610. https://doi.org/10.3390/children8070610
Biliškov AN, Ivančev B, Pogorelić Z. Effects on Recovery of Pediatric Patients Undergoing Total Intravenous Anesthesia with Propofol versus Ketofol for Short—Lasting Laparoscopic Procedures. Children. 2021; 8(7):610. https://doi.org/10.3390/children8070610
Chicago/Turabian StyleBiliškov, Ana Nevešćanin, Božena Ivančev, and Zenon Pogorelić. 2021. "Effects on Recovery of Pediatric Patients Undergoing Total Intravenous Anesthesia with Propofol versus Ketofol for Short—Lasting Laparoscopic Procedures" Children 8, no. 7: 610. https://doi.org/10.3390/children8070610
APA StyleBiliškov, A. N., Ivančev, B., & Pogorelić, Z. (2021). Effects on Recovery of Pediatric Patients Undergoing Total Intravenous Anesthesia with Propofol versus Ketofol for Short—Lasting Laparoscopic Procedures. Children, 8(7), 610. https://doi.org/10.3390/children8070610







