Effect of Dog-Assisted Therapy on Psychomotor Development of Children with Intellectual Disability
Abstract
1. Introduction
- Affective—related to feelings aroused in a person by the animal;
- Psychological stimulation—interaction with the animal, stimulating social behaviours and cognitive functions;
2. Materials and Methods
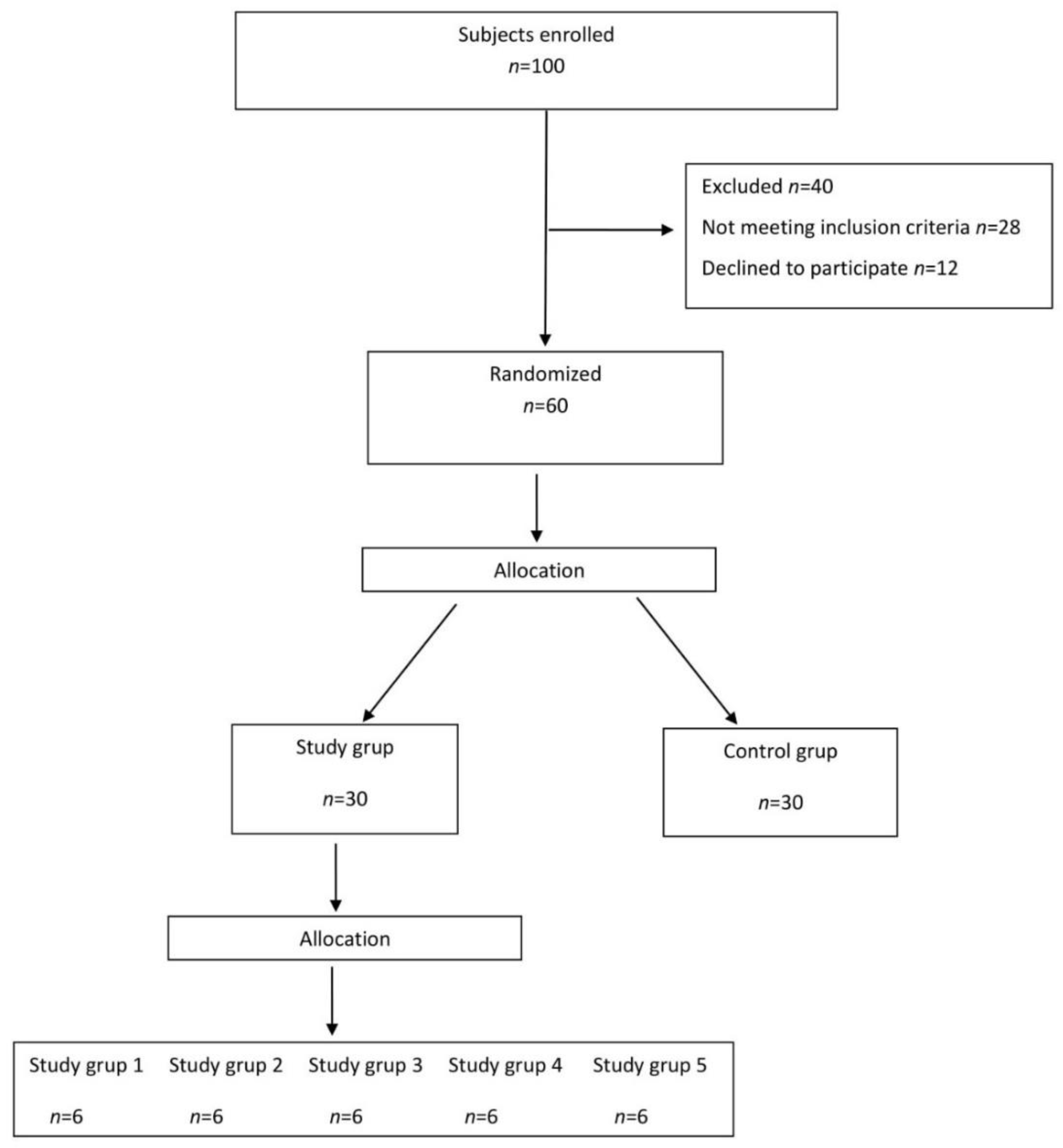
2.1. Participants
2.2. Flow of the Subjects through the Study
2.3. Study Design
2.4. Study Protocol
2.5. Measurements
- -
- Bourdon–Wiersma Dot Cancellation Test—assessing concentration and attention. The subject is shown a sheet with a sequence of various letters and digits and is asked to quickly cross out specific letters, e.g., e and r, within 3 min. The result is based on the total number of characters crossed out correctly, and a total number of characters skipped and selected mistakenly. Each type of error may be indicative of an impairment in different processes of attention, e.g., weakening. A maximum of 102 points can be scored. In the entire sheet, the digit 6 appears 102 times. One point is scored for each digit 6 crossed out correctly [29].
- -
- The Southern California Sensory Integration Tests (SCSIT Battery) proposed by J. Ayres, based on Southern California Sensory Integration Tests Manual, Los Angeles, Calif., Western Psychological Services [30,31]. The battery is designed to evaluate sensory integration by assessing e.g., kinaesthetic sense, perception of tactile stimuli, ability to visualise tactile sensations without visual control, ability to sense the location of tactile stimuli, as well as movement planning. The subtests used in the present study include postural imitation test—assessing motor planning and sequencing. A maximum of 12 points can be scored. The child is awarded 1 point if they correctly assume a position or 0 points if they fail to assume the position; finger identification test—assessing the sense of touch, attention and concentration. A maximum of 16 points can be scored, one point for each correct answer; as well as kinaesthesia test—assessing kinaesthetic sense, spatial positioning of extremities, and memory. A maximum of 24 points can be scored. The child is to indicate 12 locations where tactile stimuli were applied. Two points are scored for each correctly indicated location. If the child points to the correct part of the limb but not to the precise location, they score 1 point. If they fail to correctly indicate the location or the part of the limb, they score 0 points.
2.6. Procedure
2.7. Statistical Analysis
2.8. Sample Size
- Nmin—minimum sample size
- NP—size of the population sampled
- α—confidence level for the results, value of Z-score in normal distribution for the assumed significance level, e.g., 1.96
- f—fraction size
- e—assumed maximum error expressed with a fractional number, e.g., 3% is expressed as 0.03
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Widmar, D.H.; Feuillan, K.A. Animal-assisted therapy. In Physical Medicine and Rehabilitation—The Complete Approach; Grabois, M., Garrison, S.J., Hart, K.A., Lehmkuhl, L.D., Eds.; Blackwell: Oxford, UK, 2000; Volume 733, p. 61. [Google Scholar]
- Maber-Aleksandrowicz, S.; Avent, C.; Hassiotis, A. A systematic review of animal-assisted therapy on psychosocial outcomes in people with intellectual disability. Res. Dev. Disabil. 2016, 49–50, 322–338. [Google Scholar] [CrossRef] [PubMed]
- Silkwood-Sherer, D.J.; Killian, C.B.; Long, T.M.; Martin, K.S. Hip-potherapy—An intervention to habilitate balance deficits in children with movement disorders: A clinical trial. Phys. Ther. 2012, 92, 17–707. [Google Scholar] [CrossRef] [PubMed]
- Tseng, S.H.; Cheng, H.C.; Tam, K.W. Systematic review and meta-analysis of the effect of equine assisted activities and therapies on gross motor outcome in children with cerebral palsy. Disabil. Rehabil. 2013, 35, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Zadnikar, M.; Kastrin, A. Effects of hippotherapy and therapeutic horseback riding on postural control or balance in children with cerebral palsy:a meta-analysis. Dev. Med. Child Neurol. 2011, 53, 684–691. [Google Scholar] [CrossRef]
- Schuck, S.E.B.; Emmerson, N.A.; Fine, A.H.; Lakes, K.D. Canine-assisted therapy for children with ADHD: Preliminary findings from the Positive Assertive Cooperative Kids study. J. Attent. Disor. 2015, 19, 125–137. [Google Scholar] [CrossRef]
- Kwon, J.Y.; Chang, H.J.; Lee, J.Y.; Ha, Y.; Lee, P.K.; Kim, Y.H. Effects of hippotherapy on gait parameters in children with bilateral spastic cerebral palsy. Arch. Phys. Med. Rehabil. 2011, 92, 774–779. [Google Scholar] [CrossRef]
- Lundqvist, M.; Carlsson, P.; Sjödahl, R.; Theodorsson, E.; Levin, L.Å. Patient benefit of dog-assisted interventions in health care: A systematic review. BMC Complementary Altern. Med. 2017, 17, 358. [Google Scholar] [CrossRef]
- Muñoz Lasaa, S.; Máximo Bocanegrab, N.; Valero Alcaidea, R. Animal assisted interventions in neurorehabilitation: A review of the most recent literature. Neurology 2015, 30, 1–7. [Google Scholar] [CrossRef]
- Piek, J.P.; McLaren, S.; Kane, R.; Jensen, L.; Dender, A.; Roberts, C.; Rooney, R.; Packer, T.; Straker, L. Does the Animal Fun program improve motor performance in children aged 4–6 years? Hum. Mov. Sci. 2013, 32, 1086–1096. [Google Scholar] [CrossRef]
- Jorge, A.; Rigoli, D.; Kane, R.; Melaren, S. Does Animal Fun improve aiming and catching and balance skills in youn children. Res. Dev. Disabil. 2019, 84, 122–130. [Google Scholar]
- Richeson, N. Effects of animal-assisted therapy on agitated behaviors and social interactions of older adults with dementia. Am. J. Alzheimers Dis. Other Demen. 2003, 18, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Kongable, L.G.; Buckwalter, K.C.; Stolley, J.M. The effects of pet therapy on the social behavior of institutionalized Alzheimer’s clients. Arch. Psychiatr. Nurse 1989, 3, 191–198. [Google Scholar]
- Scorzato, I.; Zaninotto, L.; Chiara, M.; Cavedon, L. Effects of Dog-Assisted Therapy on Comunication and Basic Social Skills of Adults with Intellectual Disabilities: A piot Study. Intellectal Dev. Disabil. 2017, 55, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Gochewa, V.; Hund-Georgiadis, M.; Hediger, K. Effects of animal assisted therapy on concentration and attention span in patients with acquired brain injury: A randomized controlled trial. Neuropsychology 2018, 32, 54–64. [Google Scholar] [CrossRef] [PubMed]
- McCullough, A.; Ruehrdanz, A.; Jenkins, M.A.; Gilmer, M.J.; Olson, J.; Pawar, A.; Holley, L.; Sierra-Rivera, S.; Linder, D.E.; Pichette, D.; et al. Measuring the Effects of an Animal-Assisted Intervention for Pediatric Oncology Patients and Their Parents: A Multisite Randomized Controlled Trial. J. Pediatr. Oncol. Nurs. 2018, 35, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Muela, A.; Balluerka, N.; Amiano, N.; Caldentey, M.A.; Aliri, J. Animal-assisted psychotherapy for young people with behavioural problems in residential care. Clin. Psychol. Psychother. 2017, 24, O1485–O1494. [Google Scholar] [CrossRef]
- Schuck, S.E.; Johnson, H.L.; Abdullah, M.M.; Stehli, A.; Fine, A.H.; Lakes, K.D. The Role of Animal Assisted Intervention on Improving Self-Esteem in Children with Attention Deficit/Hyperactivity Disorder. Front. Pediatr. 2018, 6, 300. [Google Scholar] [CrossRef]
- Hill, J.; Ziviani, J.; Driscoll, C.; Cawdell-Smith, J. Can Canine-Assisted Interventions Affect the Social Behaviours of Children on the Autism Spectrum? Rev. J. Autism. Dev. Disord. 2019, 6, 13–25. [Google Scholar] [CrossRef]
- Boguszewski, D.; Świderska, B.; Adamczyk, J.G.; Białoszewski, D. Evaluation of the effectiveness of the dog-assisted therapy in the rehabilitation of children with Down syndrome. Prelim. Rep. Eur. J. Clin. Exp. Med. 2013, 2, 194–202. [Google Scholar]
- Drwięga, G.; Pietruczuk, Z. Dog therapy as a form of supporting the development of a disabled child. Niepełnosprawność Zagadnienia Probl. Rozw. 2015, 3, 57–68. [Google Scholar]
- Grandgeorge, M.; Hausberger, M. Human-animal relationships: From daily life to animal-assisted therapies. Ann. Dell’ Inst. Super. Sanita 2011, 47, 397–408. [Google Scholar]
- Friedmann, E.; Katcher, A.H.; Lynch, J.J.; Thomas, S.A. Animal companions and one-year survival of patients after discharge from a coronary care unit. Public Health Rep. 1980, 95, 307–312. [Google Scholar] [PubMed]
- De Rose, P.; Cannas, E.; Cantiello, P.R. Donkey-assisted rehabilitation program for children: A pilot study. Ann. Dell’ Inst. Super. Sanita 2011, 47, 391–396. [Google Scholar]
- Ballarini, G. Pet therapy. Animals in human therapy. Acta Biol. Med. 2003, 74, 97–100. [Google Scholar]
- Keith, T.Z.; Fine, J.G.; Taub, G.E.; Reynolds, M.R.; Kranzler, J.H. Higher order, multisample, confirmatory factor analysis of the Wechsler Intelligence Scale for Children--Fourth Edition: What does it measure? Sch. Psychol. Rev. 2006, 35, 108–127. [Google Scholar] [CrossRef]
- Hrabok, M.; Brooks, B.L.; Fay-McClymont, T.B.; Sherman, E.M.S. Wechsler Intelligence Scale for Children-Fourth Edition (WISC-IV) Short-form validity: A comparison study in pediatric epilepsy. Child. Neuropsychol. 2012, 20, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Suresh, K. An overview of randomization techniques: An unbiased assessment of outcome in clinical research. J. Hum. Reprod Sci. 2011, 4, 8–11. [Google Scholar] [CrossRef]
- Grewelf, F. The Bourdon-Wiersma test. Folia Psychiatr. Neurol. Psychiatr. 1953, 56, 694–703. [Google Scholar]
- Ayres, A.J. Sensory Integration and the Child, 2nd ed.; Western Psychological Services: Los Angeles, CA, USA, 2004. [Google Scholar]
- Kilroy, E.; Aziz-Zadeh, L.; Cermak, S. Ayres theories of autism and sensory integration revisited: What contemporary neuroscience has to say. Brain Sci. 2019, 9, 68. [Google Scholar] [CrossRef]
- Czenczek-Lewandowska, E.; Leszczak, J.; Baran, J.; Weres, A.; Wyszyńska, J.; Lewandowski, B.; Dąbrowski, M.; Mazur, A. Levels of Physical Activity in Children and Adolescents with Type 1 Diabetes in Relation to the Healthy Comparators and to the Method of Insulin Therapy Used. Int. J. Environ. Res. Public Health 2019, 16, 3498. [Google Scholar] [CrossRef]
- Guzik, A.; Drużbicki, M.; Kwolek, A.; Przysada, G.; Bazarnik-Mucha, K.; Szczepanik, M.; Wolan-Nieroda, A.; Sobolewski, M. The paediatric version of Wisconsin gait scale, adaptation for children with hemiplegic cerebral palsy: A prospective observational study. BMC Pediatr. 2018, 18, 301. [Google Scholar] [CrossRef] [PubMed]
- Rusek, W.; Leszczak, J.; Baran, J.; Adamczyk, M.; Weres, A.; Baran, R.; Inglot, G.; Czenczek-Lewandowska, E.; Porada, S.; Pop, T. Role of body mass category in the development of faulty postures in school-age children from a rural area in south-eastern Poland: A cross-sectional study. BMJ Open 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Baran, J.; Weres, A.; Czenczek-Lewandowska, E.; Leszczak, J.; Kalandyk-Osinko, K.; Łuszczki, E.; Sobek, G.; Mazur, A. Excessive Gestational Weight Gain: Long-Term Consequences for the Child. J. Clin. Med. 2020, 9, 3795. [Google Scholar] [CrossRef] [PubMed]
- Dilek, T.E.; Sibel, C. Dog-assisted therapies and activities in rehabilitation of children with cerebral palsy and physical and mental disabilities. Int. J. Environ. Res. Public Health 2015, 12, 5046–5060. [Google Scholar]
- Fung, S.; Leung, A. Pilot Study Investigating the Role of Therapy Dogs in Facilitating Social Interaction among Children with Autism. J. Contemp. Psychother. 2014, 44, 253–262. [Google Scholar] [CrossRef]
- François, M.; Jennifer, F. Animal-Assisted Therapy for Children with Pervasive Developmental Disorders. West. J. Nurs. Res. 2002, 24, 657–670. [Google Scholar]
- Bülent, E.; Hlya, K.; Irem, D. Sensory integration and activities of daily living in children with developmental coordination disorder. Ital. J. Pediatr. 2012, 38, 14. [Google Scholar] [CrossRef]
- Schoemaker, M.M.; Wees, V.; Flapper, B.; Verheij-Jansen, N.; Scholten Jaegers, S.; Geuze, R.H. Perceptual skills of children with developmental coordination disorder. Hum. Mov. Sci. 2001, 20, 111–133. [Google Scholar] [CrossRef]
- Reed, R.; Ferrer, L.; Villegas, N. Natural healers: A review of animal assisted therapy and activities as complementary treatment for chronic conditions. Rev. Lat. Am. Enferm. 2012, 20, 612–618. [Google Scholar] [CrossRef][Green Version]
- Nawrocka-Rohnka, J. Dogtherapy as a method of supporting rehabilitation for child with disorder of development. Med. News 2010, 79, 304–310. [Google Scholar]
- Gee, N.R.; Harris, S.L.; Johnson, K.L. The role of therapy dogs in speed and accuracy to complete motor skills tasks for preschool children. Anthrozoos A Multidiscip. J. Interact. People Anim. 2007, 20, 375–386. [Google Scholar] [CrossRef]
- Bunker, L.K. The role of play and motor skill development in building children’s selfconfidence and self-esteem. Elem. Sch. J. Spec. Issue Sports Phys. Educ. 1991, 91, 467–471. [Google Scholar]
- Macauley, B. Animal-assisted therapy for persons with aphasia: A pilot study. J. Rehabil. Res. Dev. 2006, 43, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.F.; Teixeira, F.A.C.; Pereira, T.E. Sensorial Integration and Intellectual Disabilities: Influence of Psychomotor Intervention. IOSR J. Sports Phys. Educ. 2016, 3, 44–49. [Google Scholar]
- Lucas, B.R.; Elliott, E.J.; Coggan, S.; Pinto, R.Z.; Jirikowic, T.; McCoy, S.W.; Latimer, J. Interventions to improve gross motor performance in children with neurodevelopmental disorders: A meta-analysis. BMC Pediatr. 2016, 16, 193. [Google Scholar] [CrossRef]
- Wuang, Y.P.; Wang, C.C.; Huang, M.H.; Su, C.Y. Prospective study of the effect of sensory integration, neurodevelopmental treatment, and perceptual-motor therapy on the sensorimotor performance in children with mild mental retardation. Am. J. Occup. 2009, 63, 441–452. [Google Scholar] [CrossRef]
- Bukhovets, B.O.; Romanchuk, A.P. Bobath therapy in correction of psychomotor development of children with organic injuries CNS. J. Health Sci. 2014, 4, 71–78. [Google Scholar]
- Zanon, M.A.; Pacheco, R.L.; Latorraca, C.O.C.; Martimbianco, A.L.C.; Pachito, D.V.; Riera, R. Neurodevelopmental Treatment (Bobath) for Children With Cerebral Palsy: A Systematic Review. J. Child. Neurol. 2019, 34, 679–686. [Google Scholar] [CrossRef]
| Month/ Group 1–5 | Week | Introductory Activities, Making Contact with the Dog, Grooming and Taking Care of the Dog | Practice of Gross Motor Skills, Balance and Motor Coordination | Practice of Fine Motor Skills | Exercises Involving Memory, Attention and Concentration | Exercises Stimulating Haptic Perception. Normalisation of Muscle Tone | Improvement of Body Schema and Spatial Orientation | Duration of DAT per Week |
|---|---|---|---|---|---|---|---|---|
| I | I | 10 | 10 | 15 | 10 | 45 | ||
| II | 10 | 10 | 15 | 10 | 45 | |||
| III | 10 | 10 | 15 | 10 | 45 | |||
| IV | 10 | 10 | 15 | 10 | 45 | |||
| Total | 40 min | 40 min | 30 min | 30 min | 20 min | 20 min | ||
| II | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 min | 40 min | 20 min | ||
| III | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 min | 40 min | 20 min | ||
| IV | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 | 40 min | 20 min | ||
| V | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 | 40 min | 20 min | ||
| VI | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 | 40 min | 20 min | ||
| VII | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 | 40 min | 20 min | ||
| VIII | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 min | 40 min | 20 min | ||
| IX | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 min | 40 min | 20 min | ||
| X | I | 5 | 10 | 15 | 10 | 5 | 45 | |
| II | 5 | 10 | 15 | 10 | 5 | 45 | ||
| III | 5 | 10 | 15 | 10 | 5 | 45 | ||
| IV | 5 | 10 | 15 | 10 | 5 | 45 | ||
| Total | 20 min | 40 min | 30 min | 30 min | 40 min | 20 min | ||
| Total duration of DAT | 3 h 40 min | 6 h 40 min | 5 h | 5 h | 6 h 20 min | 3 h 20 min | ||
| Finger Identification | Basic Descriptive Statistics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Effect Size | |||
| Before DAT-I | 30 | 8.57 | 7.35 | 9.79 | 8.00 | 4.00 | 13.00 | 6.00 | 12.00 | 3.27 | |||
| Difference II-I | 30 | 1.20 | 0.50 | 1.90 | 1.50 | −6.00 | 4.00 | 0.00 | 2.00 | 1.86 | 0.47 | ||
| Immediately after DAT-II | 30 | 9.77 | 8.62 | 10.91 | 9.50 | 4.00 | 13.00 | 7.00 | 13.00 | 3.06 | |||
| Difference III-II | 30 | 1.97 | 1.16 | 2.78 | 1.00 | 0.00 | 7.00 | 0.00 | 4.00 | 2.17 | 0.84 | ||
| Two-month follow-up-III | 30 | 11.73 | 11.13 | 12.34 | 12.00 | 8.00 | 13.00 | 11,.00 | 13.00 | 1.62 | |||
| Difference III-I | 30 | 3.17 | 2.27 | 4.07 | 3.00 | 0.00 | 8.00 | 1.00 | 5.00 | 2.41 | 1.29 | ||
| p | Chi^2 Friedman’s ANOVA (N = 30, df = 2) = 41.02326 p < 0.001 Absolute differences between rank sums are (approximately) significant if > 18.5436877917081 at a significance level = 0.05 | ||||||||||||
| Before DAT | Immediately after DAT | Two-month follow-up | |||||||||||
| Before DAT | --- | 21 | 42 | ||||||||||
| Immediately after DAT | 21 | --- | 21 | ||||||||||
| Two-month follow-up | 42 | 21 | --- | ||||||||||
| Postural Imitation | Basic Descriptive Statistics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Effect Size | |||
| Before DAT-I | 30 | 7.57 | 6.32 | 8.81 | 8.50 | 3.00 | 11.00 | 4.00 | 11.00 | 3.34 | |||
| Difference II-I | 30 | 0.83 | 0.51 | 1.16 | 1.00 | 0.00 | 3.00 | 0.00 | 1.00 | 0.87 | 0.26 | ||
| Immediately after DAT-II | 30 | 8.40 | 7.26 | 9.54 | 10.50 | 3.00 | 12.00 | 5.00 | 11.00 | 3.05 | |||
| Difference III-II | 30 | 3.03 | 1.88 | 4.19 | 1.50 | −3.00 | 11.00 | 1.00 | 5.00 | 3.09 | 1.29 | ||
| Two-month follow-up-III | 30 | 11.43 | 10.82 | 12.04 | 12.00 | 6.00 | 15.00 | 12.00 | 12.00 | 1.63 | |||
| Difference III-I | 30 | 3.87 | 2.58 | 5.15 | 3.00 | −3.00 | 12.00 | 1.00 | 6.00 | 3.44 | 1.55 | ||
| p | Chi^2 Friedman’s ANOVA (N = 30, df = 2) = 46.85437 p < 0.001 Absolute differences between rank sums are (approximately) significant if > 18.5436877917081 at a significance level = 0.05 | ||||||||||||
| Before DAT | Immediately after DAT | Two-month follow-up | |||||||||||
| Before DAT | --- | 17.5 | 48.5 | ||||||||||
| Immediately after DAT | 17.5 | --- | 31 | ||||||||||
| Two-month follow-up | 48.5 | 31 | --- | ||||||||||
| Kinaesthesia | Basic Descriptive Statistics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Effect Size | |||
| Before DAT-I | 30 | 15.07 | 12.54 | 17.60 | 17.00 | 1.00 | 24.00 | 8.00 | 20.00 | 6.77 | |||
| Difference II-I | 30 | 1.70 | 1.07 | 2.33 | 1.00 | 0.00 | 6.00 | 0.00 | 3.00 | 1.68 | 0.25 | ||
| Immediately after DAT-II | 30 | 16.77 | 14.19 | 19.34 | 19.00 | 2.00 | 24.00 | 11.00 | 24.00 | 6.90 | |||
| Difference III-II | 30 | −0.50 | −2.07 | 1.07 | 0.00 | −12.00 | 8.00 | −3.00 | 2.00 | 4.22 | 0.08 | ||
| Two-month follow-up-III | 30 | 16.27 | 14.09 | 18.44 | 17.50 | 4.00 | 24.00 | 12.00 | 21.00 | 5.82 | |||
| Difference III-I | 30 | 1.20 | −0.48 | 2.88 | 2.00 | −12.00 | 9.00 | 0.00 | 4.00 | 4.51 | 0.19 | ||
| p | Chi^2 Friedman’s ANOVA (N = 30, df = 2) = 19.38947 p < 0.001 Absolute differences between rank sums are (approximately) significant if > 18.5436877917081 at a significance level = 0.05 | ||||||||||||
| Before DAT | Immediately after DAT | Two-month follow-up | |||||||||||
| Before DAT | --- | 27 | 25.5 | ||||||||||
| Immediately after DAT | 27 | --- | 1.5 | ||||||||||
| Two-month follow-up | 25.5 | 1.5 | --- | ||||||||||
| Bourdon-Wiersma Dot Cancellation Test | Basic Descriptive Statistics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Effect Size | |||
| Before DAT-I | 30 | 25.70 | 18.41 | 32.99 | 19.50 | 2.00 | 54.00 | 8.00 | 52.00 | 19.52 | |||
| Difference II-I | 30 | 3.30 | 2.21 | 4.39 | 3.50 | −1.00 | 10.00 | 1.00 | 5.00 | 2.91 | 0.16 | ||
| Immediately after DAT-II | 30 | 29.00 | 21.20 | 36.80 | 24.00 | 2.00 | 60.00 | 9.00 | 54.00 | 20.90 | |||
| Difference III-II | 30 | 4.50 | −1.59 | 10.59 | 5.50 | −54.00 | 45.00 | 0.00 | 8.00 | 16.32 | 0.21 | ||
| Two-month follow-up-III | 30 | 33.50 | 25.56 | 41.44 | 33.00 | 5.00 | 60.00 | 10.00 | 59.00 | 21.26 | |||
| Difference III-I | 30 | 7.80 | 1.85 | 13.75 | 8.00 | −47.00 | 50.00 | 2.00 | 12.00 | 15.94 | 0.38 | ||
| p | Chi^2 Friedman’s ANOVA (N = 30, df = 2) = 29.05556 p < 0.001 Absolute differences between rank sums are (approximately) significant if > 18.5436877917081 at a significance level = 0.05 | ||||||||||||
| Before DAT | Immediately after DAT | Two-month follow-up | |||||||||||
| Before DAT | --- | 25.5 | 39 | ||||||||||
| Immediately after DAT | 25.5 | --- | 13.5 | ||||||||||
| Two-month follow-up | 39 | 13.5 | --- | ||||||||||
| Finger Identification | Basic Descriptive Statistics | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Cohen’s d | Mann-Whitney U-Test–Z | p | Effect Size | |
| DAT group—before therapy | 30 | 8.57 | 7.35 | 9.79 | 8.00 | 4.00 | 13.00 | 6.00 | 12.00 | 3.27 | 0.02 | 0.04 | 0.971 | 0.02 |
| Control group | 30 | 8.50 | 7.53 | 9.47 | 8.00 | 4.00 | 13.00 | 6.00 | 11.00 | 2.60 | ||||
| Immediately after DAT | 30 | 9.40 | 8.49 | 10.31 | 10.00 | 4.00 | 14.00 | 8.00 | 11.00 | 2.43 | 0.04 | 0.57 | 0.564 | 0.04 |
| Control group | 30 | 9.50 | 8.49 | 10.51 | 9.50 | 5.00 | 15.00 | 8.00 | 12.00 | 2.70 | ||||
| Two-month follow-up | 30 | 11.73 | 11.13 | 12.34 | 12.00 | 8.00 | 13.00 | 11.00 | 13.00 | 1.62 | 1.15 | 3.89 | <0.001 | 1.15 |
| Control group | 30 | 9.40 | 8.49 | 10.31 | 10.00 | 4.00 | 14.00 | 8.00 | 11.00 | 2.43 | ||||
| Kinaesthesia | Basic Descriptive Statistics | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Cohen’s d | Mann-Whitney U-Test–Z | p | Effect Size | |
| DAT group—before therapy | 30 | 15.07 | 12.54 | 17.60 | 17.00 | 1.00 | 24.00 | 8.00 | 20.00 | 6.77 | 0.30 | 1.18 | 0.240 | 0.30 |
| Control group | 30 | 13.23 | 12.00 | 6.00 | 24.00 | 8.00 | 17.00 | 5.35 | 17.00 | 5.35 | ||||
| Immediately after DAT | 30 | 16.77 | 14.19 | 19.34 | 19.00 | 2.00 | 24.00 | 11.00 | 24.00 | 6.90 | 0.40 | 1.63 | 0.104 | 0.40 |
| Control group | 30 | 14.30 | 12.25 | 16.35 | 14.00 | 6.00 | 25.00 | 10.00 | 18.00 | 5.48 | ||||
| Two-month follow-up | 30 | 16.27 | 14.09 | 18.44 | 17.50 | 4.00 | 24.00 | 12.00 | 21.00 | 5.82 | 0.41 | 1.60 | 0.110 | 0.41 |
| Control group | 30 | 14.00 | 12.04 | 15.96 | 13.50 | 5.00 | 25.00 | 10.00 | 17.00 | 5.26 | ||||
| Postural Imitation | Basic Descriptive Statistics | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Cohen’s d | Mann-Whitney U-Test–Z | p | Effect Size | |
| DAT group—before therapy | 30 | 7.57 | 6.32 | 8.81 | 8.50 | 3.00 | 11.00 | 4.00 | 11.00 | 3.34 | 0.35 | −0.58 | 0.559 | 0.35 |
| Control group | 30 | 8.47 | 7.77 | 9.17 | 8.00 | 4.00 | 12.00 | 7.00 | 10.00 | 1.87 | ||||
| Immediately after DAT | 30 | 8.40 | 7.26 | 9.54 | 10.50 | 3.00 | 12.00 | 5.00 | 11.00 | 3.05 | 0.50 | −0.86 | 0.387 | 0.51 |
| Control group | 30 | 9.80 | 8.88 | 10.72 | 10.00 | 5.00 | 15.00 | 9.00 | 11.00 | 2.46 | ||||
| Two-month follow-up | 30 | 11.43 | 10.82 | 12.04 | 12.00 | 6.00 | 15.00 | 12.00 | 12.00 | 1.63 | 0.94 | 3.78 | <0.001 | 0.94 |
| Control group | 30 | 9.37 | 8.34 | 10.39 | 9.00 | 4.00 | 15.00 | 8.00 | 11.00 | 2.75 | ||||
| Bourdon-Wiersma Dot Cancellation Test | Basic Descriptive Statistics | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Cohen’s d | Mann-Whitney U-Test–Z | p | Effect Size | |
| DAT group—before therapy | 30 | 25.70 | 18.41 | 32.99 | 19.50 | 2.00 | 54.00 | 8.00 | 52.00 | 19.52 | 0.12 | −1.03 | 0.301 | 0.12 |
| Control group | 30 | 27.67 | 23.01 | 32.32 | 23.50 | 6.00 | 54.00 | 19.00 | 36.00 | 12.47 | ||||
| Immediately after DAT | 30 | 29.00 | 21.20 | 36.80 | 24.00 | 2.00 | 60.00 | 9.00 | 54.00 | 20.90 | 0.00 | −0.40 | 0.690 | 0.00 |
| Control group | 30 | 28.93 | 24.33 | 33.53 | 26.00 | 7.00 | 54.00 | 20.00 | 37.00 | 12.32 | ||||
| Two-month follow-up | 30 | 33.50 | 25.56 | 41.44 | 33.00 | 5.00 | 60.00 | 10.00 | 59.00 | 21.26 | 0.31 | 0.79 | 0.429 | 0.31 |
| Control group | 30 | 28.17 | 23.42 | 32.91 | 26.00 | 7.00 | 54.00 | 19.00 | 36.00 | 12.71 | ||||
| Basic Descriptive Statistics | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Mean | −95% CI | +95% CI | Median | Min. | Max. | First Quartile | Third Quartile | StDev | Cohen’s d | Paired Samples Wilcoxon Test–Z | p | Effect Size | ||
| Finger identification | Difference II-I | 30 | 1.20 | 0.50 | 1.90 | 1.50 | −6.00 | 4.00 | 0.00 | 2.00 | 1.86 | −0.38 | 0.99 | 0.322 | 0.38 |
| Difference III-II | 30 | 1.97 | 1.16 | 2.78 | 1.00 | 0.00 | 7.00 | 0.00 | 4.00 | 2.17 | |||||
| Kinaesthesia | Difference II-I | 30 | 1.70 | 1.07 | 2.33 | 1.00 | 0.00 | 6.00 | 0.00 | 3.00 | 1.68 | 0.75 | 2.60 | 0.009 | 0.75 |
| Difference III-II | 30 | −0.50 | −2.07 | 1.07 | 0.00 | −12.00 | 8.00 | −3.00 | 2.00 | 4.22 | |||||
| Postural imitation | Difference II-I | 30 | 0.83 | 0.51 | 1.16 | 1.00 | 0.00 | 3.00 | 0.00 | 1.00 | 0.87 | 1.11 | 3.41 | 0.001 | 1.11 |
| Difference III-II | 30 | 3.03 | 1.88 | 4.19 | 1.50 | −3.00 | 11.00 | 1.00 | 5.00 | 3.09 | |||||
| Bourdon- Wierman Test | Difference II-I | 30 | 3.30 | 2.21 | 4.39 | 3.50 | −1.00 | 10.00 | 1.00 | 5.00 | 2.91 | 0.12 | 0.84 | 0.400 | 0.12 |
| Difference III-II | 30 | 4.50 | −1.59 | 10.59 | 5.50 | −54.00 | 45.00 | 0.00 | 8.00 | 16.32 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wolan-Nieroda, A.; Dudziak, J.; Drużbicki, M.; Pniak, B.; Guzik, A. Effect of Dog-Assisted Therapy on Psychomotor Development of Children with Intellectual Disability. Children 2021, 8, 13. https://doi.org/10.3390/children8010013
Wolan-Nieroda A, Dudziak J, Drużbicki M, Pniak B, Guzik A. Effect of Dog-Assisted Therapy on Psychomotor Development of Children with Intellectual Disability. Children. 2021; 8(1):13. https://doi.org/10.3390/children8010013
Chicago/Turabian StyleWolan-Nieroda, Andżelina, Jadwiga Dudziak, Mariusz Drużbicki, Bogumiła Pniak, and Agnieszka Guzik. 2021. "Effect of Dog-Assisted Therapy on Psychomotor Development of Children with Intellectual Disability" Children 8, no. 1: 13. https://doi.org/10.3390/children8010013
APA StyleWolan-Nieroda, A., Dudziak, J., Drużbicki, M., Pniak, B., & Guzik, A. (2021). Effect of Dog-Assisted Therapy on Psychomotor Development of Children with Intellectual Disability. Children, 8(1), 13. https://doi.org/10.3390/children8010013






