Screening of Sensitive Items on Gross Motor Development in Toddlers Aged 24~36 Months and Analysis of the Factors Influencing the Passing Rate
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Measurement
2.3. Screening of Items
2.4. Statistical Analysis
3. Results
3.1. Basic Demographic Information
3.2. Screening of the Main Sensitive Indicators of Gross Motor Development
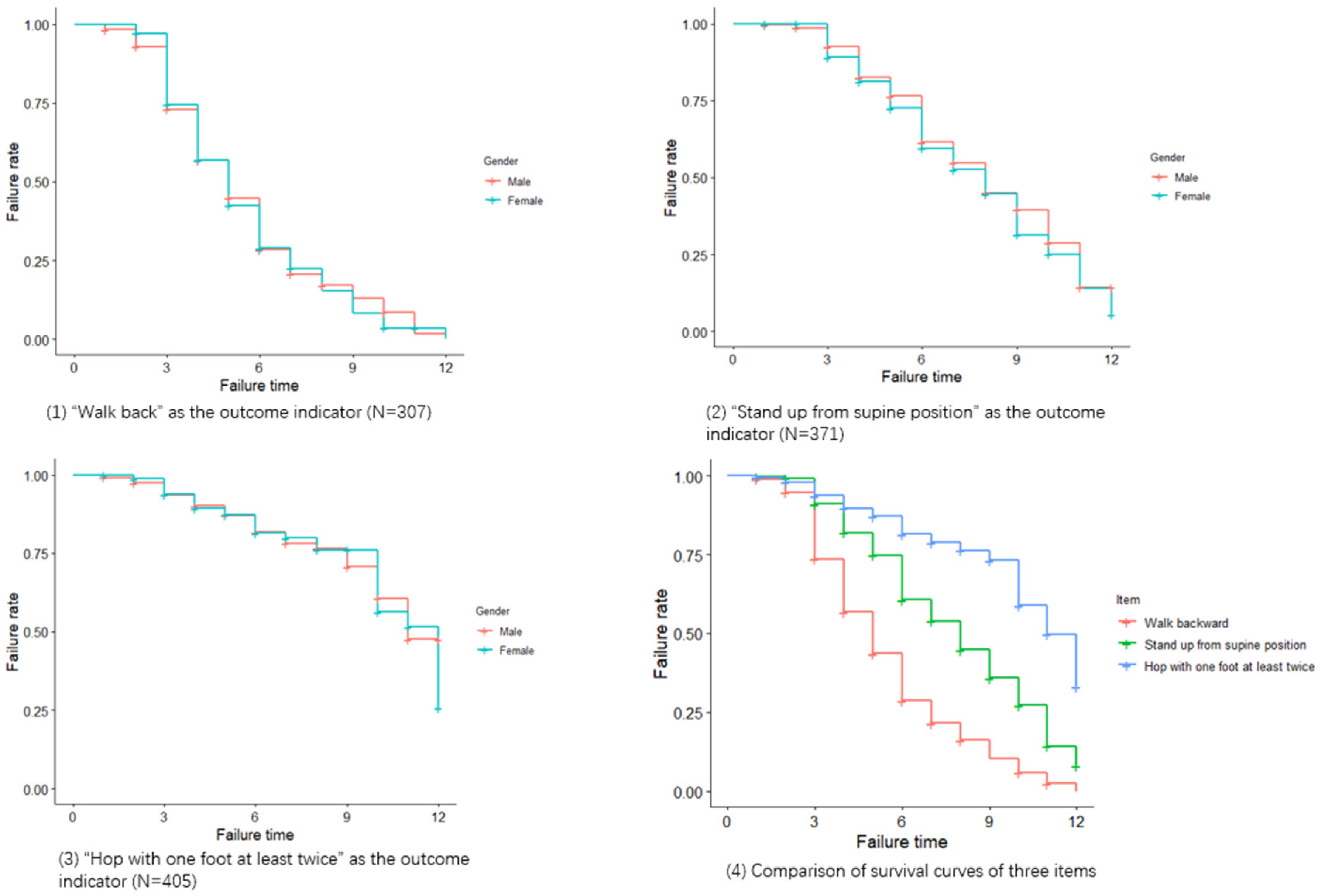
3.3. Survival Analysis
3.4. Cox Regression Analysis
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| C-LAP | Chinese Learning Accomplishment Profile |
| ASD | Autism Spectrum Disorder |
| CP | Cerebral Palsy |
| ATNR | Asymmetrical Tonic Neck Reflex |
| DS | Down Syndrome |
| RMST | Restricted Mean Survival Time |
| KTK | Körperkoordinationstest für Kinder |
| DD | Development Delay |
References
- Aparicio, E.H.; Sobrino, J.C.; Rodríguez, E.F.; de la Torre, J.P.; de Mendívil, R.F.O.; Muñoz, A.M. The importance of motor development in a child. In Proceedings of the International Conference on Education and New Learning Technology (Edulearning10), Barcelona, Spain, 6–7 July 2010. [Google Scholar]
- Liu, W.-Y.; Hou, Y.-J.; Wong, A.M.-K.; Lin, P.-S.; Lin, Y.-H.; Chen, C.-L. Relationships between Gross Motor Functions and Health-Related Quality of Life of Taiwanese Children with Cerebral Palsy. Am. J. Phys. Med. Rehabil. 2009, 88, 473–483. [Google Scholar] [CrossRef]
- Janssen, C.G.C.; Voorman, J.M.; Becher, J.G.; Dallmeijer, A.J.; Schuengel, C. Course of health-related quality of life in 9–16-year-old children with cerebral palsy: Associations with gross motor abilities and mental health. Disabil. Rehabil. 2010, 32, 344–351. [Google Scholar] [CrossRef]
- Bunker, L.K. The Role of Play and Motor Skill Development in Building Children’s Self-Confidence and Self-Esteem. Elem. Sch. J. 1991, 91, 467–471. [Google Scholar] [CrossRef]
- Westendorp, M.; Hartman, E.; Houwen, S.; Smith, J.; Visscher, C. The relationship between gross motor skills and academic achievement in children with learning disabilities. Res. Dev. Disabil. 2011, 32, 2773–2779. [Google Scholar] [CrossRef]
- Kim, H.; Carlson, A.G.; Curby, T.W.; Winsler, A. Relations among motor, social, and cognitive skills in pre-kindergarten children with developmental disabilities. Res. Dev. Disabil. 2016, 53–54, 43–60. [Google Scholar] [CrossRef]
- Alcock, K.J.; Krawczyk, K. Individual differences in language development: Relationship with motor skill at 21 months. Dev. Sci. 2010, 13, 677–691. [Google Scholar] [CrossRef]
- Qiong, D.; Haiqing, X.; Ming, W.; Zhiwei, Z.; Xuan, Z.; Aiqin, Z. Meta analysis of prevalence of autism spectrum disorder in Children in China from 2000 to 2016. Chin. J. Child Health 2017, 25, 1243–1246. (In Chinese) [Google Scholar]
- Sun, X.; Allison, C.; Wei, L.; Matthews, F.E.; Auyeung, B.; Wu, Y.Y.; Griffiths, S.; Zhang, J.; Baron-Cohen, S.; Brayne, C. Autism prevalence in China is comparable to Western prevalence. Mol. Autism 2019, 10, 7. [Google Scholar] [CrossRef]
- Mengmeng, Q.; Xiuhua, L.; Zekai, L.; Hansheng, L. A meta-analysis of the prevalence of cerebral palsy in Children in China. J. Chin. Nurs. Manag. 2015, 1, 63–67. [Google Scholar]
- Yuan, J.; Wang, J.; Ma, J.; Zhu, D.; Zhang, Z.; Li, J. Peadiatric cerebral palsy prevalence and high-risk factors in Henan Province, Central China. J. Rehabil. Med. 2019, 51, 47–53. [Google Scholar] [CrossRef]
- Li, S.; Lin, Q.; Liu, J. Prevalence of childhood cerebral palsy in six provinces in China. Zhonghua Yi Xue Za Zhi 2001, 81, 1220–1223. [Google Scholar]
- Deng, C.; Yi, L.; Mu, Y.; Zhu, J.; Qin, Y.; Fan, X.; Wang, Y.; Li, Q.; Dai, L. Recent trends in the birth prevalence of Down syndrome in China: Impact of prenatal diagnosis and subsequent terminations. Prenat. Diagn. 2015, 35, 311–318. [Google Scholar] [CrossRef]
- West, K.L. Infant Motor Development in Autism Spectrum Disorder: A Synthesis and Meta-analysis. Child Dev. 2018, 90, 2053–2070. [Google Scholar] [CrossRef]
- Allen, M.C.; Alexander, G.R. Using gross motor milestones to identify very preterm infants at risk for cerebral palsy. Dev. Med. Child Neurol. 2008, 34, 226–232. [Google Scholar] [CrossRef]
- Malak, R.; Kostiukow, A.; Krawczyk-Wasielewska, A.; Mojs, E.; Samborski, W. Delays in Motor Development in Children with Down Syndrome. Med. Sci. Monit. 2015, 21, 1904–1910. [Google Scholar] [CrossRef]
- Ozonoff, S.; Young, G.S.; Goldring, S.; Greiss-Hess, L.; Herrera, A.M.; Steele, J.; Macari, S.; Hepburn, S.; Rogers, S.J. Gross Motor Development, Movement Abnormalities, and Early Identification of Autism. J. Autism Dev. Disord. 2008, 38, 644–656. [Google Scholar] [CrossRef]
- Teitelbaum, P.; Teitelbaum, O.; Nye, J.; Fryman, J.; Maurer, R.G. Movement analysis in infancy may be useful for early diagnosis of autism. Proc. Natl. Acad. Sci. USA 1998, 95, 13982–13987. [Google Scholar] [CrossRef]
- Teitelbaum, O.; Benton, T.; Shah, P.K.; Prince, A.; Kelly, J.L.; Teitelbaum, P. Eshkol–Wachman movement notation in diagnosis: The early detection of Asperger’s syndrome. Proc. Natl. Acad. Sci. USA 2004, 101, 11909–11914. [Google Scholar] [CrossRef]
- Piper, M.C.; Darrah, J. Motor Assessment of the Developing Infant; WB Saunders Company: Philadelphia, PA, USA, 1994; Volume 81, p. 244. [Google Scholar]
- Valentini, N.C.; Rudisill, M.E.; Bandeira, P.F.R.; Hastie, P.A. The development of a short form of the Test of Gross Motor Development-2 in Brazilian children: Validity and reliability. Child Care Health Dev. 2018, 44, 759–765. [Google Scholar] [CrossRef]
- Kakebeeke, T.H.; Caflisch, J.; Chaouch, A.; Rousson, V.; Largo, R.H.; Jenni, O.G. Neuromotor development in children. Part 3: Motor performance in 3- to 5-year-olds. Dev. Med. Child Neurol. 2013, 55, 248–256. [Google Scholar] [CrossRef]
- Russell, D.J.; Avery, L.; Walter, S.D.; Hanna, S.E.; Bartlett, D.; Rosenbaum, P.L.; Palisano, R.J.; Gorter, J.W. Development and validation of item sets to improve efficiency of administration of the 66-item Gross Motor Function Measure in children with cerebral palsy. Dev. Med. Child Neurol. 2009, 52, e48–e54. [Google Scholar] [CrossRef] [PubMed]
- Norman, G. Chapter 7: From items to scales. In Health Measurement Scales: A Practical Guide to Their Development and Use; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Coster, W.J.; Mancini, M.C.; Ludlow, L.H. Factor Structure of the School Function Assessment. Educ. Psychol. Meas. 1999, 59, 665–677. [Google Scholar] [CrossRef]
- Brunton, L.K.; Bartlett, D.J. Validity and Reliability of Two Abbreviated Versions of the Gross Motor Function Measure. Phys. Ther. 2011, 91, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Darrah, J.; Piper, M.; Watt, M.-J. Assessment of gross motor skills of at-risk infants: Predictive validity of the Alberta Infant Motor Scale. Dev. Med. Child Neurol. 2008, 40, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Chen, S.; Yu, J. Reliability, Validity, and Responsiveness of the Chinese Learning Accomplishment Profile (C-LAP). Sci. Rep. under review.
- Sanford, A.R. A Manual for Use of the Learning Accomplishment Profile; Kaplan Press: Winston-Salem, NC, USA, 1976. [Google Scholar]
- Vandorpe, B.; Vandendriessche, J.; Lefevre, J.; Pion, J.; Vaeyens, R.; Matthys, S.; Philippaerts, R.; Lenoir, M. The KörperkoordinationsTest für Kinder: Reference values and suitability for 6–12-year-old children in Flanders. Scand. J. Med. Sci. Sports 2011, 21, 378–388. [Google Scholar] [CrossRef]
- Gorla, J.I.; Lifante, S.M.; Souza, A.N. Análise da tarefa saltos laterais, da bateria KTK, em pessoas com deficiência mental. Rev. Mov. Percepção 2007, 8, 147–154. (In Portugues) [Google Scholar]
- Germana, C.; Francesca, S.L.; Maclellan, M.J.; Annalisa, S.; Daniela, M.; Francesco, L.; Yury, I. Backward walking highlights gait asymmetries in children with cerebral palsy. J. Neurophysiol. 2018, 119, 1153–1165. [Google Scholar]
- Buderath, P.; Gärtner, K.; Frings, M.; Christiansen, H.; Schoch, B.; Konczak, J.; Gizewski, E.R.; Hebebrand, J.; Timmann, D.; Konczak, J. Postural and gait performance in children with attention deficit/hyperactivity disorder. Gait Posture 2009, 29, 249–254. [Google Scholar] [CrossRef]
- Cole, M. Right in the Middle: Selective Trunk Activity in the Treatment of Adult Hemiplegia. Neurology 1990, 40, 1911. [Google Scholar] [CrossRef]
- Kim, S.-G.; Ryu, Y.U.; Je, H.D.; Jeong, J.H.; Kim, H.-D. Backward walking treadmill therapy can improve walking ability in children with spastic cerebral palsy. Int. J. Rehabil. Res. 2013, 36, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aziem, A.A.; El-Basatiny, H.M.J.C.R. Effectiveness of backward walking training on walking ability in children with hemiparetic cerebral palsy: A randomized controlled trial. Clin. Rehabil. 2016, 31, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Hoogkamer, W.; Meyns, P.; Duysens, J. Steps Forward in Understanding Backward Gait. Exerc. Sport Sci. Rev. 2014, 42, 23–29. [Google Scholar] [CrossRef] [PubMed]
- VanSant, A.F. Age Differences in Movement Patterns Used by Children to Rise from a Supine Position to Erect Stance. Phys. Ther. 1988, 68, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Marsala, G.; VanSant, A.F. Age-related differences in movement patterns used by toddlers to rise from a supine position to erect stance. Phys. Ther. 1998, 78, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, C.; Shiba, Y.; Sakamoto, M.; Sato, H. The Relationship between the Movement Patterns of Rising from a Supine Position to an Erect Stance and Physical Functions in Healthy Children. Adv. Phys. Educ. 2013, 3, 92–97. [Google Scholar] [CrossRef][Green Version]
- Mewasingh, L.D.; Sékhara, T.; Pelc, K.; Missa, A.-M.; Cheron, G.; Dan, B. Motor strategies in standing up in children with hemiplegia. Pediatr. Neurol. 2004, 30, 257–261. [Google Scholar] [CrossRef]
- Hsue, B.-J.; Wang, Y.-E.; Chen, Y.-J. The movement patterns used to rise from a supine position by children with developmental delay and age-related differences in these. Res. Dev. Disabil. 2014, 35, 2205–2214. [Google Scholar] [CrossRef]
- Tanaka, H.; Borres, M.; Thulesius, O.; Tamai, H.; Ericson, M.O.; Lindblad, L.-E. Evidence of decreased sympathetic function in children with psychosomatic symptoms. Clin. Auton. Res. 2002, 12, 477–482. [Google Scholar] [CrossRef]
- Halverson, L.; Williams, K. Developmental Sequences for Hopping over Distance: A Prelongitudinal Screening. Res. Q. Exerc. Sport 1985, 56, 37–44. [Google Scholar] [CrossRef]
- Haywood, K.; Getchell, N. Life Span Motor Development; Human Kinetics Publishers: Champaign, IL, USA, 2005. [Google Scholar]
- Ghanem, I.; Seringe, R. Comparison of evaluation methods of the results of congenital clubfoot treatment. Rev. Chir. Orthopédique Réparatrice l’Appareil Mot. 1995, 81, 615. [Google Scholar]
- Kim, Y. Effects of Taekwondo Intervention on Postural Control in Youth with Autism Spectrum Disorder. Doctoral Dissertation, California State University, Los Angeles, CA, USA, 2015. [Google Scholar]
- Brudnak, M.A.; Dundero, D.; Van Hecke, F.M. Are the ‘hard’ martial arts, such as the Korean martial art, TaeKwon-Do, of benefit to senior citizens? Med. Hypotheses 2002, 59, 485–491. [Google Scholar] [CrossRef]
- Holm, I.; Tveter, A.T.; Fredriksen, P.M.; Vøllestad, N. A normative sample of gait and hopping on one leg parameters in children 7–12 years of age. Gait Posture 2009, 29, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Ahnert, J.; Schneider, W.; Bös, K. Developmental Changes and Individual Stability of Motor Abilities from the Preschool Period to Young Adulthood; Psychology Press: Hove, UK, 2009; pp. 35–62. [Google Scholar]
- Smits-Engelsman, B.C.; Henderson, S.E.; Michels, C.G. The assessment of children with Developmental Coordination Disorders in the Netherlands: The relationship between the Movement Assessment Battery for Children and the Körperkoordinations Test für Kinder. Hum. Mov. Sci. 1998, 17, 699–709. [Google Scholar] [CrossRef]
- Prätorius, B.; Milani, T.L. Motor abilities of children: Abilities of coordination and balance: Examination of differences between children of different social groups. Dtsch. Z. Sportmed. 2004, 55, 172–176. [Google Scholar]
- Hultman, C.M.; Sandin, S.; Levine, S.; Lichtenstein, P.; Reichenberg, A. (Avi) Advancing paternal age and risk of autism: New evidence from a population-based study and a meta-analysis of epidemiological studies. Mol. Psychiatry 2011, 16, 1203–1212. [Google Scholar] [CrossRef]
- Lung, F.-W.; Shu, B.-C.; Chiang, T.-L.; Lin, S.-J. Parental mental health, education, age at childbirth and child development from six to 18 months. Acta Paediatr. 2009, 98, 834–841. [Google Scholar] [CrossRef]
- Kuwabara, M.; Smith, L.B. Cross-cultural differences in cognitive development: Attention to relations and objects. J. Exp. Child Psychol. 2012, 113, 20–35. [Google Scholar] [CrossRef]
- Angulo-Barroso, R.M.; Schapiro, L.; Liang, W.; Rodrigues, O.; Shafir, T.; Kaciroti, N.; Jacobson, S.W.; Lozoff, B. Motor development in 9-month-old infants in relation to cultural differences and iron status. Dev. Psychobiol. 2010, 53, 196–210. [Google Scholar] [CrossRef]

| Item | Passing Number (Total) | Passing Rate (Total) (95% CI) | Passing Number (34~36 Months Age) | Passing Rate (34~36 Months Age) (95% CI) | Passing Number (31~33 Months Age) | Passing Rate (31~33 Months Age) (95% CI) | Passing Number (28~30 Months Age) | Passing Rate (28~30 Months Age) (95% CI) | Passing Number (24~27 Months Age) | Passing Rate (24~27 Months Age) (95% CI) |
|---|---|---|---|---|---|---|---|---|---|---|
| Walk in a straight line | 1142 | 84.3 (82.3, 86.2) | 288 | 92.6 (89.1, 95.3) | 306 | 94.2 (91.0, 96.4) | 285 | 87.2 (83.0, 90.6) | 263 | 67.3 (62.4, 71.9) |
| Jump down a step | 993 | 73.3 (70.9, 75.7) | 282 | 90.7 (86.9, 93.7) | 278 | 85.5 (81.2, 89.2) | 248 | 75.8 (70.8, 80.4) | 185 | 47.3 (42.3, 52.4) |
| Walk backward | 742 | 54.8 (52.1, 57.5) | 239 | 76.8 (71.8, 81.4) | 241 | 74.2 (69.0, 78.8) | 186 | 56.9 (51.3, 62.3) | 76 | 19.4 (15.6, 23.7) |
| Stand up from supine position | 477 | 35.2 (32.7, 37.8) | 215 | 69.1 (63.7, 74.2) | 160 | 49.2 (43.7, 54.8) | 76 | 23.2 (18.8, 28.2) | 26 | 6.6 (4.4, 9.6) |
| Change feet to climb stairs | 353 | 26.1 (23.7, 28.5) | 160 | 51.4 (45.7, 57.1) | 109 | 33.5 (28.4, 39.0) | 65 | 19.9 (15.7, 24.6) | 19 | 4.9 (3.0, 7.5) |
| Balance on one foot | 196 | 14.5 (12.6, 16.5) | 101 | 32.5 (27.3, 38.0) | 59 | 18.2 (14.1, 22.8) | 27 | 8.3 (5.5, 11.8) | 9 | 2.3 (1.1, 4.3) |
| Hop with one foot at least twice | 176 | 13.0 (11.3, 14.9) | 89 | 28.6 (23.7, 34.0) | 47 | 14.5 (10.8, 18.8) | 31 | 9.5 (6.5, 13.2) | 9 | 2.3 (1.1, 4.3) |
| β Coefficient (95% CI) | Std. Error | t Value | p Value | |
|---|---|---|---|---|
| Intercept | 22.08 (21.78, 22.38) | 0.15 | 143.20 | <0.001 |
| Walk in a straight line | 0.91 (0.54, 1.28) | 0.19 | 4.85 | <0.001 |
| Jump down a step | 1.56 (1.25, 1.88) | 0.16 | 9.61 | <0.001 |
| Walk backward | 2.71 (2.42, 3) | 0.15 | 18.60 | <0.001 |
| Stand up from supine position | 3.47 (3.18, 3.75) | 0.15 | 23.91 | <0.001 |
| Change feet to climb stairs | 2.97 (2.65, 3.3) | 0.17 | 17.77 | <0.001 |
| Balance on one foot | −1.63 (−2.04, −1.21) | 0.21 | −7.70 | <0.001 |
| Hop with one foot at least twice | 3.29 (2.86, 3.71) | 0.22 | 15.27 | <0.001 |
| Item | Weight Coefficient |
|---|---|
| Stand up from supine position | 23.91 |
| Walk backward | 18.60 |
| Change feet to climb stairs | 17.77 |
| Hop with one foot at least twice | 15.27 |
| Jump down a step | 9.61 |
| Balance on one foot | 7.70 |
| Walk in a straight line | 4.85 |
| Item | Evaluation Result |
|---|---|
| Walk backward | Not bad |
| Stand up from supine position | Good |
| Hop with one foot at least twice | Very good |
| Item | Time Point (Month) | Number of Subjects That May Pass | Number of Passing | Failure Rate | Std. Error | 95% CI |
|---|---|---|---|---|---|---|
| Walk backward | 1 | 307 | 3 | 0.99 | 0.01 | (0.98, 1) |
| 3 | 278 | 75 | 0.74 | 0.03 | (0.69, 0.79) | |
| 6 | 96 | 108 | 0.29 | 0.03 | (0.23, 0.35) | |
| 9 | 25 | 32 | 0.10 | 0.02 | (0.07, 0.16) | |
| 12 | 2 | 13 | 0 | |||
| Stand up from supine position | 1 | 371 | 1 | 1 | 0 | (0.99, 1) |
| 3 | 340 | 30 | 0.91 | 0.02 | (0.88, 0.94) | |
| 6 | 159 | 73 | 0.61 | 0.03 | (0.55, 0.67) | |
| 9 | 60 | 37 | 0.36 | 0.04 | (0.29, 0.44) | |
| 12 | 7 | 21 | 0.08 | 0.03 | (0.04, 0.19) | |
| Hop with one foot at least twice | 1 | 405 | 2 | 1 | 0 | (0.99, 1) |
| 3 | 367 | 22 | 0.94 | 0.01 | (0.91, 0.96) | |
| 6 | 169 | 29 | 0.82 | 0.02 | (0.77, 0.86) | |
| 9 | 74 | 10 | 0.73 | 0.03 | (0.67, 0.80) | |
| 12 | 12 | 18 | 0.33 | 0.08 | (0.21, 0.53) |
| Walk Backward (N = 307) | Stand up from Supine Position (N = 371) | Hop with One Foot at Least Twice (N = 405) | ||||
|---|---|---|---|---|---|---|
| β Coefficient (OR, 95% CI) | p Value | β Coefficient (OR, 95% CI) | p Value | β Coefficient (OR, 95% CI) | p Value | |
| Child’s physiological age at first measurement | 0.15 (1.17, 1.09~1.25) | <0.001 | 0.36 (1.43, 1.31~1.57) | <0.001 | 0.37 (1.45, 1.31~1.61) | <0.001 |
| Paternal age | ||||||
| ≤30 yrs | Ref | Ref | ||||
| >30 yrs | −0.45 (0.64, 0.48~0.85) | 0.002 | −0.67 (0.51, 0.32~0.82) | 0.005 | ||
| Region | ||||||
| Beijing | Ref | Ref | Ref | |||
| Shanghai | 0.33 (1.40, 0.90~2.17) | 0.14 | −0.66 (0.52, 0.31~0.86) | 0.01 | 1.72 (5.61, 1.35~23.23) | 0.02 |
| Guangdong | 0.65 (1.91, 1.03~3.56) | 0.04 | −0.21 (0.81, 0.42~1.59) | 0.55 | 1.97 (7.17, 1.51~34.08) | 0.01 |
| Paternal education | ||||||
| bachelor’s degree or above | Ref | |||||
| Below bachelor’s degree | −0.48 (0.62, 0.38~1.00) | 0.05 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, D.; Yu, J.; Wang, J.; Bai, Y.; Zhang, Y.; Lu, X.; Che, B.; Chen, S.; Huang, Y.; Yang, X. Screening of Sensitive Items on Gross Motor Development in Toddlers Aged 24~36 Months and Analysis of the Factors Influencing the Passing Rate. Children 2020, 7, 226. https://doi.org/10.3390/children7110226
Chen D, Yu J, Wang J, Bai Y, Zhang Y, Lu X, Che B, Chen S, Huang Y, Yang X. Screening of Sensitive Items on Gross Motor Development in Toddlers Aged 24~36 Months and Analysis of the Factors Influencing the Passing Rate. Children. 2020; 7(11):226. https://doi.org/10.3390/children7110226
Chicago/Turabian StyleChen, Deng, Jinming Yu, Jiwei Wang, Yue Bai, Yaxuan Zhang, Xinyuan Lu, Beibei Che, Sikun Chen, Yilu Huang, and Xiaoguang Yang. 2020. "Screening of Sensitive Items on Gross Motor Development in Toddlers Aged 24~36 Months and Analysis of the Factors Influencing the Passing Rate" Children 7, no. 11: 226. https://doi.org/10.3390/children7110226
APA StyleChen, D., Yu, J., Wang, J., Bai, Y., Zhang, Y., Lu, X., Che, B., Chen, S., Huang, Y., & Yang, X. (2020). Screening of Sensitive Items on Gross Motor Development in Toddlers Aged 24~36 Months and Analysis of the Factors Influencing the Passing Rate. Children, 7(11), 226. https://doi.org/10.3390/children7110226





