Use of Lisdexamfetamine to Treat Obesity in an Adolescent with Severe Obesity and Binge Eating
Abstract
:1. Introduction
2. Case Presentation
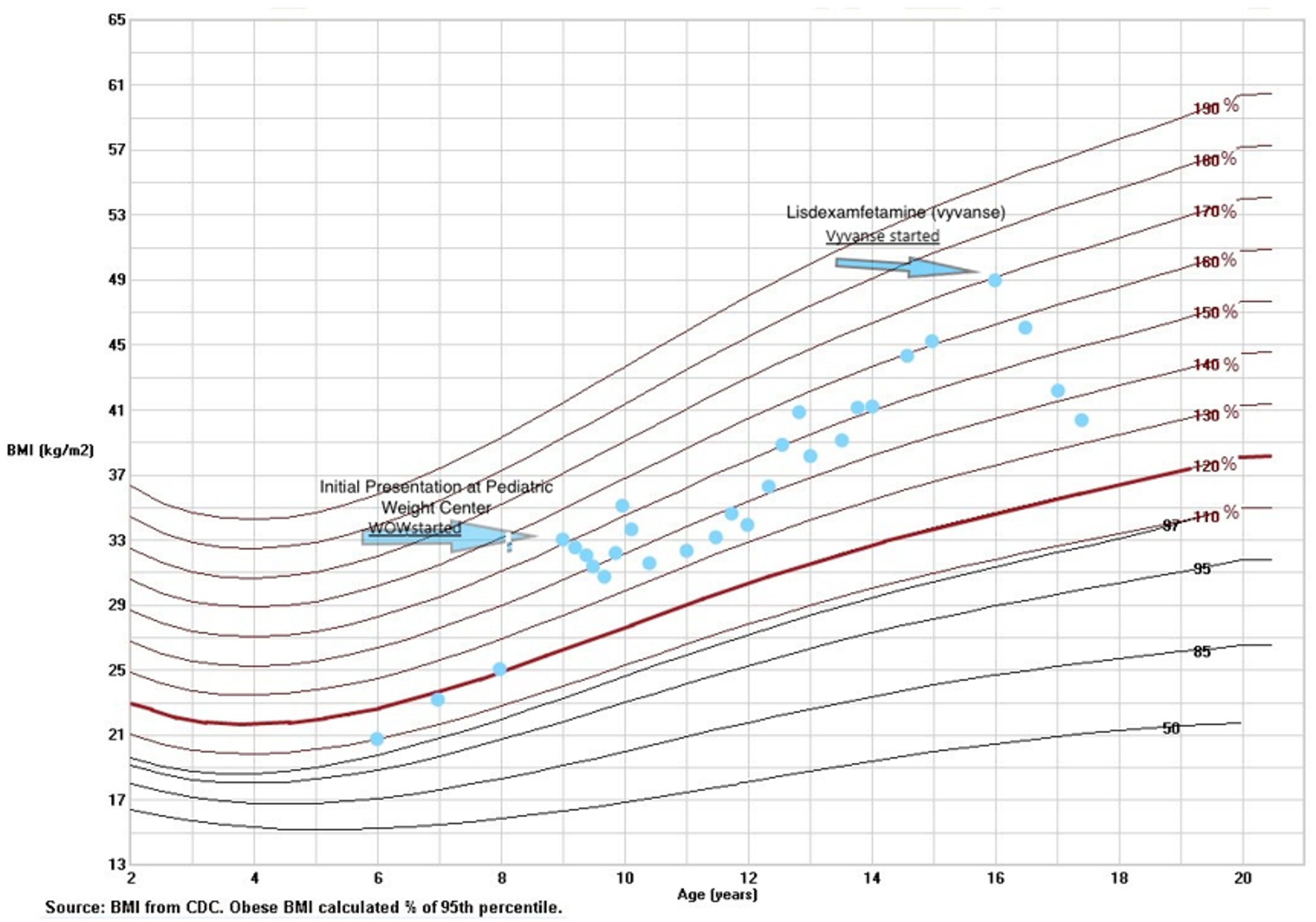
2.1. Assessment and Plan
2.2. Re-Evaluation and New Plan: Decision to Inititate Lisdexamfetamine Therapy
2.3. Consent for Publication
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Skinner, A.C.; Ravanbakht, S.N.; Skelton, J.A.; Perrin, E.M.; Armstrong, S.C. Prevalence of Obesity and Severe Obesity in US Children, 1999–2016. Pediatrics 2018, 141. [Google Scholar] [CrossRef] [PubMed]
- Twig, G.; Tirosh, A.; Leiba, A.; Levine, H.; Ben-Ami Shor, D.; Derazne, E.; Haklai, Z.; Goldberger, N.; Kasher-Meron, M.; Yifrach, D.; et al. BMI at Age 17 Years and Diabetes Mortality in Midlife: A Nationwide Cohort of 2.3 Million Adolescents. Diabetes Care 2016, 39, 1996–2003. [Google Scholar] [CrossRef] [PubMed]
- Twig, G.; Yaniv, G.; Levine, H.; Leiba, A.; Goldberger, N.; Derazne, E.; Shor, D.B.-A.; Tzur, D.; Afek, A.; Shamiss, A.; et al. Body-Mass Index in 2.3 Million Adolescents and Cardiovascular Death in Adulthood. N. Engl. J. Med. 2016, 374, 2430–2440. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, G.; Fox, C.K.; Kelly, A.S.; Jastreboff, A.M.; Browne, A.F.; Browne, N.T.; Pratt, J.S.A.; Bolling, C.; Michalsky, M.P.; Cook, S.; et al. Clinical Considerations Regarding the Use of Obesity Pharmacotherapy in Adolescents with Obesity. Obesity 2019, 27, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Pratt, J.S.A.; Browne, A.; Browne, N.T.; Bruzoni, M.; Cohen, M.; Desai, A.; Inge, T.; Linden, B.C.; Mattar, S.G.; Michalsky, M.; et al. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg. Obes. Relat. Dis. 2018, 14, 882–901. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.S.; Fox, C.K.; Rudser, K.D.; Gross, A.C.; Ryder, J.R. Pediatric obesity pharmacotherapy: Current state of the field, review of the literature and clinical trial considerations. Int. J. Obes. 2016, 40, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, P.; Kowalski, J.; Ekblom, O.; Marcus, C. Response of severely obese children and adolescents to behavioral treatment. Arch. Pediatr. Adolesc. Med. 2012, 166, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, G.; Apovian, C.M. Current pharmacotherapy for obesity. Nat. Rev. Endocrinol. 2017, 14, 12–24. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture. ChooseMyPlate.gov Website. Washington DC. My Plate. Available online: https://www.cnpp.usda.gov/MyPlate (accessed on 27 January 2019).
- Gormally, J.; Black, S.; Daston, S.; Rardin, D. The assessment of binge eating severity among obese persons. Addict. Behav. 1982, 7, 47–55. [Google Scholar] [CrossRef]
- National Institute for Children’s Health Quality. Vanderbilt Scale NICHQ.org Website. NICHQ Vanderbilt Assessment Scales: Used for Diagnosing ADHD. Boston, MA. Available online: https://www.nichq.org/sites/default/files/resource-file/NICHQ_Vanderbilt_Assessment_Scales.pdf (accessed on 27 January 2019).
- Frattarelli, D.A.; Galinkin, J.L.; Green, T.P.; Johnson, T.D.; Neville, K.A.; Paul, I.M.; Van Den Anker, J.N. Off-label use of drugs in children. Pediatrics 2014, 133, 563–567. [Google Scholar] [PubMed]
- Schwimmer, J.B.; Burwinkle, T.M.; Varni, J.W. Health-related quality of life of severely obese children and adolescents. JAMA 2003, 289, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- May, A.L.; Kuklina, E.V.; Yoon, P.W. Prevalence of cardiovascular disease risk factors among US adolescents, 1999–2008. Pediatrics 2012, 129, 1035–1041. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.T.; Lin, C.Y.; Strong, C.; Lin, Y.F.; Chou, Y.Y.; Tsai, M.C. Metabolic correlates of health-related quality of life among overweight and obese adolescents. BMC Pediatr. 2018, 18, 25. [Google Scholar] [CrossRef] [PubMed]
- Seymour, K.E.; Reinblatt, S.P.; Benson, L.; Carnell, S. Overlapping neurobehavioral circuits in ADHD, obesity, and binge eating: Evidence from neuroimaging research. CNS Spectr. 2015, 20, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Quesada, D.; Ahmed, N.U.; Fennie, K.P.; Gollub, E.L.; Ibrahimou, B. A Review: Associations Between Attention-deficit/hyperactivity Disorder, Physical Activity, Medication Use, Eating Behaviors and Obesity in Children and Adolescents. Arch. Psychiatr. Nurs. 2018, 32, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Knop, C.; Singer, V.; Uysal, Y.; Schaefer, A.; Wolters, B.; Reinehr, T. Extremely obese children respond better than extremely obese adolescents to lifestyle interventions. Pediatr. Obes. 2015, 10, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.S.; Barlow, S.E.; Rao, G.; Inge, T.H.; Hayman, L.L.; Steinberger, J.; Urbina, E.M.; Ewing, L.J.; Daniels, S.R.; American Heart Association Atherosclerosis, Hypertension, and Obesity in the Young; Committee of the Council on Cardiovascular Disease in the Young; Council on Nutrition, Physical Activity and Metabolism; Council on Clinical Cardiology. Severe obesity in children and adolescents: Identification, associated health risks, and treatment approaches: A scientific statement from the American Heart Association. Circulation 2013, 128, 1689–1712. [Google Scholar] [CrossRef] [PubMed]
- Kolotkin, R.L.; Gabriel Smolarz, B.; Meincke, H.H.; Fujioka, K. Improvements in health-related quality of life over 3 years with liraglutide 3.0 mg compared with placebo in participants with overweight or obesity. Clin. Obes. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Vyvanse (Lisdexamfetamine Dimesylate) [Package Insert]; Shire US: Lexington, MA, USA, 2017.
- Frampton, J.E. Lisdexamfetamine Dimesylate: A Review in Paediatric ADHD. Drugs 2018, 78, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
| Weight (lbs) | BMI | Lisdexamfetamine Dose | Side Effects | Clinical Progress and Comments | |
|---|---|---|---|---|---|
| First visit Time 0 | 275 | 48.89 (165th BMI%) | 20 mg once daily | Started at lowest 20 mg dose as recommended for ADD to monitor response and side effects. Family was more comfortable starting at the lowest dose. Dose would need to be titrated up in order to provide effect. | |
| 2 weeks | 264 | 46.93 | 20 mg once daily | None | Patient was "beaming" and shared many positive correlations: easier choosing healthier options, decrease in snacking behavior when home alone, and improved focus on school and tasks. |
| 1 month | 257.4 | 45.76 | Dose adjusted to 30 mg once daily | None | Patient was consuming less soda. She was applying for a job for the first time. She noted smaller portion sizes and a decrease in snacking behavior. However, dose was becoming less effective by afternoon or early evening. |
| 2 month | 253.6 | 45.09 | 30 mg once daily | None | Using FitBit to track physical activity and now rarely drinking soda. |
| 3 months | 253.8 | 45.10 | 30 mg once daily | None | Feeling more confident at school and with implementing ILT goals. Transitioned to a new school and tried out for a school play. |
| 4 months | 253.4 | 45.06 | 30 mg once daily | None | BMI had approached stability. The patient was off the medication for 1 week with return of her symptoms and increase in soda intake. Compliance and environmental triggers were reviewed. |
| 4.5 months | 244.6 | 43.49 | 30 mg once daily | None | Happier at school and making new friends. |
| 6 months | 242.8 | 43.17 | 30 mg once daily | None | Nominated Student of the Month. School grades had improved. Cravings were now well controlled. Repeat polysomnograms showed improvement in AHI down to 0.3 (previous 1.1). Repeat BED scales showed improvement from 21 to 5. |
| 7.5 months | 240 | 41.67 | 30 mg once daily | None | Patient continued to show stability. |
| 8.5 months | 241.6 | 41.95 | 30 mg once daily | None | Mild compliance issues with medication (missing one dose/week on average) with notable differences and worsening cravings upon discontinuation of the medication. |
| 10 months | 240 | 41.67 | 30 mg once daily | None | Stable progress noted. |
| 11 months | 242.8 | 42.16 | Dose increase to 40mg once daily | Patient is now 17 years and 2 months old. Increase in soda consumption with bullying recently addressed directly with school official independently. Planning ahead with expected changes to summer schedule. | |
| 12 months | 238.4 | 41.39 | 40 mg once daily | None | Increased compliance noted with improved physical activity and healthier choices. Applying for driver’s permit. |
| 13 months | 235.6 | 40.90 | 40 mg once daily | None | "I think the medication helps me tremendously." Decrease in binge eating patterns, cravings, hunger and more focus with school/home tasks. No longer at alternative locations, therefore less triggers. |
| 15 months | 239.6 | 41.60 | 40 mg once daily | None | Temporal anxiety and stressors noted. Referral to pediatric weight management psychologist with improvements noted. |
| 16 months | 233.6 | 40.84 | 40 mg once daily | None | Temporal stressors and anxiety alleviated. |
| 17 months | 235.2 | 40.84 | Dose increase to 50mg once daily | None | Stressors and anxiety related to senior year high school schedule and increase in emotional eating. |
| 18 months | 235.6 | 40.91 (135th BMI%) | 50 mg once daily | None | Graduating from high school and accepted into local college. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srivastava, G.; O’Hara, V.; Browne, N. Use of Lisdexamfetamine to Treat Obesity in an Adolescent with Severe Obesity and Binge Eating. Children 2019, 6, 22. https://doi.org/10.3390/children6020022
Srivastava G, O’Hara V, Browne N. Use of Lisdexamfetamine to Treat Obesity in an Adolescent with Severe Obesity and Binge Eating. Children. 2019; 6(2):22. https://doi.org/10.3390/children6020022
Chicago/Turabian StyleSrivastava, Gitanjali, Valerie O’Hara, and Nancy Browne. 2019. "Use of Lisdexamfetamine to Treat Obesity in an Adolescent with Severe Obesity and Binge Eating" Children 6, no. 2: 22. https://doi.org/10.3390/children6020022
APA StyleSrivastava, G., O’Hara, V., & Browne, N. (2019). Use of Lisdexamfetamine to Treat Obesity in an Adolescent with Severe Obesity and Binge Eating. Children, 6(2), 22. https://doi.org/10.3390/children6020022





