Chest Compressions for Bradycardia during Neonatal Resuscitation—Do We Have Evidence?
Abstract
1. Introduction
2. Discussion
2.1. Current Practices of Neonatal and Infant Resuscitation
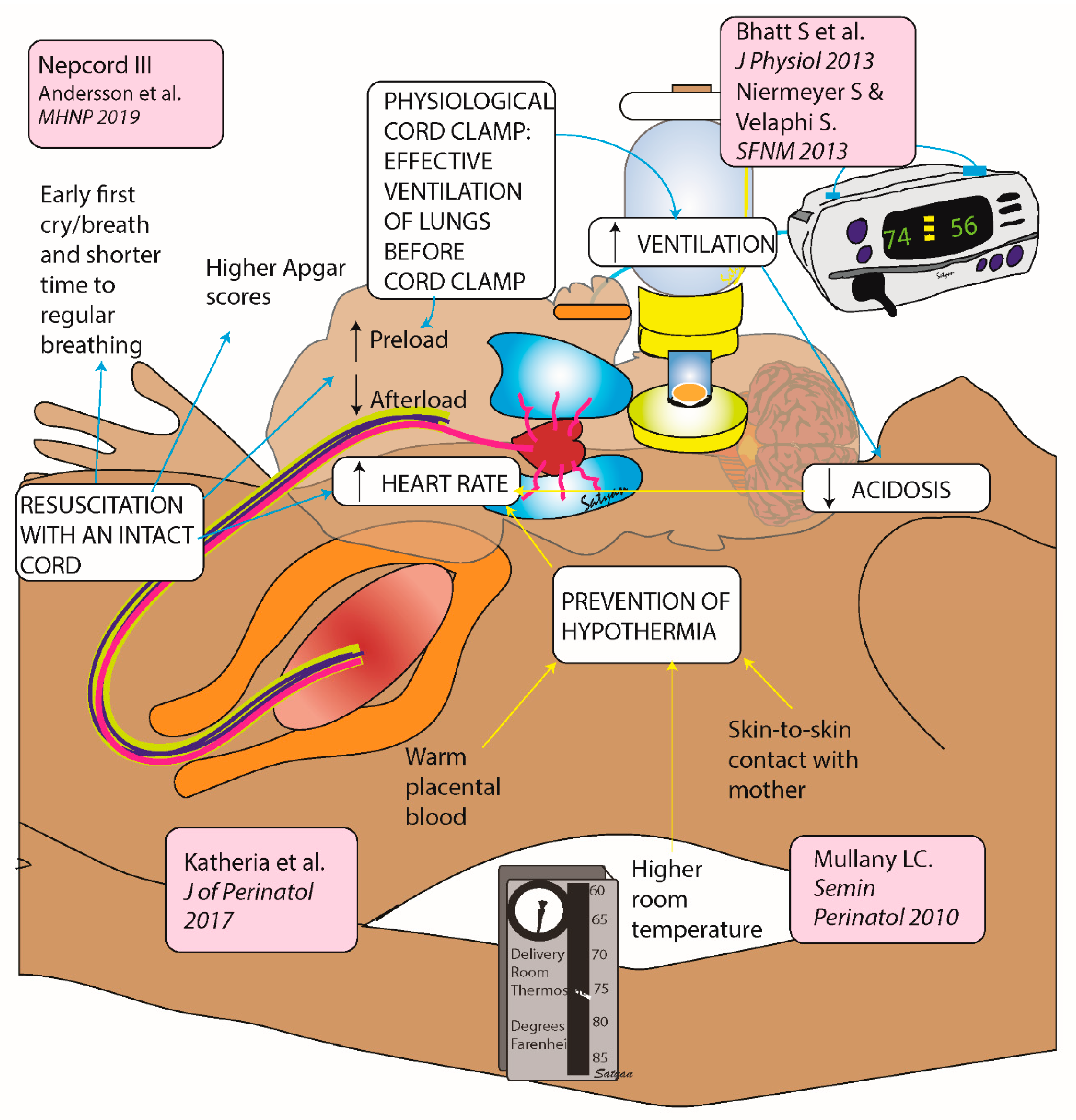
2.2. Effective Strategies to Prevent Worsening of Neonatal Bradycardia at Birth
2.3. Outcomes of Performing CC for Bradycardia in Pediatric in-Hospital Patients
2.4. Research on Performing Chest Compressions during Neonatal Resuscitation
2.5. Characteristics of Coronary and Cerebral Blood Flow during Asphyxia
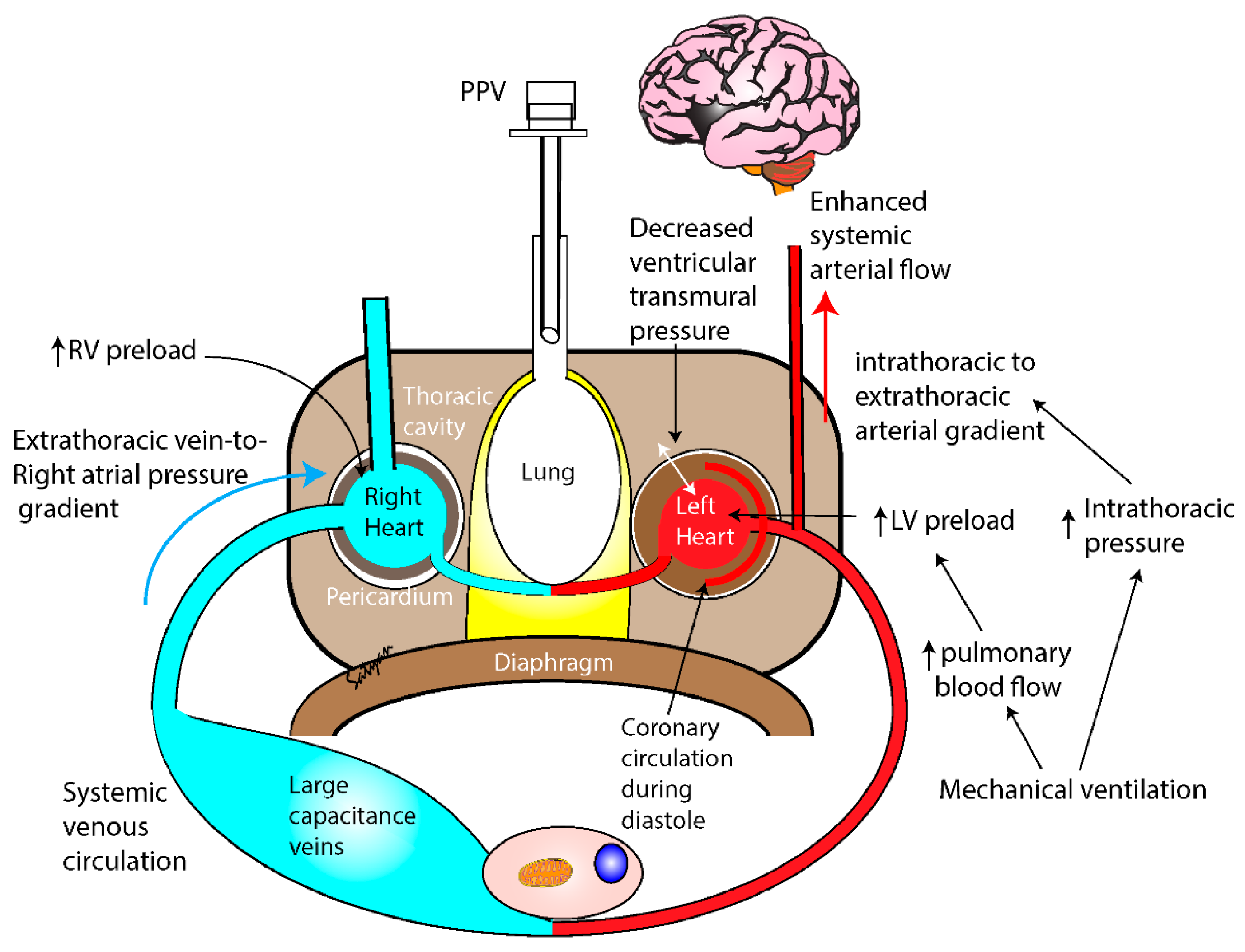
2.6. Positive Pressure Ventilation and Its Effect on Cerebral and Coronary Perfusion during Neonatal Bradycardia
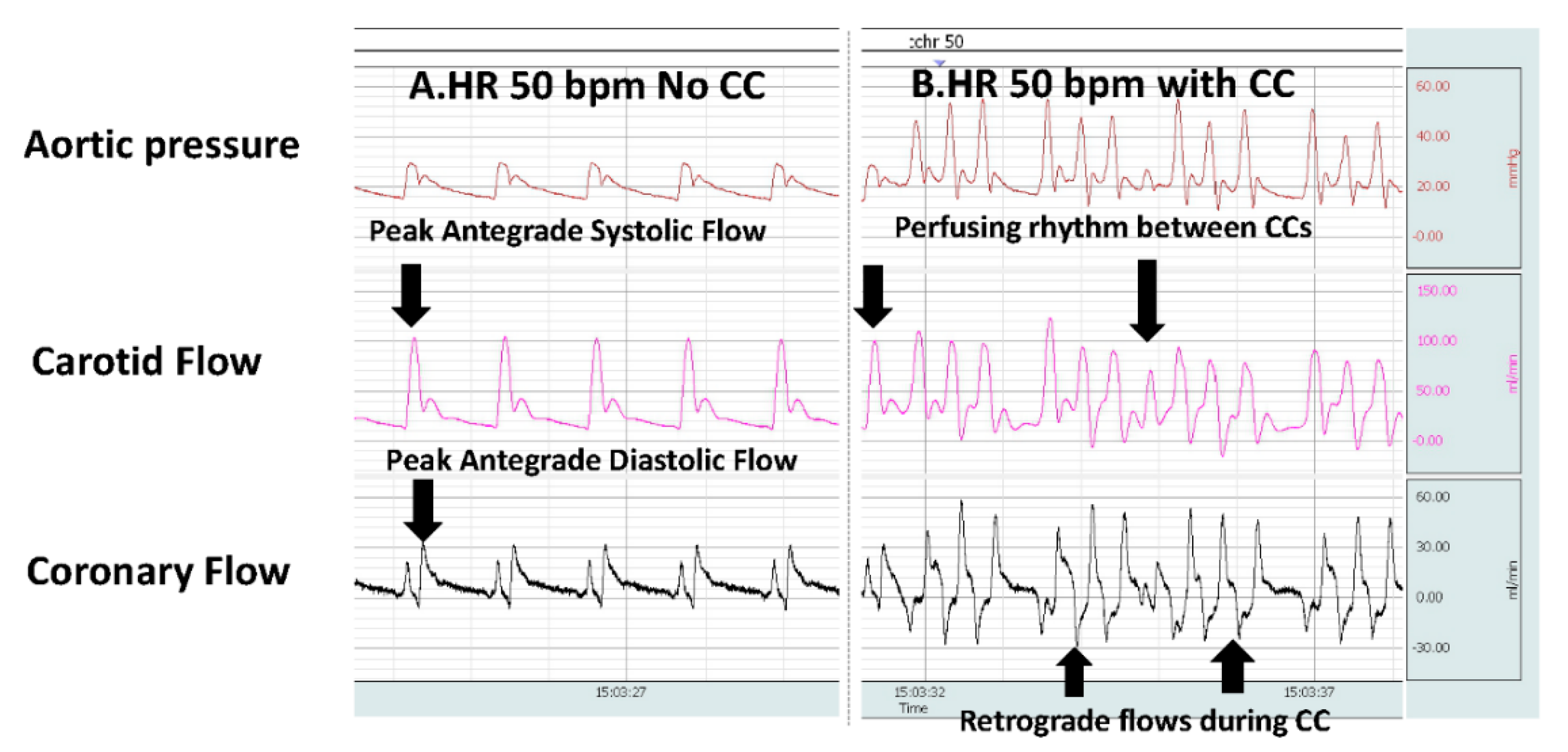
2.7. Effect of Chest Compressions during Bradycardia
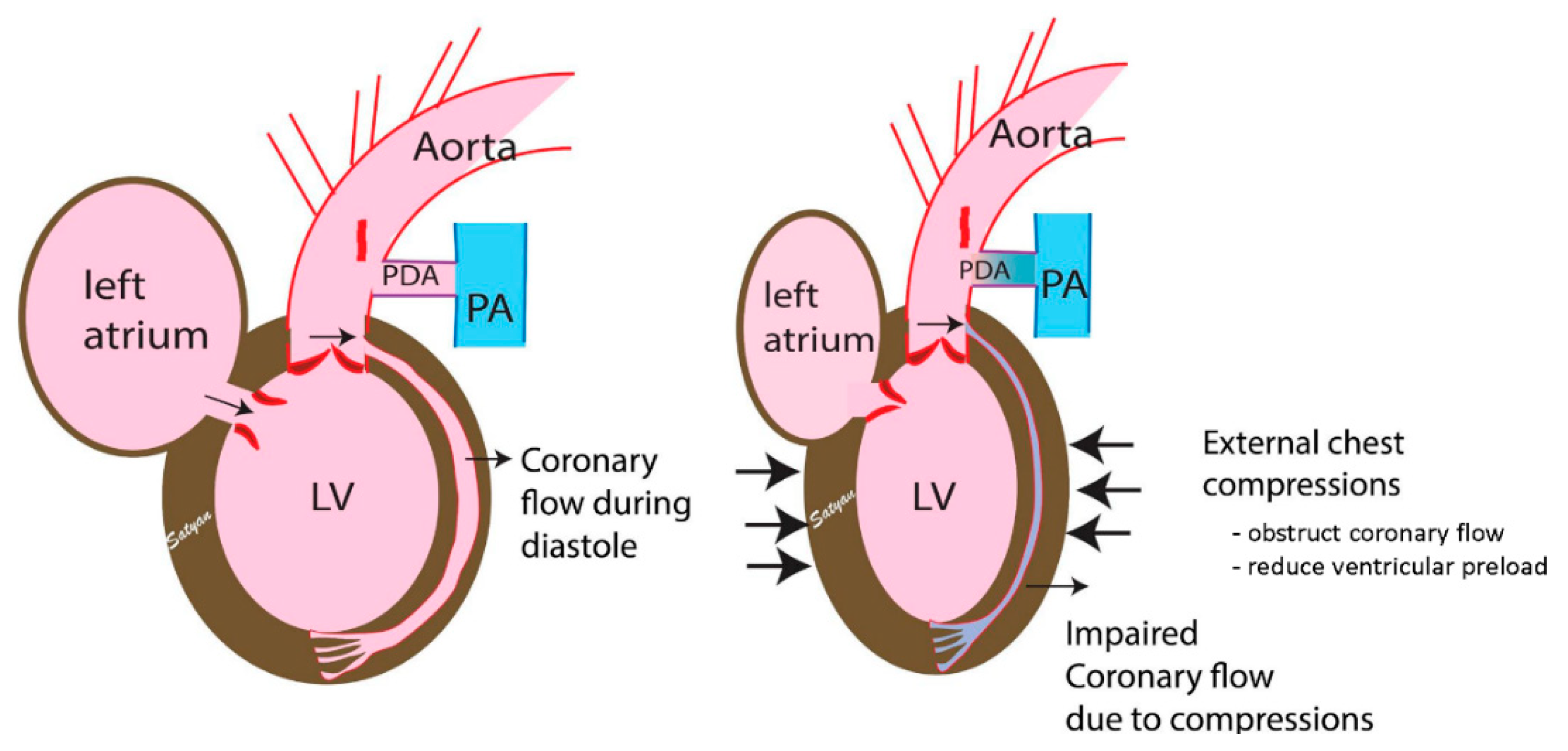
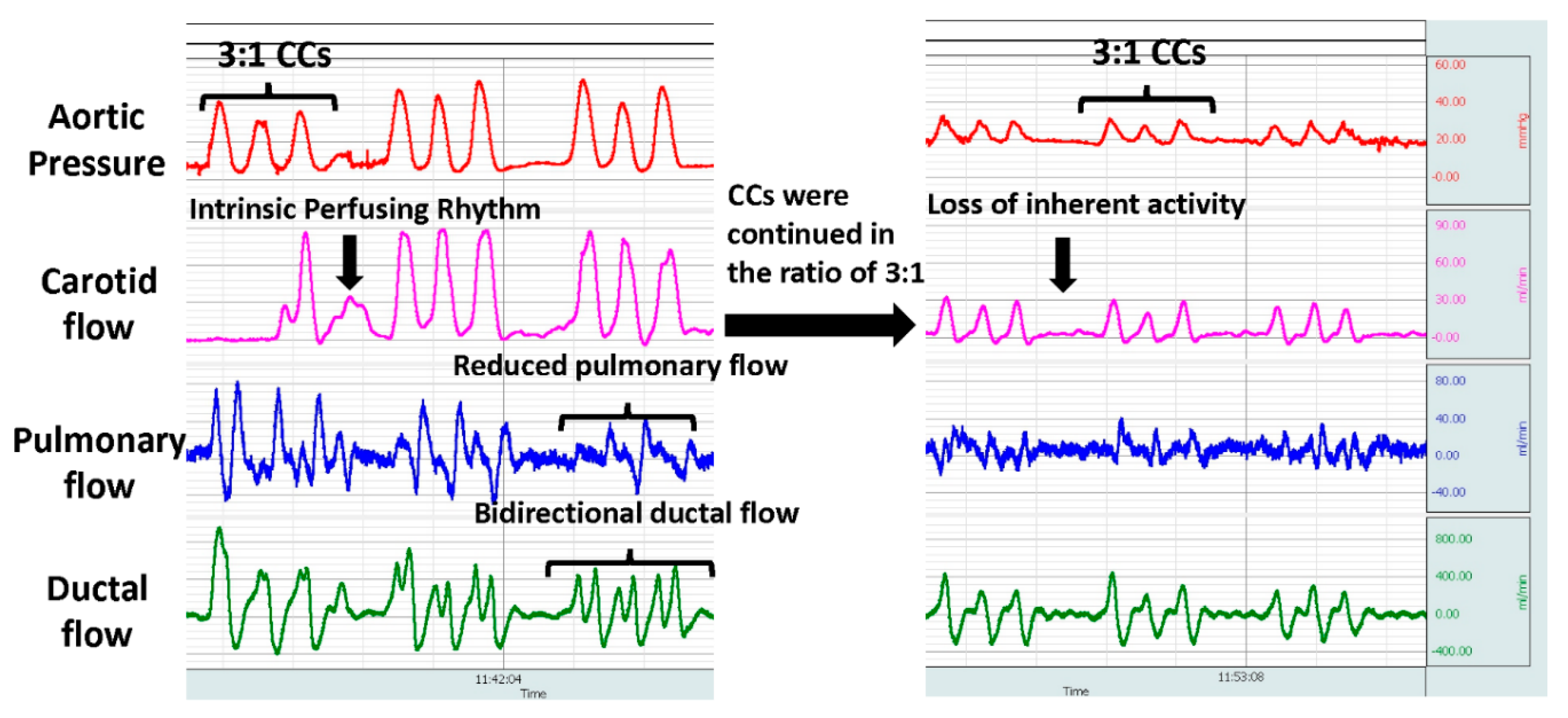
2.8. Could Performing Chest Compressions in the Presence of Perfusing Rhythm Lead to Cardiac Injury and Arrest?
2.9. Is an HR of <60 bpm an Ideal Cut-Off to Initiate CC in Neonates?
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Academy of Pediatrics and American Heart Association Textbook of Neonatal Resuscitation (NRP). Available online: https://ebooks.aappublications.org/content/textbook-of-neonatal-resuscitation-nrp-7th-ed (accessed on 22 October 2019).
- World Health Organization. Guidelines on Basic Newborn Resuscitation. Available online: https://apps.who.int/iris/handle/10665/75157 (accessed on 22 October 2019).
- Black, R.E.; Cousens, S.; Johnson, H.L.; Lawn, J.E.; Rudan, I.; Bassani, D.G.; Jha, P.; Campbell, H.; Walker, C.F.; Cibulskis, R.; et al. Global, regional, and national causes of child mortality in 2008: A systematic analysis. Lancet 2010, 375, 1969–1987. [Google Scholar] [CrossRef]
- WHO. Newborns: Reducing Mortality. Available online: https://www.who.int/news-room/fact-sheets/detail/newborns-reducing-mortality. (accessed on 22 October 2019).
- Dawes, G.S.; Jacobson, H.N.; Mott, J.C.; Shelley, H.J.; Stafford, A. The Treatment of Asphyxiated, Mature Foetal Lambs and Rhesus Monkeys with Intravenous Glucose and Sodium Carbonate. J. Physiol. 1963, 169, 167–184. [Google Scholar] [CrossRef] [PubMed]
- Dawes, G.S.; Mott, J.C.; Shelley, H.J.; Stafford, A. The Prolongation of Survival Time in Asphyxiated Immature Foetal Lambs. J. Physiol. 1963, 168, 43–64. [Google Scholar] [CrossRef] [PubMed]
- Gupta, J.M.; Tizard, J.P. The sequence of events in neonatal apnoea. Lancet 1967, 2, 55–59. [Google Scholar] [CrossRef]
- Wyllie, J. Resuscitation of the depressed newborn. Semin. Fetal Neonatal Med. 2006, 11, 158–165. [Google Scholar] [CrossRef]
- Wyckoff, M.H.; Aziz, K.; Escobedo, M.B.; Kapadia, V.S.; Kattwinkel, J.; Perlman, J.M.; Simon, W.M.; Weiner, G.M.; Zaichkin, J.G. Part 13: Neonatal Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015, 132, S543–S560. [Google Scholar] [CrossRef]
- Wyllie, J.; Perlman, J.M.; Kattwinkel, J.; Wyckoff, M.H.; Aziz, K.; Guinsburg, R.; Kim, H.S.; Liley, H.G.; Mildenhall, L.; Simon, W.M.; et al. Part 7: Neonatal resuscitation: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Resuscitation 2015, 95, e169–e201. [Google Scholar] [CrossRef]
- Shah, B.A.; Wlodaver, A.G.; Escobedo, M.B.; Ahmed, S.T.; Blunt, M.H.; Anderson, M.P.; Szyld, E.G. Impact of electronic cardiac (ECG) monitoring on delivery room resuscitation and neonatal outcomes. Resuscitation 2019, 143, 10–16. [Google Scholar] [CrossRef]
- Kamath-Rayne, B.D.; Thukral, A.; Visick, M.K.; Schoen, E.; Amick, E.; Deorari, A.; Cain, C.J.; Keenan, W.J.; Singhal, N.; Little, G.A.; et al. Helping Babies Breathe, Second Edition: A Model for Strengthening Educational Programs to Increase Global Newborn Survival. Glob. Health Sci. Pract. 2018, 6, 538–551. [Google Scholar] [CrossRef]
- Koso-Thomas, M.; McClure, E.M.; Global Network for Women’s and Children’s Health Research Investigators. The Global Network for Women’s and Children’s Health Research: A model of capacity-building research. Semin. Fetal Neonatal Med. 2015, 20, 293–299. [Google Scholar] [CrossRef]
- Niermeyer, S. From the Neonatal Resuscitation Program to Helping Babies Breathe: Global impact of educational programs in neonatal resuscitation. Semin. Fetal Neonatal Med. 2015, 20, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Vidyasagar, D. Global issues in perinatal medicine. Semin. Fetal Neonatal Med. 2015, 20, 283–284. [Google Scholar] [CrossRef] [PubMed]
- De Caen, A.R.; Berg, M.D.; Chameides, L.; Gooden, C.K.; Hickey, R.W.; Scott, H.F.; Sutton, R.M.; Tijssen, J.A.; Topjian, A.; van der Jagt, E.W.; et al. Part 12: Pediatric Advanced Life Support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015, 132, S526–S542. [Google Scholar] [CrossRef] [PubMed]
- Part 11: Neonatal Resuscitation. Available online: https://www.ahajournals.org/doi/abs/10.1161/circ.102.suppl_1.I-343 (accessed on 22 October 2019).
- Bhatt, S.; Alison, B.J.; Wallace, E.M.; Crossley, K.J.; Gill, A.W.; Kluckow, M.; te Pas, A.B.; Morley, C.J.; Polglase, G.R.; Hooper, S.B. Delaying cord clamping until ventilation onset improves cardiovascular function at birth in preterm lambs. J. Physiol. 2013, 591, 2113–2126. [Google Scholar] [CrossRef]
- Hutchon, D.J. Ventilation before Umbilical Cord Clamping Improves Physiological Transition at Birth or “Umbilical Cord Clamping before Ventilation is Established Destabilizes Physiological Transition at Birth”. Front. Pediatr. 2015, 3, 29. [Google Scholar] [CrossRef]
- Katheria, A.C.; Lakshminrusimha, S.; Rabe, H.; McAdams, R.; Mercer, J.S. Placental transfusion: A review. J. Perinatol. 2017, 37, 105–111. [Google Scholar] [CrossRef]
- Martinello, K.; Hart, A.R.; Yap, S.; Mitra, S.; Robertson, N.J. Management and investigation of neonatal encephalopathy: 2017 update. Arch. Dis. Child. Fetal Neonatal Ed. 2017, 102, F346–F358. [Google Scholar] [CrossRef]
- Mercer, J.S.; Erickson-Owens, D.A. Is it time to rethink cord management when resuscitation is needed? J. Midwifery Womens Health 2014, 59, 635–644. [Google Scholar] [CrossRef]
- Niermeyer, S.; Velaphi, S. Promoting physiologic transition at birth: Re-examining resuscitation and the timing of cord clamping. Semin. Fetal Neonatal Med. 2013, 18, 385–392. [Google Scholar] [CrossRef]
- Donoghue, A.; Berg, R.A.; Hazinski, M.F.; Praestgaard, A.H.; Roberts, K.; Nadkarni, V.M.; American Heart Association National Registry of CPR Investigators. Cardiopulmonary Resuscitation Investigators) Cardiopulmonary resuscitation for bradycardia with poor perfusion versus pulseless cardiac arrest. Pediatrics 2009, 124, 1541–1548. [Google Scholar] [CrossRef]
- Khera, R.; Tang, Y.; Girotra, S.; Nadkarni, V.M.; Link, M.S.; Raymond, T.T.; Guerguerian, A.M.; Berg, R.A.; Chan, P.S.; American Heart Association′s Get with the Guidelines-Resuscitation Investigators. Pulselessness After Initiation of Cardiopulmonary Resuscitation for Bradycardia in Hospitalized Children. Circulation 2019, 140, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Kattwinkel, J.; Perlman, J.M.; Aziz, K.; Colby, C.; Fairchild, K.; Gallagher, J.; Hazinski, M.F.; Halamek, L.P.; Kumar, P.; Little, G.; et al. Part 15: Neonatal resuscitation: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010, 122, S909–S919. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, V.; Wyckoff, M.H. Chest compressions for bradycardia or asystole in neonates. Clin. Perinatol. 2012, 39, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Rawat, M.; Chandrasekharan, P.; Gugino, S.; Koenigsknecht, C.; Helman, J.; Alsaleem, M.; Mathew, B.; Nair, J.; Berkelhamer, S.; Vali, P.; et al. Oxygenation and Hemodynamics during Chest Compressions in a Lamb Model of Perinatal Asphyxia Induced Cardiac Arrest. Child 2019, 6, 52. [Google Scholar] [CrossRef] [PubMed]
- Vento, M.; Asensi, M.; Sastre, J.; Lloret, A.; Garcia-Sala, F.; Minana, J.B.; Vina, J. Hyperoxemia caused by resuscitation with pure oxygen may alter intracellular redox status by increasing oxidized glutathione in asphyxiated newly born infants. Semin. Perinatol. 2002, 26, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Vento, M.; Asensi, M.; Sastre, J.; Lloret, A.; Garcia-Sala, F.; Vina, J. Oxidative stress in asphyxiated term infants resuscitated with 100% oxygen. J. Pediatr. 2003, 142, 240–246. [Google Scholar] [CrossRef]
- Chandrasekharan, P.; Vali, P.; Rawat, M.; Mathew, B.; Gugino, S.F.; Koenigsknecht, C.; Helman, J.; Nair, J.; Berkelhamer, S.; Lakshminrusimha, S. Continuous capnography monitoring during resuscitation in a transitional large mammalian model of asphyxial cardiac arrest. Pediatr. Res. 2017, 81, 898. [Google Scholar] [CrossRef]
- Chandrasekharan, P.K.; Rawat, M.; Nair, J.; Gugino, S.F.; Koenigsknecht, C.; Swartz, D.D.; Vali, P.; Mathew, B.; Lakshminrusimha, S. Continuous End-Tidal Carbon Dioxide Monitoring during Resuscitation of Asphyxiated Term Lambs. Neonatology 2016, 109, 265–273. [Google Scholar] [CrossRef]
- Garcia-Hidalgo, C.; Schmolzer, G.M. Chest Compressions in the Delivery Room. Child 2019, 6, 4. [Google Scholar] [CrossRef]
- Katheria, A.; Rich, W.; Finer, N. Electrocardiogram provides a continuous heart rate faster than oximetry during neonatal resuscitation. Pediatrics 2012, 130, e1177–e1181. [Google Scholar] [CrossRef]
- Delguercio, L.R.; Coomaraswamy, R.P.; State, D. Cardiac Output and Other Hemodynamic Variables during External Cardiac Massage in Man. N. Engl. J. Med. 1963, 269, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Sobotka, K.S.; Polglase, G.R.; Schmolzer, G.M.; Davis, P.G.; Klingenberg, C.; Hooper, S.B. Effects of chest compressions on cardiovascular and cerebral hemodynamics in asphyxiated near-term lambs. Pediatr. Res. 2015, 78, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Voorhees, W.D.; Babbs, C.F.; Tacker, W.A., Jr. Regional blood flow during cardiopulmonary resuscitation in dogs. Crit Care Med. 1980, 8, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Cohn, H.E.; Sacks, E.J.; Heymann, M.A.; Rudolph, A.M. Cardiovascular responses to hypoxemia and acidemia in fetal lambs. Am. J. Obs. Gynecol. 1974, 120, 817–824. [Google Scholar] [CrossRef]
- Itskovitz, J.; LaGamma, E.F.; Rudolph, A.M. The effect of reducing umbilical blood flow on fetal oxygenation. Am. J. Obs. Gynecol. 1983, 145, 813–818. [Google Scholar] [CrossRef]
- Jensen, A.; Hohmann, M.; Kunzel, W. Dynamic changes in organ blood flow and oxygen consumption during acute asphyxia in fetal sheep. J. Dev. Physiol. 1987, 9, 543–559. [Google Scholar]
- Jensen, A.; Roman, C.; Rudolph, A.M. Effects of reducing uterine blood flow on fetal blood flow distribution and oxygen delivery. J. Dev. Physiol. 1991, 15, 309–323. [Google Scholar]
- Lakshminrusimha, S.; Carrion, V. Perinatal Physiology and Principles of Neonatal Resuscitation. Clin. Pediatr. Emerg. Med. 2008, 9, 131–139. [Google Scholar] [CrossRef]
- Chandrasekharan, P.; Gugino, S.; Koenigsknecht, C.; Helman, J.; Rawat, M.; Nair, J.; Sankaran, D.; Agrawal, V.; Lakshminrusimha, S. Is Heart Rate of 60/min the Optimal Cut-off for Initiating Chest Compressions?-Characteristics of Coronary Blood Flow during Asphyxia and Neonatal Resuscitation. Available online: https://www.xcdsystem.com/pas2019/program/2019/index.cfm?pgid=156&sid=1088 (accessed on 22 October 2019).
- Lang, J.A.; Pearson, J.T.; Binder-Heschl, C.; Wallace, M.J.; Siew, M.L.; Kitchen, M.J.; te Pas, A.B.; Fouras, A.; Lewis, R.A.; Polglase, G.R.; et al. Increase in pulmonary blood flow at birth: Role of oxygen and lung aeration. J. Physiol. 2016, 594, 1389–1398. [Google Scholar] [CrossRef]
- Chandrasekharan, P.; Rawat, M.; Gugino, S.F.; Koenigsknecht, C.; Helman, J.; Nair, J.; Vali, P.; Lakshminrusimha, S. Effect of various inspired oxygen concentrations on pulmonary and systemic hemodynamics and oxygenation during resuscitation in a transitioning preterm model. Pediatr. Res. 2018, 84, 743–750. [Google Scholar] [CrossRef]
- Rawat, M.; Chandrasekharan, P.K.; Swartz, D.D.; Mathew, B.; Nair, J.; Gugino, S.F.; Koenigsknecht, C.; Vali, P.; Lakshminrusimha, S. Neonatal resuscitation adhering to oxygen saturation guidelines in asphyxiated lambs with meconium aspiration. Pediatr. Res. 2016, 79, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Chandra, N.; Weisfeldt, M.L.; Tsitlik, J.; Vaghaiwalla, F.; Snyder, L.D.; Hoffecker, M.; Rudikoff, M.T. Augmentation of carotid flow during cardiopulmonary resuscitation by ventilation at high airway pressure simultaneous with chest compression. Am. J. Cardiol. 1981, 48, 1053–1063. [Google Scholar] [CrossRef]
- Schmolzer, G.M.; O’Reilly, M.; Labossiere, J.; Lee, T.F.; Cowan, S.; Qin, S.; Bigam, D.L.; Cheung, P.Y. Cardiopulmonary resuscitation with chest compressions during sustained inflations: A new technique of neonatal resuscitation that improves recovery and survival in a neonatal porcine model. Circulation 2013, 128, 2495–2503. [Google Scholar] [CrossRef] [PubMed]
- Vali, P.; Chandrasekharan, P.; Rawat, M.; Gugino, S.; Koenigsknecht, C.; Helman, J.; Mathew, B.; Berkelhamer, S.; Nair, J.; Wyckoff, M.; et al. Hemodynamics and gas exchange during chest compressions in neonatal resuscitation. PLoS ONE 2017, 12, e0176478. [Google Scholar] [CrossRef]
- Redberg, R.F.; Tucker, K.J.; Cohen, T.J.; Dutton, J.P.; Callaham, M.L.; Schiller, N.B. Physiology of blood flow during cardiopulmonary resuscitation. A transesophageal echocardiographic study. Circulation 1993, 88, 534–542. [Google Scholar] [CrossRef]
- Anderson, P.A.; Glick, K.L.; Killam, A.P.; Mainwaring, R.D. The effect of heart rate on in utero left ventricular output in the fetal sheep. J. Physiol. 1986, 372, 557–573. [Google Scholar] [CrossRef]
- Anderson, P.A.; Killam, A.P.; Mainwaring, R.D.; Oakeley, A.E. In utero right ventricular output in the fetal lamb: The effect of heart rate. J. Physiol. 1987, 387, 297–316. [Google Scholar] [CrossRef]
- Kirkpatrick, S.E.; Pitlick, P.T.; Naliboff, J.; Friedman, W.F. Frank-Starling relationship as an important determinant of fetal cardiac output. Am. J. Physiol. 1976, 231, 495–500. [Google Scholar] [CrossRef]
- Polena, S.; Shen, K.H.; Mamakos, E.; Chuang, P.J.; Sharma, M.; Griciene, P.; Ponomarev, A.A.; Gintautas, J.; Maniar, R. Correlation between cardiac enzyme elevation and the duration of cardiopulmonary resuscitation. Proc. West. Pharm. Soc. 2005, 48, 136–138. [Google Scholar]
- Gilbert, R.D. Control of fetal cardiac output during changes in blood volume. Am. J. Physiol. 1980, 238, H80–H86. [Google Scholar] [CrossRef]
- Kenny, J.; Plappert, T.; Doubilet, P.; Salzman, D.; Sutton, M.G. Effects of heart rate on ventricular size, stroke volume, and output in the normal human fetus: A prospective Doppler echocardiographic study. Circulation 1987, 76, 52–58. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Heart Rate (bpm) | Peak Diastolic Coronary Flow (mL/kg/min) | Peak Systolic Carotid Flow (mL/kg/min) | Peak Systolic Pulmonary Flow (mL/kg/min) |
|---|---|---|---|---|
| Baseline | 167 ± 21 | 17 ± 12 | 52 ± 27 | 110 ± 40 |
| HR > 100 | 140 ± 28 | 20 ± 13 | 46 ± 17 | 117 ± 41 |
| HR 100–80 | 89 ± 6 | 30 ± 16 * | 28 ± 13 | 86 ± 67 |
| HR 79–60 | 72 ± 6 | 20 ± 10 | 58 ± 30 * | 86 ± 68 |
| HR < 60 | 51 ± 7 | 22 ± 11 | 26 ± 11 | 86 ± 52 |
| CC | 100 ± 15 | 5 ± 2 | 20 ± 13 | 19 ± 15 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agrawal, V.; Lakshminrusimha, S.; Chandrasekharan, P. Chest Compressions for Bradycardia during Neonatal Resuscitation—Do We Have Evidence? Children 2019, 6, 119. https://doi.org/10.3390/children6110119
Agrawal V, Lakshminrusimha S, Chandrasekharan P. Chest Compressions for Bradycardia during Neonatal Resuscitation—Do We Have Evidence? Children. 2019; 6(11):119. https://doi.org/10.3390/children6110119
Chicago/Turabian StyleAgrawal, Vikash, Satyan Lakshminrusimha, and Praveen Chandrasekharan. 2019. "Chest Compressions for Bradycardia during Neonatal Resuscitation—Do We Have Evidence?" Children 6, no. 11: 119. https://doi.org/10.3390/children6110119
APA StyleAgrawal, V., Lakshminrusimha, S., & Chandrasekharan, P. (2019). Chest Compressions for Bradycardia during Neonatal Resuscitation—Do We Have Evidence? Children, 6(11), 119. https://doi.org/10.3390/children6110119





