Pediatric Integrative Medicine in Academia: Stanford Children’s Experience
Abstract
1. Background
2. Pediatric Integrative Subspecialties in Academia
2.1. Gastroenterology
2.2. Pain/Perioperative
2.3. Neurology/Neurodevelopmental Pediatrics
2.4. Pediatric Oncology
2.5. Asthma and Allergy
2.6. Other Subspecialties
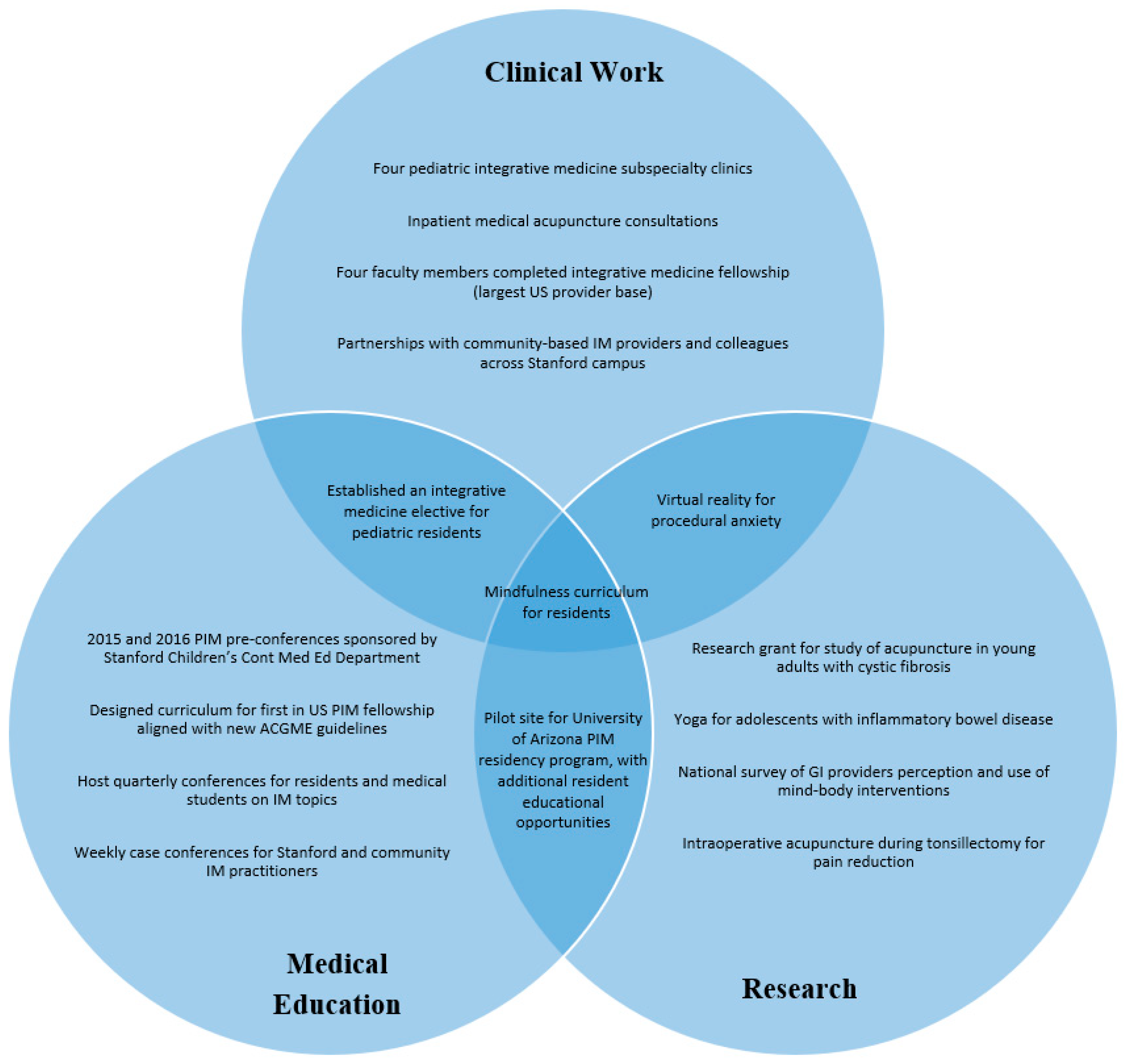
3. Integrative Medicine at Stanford Children’s Health
3.1. Program Background and History
3.2. Clinical Work
3.3. Research
3.4. Medical Education
Sample Case Presentation (Identifying Details Changed for Patient Privacy)
4. Discussion
4.1. Pediatric Integrative Medicine in Academia
4.2. Drivers for Success
4.3. Challenges and Financial Considerations
4.4. Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Misra, S.M.; Guffey, D.; Roth, I.; Giardino, A.P. Complementary and Alternative Medicine Use in Uninsured Children in Texas. Clin. Pediatr. (Phila.) 2017, 56, 866–869. [Google Scholar] [CrossRef] [PubMed]
- Kemper, K.J.; Vohra, S.; Walls, R. The Use of Complementary and Alternative Medicine in Pediatrics. Pediatrics 2008, 122, 1374–1386. [Google Scholar] [CrossRef] [PubMed]
- McClafferty, H.; Vohra, S.; Bailey, M.; Brown, M.; Esparham, A.; Gerstbacher, D.; Golianu, B.; Niemi, A.-K.; Sibinga, E.; Weydert, J.; et al. Pediatric Integrative Medicine. Pediatrics 2017, 140, e20171961. [Google Scholar] [CrossRef]
- Complementary, Alternative, or Integrative Health: What’s In a Name? Available online: https://nccih.nih.gov/health/integrative-health (accessed on 20 July 2018).
- Black, L.I.; Clarke, T.C.; Barnes, P.M.; Stussman, B.J.; Nahin, R.L. Use of Complementary Health Approaches Among Children Aged 4–17 Years in the United States: National Health Interview Survey, 2007–2012. Natl. Health Stat. Rep. 2015, 1–19. [Google Scholar]
- Clarke, T.C.; Black, L.I.; Stussman, B.J.; Barnes, P.M.; Nahin, R.L. Trends in the use of complementary health approaches among adults: United States, 2002–2012. Natl. Health Stat. Rep. 2015, 1–16. [Google Scholar]
- McCann, L.J.; Newell, S.J. Survey of paediatric complementary and alternative medicine use in health and chronic illness. Arch. Dis. Child. 2006, 91, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, D.M.; Davis, R.B.; Ettner, S.L.; Appel, S.; Wilkey, S.; Rompay, M.V.; Kessler, R.C. Trends in Alternative Medicine Use in the United States, 1990-1997: Results of a Follow-up National Survey. JAMA 1998, 280, 1569–1575. [Google Scholar] [CrossRef]
- Taw, M.B. Integrative medicine, or not integrative medicine: That is the question. J. Integr. Med. 2015, 13, 350–352. [Google Scholar] [CrossRef]
- Herman, P.M.; Poindexter, B.L.; Witt, C.M.; Eisenberg, D.M. Are complementary therapies and integrative care cost-effective? A systematic review of economic evaluations. BMJ Open 2012, 2, e001046. [Google Scholar] [CrossRef]
- Ali, A.; Katz, D.L. Disease Prevention and Health Promotion: How Integrative Medicine Fits. Am. J. Prev. Med. 2015, 49, S230–S240. [Google Scholar] [CrossRef]
- Vohra, S.; Schlegelmilch, M.; Jou, H.; Hartfield, D.; Mayan, M.; Ohinmaa, A.; Wilson, B.; Spavor, M.; Grundy, P. Comparative effectiveness of pediatric integrative medicine as an adjunct to usual care for pediatric inpatients of a North American tertiary care centre: A study protocol for a pragmatic cluster controlled trial. Contemp. Clin. Trials Commun. 2017, 5, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Cooney, G.; Dwan, K.; Mead, G. Exercise for Depression. JAMA 2014, 311, 2432–2433. [Google Scholar] [CrossRef] [PubMed]
- Ornish, D.; Scherwitz, L.W.; Billings, J.H.; Gould, K.L.; Merritt, T.A.; Sparler, S.; Armstrong, W.T.; Ports, T.A.; Kirkeeide, R.L.; Hogeboom, C.; et al. Intensive Lifestyle Changes for Reversal of Coronary Heart Disease. JAMA 1998, 280, 2001–2007. [Google Scholar] [CrossRef] [PubMed]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Reduction in the Incidence of Type 2 Diabetes with Lifestyle Intervention or Metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar]
- Loef, M.; Walach, H. The combined effects of healthy lifestyle behaviors on all cause mortality: A systematic review and meta-analysis. Prev. Med. 2012, 55, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Ornish, D.; Lin, J.; Chan, J.M.; Epel, E.; Kemp, C.; Weidner, G.; Marlin, R.; Frenda, S.J.; Magbanua, M.J.M.; Daubenmier, J.; et al. Effect of comprehensive lifestyle changes on telomerase activity and telomere length in men with biopsy-proven low-risk prostate cancer: 5-year follow-up of a descriptive pilot study. Lancet Oncol. 2013, 14, 1112–1120. [Google Scholar] [CrossRef]
- Ornish, D.; Magbanua, M.J.M.; Weidner, G.; Weinberg, V.; Kemp, C.; Green, C.; Mattie, M.D.; Marlin, R.; Simko, J.; Shinohara, K.; et al. Changes in prostate gene expression in men undergoing an intensive nutrition and lifestyle intervention. Proc. Natl. Acad. Sci. USA 2008, 105, 8369–8374. [Google Scholar] [CrossRef]
- He, Z.-Y.; Shi, C.-B.; Wen, H.; Li, F.-L.; Wang, B.-L.; Wang, J. Upregulation of p53 Expression in Patients with Colorectal Cancer by Administration of Curcumin. Cancer Investig. 2011, 29, 208–213. [Google Scholar] [CrossRef]
- Supic, G.; Jagodic, M.; Magic, Z. Epigenetics: A New Link Between Nutrition and Cancer. Nutr. Cancer 2013, 65, 781–792. [Google Scholar] [CrossRef]
- Patel, V.H. Nutrition and prostate cancer: An overview. Expert Rev. Anticancer Ther. 2014, 14, 1295–1304. [Google Scholar] [CrossRef]
- Riccio, P.; Rossano, R. Nutrition Facts in Multiple Sclerosis. ASN Neuro 2015, 7, 1759091414568185. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, D.R.; Clare Morris, M.; Scarmeas, N.; Shah, R.C.; Sijben, J.; Yaffe, K.; Zhu, X. New Perspectives on Alzheimer’s Disease and Nutrition. J. Alzheimer’s Dis. 2015, 46, 1111–1127. [Google Scholar] [CrossRef] [PubMed]
- Kawicka, A.; Regulska-Ilow, B. How nutritional status, diet and dietary supplements can affect autism. A review. Roczniki Państwowego Zakładu Higieny 2013, 64, 1–12. [Google Scholar] [PubMed]
- Eilat-Adar, S.; Sinai, T.; Yosefy, C.; Henkin, Y. Nutritional Recommendations for Cardiovascular Disease Prevention. Nutrients 2013, 5, 3646–3683. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Leyva, D.; Weighell, W.; Edel, A.L.; LaVallee, R.; Dibrov, E.; Pinneker, R.; Maddaford, T.G.; Ramjiawan, B.; Aliani, M.; Guzman, R.; et al. Potent Antihypertensive Action of Dietary Flaxseed in Hypertensive PatientsNovelty and Significance. Hypertension 2013, 62, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Yan, F.; Qian, J.; Guo, M.; Zhang, H.; Tang, X.; Chen, F.; Stoner, G.D.; Wang, X. Randomized Phase II Trial of Lyophilized Strawberries in Patients with Dysplastic Precancerous Lesions of the Esophagus. Cancer Prev. Res. 2012, 5, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, F.; Martucci, K.T.; Kraft, R.A.; Gordon, N.S.; McHaffie, J.G.; Coghill, R.C. Brain Mechanisms Supporting the Modulation of Pain by Mindfulness Meditation. J. Neurosci. 2011, 31, 5540–5548. [Google Scholar] [CrossRef]
- Jacob, J.A. As Opioid Prescribing Guidelines Tighten, Mindfulness Meditation Holds Promise for Pain Relief. JAMA 2016, 315, 2385–2387. [Google Scholar] [CrossRef]
- Cherkin, D.C.; Sherman, K.J.; Balderson, B.H.; Cook, A.J.; Anderson, M.L.; Hawkes, R.J.; Hansen, K.E.; Turner, J.A. Effect of Mindfulness-Based Stress Reduction vs Cognitive Behavioral Therapy or Usual Care on Back Pain and Functional Limitations in Adults With Chronic Low Back Pain: A Randomized Clinical Trial. JAMA 2016, 315, 1240–1249. [Google Scholar] [CrossRef]
- Nidich, S.I.; Rainforth, M.V.; Haaga, D.A.F.; Hagelin, J.; Salerno, J.W.; Travis, F.; Tanner, M.; Gaylord-King, C.; Grosswald, S.; Schneider, R.H. A Randomized Controlled Trial on Effects of the Transcendental Meditation Program on Blood Pressure, Psychological Distress, and Coping in Young Adults. Am. J. Hypertens. 2009, 22, 1326–1331. [Google Scholar] [CrossRef]
- Davidson, R.J.; Kabat-Zinn, J.; Schumacher, J.; Rosenkranz, M.; Muller, D.; Santorelli, S.F.; Urbanowski, F.; Harrington, A.; Bonus, K.; Sheridan, J.F. Alterations in Brain and Immune Function Produced by Mindfulness Meditation. Psychosom. Med. 2003, 65, 564. [Google Scholar] [CrossRef] [PubMed]
- Cotton, S.; Luberto, C.M.; Sears, R.W.; Strawn, J.R.; Stahl, L.; Wasson, R.S.; Blom, T.J.; Delbello, M.P. Mindfulness-based cognitive therapy for youth with anxiety disorders at risk for bipolar disorder: A pilot trial. Early Interv. Psychiatry 2016, 10, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Spigelblatt, L.; Laîné-Ammara, G.; Pless, I.B.; Guyver, A. The Use of Alternative Medicine by Children. Pediatrics 1994, 94, 811–814. [Google Scholar] [PubMed]
- Sawni-Sikand, A.; Schubiner, H.; Thomas, R.L. Use of Complementary/Alternative Therapies Among Children in Primary Care Pediatrics. Ambul. Pediatr. 2002, 2, 99–103. [Google Scholar] [CrossRef]
- Sibinga, E.M.S.; Ottolini, M.C.; Duggan, A.K.; Wilson, M.H. Parent-Pediatrician Communication about Complementary and Alternative Medicine Use for Children. Clin. Pediatr. (Phila.) 2004, 43, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Pappas, S.; Perlman, A. Complementary and alternative medicine: The importance of doctor-patient communication. Med. Clin. N. Am. 2002, 86, 1–10. [Google Scholar] [CrossRef]
- Eisenberg, D.M. Advising Patients Who Seek Alternative Medical Therapies. Ann. Intern. Med. 1997, 127, 61. [Google Scholar] [CrossRef]
- Perlman, A.I.; Eisenberg, D.M.; Panush, R.S. Talking with Patients about Alternative and Complementary Medicine. Rheum. Dis. Clin. N. Am. 1999, 25, 815–822. [Google Scholar] [CrossRef]
- Sugarman, J.; Burk, L. Physicians’ Ethical Obligations Regarding Alternative Medicine. JAMA 1998, 280, 1623–1625. [Google Scholar] [CrossRef]
- Kemper, K.J.; O’Connor, K.G. Pediatricians’ Recommendations for Complementary and Alternative Medical (CAM) Therapies. Ambul. Pediatr. 2004, 4, 482–487. [Google Scholar] [CrossRef]
- Sawni, A.; Thomas, R. Pediatricians’ attitudes, experience and referral patterns regarding complementary/alternative medicine: A national survey. BMC Complement. Altern. Med. 2007, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Winslow, L.C.; Shapiro, H. Physicians Want Education About Complementary and Alternative Medicine to Enhance Communication With Their Patients. Arch. Intern. Med. 2002, 162, 1176–1181. [Google Scholar] [CrossRef]
- Vohra, S.; Surette, S.; Mittra, D.; Rosen, L.D.; Gardiner, P.; Kemper, K.J. Pediatric integrative medicine: Pediatrics’ newest subspecialty? BMC Pediatr. 2012, 12, 123. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Committee on the Use of Complementary and Alternative Medicine by the American Public. Complementary and Alternative Medicine in the United States; The National Academies Collection: Reports funded by National Institutes of Health; National Academies Press (US): Washington, DC, USA, 2005; ISBN 978-0-309-09270-8. [Google Scholar]
- McClafferty, H.; Dodds, S.; Brooks, A.J.; Brenner, M.G.; Brown, M.L.; Frazer, P.; Mark, J.D.; Weydert, J.A.; Wilcox, G.M.G.; Lebensohn, P.; et al. Pediatric Integrative Medicine in Residency (PIMR): Description of a New Online Educational Curriculum. Children 2015, 2, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Frank, E.; Breyan, J.; Elon, L. Physician disclosure of healthy personal behaviors improves credibility and ability to motivate. Arch. Fam. Med. 2000, 9, 287–290. [Google Scholar] [CrossRef] [PubMed]
- McClafferty, H.; Brown, O.W.; Section on Integrative Medicine, Committee on Practice and Ambulatory Medicine. Physician Health and Wellness. Pediatrics 2014, 134, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Dewa, C.S.; Loong, D.; Bonato, S.; Thanh, N.X.; Jacobs, P. How does burnout affect physician productivity? A systematic literature review. BMC Health Serv. Res. 2014, 14, 325. [Google Scholar] [CrossRef]
- Tawfik, D.S.; Profit, J.; Morgenthaler, T.I.; Satele, D.V.; Sinsky, C.A.; Dyrbye, L.N.; Tutty, M.A.; West, C.P.; Shanafelt, T.D. Physician Burnout, Well-being, and Work Unit Safety Grades in Relationship to Reported Medical Errors. Mayo Clin. Proc. 2018, 93, 1571–1580. [Google Scholar] [CrossRef]
- Howe, A.; Smajdor, A.; Stöckl, A. Towards an understanding of resilience and its relevance to medical training. Med. Educ. 2012, 46, 349–356. [Google Scholar] [CrossRef]
- Frank, E.; Rothenberg, R.; Lewis, C.; Belodoff, B.F. Correlates of physicians’ prevention-related practices. Findings from the Women Physicians’ Health Study. Arch. Fam. Med. 2000, 9, 359–367. [Google Scholar] [CrossRef]
- Howe, M.; Leidel, A.; Krishnan, S.M.; Weber, A.; Rubenfire, M.; Jackson, E.A. Patient-Related Diet and Exercise Counseling: Do Providers’ Own Lifestyle Habits Matter? Prev. Cardiol. 2010, 13, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.T. Integrative Medicine as a Bridge to Physician Wellness. Otolaryngol.-Head Neck Surg. 2018, 158, 987–988. [Google Scholar] [CrossRef] [PubMed]
- Vlieger, A.M.; Blink, M.; Tromp, E.; Benninga, M.A. Use of Complementary and Alternative Medicine by Pediatric Patients With Functional and Organic Gastrointestinal Diseases: Results From a Multicenter Survey. Pediatrics 2008, 122, e446–e451. [Google Scholar] [CrossRef] [PubMed]
- Serpico, M.; Boyle, B.; Kemper, K.J.; Kim, S. Complementary and Alternative Medicine Use in Children with Inflammatory Bowel Diseases: A Single Center Survey. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Surette, S.; Vohra, S. Complementary, Holistic, and Integrative Medicine: Utilization Surveys of the Pediatric Literature. Pediatr. Rev. 2014, 35, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.M. Integrative Therapies and Pediatric Inflammatory Bowel Disease: The Current Evidence. Children 2014, 1, 149–165. [Google Scholar] [CrossRef]
- Miele, E.; Pascarella, F.; Giannetti, E.; Quaglietta, L.; Baldassano, R.N.; Staiano, A. Effect of a Probiotic Preparation (VSL#3) on Induction and Maintenance of Remission in Children With Ulcerative Colitis. Am. J. Gastroenterol. 2009, 104, 437–443. [Google Scholar]
- Hanai, H.; Iida, T.; Takeuchi, K.; Watanabe, F.; Maruyama, Y.; Andoh, A.; Tsujikawa, T.; Fujiyama, Y.; Mitsuyama, K.; Sata, M.; et al. Curcumin Maintenance Therapy for Ulcerative Colitis: Randomized, Multicenter, Double-Blind, Placebo-Controlled Trial. Clin. Gastroenterol. Hepatol. 2006, 4, 1502–1506. [Google Scholar] [CrossRef]
- Suskind, D.L.; Wahbeh, G.; Burpee, T.; Cohen, M.; Christie, D.; Weber, W. Tolerability of Curcumin in Pediatric Inflammatory Bowel Disease: A Forced-Dose Titration Study. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 277. [Google Scholar] [CrossRef]
- Ji, J.; Lu, Y.; Liu, H.; Feng, H.; Zhang, F.; Wu, L.; Cui, Y.; Wu, H. Acupuncture and Moxibustion for Inflammatory Bowel Diseases: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Evid.-Based Complement. Altern. Med. 2013, 2013, 158352. [Google Scholar] [CrossRef]
- Timmer, A.; Preiss, J.C.; Motschall, E.; Rücker, G.; Jantschek, G.; Moser, G. Psychological interventions for treatment of inflammatory bowel disease. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef] [PubMed]
- Cotton, S.; Roberts, Y.H.; Tsevat, J.; Britto, M.T.; Succop, P.; McGrady, M.E.; Yi, M.S. Mind-Body Complementary Alternative Medicine Use and Quality of Life in Adolescents with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2010, 16, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Mahler, T. Education and Hypnosis for Treatment of Functional Gastrointestinal Disorders (FGIDs) in Pediatrics. Am. J. Clin. Hypn. 2015, 58, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Tillisch, K. The brain-gut axis in abdominal pain syndromes. Annu. Rev. Med. 2011, 62, 381–396. [Google Scholar] [CrossRef] [PubMed]
- Rutten, J.M.T.M.; Reitsma, J.B.; Vlieger, A.M.; Benninga, M.A. Gut-directed hypnotherapy for functional abdominal pain or irritable bowel syndrome in children: A systematic review. Arch. Dis. Child. 2013, 98, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Kline, R.M.; Kline, J.J.; Di Palma, J.; Barbero, G.J. Enteric-coated, pH-dependent peppermint oil capsules for the treatment of irritable bowel syndrome in children. J. Pediatr. 2001, 138, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Lee, A.C.C.; Kemper, K.J.; Berde, C.B. Use of Complementary and Alternative Medicine in Pediatric Pain Management Service: A Survey. Pain Med. 2005, 6, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Young, L.; Kemper, K.J. Integrative care for pediatric patients with pain. J. Altern. Complement. Med. 2013, 19, 627–632. [Google Scholar] [CrossRef]
- Palermo, T.M. Impact of recurrent and chronic pain on child and family daily functioning: A critical review of the literature. J. Dev. Behav. Pediatr. 2000, 21, 58–69. [Google Scholar] [CrossRef]
- von Stülpnagel, C.; Reilich, P.; Straube, A.; Schäfer, J.; Blaschek, A.; Lee, S.-H.; Müller-Felber, W.; Henschel, V.; Mansmann, U.; Heinen, F. Myofascial trigger points in children with tension-type headache: A new diagnostic and therapeutic option. J. Child Neurol. 2009, 24, 406–409. [Google Scholar] [CrossRef]
- Calvo-Muñoz, I.; Gómez-Conesa, A.; Sánchez-Meca, J. Physical therapy treatments for low back pain in children and adolescents: A meta-analysis. BMC Musculoskelet. Disord. 2013, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Scharff, L.; Sethna, N.F.; McCarthy, C.F.; Scott-Sutherland, J.; Shea, A.M.; Sullivan, P.; Meier, P.; Zurakowski, D.; Masek, B.J.; et al. Physical therapy and cognitive-behavioral treatment for complex regional pain syndromes. J. Pediatr. 2002, 141, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Cotton, S.; Luberto, C.M.; Bogenschutz, L.H.; Pelley, T.J.; Dusek, J. Integrative Care Therapies and Pain in Hospitalized Children and Adolescents: A Retrospective Database Review. J. Altern. Complement. Med. 2013, 20, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Wan, L.; Jamison, R.N. Using Integrative Medicine in Pain Management: An Evaluation of Current Evidence. Anesth. Analg. 2017, 125, 2081. [Google Scholar]
- Tsao, G.J.; Messner, A.H.; Seybold, J.; Sayyid, Z.N.; Cheng, A.G.; Golianu, B. Intraoperative acupuncture for posttonsillectomy pain: A randomized, double-blind, placebo-controlled trial. Laryngoscope 2015, 125, 1972–1978. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, G.H.; DuHamel, K.N.; Redd, W.H. A meta-analysis of hypnotically induced analgesia: How effective is hypnosis? Int. J. Clin. Exp. Hypn. 2000, 48, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Golianu, B.; Seybold, J.; D’Souza, G. Acupuncture as an Anesthetic Adjuvant for Pediatric Orthopedic Patients: A Pilot Study and Protocol Description. Med. Acupunct. 2015, 27, 475–480. [Google Scholar] [CrossRef]
- Won, A.S.; Bailey, J.; Bailenson, J.; Tataru, C.; Yoon, I.A.; Golianu, B. Immersive Virtual Reality for Pediatric Pain. Children 2017, 4, 52. [Google Scholar] [CrossRef]
- Yuan, J.C.; Rodriguez, S.; Caruso, T.J.; Tsui, J.H. Provider-controlled virtual reality experience may adjust for cognitive load during vascular access in pediatric patients. Can. J. Anaesth. 2017, 64, 1275–1276. [Google Scholar] [CrossRef]
- Soo, I.; Mah, J.K.; Barlow, K.; Hamiwka, L.; Wirrell, E. Use of complementary and alternative medical therapies in a pediatric neurology clinic. Can. J. Neurol. Sci. 2005, 32, 524–528. [Google Scholar] [CrossRef]
- Treat, L.; Liesinger, J.; Ziegenfuss, J.Y.; Humeniuk, K.; Prasad, K.; Tilburt, J.C. Patterns of Complementary and Alternative Medicine Use in Children with Common Neurological Conditions. Glob. Adv. Health Med. 2014, 3, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Kenney, D.; Jenkins, S.; Youssef, P.; Kotagal, S. Patient Use of Complementary and Alternative Medicines in an Outpatient Pediatric Neurology Clinic. Pediatr. Neurol. 2016, 58, 48–52.e7. [Google Scholar] [CrossRef] [PubMed]
- Galicia-Connolly, E.; Adams, D.; Bateman, J.; Dagenais, S.; Clifford, T.; Baydala, L.; King, W.J.; Vohra, S. CAM Use in Pediatric Neurology: An Exploration of Concurrent Use with Conventional Medicine. PLoS ONE 2014, 9, e94078. [Google Scholar] [CrossRef] [PubMed]
- Perrin, J.M.; Coury, D.L.; Hyman, S.L.; Cole, L.; Reynolds, A.M.; Clemons, T. Complementary and Alternative Medicine Use in a Large Pediatric Autism Sample. Pediatrics 2012, 130, S77–S82. [Google Scholar] [CrossRef] [PubMed]
- Condò, M.; Posar, A.; Arbizzani, A.; Parmeggiani, A. Riboflavin prophylaxis in pediatric and adolescent migraine. J. Headache Pain 2009, 10, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Van Den Eeden, S.K.; Ackerson, L.M.; Salk, S.E.; Reince, R.H.; Elin, R.J. Oral magnesium oxide prophylaxis of frequent migrainous headache in children: A randomized, double-blind, placebo-controlled trial. Headache 2003, 43, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Guidetti, V.; Dosi, C.; Bruni, O. The relationship between sleep and headache in children: Implications for treatment. Cephalalgia 2014, 34, 767–776. [Google Scholar] [CrossRef]
- Legarda, S.B.; McMahon, D.; Othmer, S.; Othmer, S. Clinical Neurofeedback: Case Studies, Proposed Mechanism, and Implications for Pediatric Neurology Practice. J. Child Neurol. 2011, 26, 1045–1051. [Google Scholar] [CrossRef]
- Tan, G.; Thornby, J.; Hammond, D.C.; Strehl, U.; Canady, B.; Arnemann, K.; Kaiser, D.A. Meta-Analysis of EEG Biofeedback in Treating Epilepsy. Clin. Eeg. Neurosci. 2009, 40, 173–179. [Google Scholar] [CrossRef]
- Kanitz, J.L.; Camus, M.E.M.; Seifert, G. Keeping the balance—An overview of mind–body therapies in pediatric oncology. Complement. Ther. Med. 2013, 21, S20–S25. [Google Scholar] [CrossRef]
- Laengler, A.; Spix, C.; Seifert, G.; Gottschling, S.; Graf, N.; Kaatsch, P. Complementary and alternative treatment methods in children with cancer: A population-based retrospective survey on the prevalence of use in Germany. Eur. J. Cancer 2008, 44, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Bishop, F.L.; Prescott, P.; Chan, Y.K.; Saville, J.; von Elm, E.; Lewith, G.T. Prevalence of Complementary Medicine Use in Pediatric Cancer: A Systematic Review. Pediatrics 2010, 125, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.S. Integrative Therapy Use for Management of Side Effects and Toxicities Experienced by Pediatric Oncology Patients. Children 2014, 1, 424–440. [Google Scholar] [CrossRef] [PubMed]
- Campbell, T.C. Cancer Prevention and Treatment by Wholistic Nutrition. J. Nat. Sci. 2017, 3. [Google Scholar]
- Campbell, T.C. Nutrition and Cancer: An Historical Perspective—The Past, Present, and Future of Nutrition and Cancer. Part 2. Misunderstanding and Ignoring Nutrition. Nutr. Cancer 2017, 69, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.; Howell, A.; Evans, D.G. Can diet and lifestyle prevent breast cancer: What is the evidence? Am. Soc. Clin. Oncol. Educ. Book 2015, e66-73. [Google Scholar] [CrossRef]
- Lee, J.; Jeon, J.Y.; Meyerhardt, J.A. Diet and Lifestyle in Survivors of Colorectal Cancer. Hematol. Oncol. Clin. N. Am. 2015, 29, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Correa, P. Human Gastric Carcinogenesis: A Multistep and Multifactorial Process—First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992, 52, 6735–6740. [Google Scholar]
- Vance, T.M.; Su, J.; Fontham, E.T.H.; Koo, S.I.; Chun, O.K. Dietary Antioxidants and Prostate Cancer: A Review. Nutr. Cancer 2013, 65, 793–801. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a Preventable Disease that Requires Major Lifestyle Changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef]
- Chatzi, L.; Apostolaki, G.; Bibakis, I.; Skypala, I.; Bibaki-Liakou, V.; Tzanakis, N.; Kogevinas, M.; Cullinan, P. Protective effect of fruits, vegetables and the Mediterranean diet on asthma and allergies among children in Crete. Thorax 2007, 62, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Lozinsky, A.C.; Meyer, R.; Koker, C.D.; Dziubak, R.; Godwin, H.; Reeve, K.; Ortega, G.D.; Shah, N. Time to symptom improvement using elimination diets in non-IgE-mediated gastrointestinal food allergies. Pediatr. Allergy Immunol. 2015, 26, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Castro-Rodriguez, J.A.; Ramirez-Hernandez, M.; Padilla, O.; Pacheco-Gonzalez, R.M.; Pérez-Fernández, V.; Garcia-Marcos, L. Effect of foods and Mediterranean diet during pregnancy and first years of life on wheezing, rhinitis and dermatitis in preschoolers. Allergol. Immunopathol. (Madrid) 2016, 44, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Cramer, H.; Posadzki, P.; Dobos, G.; Langhorst, J. Yoga for asthma: A systematic review and meta-analysis. Ann. Allergy Asthma Immunol. 2014, 112, 503–510.e5. [Google Scholar]
- Sankar, J.; Das, R.R. Asthma—A Disease of How We Breathe: Role of Breathing Exercises and Pranayam. Indian J. Pediatr. 2018, 85, 905–910. [Google Scholar] [CrossRef] [PubMed]
- Burgess, J.; Ekanayake, B.; Lowe, A.; Dunt, D.; Thien, F.; Dharmage, S.C. Systematic review of the effectiveness of breathing retraining in asthma management. Expert Rev. Respir. Med. 2011, 5, 789–807. [Google Scholar] [CrossRef] [PubMed]
- Mark, J.D. Pediatric Asthma. Nutr. Clin. Pract. 2009, 24, 578–588. [Google Scholar] [CrossRef]
- Misra, S.M. The Current Evidence of Integrative Approaches to Pediatric Asthma. Curr. Probl. Pediatr. Adolesc. Health Care 2016, 46, 190–194. [Google Scholar] [CrossRef]
- Guardamagna, O.; Abello, F.; Cagliero, P.; Lughetti, L. Impact of nutrition since early life on cardiovascular prevention. Ital. J. Pediatr. 2012, 38, 73. [Google Scholar] [CrossRef]
- Kaikkonen, J.E.; Mikkilä, V.; Raitakari, O.T. Role of Childhood Food Patterns on Adult Cardiovascular Disease Risk. Curr. Atheroscler. Rep. 2014, 16, 443. [Google Scholar] [CrossRef]
- Kavey, R.-E.W.; Allada, V.; Daniels, S.R.; Hayman, L.L.; McCrindle, B.W.; Newburger, J.W.; Parekh, R.S.; Steinberger, J. Cardiovascular Risk Reduction in High-Risk Pediatric Patients*: A Scientific Statement From the American Heart Association Expert Panel on Population and Prevention Science; the Councils on Cardiovascular Disease in the Young, Epidemiology and Prevention, Nutrition, Physical Activity and Metabolism, High Blood Pressure Research, Cardiovascular Nursing, and the Kidney in Heart Disease; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research Endorsed by the American Academy of Pediatrics. J. Cardiovasc. Nurs. 2007, 22, 218–253. [Google Scholar] [PubMed]
- Guarneri, M.; Mercado, N.; Suhar, C. Integrative Approaches for Cardiovascular Disease. Nutr. Clin. Pract. 2009, 24, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Boisvert, J.A.; Harrell, W.A. Integrative Treatment of Pediatric Obesity: Psychological and Spiritual Considerations. Integr. Med. (Encinitas) 2015, 14, 40–47. [Google Scholar] [PubMed]
- Sibinga, E.M.S.; Webb, L.; Ghazarian, S.R.; Ellen, J.M. School-Based Mindfulness Instruction: An RCT. Pediatrics 2016, 137, e20152532. [Google Scholar] [CrossRef] [PubMed]
- Nesvold, A.; Fagerland, M.W.; Davanger, S.; Ellingsen, Ø.; Solberg, E.E.; Holen, A.; Sevre, K.; Atar, D. Increased heart rate variability during nondirective meditation. Eur. J. Prev. Cardiol. 2012, 19, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Nijjar, P.S.; Puppala, V.K.; Dickinson, O.; Duval, S.; Duprez, D.; Kreitzer, M.J.; Benditt, D.G. Modulation of the autonomic nervous system assessed through heart rate variability by a mindfulness based stress reduction program. Int. J. Cardiol. 2014, 177, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Olex, S.; Newberg, A.; Figueredo, V.M. Meditation: Should a cardiologist care? Int. J. Cardiol. 2013, 168, 1805–1810. [Google Scholar] [CrossRef]
- Painovich, J.; Longhurst, J. Integrating acupuncture into the cardiology clinic: Can it play a role? Sheng Li Xue Bao 2015, 67, 19–31. [Google Scholar]
- McClafferty, H.; Brooks, A.J.; Chen, M.-K.; Brenner, M.; Brown, M.; Esparham, A.; Gerstbacher, D.; Golianu, B.; Mark, J.; Weydert, J.; et al. Pediatric Integrative Medicine in Residency Program: Relationship between Lifestyle Behaviors and Burnout and Wellbeing Measures in First-Year Residents. Children 2018, 5, 54. [Google Scholar] [CrossRef]
- Won, A.S.; Tataru, C.A.; Cojocaru, C.M.; Krane, E.J.; Bailenson, J.N.; Niswonger, S.; Golianu, B. Two Virtual Reality Pilot Studies for the Treatment of Pediatric CRPS. Pain Med. 2015, 16, 1644–1647. [Google Scholar] [CrossRef]
- Arruda, J.M.; Bogetz, A.L.; Vellanki, S.; Wren, A.; Yeh, A.M. Yoga as adjunct therapy for adolescents with inflammatory bowel disease: A pilot clinical trial. Complement. Ther. Med. 2018, 41, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Bodner, K.; D’Amico, S.; Luo, M.; Sommers, E.; Goldstein, L.; Neri, C.; Gardiner, P. A cross-sectional review of the prevalence of integrative medicine in pediatric pain clinics across the United States. Complement. Ther. Med. 2018, 38, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Kemper, K.J. Integrative medicine is becoming mainstream: Research on multimodal interventions needs to catch up. Complement. Ther. Med. 2018, 39, A1. [Google Scholar] [CrossRef] [PubMed]
- Mark, J.D.; Chung, Y. Complementary and alternative medicine in pulmonology. Curr. Opin. Pediatr. 2015, 27, 334–340. [Google Scholar] [CrossRef]
- Mark, J.D. Integrative medicine and asthma. Pediatr. Clin. N. Am. 2007, 54, 1007–1023. [Google Scholar] [CrossRef]
- Coates, A.C.; Mark, J.D. Nutrition in Cystic Fibrosis. In Nutrition in Pediatric Pulmonary Disease; Dumont, R., Chung, Y., Eds.; Nutrition and Health; Springer: New York, NY, USA, 2014; pp. 81–97. ISBN 978-1-4614-8474-5. [Google Scholar]
- Mark, J.D. Chapter 29—Asthma. In Integrative Medicine (Fourth Edition); Rakel, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 288–299.e2. ISBN 978-0-323-35868-2. [Google Scholar]
- Yeh, A.M.; Wren, A.; Golianu, B. Mind-Body Interventions for Pediatric Inflammatory Bowel Disease. Children 2017, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Kanak, M.; Park, K.T.; Yeh, A.M. Acupuncture and Integrative Medicine for Pediatric Gastroesophageal Reflux and Functional Dyspepsia. Med. Acupunct. 2015, 27, 467–474. [Google Scholar] [CrossRef]
- Karamian, A.S.; Yeh, A.M.; Wusthoff, C. Integrative Medicine in Child Neurology: What do providers think and what do they need to learn? In Proceedings of the Annual Meeting of American Academy of Neurology, Los Angeles, CA, USA, 21 April 2018. [Google Scholar]
- Staveski, S.L.; Boulanger, K.; Erman, L.; Lin, L.; Almgren, C.; Journel, C.; Roth, S.J.; Golianu, B. The Impact of Massage and Reading on Children’s Pain and Anxiety After Cardiovascular Surgery: A Pilot Study. Pediatr. Crit. Care Med. 2018, 19, 725–732. [Google Scholar] [CrossRef]
- Golianu, B.; Krane, E.; Seybold, J.; Almgren, C.; Anand, K.J.S. Non-pharmacological techniques for pain management in neonates. Semin. Perinatol. 2007, 31, 318–322. [Google Scholar] [CrossRef]
- Sparrow, K.; Golianu, B. Does Acupuncture Reduce Stress Over Time? A Clinical Heart Rate Variability Study in Hypertensive Patients. Med. Acupunct. 2014, 26, 286–294. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Hall, R.W.; Golianu, B.; Yates, C.; Williams, D.K.; Chang, J.; Anand, K.J.S. Does noninvasive electrical stimulation of acupuncture points reduce heelstick pain in neonates? Acta Paediatr. 2016, 105, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- NS-CSHCN Compare All Years: How Many Children/Youth Have Special Health Care Needs? Nationwide. Available online: http://childhealthdata.org/browse/survey/results?q=1792&r=1&t=1&ta=116 (accessed on 27 November 2018).
- Eckert, M.; Amarell, C.; Anheyer, D.; Cramer, H.; Dobos, G. Integrative Pediatrics: Successful Implementation of Integrative Medicine in a German Hospital Setting-Concept and Realization. Children 2018, 5, 122. [Google Scholar] [CrossRef] [PubMed]
- Esparham, A.; Misra, S.; Sibinga, E.; Culbert, T.; Kemper, K.; McClafferty, H.; Vohra, S.; Rosen, L. Pediatric Integrative Medicine: Vision for the Future. Children 2018, 5, 111. [Google Scholar] [CrossRef] [PubMed]
- Bell, I.R.; Caspi, O.; Schwartz, G.E.R.; Grant, K.L.; Gaudet, T.W.; Rychener, D.; Maizes, V.; Weil, A. Integrative medicine and systemic outcomes research: Issues in the emergence of a new model for primary health care. Arch. Intern. Med. 2002, 162, 133–140. [Google Scholar] [CrossRef] [PubMed]
| Category | Name of Study | Study Type | Findings | Funding |
|---|---|---|---|---|
| Pediatric integrative medicine | Pediatric Integrative Medicine [3] | Position Statement | Position Statement of Pediatric Integrative Medicine | N/A |
| Medical education | Pediatric Integrative Medicine in Residency (PIMR): Description of a New Online Educational Curriculum [46] | Retrospective review | Online curriculum targets integrative medicine (IM) knowledge gaps in pediatric residents | Funding received from foundation grants outside of Stanford |
| Pediatric Integrative Medicine in Residency Program: Relationship between Lifestyle Behaviors and Burnout and Wellbeing Measures in First-Year Residents [121] | Retrospective review | Details burnout wellbeing in PIMR participants | Funding received from foundation grants outside of Stanford | |
| Pediatric pulmonary | Complementary and Alternative Medicine in Pulmonology [126] | Literature review | Examines complementary and integrative medicine (CAM) use and effectiveness in children with pulmonary disorders | N/A |
| Integrative Medicine in Asthma [127] | Literature review | Details integrative approach for children with asthma | N/A | |
| Nutrition in Pediatric Cystic Fibrosis [128] | Book chapter | Details evidence of nutritional therapies in children with cystic fibrosis | N/A | |
| Integrated Medicine and Asthma [129] | Book chapter | Details evidence of integrative approach to asthma | N/A | |
| Pediatric gastroenterology | Yoga as adjunct therapy for adolescents with inflammatory bowel disease: A pilot clinical trial [127] | Prospective pilot | Yoga is acceptable, safe and feasible for adolescents with IBD | Tracie Lawlor Foundation, prAna |
| Mind-Body Interventions for Pediatric Inflammatory Bowel Disease [130] | Literature review | Review of evidence on mind-body interventions for IBD | N/A | |
| Integrative Treatment of Reflux and Functional Dyspepsia in Children [130] | Case study and literature review | Describes integrative approach to children with reflux and functional dyspepsia | N/A | |
| Acupuncture and Integrative Medicine for Pediatric Gastroesophageal Reflux and Functional Dyspepsia [131] | Retrospective case series | Describes effect of acupuncture on children with GERD and functional dyspepsia | Stanford Medical Scholars Program | |
| Pediatric neurology | Integrative Medicine in Child Neurology: what do providers think and what do they need to learn? [132] | National survey | Describes results of a national survey of IM modalities used in pediatric neurology | N/A |
| Pediatric pain | The Impact of Massage and Reading on Children’s Pain and Anxiety After Cardiovascular Surgery: A Pilot Study [133] | Randomized prospective trial | Massage was safe and feasible for children undergoing cardiac surgery. Massage decreased anxiety scores and lowered exposure to benzodiazepines. | N/A |
| Immersive Virtual Reality for Pediatric Pain [80] | Literature review | Review of evidence on using virtual reality for acute, chronic, and procedural pain | Mayday Foundation | |
| Two Virtual Reality Pilot Studies for the Treatment of Pediatric CRPS [122] | Prospective pilots | Virtual Reality feasible and effective for treating pediatric CRPS | Mayday Foundation | |
| Non-Pharmacological Techniques for Pain Management in Neonates [134] | Literature review | Details evidence of non-pharmacologic techniques to treat neonatal pain | N/A | |
| Acupuncture | Acupuncture as an Anesthetic Adjuvant for Pediatric Orthopedic Patients: A Pilot Study and Protocol Description [79] | Prospective pilot | Acupuncture was associated with low pain scores and levels of nausea in patients undergoing orthopedic surgery | N/A |
| Does Acupuncture Reduce Stress Over Time? A Clinical Heart Rate Variability Study in Hypertensive Patients [135] | Retrospective case study | Acupuncture increased heart rate variability after acupuncture treatment in adults undergoing treatment for hypertension | N/A | |
| Does Noninvasive Electrical Stimulation of Acupuncture Points Reduce Heelstick Pain in Neonates [136] | Randomized prospective trial | Noninvasive electrical stimulation at acupuncture points was not effective to decrease heelstick pain in neonates | Mayday Foundation | |
| Intraoperative acupuncture for post-tonsillectomy pain: a randomized, double-blind, placebo-controlled trial. [77] | Randomized prospective trial | Intraoperative acupuncture is feasible, well tolerated, and results in improved pain and earlier return of diet after tonsillectomy. | Stanford Medical Scholars Program, Howard Hughes Medical Institute Medical Fellows Program, Stanford Children’s Health Research Institute Akiko Yamazaki and Jerry Yang Faculty Scholar |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramesh, G.; Gerstbacher, D.; Arruda, J.; Golianu, B.; Mark, J.; Yeh, A.M. Pediatric Integrative Medicine in Academia: Stanford Children’s Experience. Children 2018, 5, 168. https://doi.org/10.3390/children5120168
Ramesh G, Gerstbacher D, Arruda J, Golianu B, Mark J, Yeh AM. Pediatric Integrative Medicine in Academia: Stanford Children’s Experience. Children. 2018; 5(12):168. https://doi.org/10.3390/children5120168
Chicago/Turabian StyleRamesh, Gautam, Dana Gerstbacher, Jenna Arruda, Brenda Golianu, John Mark, and Ann Ming Yeh. 2018. "Pediatric Integrative Medicine in Academia: Stanford Children’s Experience" Children 5, no. 12: 168. https://doi.org/10.3390/children5120168
APA StyleRamesh, G., Gerstbacher, D., Arruda, J., Golianu, B., Mark, J., & Yeh, A. M. (2018). Pediatric Integrative Medicine in Academia: Stanford Children’s Experience. Children, 5(12), 168. https://doi.org/10.3390/children5120168





