Multidisciplinary Pain Management for Pediatric Patients with Acute and Chronic Pain: A Foundational Treatment Approach When Prescribing Opioids
Abstract
1. Introduction
“Nonpharmacologic therapy and nonopioid pharmacologic therapy are preferred for chronic pain. Clinicians should consider opioid therapy only if expected benefits for both pain and function are anticipated to outweigh risks to the patient. If opioids are used, they should be combined with nonpharmacologic therapy and nonopioid pharmacologic therapy, as appropriate.”
Aims
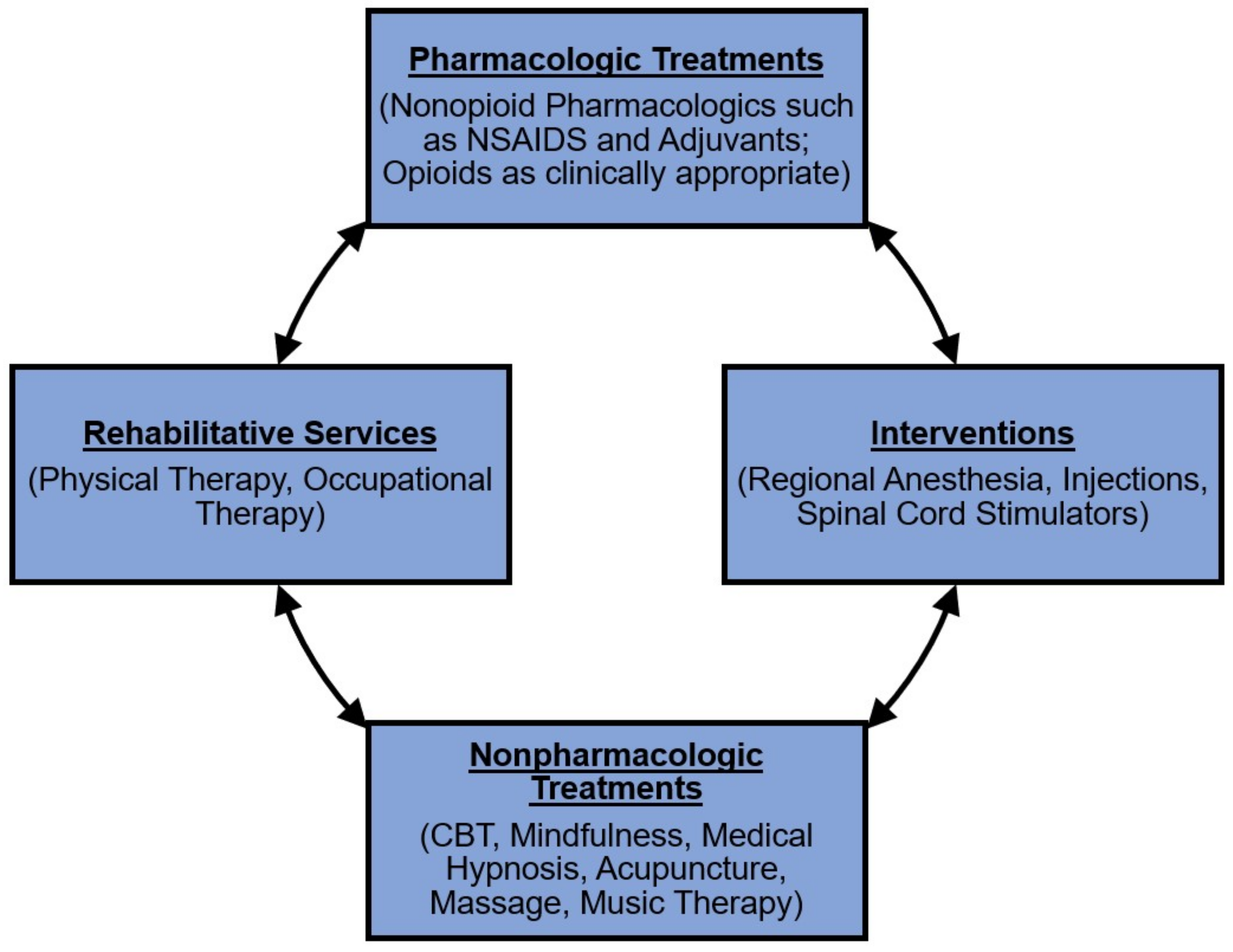
2. Multidisciplinary Pain Management Overview
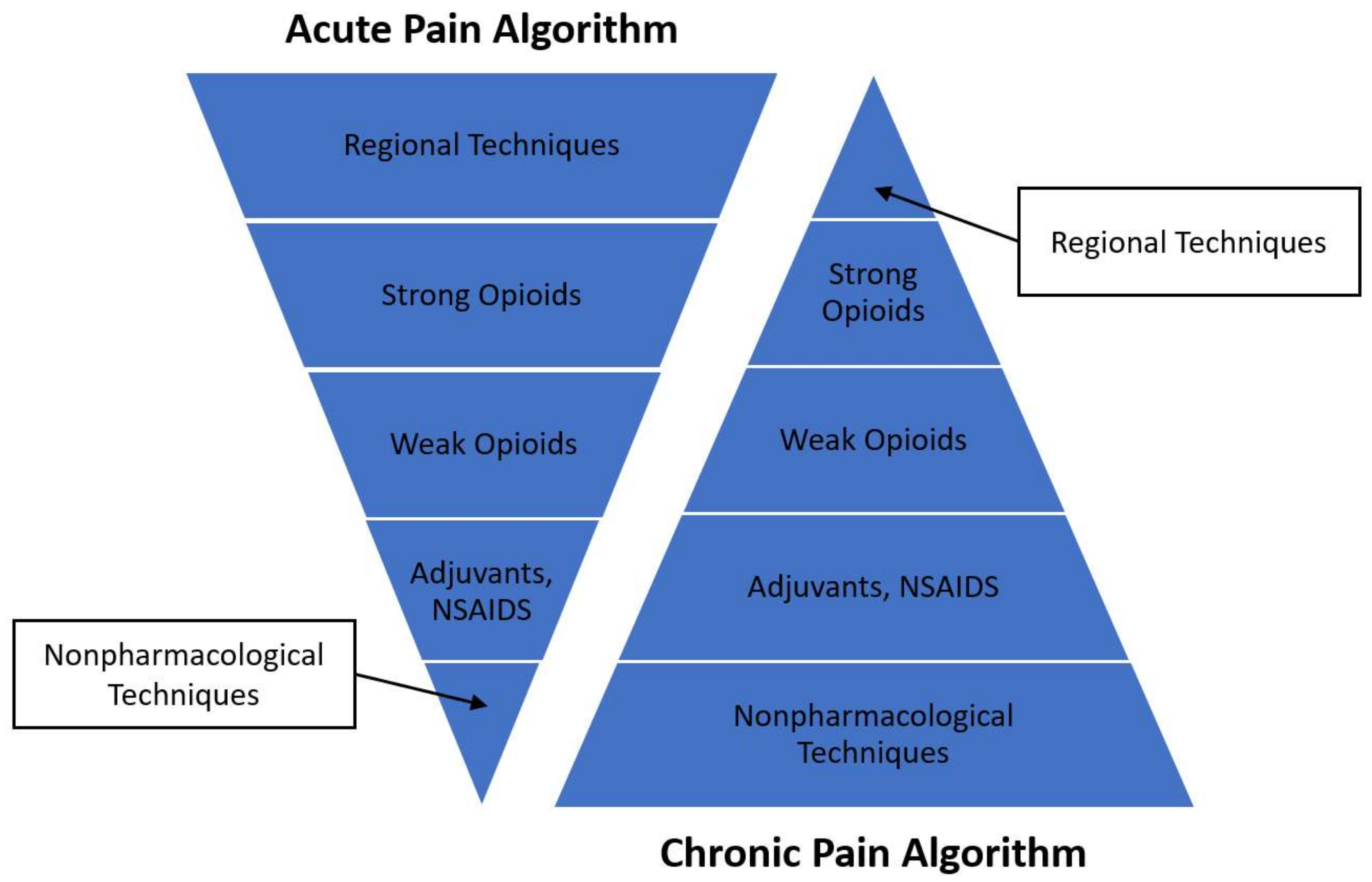
2.1. Acute Multidisciplinary Pain Management
2.2. Chronic Multidisciplinary Pain Management
3. Integrative Non-Pharmacological Therapies in Pediatric Multidisciplinary Pain Management
3.1. Cognitive Behavioral Therapy
3.1.1. Evidence for CBT and Pain Management
3.1.2. CBT Summary
3.2. Mindfulness
3.2.1. Evidence for MBIs and Pain Management
3.2.2. Mindfulness Summary
3.3. Hypnosis
3.3.1. Evidence for Hypnosis and Pain Management
3.3.2. Hypnosis Summary
3.4. Acupuncture
3.4.1. Evidence for Acupuncture and Pain Management
3.4.2. Acupuncture Summary
4. Intensive Interdisciplinary Pediatric Pain Rehabilitative Programs
5. Discussion
Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Therapy Type | Authors (Reference Number) | Study Type (Grade of Evidence) | Study Population | Outcome | Key Results |
|---|---|---|---|---|---|
| CBT | Eccleston et al. [37] | Cochrane systematic review (1) | Cochrane systematic review of psychological therapies (37 RCTs; 2111 participants, mean age 12.45, SD 2.2) | Pain intensity; Disability | Reduced headache pain (RR 2.47, CI 1.97–3.09, p < 0.01; Reduced disability in headache pain (SMD −0.49, CI −0.74 to −0.24, p < 0.01); Reduced nonheadache pain (SMD −0.57, CI −0.86 to −0.27, p < 0.01); Reduced disability in nonheadache pain (SMD −0.45, CI −0.71 to −0.19, p < 0.01 |
| Palermo et al. [61] | Meta-analysis (1) | Meta-analysis (25 RCTs; 1247 participants, 9–17 yo) | Pain intensity | Decreased headache (OR 6.1, CI 4.06 to 9.15, p < 0.0001); Decreased abdominal pain (OR 7.5, CI 3.29 to 17.16, p < 0.001) | |
| Fisher et al. [62] | Systematic review and meta-analysis (1) | Meta-analysis (37 RCTs; 1005 participants, mean age 9.4, SD 1.14) | Pain intensity; Disability | Decreased non-headache pain (SMD −0.60, CI −0.91to −0.29, p < 0.001); Decreased nonheadache pain disability (SMD −0.27, CI −0.46 to −0.08, p < 0.01); Decreased headache (RR 2.9, CI 2.25 to 3.73, p < 0.001); Decreased headache at follow-up (RR 3.34, CI 2.02 to 5.53, p < 0.001 | |
| Mindfulness | Ruskin et al. [82] | Prospective pre-post interventional study (2) | 21 adolescents, 12–18 yo with chronic pain | Feasibility; Acceptability | 90.5% treatment completion rate; No dropouts; Compliance with home practice (M = 60 min/week); Would recommend group to a friend |
| Chadi et al. [83] | RCT (2) | 19 adolescents, 13.9–17.8 yo with chronic pain | Quality of life; Depression; Anxiety; Pain perception; Psychological distress; Salivary cortisol | No significant changes in quality of life, depression, anxiety, pain perception, or psychological distress; Cortisol levels decreased from an average of 3.37 (±1.72) pre-intervention nmol/L to 1.95 (±1.13) nmol/L post-intervention; Cohen’s d = 0.77, p < 0.001). | |
| Ali et al. [84] | Pilot (3) | 15 adolescents, 10–18 yo with fibromyalgia, chronic fatigue, musculoskeletal pain, headache, or abdominal pain | Functional disability; Fibromyalgia symptoms; Quality of life; Mindfulness | Decreased functional disability (33% improvement, p = 0.026); improved fibromyalgia symptoms (26% improvement, p = 0.03), and improved mindfulness (child: 12% improvement, p = 0.02; parent: 17% improvement, p = 0.03). | |
| Waelde et al. [85] | Pilot (3) | 20 adolescents, 13–17 yo with chronic pain | Feasibility and Acceptability Parental assessment | Feasible and acceptable; Decreased parental worry about child (p < 0.01) | |
| Hesse et al. [86] | Pilot (3) | 20 adolescent females, 11–16 yo, with recurrent headaches | Quality of life; Pain acceptance; Depression | Improved quality of life, Parent assess PedsQL, p = 0.049; Improved child acceptance of pain, CPAQ-A p = −0.005; Improved child depression, CES-DC p = 0.009 | |
| Hypnosis | Manworren et al. [35] | RCT (2) | 24 adolescents, 10–18 yo undergoing Nuss procedure | Pain intensity; Morphine equivalent/hour; Length of stay (LOS) | Lower mean pain intensity (−1.72, 95% CI 2.89–0.55, p = 0.0047); Lower maximum pain intensity (−2.27, 95% CI 3.73–0.82, p = 0.0028); Less morphine equivalents/hour (−0.13, 95% CI −0.25–0.00, p = 0.046); Shorter LOS (p = 0.0013) |
| Acupuncture | Tsao et al. [122] | RCT (2) | 59 children, 3–12 yo undergoing tonsillectomy | Pain, Oral food intake | Improvement in pain control and oral intake (p = 0.0065 and 0.001, respectively) |
| Raith et al. [123] | RCT (2) | 28 newborns with neonatal abstinence syndrome | Duration of morphine treatment; Length of stay | Shorter duration of morphine treatment (28 days vs. 39 days; p = 0.019) and hospital stay (35 vs. 50 days; p = 0.048) | |
| Rehabilitation | Hechler, et al. [38] | Systematic review (1) | 10 studies (1020 adolescents, mean age 13.9, SD 1.5) | Pain intensity; Pain-related disability | Decreased pain intensity (d = −1.33, CI −2.28 to −0.38, p = 0.01); Decreased pain related disability (d = −1.09, CI −1.71 to −0.48, p < 0.001) |
| Logan et al. [126] | Longitudinal case series (3) | 56 children and adolescents, 8–18 yo with CRPS | Pain intensity; Functional disability; Subjective report of limb function, timed running, occupational performance, medication use, use of assistive devices, emotional functioning, anxiety and depression | Statistically significant improvements from admission to discharge in pain intensity, functional disability, subjective report of limb function, timed running, occupational performance, medication use, use of assistive devices, emotional functioning, anxiety, and depression. (all p < 0.01). (multiple outcomes) | |
| Simons et al. [127] | Comparative study (case-controlled) (3) | 100 children and adolescents, mean age 13.9 (SD 2.17) | Functional disability; Fear of pain; Readiness to change | Improved functional disability (p = 0.00); Improved fear of pain (p = 0.00); Increased readiness to change (precontemplation p = 0.01, action/maintenance p = 0.00) (multiple outcomes) | |
| Bruce et al. [51] | Prospective longitudinal case series (3) | 171 adolescents 12–18, mean age 15.3 (SD 1.73) | Functional disability; depressive symptoms; catastrophizing; Changes in opioid medication | Improved functional disability (p < 0.001) Improved depressive symptoms (p < 0.0001) Improved catastrophizing (p < 0.0001) Reduced pain severity (p = 0.0001) Decreased opioid medication (Out of 30 patients on opioids, 21 were weaned off medication, 1 did not finish program) |
References
- Pain terms: A list with definitions and notes on usage. Recommended by the IASP Subcommittee on Taxonomy. Pain 1979, 6, 249. [Google Scholar]
- Anwar, K. Pathophysiology of pain. Dis. Mon. 2016, 62, 324–329. [Google Scholar] [CrossRef] [PubMed]
- IASP Pain Terminology. International Association for the Study of Pain Committee on Taxonomy. Available online: http://www.iasp-pain.org/Taxonomy#Pain (accessed on 1 December 2018).
- Slater, M.E.; De Lima, J.; Campbell, K.; Lane, L.; Collins, J. Opioids for the management of severe chronic nonmalignant pain in children: A retrospective 1-year practice survey in a children’s hospital. Pain Med. 2010, 11, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Berde, C.; Nurko, S. Opioid side effects—Mechanism-based therapy. N. Engl. J. Med. 2008, 358, 2400–2402. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.A.; McQuay, H.J. Prevalence of opioid adverse events in chronic non-malignant pain: Systematic review of randomised trials of oral opioids. Arthritis Res. Ther. 2005, 7, R1046–R1051. [Google Scholar] [CrossRef] [PubMed]
- Gomes, T.; Mamdani, M.M.; Dhalla, I.A.; Paterson, J.M.; Juurlink, D.N. Opioid dose and drug-related mortality in patients with nonmalignant pain. Arch. Intern. Med. 2011, 171, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Grunkemeier, D.M.; Cassara, J.E.; Dalton, C.B.; Drossman, D.A. The narcotic bowel syndrome: Clinical features, pathophysiology, and management. Clin. Gastroenterol. Hepatol. 2007, 5, 1126–1139. [Google Scholar] [CrossRef]
- Fudin, J.; Raouf, M.; Wegrzyn, E.L. Opioid Dosing Policy: Pharmacological Considerations Regarding Equianalgesic Dosing; American Academy of Integrative Pain Management: Lenexa, KS, USA, 2017. [Google Scholar]
- Lee, M.; Silverman, S.M.; Hansen, H.; Patel, V.B.; Manchikanti, L. A comprehensive review of opioid-induced hyperalgesia. Pain Physician 2011, 14, 145–161. [Google Scholar]
- Task Force on Multimodal Pain Treatment Defines Terms for Chronic Pain Care. Available online: http://www.iasp-pain.org/PublicationsNews/NewsDetail.aspx?ItemNumber=6981 (accessed on 1 December 2018).
- Friedrichsdorf, S.J. Contemporary pediatric palliative care: Myths and barriers to integration into clinical care. Curr. Pediatr. Rev. 2016, 13, 8–12. [Google Scholar] [CrossRef]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm. Rep. 2016, 65, 1–49. [Google Scholar] [CrossRef]
- Dash, G.F.; Wilson, A.C.; Morasco, B.J.; Feldstein Ewing, S.W. A Model of the Intersection of Pain and Opioid Misuse in Children and Adolescents. Clin. Psychol. Sci. 2018, 6, 629–646. [Google Scholar] [CrossRef] [PubMed]
- Schechter, N.L.; Walco, G.A. The Potential Impact on Children of the CDC Guideline for Prescribing Opioids for Chronic Pain: Above All, Do No Harm. JAMA Pediatr. 2016, 170, 425–426. [Google Scholar] [CrossRef] [PubMed]
- Friedrichsdorf, S.J.; Giordano, J.; Desai Dakoji, K.; Warmuth, A.; Daughtry, C.; Schulz, C.A. Chronic Pain in Children and Adolescents: Diagnosis and Treatment of Primary Pain Disorders in Head, Abdomen, Muscles and Joints. Children (Basel) 2016, 3, 42. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.P.; Callahan, S.T.; Cooper, W.O.; Dupont, W.D.; Murray, K.T.; Franklin, A.D.; Hall, K.; Dudley, J.A.; Stein, C.M.; Ray, W.A. Outpatient Opioid Prescriptions for Children and Opioid-Related Adverse Events. Pediatrics 2018, 142. [Google Scholar] [CrossRef] [PubMed]
- Krane, E.J.; Weisman, S.J.; Walco, G.A. The National Opioid Epidemic and the Risk of Outpatient Opioids in Children. Pediatrics 2018, 142. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Lee, A.C.; Kemper, K.J.; Berde, C.B. Use of complementary and alternative medicine in pediatric pain management service: A survey. Pain Med. 2005, 6, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.; Law, E.; Dudeney, J.; Palermo, T.M.; Stewart, G.; Eccleston, C. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database Syst. Rev. 2018, 9. [Google Scholar] [CrossRef]
- Kamper, S.J.; Apeldoorn, A.T.; Chiarotto, A.; Smeets, R.J.; Ostelo, R.W.; Guzman, J.; van Tulder, M.W. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: Cochrane systematic review and meta-analysis. BMJ 2015, 350, h444. [Google Scholar] [CrossRef]
- Lee, C.; Crawford, C.; Swann, S. Active Self-Care Therapies for Pain (PACT) Working Group. Multimodal, integrative therapies for the self-management of chronic pain symptoms. Pain Med. 2014, 15 Suppl. 1, S76–S85. [Google Scholar] [CrossRef]
- Nicol, A.L.; Hurley, R.W.; Benzon, H.T. Alternatives to opioids in the pharmacologic management of chronic pain syndromes: A narrative review of randomized, controlled, and blinded clinical trials. Anesth. Analg. 2017, 125, 1682–1703. [Google Scholar] [CrossRef]
- Gardiner, P.; Lestoquoy, A.S.; Gergen-Barnett, K.; Penti, B.; White, L.F.; Saper, R.; Fredman, L.; Stillman, S.; Lily Negash, N.; Adelstein, P.; et al. Design of the integrative medical group visits randomized control trial for underserved patients with chronic pain and depression. Contemp. Clin. Trials 2017, 54, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Tick, H.; Nielsen, A.; Pelletier, K.R.; Bonakdar, R.; Simmons, S.; Glick, R.; Ratner, E.; Lemmon, R.L.; Wayne, P.; Zador, V.; et al. Evidence-Based Nonpharmacologic Strategies for Comprehensive Pain Care: The Consortium Pain Task Force White Paper. Explore (NY) 2018, 14, 177–211. [Google Scholar] [CrossRef] [PubMed]
- Waldman, S. Pain Management, 2nd ed.; Elsevier: Philadelphia, PA, USA, 2011; ISBN 9781437736038. [Google Scholar]
- Odell, S.; Logan, D.E. Pediatric pain management: The multidisciplinary approach. J. Pain Res. 2013, 6, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Gritsenko, K.; Khelemsky, Y.; Kaye, A.D.; Vadivelu, N.; Urman, R.D. Multimodal therapy in perioperative analgesia. Best Pract. Res. Clin. Anaesthesiol. 2014, 28, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.R.; Golianu, B. Perioperative management in children with chronic pain. Paediatr. Anaesth. 2016, 26, 794–806. [Google Scholar] [CrossRef] [PubMed]
- Panella, J.J. Preoperative care of children: Strategies from a child life perspective. AORN J. 2016, 104, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S.; Wang, S.; Porfyris, S.; Kamasinski-Sol, R.; Steinhorn, D.M. Massage therapy in outpatient pediatric chronic pain patients: Do they facilitate significant reductions in levels of distress, pain, tension, discomfort, and mood alterations? Paediatr. Anaesth. 2008, 18, 884–887. [Google Scholar] [CrossRef]
- Wang, S.M.; Escalera, S.; Lin, E.C.; Maranets, I.; Kain, Z.N. Extra-1 acupressure for children undergoing anesthesia. Anesth. Analg. 2008, 107, 811–816. [Google Scholar] [CrossRef]
- Brewer, S.; Gleditsch, S.L.; Syblik, D.; Tietjens, M.E.; Vacik, H.W. Pediatric anxiety: Child life intervention in day surgery. J. Pediatr. Nurs. 2006, 21, 13–22. [Google Scholar] [CrossRef]
- Yip, P.; Middleton, P.; Cyna, A.M.; Carlyle, A.V. Non-pharmacological interventions for assisting the induction of anaesthesia in children. Cochrane Database Syst. Rev. 2009, CD006447. [Google Scholar] [CrossRef]
- Manworren, R.C.B.; Girard, E.; Verissimo, A.M.; Ruscher, K.A.; Santanelli, J.P.; Weiss, R.; Hight, D. Hypnosis for postoperative pain management of thoracoscopic approach to repair pectus excavatum: Retrospective analysis. J. Pediatr. Surg. Nurs. 2015, 4, 60–69. [Google Scholar] [CrossRef]
- Jensen, B.; Chen, J.; Furnish, T.; Wallace, M. Medical marijuana and chronic pain: A review of basic science and clinical evidence. Curr. Pain Headache Rep. 2015, 19, 50. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Palermo, T.M.; Williams, A.C.; Lewandowski, H.A.; Morley, S.; Fisher, E.; Law, E. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database Syst. Rev. 2014, CD003968. [Google Scholar] [CrossRef] [PubMed]
- Hechler, T.; Kanstrup, M.; Holley, A.L.; Simons, L.E.; Wicksell, R.; Hirschfeld, G.; Zernikow, B. Systematic review on intensive interdisciplinary pain treatment of children with chronic pain. Pediatrics 2015, 136, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Scharff, L.; Sethna, N.F.; McCarthy, C.F.; Scott-Sutherland, J.; Shea, A.M.; Sullivan, P.; Meier, P.; Zurakowski, D.; Masek, B.J.; et al. Physical therapy and cognitive-behavioral treatment for complex regional pain syndromes. J. Pediatr. 2002, 141, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Kuttner, L. Pediatric hypnosis: Pre-, peri-, and post-anesthesia. Paediatr. Anaesth. 2012, 22, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L. Disrupting the downward spiral of chronic pain and opioid addiction with mindfulness-oriented recovery enhancement: A review of clinical outcomes and neurocognitive targets. J. Pain Palliat. Care Pharmacother. 2014, 28, 122–129. [Google Scholar] [CrossRef]
- Agoston, A.M.; Sieberg, C.B. Nonpharmacologic treatment of pain. Semin. Pediatr. Neurol. 2016, 23, 220–223. [Google Scholar] [CrossRef]
- Golianu, B.; Seybold, J.; Almgren, C. Acupucture helps reduce need for sedative medications in neonates and infants undergoing treatment in the intensive care unit. Med. Acupunct. 2014, 26, 279–285. [Google Scholar] [CrossRef]
- Schmitt, Y.S.; Hoffman, H.G.; Blough, D.K.; Patterson, D.R.; Jensen, M.P.; Soltani, M.; Carrougher, G.J.; Nakamura, D.; Sharar, S.R. A randomized, controlled trial of immersive virtual reality analgesia, during physical therapy for pediatric burns. Burns 2011, 37, 61–68. [Google Scholar] [CrossRef]
- Brown, M.L.; Rojas, E.; Gouda, S. A Mind-body approach to pediatric pain management. Children (Basel) 2017, 4, 50. [Google Scholar] [CrossRef] [PubMed]
- Rabin, J.; Brown, M.; Alexander, S. Update in the treatment of chronic pain within pediatric patients. Curr. Probl. Pediatr. Adolesc. Health Care 2017, 47, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Dhond, R.P.; Yeh, C.; Park, K.; Kettner, N.; Napadow, V. Acupuncture modulates resting state connectivity in default and sensorimotor brain networks. Pain 2008, 136, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; White, M.P.; Greicius, M.D.; Waelde, L.C.; Spiegel, D. Brain activity and functional connectivity associated with hypnosis. Cereb. Cortex 2017, 27, 4083–4093. [Google Scholar] [CrossRef] [PubMed]
- Kucyi, A.; Salomons, T.V.; Davis, K.D. Cognitive behavioral training reverses the effect of pain exposure on brain network activity. Pain 2016, 157, 1895–1904. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.C.; Dorflinger, L.; Edmond, S.N.; Islam, L.; Heapy, A.A.; Fraenkel, L. Barriers and facilitators to use of non-pharmacological treatments in chronic pain. BMC Fam. Pract. 2017, 18, 41. [Google Scholar] [CrossRef] [PubMed]
- Bruce, B.K.; Ale, C.M.; Harrison, T.E.; Bee, S.; Luedtke, C.; Geske, J.; Weiss, K.E. Getting back to living: Further evidence for the efficacy of an interdisciplinary pediatric pain treatment program. Clin. J. Pain 2017, 33, 535–542. [Google Scholar] [CrossRef]
- Chambless, D.L.; Hollon, S.D. Defining empirically supported therapies. J. Consult. Clin. Psychol. 1998, 66, 7–18. [Google Scholar] [CrossRef]
- Butler, A.C.; Chapman, J.E.; Forman, E.M.; Beck, A.T. The empirical status of cognitive-behavioral therapy: A review of meta-analyses. Clin. Psychol. Rev. 2006, 26, 17–31. [Google Scholar] [CrossRef]
- Beck, J.S. Cognitive Behavior Therapy: Basics and Beyond; Guilford Press: New York, NY, USA, 2011. [Google Scholar]
- Wicksell, R.K.; Greco, L.A. Acceptance and commitment therapy for pediatric chronic pain. In Acceptance and Mindfulness Treatments for Children and Adolescents: A Practitioner’s Guide; New Harbinger Publications: Oakland, CA, USA, 2008; pp. 89–113. ISBN 978-1-57224-541-9. [Google Scholar]
- Grave, J.; Blissett, J. Is cognitive behavior therapy developmentally appropriate for young children? A critical review of the evidence. Clin. Psychol. Rev. 2004, 24, 399–420. [Google Scholar] [CrossRef]
- Seminowicz, D.A.; Shpaner, M.; Keaser, M.L.; Krauthamer, G.M.; Mantegna, J.; Dumas, J.A.; Newhouse, P.A.; Filippi, C.G.; Keefe, F.J.; Naylor, M.R. Cognitive-behavioral therapy increases prefrontal cortex gray matter in patients with chronic pain. J. Pain 2013, 14, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.B.; Kosek, E.; Wicksell, R.; Kemani, M.; Olsson, G.; Merle, J.V.; Kadetoff, D.; Ingvar, M. Cognitive Behavioral Therapy increases pain-evoked activation of the prefrontal cortex in patients with fibromyalgia. Pain 2012, 153, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C.; Eccleston, C.; Morley, S. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochrane Database Syst. Rev. 2012, 11, CD007407. [Google Scholar] [CrossRef] [PubMed]
- Coakley, R.; Wihak, T. Evidence-based psychological interventions for the management of pediatric chronic pain: New directions in research and clinical practice. Children (Basel) 2017, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Palermo, T.M.; Eccleston, C.; Lewandowski, A.S.; Williams, A.C.; Morley, S. Randomized controlled trials of psychological therapies for management of chronic pain in children and adolescents: An updated meta-analytic review. Pain 2010, 148, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.; Heathcote, L.; Palermo, T.M.; de C Williams, A.C.; Lau, J.; Eccleston, C. Systematic review and meta-analysis of psychological therapies for children with chronic pain. J. Pediatr. Psychol. 2014, 39, 763–782. [Google Scholar] [CrossRef] [PubMed]
- Palermo, T.M.; Wilson, A.C.; Peters, M.; Lewandowski, A.; Somhegyi, H. Randomized controlled trial of an Internet-delivered family cognitive-behavioral therapy intervention for children and adolescents with chronic pain. Pain 2009, 146, 205–213. [Google Scholar] [CrossRef]
- Huestis, S.E.; Kao, G.; Dunn, A.; Hilliard, A.T.; Yoon, I.A.; Golianu, B.; Bhandari, R.P. Multi-Family Pediatric Pain Group Therapy: Capturing Acceptance and Cultivating Change. Children (Basel) 2017, 4, 106. [Google Scholar] [CrossRef]
- Coakley, R.; Wihak, T.; Kossowsky, J.; Iversen, C.; Donado, C. The Comfort Ability Pain Management Workshop: A Preliminary, Nonrandomized Investigation of a Brief, Cognitive, Biobehavioral, and Parent Training Intervention for Pediatric Chronic Pain. J. Pediatr. Psychol. 2018, 43, 252–265. [Google Scholar] [CrossRef]
- Kabat-Zinn, J. Wherever You Go, There You Are; Hyperion: New York, NY, USA, 1994. [Google Scholar]
- Black, D.S.; Slavich, G.M. Mindfulness meditation and the immune system: A systematic review of randomized controlled trials. Ann. N. Y. Acad. Sci. 2016, 1373, 13–24. [Google Scholar] [CrossRef]
- Grossman, P.; Niemann, L.; Schmidt, S.; Walach, H. Mindfulness-based stress reduction and health benefits. A meta-analysis. J. Psychosom. Res. 2004, 57, 35–43. [Google Scholar] [CrossRef]
- Ahola Kohut, S.; Stinson, J.; Davies-Chalmers, C.; Ruskin, D.; van Wyk, M. Mindfulness-based interventions in clinical samples of adolescents with chronic illness: A systematic review. J. Altern. Complement. Med. 2017, 23, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Baer, R.A. Mindfulness training as a clinical intervention: A conceptual and empirical review. Clin. Psychol. Sci. Pract. 2003, 10, 125–143. [Google Scholar] [CrossRef]
- Garland, E.L.; Froeliger, B.; Zeidan, F.; Partin, K.; Howard, M.O. The downward spiral of chronic pain, prescription opioid misuse, and addiction: Cognitive, affective, and neuropsychopharmacologic pathways. Neurosci. Biobehav. Rev. 2013, 37, 2597–2607. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L.; Gaylord, S.A.; Palsson, O.; Faurot, K.; Douglas Mann, J.; Whitehead, W.E. Therapeutic mechanisms of a mindfulness-based treatment for IBS: Effects on visceral sensitivity, catastrophizing, and affective processing of pain sensations. J. Behav. Med. 2012, 35, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Holzel, B.K.; Lazar, S.W.; Gard, T.; Schuman-Olivier, Z.; Vago, D.R.; Ott, U. How does mindfulness meditation work? Proposing mechanisms of action from a conceptual and neural perspective. Perspect. Psychol. Sci. 2011, 6, 537–559. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, F.; Martucci, K.T.; Kraft, R.A.; Gordon, N.S.; McHaffie, J.G.; Coghill, R.C. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J. Neurosci. 2011, 31, 5540–5548. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, F.; Vago, D.R. Mindfulness meditation-based pain relief: A mechanistic account. Ann. N. Y. Acad. Sci. 2016, 1373, 114–127. [Google Scholar] [CrossRef]
- Wager, T.D.; Scott, D.J.; Zubieta, J.K. Placebo effects on human mu-opioid activity during pain. Proc. Natl. Acad. Sci. USA 2007, 104, 11056–11061. [Google Scholar] [CrossRef]
- Sharon, H.; Maron-Katz, A.; Ben Simon, E.; Flusser, Y.; Hendler, T.; Tarrasch, R.; Brill, S. Mindfulness meditation modulates pain through endogenous opioids. Am. J. Med. 2016, 129, 755–758. [Google Scholar] [CrossRef]
- Zeidan, F.; Adler-Neal, A.L.; Wells, R.E.; Stagnaro, E.; May, L.M.; Eisenach, J.C.; McHaffie, J.G.; Coghill, R.C. Mindfulness-meditation-based pain relief is not mediated by endogenous opioids. J. Neurosci. 2016, 36, 3391–3397. [Google Scholar] [CrossRef] [PubMed]
- Veehof, M.M.; Trompetter, H.R.; Bohlmeijer, E.T.; Schreurs, K.M. Acceptance- and mindfulness-based interventions for the treatment of chronic pain: A meta-analytic review. Cogn. Behav. Ther. 2016, 45, 5–31. [Google Scholar] [CrossRef] [PubMed]
- Hilton, L.; Hempel, S.; Ewing, B.A.; Apaydin, E.; Xenakis, L.; Newberry, S.; Colaiaco, B.; Maher, A.R.; Shanman, R.M.; Sorbero, M.E.; et al. Mindfulness meditation for chronic pain: Systematic review and meta-analysis. Ann. Behav. Med. 2017, 51, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Anheyer, D.; Haller, H.; Barth, J.; Lauche, R.; Dobos, G.; Cramer, H. Mindfulness-based stress reduction for treating low back pain: A systematic review and meta-analysis. Ann. Intern. Med. 2017, 166, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Ruskin, D.A.; Gagnon, M.M.; Kohut, S.A.; Stinson, J.N.; Walker, K.S. A mindfulness program adapted for adolescents with chronic pain: Feasibility, acceptability, and initial outcomes. Clin. J. Pain 2017. [Google Scholar] [CrossRef] [PubMed]
- Chadi, N.; McMahon, A.; Vadnais, M.; Malboeuf-Hurtubise, C.; Djemli, A.; Dobkin, P.L.; Lacroix, J.; Luu, T.M.; Haley, N. Mindfulness-based intervention for female adolescents with chronic pain: A pilot randomized trial. J. Can. Acad. Child Adolesc. Psychiatry 2016, 25, 159–168. [Google Scholar] [PubMed]
- Ali, A.; Weiss, T.R.; Dutton, A.; McKee, D.; Jones, K.D.; Kashikar-Zuck, S.; Silverman, W.K.; Shapiro, E.D. Mindfulness-based stress reduction for adolescents with functional somatic syndromes: A pilot cohort study. J. Pediatr. 2017, 183, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Waelde, L.C.; Feinstein, A.B.; Bhandari, R.; Griffin, A.; Yoon, I.A.; Golianu, B. A pilot study of mindfulness meditation for pediatric chronic pain. Children (Basel) 2017, 4, 32. [Google Scholar] [CrossRef]
- Hesse, T.; Holmes, L.G.; Kennedy-Overfelt, V.; Kerr, L.M.; Giles, L.L. Mindfulness-based intervention for adolescents with recurrent headaches: A pilot feasibility study. Evid. Based Complement. Alternat. Med. 2015, 2015, 508958. [Google Scholar] [CrossRef]
- Jastrowski Mano, K.E.; Salamon, K.S.; Hainsworth, K.R.; Anderson Khan, K.J.; Ladwig, R.J.; Davies, W.H.; Weisman, S.J. A randomized, controlled pilot study of mindfulness-based stress reduction for pediatric chronic pain. Altern. Ther. Health Med. 2013, 19, 8–14. [Google Scholar]
- Ruskin, D.; Lalloo, C.; Amaria, K.; Stinson, J.N.; Kewley, E.; Campbell, F.; Brown, S.C.; Jeavons, M.; McGrath, P.A. Assessing pain intensity in children with chronic pain: Convergent and discriminant validity of the 0 to 10 numerical rating scale in clinical practice. Pain Res. Manag. 2014, 19, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Zoogman, S.; Goldberg, S.B.; Hoyt, W.T.; Miller, L. Mindfulness interventions with youth: A meta-analysis. Mindfulness 2015, 6, 290–302. [Google Scholar] [CrossRef]
- Zenner, C.; Herrnleben-Kurz, S.; Walach, H. Mindfulness-based interventions in schools-a systematic review and meta-analysis. Front. Psychol. 2014, 5, 603. [Google Scholar] [CrossRef] [PubMed]
- Sibinga, E.M.; Webb, L.; Ghazarian, S.R.; Ellen, J.M. School-Based Mindfulness Instruction: An RCT. Pediatrics 2016, 137. [Google Scholar] [CrossRef] [PubMed]
- Mani, M.; Kavanagh, D.J.; Hides, L.; Stoyanov, S.R. Review and Evaluation of Mindfulness-Based iPhone Apps. JMIR Mhealth Uhealth 2015, 3, e82. [Google Scholar] [CrossRef] [PubMed]
- Hilgard, E.R. Hypnotic Susceptibility; Harcourt, Brace & World Inc.: New York, NY, USA, 1965. [Google Scholar]
- Spiegel, H.; Spiegel, D. Trance and Treatment: Clinical Uses of Hypnosis; American Psychistric Publishing: Washington, DC, USA, 1987; ISBN 978-1585621903. [Google Scholar]
- Lobe, T.E. Perioperative hypnosis reduces hospitalization in patients undergoing the Nuss procedure for pectus excavatum. J. Laparoendosc. Adv. Surg. Tech. A 2006, 16, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Gruzelier, J.H. A working model of the neurophysiology of hypnosis: A review of the evidence. Contemp. Hypn. 1998, 15, 3–21. [Google Scholar] [CrossRef]
- Jensen, M.P.; Patterson, D.R. Hypnotic approaches for chronic pain management: Clinical implications of recent research findings. Am. Psychol. 2014, 69, 167–177. [Google Scholar] [CrossRef]
- Crawford, H.J.; Horton, J.E.; Harrington, G.C.; Vendemia, J.M.C.; Plantec, M.B.; Jung, S.; Shamro, C.; Downs, J.H., III. Hypnotic analgesia (Disattending pain) impacts neuronal network activation: An fMRI study of noxious somatosensory TENS stimuli. Neuroimage 1998, 7, S436. [Google Scholar] [CrossRef]
- Rainville, P.; Duncan, G.H.; Price, D.D.; Carrier, B.; Bushnell, M.C. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science 1997, 277, 968–971. [Google Scholar] [CrossRef]
- Montgomery, G.H.; DuHamel, K.N.; Redd, W.H. A meta-analysis of hypnotically induced analgesia: How effective is hypnosis? Int. J. Clin. Exp. Hypn. 2000, 48, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Adachi, T.; Fujino, H.; Nakae, A.; Mashimo, T.; Sasaki, J. A meta-analysis of hypnosis for chronic pain problems: A comparison between hypnosis, standard care, and other psychological interventions. Int. J. Clin. Exp. Hypn. 2014, 62, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Lambert, S.A. The effects of hypnosis/guided imagery on the postoperative course of children. J. Dev. Behav. Pediatr. 1996, 17, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Liossi, C.; Hatira, P. Clinical hypnosis versus cognitive behavioral training for pain management with pediatric cancer patients undergoing bone marrow aspirations. Int. J. Clin. Exp. Hypn. 1999, 47, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Vlieger, A.M.; Menko-Frankenhuis, C.; Wolfkamp, S.C.; Tromp, E.; Benninga, M.A. Hypnotherapy for children with functional abdominal pain or irritable bowel syndrome: A randomized controlled trial. Gastroenterology 2007, 133, 1430–1436. [Google Scholar] [CrossRef] [PubMed]
- Vlieger, A.M.; Rutten, J.M.; Govers, A.M.; Frankenhuis, C.; Benninga, M.A. Long-term follow-up of gut-directed hypnotherapy vs. standard care in children with functional abdominal pain or irritable bowel syndrome. Am. J. Gastroenterol. 2012, 107, 627–631. [Google Scholar] [CrossRef]
- Anbar, R.D. Self-hypnosis for the treatment of functional abdominal pain in childhood. Clin. Pediatr. (Phila) 2001, 40, 447–451. [Google Scholar] [CrossRef]
- Olness, K. Imagery (self-hypnosis) as adjunct therapy in childhood cancer: Clinical experience with 25 patients. Am. J. Pediatr. Hematol. Oncol. 1981, 3, 313–321. [Google Scholar]
- Kohen, D.P.; Zajac, R. Self-hypnosis training for headaches in children and adolescents. J. Pediatr. 2007, 150, 635–639. [Google Scholar] [CrossRef]
- Olness, K.; MacDonald, J.T.; Uden, D.L. Comparison of self-hypnosis and propranolol in the treatment of juvenile classic migraine. Pediatrics 1987, 79, 593–597. [Google Scholar]
- Lin, Y.C.; Wan, L.; Jamison, R.N. Using integrative medicine in pain management: An evaluation of current evidence. Anesth. Analg. 2017, 125, 2081–2093. [Google Scholar] [CrossRef]
- Hui, K.K.; Liu, J.; Marina, O.; Napadow, V.; Haselgrove, C.; Kwong, K.K.; Kennedy, D.N.; Makris, N. The integrated response of the human cerebro-cerebellar and limbic systems to acupuncture stimulation at ST 36 as evidenced by fMRI. Neuroimage 2005, 27, 479–496. [Google Scholar] [CrossRef]
- Napadow, V.; Kettner, N.; Liu, J.; Li, M.; Kwong, K.K.; Vangel, M.; Makris, N.; Audette, J.; Hui, K.K. Hypothalamus and amygdala response to acupuncture stimuli in Carpal Tunnel Syndrome. Pain 2007, 130, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.M.; Kain, Z.N.; White, P. Acupuncture analgesia: I. The scientific basis. Anesth. Analg. 2008, 106, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Oke, S.L.; Tracey, K.J. The inflammatory reflex and the role of complementary and alternative medical therapies. Ann. N. Y. Acad. Sci. 2009, 1172, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Chavan, S.S.; Tracey, K.J. Regulating innate immunity with dopamine and electroacupuncture. Nat. Med. 2014, 20, 239–241. [Google Scholar] [CrossRef] [PubMed]
- NIH consensus conference: Acupuncture. JAMA 1998, 280, 1518–1524. [CrossRef]
- Haake, M.; Muller, H.H.; Schade-Brittinger, C.; Basler, H.D.; Schafer, H.; Maier, C.; Endres, H.G.; Trampisch, H.J.; Molsberger, A. German Acupuncture Trials (GERAC) for chronic low back pain: Randomized, multicenter, blinded, parallel-group trial with 3 groups. Arch. Intern. Med. 2007, 167, 1892–1898. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, J.; Li, Y.; Sun, X.; Chang, X.; Zheng, H.; Gong, B.; Huang, Y.; Yang, M.; Wu, X.; et al. The long-term effect of acupuncture for migraine prophylaxis: A randomized clinical trial. JAMA Intern. Med. 2017, 177, 508–515. [Google Scholar] [CrossRef]
- Berman, B.M.; Lao, L.; Langenberg, P.; Lee, W.L.; Gilpin, A.M.; Hochberg, M.C. Effectiveness of acupuncture as adjunctive therapy in osteoarthritis of the knee: A randomized, controlled trial. Ann. Intern. Med 2004, 141, 901–910. [Google Scholar] [CrossRef]
- Golianu, B.; Yeh, A.M.; Brooks, M. Acupuncture for pediatric pain. Children (Basel) 2014, 1, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Tassone, R.F.; Jahng, S.; Rahbar, R.; Holzman, R.S.; Zurakowski, D.; Sethna, N.F. Acupuncture management of pain and emergence agitation in children after bilateral myringotomy and tympanostomy tube insertion. Paediatr. Anaesth. 2009, 19, 1096–1101. [Google Scholar] [CrossRef] [PubMed]
- Tsao, G.J.; Messner, A.H.; Seybold, J.; Sayyid, Z.N.; Cheng, A.G.; Golianu, B. Intraoperative acupuncture for posttonsillectomy pain: A randomized, double-blind, placebo-controlled trial. Laryngoscope 2015, 125, 1972–1978. [Google Scholar] [CrossRef] [PubMed]
- Raith, W.; Schmolzer, G.M.; Resch, B.; Reiterer, F.; Avian, A.; Koestenberger, M.; Urlesberger, B. Laser acupuncture for neonatal abstinence syndrome: A randomized controlled trial. Pediatrics 2015, 136, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Gottschling, S.; Meyer, S.; Gribova, I.; Distler, L.; Berrang, J.; Gortner, L.; Graf, N.; Shamdeen, M.G. Laser acupuncture in children with headache: A double-blind, randomized, bicenter, placebo-controlled trial. Pain 2008, 137, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Wayne, P.M.; Kerr, C.E.; Schnyer, R.N.; Legedza, A.T.; Savetsky-German, J.; Shields, M.H.; Buring, J.E.; Davis, R.B.; Conboy, L.A.; Highfield, E.; et al. Japanese-style acupuncture for endometriosis-related pelvic pain in adolescents and young women: Results of a randomized sham-controlled trial. J. Pediatr. Adolesc. Gynecol. 2008, 21, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Logan, D.E.; Carpino, E.A.; Chiang, G.; Condon, M.; Firn, E.; Gaughan, V.J.; Hogan, M.; Leslie, D.S.; Olson, K.; Sager, S.; et al. A day-hospital approach to treatment of pediatric complex regional pain syndrome: Initial functional outcomes. Clin. J. Pain 2012, 28, 766–774. [Google Scholar] [CrossRef]
- Simons, L.E.; Sieberg, C.B.; Pielech, M.; Conroy, C.; Logan, D.E. What does it take? Comparing intensive rehabilitation to outpatient treatment for children with significant pain-related disability. J. Pediatr. Psychol. 2013, 38, 213–223. [Google Scholar] [CrossRef]
- American Pain Society. Pain in Infants, Children, and Adolescents SIG. Available online: http://americanpainsociety.org/get-involved/shared-interest-groups/pediatric-adolescent-pain (accessed on 1 December 2018).
| Modalities | Ovid | Embase | Prospero | Cochrane | Pubmed | Number of Articles Screened | Number of Articles Reviewed |
|---|---|---|---|---|---|---|---|
| CBT | 42 | 537 | 2 | 2 | 274 | 48 | 12 |
| Mindfulness | 37 | 53 | 2 | 17 | 26 | 45 | 25 |
| Hypnosis | 164 | 272 | 1 | 1 | 152 | 28 | 14 |
| Acupuncture | 149 | 357 | 2 | 24 | 132 | 32 | 14 |
| Intensive Rehab | 189 | 164 | 2 | 51 | 87 | 15 | 6 |
| Multidisciplinary | 324 | 1232 | 1 | 19 | 546 | 52 | 13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wren, A.A.; Ross, A.C.; D’Souza, G.; Almgren, C.; Feinstein, A.; Marshall, A.; Golianu, B. Multidisciplinary Pain Management for Pediatric Patients with Acute and Chronic Pain: A Foundational Treatment Approach When Prescribing Opioids. Children 2019, 6, 33. https://doi.org/10.3390/children6020033
Wren AA, Ross AC, D’Souza G, Almgren C, Feinstein A, Marshall A, Golianu B. Multidisciplinary Pain Management for Pediatric Patients with Acute and Chronic Pain: A Foundational Treatment Approach When Prescribing Opioids. Children. 2019; 6(2):33. https://doi.org/10.3390/children6020033
Chicago/Turabian StyleWren, Anava A., Alexandra C. Ross, Genevieve D’Souza, Christina Almgren, Amanda Feinstein, Amanda Marshall, and Brenda Golianu. 2019. "Multidisciplinary Pain Management for Pediatric Patients with Acute and Chronic Pain: A Foundational Treatment Approach When Prescribing Opioids" Children 6, no. 2: 33. https://doi.org/10.3390/children6020033
APA StyleWren, A. A., Ross, A. C., D’Souza, G., Almgren, C., Feinstein, A., Marshall, A., & Golianu, B. (2019). Multidisciplinary Pain Management for Pediatric Patients with Acute and Chronic Pain: A Foundational Treatment Approach When Prescribing Opioids. Children, 6(2), 33. https://doi.org/10.3390/children6020033





