Anxiety in Young Children with Williams Syndrome: A Longitudinal Study
Abstract
Highlights
- •
- Anxiety symptomology is highly prevalent in very young children with Williams syndrome, and seems to increase with age/over time (with many children developing co-occurring anxiety disorder symptoms).
- •
- Our longitudinal findings also provide evidence for the contribution of environmental factors on the nature, developmental course, and maintenance of anxiety, notably, chronological age, sex, and IQ.
- •
- The importance of screening the Williams syndrome population at a very early age and allows clinicians to identify those most at risk of developing an anxiety disorder.
- •
- The existing anxiety classification systems should be reviewed to reflect specific symptomology in young children with intellectual disabilities or delayed verbal skills, and implications for early intervention programmes aimed at reducing anxiety risk in Williams syndrome individuals.
Abstract
1. Anxiety in Young Children with Williams Syndrome: A Longitudinal Study
2. Anxiety in Williams Syndrome
2.1. How Are Chronological Age, Sex, and IQ Related to Anxiety in WS?
2.2. Longitudinal Studies of Anxiety in WS
2.3. Anxiety in Young Children with WS
2.4. The Current Study
3. Methods
3.1. Participants
3.2. Materials
3.3. Anxiety Measures
3.4. Combining Anxiety Data for Time 2
3.5. Developmental Measure
3.6. Analytic Approach
4. Results
4.1. Time 1—Prevalence of Anxiety in Young Children with WS
4.2. Time 2—Longitudinal Trajectory of Anxiety in Young Children with WS
4.3. Heterogeneity of Anxiety in Young Children with WS over Time
4.4. Environmental Contributions to Anxiety Change over Time
4.4.1. Relationship Between Cardiovascular Abnormalities and Symptoms of a Phobia of Blood Injectional Injury
4.4.2. Relationship Between Hyperacusis and Symptoms of a Phobia of Loud Noises
4.4.3. Relationship Between Anxiety and Chronological Age, Sex, and DQ over Time
4.4.4. Relationship Between Chronological Age and Anxiety over Time
4.4.5. Relationship Between Sex and Anxiety over Time
4.4.6. Relationship Between Verbal, Nonverbal, and Global DQ and Anxiety over Time
5. Discussion
5.1. Prevalence of Anxiety in Very Young Children with WS
5.2. Developmental Trajectory of Anxiety in Young WS Children
5.3. Environmental Influences on Anxiety in Young WS Children
5.4. The Association Between Anxiety over Time and Chronological Age, Sex, and IQ
5.5. Considerations and Future Research
5.6. Clinical Implications
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Mean (SD) | Range | |
|---|---|---|
| CA (years) a | 2.60 (0.25) | 2.21–2.98 |
| Gender distribution | 7 M, 7 F | |
| Overall DQ b | 109.45 (20.15) | 72.17–151.47 |
| Verbal DQ b | 107.15 (20.48) | 63.81–141.88 |
| Nonverbal DQ b | 106.31 (17.83) | 80.52–136.01 |
| Primary Caregiver Age (Years) | 35.11 (5.78) | 27–44 |
| PAS (raw scores) | ||
| Total Anxiety | 12.64 (9.96) | 1–33 |
| GAD c | 1.14 (2.11) | 0–8 |
| Social Anxiety | 3.64 (3.13) | 0–10 |
| OCD d | 1.21 (1.81) | 0–6 |
| Physical Injury Fears | 3.50 (3.00) | 0–11 |
| Separation Anxiety | 3.07 (3.20) | 0–12 |
Appendix B. Longitudinal Anxiety Data
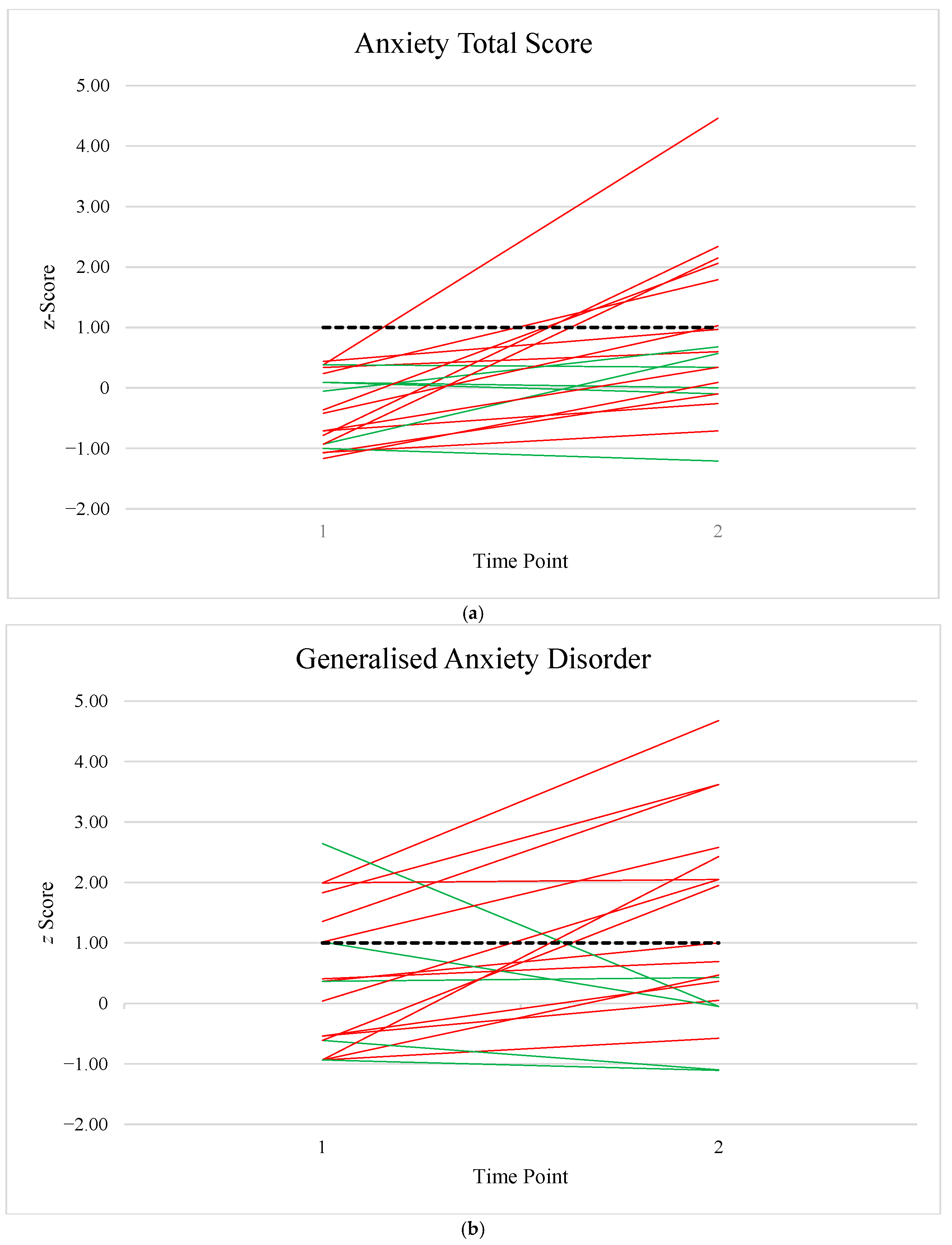
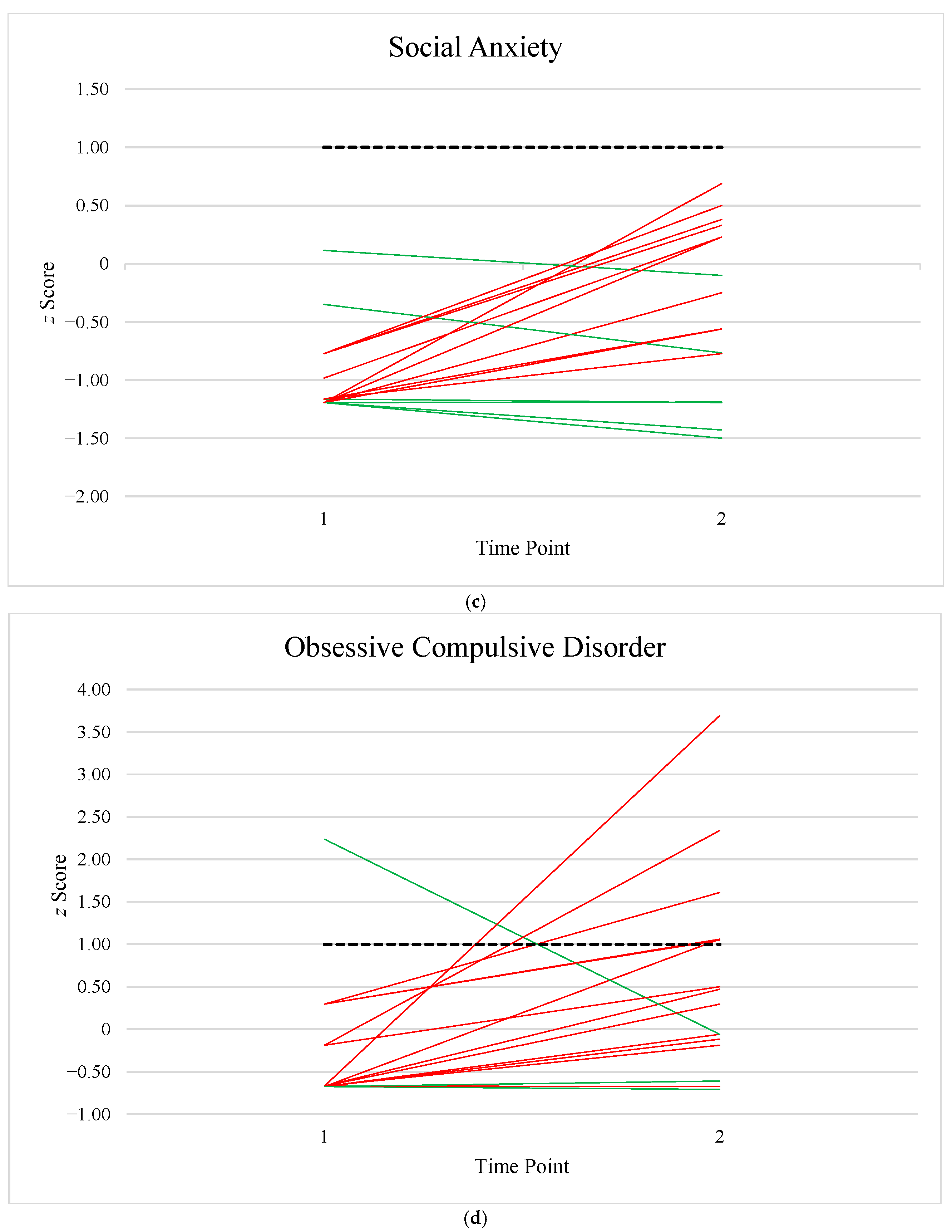
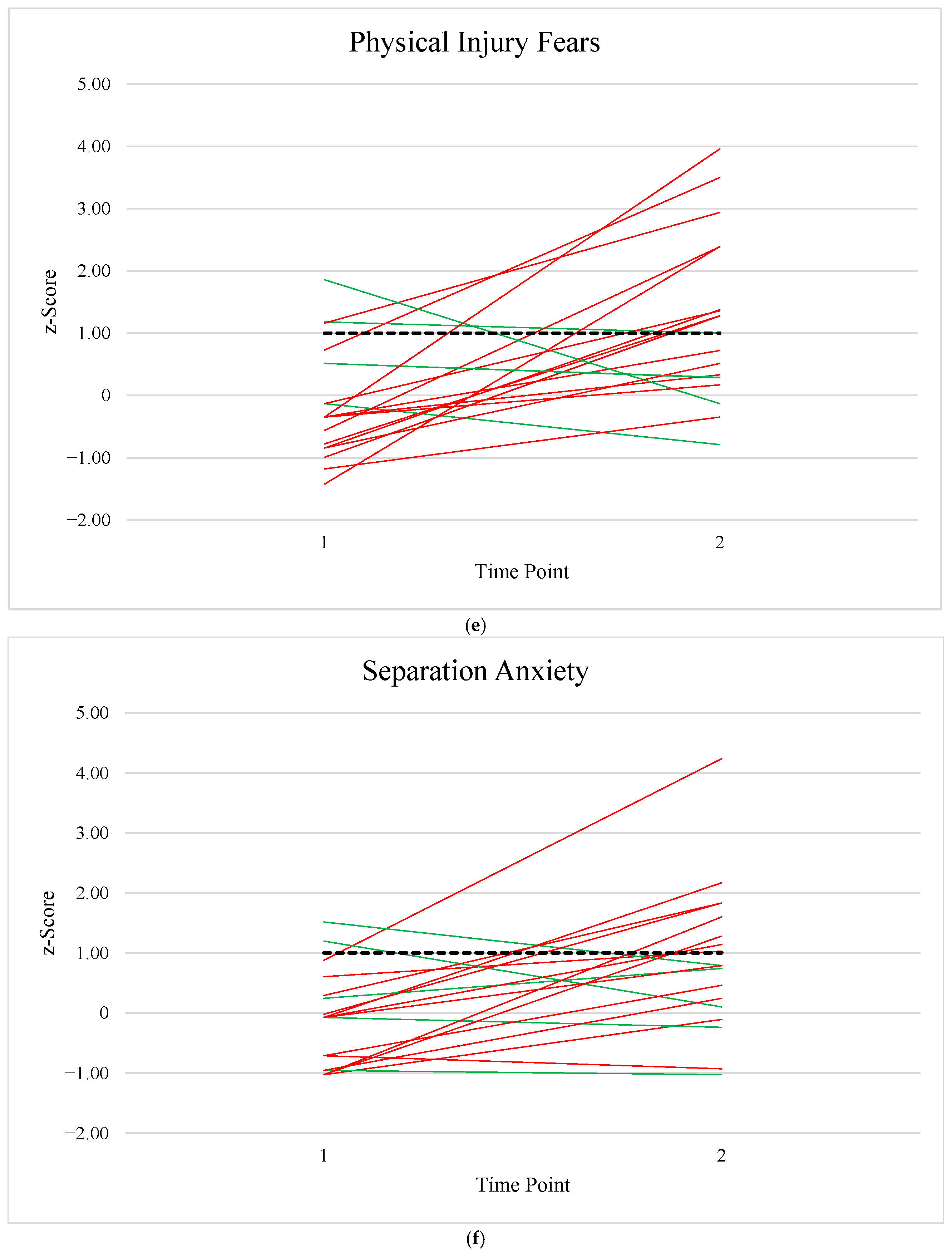
References
- Kozel, B.A.; Barak, B.; Kim, C.; Mervis, C.B.; Osborne, L.R.; Porter, M.; Pober, B.R. Williams syndrome. Nat. Rev. Dis. 2021, 7, 42. [Google Scholar] [CrossRef]
- Dodd, H.F.; Porter, M.A. Psychopathology in Williams syndrome: The effect of individual differences across the life span. J. Ment. Health Res. Intellect. Disabil. 2009, 2, 89–109. [Google Scholar] [CrossRef]
- Leyfer, O.T.; Woodruff-Bordon, J.; Klein-Tasman, B.P.; Fricke, J.S.; Mervis, C.B. Prevalence of psychiatric disorders in 4 to 6-year-olds with Williams syndrome. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2006, 141B, 615–622. [Google Scholar] [CrossRef]
- Ng-Cordell, E.; Hanley, M.; Kelly, A.; Riby, D.M. Anxiety in Williams syndrome: The role of social behaviour, executive functions, and change over time. J. Autism Dev. Disord. 2018, 48, 796–808. [Google Scholar] [CrossRef]
- Davies, M.; Udwin, O.; Howlin, P. Adults with Williams syndrome: Preliminary study if social, emotional and behavioural difficulties. Br. J. Psychiatry 1998, 172, 273–276. [Google Scholar] [CrossRef]
- Leyfer, O.; Woodruff-Borden, J.; Mervis, C.B. Anxiety disorders in children with Williams syndrome, their mothers, and their siblings: Implications for the etiology of anxiety disorders. J. Neurodev. Disord. 2009, 1, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Plissart, L.; Borghgraef, M.; Volcke, P.; Van den Berghe, H.; Fryns, J.P. Adults with Williams-Beuren syndrome: Evaluation of the medical, psychological and behavioural aspects. Clin. Genet. 1994, 46, 161–167. [Google Scholar] [CrossRef]
- Riby, D.M.; Hanley, M.; Kirk, H.; Clark, F.; Little, K.; Fleck, R.; Janes, E.; Kelso, L.; O’Kane, F.; Cole-Fletcher, R.; et al. The interplay between anxiety and social functioning in Williams syndrome. J. Autism Dev. Disord. 2014, 44, 1220–1229. [Google Scholar] [CrossRef]
- Udwin, O.; Howlin, P.; Davies, M.; Mannion, E. Community care for adults with Williams syndrome: How families cope and the availability of support networks. JIDR 1998, 42, 238–245. [Google Scholar] [CrossRef]
- Giora, A.; Gega, L.; Landau, S.; Marks, I. Adult recall of having been bullied in attenders of an anxiety disorder unit and attenders of a dental clinic: A pilot controlled study. Behav. Change 2005, 22, 44–49. [Google Scholar] [CrossRef]
- La Greca, A.M.; Harrison, H.M. Adolescent peer relations, friendships, and romantic relationships: Do they predict social anxiety and depression? J. Clin. Child. Adolesc. Psychol. 2005, 34, 49–61. [Google Scholar] [CrossRef]
- Strauss, C.C.; Frame, C.L.; Forehand, R. Psychosocial impairment associated with anxiety in children. J. Clin. Neuropsychol. 1987, 16, 235–239. [Google Scholar] [CrossRef]
- Cole, D.A.; Peeke, L.G.; Martin, J.M.; Truglio, R.; Seroczynski, A.D. A longitudinal look at the relation between depression and anxiety in children and adolescents. J. Consult. Clin. Neuropsyhol 1998, 66, 451–460. [Google Scholar] [CrossRef]
- Stinton, C.; Elison, S.; Howlin, P. Mental health problems in adults with Williams syndrome. Am. J. Intellect. Dev. Disabil. 2010, 115, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Caspi, A.; Elder, G.H., Jr.; Bem, D.J. Moving away from the world: Life-course patterns of shy children. Dev. Psychol. 1988, 24, 824–831. [Google Scholar] [CrossRef]
- Last, C.G.; Hansen, C.; Franco, N. Anxious children in adulthood: A prospective study of adjustment. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Fryssira, H.; Palmer, R.; Hallidie-Smith, K.A.; Taylor, J.; Donnai, D.; Reardon, W. Flourescent in situ hybridisation (FISH) for hemizygous deletion at the elastin locus in patients with isolated supravalvar aortic stenosis. J. Med. Genet. 1997, 34, 306–308. [Google Scholar] [CrossRef]
- Dykens, E.M. Annotation: Psychopathology in children with intellectual disability. J. Child. Psychol. Psychiatry 2000, 41, 407–417. [Google Scholar] [CrossRef]
- Jabbi, M.; Kippenhan, J.S.; Kohn, P.; Marenco, S.; Mervis, C.B.; Morris, C.A.; Meyer-Lindenberg, A.; Berman, K.F. The Williams syndrome chromosome 7q11. 23 hemideletion confers hypersocial, anxious personality coupled with altered insula structure and function. Proc. Natl. Acad. Sci. USA 2012, 109, E860–E866. [Google Scholar] [CrossRef]
- Royston, R.; Howlin, P.; Waite, J.; Oliver, C. Anxiety disorders in Williams syndrome contrasted with intellectual disability and the general population: A systematic review and meta-analysis. J. Autism Dev. Disord. 2017, 47, 3765–3777. [Google Scholar] [CrossRef]
- Dykens, E.M. Anxiety, Fears, and Phobias in persons with Williams syndrome. Dev. Neuropsychol. 2003, 23, 291–316. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Lindenberg, A.; Hariri, A.R.; Munoz, K.E.; Mervis, C.B.; Mattay, V.S.; Morris, C.A.; Berman, K.F. Neural correlates of genetically abnormal social cognition in Williams syndrome. Nat. Neurosci. 2005, 8, 991–993. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Lindenberg, A.; Mervis, C.B.; Faith Berman, K. Neural mechanisms in Williams syndrome: A unique window to genetic influences on cognition and behaviour. Nat. Rev. Neurosci. 2006, 7, 380–393. [Google Scholar] [CrossRef]
- Muñoz, K.E.; Meyer-Lindenberg, A.; Hariri, A.R.; Mervis, C.B.; Mattay, V.S.; Morris, C.A.; Berman, K.F. Abnormalities in neural processing of emotional stimuli in Williams syndrome vary according to social vs. non-social content. Neuroimage 2010, 50, 340–346. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Haas, B.W.; Mills, D.; Yam, A.; Hoeft, F.; Bellugi, U.; Reiss, A. Genetic influences on sociability: Heightened amygdala reactivity and event-related responses to positive social stimuli in Williams syndrome. J. Neurosci. 2009, 29, 1132–1139. [Google Scholar] [CrossRef]
- Dodd, H.F.; Porter, M.A. I see happy people: Attention bias towards happy but not angry facial expressions in Williams syndrome. Cogn. Neuropsychiatry 2010, 15, 549–567. [Google Scholar] [CrossRef]
- Avery, S.N.; Thornton-Wells, T.A.; Anderson, A.W.; Urbano Blackfors, J. White matter integrity deficits in prefrontal-amygdala pathways in Williams syndrome. Neuroimage 2012, 43, 887–894. [Google Scholar] [CrossRef][Green Version]
- Thornton-Wells, T.A.; Avery, S.N.; Blackford, J.U. Using novel control groups to dissect the amygdala’s role in Williams syndrome. Dev. Cogn. Neurosci. 2011, 1, 295–304. [Google Scholar] [CrossRef]
- Rapee, R.M.; Schniering, C.A.; Hudson, J.L. Anxiety disorders during childhood and adolescence: Origins and treatment. Annu. Rev. Clin. Psychol. 2009, 5, 311–341. [Google Scholar] [CrossRef]
- Lieb, R.; Wittchen, H.U.; Höfler, M.; Fuetsch, M.; Stein, M.B.; Merikangas, K.R. Parental psychopathology, parenting styles, and the risk of social phobia in offspring: A prospective-longitudinal community study. Arch. Gen. Psychiatry 2000, 57, 859–866. [Google Scholar] [CrossRef]
- Ehringer, M.A.; Rhee, S.H.; Young, S.; Corley, R.; Hewitt, J.K. Genetic and environmental contributions to common psychopathologies of childhood and adolescence: A study of twins and their siblings. J. Abnorm. Child. Psychol. 2006, 34, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.M.; Eley, T.C. Genetic influences on anxiety in children: What we’ve learned and where we’re heading. Clin. Child. Fam. Psychol. Rev. 2007, 10, 199–212. [Google Scholar] [CrossRef]
- Thapar, A.; McGuffin, P. Are anxiety symptoms in childhood heritable? J. Child Psychol. Psychiatry Allied Discip. 1995, 36, 439–447. [Google Scholar] [CrossRef]
- Smoller, J.W. The genetics of stress-related disorders: PTSD, depression, and anxiety disorders. Neuropsychopharmacology 2016, 41, 297–319. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, K.; Nishi, A.; Kondo, K.; Yanagida, K.; Kawakami, N. Screening performance of K6/K10 and other screening instruments for mood and anxiety disorders in Japan. Psychiatry Clin. Neurosci. 2011, 65, 434–441. [Google Scholar] [CrossRef]
- Schubert, C. The genomic basis of the Williams–Beuren syndrome. Cell Mol. Sci. 2009, 66, 1178–1197. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.L.; Rapee, R.M. Familial and social environments in the etiology and maintenance of anxiety disorders. In Oxford Handbook of Anxiety and Related Disorders; Antony, M.M., Stein, M.B., Eds.; Oxford University Press: New York, NY, USA, 2009; pp. 173–189. [Google Scholar]
- Green, T.; Avda, S.; Dotan, I.; Zarchi, O.; Basel-Vanagaite, L.; Zalsman, G.; Weizman, A.; Gothelf, D. Phenotypic psychiatric characterization of children with Williams syndrome and response of those with ADHD to methylphenidate treatment. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2012, 159, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Woodruff-Borden, J.; Kistler, D.J.; Henderson, D.R.; Crawford, N.A.; Mervis, C.B. Longitudinal course of anxiety in children and adolescents with Williams syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 2010, 154C, 277–290. [Google Scholar] [CrossRef]
- Uljarević, M.; Labuschagne, I.; Bobin, R.; Atkinson, A.; Hocking, D.R. Brief report: The impact of sensory hypersensitivity and intolerance of uncertainty on anxiety in Williams syndrome. J. Autism Dev. Disord. 2018, 48, 3958–3964. [Google Scholar] [CrossRef]
- Gagliardi, C.; Martelli, S.; Tavano, A.; Borgatti, R. Behavioural features of Italian infants and young adults with Williams–Beuren syndrome. J. Intellect. Disabil. Res. 2011, 55, 121–131. [Google Scholar] [CrossRef]
- Hahn, L.J.; Fidler, D.J.; Hepburn, S.L. Adaptive behavior and problem behavior in young children with Williams syndrome. Am. J. Intellect. Dev. Disabil. 2014, 119, 49–63. [Google Scholar] [CrossRef]
- Klein-Tasman, B.P.; Lee, K. Problem behaviour and psychosocial functioning in young children with Williams syndrome: Parent and teacher perspectives. J. Intellect. Disabil. Res. 2017, 61, 853–865. [Google Scholar] [CrossRef]
- Braga, A.C.; Carreiro, L.R.R.; Tafla, T.L.; Ranalli, N.M.G.; Honjo, R.S.; Kim, C.; Teixeira, M.C.T.V. Cognitive and behavioral profile of Williams syndrome toddlers. CoDAS 2018, 30, e20170188. [Google Scholar] [CrossRef]
- Neo, W.S.; Tonnsen, B.L. Brief Report: Challenging Behaviors in Toddlers and Preschoolers with Angelman, Prader–Willi, and Williams Syndromes. J. Autism Dev. Disord. 2019, 49, 1717–1726. [Google Scholar] [CrossRef] [PubMed]
- Murthy, R.S. Mass violence and mental health: Recent epidemiological findings. Int. Rev. Psychiatry 2007, 19, 183–192. [Google Scholar] [CrossRef]
- Yule, W.; Udwin, O.; Murdoch, K. The ‘Jupiter’ sinking: Effects on children’s fears, depression and anxiety. J. Child Psychol. Psychiatry Allied Discip. 1990, 31, 1051–1061. [Google Scholar] [CrossRef]
- Eley, T.C.; Stevenson, J. Specific life events and chronic experiences differentially associated with depression and anxiety in young twins. J. Abnorm. Child. PSychol 2000, 28, 383–394. [Google Scholar] [CrossRef]
- Eley, T.C.; Lau, J.Y. Genetics and the family environment. In Psychopathology and the Family; Hudson, J., Rapee, R., Eds.; Elsevier: Sydney, Australia, 2005; pp. 3–19. [Google Scholar]
- Broeren, S.; Muris, P.; Diamantopoulou, S.; Baker, J.R. The course of childhood anxiety symptoms: Developmental trajectories and child-related factors in normal children. J. Abnorm. Child. Psychol. 2013, 41, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Mian, N.D.; Wainwright, L.; Briggs-Gowan, M.J.; Carter, A.S. An ecological risk model for early childhood anxiety: The importance of early child symptoms and temperament. J. Abnorm. Child. Psychol. 2011, 39, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Chess, S. Temperament and Development; Brunner/Mazel: New York, NY, USA, 1977. [Google Scholar]
- Hudson, J.L.; Rapee, R.M. From Anxious Temperament to Disorder: An Etiological Model. In Generalized Anxiety Disorder: Advances in Research and Practice; Heimberg, R.G., Turk, C.L., Mennin, D.S., Eds.; The Guilford Press: Washington, DC, USA, 2004; pp. 51–74. [Google Scholar]
- Khreim, I.; Mikkelsen, E. Anxiety disorders in adults with mental retardation. Psychiatr. Ann. 1997, 27, 175–181. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- World Health Organisation. ICD-10 Classifications of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines; World Health Organisation: Geneva, Switzerland, 1992. [Google Scholar]
- March, J.S.; Parker, J.D.; Sullivan, K.; Stallings, P.; Conners, C.K. The Multidimensional Anxiety Scale for Children (MASC): Factor structure, reliability, and validity. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 554–565. [Google Scholar] [CrossRef]
- Muris, P.; de Jong, P.J.; Engelen, S. Relationships between neuroticism, attentional control, and anxiety disorders symptoms in non-clinical children. Pers. Individ. Dif. 2004, 37, 789–797. [Google Scholar] [CrossRef]
- Parker, G.; Hadzi-Pavlovic, D. Is the female preponderance in major depression secondary to a gender difference in specific anxiety disorders? Psychol. Med. 2004, 34, 461–470. [Google Scholar] [CrossRef][Green Version]
- Roza, S.J.; Hofstra, M.B.; Van Der Ende, J.; Verhulst, F.C. Stable prediction of mood and anxiety disorders based on behavioral and emotional problems in childhood: A 14-year follow-up during childhood, adolescence, and young adulthood. Am. J. Psychiatry 2003, 160, 2116–2121. [Google Scholar] [CrossRef]
- King, N.J.; Ollier, K.; Iacuone, R.; Schuster, S.; Bays, K.; Gullone, E.; Ollendick, T.H. Fears of children and adolescents: A cross-sectional Australian study using the revised-fear survey schedule for children. J. Child Psychol. Psychiatry Allied Discip. 1989, 30, 775–784. [Google Scholar] [CrossRef]
- Einfeld, S.L.; Tonge, B.J. Population prevalence of psychopathology in children and adolescents with intellectual disability: II epidemiological findings. J. Intellect. Disabil. Res. 1996, 40, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Ng, R.; Järvinen, A.; Bellugi, U. Characterizing associations and dissociations between anxiety, social, and cognitive phenotypes of Williams syndrome. Res. Dev. Disabil. 2014, 35, 2403–2415. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Porter, M.A.; Dodd, H.; Cairns, D. Psychopathological and behavior impairments in Williams-Beuren syndrome: The influence of gender, chronological age, and cognition. Child. Neuropsych 2009, 15, 359–374. [Google Scholar] [CrossRef]
- Woodcock, R.W.; Johnson, M.B. Woodcock-Johnson Psycho-Educational Battery—Revised; Riverside: Itasca, IL, USA, 1990. [Google Scholar]
- Osório, A.A.; Rossi, N.F.; Gonçalves, Ó.F.; Sampaio, A.; Giacheti, C.M. Psychopathology and behavior problems in children and adolescents with Williams syndrome: Distinctive relationships with cognition. Child. Neuropsych 2017, 23, 631–641. [Google Scholar] [CrossRef]
- Achenbach, T.M.; Rescorla, L.A. Manual for the ASEBA School-Age Forms & Profiles; University of Vermont, Research Center for Children, Youth, & Families: Burlington, VT, USA, 2001. [Google Scholar]
- Rzepecka, H.; McKenzie, K.; McClure, I.; Murphy, S. Sleep, anxiety and challenging behaviour in children with intellectual disability and/or Autism Spectrum Disorder. Res. Dev. Disabil. 2011, 32, 2758–2766. [Google Scholar] [CrossRef] [PubMed]
- Silverman, W.K.; Albano, A.M. Anxiety Disorders Interview Schedule for DSM-IV: Child Version; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- Kaufman, J.; Birmaher, B.; Brent, D.; Rao, U.; Flynn, C.; Moreci, P.; Williamson, D.; Ryan, N. Schedule for affective disorders and schizophrenia for school-aged children-present and lifetime version (K-SADS-PL): Initial reliability and validity data. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 980–988. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV, 4th ed.; Text rev.; American Psychiatric Publishing: Washington, DC, USA, 1994. [Google Scholar]
- Spence, S.H. Structure of anxiety symptoms among children: A confirmatory factor-analytic study. J. Abnorm. Psychol. 1997, 106, 280–297. [Google Scholar] [CrossRef]
- Papaeliou, C.; Polemikos, N.; Fryssira, E.; Kodakos, A.; Kaila, M.; Yiota, X.; Benaveli, C.; Michaelides, V.; Stroggilos, M.; Vrettopoulou, M. Behavioural profile and maternal stress in Greek young children with Williams syndrome. Child Care Health Dev. 2012, 38, 844–853. [Google Scholar] [CrossRef]
- Achenbach, T.M.; Rescorla, L.A. Manual for the ASEBA Preschool Forms & Profiles; University of Vermont, Research Center for Children, Youth, & Families: Burlington, VT, USA, 2000. [Google Scholar]
- Ferdinand, R.F. Validity of the CBCL/YSR DSM-IV scales anxiety problems and affective problems. J. Anxiety Disord. 2008, 22, 126–134. [Google Scholar] [CrossRef]
- Korenberg, J.R.; Chen, X.N.; Hirota, H.; Lai, Z.; Bellugi, U.; Burian, D.; Roe, B.; Matsuoka, R. VI. Genome structure and cognitive map of Williams syndrome. J. Cogn. Neurosci. 2000, 12, 89–107. [Google Scholar] [CrossRef] [PubMed]
- Tassabehji, M.; Metcalfe, K.; Fergusson, W.D.; Carette, M.J.; Dore, J.K.; Donnai, D.; Reed, A.P.; Pröschel, C.; Gutowski, N.J.; Mao, X.; et al. LIM–kinase deleted in Williams syndrome. Nat. Genet. 1996, 13, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Mullen, E.M. Mullen Scales of Early Learning; American Guidance Service: Circle Pines, MN, USA, 1995. [Google Scholar]
- Mervis, C.B.; Robinson, B.F.; Bertrand, J.; Morris, C.A.; Klein-Tasman, B.P.; Armstrong, S.C. The Williams syndrome cognitive profile. Brain Cogn. 2000, 44, 604–628. [Google Scholar] [CrossRef] [PubMed]
- Miezah, D.; Porter, M.; Batchelor, J.; Boulton, K.; Veloso, G.C. Cognitive abilities in Williams syndrome. Res. Dev. Disabil. 2020, 104, 103701. [Google Scholar] [CrossRef]
- Miezah, D.; Porter, M.; Rossi, A.; Kazzi, C.; Batchelor, J.; Reeve, J. Cognitive profile of young children with Williams syndrome. J. Intellect. Disabil. Res. 2021, 65, 784–794. [Google Scholar] [CrossRef] [PubMed]
- Martens, M.A.; Wilson, S.J.; Reutens, D.C. Research review: Williams syndrome: A critical review of the cognitive, behavioral, and neuroanatomical phenotype. J. Child. Psychol. Psychiatry 2008, 49, 576–608. [Google Scholar] [CrossRef]
- Porter, M.A.; Coltheart, M. Cognitive heterogeneity in Williams syndrome. Dev. Neuropsychol. 2005, 27, 275–306. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/2033.0.55.001~2016~Main%20Features~IRSAD%20Interactive%20Map~16 (accessed on 20 May 2022).
- Spence, S.H.; Rapee, R.; McDonald, C.; Ingram, M. The structure of anxiety symptoms among preschoolers. Behav. Res. Ther. 2001, 39, 1293–1316. [Google Scholar] [CrossRef]
- Galán-Luque, T.; Serrano-Ortiz, M.; Orgilés, M. Factor Structure and Psychometric Properties of the Spence Children’s Anxiety Scale: A 25-Year Systematic Review. Child. Psychiatry Hum. Dev. 2025, 56, 492–506. [Google Scholar] [CrossRef] [PubMed]
- Nauta, M.H.; Scholing, A.; Rapee, R.M.; Abbot, M.; Spence, S.H.; Waters, A. A parent-report measure of children’s anxiety: Psychometric properties and comparison with child-report in a clinic and normal sample. Behav. Res. Ther. 2004, 42, 813–839. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.; Riby, D.M.; Janes, E.; Connolly, B.; McConachie, H. Anxiety and repetitive behaviours in Autism Spectrum Disorders and Williams syndrome: A cross-syndrome comparison. J. Autism Dev. Disord. 2012, 42, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Spence, S.H.; Rapee, R. Preschool Anxiety Scale (Parent Report); University of Queensland: Brisbane, Australia, 1999. [Google Scholar]
- Kazzi, C.; Porter, M.; Zhong, Q.; Veloso, G.; Reeve, J. The relationship between anxiety and executive functioning in children with Williams syndrome. Glob. J. Intellect. Dev. Disabil. 2021, 8, 555745. [Google Scholar] [CrossRef]
- Spence, S.H. A measure of anxiety symptoms among children. Behav. Res. Ther. 1998, 36, 545–566. [Google Scholar] [CrossRef] [PubMed]
- Rogers, S.J.; Estes, A.; Lord, C.; Vismara, L.; Winter, J.; Fitzpartick, A.; Guo, M.; Dawson, G. Effects of brief Early Start Denver Model (ESDM)-based parent intervention on toddlers at risk for autism spectrum disorders: A random controlled trial. J. Am. Acad. Child Adolesc. Psychiatry 2012, 51, 1052–1065. [Google Scholar] [CrossRef]
- Campbell, L.E.; Daley, E.; Toal, F.; Stevens, A.; Azuma, R.; Karmiloff-Smith, A.; Murphy, D.G.M.; Murphy, M.K.C. Brain structural differences associated with the behavioural phenotype in children with Williams syndrome. Brain Res. 2009, 1258, 96–107. [Google Scholar] [CrossRef]
- Hocking, D.R.; Reeve, J.; Porter, M.A. Characterising the profile of everyday executive functioning and relation to IQ in adults with Williams syndrome: Is the BRIEF adult version a valid rating scale? PLoS ONE 2015, 10, e0137628. [Google Scholar] [CrossRef]
- Rhodes, S.M.; Riby, D.M.; Park, J.; Fraser, E.; Campbell, L.E. Executive neuropsychological functioning in individuals with Williams syndrome. Neuropsychologia 2010, 48, 1216–1226. [Google Scholar] [CrossRef]
- Rothman, K.J. No adjustments are needed for multiple comparisons. Epidemiology 1990, 1, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Erlbaum: Hillsdale, NJ, USA, 1988. [Google Scholar]
- The Spence Children’s Anxiety Scale. SCAS. Available online: https://www.scaswebsite.com/ (accessed on 20 May 2022).
- McAdams, P.D. Intimacy, and the Life Story: Personological Inquiries into Identity; Guilford Press: New York, NY, USA, 1985. [Google Scholar]
- Pillemer, D.B. Momentous events and the life story. Rev. Gen. Psychol. 2001, 5, 123–134. [Google Scholar] [CrossRef]
- Baxter, A.J.; Scott, K.M.; Vos, T.; Whiteford, H.A. Global prevalence of anxiety disorders: A systematic review and meta-regression. Psychol. Med. 2013, 43, 897–910. [Google Scholar] [CrossRef]
- Somers, J.M.; Goldner, E.M.; Waraich, P.; Hsu, L. Prevalence and incidence studies of anxiety disorders: A systematic review of the literature. Can. J. Psychiatry 2006, 51, 100–113. [Google Scholar] [CrossRef]
- Dennis, M.; Landry, S.H.; Barnes, M.; Fletcher, J.M. A model of neurocognitive function in spina bifida over the life span. JINS 2006, 12, 258–296. [Google Scholar] [CrossRef] [PubMed]
- Luby, J.L. Treatment of anxiety and depression in the preschool period. J. Am. Acad. Child. Adolesc. 2013, 52, 346–358. [Google Scholar] [CrossRef]
- Connolly, S.D.; Bernstein, G.A. Practice parameter for the assessment and treatment of children and adolescents with anxiety disorders. J. Am. Acad. Child. Adolesc. 2007, 46, 267–283. [Google Scholar] [CrossRef] [PubMed]
- Finelli, J.; Gleason, M.M. Psychopharmacologic considerations in early childhood. In Clinical Guide to Psychiatric Assessment of Infants and Young Children; Frankel, K., Harrison, J., Njoroge, W., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 285–326. [Google Scholar]
- Zaim, N.; Harrison, J. Pre-school mental health disorders: A review. Int. Rev. Psychiatry 2020, 32, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Fynn, G.; Porter, M.; Borchard, T.; Kazzi, C.; Zhong, Q.; Campbell, L. The effectiveness of cognitive behavioural therapy for individuals with an intellectual disability and anxiety: A systematic review. JIDR 2023, 67, 816–841. [Google Scholar] [CrossRef]
- Wittchen, H.U.; Hoyer, J. Generalized anxiety disorder: Nature and course. J. Clin. Psychiatry 2001, 62, 15–21. [Google Scholar]
- Danesh, A.A.; Howery, S.; Aazh, H.; Kaf, W.; Eshraghi, A.A. Hyperacusis in Autism Spectrum Disorders. Audiol. Res. 2021, 11, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.M.; Dadds, M.R.; Rapee, R.M. Family treatment of childhood anxiety: A controlled trial. J. Consult. Clin. Psychol. 1996, 64, 333–342. [Google Scholar] [CrossRef]
- Toscano, R.; Baillie, A.J.; Lyneham, H.J.; Kelly, A.; Kidd, T.; Hudson, J.L. Assessment of anxiety in children and adolescents: A comparative study on the validity and reliability of the Spence Children’s Anxiety Scale in children and adolescents with anxiety and Autism Spectrum Disorder. J. Affect. Disord. 2020, 260, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Finlay, W.M.; Lyons, E. Acquiescence in interviews with people who have mental retardation. Ment. Retard. 2002, 40, 14–29. [Google Scholar] [CrossRef]
- Hartley, S.L.; MacLean, W.E., Jr. A review of the reliability and validity of Likert-type scales for people with intellectual disability. JIDR 2006, 50, 813–827. [Google Scholar] [CrossRef]
- Hettema, J.M.; Neale, M.C.; Kendler, K.S. A review and meta-analysis of the genetic epidemiology of anxiety disorders. Am. J. Psychiatry 2001, 158, 1568–1578. [Google Scholar] [CrossRef] [PubMed]
- Rapee, R.M.; Spence, S.H. The etiology of social phobia: Empirical evidence and an initial model. Clin. Psych. Rev. 2004, 24, 737–767. [Google Scholar] [CrossRef]
- Dai, L.; Carter, C.S.; Ying, J.; Bellugi, U.; Pournajafi-Nazarloo, H.; Korenberg, J.R. Oxytocin and vasopressin are dysregulated in Williams Syndrome, a genetic disorder affecting social behavior. PLoS ONE 2012, 7, e38513. [Google Scholar] [CrossRef]
- Griffiths, H.; Fazel, M.S. Early intervention crucial in anxiety disorders in children. Practitioner 2016, 26, 17–20. [Google Scholar]
- Huston, J.C.; Thom, R.P.; Ravichandran, C.T.; Mullett, J.E.; Moran, C.; Waxler, J.L.; Prober, B.R.; McDougle, C.J. Repetitive Thoughts and Repetitive Behaviors in Williams Syndrome. J. Autism Dev. Disord. 2021, 52, 852–862. [Google Scholar] [CrossRef]


| Time Point | ||||
|---|---|---|---|---|
| Time 1 | Time 2 | |||
| Mean (SD) | Range | Mean (SD) | Range | |
| CA (years) a | 4.08 (1.012) | 2.24–5.97 | 7.53 (1.87) | 3.98–9.90 |
| Gender distribution | 7 M, 12 F, 0 NB b | |||
| Family SES c | 3.59 (1.54) | 1–5 | 3.59 (1.54) | 1–5 |
| Primary Caregiver Age (Years) | 35.33 (3.46) | 31–41 | 38.13 (3.87) | 33–46 |
| Global DQ d | 54.64 (11.71) | 28.62–68.25 | - | - |
| Verbal DQ d | 56.73 (16.50) | 24.81–93.00 | - | - |
| Nonverbal DQ d | 57.72 (13.83) | 31.48–77.15 | - | - |
| Current Interventions | ||||
| % Speech Therapy | 100.00 | 100.00 | ||
| % Occupational Therapy | 68.42 | 93.33 | ||
| % Physiotherapy | 84.21 | 46.67 | ||
| % Psychological/Behaviour Therapy | 0.00 | 15.79 d | ||
| Anxiety/Specific Phobia Subscales And Total Scores | Time Point | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Time 1 | Time 2 a | Difference (T2-T1) | |||||||
| M (SD) | Range | % in Clinical Range e | M (SD) | Range | % in Clinical Range e | T (18) | p | d | |
| Total Anxiety | −0.38 (0.59) | −1.17–0.44 | 0% | 0.79 (1.31) | −1.21–4.46 | 31.58% | 4.16 | 0.001 ** | 0.95 |
| GAD b | 0.37 (1.18) | −0.94–2.64 | 36.84% | 1.22 (1.66) | −1.11–4.68 | 47.37% | 2.52 | 0.021 * | 0.58 |
| Social Anxiety | −1.00 (0.35) | −1.19–0.12 | 0% | −0.39 (0.67) | −1.50–0.69 | 0% | 21.00 d | 0.003 ** | 0.48 g |
| OCD c | −0.32 (0.72) | −0.67–2.24 | 5.26% | 0.44 (1.16) | −0.71–3.69 | 31.58% | 20.00 d | 0.003 ** | 0.49 g |
| Physical Injury Fears | 0.12 (0.89) | −1.42–1.86 | 15.79% | 1.19 (1.32) | −0.79–3.96 | 52.63% | 3.58 | 0.002 ** | 0.82 |
| Separation Anxiety | −0.11 (0.78) | −1.03–1.52 | 10.53% | 0.90 (1.20) | −1.03–4.24 | 42.11% | 3.76 | 0.001 ** | 0.86 |
| Specific Phobia Total f | 63.16% | 84.21% | |||||||
| Animal | 5.26% | 26.32% | |||||||
| Natural Environment | 5.26% | 21.05% | |||||||
| Blood-Injectional-Injury | 10.53% | 26.32% | |||||||
| Situational | 26.53% | 47.37% | |||||||
| Other | 15.79% | 10.53% | |||||||
| Loud Noises | 36.84% | 52.63% | |||||||
| Clinically Elevated Anxiety Symptoms a | Significant Life Event b | Additional Psychological Symptoms or Diagnosis c | Psychological Intervention | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ID | Sex | Time 1 | Time 2 | Before Time 1 | Between Time 1 and 2 | Before Time 1 | Between Time 1 and 2 | Before Time 1 | Between Time 1 and 2 | Hyperacusis | Cardiovascular Abnormalities |
| 1 | M | None | GAD | Multiple hospital visits and surgeries | Started school | Problems concentrating; restless; fidgety | Problems concentrating; restless; fidgety | None | None | Yes | Yes |
| 2 | F | GAD; SP-blood-injury-injection; SP-Situational; SP-loud noises | GAD, OCD, Physical Injury Fears, Separation Anxiety; SP-Natural Environment; SP-blood-injury-injection; SP-Situational; SP-loud noises | Started school | New sibling | Problems concentrating; will not sleep alone | Problems concentrating; restless; fidgety | None | None | Yes | None |
| 3 | F | GAD; Separation Anxiety; SP-blood-injury-injection; SP-loud noises | OCD, SP-loud noises | Started preschool | Started school | Problems concentrating; restless; fidgety Repetitive thoughts; worries; trouble sleeping when separated from parents at night; hard to settle | Problems concentrating; restless; fidgety Repetitive thoughts; worries; trouble sleeping when separated from parents at night; hard to settle | None | None | Yes | Yes |
| 4 | F | None | SP-blood-injury-injection; SP-Situational; SP-Other; SP-loud noises | None | None | None | None | None | Therapy for anxiety and aggression | Yes | Yes |
| 5 | F | GAD; Physical; SP-loud noises | Physical Injury Fears; SP-Animals; SP-Natural Environment; SP-blood-injury-injection; SP-Other; SP-Loud Noises | Started preschool | Started school | Problems concentrating; hard to settle; depressed mood | Problems concentrating | None | None | Yes | Yes |
| 6 | F | GAD; SP-Situational | GAD; Physical Injury Fears; Separation Anxiety; SP-Animal; SP-Situational | Started school | None | Problems concentrating; restless; fidgety; hard to settle | ADHDdiagnosis; depressed mood | None | None | Yes | Yes |
| 7 | F | SP-Situational | Physical Injury Fears; SP-Situational | Started day-care | Started school; new Sibling | None | Problems concentrating; restless; fidgety | None | None | Yes | Yes |
| 8 | M | SP-Other | SP-Animals; SP-Natural Environment; SP-Situational | None | Started school; surgery | Problems concentrating; restless; fidgety; hard to settle | Problems concentrating; restless; fidgety; hard to settle | None | Therapy for anxiety | Yes | Yes |
| 9 | F | OCD | GAD | Started preschool | Started school | Restless; fidgety; hard to settle | None | None | Yes | Yes | |
| 10 | F | None | Physical Injury Fears; Separation Anxiety | Started preschool | Started school | Restless; fidgety; hard to settle | None | None | Yes | Yes | |
| 11 | M | GAD | GAD; OCD; Physical Injury Fears; Separation Anxiety; SP-Animals; SP-Loud Noises | Started preschool | None | Problems concentrating; restless; fidgety; hard to settle | Problems concentrating; restless; fidgety; hard to settle | None | None | Yes | Yes |
| 12 | M | GAD; Physical Injury Fears; SP-Natural Environment; SP-Other, | GAD; Separation Anxiety; SP-Other, SP-Situational; SP-Loud Noises | Started day-care | Parent Separation; started school | None | None | None | Therapy for mood | Yes | Yes |
| 13 | F | SP-Loud Noises | Separation Anxiety; SP- blood-injury-injection; SP-Loud Noises | Started school | None | Problems concentrating; restless; fidgety | Mild problems with concentration | None | None | Yes | None |
| 14 | F | SP-Situational; SP-Other | SP-Situational | None | Started Day-care | Problems concentrating | Problems concentrating; restless; fidgety | None | None | Yes | None |
| 15 | M | Physical Injury Fears; SP-Animal; SP-Loud Noises | Physical Injury Fears; Separation Anxiety; SP-Animal; SP-Natural Environment; SP-Situational; SP-Loud Noises | Started day-care | Started school | Hard to settle | None | None | None | Yes | Yes |
| 16 | F | GAD; Separation Anxiety; SP-Situational; SP-Loud Noises | GAD; OCD; Physical Injury Fears; SP-Situational; SP-Loud Noises | Separation of parents, started preschool | Started school | Problems concentrating; restless; fidgety; hyperactivity, hard to settle | Problems concentrating; restless; fidgety; hyperactivity, | None | None | Yes | Yes |
| 17 | M | SP-Loud Noises | GAD; OCD; Physical Injury Fears; Separation Anxiety; SP-Loud Noises | Surgery, started school | None | Problems concentrating; restless; fidgety | Problems concentrating; restless; fidgety | None | None | Yes | Yes |
| 18 | M | None | GAD; OCD; Physical Injury Fears; Separation; SP-Situational | Multiple surgeries, started school | None | Problems concentrating; restless; fidgety | Problems concentrating; restless; fidgety | None | None | Yes | Yes |
| 19 | F | None | Physical Injury Fears; Separation Anxiety; SP-Situational | Multiple surgeries, started preschool | Started school | Hard to settle | None | None | None | No | Yes |
| Anxiety Subtypes and Total Score | Chronological Age | Sex | DQ | ||
|---|---|---|---|---|---|
| Global | Verbal | Nonverbal | |||
| Total Score a | 0.13 | 0.02 | −0.16 | −0.34 | −0.20 |
| GAD b | 0.02 | −0.41 | −0.13 | −0.34 | −0.14 |
| Social Anxiety | 0.49 * | 0.22 | −0.06 | −0.17 | −0.20 |
| OCD c | −0.02 | −0.24 | 0.30 | 0.11 | 0.21 |
| Physical Injury Fears | 0.31 | 0.61 ** | −0.18 | −0.14 | −0.07 |
| Separation Anxiety | 0.36 | −0.19 | −0.11 | −0.04 | 0.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reeve, J.L.; Porter, M.A. Anxiety in Young Children with Williams Syndrome: A Longitudinal Study. Children 2025, 12, 1098. https://doi.org/10.3390/children12081098
Reeve JL, Porter MA. Anxiety in Young Children with Williams Syndrome: A Longitudinal Study. Children. 2025; 12(8):1098. https://doi.org/10.3390/children12081098
Chicago/Turabian StyleReeve, Jessica L., and Melanie A. Porter. 2025. "Anxiety in Young Children with Williams Syndrome: A Longitudinal Study" Children 12, no. 8: 1098. https://doi.org/10.3390/children12081098
APA StyleReeve, J. L., & Porter, M. A. (2025). Anxiety in Young Children with Williams Syndrome: A Longitudinal Study. Children, 12(8), 1098. https://doi.org/10.3390/children12081098






