An Update on Pharmacologic Management of Neonatal Hypotension: When, Why, and Which Medication
Abstract
1. Introduction
2. Method of Literature Search
3. Definitions of Arterial Hypotension in Neonates
4. Pathophysiology of Hemodynamic Instability in Neonates
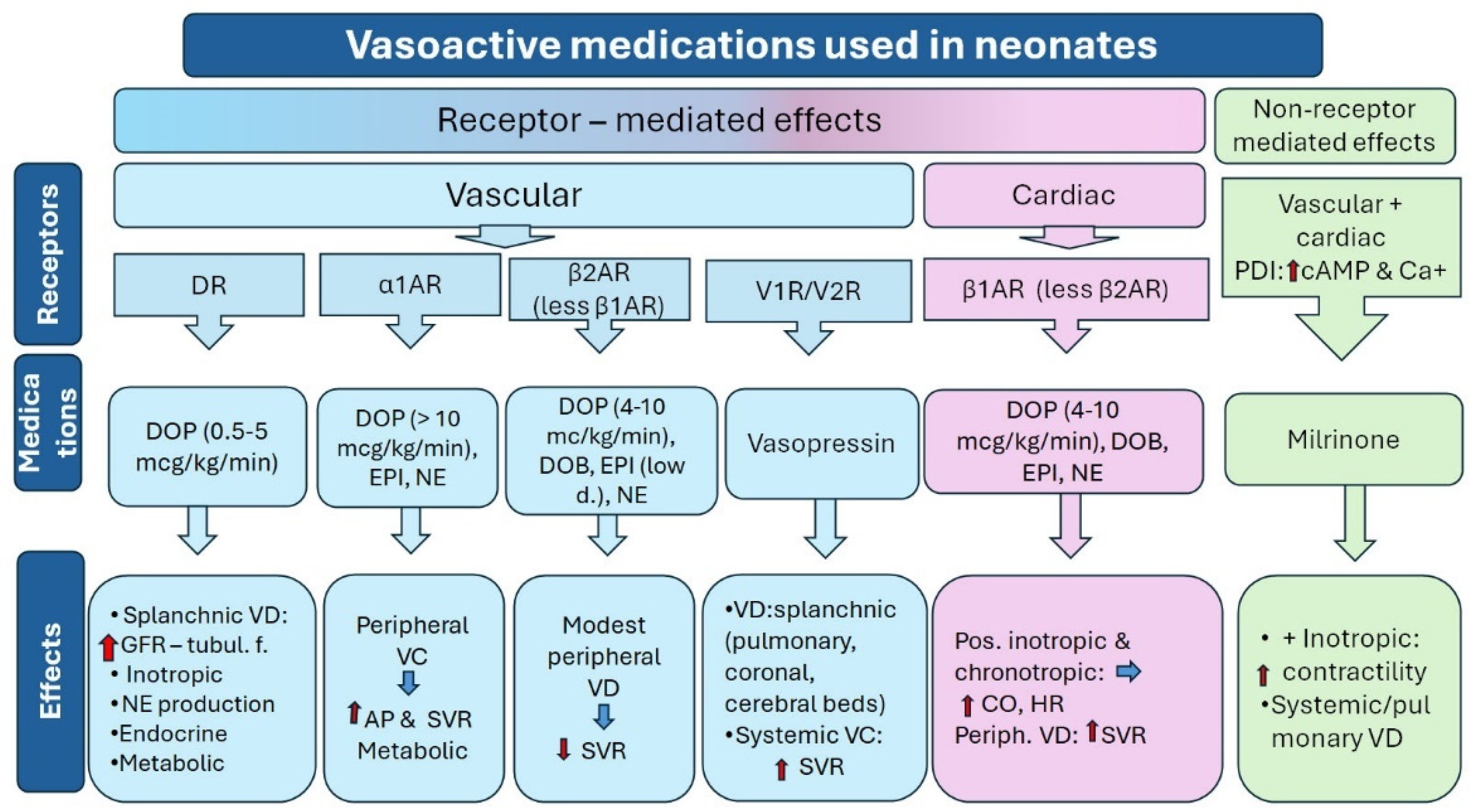
5. Anti-Hypotensive Medications Used in Neonates
5.1. Dopamine
5.2. Dobutamine
5.3. Epinephrine
5.4. Norepinephrine
5.5. Milrinone
Vasopressin and Terlipressin
5.6. Levosimendan
5.7. Corticosteroids
6. Long-Term Outcomes of Hypotensive Neonates Treated with Vasoactive Medications
7. Indications for Anti-Hypotensive Medications in Clinical Practice
8. Comparisons between Anti-Hypotensive Medications
9. Discussion
10. Future Perspectives
11. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kluckow, M. Low Systemic Blood Flow and Pathophysiology of the Preterm Transitional Circulation. Early Hum. Dev. 2005, 81, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Dempsey, E.M.; Al Hazzani, F.; Barrington, K.J. Permissive Hypotension in the Extremely Low Birthweight Infant with Signs of Good Perfusion. Arch. Dis. Child. Fetal Neonatal. Ed. 2009, 94, F241–F244. [Google Scholar] [CrossRef] [PubMed]
- Gill, A.W. Postnatal Cardiovascular Adaptation. Arch. Dis. Child. Fetal Neonatal. Ed. 2019, 104, F220–F224. [Google Scholar] [CrossRef] [PubMed]
- Burns, M.L.; Stensvold, H.J.; Risnes, K.; Guthe, H.J.; Astrup, H.; Nordhov, S.M.; Selberg, T.R.; Rønnestad, A.; Lang, A.M. Norwegian Neonatal Network Inotropic Therapy in Newborns, A Population-Based National Registry Study. Pediatr. Crit. Care Med. 2016, 17, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Aldana-Aguirre, J.C.; Deshpande, P.; Jain, A.; Weisz, D.E. Physiology of Low Blood Pressure During the First Day After Birth Among Extremely Preterm Neonates. J. Pediatr. 2021, 236, 40–46.e3. [Google Scholar] [CrossRef] [PubMed]
- Ancel, P.-Y.; Goffinet, F.; EPIPAGE-2 Writing Group; Kuhn, P.; Langer, B.; Matis, J.; Hernandorena, X.; Chabanier, P.; Joly-Pedespan, L.; Lecomte, B.; et al. Survival and Morbidity of Preterm Children Born at 22 through 34 Weeks’ Gestation in France in 2011: Results of the EPIPAGE-2 Cohort Study. JAMA Pediatr. 2015, 169, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Lalan, S.P.; Warady, B.A. Discrepancies in the Normative Neonatal Blood Pressure Reference Ranges. Blood Press. Monit. 2015, 20, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Kent, A.L.; Meskell, S.; Falk, M.C.; Shadbolt, B. Normative Blood Pressure Data in Non-Ventilated Premature Neonates from 28–36 Weeks Gestation. Pediatr. Nephrol. 2009, 24, 141–146. [Google Scholar] [CrossRef]
- Dempsey, E.; Rabe, H. The Use of Cardiotonic Drugs in Neonates. Clin. Perinatol. 2019, 46, 273–290. [Google Scholar] [CrossRef]
- Lasky, T.; Greenspan, J.; Ernst, F.R.; Gonzalez, L. Dopamine and Dobutamine Use in Preterm or Low Birth Weight Neonates in the Premier 2008 Database. Clin. Ther. 2011, 33, 2082–2088. [Google Scholar] [CrossRef]
- Batton, B.; Batton, D.; Riggs, T. Blood Pressure during the First 7 Days in Premature Infants Born at Postmenstrual Age 23 to 25 Weeks. Am. J. Perinatol. 2007, 24, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Alderliesten, T.; Lemmers, P.M.A.; van Haastert, I.C.; de Vries, L.S.; Bonestroo, H.J.C.; Baerts, W.; van Bel, F. Hypotension in Preterm Neonates: Low Blood Pressure Alone Does Not Affect Neurodevelopmental Outcome. J. Pediatr. 2014, 164, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Batton, B.; Li, L.; Newman, N.S.; Das, A.; Watterberg, K.L.; Yoder, B.A.; Faix, R.G.; Laughon, M.M.; Stoll, B.J.; Higgins, R.D.; et al. Early Blood Pressure, Antihypotensive Therapy and Outcomes at 18–22|Months’ Corrected Age in Extremely Preterm Infants. Arch. Dis. Child. Fetal Neonatal. Ed. 2016, 101, F201–F206. [Google Scholar] [CrossRef] [PubMed]
- Bravo, M.C.; López-Ortego, P.; Sánchez, L.; Díez, J.; Cabañas, F.; Pellicer, A. Randomised Trial of Dobutamine versus Placebo for Low Superior Vena Cava Flow in Preterm Infants: Long-Term Neurodevelopmental Outcome. J. Paediatr. Child Health 2021, 57, 872–876. [Google Scholar] [CrossRef]
- Doucette, S.M.; Kelly, E.N.; Church, P.T.; Lee, S.; Shah, V.; Canadian Neonatal Network (CNN) Investigators and CNFUN Investigators and Steering Committee. Association of Inotrope Use with Neurodevelopmental Outcomes in Infants <29 Weeks Gestation: A Retrospective Cohort Study. J. Matern. Fetal Neonatal. Med. 2022, 35, 6044–6052. [Google Scholar] [CrossRef] [PubMed]
- Subhedar, N.V.; Shaw, N.J. Dopamine versus Dobutamine for Hypotensive Preterm Infants. Cochrane Database Syst. Rev. 2003, CD001242. [Google Scholar] [CrossRef] [PubMed]
- Low, J.A.; Froese, A.B.; Galbraith, R.S.; Smith, J.T.; Sauerbrei, E.E.; Derrick, E.J. The Association between Preterm Newborn Hypotension and Hypoxemia and Outcome during the First Year. Acta Paediatr. 1993, 82, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Logan, J.W.; O’Shea, T.M.; Allred, E.N.; Laughon, M.M.; Bose, C.L.; Dammann, O.; Batton, D.G.; Engelke, S.C.; Leviton, A.; ELGAN Study Investigators. Early Postnatal Hypotension and Developmental Delay at 24 Months of Age among Extremely Low Gestational Age Newborns. Arch. Dis. Child. Fetal Neonatal. Ed. 2011, 96, F321–F328. [Google Scholar] [CrossRef] [PubMed]
- Kuint, J.; Barak, M.; Morag, I.; Maayan-Metzger, A. Early Treated Hypotension and Outcome in Very Low Birth Weight Infants. Neonatology 2009, 95, 311–316. [Google Scholar] [CrossRef]
- McKim, K.J.; Lucafo, S.; Bhombal, S.; Bain, L.; Chock, V.Y. Blood Pressure Goals: Is Cerebral Saturation the New Mean Arterial Pressure? Am. J. Perinatol. 2022, 41, 498–504. [Google Scholar] [CrossRef]
- Dempsey, E.M. What Should We Do about Low Blood Pressure in Preterm Infants. Neonatology 2017, 111, 402–407. [Google Scholar] [CrossRef]
- Cooke, R.W.I. Report of Working Group of the British Association of Perinatal Medicine and Neonatal Nurses Association on Categories of Babies Requiring Neonatal Care. Arch. Dis. Child. 1992, 67, 868–869. [Google Scholar] [CrossRef][Green Version]
- Faust, K.; Härtel, C.; Preuß, M.; Rabe, H.; Roll, C.; Emeis, M.; Wieg, C.; Szabo, M.; Herting, E.; Göpel, W.; et al. Short-Term Outcome of Very-Low-Birthweight Infants with Arterial Hypotension in the First 24 h of Life. Arch. Dis. Child. Fetal Neonatal. Ed. 2015, 100, F388–F392. [Google Scholar] [CrossRef] [PubMed]
- de Boode, W.-P. Clinical Monitoring of Systemic Hemodynamics in Critically Ill Newborns. Early Hum. Dev. 2010, 86, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, D.; Baik-Schneditz, N.; Schwaberger, B.; Urlesberger, B.; Pichler, G. Insights into Neonatal Cerebral Autoregulation by Blood Pressure Monitoring and Cerebral Tissue Oxygenation: A Qualitative Systematic Review. Children 2023, 10, 1304. [Google Scholar] [CrossRef] [PubMed]
- Askie, L.M.; Darlow, B.A.; Davis, P.G.; Finer, N.; Stenson, B.; Vento, M.; Whyte, R. Effects of Targeting Lower versus Higher Arterial Oxygen Saturations on Death or Disability in Preterm Infants. Cochrane Database Syst. Rev. 2017, 4, CD011190. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.L.; Pellicer, A.; Hyttel-Sørensen, S.; Ergenekon, E.; Szczapa, T.; Hagmann, C.; Naulaers, G.; Mintzer, J.; Fumagalli, M.; Dimitriou, G.; et al. Cerebral Oximetry Monitoring in Extremely Preterm Infants. N. Engl. J. Med. 2023, 388, 1501–1511. [Google Scholar] [CrossRef] [PubMed]
- Dilli, D.; Soylu, H.; Tekin, N. Neonatal Hemodynamics and Management of Hypotension in Newborns. Turk. Pediatri. Ars. 2018, 53, S65–S75. [Google Scholar] [CrossRef] [PubMed]
- Hooper, S.B.; Te Pas, A.B.; Lang, J.; van Vonderen, J.J.; Roehr, C.C.; Kluckow, M.; Gill, A.W.; Wallace, E.M.; Polglase, G.R. Cardiovascular Transition at Birth: A Physiological Sequence. Pediatr. Res. 2015, 77, 608–614. [Google Scholar] [CrossRef]
- Giesinger, R.E.; McNamara, P.J. Hemodynamic Instability in the Critically Ill Neonate: An Approach to Cardiovascular Support Based on Disease Pathophysiology. Semin. Perinatol. 2016, 40, 174–188. [Google Scholar] [CrossRef]
- Xu, A.; Hawkins, C.; Narayanan, N. Ontogeny of Sarcoplasmic Reticulum Protein Phosphorylation by Ca2+--Calmodulin-Dependent Protein Kinase. J. Mol. Cell. Cardiol. 1997, 29, 405–418. [Google Scholar] [CrossRef]
- Singh, Y.; Katheria, A.C.; Vora, F. Advances in Diagnosis and Management of Hemodynamic Instability in Neonatal Shock. Front. Pediatr. 2018, 6, 2. [Google Scholar] [CrossRef]
- Cox, D.J.; Groves, A.M. Inotropes in Preterm Infants--Evidence for and Against. Acta Paediatr. 2012, 101, 17–23. [Google Scholar] [CrossRef]
- Gao, Y.; Raj, J.U. Regulation of the Pulmonary Circulation in the Fetus and Newborn. Physiol. Rev. 2010, 90, 1291–1335. [Google Scholar] [CrossRef]
- Patra, A.; Thakkar, P.S.; Makhoul, M.; Bada, H.S. Objective Assessment of Physiologic Alterations Associated with Hemodynamically Significant Patent Ductus Arteriosus in Extremely Premature Neonates. Front. Pediatr. 2021, 9, 648584. [Google Scholar] [CrossRef]
- Pellicer, A.; Riera, J.; Lopez-Ortego, P.; Bravo, M.C.; Madero, R.; Perez-Rodriguez, J.; Labrandero, C.; Quero, J.; Buño, A.; Castro, L.; et al. Phase 1 Study of Two Inodilators in Neonates Undergoing Cardiovascular Surgery. Pediatr. Res. 2013, 73, 95–103. [Google Scholar] [CrossRef]
- Petrova, A.; Bhatt, M.; Mehta, R. Regional Tissue Oxygenation in Preterm Born Infants in Association with Echocardiographically Significant Patent Ductus Arteriosus. J. Perinatol. 2011, 31, 460–464. [Google Scholar] [CrossRef]
- Barlow, A.J.; Ward, C.; Webber, S.A.; Sinclair, B.G.; Potts, J.E.; Sandor, G.G.S. Myocardial Contractility in Premature Neonates with and without Patent Ductus Arteriosus. Pediatr. Cardiol. 2004, 25, 102–107. [Google Scholar] [CrossRef]
- Noori, S.; Friedlich, P.; Seri, I.; Wong, P. Changes in Myocardial Function and Hemodynamics after Ligation of the Ductus Arteriosus in Preterm Infants. J. Pediatr. 2007, 150, 597–602. [Google Scholar] [CrossRef]
- Noori, S.; Seri, I. Neonatal Blood Pressure Support: The Use of Inotropes, Lusitropes, and Other Vasopressor Agents. Clin. Perinatol. 2012, 39, 221–238. [Google Scholar] [CrossRef]
- Durrmeyer, X.; Marchand-Martin, L.; Porcher, R.; Gascoin, G.; Roze, J.-C.; Storme, L.; Favrais, G.; Ancel, P.-Y.; Cambonie, G. Hemodynamic EPIPAGE 2 Study Group Abstention or Intervention for Isolated Hypotension in the First 3 Days of Life in Extremely Preterm Infants: Association with Short-Term Outcomes in the EPIPAGE 2 Cohort Study. Arch. Dis. Child. Fetal Neonatal. Ed. 2017, 102, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Ng, P.C.; Lee, C.H.; Lam, C.W.K.; Ma, K.C.; Fok, T.F.; Chan, I.H.S.; Wong, E. Transient Adrenocortical Insufficiency of Prematurity and Systemic Hypotension in Very Low Birthweight Infants. Arch. Dis. Child. Fetal Neonatal. Ed. 2004, 89, F119–F126. [Google Scholar] [CrossRef]
- Fernandez, E.; Schrader, R.; Watterberg, K. Prevalence of Low Cortisol Values in Term and Near-Term Infants with Vasopressor-Resistant Hypotension. J. Perinatol. 2005, 25, 114–118. [Google Scholar] [CrossRef]
- Aoki, M.; Urakami, T.; Nagano, N.; Aoki, R.; Morioka, I. Association of Plasma Cortisol Levels with Gestational Age and Anthropometric Values at Birth in Preterm Infants. Int. J. Environ. Res. Public Health 2022, 19, 11448. [Google Scholar] [CrossRef]
- Watterberg, K.L.; Shaffer, M.L.; Mishefske, M.J.; Leach, C.L.; Mammel, M.C.; Couser, R.J.; Abbasi, S.; Cole, C.H.; Aucott, S.W.; Thilo, E.H.; et al. Growth and Neurodevelopmental Outcomes after Early Low-Dose Hydrocortisone Treatment in Extremely Low Birth Weight Infants. Pediatrics 2007, 120, 40–48. [Google Scholar] [CrossRef]
- Huysman, M.W.; Hokken-Koelega, A.C.; De Ridder, M.A.; Sauer, P.J. Adrenal Function in Sick Very Preterm Infants. Pediatr. Res. 2000, 48, 629–633. [Google Scholar] [CrossRef]
- Foote, H.P.; Benjamin, D.K.; Greenberg, R.G.; Clark, R.H.; Hornik, C.P. Use of Vasopressors for Septic Shock in the Neonatal Intensive Care Unit. J. Perinatol. 2023, 43, 1274–1280. [Google Scholar] [CrossRef]
- Sahni, M.; Jain, S. Hypotension in Neonates. NeoReviews 2016, 17, e579–e589. [Google Scholar] [CrossRef]
- Stranak, Z.; Semberova, J.; Barrington, K.; O’Donnell, C.; Marlow, N.; Naulaers, G.; Dempsey, E.; HIP Consortium. International Survey on Diagnosis and Management of Hypotension in Extremely Preterm Babies. Eur. J. Pediatr. 2014, 173, 793–798. [Google Scholar] [CrossRef]
- Phad, N.; de Waal, K. What Inotrope and Why? Clin. Perinatol. 2020, 47, 529–547. [Google Scholar] [CrossRef]
- Joynt, C.; Cheung, P.-Y. Treating Hypotension in Preterm Neonates with Vasoactive Medications. Front. Pediatr. 2018, 6, 86. [Google Scholar] [CrossRef]
- Seri, I.; Rudas, G.; Bors, Z.; Kanyicska, B.; Tulassay, T. Effects of Low-Dose Dopamine Infusion on Cardiovascular and Renal Functions, Cerebral Blood Flow, and Plasma Catecholamine Levels in Sick Preterm Neonates. Pediatr. Res. 1993, 34, 742–749. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jentzer, J.C.; Coons, J.C.; Link, C.B.; Schmidhofer, M. Pharmacotherapy Update on the Use of Vasopressors and Inotropes in the Intensive Care Unit. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 249–260. [Google Scholar] [CrossRef]
- Jentzer, J.C.; Hollenberg, S.M. Vasopressor and Inotrope Therapy in Cardiac Critical Care. J. Intensive Care Med. 2021, 36, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Kent, A.L.; Chaudhari, T. Determinants of Neonatal Blood Pressure. Curr. Hypertens. Rep. 2013, 15, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Rozé, J.C.; Tohier, C.; Maingueneau, C.; Lefèvre, M.; Mouzard, A. Response to Dobutamine and Dopamine in the Hypotensive Very Preterm Infant. Arch. Dis. Child. 1993, 69, 59–63. [Google Scholar] [CrossRef]
- Seri, I. Cardiovascular, Renal, and Endocrine Actions of Dopamine in Neonates and Children. J. Pediatr. 1995, 126, 333–344. [Google Scholar] [CrossRef]
- Osborn, D.; Evans, N.; Kluckow, M. Randomized Trial of Dobutamine versus Dopamine in Preterm Infants with Low Systemic Blood Flow. J. Pediatr. 2002, 140, 183–191. [Google Scholar] [CrossRef]
- Pellicer, A.; Bravo, M.d.C.; Madero, R.; Salas, S.; Quero, J.; Cabañas, F. Early Systemic Hypotension and Vasopressor Support in Low Birth Weight Infants: Impact on Neurodevelopment. Pediatrics 2009, 123, 1369–1376. [Google Scholar] [CrossRef]
- Osborn, D.A.; Evans, N.; Kluckow, M.; Bowen, J.R.; Rieger, I. Low Superior Vena Cava Flow and Effect of Inotropes on Neurodevelopment to 3 Years in Preterm Infants. Pediatrics 2007, 120, 372–380. [Google Scholar] [CrossRef]
- Sarafidis, K.; Verykouki, E.; Nikopoulos, S.; Apostolidou-Kiouti, F.; Diakonidis, T.; Agakidou, E.; Kontou, A.; Haidich, A.-B. Systematic Review and Meta-Analysis of Cardiovascular Medications in Neonatal Hypotension. Biomed. Hub. 2022, 7, 70–79. [Google Scholar] [CrossRef]
- Driscoll, D.J.; Gillette, P.C.; Duff, D.F.; Nihill, M.R.; Gutgesell, H.P.; Vargo, T.A.; Mullins, C.E.; McNamara, D.G. Hemodynamic Effects of Dobutamine in Children. Am. J. Cardiol. 1979, 43, 581–585. [Google Scholar] [CrossRef]
- Mahoney, L.; Shah, G.; Crook, D.; Rojas-Anaya, H.; Rabe, H. A Literature Review of the Pharmacokinetics and Pharmacodynamics of Dobutamine in Neonates. Pediatr. Cardiol. 2016, 37, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Hallik, M.; Ilmoja, M.-L.; Standing, J.F.; Soeorg, H.; Jalas, T.; Raidmäe, M.; Uibo, K.; Köbas, K.; Sõnajalg, M.; Takkis, K.; et al. Population Pharmacokinetics and Pharmacodynamics of Dobutamine in Neonates on the First Days of Life. Br. J. Clin. Pharmacol. 2020, 86, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Pellicer, A.; Valverde, E.; Elorza, M.D.; Madero, R.; Gayá, F.; Quero, J.; Cabañas, F. Cardiovascular Support for Low Birth Weight Infants and Cerebral Hemodynamics: A Randomized, Blinded, Clinical Trial. Pediatrics 2005, 115, 1501–1512. [Google Scholar] [CrossRef] [PubMed]
- Valverde, E.; Pellicer, A.; Madero, R.; Elorza, D.; Quero, J.; Cabañas, F. Dopamine versus Epinephrine for Cardiovascular Support in Low Birth Weight Infants: Analysis of Systemic Effects and Neonatal Clinical Outcomes. Pediatrics 2006, 117, e1213–e1222. [Google Scholar] [CrossRef] [PubMed]
- Heckmann, M.; Trotter, A.; Pohlandt, F.; Lindner, W. Epinephrine Treatment of Hypotension in Very Low Birthweight Infants. Acta Paediatr. 2002, 91, 566–570. [Google Scholar] [CrossRef] [PubMed]
- Rizk, M.Y.; Lapointe, A.; Lefebvre, F.; Barrington, K.J. Norepinephrine Infusion Improves Haemodynamics in the Preterm Infants during Septic Shock. Acta Paediatr. 2018, 107, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Nissimov, S.; Joye, S.; Kharrat, A.; Zhu, F.; Ripstein, G.; Baczynski, M.; Choudhury, J.; Jasani, B.; Deshpande, P.; Ye, X.Y.; et al. Dopamine or Norepinephrine for Sepsis-Related Hypotension in Preterm Infants: A Retrospective Cohort Study. Eur. J. Pediatr. 2023, 182, 1029–1038. [Google Scholar] [CrossRef]
- Hong, L.; Davies, M.; Whitfield, K. Noradrenaline Use for Neonatal Circulatory Support. J. Paediatr. Child Health 2022, 58, 2084–2090. [Google Scholar] [CrossRef]
- Rowcliff, K.; de Waal, K.; Mohamed, A.-L.; Chaudhari, T. Noradrenaline in Preterm Infants with Cardiovascular Compromise. Eur. J. Pediatr. 2016, 175, 1967–1973. [Google Scholar] [CrossRef] [PubMed]
- Tourneux, P.; Rakza, T.; Abazine, A.; Krim, G.; Storme, L. Noradrenaline for Management of Septic Shock Refractory to Fluid Loading and Dopamine or Dobutamine in Full-Term Newborn Infants. Acta Paediatr. 2008, 97, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Lozada Martinez, I.D.; Bayona-Gamboa, A.J.; Meza-Fandiño, D.F.; Paz-Echeverry, O.A.; Ávila-Bonilla, Á.M.; Paz-Echeverry, M.J.; Pineda-Trujillo, F.J.; Rodríguez-García, G.P.; Covaleda-Vargas, J.E.; Narvaez-Rojas, A.R. Inotropic Support in Cardiogenic Shock: Who Leads the Battle, Milrinone or Dobutamine? Ann. Med. Surg. 2022, 82, 104763. [Google Scholar] [CrossRef] [PubMed]
- Giaccone, A.; Zuppa, A.F.; Sood, B.; Cohen, M.S.; O’Byrne, M.L.; Moorthy, G.; Mathur, A.; Kirpalani, H. Milrinone Pharmacokinetics and Pharmacodynamics in Neonates with Persistent Pulmonary Hypertension of the Newborn. Am. J. Perinatol. 2017, 34, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Paradisis, M.; Jiang, X.; McLachlan, A.J.; Evans, N.; Kluckow, M.; Osborn, D. Population Pharmacokinetics and Dosing Regimen Design of Milrinone in Preterm Infants. Arch. Dis. Child. Fetal Neonatal. Ed. 2007, 92, F204–F209. [Google Scholar] [CrossRef] [PubMed]
- Hallik, M.; Tasa, T.; Starkopf, J.; Metsvaht, T. Dosing of Milrinone in Preterm Neonates to Prevent Postligation Cardiac Syndrome: Simulation Study Suggests Need for Bolus Infusion. Neonatology 2017, 111, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Samiee-Zafarghandy, S.; Raman, S.R.; van den Anker, J.N.; McHutchison, K.; Hornik, C.P.; Clark, R.H.; Brian Smith, P.; Best Pharmaceuticals for Children Act—Pediatric Trials Network Administrative Core Committee. Safety of Milrinone Use in Neonatal Intensive Care Units. Early Hum. Dev. 2015, 91, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Rios, D.R.; Kaiser, J.R. Vasopressin versus Dopamine for Treatment of Hypotension in Extremely Low Birth Weight Infants: A Randomized, Blinded Pilot Study. J. Pediatr. 2015, 166, 850–855. [Google Scholar] [CrossRef]
- Khare, C.; Adhisivam, B.; Bhat, B.V.; Vaishnav, D. Utility of Low Dose Vasopressin for Persistent Pulmonary Hypertension of Newborn with Catecholamine Refractory Shock. Indian J. Pediatr. 2021, 88, 450–454. [Google Scholar] [CrossRef]
- Meyer, S. Vasopressin Infusion Therapy for Refractory Hypotension in Extremely Low Birthweight Neonates. Pediatr. Int. 2011, 53, 287. [Google Scholar] [CrossRef]
- Alakeel, Y.S.; Alkahtani, M.M.; Hijazi, O.M.; Algahtani, M.M. Vasopressin Associated Hyponatremia in Critically Ill Children: A Cross-Sectional Study. Saudi Pharm. J. 2022, 30, 1107–1112. [Google Scholar] [CrossRef]
- Ikegami, H.; Funato, M.; Tamai, H.; Wada, H.; Nabetani, M.; Nishihara, M. Low-Dose Vasopressin Infusion Therapy for Refractory Hypotension in ELBW Infants. Pediatr. Int. 2010, 52, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Al-Saadi, A.; Sushko, K.; Bui, V.; van den Anker, J.; Razak, A.; Samiee-Zafarghandy, S. Efficacy and Safety of Vasopressin and Terlipressin in Preterm Neonates: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 13760. [Google Scholar] [CrossRef] [PubMed]
- López-Suárez, O.; Pérez-Muñuzuri, A.; Crespo-Suárez, P.; Fernández-Lorenzo, J. Terlipressin as Rescue Therapy for Amine-Resistant Shock in Premature Infants: A Report on Two Cases. J. Neonatal-Perinat. Med. 2009, 2, 57–60. [Google Scholar] [CrossRef]
- Mohamed, A.A.; Louis, D.; Surak, A.; Weisz, D.E.; McNamara, P.J.; Jain, A. Vasopressin for Refractory Persistent Pulmonary Hypertension of the Newborn in Preterm Neonates—A Case Series. J. Matern. Fetal Neonatal. Med. 2022, 35, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Filippi, L.; Gozzini, E.; Daniotti, M.; Pagliai, F.; Catarzi, S.; Fiorini, P. Rescue Treatment with Terlipressin in Different Scenarios of Refractory Hypotension in Newborns and Infants. Pediatr. Crit. Care Med. 2011, 12, e237–e241. [Google Scholar] [CrossRef] [PubMed]
- Oulego-Erroz, I.; Terroba-Seara, S.; Castanon-Lopez, L.; Rodriguez-Nunez, A. Rescue Treatment with Terlipressin for Persistent Pulmonary Hypertension and Refractory Shock in a Preterm Infant. Indian Pediatr. 2020, 57, 864–865. [Google Scholar] [CrossRef]
- Stathopoulos, L.; Nicaise, C.; Michel, F.; Thomachot, L.; Merrot, T.; Lagier, P.; Martin, C. Terlipressin as Rescue Therapy for Refractory Pulmonary Hypertension in a Neonate with a Congenital Diaphragmatic Hernia. J. Pediatr. Surg. 2011, 46, e19–e21. [Google Scholar] [CrossRef]
- Bissolo, F.; Forcellini, C.A.; Spaggiari, S.; Gaffuri, M.; Pagano, G.; Biban, P. Is There Any Role for Terlipressin in the Extremely Low Birth Weight Infant with Refractory Septic Shock? J. Pediatr. Neonatal Individ. Med. (JPNIM) 2012, 1, 103–106. [Google Scholar] [CrossRef]
- Matok, I.; Leibovitch, L.; Vardi, A.; Adam, M.; Rubinshtein, M.; Barzilay, Z.; Paret, G. Terlipressin as Rescue Therapy for Intractable Hypotension during Neonatal Septic Shock. Pediatr. Crit. Care Med. 2004, 5, 116–118. [Google Scholar] [CrossRef]
- Papoff, P.; Mancuso, M.; Barbara, C.S.; Moretti, C. The Role of Terlipressin in Pediatric Septic Shock: A Review of the Literature and Personal Experience. Int. J. Immunopathol. Pharmacol. 2007, 20, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Heringlake, M.; Alvarez, J.; Bettex, D.; Bouchez, S.; Fruhwald, S.; Girardis, M.; Grossini, E.; Guarracino, F.; Herpain, A.; Toller, W.; et al. An Update on Levosimendan in Acute Cardiac Care: Applications and Recommendations for Optimal Efficacy and Safety. Expert Rev. Cardiovasc. Ther. 2021, 19, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Ricci, Z.; Garisto, C.; Favia, I.; Vitale, V.; Di Chiara, L.; Cogo, P.E. Levosimendan Infusion in Newborns after Corrective Surgery for Congenital Heart Disease: Randomized Controlled Trial. Intensive Care Med. 2012, 38, 1198–1204. [Google Scholar] [CrossRef]
- Takahashi, R.; Talukder, M.A.; Endoh, M. Inotropic Effects of OR-1896, an Active Metabolite of Levosimendan, on Canine Ventricular Myocardium. Eur. J. Pharmacol. 2000, 400, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Seri, I.; Tan, R.; Evans, J. Cardiovascular Effects of Hydrocortisone in Preterm Infants with Pressor-Resistant Hypotension. Pediatrics 2001, 107, 1070–1074. [Google Scholar] [CrossRef]
- Kumbhat, N.; Noori, S. Corticosteroids for Neonatal Hypotension. Clin. Perinatol. 2020, 47, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, K.; Szakmar, E.; Meder, U.; Szakacs, L.; Cseko, A.; Vatai, B.; Szabo, A.J.; McNamara, P.J.; Szabo, M.; Jermendy, A. A Randomized Controlled Study of Low-Dose Hydrocortisone Versus Placebo in Dopamine-Treated Hypotensive Neonates Undergoing Hypothermia Treatment for Hypoxic-Ischemic Encephalopathy. J. Pediatr. 2019, 211, 13–19.e3. [Google Scholar] [CrossRef]
- Ng, P.C.; Lee, C.H.; Bnur, F.L.; Chan, I.H.S.; Lee, A.W.Y.; Wong, E.; Chan, H.B.; Lam, C.W.K.; Lee, B.S.C.; Fok, T.F. A Double-Blind, Randomized, Controlled Study of a “Stress Dose” of Hydrocortisone for Rescue Treatment of Refractory Hypotension in Preterm Infants. Pediatrics 2006, 117, 367–375. [Google Scholar] [CrossRef]
- Matt, S.M.; Gaskill, P.J. Where Is Dopamine and How Do Immune Cells See It? Dopamine-Mediated Immune Cell Function in Health and Disease. J. Neuroimmune Pharmacol. 2020, 15, 114–164. [Google Scholar] [CrossRef]
- Channer, B.; Matt, S.M.; Nickoloff-Bybel, E.A.; Pappa, V.; Agarwal, Y.; Wickman, J.; Gaskill, P.J. Dopamine, Immunity, and Disease. Pharmacol. Rev. 2023, 75, 62–158. [Google Scholar] [CrossRef]
- Seri, I.; Evans, J. Controversies in the Diagnosis and Management of Hypotension in the Newborn Infant. Curr. Opin. Pediatr. 2001, 13, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Rios, D.R.; Moffett, B.S.; Kaiser, J.R. Trends in Pharmacotherapy for Neonatal Hypotension. J. Pediatr. 2014, 165, 697–701.e1. [Google Scholar] [CrossRef] [PubMed]
- Eldadah, M.K.; Schwartz, P.H.; Harrison, R.; Newth, C.J. Pharmacokinetics of Dopamine in Infants and Children. Crit. Care Med. 1991, 19, 1008–1011. [Google Scholar] [CrossRef] [PubMed]
- Padbury, J.F.; Agata, Y.; Baylen, B.G.; Ludlow, J.K.; Polk, D.H.; Goldblatt, E.; Pescetti, J. Dopamine Pharmacokinetics in Critically Ill Newborn Infants. J. Pediatr. 1987, 110, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Bhatt-Mehta, V.; Nahata, M.C.; McClead, R.E.; Menke, J.A. Dopamine Pharmacokinetics in Critically Ill Newborn Infants. Eur. J. Clin. Pharmacol. 1991, 40, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Evans, N. Which Inotrope for Which Baby? Arch. Dis. Child. Fetal Neonatal. Ed. 2006, 91, F213–F220. [Google Scholar] [CrossRef]
- Sassano-Higgins, S.; Friedlich, P.; Seri, I. A Meta-Analysis of Dopamine Use in Hypotensive Preterm Infants: Blood Pressure and Cerebral Hemodynamics. J. Perinatol. 2011, 31, 647–655. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, J.; Penny, D.J.; Kim, N.S.; Yu, V.Y.; Smolich, J.J. Mechanisms of Blood Pressure Increase Induced by Dopamine in Hypotensive Preterm Neonates. Arch. Dis. Child. Fetal Neonatal. Ed. 1999, 81, F99–F104. [Google Scholar] [CrossRef]
- Kirupakaran, K.; de Sousa, P.; Le Roux, C.; Redwood, L.; Rabe, H.; Patel, B.A. Evaluation of the Causal Effects between Dopamine Infusion Changeover and Fluctuations in Mean Arterial Pressure in Neonates. Arch. Dis. Child. 2020, 105, 390–394. [Google Scholar] [CrossRef]
- Eriksen, V.R.; Hahn, G.H.; Greisen, G. Dopamine Therapy Is Associated with Impaired Cerebral Autoregulation in Preterm Infants. Acta Paediatr. 2014, 103, 1221–1226. [Google Scholar] [CrossRef]
- Garner, R.S.; Burchfield, D.J. Treatment of Presumed Hypotension in Very Low Birthweight Neonates: Effects on Regional Cerebral Oxygenation. Arch. Dis. Child. Fetal Neonatal. Ed. 2013, 98, F117–F121. [Google Scholar] [CrossRef] [PubMed]
- Wong, F.Y.; Barfield, C.P.; Horne, R.S.C.; Walker, A.M. Dopamine Therapy Promotes Cerebral Flow-Metabolism Coupling in Preterm Infants. Intensive Care Med. 2009, 35, 1777–1782. [Google Scholar] [CrossRef] [PubMed]
- Bravo, M.C.; López-Ortego, P.; Sánchez, L.; Riera, J.; Madero, R.; Cabañas, F.; Pellicer, A. Randomized, Placebo-Controlled Trial of Dobutamine for Low Superior Vena Cava Flow in Infants. J. Pediatr. 2015, 167, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, M.B.; Chu, T.G.; Murphy, F.M.; Willits, N.; Morse, L.S. Dopamine Use Is an Indicator for the Development of Threshold Retinopathy of Prematurity. Br. J. Ophthalmol. 1999, 83, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Berg, R.A.; Donnerstein, R.L.; Padbury, J.F. Dobutamine Infusions in Stable, Critically Ill Children: Pharmacokinetics and Hemodynamic Actions. Crit. Care Med. 1993, 21, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Filippi, L.; Pezzati, M.; Poggi, C.; Rossi, S.; Cecchi, A.; Santoro, C. Dopamine versus Dobutamine in Very Low Birthweight Infants: Endocrine Effects. Arch. Dis. Child. Fetal Neonatal. Ed. 2007, 92, F367–F371. [Google Scholar] [CrossRef] [PubMed]
- Schranz, D.; Stopfkuchen, H.; Jüngst, B.K.; Clemens, R.; Emmrich, P. Hemodynamic Effects of Dobutamine in Children with Cardiovascular Failure. Eur. J. Pediatr. 1982, 139, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Sakata, Y.; Anzai, T.; Iijima, T.; Yoshida, I.; Hamada, Y.; Obayashi, T.; Ishikawa, S.; Yanagisawa, H.; Kodama, H.; Otaki, A. Effects of dobutamine and isoproterenol on systemic hemodynamics and myocardial metabolism in children after open heart surgery. Kokyu Junkan 1992, 40, 65–70. [Google Scholar]
- Osborn, D.A.; Evans, N.; Kluckow, M. Left Ventricular Contractility in Extremely Premature Infants in the First Day and Response to Inotropes. Pediatr. Res. 2007, 61, 335–340. [Google Scholar] [CrossRef]
- Robel-Tillig, E.; Knüpfer, M.; Pulzer, F.; Vogtmann, C. Cardiovascular Impact of Dobutamine in Neonates with Myocardial Dysfunction. Early Hum. Dev. 2007, 83, 307–312. [Google Scholar] [CrossRef]
- Pellicer, A.; Fernández, R.; Jullien, V.; Gleeson, C.; Bravo, M.C.; Ortego, P.L.; Sánchez, L.; Ybarra, M.; Rojas-Anaya, H.; Cabañas, F.; et al. Pharmacokinetic Study (Phase I–II) of a New Dobutamine Formulation in Preterm Infants Immediately after Birth. Pediatr. Res. 2021, 89, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, K.M.; Ammar, A.S. Norepinephrine Supplemented with Dobutamine or Epinephrine for the Cardiovascular Support of Patients with Septic Shock. Indian J. Crit. Care Med. 2012, 16, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.E.; Byrne, P.J. Cardiopulmonary Resuscitation and Epinephrine Infusion in Extremely Low Birth Weight Infants in the Neonatal Intensive Care Unit. J. Perinatol. 2004, 24, 691–695. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.G.; Schwartz, P.H.; Davis, A.L. Pharmacokinetics of Exogenous Epinephrine in Critically Ill Children. Crit. Care Med. 1993, 21, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Oualha, M.; Urien, S.; Spreux-Varoquaux, O.; Bordessoule, A.; D’Agostino, I.; Pouard, P.; Tréluyer, J.-M. Pharmacokinetics, Hemodynamic and Metabolic Effects of Epinephrine to Prevent Post-Operative Low Cardiac Output Syndrome in Children. Crit. Care 2014, 18, R23. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Kaiser, J.R.; Moffett, B.S.; Rodman, E.; Toy, C.; Rios, D.R. Efficacy of Low-Dose Epinephrine Continuous Infusion in Neonatal Intensive Care Unit Patients. J. Pediatr. Pharmacol. Ther. 2021, 26, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Garvey, A.A.; Kooi, E.M.W.; Dempsey, E.M. Inotropes for Preterm Infants: 50 Years on Are We Any Wiser? Front. Pediatr. 2018, 6, 88. [Google Scholar] [CrossRef]
- Fage, N.; Asfar, P.; Radermacher, P.; Demiselle, J. Norepinephrine and Vasopressin in Hemorrhagic Shock: A Focus on Renal Hemodynamics. Int. J. Mol. Sci. 2023, 24, 4103. [Google Scholar] [CrossRef]
- Gupta, S.; Agrawal, G.; Thakur, S.; Gupta, A.; Wazir, S. The Effect of Norepinephrine on Clinical and Hemodynamic Parameters in Neonates with Shock: A Retrospective Cohort Study. Eur. J. Pediatr. 2022, 181, 2379–2387. [Google Scholar] [CrossRef]
- Lu, P.; Sun, Y.; Gong, X.; Li, Z.; Hong, W. Use of Norepinephrine in Preterm Neonates with Dopamine-Resistant Shock: A Retrospective Single-Centre Cross-Sectional Study. BMJ Paediatr. Open 2023, 7, e001804. [Google Scholar] [CrossRef]
- Banothu, K.K.; Sankar, J.; Kumar, U.V.; Gupta, P.; Pathak, M.; Jat, K.R.; Kabra, S.K.; Lodha, R. A Randomized Controlled Trial of Norepinephrine Plus Dobutamine Versus Epinephrine as First-Line Vasoactive Agents in Children with Fluid Refractory Cold Septic Shock. Crit. Care Explor. 2023, 5, e0815. [Google Scholar] [CrossRef] [PubMed]
- Tourneux, P.; Rakza, T.; Bouissou, A.; Krim, G.; Storme, L. Pulmonary Circulatory Effects of Norepinephrine in Newborn Infants with Persistent Pulmonary Hypertension. J. Pediatr. 2008, 153, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit. Care Med. 2017, 45, 486–552. [Google Scholar] [CrossRef] [PubMed]
- Na, S.J.; Yang, J.H.; Ko, R.-E.; Chung, C.R.; Cho, Y.H.; Choi, K.H.; Kim, D.; Park, T.K.; Lee, J.M.; Song, Y.B.; et al. Dopamine versus Norepinephrine as the First-Line Vasopressor in the Treatment of Cardiogenic Shock. PLoS ONE 2022, 17, e0277087. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Gurumurthy, G.; Sridharan, L.; Gupta, D.; Nicholson, W.J.; Jaber, W.A.; Vallabhajosyula, S. A Clinical Update on Vasoactive Medication in the Management of Cardiogenic Shock. Clin. Med. Insights Cardiol. 2022, 16, 11795468221075064. [Google Scholar] [CrossRef] [PubMed]
- Halliday, M.; Kavarana, M.; Ebeling, M.; Kiger, J. Milrinone Use for Hemodynamic Instability in Patent Ductus Arteriosus Ligation. J. Matern. Fetal Neonatal. Med. 2017, 30, 529–533. [Google Scholar] [CrossRef] [PubMed]
- van Diepen, S.; Katz, J.N.; Albert, N.M.; Henry, T.D.; Jacobs, A.K.; Kapur, N.K.; Kilic, A.; Menon, V.; Ohman, E.M.; Sweitzer, N.K.; et al. Contemporary Management of Cardiogenic Shock: A Scientific Statement from the American Heart Association. Circulation 2017, 136, e232–e268. [Google Scholar] [CrossRef]
- Carceles, M.D.; Fuentes, T.; Aroca, V.; Lopez, J.; Hernández, J. Effects of Milrinone on Contractility and Cyclic Adenosine Monophosphate Production Induced by Beta1- and Beta2-Adrenergic Receptor Activation in Human Myocardium. Clin. Ther. 2007, 29, 1718–1724. [Google Scholar] [CrossRef] [PubMed]
- Chong, L.Y.Z.; Satya, K.; Kim, B.; Berkowitz, R. Milrinone Dosing and a Culture of Caution in Clinical Practice. Cardiol. Rev. 2018, 26, 35–42. [Google Scholar] [CrossRef]
- Hoffman, T.M.; Wernovsky, G.; Atz, A.M.; Kulik, T.J.; Nelson, D.P.; Chang, A.C.; Bailey, J.M.; Akbary, A.; Kocsis, J.F.; Kaczmarek, R.; et al. Efficacy and Safety of Milrinone in Preventing Low Cardiac Output Syndrome in Infants and Children after Corrective Surgery for Congenital Heart Disease. Circulation 2003, 107, 996–1002. [Google Scholar] [CrossRef]
- Bassler, D.; Choong, K.; McNamara, P.; Kirpalani, H. Neonatal Persistent Pulmonary Hypertension Treated with Milrinone: Four Case Reports. Biol. Neonate 2006, 89, 1–5. [Google Scholar] [CrossRef] [PubMed]
- McNamara, P.J.; Shivananda, S.P.; Sahni, M.; Freeman, D.; Taddio, A. Pharmacology of Milrinone in Neonates with Persistent Pulmonary Hypertension of the Newborn and Suboptimal Response to Inhaled Nitric Oxide. Pediatr. Crit. Care Med. 2013, 14, 74–84. [Google Scholar] [CrossRef] [PubMed]
- James, A.T.; Bee, C.; Corcoran, J.D.; McNamara, P.J.; Franklin, O.; El-Khuffash, A.F. Treatment of Premature Infants with Pulmonary Hypertension and Right Ventricular Dysfunction with Milrinone: A Case Series. J. Perinatol. 2015, 35, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, A.; Francis, J.V.; Lewis, A.I. Use of Milrinone in the Management of Haemodynamic Instability Following Duct Ligation. Eur. J. Pediatr. 2011, 170, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Paradisis, M.; Evans, N.; Kluckow, M.; Osborn, D. Randomized Trial of Milrinone versus Placebo for Prevention of Low Systemic Blood Flow in Very Preterm Infants. J. Pediatr. 2009, 154, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Sahni, M.; El-Khuffash, A.; Khadawardi, E.; Sehgal, A.; McNamara, P.J. Use of Targeted Neonatal Echocardiography to Prevent Postoperative Cardiorespiratory Instability after Patent Ductus Arteriosus Ligation. J. Pediatr. 2012, 160, 584–589.e1. [Google Scholar] [CrossRef] [PubMed]
- El-Khuffash, A.F.; Jain, A.; Weisz, D.; Mertens, L.; McNamara, P.J. Assessment and Treatment of Post Patent Ductus Arteriosus Ligation Syndrome. J. Pediatr. 2014, 165, 46–52.e1. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, B.E.U.; Rücker, G.; Stiller, B. Prophylactic Milrinone for the Prevention of Low Cardiac Output Syndrome and Mortality in Children Undergoing Surgery for Congenital Heart Disease. Cochrane Database Syst. Rev. 2015, CD009515. [Google Scholar] [CrossRef]
- Cavigelli-Brunner, A.; Hug, M.I.; Dave, H.; Baenziger, O.; Buerki, C.; Bettex, D.; Cannizzaro, V.; Balmer, C. Prevention of Low Cardiac Output Syndrome After Pediatric Cardiac Surgery: A Double-Blind Randomized Clinical Pilot Study Comparing Dobutamine and Milrinone. Pediatr. Crit. Care Med. 2018, 19, 619–625. [Google Scholar] [CrossRef]
- Lechner, E.; Hofer, A.; Leitner-Peneder, G.; Freynschlag, R.; Mair, R.; Weinzettel, R.; Rehak, P.; Gombotz, H. Levosimendan versus Milrinone in Neonates and Infants after Corrective Open-Heart Surgery: A Pilot Study. Pediatr. Crit. Care Med. 2012, 13, 542–548. [Google Scholar] [CrossRef]
- Yoshimura, M.; Conway-Campbell, B.; Ueta, Y. Arginine Vasopressin: Direct and Indirect Action on Metabolism. Peptides 2021, 142, 170555. [Google Scholar] [CrossRef] [PubMed]
- Watts, J.A.; Arroyo, J.P. Rethinking Vasopressin: New Insights into Vasopressin Signaling and Its Implications. Kidney360 2023, 4, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Treschan, T.A.; Peters, J. The Vasopressin System: Physiology and Clinical Strategies. Anesthesiology 2006, 105, 599–612; quiz 639–640. [Google Scholar] [CrossRef] [PubMed]
- Bankir, L.; Bichet, D.G.; Morgenthaler, N.G. Vasopressin: Physiology, Assessment and Osmosensation. J. Intern. Med. 2017, 282, 284–297. [Google Scholar] [CrossRef] [PubMed]
- Ertmer, C.; Rehberg, S.; Westphal, M. Vasopressin Analogues in the Treatment of Shock States: Potential Pitfalls. Best. Pract. Res. Clin. Anaesthesiol. 2008, 22, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Lolait, S.J.; O’Carroll, A.M.; McBride, O.W.; Konig, M.; Morel, A.; Brownstein, M.J. Cloning and Characterization of a Vasopressin V2 Receptor and Possible Link to Nephrogenic Diabetes Insipidus. Nature 1992, 357, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Acker, S.N.; Kinsella, J.P.; Abman, S.H.; Gien, J. Vasopressin Improves Hemodynamic Status in Infants with Congenital Diaphragmatic Hernia. J. Pediatr. 2014, 165, 53–58.e1. [Google Scholar] [CrossRef] [PubMed]
- Shivanna, B.; Rios, D.; Rossano, J.; Fernandes, C.J.; Pammi, M. Vasopressin and Its Analogues for the Treatment of Refractory Hypotension in Neonates. Cochrane Database Syst. Rev. 2013, CD009171. [Google Scholar] [CrossRef] [PubMed]
- Kam, P.C.A.; Williams, S.; Yoong, F.F.Y. Vasopressin and Terlipressin: Pharmacology and Its Clinical Relevance. Anaesthesia 2004, 59, 993–1001. [Google Scholar] [CrossRef]
- Alten, J.A.; Borasino, S.; Toms, R.; Law, M.A.; Moellinger, A.; Dabal, R.J. Early Initiation of Arginine Vasopressin Infusion in Neonates after Complex Cardiac Surgery. Pediatr. Crit. Care Med. 2012, 13, 300–304. [Google Scholar] [CrossRef]
- Ni, M.; Kaiser, J.R.; Moffett, B.S.; Rhee, C.J.; Placencia, J.; Dinh, K.L.; Hagan, J.L.; Rios, D.R. Use of Vasopressin in Neonatal Intensive Care Unit Patients with Hypotension. J. Pediatr. Pharmacol. Ther. 2017, 22, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.N.; Miller, J.L.; Ranallo, C. Dosing of Vasopressin in Critically Ill Children: Implications for Medication Safety. Pediatr. Crit. Care Med. 2016, 17, 277–278. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Gottschling, S.; Baghai, A.; Wurm, D.; Gortner, L. Arginine-Vasopressin in Catecholamine-Refractory Septic versus Non-Septic Shock in Extremely Low Birth Weight Infants with Acute Renal Injury. Crit. Care 2006, 10, R71. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Quinones Cardona, V.; Menkiti, O.R. Use of Vasopressin in Persistent Pulmonary Hypertension of the Newborn: A Case Series. SAGE Open Med. Case Rep. 2022, 10, 2050313X221102289. [Google Scholar] [CrossRef] [PubMed]
- Siefkes, H.M.; Lakshminrusimha, S. Management of Systemic Hypotension in Term Infants with Persistent Pulmonary Hypertension of the Newborn: An Illustrated Review. Arch. Dis. Child. Fetal Neonatal. Ed. 2021, 106, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Dhalait, S.; Fursule, A.; Khandare, J.; Kaul, A. Use of Vasopressin as Rescue Therapy in Refractory Hypoxia and Refractory Systemic Hypotension in Term Neonates with Severe Persistent Pulmonary Hypertension-A Prospective Observational Study. Am. J. Perinatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Capolupo, I.; De Rose, D.U.; Mazzeo, F.; Monaco, F.; Giliberti, P.; Landolfo, F.; Di Pede, A.; Toscano, A.; Conforti, A.; Bagolan, P.; et al. Early Vasopressin Infusion Improves Oxygenation in Infants with Congenital Diaphragmatic Hernia. Front. Pediatr. 2023, 11, 1104728. [Google Scholar] [CrossRef] [PubMed]
- Ouellet, S.; Drolet, C.; Morissette, G.; Pellerin, A.; Hébert, A. Vasopressin in Newborns with Refractory Acute Pulmonary Hypertension. Pediatr. Res. 2024. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, G.; Lindblom, P.; Ohlin, M.; Berling, R.; Vernersson, E. Pharmacokinetics of Terlipressin after Single i.v. Doses to Healthy Volunteers. Drugs Exp. Clin. Res. 1990, 16, 307–314. [Google Scholar]
- Chen, Q.; Huang, Z.; Chen, Y. Terlipressin in the Treatment of Neonatal Refractory Hypotension Caused by Septic Shock: A Case Report. Turk. J. Pediatr. 2020, 62, 668–672. [Google Scholar] [CrossRef]
- Masarwa, R.; Paret, G.; Perlman, A.; Reif, S.; Raccah, B.H.; Matok, I. Role of Vasopressin and Terlipressin in Refractory Shock Compared to Conventional Therapy in the Neonatal and Pediatric Population: A Systematic Review, Meta-Analysis, and Trial Sequential Analysis. Crit. Care 2017, 21, 1. [Google Scholar] [CrossRef]
- Rodríguez-Núñez, A.; López-Herce, J.; Gil-Antón, J.; Hernández, A.; Rey, C.; RETSPED Working Group of the Spanish Society of Pediatric Intensive Care. Rescue Treatment with Terlipressin in Children with Refractory Septic Shock: A Clinical Study. Crit. Care 2006, 10, R20. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.J.; Booth, R.A.; Petros, A.J. Terlipressin Bolus Induces Systemic Vasoconstriction in Septic Shock. Pediatr. Crit. Care Med. 2004, 5, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Barrett, L.K.; Singer, M.; Clapp, L.H. Vasopressin: Mechanisms of Action on the Vasculature in Health and in Septic Shock. Crit. Care Med. 2007, 35, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Berg, R.A. A Long-Acting Vasopressin Analog for Septic Shock: Brilliant Idea or Dangerous Folly? Pediatr. Crit. Care Med. 2004, 5, 188–189. [Google Scholar] [CrossRef] [PubMed]
- Papp, Z.; Édes, I.; Fruhwald, S.; De Hert, S.G.; Salmenperä, M.; Leppikangas, H.; Mebazaa, A.; Landoni, G.; Grossini, E.; Caimmi, P.; et al. Levosimendan: Molecular Mechanisms and Clinical Implications: Consensus of Experts on the Mechanisms of Action of Levosimendan. Int. J. Cardiol. 2012, 159, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Yang, Y.-M.; Zhu, J.-Y.; Lu, Y.-Q. Multiorgan Drug Action of Levosimendan in Critical Illnesses. Biomed. Res. Int. 2019, 2019, 9731467. [Google Scholar] [CrossRef] [PubMed]
- Antila, S.; Kivikko, M.; Lehtonen, L.; Eha, J.; Heikkilä, A.; Pohjanjousi, P.; Pentikäinen, P.J. Pharmacokinetics of Levosimendan and Its Circulating Metabolites in Patients with Heart Failure after an Extended Continuous Infusion of Levosimendan. Br. J. Clin. Pharmacol. 2004, 57, 412–415. [Google Scholar] [CrossRef]
- Louhelainen, M.; Merasto, S.; Finckenberg, P.; Vahtola, E.; Kaheinen, P.; Leskinen, H.; Levijoki, J.; Pollesello, P.; Haikala, H.; Mervaala, E.M.A. Effects of Calcium Sensitizer OR-1986 on a Cardiovascular Mortality and Myocardial Remodelling in Hypertensive Dahl/Rapp Rats. J. Physiol. Pharmacol. 2009, 60, 41–47. [Google Scholar]
- Kivikko, M.; Lehtonen, L.; Colucci, W.S. Sustained Hemodynamic Effects of Intravenous Levosimendan. Circulation 2003, 107, 81–86. [Google Scholar] [CrossRef]
- Bourgoin, P.; Lecomte, J.; Oualha, M.; Berthomieu, L.; Pereira, T.; Davril, E.; Lamoureux, F.; Joram, N.; Chenouard, A.; Duflot, T. Population Pharmacokinetics of Levosimendan and Its Metabolites in Critically Ill Neonates and Children Supported or Not by Extracorporeal Membrane Oxygenation. Clin. Pharmacokinet. 2023, 62, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Bravo, M.C.; López, P.; Cabañas, F.; Pérez-Rodríguez, J.; Pérez-Fernández, E.; Quero, J.; Pellicer, A. Acute Effects of Levosimendan on Cerebral and Systemic Perfusion and Oxygenation in Newborns: An Observational Study. Neonatology 2011, 99, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Momeni, M.; Rubay, J.; Matta, A.; Rennotte, M.-T.; Veyckemans, F.; Poncelet, A.J.; Clement de Clety, S.; Anslot, C.; Joomye, R.; Detaille, T. Levosimendan in Congenital Cardiac Surgery: A Randomized, Double-Blind Clinical Trial. J. Cardiothorac. Vasc. Anesth. 2011, 25, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, L.; Holcher, S.; Leyens, J.; Geipel, A.; Strizek, B.; Dresbach, T.; Mueller, A.; Kipfmueller, F. Evaluation of Levosimendan as Treatment Option in a Large Case-Series of Preterm Infants with Cardiac Dysfunction and Pulmonary Hypertension. Eur. J. Pediatr. 2023, 182, 3165–3174. [Google Scholar] [CrossRef]
- Angadi, U.; Westrope, C.; Chowdhry, M.F. Is Levosimendan Effective in Paediatric Heart Failure and Post-Cardiac Surgeries? Interact. Cardiovasc. Thorac. Surg. 2013, 17, 710–714. [Google Scholar] [CrossRef] [PubMed]
- Hummel, J.; Rücker, G.; Stiller, B. Prophylactic Levosimendan for the Prevention of Low Cardiac Output Syndrome and Mortality in Paediatric Patients Undergoing Surgery for Congenital Heart Disease. Cochrane Database Syst. Rev. 2017, 3, CD011312. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.P.; Dasnadi, S.; Zhao, Y.; Chen, H.H. A Comparative Analysis of Ante- and Postnatal Clinical Characteristics of Extremely Premature Neonates Suffering from Refractory and Non-Refractory Hypotension: Is Early Clinical Differentiation Possible? Early Hum. Dev. 2017, 113, 49–54. [Google Scholar] [CrossRef]
- Noori, S.; Siassi, B.; Durand, M.; Acherman, R.; Sardesai, S.; Ramanathan, R. Cardiovascular Effects of Low-Dose Dexamethasone in Very Low Birth Weight Neonates with Refractory Hypotension. Biol. Neonate 2006, 89, 82–87. [Google Scholar] [CrossRef]
- Watterberg, K.L. Hydrocortisone Dosing for Hypotension in Newborn Infants: Less Is More. J. Pediatr. 2016, 174, 23–26.e1. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.F.; Watterberg, K.L. Relative Adrenal Insufficiency in the Preterm and Term Infant. J. Perinatol. 2009, 29 (Suppl. S2), S44–S49. [Google Scholar] [CrossRef]
- Kantake, M.; Ohkawa, N.; Iwasaki, T.; Ikeda, N.; Awaji, A.; Saito, N.; Shoji, H.; Shimizu, T. Postnatal Relative Adrenal Insufficiency Results in Methylation of the Glucocorticoid Receptor Gene in Preterm Infants: A Retrospective Cohort Study. Clin. Epigenet. 2018, 10, 66. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, K.; Sato, M.; Hashimoto, K.; Imamura, T.; Go, H.; Hosoya, M. A Polymorphism in the Glucocorticoid Receptor Gene Is Associated with Refractory Hypotension in Premature Infants. Pediatr. Neonatol. 2018, 59, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Vezina, H.E.; Ng, C.M.; Vazquez, D.M.; Barks, J.D.; Bhatt-Mehta, V. Population Pharmacokinetics of Unbound Hydrocortisone in Critically Ill Neonates and Infants with Vasopressor-Resistant Hypotension. Pediatr. Crit. Care Med. 2014, 15, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Ando, M.; Park, I.-S.; Wada, N.; Takahashi, Y. Steroid Supplementation: A Legitimate Pharmacotherapy after Neonatal Open Heart Surgery. Ann. Thorac. Surg. 2005, 80, 1672–1678; discusison 1678. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.L.; Carcillo, J.A.; Aneja, R.K.; Deymann, A.J.; Lin, J.C.; Nguyen, T.C.; Okhuysen-Cawley, R.S.; Relvas, M.S.; Rozenfeld, R.A.; Skippen, P.W.; et al. American College of Critical Care Medicine Clinical Practice Parameters for Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit. Care Med. 2017, 45, 1061–1093. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.; Sinha, I.P.; Subhedar, N.V. Corticosteroids for Treating Hypotension in Preterm Infants. Cochrane Database Syst. Rev. 2011, 2011, CD003662. [Google Scholar] [CrossRef] [PubMed]
- Noori, S.; Friedlich, P.; Wong, P.; Ebrahimi, M.; Siassi, B.; Seri, I. Hemodynamic Changes after Low-Dosage Hydrocortisone Administration in Vasopressor-Treated Preterm and Term Neonates. Pediatrics 2006, 118, 1456–1466. [Google Scholar] [CrossRef]
- Higgins, S.; Friedlich, P.; Seri, I. Hydrocortisone for Hypotension and Vasopressor Dependence in Preterm Neonates: A Meta-Analysis. J. Perinatol. 2010, 30, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Alsaleem, M.; Malik, A.; Lakshminrusimha, S.; Kumar, V.H. Hydrocortisone Improves Oxygenation Index and Systolic Blood Pressure in Term Infants with Persistent Pulmonary Hypertension. Clin. Med. Insights Pediatr. 2019, 13, 1179556519888918. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.W.; Cheong, J.L.; Hay, S.; Manley, B.J.; Halliday, H.L. Early (<7 Days) Systemic Postnatal Corticosteroids for Prevention of Bronchopulmonary Dysplasia in Preterm Infants. Cochrane Database Syst. Rev. 2021, 10, CD001146. [Google Scholar] [CrossRef]
- Parikh, N.A.; Lasky, R.E.; Kennedy, K.A.; Moya, F.R.; Hochhauser, L.; Romo, S.; Tyson, J.E. Postnatal Dexamethasone Therapy and Cerebral Tissue Volumes in Extremely Low Birth Weight Infants. Pediatrics 2007, 119, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Melan, N.; Pradat, P.; Godbert, I.; Pastor-Diez, B.; Basson, E.; Picaud, J.-C. Neurodevelopment at 24 Months Corrected Age in Extremely Preterm Infants Treated with Dexamethasone Alternatives during the Late Postnatal Period: A Cohort Study. Eur. J. Pediatr. 2024, 183, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Baud, O.; Trousson, C.; Biran, V.; Leroy, E.; Mohamed, D.; Alberti, C.; PREMILOC Trial Group. Association Between Early Low-Dose Hydrocortisone Therapy in Extremely Preterm Neonates and Neurodevelopmental Outcomes at 2 Years of Age. JAMA 2017, 317, 1329–1337. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.W.; Cheong, J.L.; Ehrenkranz, R.A.; Halliday, H.L. Early (<8 Days) Systemic Postnatal Corticosteroids for Prevention of Bronchopulmonary Dysplasia in Preterm Infants. Cochrane Database Syst. Rev. 2017, 10, CD001146. [Google Scholar] [CrossRef] [PubMed]
- Batton, B.; Zhu, X.; Fanaroff, J.; Kirchner, H.L.; Berlin, S.; Wilson-Costello, D.; Walsh, M. Blood Pressure, Anti-Hypotensive Therapy, and Neurodevelopment in Extremely Preterm Infants. J. Pediatr. 2009, 154, 351–357.e1. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.M.; West, C.R.; Cooke, R.W. Blood Pressure and Cerebral Haemorrhage and Ischaemia in Very Low Birthweight Infants. Early Hum. Dev. 1989, 19, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Fanaroff, A.A.; Fanaroff, J.M. Short- and Long-Term Consequences of Hypotension in ELBW Infants. Semin. Perinatol. 2006, 30, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Bouyssi-Kobar, M.; Brossard-Racine, M.; Jacobs, M.; Murnick, J.; Chang, T.; Limperopoulos, C. Regional Microstructural Organization of the Cerebral Cortex Is Affected by Preterm Birth. Neuroimage Clin. 2018, 18, 871–880. [Google Scholar] [CrossRef]
- DiSessa, T.G.; Leitner, M.; Ti, C.C.; Gluck, L.; Coen, R.; Friedman, W.F. The Cardiovascular Effects of Dopamine in the Severely Asphyxiated Neonate. J. Pediatr. 1981, 99, 772–776. [Google Scholar] [CrossRef]
- Rasmussen, M.I.; Hansen, M.L.; Pellicer, A.; Gluud, C.; Dempsey, E.; Mintzer, J.; Hyttel-Sørensen, S.; Heuchan, A.M.; Hagmann, C.; Ergenekon, E.; et al. Cerebral Oximetry Monitoring versus Usual Care for Extremely Preterm Infants: A Study Protocol for the 2-Year Follow-up of the SafeBoosC-III Randomised Clinical Trial. Trials 2023, 24, 653. [Google Scholar] [CrossRef]
- Baske, K.; Saini, S.S.; Dutta, S.; Sundaram, V. Epinephrine versus Dopamine in Neonatal Septic Shock: A Double-Blind Randomized Controlled Trial. Eur. J. Pediatr. 2018, 177, 1335–1342. [Google Scholar] [CrossRef]
- Plomgaard, A.M.; Alderliesten, T.; van Bel, F.; Benders, M.; Claris, O.; Cordeiro, M.; Dempsey, E.; Fumagalli, M.; Gluud, C.; Hyttel-Sorensen, S.; et al. No Neurodevelopmental Benefit of Cerebral Oximetry in the First Randomised Trial (SafeBoosC II) in Preterm Infants during the First Days of Life. Acta Paediatr. 2019, 108, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Osborn, D.A. Diagnosis and Treatment of Preterm Transitional Circulatory Compromise. Early Hum. Dev. 2005, 81, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, L.; Crook, D.; Walter, K.N.; Sherman, E.; Rabe, H. What Is the Evidence for the Use of Adrenaline in the Treatment of Neonatal Hypotension? Cardiovasc. Hematol. Agents Med. Chem. 2012, 10, 50–98. [Google Scholar] [CrossRef]
- De Backer, D.; Aldecoa, C.; Njimi, H.; Vincent, J.-L. Dopamine versus Norepinephrine in the Treatment of Septic Shock: A Meta-Analysis*. Crit. Care Med. 2012, 40, 725–730. [Google Scholar] [CrossRef]
- Russell, J.A.; Walley, K.R.; Singer, J.; Gordon, A.C.; Hébert, P.C.; Cooper, D.J.; Holmes, C.L.; Mehta, S.; Granton, J.T.; Storms, M.M.; et al. Vasopressin versus Norepinephrine Infusion in Patients with Septic Shock. N. Engl. J. Med. 2008, 358, 877–887. [Google Scholar] [CrossRef]
- Vasu, T.S.; Cavallazzi, R.; Hirani, A.; Kaplan, G.; Leiby, B.; Marik, P.E. Norepinephrine or Dopamine for Septic Shock: Systematic Review of Randomized Clinical Trials. J. Intensive Care Med. 2012, 27, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, H.; Zhang, D. Timing of Norepinephrine Initiation in Patients with Septic Shock: A Systematic Review and Meta-Analysis. Crit. Care 2020, 24, 488. [Google Scholar] [CrossRef]
- Elbouhy, M.A.; Soliman, M.; Gaber, A.; Taema, K.M.; Abdel-Aziz, A. Early Use of Norepinephrine Improves Survival in Septic Shock: Earlier than Early. Arch. Med. Res. 2019, 50, 325–332. [Google Scholar] [CrossRef]
- Avni, T.; Lador, A.; Lev, S.; Leibovici, L.; Paul, M.; Grossman, A. Vasopressors for the Treatment of Septic Shock: Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0129305. [Google Scholar] [CrossRef]
- Gorantiwar, S.; de Waal, K. Progression from Sepsis to Septic Shock and Time to Treatments in Preterm Infants with Late-Onset Sepsis. J. Paediatr. Child Health 2021, 57, 1905–1911. [Google Scholar] [CrossRef]
- Cheng, L.; Yan, J.; Han, S.; Chen, Q.; Chen, M.; Jiang, H.; Lu, J. Comparative Efficacy of Vasoactive Medications in Patients with Septic Shock: A Network Meta-Analysis of Randomized Controlled Trials. Crit. Care 2019, 23, 168. [Google Scholar] [CrossRef]
- Botha, P.; Parry, G.; Dark, J.H.; Macgowan, G.A. Acute Hemodynamic Effects of Intravenous Sildenafil Citrate in Congestive Heart Failure: Comparison of Phosphodiesterase Type-3 and -5 Inhibition. J. Heart Lung Transplant. 2009, 28, 676–682. [Google Scholar] [CrossRef]
- Beam, K.; Sharma, P.; Levy, P.; Beam, A.L. Artificial Intelligence in the Neonatal Intensive Care Unit: The Time Is Now. J. Perinatol. 2024, 44, 131–135. [Google Scholar] [CrossRef]
- McAdams, R.M.; Kaur, R.; Sun, Y.; Bindra, H.; Cho, S.J.; Singh, H. Predicting Clinical Outcomes Using Artificial Intelligence and Machine Learning in Neonatal Intensive Care Units: A Systematic Review. J. Perinatol. 2022, 42, 1561–1575. [Google Scholar] [CrossRef]
- Bravo, M.C.; Jiménez, R.; Parrado-Hernández, E.; Fernández, J.J.; Pellicer, A. Predicting the Effectiveness of Drugs Used for Treating Cardiovascular Conditions in Newborn Infants. Pediatr. Res. 2023, 95, 1124–1131. [Google Scholar] [CrossRef]
- Gill, A.B.; Weindling, A.M. Randomised Controlled Trial of Plasma Protein Fraction versus Dopamine in Hypotensive Very Low Birthweight Infants. Arch. Dis. Child. 1993, 69, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Klarr, J.M.; Faix, R.G.; Pryce, C.J.; Bhatt-Mehta, V. Randomized, Blind Trial of Dopamine versus Dobutamine for Treatment of Hypotension in Preterm Infants with Respiratory Distress Syndrome. J. Pediatr. 1994, 125, 117–122. [Google Scholar] [CrossRef]
- Bourchier, D.; Weston, P.J. Randomised Trial of Dopamine Compared with Hydrocortisone for the Treatment of Hypotensive Very Low Birthweight Infants. Arch. Dis. Child. Fetal Neonatal. Ed. 1997, 76, F174–F178. [Google Scholar] [CrossRef]
- Paradisis, M.; Osborn, D.A. Adrenaline for Prevention of Morbidity and Mortality in Preterm Infants with Cardiovascular Compromise. Cochrane Database Syst. Rev. 2004, CD003958. [Google Scholar] [CrossRef]
- Gaissmaier, R.E.; Pohlandt, F. Single-Dose Dexamethasone Treatment of Hypotension in Preterm Infants. J. Pediatr. 1999, 134, 701–705. [Google Scholar] [CrossRef]
- Bhayat, S.I.; Gowda, H.M.S.; Eisenhut, M. Should Dopamine Be the First Line Inotrope in the Treatment of Neonatal Hypotension? Review of the Evidence. World J. Clin. Pediatr. 2016, 5, 212–222. [Google Scholar] [CrossRef]
- Miall-Allen, V.M.; Whitelaw, A.G. Response to Dopamine and Dobutamine in the Preterm Infant Less than 30 Weeks Gestation. Crit. Care Med. 1989, 17, 1166–1169. [Google Scholar] [CrossRef]
- Wen, L.; Xu, L. The Efficacy of Dopamine versus Epinephrine for Pediatric or Neonatal Septic Shock: A Meta-Analysis of Randomized Controlled Studies. Ital. J. Pediatr. 2020, 46, 6. [Google Scholar] [CrossRef]
- Jothinath, K.; Balakrishnan, S.; Raju, V.; Menon, S.; Osborn, J. Clinical Efficacy of Levosimendan vs Milrinone in Preventing Low Cardiac Output Syndrome Following Pediatric Cardiac Surgery. Ann. Card. Anaesth. 2021, 24, 217–223. [Google Scholar] [CrossRef]
- Wang, A.; Cui, C.; Fan, Y.; Zi, J.; Zhang, J.; Wang, G.; Wang, F.; Wang, J.; Tan, Q. Prophylactic Use of Levosimendan in Pediatric Patients Undergoing Cardiac Surgery: A Prospective Randomized Controlled Trial. Crit. Care 2019, 23, 428. [Google Scholar] [CrossRef]
- Laughon, M.; Bose, C.; Allred, E.; O’Shea, T.M.; Van Marter, L.J.; Bednarek, F.; Leviton, A.; ELGAN Study Investigators. Factors Associated with Treatment for Hypotension in Extremely Low Gestational Age Newborns during the First Postnatal Week. Pediatrics 2007, 119, 273–280. [Google Scholar] [CrossRef]
- Wong, J.; Shah, P.S.; Yoon, E.W.; Yee, W.; Lee, S.; Dow, K. Inotrope Use among Extremely Preterm Infants in Canadian Neonatal Intensive Care Units: Variation and Outcomes. Am. J. Perinatol. 2015, 32, 9–14. [Google Scholar] [CrossRef]
- Batton, B.; Li, L.; Newman, N.S.; Das, A.; Watterberg, K.L.; Yoder, B.A.; Faix, R.G.; Laughon, M.M.; Stoll, B.J.; Van Meurs, K.P.; et al. Use of Antihypotensive Therapies in Extremely Preterm Infants. Pediatrics 2013, 131, e1865–e1873. [Google Scholar] [CrossRef]
- Thewissen, L.; Naulaers, G.; Hendrikx, D.; Caicedo, A.; Barrington, K.; Boylan, G.; Cheung, P.-Y.; Corcoran, D.; El-Khuffash, A.; Garvey, A.; et al. Cerebral Oxygen Saturation and Autoregulation during Hypotension in Extremely Preterm Infants. Pediatr. Res. 2021, 90, 373–380. [Google Scholar] [CrossRef]
- Askie, L.M.; Darlow, B.A.; Finer, N.; Schmidt, B.; Stenson, B.; Tarnow-Mordi, W.; Davis, P.G.; Carlo, W.A.; Brocklehurst, P.; Davies, L.C.; et al. Association Between Oxygen Saturation Targeting and Death or Disability in Extremely Preterm Infants in the Neonatal Oxygenation Prospective Meta-Analysis Collaboration. JAMA 2018, 319, 2190–2201. [Google Scholar] [CrossRef] [PubMed]
- Greisen, G.; Hansen, M.L.; Rasmussen, M.I.S.; Vestager, M.; Hyttel-Sørensen, S.; Hahn, G.H. Cerebral Oximetry in Preterm Infants-To Use or Not to Use, That Is the Question. Front. Pediatr. 2021, 9, 747660. [Google Scholar] [CrossRef]
- Murphy, E.; Healy, D.B.; Chioma, R.; Dempsey, E.M. Evaluation of the Hypotensive Preterm Infant: Evidence-Based Practice at the Bedside? Children 2023, 10, 519. [Google Scholar] [CrossRef] [PubMed]
- Pichler, G.; Baumgartner, S.; Biermayr, M.; Dempsey, E.; Fuchs, H.; Goos, T.G.; Lista, G.; Lorenz, L.; Karpinski, L.; Mitra, S.; et al. Cerebral Regional Tissue Oxygen Saturation to Guide Oxygen Delivery in Preterm Neonates during Immediate Transition after Birth (COSGOD III): An Investigator-Initiated, Randomized, Multi-Center, Multi-National, Clinical Trial on Additional Cerebral Tissue Oxygen Saturation Monitoring Combined with Defined Treatment Guidelines versus Standard Monitoring and Treatment as Usual in Premature Infants during Immediate Transition: Study Protocol for a Randomized Controlled Trial. Trials 2019, 20, 178. [Google Scholar] [CrossRef]
- Levy, P.T.; Pellicer, A.; Schwarz, C.E.; Neunhoeffer, F.; Schuhmann, M.U.; Breindahl, M.; Fumagelli, M.; Mintzer, J.; de Boode, W.; ESPR Special Interest Group “Near InfraRed Spectroscopy” (NIRS). Near-Infrared Spectroscopy for Perioperative Assessment and Neonatal Interventions. Pediatr. Res. 2021. [Google Scholar] [CrossRef]
- Dempsey, E.M.; Barrington, K.J.; Marlow, N.; O’Donnell, C.P.F.; Miletin, J.; Naulaers, G.; Cheung, P.-Y.; Corcoran, J.D.; El-Khuffash, A.F.; Boylan, G.B.; et al. Hypotension in Preterm Infants (HIP) Randomised Trial. Arch. Dis. Child. Fetal Neonatal. Ed. 2021, 106, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Batton, B.J.; Li, L.; Newman, N.S.; Das, A.; Watterberg, K.L.; Yoder, B.A.; Faix, R.G.; Laughon, M.M.; Van Meurs, K.P.; Carlo, W.A.; et al. Feasibility Study of Early Blood Pressure Management in Extremely Preterm Infants. J. Pediatr. 2012, 161, 65–69.e1. [Google Scholar] [CrossRef] [PubMed]
- Watterberg, K.L.; Fernandez, E.; Walsh, M.C.; Truog, W.E.; Stoll, B.J.; Sokol, G.M.; Kennedy, K.A.; Fraga, M.V.; Beauman, S.S.; Carper, B.; et al. Barriers to Enrollment in a Randomized Controlled Trial of Hydrocortisone for Cardiovascular Insufficiency in Term and Late Preterm Newborn Infants. J. Perinatol. 2017, 37, 1220–1223. [Google Scholar] [CrossRef]
- Noori, S.; Seri, I. Evidence-Based versus Pathophysiology-Based Approach to Diagnosis and Treatment of Neonatal Cardiovascular Compromise. Semin. Fetal Neonatal. Med. 2015, 20, 238–245. [Google Scholar] [CrossRef]
- Ruggieri, L.; Giannuzzi, V.; Baiardi, P.; Bonifazi, F.; Davies, E.H.; Giaquinto, C.; Bonifazi, D.; Felisi, M.; Chiron, C.; Pressler, R.; et al. Successful Private-Public Funding of Paediatric Medicines Research: Lessons from the EU Programme to Fund Research into off-Patent Medicines. Eur. J. Pediatr. 2015, 174, 481–491. [Google Scholar] [CrossRef]
- Pratico, A.D.; Longo, L.; Mansueto, S.; Gozzo, L.; Barberi, I.; Tiralongo, V.; Salvo, V.; Falsaperla, R.; Vitaliti, G.; La Rosa, M.; et al. Off-Label Use of Drugs and Adverse Drug Reactions in Pediatric Units: A Prospective, Multicenter Study. Curr. Drug. Saf. 2018, 13, 200–207. [Google Scholar] [CrossRef]
- Conroy, S.; Choonara, I.; Impicciatore, P.; Mohn, A.; Arnell, H.; Rane, A.; Knoeppel, C.; Seyberth, H.; Pandolfini, C.; Raffaelli, M.P.; et al. Survey of Unlicensed and off Label Drug Use in Paediatric Wards in European Countries. European Network for Drug Investigation in Children. BMJ 2000, 320, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Regulation (EC) No 1901/2006 of the European Parliament and of the Council of 12 December 2006 on Medicinal Products for Paediatric Use and Amending Regulation (EEC) No 1768/92, Directive 2001/20/EC, Directive 2001/83/EC and Regulation (EC) No 726/2004 (Text with EEA Relevance).
- Hallik, M.; Soeorg, H.; Kahre, T.; Murumets, Ü.; Ilmoja, M.-L.; Kipper, K.; Metsvaht, T. Pharmacogenetics May Explain Part of the Interindividual Variability of Dobutamine Pharmacodynamics in Neonates. Br. J. Clin. Pharmacol. 2022, 88, 4155–4162. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, C.E.; Dempsey, E.M. Management of Neonatal Hypotension and Shock. Semin. Fetal Neonatal. Med. 2020, 25, 101121. [Google Scholar] [CrossRef] [PubMed]
- Subhedar, N.V. Treatment of Hypotension in Newborns. Semin. Neonatol. 2003, 8, 413–423. [Google Scholar] [CrossRef] [PubMed]
| Medication [References] | Mechanisms of Action/Receptors | Hemodynamic Effects | Dose Regimen (Range) * | Main Indications | Potential Side Effects |
|---|---|---|---|---|---|
| Dopamine [9,40,51,56,57,58,59,60] | D1R/D2R | VD of peripheral vascular beds. Increases GFR and tubular functions; endocrine effects; induces EPI and NE production. | ≤2–5 (2–20) | Most common first-line agent for systemic AH in the absence of cardiac dysfunction. | Tachycardia: at doses ≥10, may cause PPHN; decreased production of TSH, GH, and prolactin. |
| DR > β-1R > α-1R | Positive inotropic and chronotropic; increase HR, AP, myocardiac contractility, and CBF. | ≥5–10 (2–20) | |||
| α-1R > β-1R | VC; increase AP and PVS and SVR. | ≥10–20 (2–20) | |||
| Dobutamine [9,14,49,51,56,61,62,63,64] | β-1R > β-2R > αR | Inotropic effects and VD; increase cardiac contractility and output. Decrease PVR and SVR. | 2.5–5 (2.5–10) | First- or second-line agent or combined with DOP; AH with cardiac dysfunction; transitional AH with low cardiac output and increased SVR or large PDA; PPHN with right ventricular dysfunction. | Tachycardia |
| Epinephrine [9,40,50,65,66,67] | β-1R > β-2R | Inotropic action: increase cardiac contractility and SVR. | 0.01 to 0.1 (0.05–2.5) | Adjuvant or rescue treatment for inotrope—refractory shock; PPHN with systemic AH; warm septic shock with decreased contractility; transitional AH with large PDA; rarely, first-line agent. | High doses (over 0.5): increased plasma glucose & lactate |
| α-1R > β-1R = β-2R | VC. Increase myocardiac contractility, SVR, HR, AP. | 0.1 to 0.2 (0.05–2.5) | |||
| Norepinephrine [51,53,68,69,70,71,72] | α-1R and α-2R | Vasopressor effects; inotropic action and systemic VC. Increases AP and decreases inotrope score. Mild pulmonary VD. | 0.01–0.04 (0.04–1) | Adjuvant in catecholamine refractory shock: warm septic shock, PPHN, post-cardiac surgery, perinatal asphyxia; AH with cardiac dysfunction; Maybe 1st choice for septic shock. | VC; transient systemic hypertension; potential tissue necrosis if extravasated. |
| Milrinone [51,73,74,75,76,77] | Phosphodiesterase inhibitor: increases intracellular cyclic AMP and calcium. | Inotropic and lusitropic actions; VD; decrease in pulmonary and systemic vascular resistance. | LD: 50; Infusion: 0.75 for 3 h. (0.25–0.75) | PPHN post-cardiac surgery; prevention of low LVOS after PDA ligation. | Tachycardia and hypotension requiring vasopressors. |
| Vasopressin (AVP) [51,78,79,80,81,82] | Antidiuretic hormone. V1R: systemic VD. V2R: selective VD of pulmonary, cerebral, and coronal vascular bed. | Increase AP in catecholamine-resistant shock and cardiac output. Potentially decrease pulmonary vascular resistance. | 0.00001 and 0.003 units/kg/min and 0.003 U/kg/min. (0.00001 and 0.003 U/kg/min) | Catecholamine- and steroid-refractory or VD shock, warm septic shock, PPHN, post-cardiac surgery. | Tachy-arrhythmias, hyponatremia; transient thrombocytopenia; liver necrosis. High doses: reductions in cardiac output and oxygen delivery. |
| Terlipressin [80,83,84,85,86,87,88,89,90,91] | Comparable to AVP. Higher affinity to V1R, longer half-life | Comparable to AVP | LD: 5–20 MD: 5–20 every 4 h | Volume and catecholamine–refractory shock. | Potentially comparable to AVP. Potential limb necrosis. Well-tolerated. |
| Levosimendan [36,92,93,94] | Calcium sensitizer; inotropic action via binding to cardiac troponin; VD action. | Increase myocardial contractility, AP, tissue perfusion and oxygenation; VD; decrease lactate and HR. | Infusion of 0.2 over 24 h. | Severe, refractory warm septic shock; LCOS; severe cardiac dysfunction and PPHN. | Tachycardia, arrythmia, hypotension potentially requiring intervention. Well tolerated. |
| Hydrocortisone [39,45,95,96,97,98] | Hormone; stimulation or reverse pf desensitization of AR; improvement in capillary integrity. | Increases AP, improves left ventricular function and oxygenation index, and decreases the need for vasopressors. | Initial: 1–2 mg/kg; MD: 0.5–1 mg/kg every 8–12 h | Adjuvant or rescue therapy for refractory AH (e.g., septic shock, perinatal asphyxia). PPHN resistant to iNO and inotropes. | Transient hyperglycemia, hypertension, spontaneous intestinal perforation, rare myocardial hypertrophy. |
| First Author (Year) [Reference] | Design/Aim | Population | Age at Exposure/Age at FU | Main Results | Authors’ Conclusions/Comments |
|---|---|---|---|---|---|
| DiSessa et al. (1981) [209] | RCT/AIM: Cardiovascular effects of DOP vs. PL in asphyxiated neonates. | 14 asphyxiated term neonates | Early postnatally/1–24 mo | DOP vs. PL: higher increase in AP, improved caECHO indices; 5/6 infants had a normal ND on FU. | DOP raises systemic AP and improves cardiac function. |
| Watkins et al. (1989) [206] | Cohort/AIM: Assoc. of AH periods with PIVH and ischemic lesions. | 131 VLBWI (GA 24–34 weeks); 56 (58% hypotensive) | First 4 DoL/2 years of age. | No association of AH with subsequent NDI in survivors. | Ischemic lesions did not correlate with periods of hypotension. |
| Low et al. (1993) [17] | Cohort/AIM: AH/hypoxemia assoc. with ND at 1 year. | 93 neonates (GA < 34 weeks), 2 Gr: 49 normal ND; 49 abnormal ND. | First 4 DoL/1 year | Major NDI: 8% without AH or hypoxemia vs. 53% with both AH and hypoxemia. | Combination of hypotension and hypoxemia are associated with long-term NDI. |
| Fanaroff et al. (2006) [207] | Retrospective/AIM: Assoc. of treated AH with ND at 20 mo CA. | 156 ELBWI (birth weight 400–999 g). | First 72 hoL/20 mo CA | Early outcomes: Treated AH assoc. with severe PIVH, longer hospital stay, and death. 20 mo CA outcome: Treated AH was assoc. with MDI < 70 and hearing loss. | Infants with treated hypotension are more likely to have delayed motor development, hearing loss, and death. |
| Osborn et al. (2007) [60] | RCT/AIM: Assoc. of early SVC flow (caECHO) and DOP vs. DOB with ND at 1 and 3 years. | 84 neonates (GA < 30 weeks) in 2 Grs: 84 normal SVC flow Gr vs. 44 low SVC flow Gr.: DOP-Gr vs. DOB-Gr. | 24 hoL/1 & 3 years. | (A) DOB vs. DOP: sign. lower rate of severe PIVH & disability at 3 year. No sign. dif. in clinical outcomes. (B) Low vs. normal SVC flow: increased mortality, morbidity, severe PIVH, and NDI at 3 year CA. | (A) No sign. dif. in combined death and disability between DOP & DOB. (B) Early low SVC flow was assoc. with increased rates of death, morbidity, and NDI. |
| Batton et al. (2009) [205] | Retrospective cohort/AIM: Assoc. of early AP treatment with NDI. | 168 ELBW (GA 23–25 weeks) in 3 Grs | First 72 h of life/18 to 22 mo PMA | Compared to untreated-normotensive, the untreated-low AP had more CP, deafness, or any NDI and the treated-low AP had worse NDI and less survival without NDI. | Early low AP alone is associated with adverse long-term ND outcomes regardless of treatment. |
| Kuint et al. (2009) [19] | Retrospective, case control/AIM: Identify risk factors for AH and relations to short- and long-term outcomes. | 218 survived VLBWI: TGr (n = 109) vs. CGr (normotensive, n = 109). | 24 h of life/ > 2 year | TGr vs. CGr: PIVH 2–4/PVL 11% vs. 2.7%; major disability: 20% vs. 5% | Early anti-hypotensive therapy is related to PIVH, PVL, and major NDI. |
| Pellicer et al. (2009) [59] | RCT (3 Grs). AIM: Effect of early treatment with DOP or EPI vs. no treatment on ND. | 60 hypotensive VLBWI (GA < 32 weeks) and 70 normotensive (CGr). | First 96 hoL/2–3 years of age. | DOP vs. EPI: No differences in NDI (death or CP or severe NDI). | Cautious use of cardiovascular support to treat early AH in LBWI seems to be safe. |
| Logan et al. (2011) [18] | Prospective cohort/AIM: Assoc. of AH and treatment with ND. | 945 neonates (GA < 28 weeks). | First 24 hoL/24 mo CA. | Adjusted AH was not associated with MDI < 70 or PDI < 70. | There is little evidence that early AH is assoc. with NDI at 24 mo CA. |
| Alderliesten et al. (2014) [12] | Prospective, observational case-control/AIM: Relation of early DOP and cerebral oxygenation with NDI. | 132 neonates (GA < 32 weeks, TGr): 66 hypotensive (volume vs. DOP); and CGr, 66 normotensive. | First 72 hoL/18 and 24 mo CA. | NDI was comparable between TGr & CGr but was assoc. with duration of low cerebral oxygenation. | Early hypotension was not assoc. with lower cerebral oxygenation or NDI. |
| Batton et al. (2016) [13] | Prospective observational/AIM: Relation of early AP and its therapy, with ND. | 158 survivors ELBWI (GA 23–26 weeks) in 4 groups. | First 24 h of life/18–22 mo CA. | Death or NDI was sign. higher in the treated as compared to untreated infants irrespective of AP changes. | Exposure to anti-hypotensive therapy was assoc. with increased risk of combined death and NDI at 18–22 months’ CA. |
| Bravo et al. (2021) [14] | RCT/AIM: Assoc. of early low SVC flow within with long-term outcome. | 28 neonates (GA < 31 weeks) with (a) low SVC flow; (b) 98 with normal SVC flow. | First 24 hoL/2 and 6 years. | No sign. dif. in combined outcome (mortality or NDI) between the DOB, PL, and normal flow groups. | No dif. in long-term outcome related to SVC or its treatment early after birth. SVC flow was not associated with long-term NDI |
| Doucette et al. (2022) [15] | Retrospective cohort/AIM: Examine ND relation with inotrope treatment. | 1394 survivors (GA < 29 weeks): TGr, 245 treated with inotropes. | First week of life/18–24 mo of age | Compared to CGr, the TGr had higher rates of hearing loss. No difference in the risk of adjusted NDI. | Inotrope treatment in the first WoL were at increased risk for hearing loss. There was no diff. in NDI. |
| Compared Medications [References] | Author (Year)/Design | Population | Results & Authors’ Conclusions |
|---|---|---|---|
| DOP vs. PL [209] | DiSessa et al. (1981) [209]/RCT. | 14 asphyxiated term neonates. | DOP raises AP and improve cardiac function in asphyxiated neonates. |
| DOP vs. PL [227] | Gilli et al. (1993) [227]/RCT. | 39 VLBWI (<24 hol). | Response rate was higher in DOP. DOP treatment should be used earlier in hypotensive neonates. |
| DOP vs. DOB [56] | Roze et al. (1993) [56]/RCT. | 20 hypotensive neonates (GA < 32 weeks) in first DoL. | DOP increased AP more efficiently, while only DOB increased the LVO. |
| DOP vs. DOB [228] | Klarr et al. (1994) [228]/RCT. | 63 hypotensive neonates (GA < 35 weeks) in the first 24 hol. | DOP is more effective than DOB for the early treatment of AH. |
| DOP vs. DOB [58,60] | Osborn et al. (2002 and 2007) follow up at 3 Years [58,60]/RCT. | 42 neonates (GA < 30 weeks) with low SVC flow in the first 24 hol. | DOB was more efficient in increasing blood flow, but less efficient in increasing AP. No dif. in long-term ND at 3 YoL. |
| DOP vs. DOB [10] | Lasky et al. (2011) [10]/retrospective cohort study. | 287 LBWI < 1 mo of age. | No dif. in mortality between DOP and DOB. Treatment with DOP alone was more common. |
| DOP vs. DOB [116] | Filippi et al. (2007) [116]/non-blind RCT. | 35 hypotensive VLBWI. | DOP is more effective than DOB in increasing systemic AP. DOP reduces TSH, T4, and prolactin. |
| DOP vs. DOB [16] | Subhedar et al. (2003) [16]/Cochrane review. | 209 infants (GA 23–36 weeks) < 28 days. | DOP was more effective than DOB for short-term treatment. The long-term effect on ND is unknown. |
| DOP vs. DOB vs. EPI vs. NE vs. MIL vs. AVP vs. LEVO vs. corticosteroids vs. volume. | Sarafidis et al. (2022) [61]/Systematic review and pairwise meta-analysis. | 19 studies in 758 hypotensive term and preterm neonates. | DOP more effectively increased AP than DOB. |
| DOP vs. EPI [65] | Pellicer et al. (2005) [65]/RCT. | 59 hypotensive neonates (GA < 32 weeks) aged 2–16 h. | Both medications showed comparable increases in BP, cerebral oxygenation, CBF, response rate, and need for rescue therapy. |
| DOP vs. EPI [66] | Valverde et al. (2006) [66]/RCT. | 60 hypotensive LBWI (GA < 32 weeks) < 24 hoL. | EPI is as effective as DOP for the treatment of AH in LBWI, but it is associated with more adverse effects. |
| DOP vs. EPI [59] | Pellicer et al. (2009) [59]/RCT. | 130 LBWI (GA < 32 weeks) < 24 hoL. | Cautious use of CV support for early systemic AH in LBWI seems to be safe. |
| DOP vs. NE as 1st-line treatment [69] | Nissimov et al. (2023) [69]/retrospective study. | 156 neonates (<35 weeks PMA) with sepsis or NEC. | NE was associated with decreased mortality, neurologic injury, and occurrence of NEC/sepsis among the survivors. |
| DOP vs. AVP [78] | Rios et al. (2015) [78]/RCT. | Hypotensive ELBWI (GA < 30 weeks) first 24 hoL. | AVP-Gr received fewer doses of surfactant, had lower PaCO2, and were not tachycardic. |
| DOP vs. Hydrocortisone [229] | Bourchier and Weston (1997) [229]/RCT. | 40 hypotensive VLBWI (GA 27 weeks). | Successful treatment: hydrocortisone 81% vs. DOP 100%. No dif. in any clinical outcome. |
| DOB vs. PL [14,113] | Bravo et al. (2015 & 2021) [14,113]/RCT exploratory short-term outcome and long-term studies. | 127 infants (GA < 31 weeks); 28 with low SVC flow and 98 normal SVC flow, within the first 24 hol. | SVC flow increased in the entire cohort. No dif. in AP and other clinical and biochemical parameters. No dif. in the combined outcome (mortality or NDI at 6 years). |
| DOB vs. MIL [149] | No study in neonates. Cavigelli-Brunner et al. (2018) [149]/pilot RCT in children. | 50 children (age 2.5 mo to 14.2 years). | DOB and MIL are safe, well tolerated, and equally effective in prevention of LCOS after pediatric cardiac surgery. |
| EPI vs. no treatment vs. other inotropes [230] | Paradisis et al. (2004) [230]/Cochrane review. | No published study was found. | |
| EPI vs. hydrocortisone as adjuvant treatments [47] | Foote et al. (2023) [47]/multicenter RCT. | 1592 infants with septic shock refractory to DOP. | The use of hydrocortisone as an adjuvant treatment was associated with decreased mortality. EPI alone or in combination therapy was associated with worse outcomes. |
| MIL vs. placebo [140] | Hoffman et al. (2003) [140]/RCT. | 238 neonates and children (aged 2 days to 6.9 years) in high-risk for LCOS after corrective cardiac surgery. | High-doses of MIL reduces the risk of LCOS after cardiac surgery. |
| MIL vs. PL [145] | Paradisis et al. (2009) [145]/RCT. | 90 infants (GA < 30 weeks; age < 6 h) in high risk of low SVC flow. | MIL did not prevent low systemic blood flow during the first 24 h. MIL had higher HR. No dif. in AP, inotrope use, PIVH, other clinical outcomes, and mortality or side effects. |
| MIL vs. no prophylaxis [136] | Halliday et al. (2017) [136]/Retrospective study. | 45 preterm neonates (GA 23–26 weeks) subjected to PDA ligation. | MIL-Gr had higher AP. No dif. in inotrope and hydrocortisone use, or clinical outcomes. Prophylactic after PDA ligation does not sign. affect CV stability or long-term outcome. |
| MIL vs. LEVO [183] | Momeni et al. (2011) [183]/RCT. | 36 infants and children (age range 7–977 d) operated for CHD. | LEVO-Gr had lower myocardial oxygen demands and troponin levels postoperatively. LEVO is at least as efficacious as MIL. |
| MIL vs. LEVO [150] | Lechner et al. (2012) [150]/RCT; prophylactic LEVO vs. MIL to prevent LCOS. | 40 term infants undergoing repair of CHD. | Postoperative cardiac output and index were similar in LEVO vs. MIL. Improvement in cardiac function in the MIL-Gr. Both drugs were well tolerated; no death or serious adverse event. |
| MIL vs. LEVO [36] | Pellicer et al. (2013) [36]/RCT. | 20 term neonates undergoing surgical repair for CHD. | MIL Gr had lower pH and higher blood glucose and inotrope score. Study drug withdrawal at 96 h was more frequent in LEVO-Gr. LEVO is well tolerated and may have advantages over MIL in terms of the dosing regimen. |
| LEVO vs. standard inotrope treatment [93] | Ricci et al. (2012) [93]/RCT. | 63 neonates (<30 days) at risk of low SVC flow post-surgery for CHD. | The occurrence of LCOS, HR, lactate levels, and inotrope score were sign. lower in LEVO Gr. No dif. in mortality and clinical outcomes. LEVO was well tolerated with a potential benefit on postoperative hemodynamic and metabolic parameters. |
| LEVO vs. standard inotrope treatment [186] | Hummel et al. (2017) [186]/Cochrane. | Five RCTs with a total of 212 neonates and children under 5 years undergoing surgery for CHD. | LEVO showed no clear effect on mortality and clinical outcomes. Current evidence is insufficient to suggest LEVO for prevention of LCOS and mortality. |
| Hydrocortisone vs. placebo as rescue treatment [98] | Ng et al. (2006) [98]/RCT. | 48 VLBW infants with refractory AH. | A stress dose of hydrocortisone was effective in treating refractory AH in VLBW infants. However, routine corticosteroids could not be recommended because of their potential adverse effects. |
| Hydrocortisone vs. placebo as rescue treatment [97] | Kovacs et al. (2019) [97]/RCT. | 35 cooled asphyxiated term neonates with volume-resistant AH. | Hydrocortisone use effectively increased the AP and decreased the inotrope needs in cooled asphyxiated neonates with resistant AH. |
| Hydrocortisone vs. placebo [194] | Ando et al. (2005) [194]/RCT. | Twenty neonates (age < 28 days) undergoing biventricular repair. | Hydrocortisone improved hemodynamic profile and decreased the inotrope score without increasing the risk of complications. Adrenal insufficiency may occur after neonatal open-heart surgery. |
| Dexamethasone vs. placebo as adjuvant therapy [231] | Gaissmaier et al. (1999) [231]/RCT. | 20 neonates (GA 25–36 vertical alignment) < 1 mo, who with inotrope-refractory AH. | DXM was effective for the management of severe AH in preterm infants not responding to standardized treatment. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agakidou, E.; Chatziioannidis, I.; Kontou, A.; Stathopoulou, T.; Chotas, W.; Sarafidis, K. An Update on Pharmacologic Management of Neonatal Hypotension: When, Why, and Which Medication. Children 2024, 11, 490. https://doi.org/10.3390/children11040490
Agakidou E, Chatziioannidis I, Kontou A, Stathopoulou T, Chotas W, Sarafidis K. An Update on Pharmacologic Management of Neonatal Hypotension: When, Why, and Which Medication. Children. 2024; 11(4):490. https://doi.org/10.3390/children11040490
Chicago/Turabian StyleAgakidou, Eleni, Ilias Chatziioannidis, Angeliki Kontou, Theodora Stathopoulou, William Chotas, and Kosmas Sarafidis. 2024. "An Update on Pharmacologic Management of Neonatal Hypotension: When, Why, and Which Medication" Children 11, no. 4: 490. https://doi.org/10.3390/children11040490
APA StyleAgakidou, E., Chatziioannidis, I., Kontou, A., Stathopoulou, T., Chotas, W., & Sarafidis, K. (2024). An Update on Pharmacologic Management of Neonatal Hypotension: When, Why, and Which Medication. Children, 11(4), 490. https://doi.org/10.3390/children11040490







