Avoidance Measures for Patients with Allergic Rhinitis: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
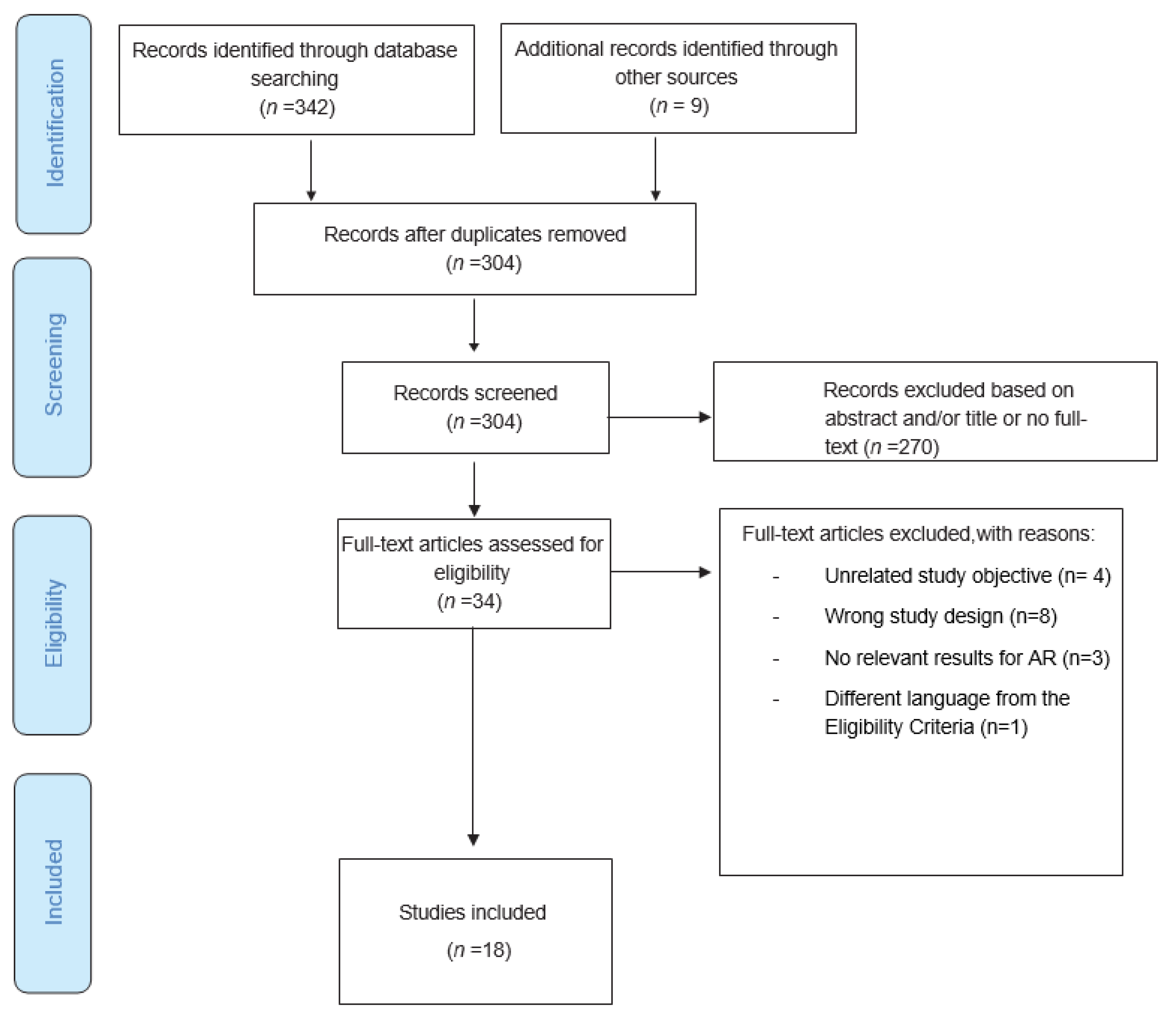
3. Results
3.1. Environmental Control Measures
3.2. Individual Control Measures
3.3. Combined Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Greiner, A.N.; Hellings, P.W.; Rotiroti, G.; Scadding, G.K. Allergic rhinitis. Lancet 2011, 378, 2112–2122. [Google Scholar] [CrossRef]
- Caldeira, L.E.; Silva, M.I.T.; Martins-Dos-santos, G.; Pereira, A.M. Allergic rhinitis—Classification, pathophysiology, diagnosis and treatment. Rev. Port. Imunoalergol. 2021, 29, 95–106. [Google Scholar] [CrossRef]
- Cox, L. Approach to Patients with Allergic Rhinitis: Testing and Treatment. Med. Clin. 2020, 104, 77–94. [Google Scholar] [CrossRef]
- Bernstein, D.I.; Schwartz, G.; Bernstein, J.A. Allergic Rhinitis: Mechanisms and Treatment. Immunol. Allergy Clin. 2016, 36, 261–278. [Google Scholar] [CrossRef]
- Schuler, C.F., IV; Montejo, J.M. Allergic Rhinitis in Children and Adolescents. Pediatr. Clin. N. Am. 2019, 66, 981–993. [Google Scholar] [CrossRef]
- Andiappan, A.K.; Puan, K.J.; Lee, B.; Nardin, A.; Poidinger, M.; Connolly, J.; Chew, F.T.; Wang, D.Y.; Rotzschke, O.; Wang, D.-Y. Allergic airway diseases in a tropical urban environment are driven by dominant mono-specific sensitization against house dust mites. Allergy 2014, 69, 501–509. [Google Scholar] [CrossRef]
- Toppila-Salmi, S.; Huhtala, H.; Karjalainen, J.; Renkonen, R.; Mäkelä, M.J.; Wang, D.Y.; Pekkanen, J. Sensitization pattern affects the asthma risk in Finnish adult population. Allergy 2015, 70, 1112–1120. [Google Scholar] [CrossRef]
- Siroux, V.; Ballardini, N.; Soler, M.; Lupinek, C.; Boudier, A.; Pin, I.; Just, J.; Nadif, R.; Anto, J.M.; Melen, E.; et al. The asthma-rhinitis multimorbidity is associated with IgE polysensitization in adolescents and adults. Allergy 2018, 73, 1447–1458. [Google Scholar] [CrossRef]
- Bush, R.K. Indoor allergens, environmental avoidance, and allergic respiratory disease. Allergy Asthma Proc. 2016, 29, 575–579. [Google Scholar] [CrossRef]
- Platts-Mills, T.A.E. Allergen Avoidance in the Treatment of Asthma and Rhinitis. N. Engl. J. Med. 2003, 349, 207–208. [Google Scholar] [CrossRef]
- Burbach, G.J.; Heinzerling, L.M.; Edenharter, G.; Bachert, C.; Bindslev-Jensen, C.; Bonini, S.; Bousquet, J.; Bousquet-Rouanet, L.; Bousquet, P.J.; Bresciani, M.; et al. GA(2)LEN skin test study II: Clinical relevance of inhalant allergen sensitizations in Europe. Allergy 2009, 64, 1507–1515. [Google Scholar] [CrossRef]
- Bousquet, J.; Pfaar, O.; Togias, A.; Schünemann, H.J.; Ansotegui, I.; Papadopoulos, N.G.; Tsiligianni, I.; Agache, I.; Anto, J.M.; Bachert, C.; et al. 2019 ARIA Care pathways for allergen immunotherapy. Allergy Eur. J. Allergy Clin. Immunol. 2019, 74, 2087–2102. [Google Scholar] [CrossRef]
- Peters, M.D.; Godfrey, C.M.; Khalil, H.; McInerney, P.; Parker, D.; Soares, C.B. Guidance for conducting systematic scoping reviews. JBI Evid. Implement. 2015, 13, 141–146. [Google Scholar] [CrossRef]
- Kniest, F.M.; Young, E.; Van Praag, M.C.G.; Vos, H.; Kort, H.S.M.; Koers, W.J.; De Maat-Bleeker, F.; Van Bronswijk, J.E.M.H. Clinical evaluation of a double-blind dust-mite avoidance trial with mite-allergic rhinitic patients. Clin. Exp. Allergy 1991, 21, 39–47. [Google Scholar] [CrossRef]
- Terreehorst, I.; Hak, E.; Oosting, A.J.; Tempels-Pavlica, Z.; de Monchy, J.G.R.; Bruijnzeel-Koomen, C.A.F.M.; Aalberse, R.C.; Gerth van Wijk, R. Evaluation of Impermeable Covers for Bedding in Patients with Allergic Rhinitis. N. Engl. J. Med. 2003, 349, 237–246. [Google Scholar] [CrossRef]
- Li, L.; Zhang, L.; Mo, J.H.; Li, Y.Y.; Xia, J.Y.; Bai, X.B.; Xie, P.F.; Liang, J.Y.; Yang, Z.F.; Chen, Q.Y. Efficacy of indoor air purification in the treatment of Artemisia pollen-allergic rhinitis: A randomised, double-blind, clinical controlled trial. Clin. Otolaryngol. 2020, 45, 394–401. [Google Scholar] [CrossRef]
- Moon, J.S.; Choi, S.O. Environmental Controls in Reducing House Dust Mites and Nasal Symptoms in Patients with Allergic Rhinitis. Yonsei Med. J. 1999, 40, 238–243. [Google Scholar] [CrossRef]
- Mohan, L.; Hanna, H.; Warner, J.; Boyle, R. Effects of temperature-controlled laminar airflow on sleep quality in children with perennial allergic asthma and rhinitis. Allergy 2011, 66, 74–75. [Google Scholar]
- O’Meara, T.J.; Sercombe, J.K.; Morgan, G.; Reddel, H.K.; Xuan, W.; Tovey, E.R. The reduction of rhinitis symptoms by nasal filters during natural exposure to ragweed and grass pollen. Allergy 2005, 60, 529–532. [Google Scholar] [CrossRef]
- Ojeda, P.; Pique, N.; Alonso, A.; Delgado, J.; Feo, F.; Igea, J.M.; Navarro, A.; Olaguibel, J.M.; Subiza, J.; Nieto, C.; et al. A topical microemulsion for the prevention of allergic rhinitis symptoms: Results of a randomized, controlled, double-blind, parallel group, multicentre, multinational clinical trial (Nares study). Allergy Asthma Clin. Immunol. 2013, 9, 32. [Google Scholar] [CrossRef]
- Stillerman, A.; Nachtsheim, C.; Li, W.; Albrecht, M.; Waldman, J. Efficacy of a novel air filtration pillow for avoidance of perennial allergens in symptomatic adults. Ann. Allergy Asthma Immunol. 2010, 104, 440–449. [Google Scholar] [CrossRef]
- Kenney, P.; Hilberg, O.; Laursen, A.C.; Peel, R.G.; Sigsgaard, T. Preventive effect of nasal filters on allergic rhinitis: A randomized, double-blind, placebo-controlled crossover park study. J. Allergy Clin. Immunol. 2015, 136, 1566–1572.e5. [Google Scholar] [CrossRef]
- Berings, M.; Jult, A.; Vermeulen, H.; De Ruyck, N.; Derycke, L.; Ucar, H.; Ghekiere, P.; Temmerman, R.; Ellis, J.; Bachert, C.; et al. Probiotics-impregnated bedding covers for house dust mite allergic rhinitis: A pilot randomized clinical trial. Clin. Exp. Allergy 2017, 47, 1092–1096. [Google Scholar] [CrossRef]
- Chen, M.; Wu, Y.; Yuan, S.; Tang, M.; Zhang, L.; Chen, J.; Li, L.; Wu, J.; Zhang, J.; Yin, Y. Allergic Rhinitis Improvement in Asthmatic Children After Using Acaricidal Bait: A Randomized, Double-Blind, Cross-Placebo Study. Front. Pediatr. 2021, 9, 1015. [Google Scholar] [CrossRef] [PubMed]
- Brehler, R.; Kütting, B.; Biel, K.; Luger, T. Positive effects of a fresh air filtration system on hay fever symptoms. Int. Arch. Allergy Immunol. 2003, 130, 60–65. [Google Scholar] [CrossRef]
- Andersson, M.; Greiff, L.; Wollmer, P. Effects of a Topical Microemulsion in House Dust Mite Allergic Rhinitis. Basic Clin. Pharmacol. Toxicol. 2011, 108, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Reisman, R.E.; Mauriello, P.M.; Davis, G.B.; Georgitis, J.W.; DeMasi, J.M. A double-blind study of the effectiveness of a high-efficiency particulate air (HEPA) filter in the treatment of patients with perennial allergic rhinitis and asthma. J. Allergy Clin. Immunol. 1990, 85, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Antonicelli, L.; Bilò, M.B.; Pucci, S.; Schou, C.; Bonifazi, F. Efficacy of an air-cleaning device equipped with a high efficiency particulate air filter in house dust mite respiratory allergy. Allergy 1991, 46, 594–600. [Google Scholar] [CrossRef]
- Jeon, Y.H.; Lee, Y.J.; Sohn, M.H.; Lee, H.R. Effects of Vacuuming Mattresses on Allergic Rhinitis Symptoms in Children. Allergy Asthma Immunol. Res. 2019, 11, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Manuyakorn, W.; Padungpak, S.; Luecha, O.; Kamchaisatian, W.; Sasisakulporn, C.; Vilaiyuk, S.; Monyakul, V.; Benjaponpitak, S. Assessing the efficacy of a novel temperature and humidity control machine to minimize house dust mite allergen exposure and clinical symptoms in allergic rhinitis children sensitized to dust mites: A pilot study. Asian Pac. J. Allergy Immunol. 2015, 33, 129–135. [Google Scholar] [CrossRef]
- Incorvaia, C.; Yacoub, M.R.; Rapetti, A.; Zapelli, I.; Qualizza, R.; Brame, B. Do environmental measures improve quality of life in mite-induced allergic rhinitis? Ital. J. Allergy Clin. Immunol. 2008, 18, 18–21. Available online: https://www.researchgate.net/publication/286613670_Do_environmental_measures_improve_quality_of_life_in_mite-induced_allergic_rhinitis (accessed on 7 November 2022).
- Gøtzsche, P.C.; Johansen, H.K. House dust mite control measures for asthma: Systematic review. Allergy Eur. J. Allergy Clin. Immunol. 2008, 63, 646–659. [Google Scholar] [CrossRef]
- Bumbacea, R.; Corcea, S.; Ali, S.; Dinica, L.; Fanfaret, I.; Boda, D. Mite allergy and atopic dermatitis: Is there a clear link? Exp. Ther. Med. 2020, 20, 3554–3560. Available online: https://pubmed.ncbi.nlm.nih.gov/32905207/ (accessed on 30 January 2022).
- Tam, H.; Calderon, M.A.; Manikam, L.; Nankervis, H.; García Núñez, I.; Williams, H.C.; Durham, S.; Boyle, R.J. Specific allergen immunotherapy for the treatment of atopic eczema. Cochrane Database Syst. Rev. 2016, 2, CD008774. [Google Scholar] [CrossRef]
- Sheikh, A.; Hurwitz, B. House dust mite avoidance measures for perennial allergic rhinitis. Cochrane Database Syst. Rev. 2001, 67, 158–165. [Google Scholar]
- Nurmatov, U.; Van Schayck, C.P.; Hurwitz, B.; Sheikh, A. House dust mite avoidance measures for perennial allergic rhinitis: An updated Cochrane systematic review. Allergy Eur. J. Allergy Clin. Immunol. 2012, 67, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Dror, A.A.; Eisenbach, N.; Marshak, T.; Layous, E.; Zigron, A.; Shivatzki, S.; Morozov, N.G.; Taiber, S.; Alon, E.E.; Ronen, O.; et al. Reduction of allergic rhinitis symptoms with face mask usage during the COVID-19 pandemic. J Allergy Clin. Immunol. Pract. 2020, 8, 3590–3593. [Google Scholar] [CrossRef]
- Lin, J.S.; Tan, B.; Yeh, C.; Kern, R.; Conley, D.; Welch, K.; Peters, A.; Smith, S. Comparing the severity of chronic rhinosinusitis symptoms before versus during the COVID-19 pandemic. Laryngoscope Investig. Otolaryngol. 2022, 7, 1704. [Google Scholar] [CrossRef]
| Author, Year | Study Design | Patient Group (n; Age (mean = ±std)) | Intervention(s) | Outcomes Evaluated | Results |
|---|---|---|---|---|---|
| Andersson M, 2011 [26] | Randomized, double-blinded, crossover trial | 20 patients with PAR sensitized to HDM Age = (19–48) | Nasal topical microemulsion | Nasal symptoms score (four-point scale) TNSS | . reduction in nasal symptoms with the microemulsion from baseline . reduction in symptoms when comparing microemulsion and placebo on treatment days 3, 4, and 6 |
| Antonicelli L, 1991 [28] | Randomized cross-over study | 9 patients with AR sensitized to HDM Age = 16 yrs (std not given) | HEPA filter | HDM (Der p1, Der f1 and Der m1) allergen level rhinitis and asthma symptom scores | . no significant difference in floor allergen levels with HEPA filter use . no difference in rhinitis symptom scores |
| Berings M, 2017 [23] | Randomized, double-blind, placebo-controlled, crossover trial | 24 patients with AR sensitized to HDM Age = not given. | Probiotics-impregnated bed covers | Der p1 levels in mattress and pillow dust samples Symptoms score (VAS) QoL (RQLQ and NRQLQ) Medication use | . no difference in Der p1 levels between groups . significant improvement compared to the baseline of symptoms and QoL scores in the intervention group . no significant changes in medication use |
| Brehler R, 2003 [25] | Placebo-controlled, double-blind, crossover study | 44 patients with seasonal AR, allergic conjunctivitis, and asthma Age = not given | Active filter was used | Symptom score PEFR Medication use ECP levels | . reductions in symptoms, use of medication, and increase in PEFR were bigger in volunteers with exclusively hay fever. . no effect on ECP levels |
| Chen M, 2021 [24] | Randomized, double-blind, placebo-controlled, crossover study | 50 patients with AR and asthma sensitized to HDM Age = 6 (std not given) | Acaricidal bait | Symptoms score (VAS) AR control (RCAT) Asthma control (ACQ-5) QoL (RQLQ) HDM allergen levels | . significant improvement in VAS, RCAT, and RQLQ scores in the acaricidal bait . no significant improvement in ACQ-5 . HDM allergen levels significantly decreased |
| Incorvaia C, 2008 [31] | Randomized placebo-controlled 2 x 2 factorial trial | 25 patients with persistent AR sensitized to HDM IG: 12 CG: 13 Age = not given. | Bed casings + acaricide | QoL (RQLQ) | . significant improvement in RQLQ compared to the baseline . no difference when compared to the placebo |
| Jeon YH, 2019 [29] | Single-blind, randomized parallel trial | 40 patients with mild PAR sensitized only to HDM IG: 20; 8.90 ± 2.2 yrs CG: 20; 8.45 ± 2.2 yrs | Daily vacuuming of mattresses | Symptoms score (VAS) dust weight and concentration of Der p1 and Der f1 | . significant improvement in all symptom scores after 2 weeks in the intervention group . reduction in the collected dust weight . no significant change in Der p1 and Der f1 concentrations |
| Kenney P, 2015 [22] | Randomized, double-blind, placebo-controlled crossover clinical trial | 65 AR patients sensitized to grass pollen Age = 24.8 ± 6.1 yrs | Nasal filters | TNSS TOSS FEV1 | . significant reduction in daily TNSS and maximum TNSS compared with placebo . no difference in daily TOSS . no difference in FEV1 between placebo and filter |
| Kniest F, 1991 [14] | Double-blind matched pair controlled trial | 20 patients with PAR sensitized to HDM IG: 10; 19 yrs (std not given) CG: 10; 21.5 yrs (std not given) | intensive home cleaning with or without acaricide (solidified benzyl benzoate) | Daily symptom score Medication scores Physician assessment Total and mite-specific serum IgE Nose eosinophils Guanine exposure | . reduction in daily symptoms and total IgE with the use of acaricide . no effects with intensive cleaning ‘only’ |
| Li L, 2020 [16] | Randomized, double-blind, controlled trial | 90 AR patients sensitized to Artemisia pollen IG: 45; 35.5 ± 8,2 yrs CG: 45; 36.1 ± 9,2 yrs | HEPA filter | Symptom severity (VAS) QoL (VAS) RQLQ Epworth Sleepiness Scale | . rhinitis symptom scores showed significant differences between IG and CG . no differences in RQLQ between groups . nasal symptom score, allergy symptom score (VAS), Epworth Sleepiness Scale score decreased progressively each week |
| Manuyakorn W, 2015 [30] | Observational study | 7 Patients with persistent AR who were skin prick test positive for HDM Age = 9.8 ± 1,7 yrs. | Use of a temperature and humidity control machine | TNSS and the level of dust mite allergens (Der p1 and Der f1) | There was a significant reduction in TNSS at 2 and 4 months and 70% of the patients were able to stop using their intranasal corticosteroids by the end of the experiment. There was a notable reduction in the levels of Der f1 as early as 2 months after installing the machine but this reduction became significant only after 4 months and Der f 1 level remained low until the end of the experiment. |
| Mohan L, 2011 [18] | Randomized control-trial | 52 patients with allergic asthma and rhinitis sensitized to a perennial allergen IG = 36 CG = 16 Age = 8–16 | Nocturnal temperature-controlled laminar airflow (TLA) | Sleep quality (PSQ; CSHQ and wristwatch actigraphy | . no significant improvement in sleep quality |
| Moon JS, 1999 [17] | Open randomized controlled trial | 30 patients with AR sensitized to HDM IG: 15 CG: 15 Age = 15.6 yrs (for total patients) | Bed covers + vinyl mattress cover * daily wet cleaning of floors + fortnight washing of top bedding cover + removal of soft furnishings | Daily rhinitis symptoms scores HDM load | . HDM loads were significantly reduced in the intervention group compared to the control group . significant improvement in symptoms scores in the intervention group |
| Ojeda P, 2013 [20] | Randomized controlled double-blind clinical trial | 110 patients with moderate to severe AR or rhino-conjunctivitis sensitized to grass, birch, or olive tree pollens IG: 55; 32.6 ± 9.9 yrs CG: 55; 34.9 ± 11.5 yrs | Nasal topical microemulsion | QoL (RQLQ) Nasal, ocular, and lung symptoms Medication usage | . reduction in symptoms in the intervention group . improvement in QoL although without statistically significant differences between groups |
| O’Meara TJ, 2005 [19] | Double-blind placebo-controlled trial | 46 patients with AR sensitized to mixed ragweed IG: 22; 51.1 yrs (std not given) CG: 24; 50.3 yrs (std not given) | Nasal filters | Symptoms score (MSC and TSC) Peak nasal inspiratory flow | . MSC scores decreased in the intervention group and increased in the placebo group compared with baseline scores . TSC scores decreased in the intervention group when compared with the placebo group . no difference between groups in peak nasal inspiratory flow |
| Reisman R, 1990 [27] | Double-blind cross-over randomized controlled trial | 32 patients with PAR and/or asthma sensitized to HDM Age = 27.5 yrs (std not given) | HEPA filter | Particulate counts in bedroom air Symptom score Medication score Patient’s subjective response to treatment | . reduction in particulate counts in bedroom air . reduction in symptom and medication scores |
| Stillerman A et al., 2010 [21] | Randomized, double-blind, placebo-controlled, crossover trial | 35 PARC patients sensitized to HDM, dog or cat Age = 39.1 ± 11.9 yrs | HEPA filter + pillow encasement | TSS QoL (NRQLQ) | . significant improvements in TSS and QoL |
| Terreehorst I, 2003 [15] | Double-blind randomized controlled trial | 232 patients with AR and/or asthma IG: 114; 25.7 ± 1.1 yrs CG: 118; 26.9 ± 1.1 yrs | Bed covers | Daily symptom score (VAS) Nasal allergen-provocation test score Der p1 and Der f1 concentration | . concentrations of Der p1 and Der f1 in the mattress sample was significantly lower in the impermeable cover group when compared with the control group. . no significant difference between groups on daily symptom score, or nasal allergen provocation test |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomé, M.; Lourenço, O. Avoidance Measures for Patients with Allergic Rhinitis: A Scoping Review. Children 2023, 10, 300. https://doi.org/10.3390/children10020300
Tomé M, Lourenço O. Avoidance Measures for Patients with Allergic Rhinitis: A Scoping Review. Children. 2023; 10(2):300. https://doi.org/10.3390/children10020300
Chicago/Turabian StyleTomé, Miguel, and Olga Lourenço. 2023. "Avoidance Measures for Patients with Allergic Rhinitis: A Scoping Review" Children 10, no. 2: 300. https://doi.org/10.3390/children10020300
APA StyleTomé, M., & Lourenço, O. (2023). Avoidance Measures for Patients with Allergic Rhinitis: A Scoping Review. Children, 10(2), 300. https://doi.org/10.3390/children10020300










