Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome
Abstract
1. Introduction
2. Marfan’s Syndrome (MFS)
2.1. Cause of Marfan’s Syndrome
2.2. Marfan’s Syndrome Manifestations
3. Oxidative Stress
4. Aortic Aneurysm Formation in Marfan’s Syndrome and the Role of Oxidative Stress
4.1. Aortic Aneurysm Formation in Marfan’s Syndrome
4.2. Role of Oxidative Stress in Aortic Aneurysm Formation
5. Oxidative Stress-Reducing Strategies
5.1. Resveratrol
5.2. n-3 polyunsaturated fatty acids (PUFAs) (n-3 PUFA): Docosahexaenoic and Lipoic Acids
5.3. Hibiscus sabdariffa Linne (HSL)
5.4. Pharmacological Management of MFS
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marx, J.; Izumo, S.; Nadal-Ginard, B.; Mahdavi, V. Oxygen free radicals linked to many diseases. Science 1987, 235, 529–531. [Google Scholar] [CrossRef] [PubMed]
- Griendling, K.K.; Sorescu, D.; Ushio-Fukai, M. NAD (P) H oxidase: Role in cardiovascular biology and disease. Circ. Res. 2000, 86, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.Y.; Griendling, K. Redox signaling, vascular function, and hypertension. Antioxid. Redox Signal. 2008, 10, 1045–1059. [Google Scholar] [CrossRef] [PubMed]
- Faraci, F.M.; Didion, S.P. Vascular protection: Superoxide dismutase isoforms in the vessel wall. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.E.; Soria-Castro, E.; Guarner-Lans, V.; Ontiveros, E.M.; Mejía, B.I.H.; Hernandez, H.J.M.; García, R.B.; Herrera, V.; Pérez-Torres, I. Analysis of oxidative stress enzymes and structural and functional proteins on human aortic tissue from different aortopathies. Oxidative Med. Cell Longev. 2014, 2014, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Schulz, E.; Anter, E.; Keaney, K., Jr. Oxidative stress, antioxidants, and endothelial function. Curr. Med. Chem. 2004, 11, 1093–1104. [Google Scholar] [CrossRef]
- Cai, H.; Harrison, D.G. Endothelial dysfunction in cardiovascular diseases: The role of oxidant stress. Circ. Res. 2000, 87, 840–844. [Google Scholar] [CrossRef]
- Boullier, A.; Bird, D.A.; Chang, M.-K.; Dennis, E.A.; Friedman, P.; Gillotte-Taylor, K.; Hörkkö, S.; Palinski, W.; Quehenberger, O.; Shaw, P.; et al. Scavenger receptors, oxidized LDL, and atherosclerosis. Ann. N. Y. Acad. Sci. 2006, 947, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.C.; van Breemen, C.; Chung, A.W. Vasomotor dysfunction in the thoracic aorta of Marfan syndrome is associated with accumulation of oxidative stress. Vasc. Pharmacol. 2010, 52, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Maiellaro-Rafferty, K.; Weiss, D.; Joseph, G.; Wan, W.; Gleason, R.L.; Taylor, W.R. Catalase overexpression in aortic smooth muscle prevents pathological mechanical changes underlying abdominal aortic aneurysm formation. Am. J. Physiol. Circ. Physiol. 2011, 301, H355–H362. [Google Scholar] [CrossRef]
- Zhang, P.-Y.; Xu, X.; Li, X.-C. Cardiovascular diseases: Oxidative damage and antioxidant protection. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3091–3096. [Google Scholar] [PubMed]
- Judge, D.; Dietz, H.C. Marfan’s syndrome. Lancet 2005, 366, 1965–1976. [Google Scholar] [CrossRef]
- Kinoshita, N.; Mimura, J.; Obayashi, C.; Katsukawa, F.; Onishi, S.; Yamazaki, H. Aortic root dilatation among young competitive athletes: Echocardiographic screening of 1929 athletes between 15 and 34 years of age. Am. Heart J. 2000, 139, 723–728. [Google Scholar] [CrossRef]
- Collod-Béroud, G.; Le Bourdelles, S.; Ades, L.; Ala-Kokko, L.; Booms, P.; Boxer, M.; Child, A.; Comeglio, P.; De Paepe, A.; Hyland, J.C.; et al. Update of the UMD-FBN1mutation database and creation of anFBN1polymorphism database. Hum. Mutat. 2003, 22, 199–208. [Google Scholar] [CrossRef]
- Dietz, H.C.; Cutting, C.R.; Pyeritz, R.E.; Maslen, C.L.; Sakai, L.Y.; Corson, G.M.; Puffenberger, E.; Hamosh, A.; Nanthakumar, E.J.; Curristin, S.M.; et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature 1991, 352, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, D.P.; Ono, R.N.; Sakai, L.Y. Calcium Stabilizes Fibrillin-1 against Proteolytic Degradation. J. Biol. Chem. 1997, 272, 1231–1236. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, D.P.; Mechling, D.E.; Boswell, B.A.; Keene, D.R.; Sakai, L.Y.; Bächinger, H.P. Calcium Determines the Shape of Fibrillin. J. Biol. Chem. 1997, 272, 7368–7373. [Google Scholar] [CrossRef]
- De la Fuente-Alonso, A.; Toral, M.; Alfayate, A.; Ruiz-Rodríguez, M.J.; Bonzón-Kulichenko, E.; Teixido-Tura, G.; Martínez-Martínez, S.; Méndez-Olivares, M.J.; López-Maderuelo, D.; González-Valdés, I.; et al. Aortic disease in Marfan syndrome is caused by overactivation of sGC-PRKG signaling by NO. Nat. Commun. 2021, 12, 1–18. [Google Scholar] [CrossRef]
- Cañadas, V.; Vilacosta, I.; Bruna, I.; Fuster, V. Marfan syndrome Part 1: Pathophysiology and diagnosis. Nat. Rev. Cardiol. 2010, 7, 256–265. [Google Scholar] [CrossRef]
- Yuan, S.-M.; Jing, H. Cystic medial necrosis: Pathological findings and clinical implications. Braz. J. Cardiovasc. Surg. 2011, 26, 107–115. [Google Scholar] [CrossRef]
- Chung, A.W.Y.; Yeung, K.A.; Cortes, S.; Sandor, G.; Judge, D.; Dietz, H.C.; Van Breemen, C. Endothelial dysfunction and compromised eNOS/Akt signaling in the thoracic aorta during the progression of Marfan syndrome. Br. J. Pharmacol. 2007, 150, 1075–1083. [Google Scholar] [CrossRef]
- Bellini, C.; Bersi, M.; Caulk, A.W.; Ferruzzi, J.; Milewicz, D.M.; Ramirez, F.; Rifkin, D.B.; Tellides, G.; Yanagisawa, H.; Humphrey, J.D. Comparison of 10 murine models reveals a distinct biomechanical phenotype in thoracic aortic aneurysms. J. R. Soc. Interface 2017, 14, 20161036. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, J.D.; Tellides, G. Central artery stiffness and thoracic aortopathy. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H169–H182. [Google Scholar] [CrossRef] [PubMed]
- Mizuguchi, T.; Collod-Beroud, G.; Akiyama, T.; Abifadel, M.; Harada, N.; Morisaki, T.; Allard, D.; Varret, M.; Claustres, M.; Morisaki, H.; et al. Heterozygous TGFBR2 mutations in Marfan syndrome. Nat. Genet. 2004, 36, 855–860. [Google Scholar] [CrossRef]
- Singh, K.K.; Rommel, K.; Mishra, A.; Karck, M.; Haverich, A.; Schmidtke, J.; Arslan-Kirchner, M. TGFBR1andTGFBR2mutations in patients with features of Marfan syndrome and Loeys-Dietz syndrome. Hum. Mutat. 2006, 27, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.-C.; Chen, C.-M.; Hamdin, C.D.; Orekhov, A.N.; Sobenin, I.A.; Layne, M.D.; Yet, S.-F. Therapeutic Potential of Heme Oxygenase-1 in Aneurysmal Diseases. Antioxidants 2020, 9, 1150. [Google Scholar] [CrossRef]
- Zúñiga-Muñoz, A.M.; Pérez-Torres, I.; Guarner-Lans, V.; Núñez-Garrido, E.; Espejel, R.V.; Huesca-Gómez, C.; Gamboa-Ávila, R.; Soto, M.E. Glutathione system participation in thoracic aneurysms from patients with Marfan syndrome. Vasa 2017, 46, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Mandelbaum, K.; Pereira, L.; Perez, A. Candidate gene linkage analysis indicates genetic heterogeneity in Marfan syndrome. Braz. J. Med. Biol. Res. 2011, 44, 793–800. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kainulainen, K.; Steinmann, B.; Collins, F.; Dietz, H.C.; Francomano, C.A.; Child, A.; Kilpatrick, M.W.; Brock, D.J.H.; Keston, M.; Pyeritz, R.E.; et al. Marfan syndrome: No evidence for heterogeneity in different populations, and more precise mapping of the gene. Am. J. Hum. Genet. 1991, 49, 662–667. [Google Scholar]
- Tsipouras, P.; Del Mastro, R.; Sarfarazi, M.; Lee, B.; Vitale, E.; Child, A.H.; Godfrey, M.; Devereux, R.B.; Hewett, D.; Steinmann, B.; et al. Genetic linkage of the marfan syndrome, ectopia lentis, and congenital contractural arachnodactyly to the fibrillin genes on chromosomes 15 and 5. N. Engl. J. Med. 1992, 326, 905–909. [Google Scholar] [CrossRef]
- Mckusick, V.A. The cardiovascular aspects of Marfan’s syndrome: A heritable disorder of connective tissue. Circulation 1955, 11, 321–342. [Google Scholar] [CrossRef] [PubMed]
- Pyeritz, R.E.; Wappel, M.A. Mitral valve dysfunction in the Marfan syndrome: Clinical and echocardiographic study of prevalence and natural history. Am. J. Med. 1983, 74, 797–807. [Google Scholar] [CrossRef]
- Roberts, W.C.; Honig, H.S. The spectrum of cardiovascular disease in the Marfan syndrome: A clinico-morphologic study of 18 necropsy patients and comparison to 151 previously reported necropsy patients. Am. Hear. J. 1982, 104, 115–135. [Google Scholar] [CrossRef]
- Hagerty, T.; Geraghty, P.; Braverman, A.C. Abdominal aortic aneurysm in Marfan syndrome. Ann. Vasc. Surg. 2017, 40, 294.e1–294.e6. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, Q.; Zhu, J.; Xiao, Q.; Zhang, L. Reactive oxygen species: Key regulators in vascular health and diseases. Br. J. Pharmacol. 2017, 175, 1279–1292. [Google Scholar] [CrossRef] [PubMed]
- Lassègue, B.; Griendling, K. NADPH Oxidases: Functions and pathologies in the vasculature. Arter. Thromb. Vasc. Biol. 2010, 30, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Drummond, G.R.; Dusting, G.J. Suppression of Oxidative Stress in the Endothelium and Vascular Wall. Endothelium 2004, 11, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Inoue, N.; Kawashima, S. Role of the vascular NADH/NADPH oxidase system in atherosclerosis. Ann. N. Y. Acad. Sci. 2006, 902, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Lounsbury, K.M.; Hu, Q.; Ziegelstein, R.C. Calcium signaling and oxidant stress in the vasculature. Free Radic. Biol. Med. 2000, 28, 1362–1369. [Google Scholar] [CrossRef]
- Walia, M.; Sormaz, L.E.; Samson, S.; Lee, R.M.; Grover, A.K. Effects of hydrogen peroxide on pig coronary artery endothelium. Eur. J. Pharmacol. 2000, 400, 249–253. [Google Scholar] [CrossRef]
- Lyle, A.; Griendling, K. Modulation of vascular smooth muscle signaling by reactive oxygen species. Physiology 2006, 21, 269–280. [Google Scholar] [CrossRef]
- Zanetti, M.; Sato, J.; Katusic, Z.S.; O’Brien, T. Gene transfer of superoxide dismutase isoforms reverses endothelial dysfunction in diabetic rabbit aorta. Am. J. Physiol. Circ. Physiol. 2001, 280, H2516–H2523. [Google Scholar] [CrossRef]
- Xu, J.; Wang, S.; Zhang, M.; Wang, Q.; Asfa, S.; Zou, M.-H. Tyrosine nitration of PA700 links proteasome activation to endothelial dysfunction in mouse models with cardiovascular risk factors. PLoS ONE 2012, 7, e29649. [Google Scholar] [CrossRef]
- MacMillan-Crow, L.A.; Cruthirds, D.L. Manganese superoxide dismutase in disease. Free Radic. Res. 2001, 34, 325–336. [Google Scholar] [CrossRef]
- Visner, G.A.; Chesrown, S.E.; Monnier, J.; Ryan, U.S.; Nick, H.S. Regulation of manganese superoxide dismutase: IL-1 and TNF induction in pulmonary artery and microvascular endothelial cells. Biochem. Biophys. Res. Commun. 1992, 188, 453–462. [Google Scholar] [CrossRef]
- Ülker, S.; McMaster, D.; McKeown, P.P.; Bayraktutan, U. Impaired activities of antioxidant enzymes elicit endothelial dysfunction in spontaneous hypertensive rats despite enhanced vascular nitric oxide generation. Cardiovasc. Res. 2003, 59, 488–500. [Google Scholar] [CrossRef]
- Kanie, N.; Kamata, K. Contractile responses in spontaneously diabetic mice: I. Involvement of superoxide anion in enhanced contractile response of aorta to norepinephrine in C57BL/KsJ(db/db) mice. Gen. Pharmacol. Vasc. Syst. 2000, 35, 311–318. [Google Scholar] [CrossRef]
- Dhaunsi, G.S.; Yousif, M.H.M.; Akhtar, S.; Chappell, M.C.; Diz, D.I.; Benter, I.F. Angiotensin-(1–7) prevents diabetes-induced attenuation in PPAR-γ and catalase activities. Eur. J. Pharmacol. 2010, 638, 108–114. [Google Scholar] [CrossRef]
- Eldadah, Z.A.; Brenn, T.; Furthmayr, H.; Dietz, H.C. Expression of a mutant human fibrillin allele upon a normal human or murine genetic background recapitulates a Marfan cellular phenotype. J. Clin. Investig. 1995, 95, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Dietz, H.C.; Mc Intosh, I.; Sakai, L.Y.; Corson, G.M.; Chalberg, S.C.; Pyeritz, R.E.; Francomano, C.A. Four novel FBN1 mutations: Significance for mutant transcript level and EGF-like domain calcium binding in the pathogenesis of Marfan syndrome. Genomics 1993, 17, 468–475. [Google Scholar] [CrossRef]
- Padang, R.; Bannon, P.G.; Jeremy, R.; Richmond, D.R.; Semsarian, C.; Vallely, M.; Wilson, M.; Yan, T.D. The genetic and molecular basis of bicuspid aortic valve associated thoracic aortopathy: A link to phenotype heterogeneity. Ann. Cardiothorac. Surg. 2013, 2, 83–91. [Google Scholar] [CrossRef]
- Robertson, E.; Dilworth, C.; Lu, Y.; Hambly, B.; Jeremy, R. Molecular mechanisms of inherited thoracic aortic disease—From gene variant to surgical aneurysm. Biophys. Rev. 2014, 7, 105–115. [Google Scholar] [CrossRef]
- Jones, J.A.; Ikonomidis, J.S. The pathogenesis of aortopathy in Marfan syndrome and related diseases. Curr. Cardiol. Rep. 2010, 12, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Jondeau, G.; Michel, J.B.; Boileau, C. The translational science of Marfan syndrome. Heart 2011, 97, 1206–1214. [Google Scholar] [CrossRef]
- Lillie, M.A.; David, G.J.; Gosline, J.M. Mechanical role of elastin-associated microfibrils in pig aortic elastic tissue. Connect. Tissue Res. 1998, 37, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.Z.; Tehrani, A.Y.; Jett, K.A.; Bernatchez, P.; van Breemen, C.; Esfandiarei, M. Quantification of aortic and cutaneous elastin and collagen morphology in Marfan syndrome by multiphoton microscopy. J. Struct. Biol. 2014, 187, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Schwaerzer, G.K.; Kalyanaraman, H.; Casteel, D.E.; Dalton, N.D.; Gu, Y.; Lee, S.; Zhuang, S.; Wahwah, N.; Schilling, J.M.; Patel, H.H.; et al. Aortic pathology from protein kinase G activation is prevented by an antioxidant vitamin B12 analog. Nat. Commun. 2019, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- El-Hamamsy, I.; Yacoub, M.H.; El-Hamamsy, I.; Yacoub, M.H. Cellular and molecular mechanisms of thoracic aortic aneurysms. Nat. Rev. Cardiol. 2009, 6, 771–786. [Google Scholar] [CrossRef] [PubMed]
- Daugherty, A.; Chen, Z.; Sawada, H.; Rateri, D.L.; Sheppard, M.B. Transforming growth factor-beta in thoracic aortic Aneurysms: Good, bad, or irrelevant? J. Am. Heart Assoc. 2017, 6, e005221. [Google Scholar] [CrossRef]
- Kaartinen, V.; Warburton, D. Fibrillin controls TGF-β activation. Nat. Genet. 2003, 33, 331–332. [Google Scholar] [CrossRef]
- Carta, L.; Smaldone, S.; Zilberberg, L.; Loch, D.; Dietz, H.C.; Rifkin, D.B.; Ramirez, F. p38 MAPK is an early determinant of promiscuous Smad2/3 signaling in the aortas of Fibrillin-1 (Fbn1)-null mice. J. Biol. Chem. 2009, 284, 5630–5636. [Google Scholar] [CrossRef]
- Habashi, J.P.; Judge, D.; Holm, T.M.; Cohn, R.D.; Loeys, B.; Cooper, T.K.; Myers, L.; Klein, E.C.; Liu, G.; Calvi, C.; et al. Losartan, an AT1 antagonist, prevents aortic aneurysm in a mouse model of Marfan syndrome. Science 2006, 312, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Keane, M.G.; Pyeritz, R.E. Medical management of Marfan syndrome. Circulation 2008, 117, 2802–2813. [Google Scholar] [CrossRef]
- Rateri, D.L.; Davis, F.M.; Balakrishnan, A.; Howatt, D.A.; Moorleghen, J.J.; O’Connor, W.N.; Charnigo, R.; Cassis, L.A.; Daugherty, A. Angiotensin II induces region-specific medial disruption during evolution of ascending aortic aneurysms. Am. J. Pathol. 2014, 184, 2586–2595. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Fujii, K.; Akiyama, N.; Furutani, A.; Hoshii, Y.; Tanaka, N.; Ricci, R.; Ishihara, T.; et al. Regression of abdominal aortic aneurysm by inhibition of c-Jun N-terminal kinase. Nat. Med. 2005, 11, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Altayó, F.; Meirelles, T.; Molist, E.C.; Sorolla, M.A.; del Blanco, D.G.; Luque, J.L.; Mas-Stachurska, A.; Siegert, A.-M.; Bonorino, F.; Barberà, L.; et al. Redox stress in Marfan syndrome: Dissecting the role of the NADPH oxidase NOX4 in aortic aneurysm. Free. Radic. Biol. Med. 2018, 118, 44–58. [Google Scholar] [CrossRef]
- Wang, W.; Xu, B.; Xuan, H.; Ge, Y.; Wang, Y.; Wang, L.; Huang, J.; Fu, W.; Michie, S.A.; Dalman, R.L. Hypoxia-inducible factor 1 in clinical and experimental aortic aneurysm disease. J. Vasc. Surg. 2017, 68, 1538–1550.e2. [Google Scholar] [CrossRef]
- Oshima, Y.; Tanimoto, T.; Yuji, K.; Tojo, A. Association between aortic dissection and systemic exposure of vascular endothelial growth factor pathway inhibitors in the japanese adverse drug event report database. Circulation 2017, 135, 815–817. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.L.; Carraro, C.C.; Belló-Klein, A.; Kalil, A.N.; Aerts, N.R.; Carvalho, F.B.; Fernandes, M.C.; Zettler, C.G. Oxidative stress in aortas of patients with advanced occlusive and aneurysmal diseases. Ann. Vasc. Surg. 2018, 52, 216–224. [Google Scholar] [CrossRef]
- Clempus, R.E.; Griendling, K. Reactive oxygen species signaling in vascular smooth muscle cells. Cardiovasc. Res. 2006, 71, 216–225. [Google Scholar] [CrossRef]
- Li, P.-F.; Dietz, R.; Von Harsdorf, R. Reactive oxygen species induce apoptosis of vascular smooth muscle cell. FEBS Lett. 1997, 404, 249–252. [Google Scholar] [CrossRef]
- Martinez-Lemus, L.A.; Zhao, G.; Galiñanes, E.L.; Boone, M. Inward remodeling of resistance arteries requires reactive oxygen species-dependent activation of matrix metalloproteinases. Am. J. Physiol. Circ. Physiol. 2011, 300, H2005–H2015. [Google Scholar] [CrossRef]
- Quintana, R.A.; Taylor, W.R. Cellular Mechanisms of Aortic Aneurysm Formation. Circ. Res. 2019, 124, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Patrono, C.; Fitzgerald, G.A. Isoprostanes: Potential markers of oxidant stress in atherothrombotic disease. Arter. Thromb. Vasc. Biol. 1997, 17, 2309–2315. [Google Scholar] [CrossRef]
- Delanty, N.; Reilly, M.; Pratico, D.; Fitzgerald, D.J.; Lawson, J.A.; Fitzgerald, G.A. 8-Epi PGF2α: Specific analysis of an isoeicosanoid as an index of oxidant stress in vivo. Br. J. Clin. Pharmacol. 1996, 42, 15–19. [Google Scholar] [CrossRef]
- Fiorillo, C.; Becatti, M.; Attanasio, M.; Lucarini, L.; Nassi, N.; Evangelisti, L.; Porciani, M.; Nassi, P.; Gensini, G.; Abbate, R.; et al. Evidence for oxidative stress in plasma of patients with Marfan syndrome. Int. J. Cardiol. 2010, 145, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Drummond, G.R.; Selemidis, S.; Griendling, K.; Sobey, C.G. Combating oxidative stress in vascular disease: NADPH oxidases as therapeutic targets. Nat. Rev. Drug Discov. 2011, 10, 453–471. [Google Scholar] [CrossRef] [PubMed]
- Portelli, S.S.; Hambly, B.D.; Jeremy, R.W.; Robertson, E.N. Oxidative stress in genetically triggered thoracic aortic aneurysm: Role in pathogenesis and therapeutic opportunities. Redox Rep. 2021, 26, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Dolan, J.M.; Meng, H.; Singh, S.; Paluch, R.; Kolega, J. High fluid shear stress and spatial shear stress gradients affect endothelial proliferation, survival, and alignment. Ann. Biomed. Eng. 2011, 39, 1620–1631. [Google Scholar] [CrossRef] [PubMed]
- Branchetti, E.; Poggio, P.; Sainger, R.; Shang, E.; Grau, J.B.; Jackson, B.M.; Lai, E.K.; Parmacek, M.S.; Gorman, R.C.; Gorman, J.H.; et al. Oxidative stress modulates vascular smooth muscle cell phenotype via CTGF in thoracic aortic aneurysm. Cardiovasc. Res. 2013, 100, 316–324. [Google Scholar] [CrossRef]
- Emrich, F.; Penov, K.; Arakawa, M.; Dhablania, N.; Burdon, G.; Pedroza, A.J.; Koyano, T.K.; Kim, Y.M.; Raaz, U.; Connolly, A.J.; et al. Anatomically specific reactive oxygen species production participates in Marfan syndrome aneurysm formation. J. Cell. Mol. Med. 2019, 23, 7000–7009. [Google Scholar] [CrossRef] [PubMed]
- Miller, F.J.; Sharp, J.; Fang, X.; Oberley, L.W.; Oberley, T.D.; Weintraub, N.L. Oxidative stress in human abdominal aortic aneurysms. Arter. Thromb. Vasc. Biol. 2002, 22, 560–565. [Google Scholar] [CrossRef]
- Siu, K.L.; Miao, X.N.; Cai, H. Recoupling of eNOS with folic acid prevents abdominal aortic aneurysm formation in angiotensin II-infused apolipoprotein E null mice. PLoS ONE 2014, 9, e88899. [Google Scholar] [CrossRef] [PubMed]
- Kigawa, Y.; Miyazaki, T.; Lei, X.-F.; Nakamachi, T.; Oguchi, T.; Kim-Kaneyama, J.-R.; Taniyama, M.; Tsunawaki, S.; Shioda, S.; Miyazaki, A. NADPH oxidase deficiency exacerbates angiotensin II–induced abdominal aortic aneurysms in mice. Arter. Thromb. Vasc. Biol. 2014, 34, 2413–2420. [Google Scholar] [CrossRef] [PubMed]
- Crosas-Molist, E.; Meirelles, T.; López-Luque, J.; Serra-Peinado, C.; Selva, J.; Caja, L.; del Blanco, D.G.; Uriarte, J.J.; Bertran, E.; Mendizábal, Y.; et al. Vascular smooth muscle cell phenotypic changes in patients with Marfan syndrome. Arter. Thromb. Vasc. Biol. 2015, 35, 960–972. [Google Scholar] [CrossRef]
- Clark-Greuel, J.N.; Connolly, J.M.; Sorichillo, E.; Narula, N.R.; Rapoport, H.S.; Mohler, E.R., III; Gorman, J.H., III; Gorman, R.C.; Levy, R.J. Transforming growth factor-β1 mechanisms in aortic valve calcification: Increased alkaline phosphatase and related events. Ann. Thorac. Surg. 2007, 83, 946–953. [Google Scholar] [CrossRef]
- Griendling, K.K.; Minieri, C.A.; Ollerenshaw, J.D.; Alexander, R.W. Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ. Res. 1994, 74, 1141–1148. [Google Scholar] [CrossRef]
- Satoh, K.; Nigro, P.; Matoba, T.; O’Dell, M.R.; Cui, Z.; Shi, X.; Mohan, A.; Yan, C.; Abe, J.-I.; Illig, K.A.; et al. Cyclophilin A enhances vascular oxidative stress and the development of angiotensin II–induced aortic aneurysms. Nat. Med. 2009, 15, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Dikalov, S.I.; Nazarewicz, R.R. Angiotensin II-induced production of mitochondrial reactive oxygen species: Potential mechanisms and relevance for cardiovascular disease. Antioxid. Redox Signal. 2013, 19, 1085–1094. [Google Scholar] [CrossRef]
- Barhoumi, T.; Fraulob-Aquino, J.C.; Mian, M.O.R.; Ouerd, S.; Idris-Khodja, N.; Huo, K.-G.; Rehman, A.; Caillon, A.; Dancose-Giambattisto, B.; Ebrahimian, T.; et al. Matrix metalloproteinase-2 knockout prevents angiotensin II-induced vascular injury. Cardiovasc. Res. 2017, 113, 1753–1762. [Google Scholar] [CrossRef]
- Browatzki, M.; Larsen, D.; Pfeiffer, C.A.; Gehrke, S.G.; Schmidt, J.; Kranzhöfer, A.; Katus, H.A.; Kranzhöfer, R. Angiotensin II stimulates matrix metalloproteinase secretion in human vascular smooth muscle cells via nuclear factor-κB and activator protein 1 in a redox-sensitive manner. J. Vasc. Res. 2005, 42, 415–423. [Google Scholar] [CrossRef]
- Kim, H.W.; Blomkalns, A.L.; Ogbi, M.; Thomas, M.; Gavrila, D.; Neltner, B.S.; Cassis, L.A.; Thompson, R.W.; Weiss, R.M.; Lindower, P.D.; et al. Role of myeloperoxidase in abdominal aortic aneurysm formation: Mitigation by taurine. Am. J. Physiol. Circ. Physiol. 2017, 313, H1168–H1179. [Google Scholar] [CrossRef]
- Webb, N.R.; De Beer, M.C.; Wroblewski, J.M.; Ji, A.; Bailey, W.; Shridas, P.; Charnigo, R.J.; Noffsinger, V.P.; Witta, J.; Howatt, D.A.; et al. Deficiency of endogenous acute-phase serum amyloid a protects apoE-/-mice from angiotensin II-induced abdominal aortic aneurysm formation. Arter. Thromb. Vasc. Biol. 2015, 35, 1156–1165. [Google Scholar] [CrossRef]
- Guzik, B.; Sagan, A.; Ludew, D.; Mrowiecki, W.; Chwała, M.; Bujak-Gizycka, B.; Filip, G.; Grudzien, G.; Kapelak, B.; Żmudka, K.; et al. Mechanisms of oxidative stress in human aortic aneurysms—Association with clinical risk factors for atherosclerosis and disease severity. Int. J. Cardiol. 2013, 168, 2389–2396. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Ni, Z.; Oveisi, F.; Trnavsky-Hobbs, D.L. Effect of antioxidant therapy on blood pressure and NO synthase expression in hypertensive rats. Hypertension 2000, 36, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.E.; Manzano-Pech, L.G.; Guarner-Lans, V.; Díaz-Galindo, J.A.; Vásquez, X.; Castrejón-Tellez, V.; Gamboa, R.; Huesca, C.; Fuentevilla-Alvárez, G.; Pérez-Torres, I. Oxidant/antioxidant profile in the thoracic aneurysm of patients with the Loeys-Dietz syndrome. Oxidative Med. Cell. Longev. 2020, 2020, 5392454–5392517. [Google Scholar] [CrossRef]
- Anand, P.; Rajakumar, D.; Jeraud, M.; Felix, A.J.W.; Balasubramanian, T. Effects of taurine on glutathione peroxidase, glutathione reductase and reduced glutathione levels in rats. Pak. J. Biol. Sci. 2011, 14, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Fang, Y.-Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef]
- Satta, S.; Mahmoud, A.M.; Wilkinson, F.L.; Alexander, M.Y.; White, S.J. The Role of Nrf2 in cardiovascular function and disease. Oxid. Med. Cell. Longev. 2017, 2017, 1–18. [Google Scholar] [CrossRef]
- Xiong, W.; Mactaggart, J.; Knispel, R.; Worth, J.; Zhu, Z.; Li, Y.; Sun, Y.; Baxter, B.T.; Johanning, J. Inhibition of reactive oxygen species attenuates aneurysm formation in a murine model. Atherosclerosis 2009, 202, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D. Fish, mercury, selenium and cardiovascular risk: Current evidence and unanswered questions. Int. J. Environ. Res. Public Health 2009, 6, 1894–1916. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.E.; Hernández, A.V.I.; Guarner-Lans, V.; Zuñiga-Muñoz, A.; Fraustro, A.A.; Espejel, R.V.; Pérez-Torres, I. Participation of oleic acid in the formation of the aortic aneurysm in Marfan syndrome patients. Prostaglandins Other Lipid Mediat. 2016, 123, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Lubos, E.; Loscalzo, J.; Handy, D.E. Glutathione Peroxidase-1 in health and disease: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2011, 15, 1957–1997. [Google Scholar] [CrossRef]
- Hubackova, S.; Krejcikova, K.; Bartek, J.; Hodny, Z. IL1- and TGFβ-Nox4 signaling, oxidative stress and DNA damage response are shared features of replicative, oncogene-induced, and drug-induced paracrine ‘Bystander senescence’. Aging 2012, 4, 932–951. [Google Scholar] [CrossRef]
- Polimanti, R.; Piacentini, S.; Lazzarin, N.; Re, M.A.; Manfellotto, D.; Fuciarelli, M. Glutathione S-transferase variants as risk factor for essential hypertension in Italian patients. Mol. Cell. Biochem. 2011, 357, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Rybka, J.; Kupczyk, D.; Kędziora-Kornatowska, K.; Motyl, J.; Czuczejko, J.; Szewczyk-Golec, K.; Kozakiewicz, M.; Pawluk, H.; de Carvalho, L.A.; Kędziora, J. Glutathione-related antioxidant defense system in elderly patients treated for hypertension. Cardiovasc. Toxicol. 2010, 11, 1–9. [Google Scholar] [CrossRef]
- Chung, A.W.; Yang, H.C.; Yeung, K.A.; Van Breemen, C. Mechanical and pharmacological approaches to investigate the pathogenesis of marfan syndrome in the abdominal aorta. J. Vasc. Res. 2008, 45, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Şener, G.; Paskaloğlu, K.; Toklu, H.; Kapucu, C.; Ayanoglu-Dulger, G.; Kacmaz, A.; Sakarcan, A. Melatonin ameliorates chronic renal failure-induced oxidative organ damage in rats. J. Pineal Res. 2004, 36, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Chew, D.K.; Conte, M.S.; Khalil, R.A. Matrix metalloproteinase-specific inhibition of Ca2+ entry mechanisms of vascular contraction. J. Vasc. Surg. 2004, 40, 1001–1010. [Google Scholar] [CrossRef]
- Chung, A.W.; Yeung, K.A.; Sandor, G.; Judge, D.P.; Dietz, H.C.; Van Breemen, C. Loss of elastic fiber integrity and reduction of vascular smooth muscle contraction resulting from the upregulated activities of matrix metalloproteinase-2 and -9 in the thoracic aortic aneurysm in Marfan syndrome. Circ. Res. 2007, 101, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.W.Y.; Yang, H.H.C.; Radomski, M.W.; Van Breemen, C. Long-term doxycycline is more effective than atenolol to prevent thoracic aortic aneurysm in marfan syndrome through the inhibition of matrix metalloproteinase-2 and -9. Circ. Res. 2008, 102, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.H.; Kawase, M.; Murakami, K.; Chen, S.F.; Li, Y.; Calagui, B.; Reola, L.; Carlson, E.; Epstein, C.J. Overexpression of SOD1 in transgenic rats protects vulnerable neurons against ischemic damage after global cerebral ischemia and reperfusion. J. Neurosci. 1998, 18, 8292–8299. [Google Scholar] [CrossRef] [PubMed]
- Van Deel, E.D.; Lu, Z.; Xu, X.; Zhu, G.; Hu, X.; Oury, T.D.; Bache, R.J.; Duncker, D.J.; Chen, Y. Extracellular superoxide dismutase protects the heart against oxidative stress and hypertrophy after myocardial infarction. Free Radic. Biol. Med. 2008, 44, 1305–1313. [Google Scholar] [CrossRef]
- Hibender, S.; Franken, R.; van Roomen, C.; ter Braake, A.; van der Made, I.; Schermer, E.E.; Gunst, Q.; Hoff, M.J.V.D.; Lutgens, E.; Pinto, Y.M.; et al. Resveratrol inhibits aortic root dilatation in the Fbn1 C1039G/+ Marfan mouse model. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1618–1626. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J. Senescent cells, tumor suppression, and organismal aging: Good citizens, bad neighbors. Cell 2005, 120, 513–522. [Google Scholar] [CrossRef]
- Milewicz, D.M.; Prakash, S.K.; Ramirez, F. Therapeutics targeting drivers of thoracic aortic aneurysms and acute aortic dissections: Insights from predisposing genes and mouse models. Annu. Rev. Med. 2017, 68, 51–67. [Google Scholar] [CrossRef]
- Bunton, T.E.; Biery, N.J.; Myers, L.; Gayraud-Morel, B.; Ramirez, F.; Dietz, H.C. Phenotypic Alteration of Vascular Smooth Muscle Cells Precedes Elastolysis in a Mouse Model of Marfan Syndrome. Circ. Res. 2001, 88, 37–43. [Google Scholar] [CrossRef]
- Sung, M.M.; Dyck, J.R. Therapeutic potential of resveratrol in heart failure. Ann. N. Y. Acad. Sci. 2015, 1348, 32–45. [Google Scholar] [CrossRef]
- Van Andel, M.M.; Groenink, M.; Zwinderman, A.H.; Mulder, B.J.; De Waard, V. The potential beneficial effects of resveratrol on cardiovascular complications in Marfan syndrome patients–insights from rodent-based animal studies. Int. J. Mol. Sci. 2019, 20, 1122. [Google Scholar] [CrossRef]
- Budbazar, E.; Seta, F. Abstract 447: Resveratrol attenuates incidence of aortic aneurysm by decreasing reversible oxidation of sirtuin-1 and inhibiting excessive Tgf-b1 in a mouse model of Marfan’s syndrome. Arterioscler. Thromb. Vasc. Biol. 2020, 40, A447. [Google Scholar] [CrossRef]
- Tang, Y.; Xu, J.; Qu, W.; Peng, X.; Xin, P.; Yang, X.; Ying, C.; Sun, X.; Hao, L. Resveratrol reduces vascular cell senescence through attenuation of oxidative stress by SIRT1/NADPH oxidase-dependent mechanisms. J. Nutr. Biochem. 2012, 23, 1410–1416. [Google Scholar] [CrossRef]
- Zarzuelo, M.J.; Sepúlveda, R.L.; Sánchez, M.; Romero, M.; Gómez-Guzmán, M.; Ungvary, Z.; Perez-Vizcaino, F.; Jimenez, R.; Duarte, J. SIRT1 inhibits NADPH oxidase activation and protects endothelial function in the rat aorta: Implications for vascular aging. Biochem. Pharmacol. 2013, 85, 1288–1296. [Google Scholar] [CrossRef]
- Palmieri, D.; Pane, B.; Barisione, C.; Spinella, G.; Garibaldi, S.; Ghigliotti, G.; Brunelli, C.; Fulcheri, E.; Palombo, D. Resveratrol counteracts systemic and local inflammation involved in early abdominal aortic aneurysm development. J. Surg. Res. 2011, 171, e237–e246. [Google Scholar] [CrossRef]
- Franken, R.; Hibender, S.; Hartog, A.W.D.; Radonic, T.; De Vries, C.J.M.; Zwinderman, A.H.; Groenink, M.; Mulder, B.J.M.; De Waard, V. No beneficial effect of general and specific anti-inflammatory therapies on aortic dilatation in Marfan mice. PLoS ONE 2014, 9, e107221. [Google Scholar] [CrossRef]
- Wu, M.-L.; Ho, Y.; Lin, C.-Y.; Yet, S.-F. Heme oxygenase-1 in inflammation and cardiovascular disease. Am. J. Cardiovasc. Dis. 2011, 1, 150–158. [Google Scholar] [PubMed]
- Durante, W. Targeting heme oxygenase-1 in the arterial response to injury and disease. Antioxidants 2020, 9, 829. [Google Scholar] [CrossRef] [PubMed]
- Azuma, J.; Wong, R.J.; Morisawa, T.; Hsu, M.; Maegdefessel, L.; Zhao, H.; Kalish, F.; Kayama, Y.; Wallenstein, M.B.; Deng, A.C.; et al. Heme oxygenase-1 expression affects murine abdominal aortic aneurysm progression. PLoS ONE 2016, 11, e0149288. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.; Wu, M.-L.; Gung, P.-Y.; Chen, C.-H.; Kuo, C.-C.; Yet, S.-F. Heme oxygenase-1 deficiency exacerbates angiotensin II-induced aortic aneurysm in mice. Oncotarget 2016, 7, 67760–67776. [Google Scholar] [CrossRef] [PubMed]
- Meital, L.T.; Windsor, M.T.; Perissiou, M.; Schulze, K.; Magee, R.; Kuballa, A.; Golledge, J.; Bailey, T.G.; Askew, C.; Russell, F.D. Omega-3 fatty acids decrease oxidative stress and inflammation in macrophages from patients with small abdominal aortic aneurysm. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Manning, M.W.; Cassis, L.A.; Daugherty, A. Differential effects of doxycycline, a broad-spectrum matrix metalloproteinase inhibitor, on angiotensin ii–induced atherosclerosis and abdominal aortic aneurysms. Arter. Thromb. Vasc. Biol. 2003, 23, 483–488. [Google Scholar] [CrossRef]
- Guido, M.C.; Debbas, V.; Salemi, V.M.; Tavares, E.R.; Meirelles, T.; Araujo, T.L.S.; Nolasco, P.; Ferreira-Filho, J.C.A.; Takimura, C.K.; Pereira, L.V.; et al. Effect of the antioxidant lipoic acid in aortic phenotype in a marfan syndrome mouse model. Oxidative Med. Cell. Longev. 2018, 2018, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gorąca, A.; Huk-Kolega, H.; Piechota, A.; Kleniewska, P.; Ciejka, E.; Skibska, B. Lipoic acid—Biological activity and therapeutic potential. Pharmacol. Rep. 2011, 63, 849–858. [Google Scholar] [CrossRef]
- Tibullo, D.; Volti, G.L.; Giallongo, C.; Grasso, S.; Tomassoni, D.; Anfuso, C.D.; Lupo, G.; Amenta, F.; Avola, R.; Bramanti, V. Biochemical and clinical relevance of alpha lipoic acid: Antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflamm. Res. 2017, 66, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-L.; Hsieh, Y.-J.; Chou, F.-P.; Wang, C.-J.; Cheng, M.-T.; Tseng, T.-H. Hibiscus protocatechuic acid inhibits lipopolysaccharide-induced rat hepatic damage. Arch. Toxicol. 2003, 77, 42–47. [Google Scholar]
- Fakeye, T.; Adegoke, A.; Omoyeni, O.; Famakinde, A. Effects of water extract of Hibiscus sabdariffa, Linn (Malvaceae)’Roselle’on excretion of a diclofenac formulation. Phytother. Res. 2007, 21, 96–98. [Google Scholar] [CrossRef]
- Soto, M.E.; Zuñiga-Muñoz, A.; Guarner-Lans, V.; Duran-Hernández, E.J.; Pérez-Torres, I. Infusion of Hibiscus sabdariffa L. modulates oxidative stress in patients with Marfan syndrome. Mediat. Inflamm. 2016, 2016, 1–12. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Ruiz-Ramírez, A.; Baños, G.; El-Hafidi, M. Hibiscus sabdariffa Linnaeus (Malvaceae), curcumin and resveratrol as alternative medicinal agents against metabolic syndrome. Cardiovasc. Hematol. Agents Med. Chem. 2013, 11, 25–37. [Google Scholar] [CrossRef]
- Wang, S.-C.; Lee, S.-F.; Wang, C.-J.; Lee, C.-H.; Lee, W.-C.; Lee, H.-J. Aqueous extract from Hibiscus sabdariffa Linnaeus ameliorate diabetic nephropathy via regulating oxidative status and Akt/Bad/14-3-3γ in an experimental animal model. Evid.-Based Complement. Altern. Med. 2011, 2011, 938126. [Google Scholar]
- Jung, O.; Marklund, S.L.; Geiger, H.; Pedrazzini, T.; Busse, R.; Brandes, R.P. Extracellular superoxide dismutase is a major determinant of nitric oxide bioavailability: In vivo and ex vivo evidence from ecSOD-deficient mice. Circ. Res. 2003, 93, 622–629. [Google Scholar] [CrossRef]
- Adler, S.; Huang, H. Oxidant stress in kidneys of spontaneously hypertensive rats involves both oxidase overexpression and loss of extracellular superoxide dismutase. Am. J. Physiol. Physiol. 2004, 287, F907–F913. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Reaume, A.G.; Huang, T.-T.; Carlson, E.; Murakami, K.; Chen, S.F.; Hoffman, E.K.; Scott, R.W.; Epstein, C.J.; Chan, P.H. Reduction of CuZn-superoxide dismutase activity exacerbates neuronal cell injury and edema formation after transient focal cerebral ischemia. J. Neurosci. 1997, 17, 4180–4189. [Google Scholar] [CrossRef] [PubMed]
- Farombi, E.; Ige, O. Hypolipidemic and antioxidant effects of ethanolic extract from dried calyx of Hibiscus sabdariffa in alloxan-induced diabetic rats. Fundam. Clin. Pharmacol. 2007, 21, 601–609. [Google Scholar] [CrossRef]
- Mirochnitchenko, O.; Weisbrot-Lefkowitz, M.; Reuhl, K.; Chen, L.; Yang, C.; Inouye, M. Acetaminophen toxicity: Opposite effects of two forms of glutathione peroxidase. J. Biol. Chem. 1999, 274, 10349–10355. [Google Scholar] [CrossRef]
- Avissar, N.; Slemmon, J.R.; Palmer, I.S.; Cohen, H.J. Partial sequence of human plasma glutathione peroxidase and immunologic identification of milk glutathione peroxidase as the plasma enzyme. J. Nutr. 1991, 121, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Engle, M.R.; Singh, S.P.; Czernik, P.J.; Gaddy, D.; Montague, D.C.; Ceci, J.D.; Yang, Y.; Awasthi, S.; Awasthi, Y.C.; Zimniak, P. Physiological role of mGSTA4-4, a glutathione S-transferase metabolizing 4-hydroxynonenal: Generation and analysis of mGsta4 null mouse. Toxicol. Appl. Pharmacol. 2004, 194, 296–308. [Google Scholar] [CrossRef]
- Chapple, S.; Cheng, X.; Mann, G.E. Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol. 2013, 1, 319–331. [Google Scholar] [CrossRef]
- Wilton, E.; Bland, M.; Thompson, M.; Jahangiri, M. Matrix metalloproteinase expression in the ascending aorta and aortic valve. Interact. Cardiovasc. Thorac. Surg. 2008, 7, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.H.; Chen, J.H.; Chou, F.P.; Wang, C.J. Protocatechuic acid inhibits cancer cell metastasis involving the down-regulation of Ras/Akt/NF-κB pathway and MMP-2 production by targeting RhoB activation. Br. J. Pharmacol. 2011, 162, 237–254. [Google Scholar] [CrossRef]
- Kobayashi, H.; Matsuda, M.; Fukuhara, A.; Komuro, R.; Shimomura, I. Dysregulated glutathione metabolism links to impaired insulin action in adipocytes. Am. J. Physiol. Metab. 2009, 296, E1326–E1334. [Google Scholar] [CrossRef]
- Deponte, M. Glutathione catalysis and the reaction mechanisms of glutathione-dependent enzymes. Biochim. Biophys. Acta—Gen. Subj. 2013, 1830, 3217–3266. [Google Scholar] [CrossRef]
- Franken, R.; den Hartog, A.W.; Singh, M.; Pals, G.; Zwinderman, A.H.; Groenink, M.; Mulder, B.J. Marfan syndrome: Progress report. Prog. Pediatr. Cardiol. 2012, 34, 9–14. [Google Scholar] [CrossRef]
- Koo, H.-K.; Lawrence, K.A.; Musini, V. Beta-blockers for preventing aortic dissection in Marfan syndrome. Cochrane Database Syst. Rev. 2017, 11, CD011103. [Google Scholar] [CrossRef]
- Gersony, D.R.; McClaughlin, M.A.; Jin, Z.; Gersony, W.M. The effect of beta-blocker therapy on clinical outcome in patients with Marfan’s syndrome: A meta-analysis. Int. J. Cardiol. 2007, 114, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Shores, J.; Berger, K.R.; Murphy, E.A.; Pyeritz, R.E. Progression of aortic dilatation and the benefit of long-term β-adrenergic blockade in Marfan’s syndrome. N. Engl. J. Med. 1994, 330, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Van Andel, M.M.; Indrakusuma, R.; Jalalzadeh, H.; Balm, R.; Timmermans, J.; Scholte, A.J.; Berg, M.V.D.; Zwinderman, A.H.; Mulder, B.J.M.; De Waard, V.; et al. Long-term clinical outcomes of losartan in patients with Marfan syndrome: Follow-up of the multicentre randomized controlled COMPARE trial. Eur. Heart J. 2020, 41, 4181–4187. [Google Scholar] [CrossRef] [PubMed]
- Groenink, M.; Hartog, A.W.D.; Franken, R.; Radonic, T.; de Waard, V.; Timmermans, J.; Scholte, A.J.; Berg, M.P.V.D.; Spijkerboer, A.M.; Marquering, H.A.; et al. Losartan reduces aortic dilatation rate in adults with Marfan syndrome: A randomized controlled trial. Eur. Heart J. 2013, 34, 3491–3500. [Google Scholar] [CrossRef]
- Mullen, M.; Jin, X.Y.; Child, A.; Stuart, A.G.; Dodd, M.; Aragon-Martin, J.A.; Gaze, D.; Kiotsekoglou, A.; Yuan, L.; Hu, J.; et al. Irbesartan in Marfan syndrome (AIMS): A double-blind, placebo-controlled randomised trial. Lancet 2019, 394, 2263–2270. [Google Scholar] [CrossRef]
- Sellers, S.L.; Milad, N.; Chan, R.; Mielnik, M.; Jermilova, U.; Huang, P.L.; de Crom, R.; Hirota, J.A.; Hogg, J.C.; Sandor, G.G.; et al. Inhibition of Marfan syndrome aortic root dilation by losartan. Am. J. Pathol. 2018, 188, 574–585. [Google Scholar] [CrossRef]
- Tehrani, A.Y.; White, Z.; Milad, N.; Esfandiarei, M.; Seidman, M.A.; Bernatchez, P. Blood pressure-independent inhibition of Marfan aortic root widening by the angiotensin II receptor blocker valsartan. Physiol. Rep. 2021, 9, e14877. [Google Scholar] [CrossRef]
- Takata, M.; Amiya, E.; Watanabe, M.; Omori, K.; Imai, Y.; Fujita, D.; Nishimura, H.; Kato, M.; Morota, T.; Nawata, K.; et al. Impairment of flow-mediated dilation correlates with aortic dilation in patients with Marfan syndrome. Heart Vessel. 2013, 29, 478–485. [Google Scholar] [CrossRef]
- Gibson, C.; Nielsen, C.; Alex, R.; Cooper, K.; Farney, M.; Gaufin, D.; Cui, J.Z.; Van Breemen, C.; Broderick, T.L.; Vallejo-Elias, J.; et al. Mild aerobic exercise blocks elastin fiber fragmentation and aortic dilatation in a mouse model of Marfan syndrome associated aortic aneurysm. J. Appl. Physiol. 2017, 123, 147–160. [Google Scholar] [CrossRef]
- Xiong, W.; Knispel, R.A.; Dietz, H.C.; Ramirez, F.; Baxter, B.T. Doxycycline delays aneurysm rupture in a mouse model of Marfan syndrome. J. Vasc. Surg. 2008, 47, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.R.; Clayton, N.P.; Carta, L.; Galatioto, J.; Chiu, E.; Smaldone, S.; Nelson, C.A.; Cheng, S.H.; Wentworth, B.M.; Ramirez, F. Dimorphic effects of transforming growth factor-β signaling during aortic aneurysm progression in mice suggest a combinatorial therapy for Marfan syndrome. Arter. Thromb. Vasc. Biol. 2015, 35, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Rurali, E.; Perrucci, G.L.; Pilato, C.A.; Pini, A.; Gaetano, R.; Nigro, P.; Pompilio, G. Precise therapy for thoracic aortic aneurysm in Marfan syndrome: A puzzle nearing its solution. Prog. Cardiovasc. Dis. 2018, 61, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Longo, G.M.; Xiong, W.; Greiner, T.C.; Zhao, Y.; Fiotti, N.; Baxter, B.T. Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J. Clin. Investig. 2002, 110, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, M.W.; Phylactou, L.A. Towards an RNA-based therapy for Marfan syndrome. Mol. Med. Today 1998, 4, 376–381. [Google Scholar] [CrossRef]
- Montgomery, R.A.; Dietz, H.C. Inhibition of fibrillin 1 expression using U1 snRNA as a vehicle for the presentation of antisense targeting sequence. Hum. Mol. Genet. 1997, 6, 519–525. [Google Scholar] [CrossRef]
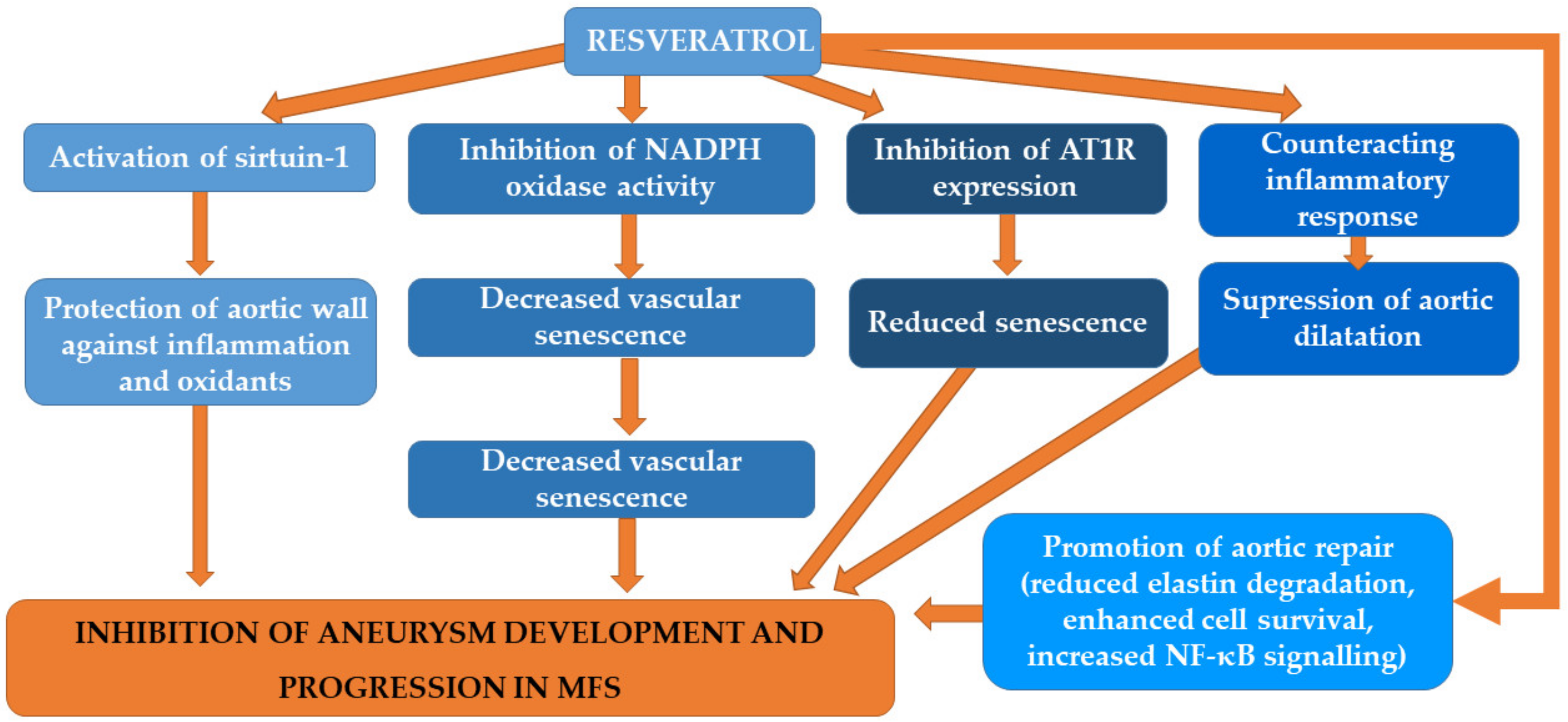
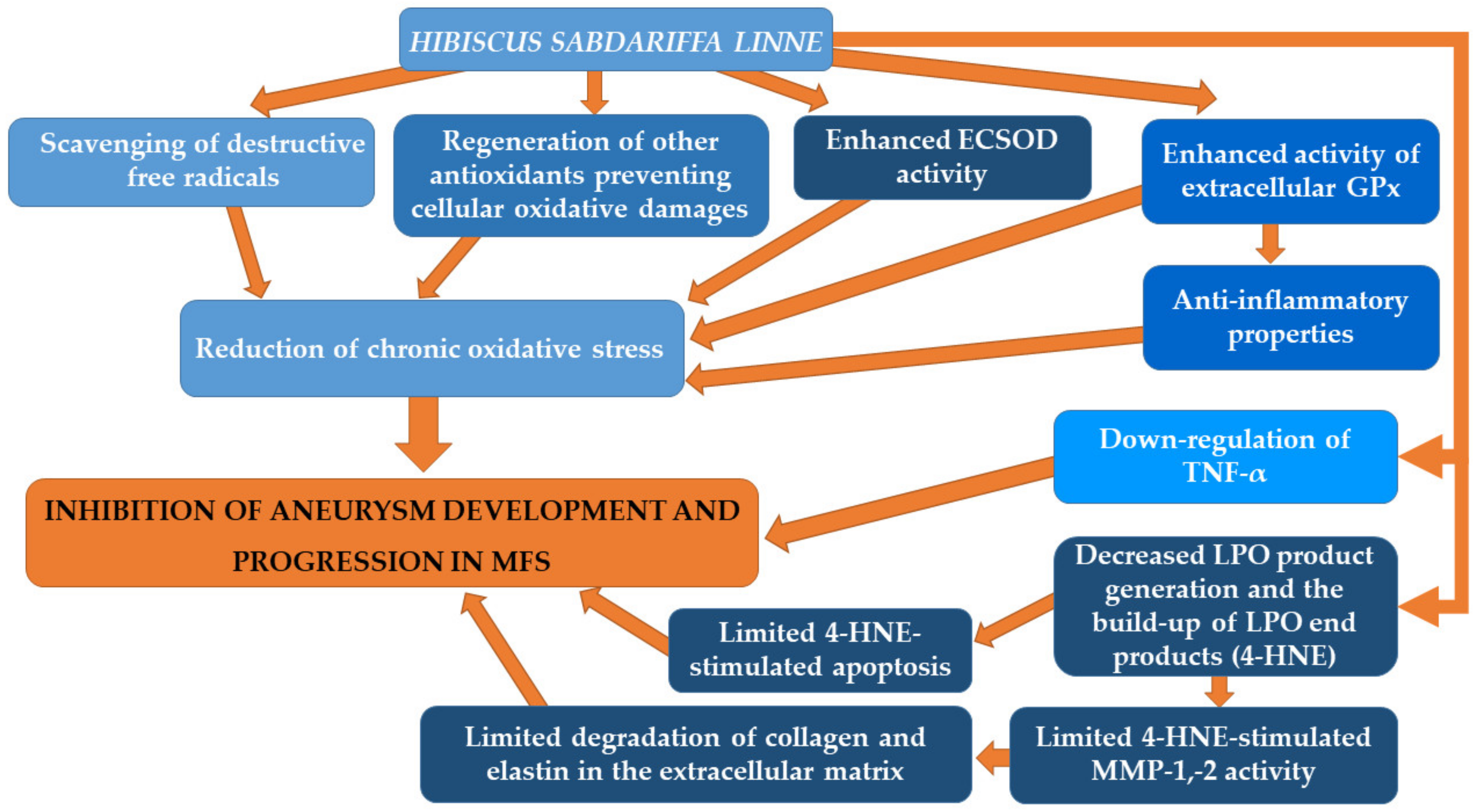
| Type of Therapy | Type of Study | Important Results | Ref. |
|---|---|---|---|
| Resveratrol (Res) | Animal study Adult male Wistar rats fed with HFS diet in the presence or absence of Res for 3 months. Cultured BAECs | Res protected against HFS- or high-glucose-induced increase in NADPH oxidase p47phox expression and decrease in SIRT1 level. Conclusion: Res can reverse the senescence process in the aorta induced by HFS in rats or by exposure to high glucose in cultured BAECs. The underlying mechanism is SIRT1/NADPH oxidase pathway-dependent. | [122] |
| Animal study Fbn1 (C1039G/+) MFS mouse model. Cultured SMC | Resveratrol enhanced the nuclear localization of sirtuin-1 in the vessel wall. It had no effect on leukocyte infiltration, activation of SMAD2 and ERK1/2. Resveratrol reduced aortic elastin breaks and decreased micro-RNA-29b expression. Resveratrol’s effect on micro-RNA-29b downregulation was endothelial cell- and nuclear factor κB-dependent. | [115] | |
| Animal study Male Sprague-Dawley rats on Resveratrol (10 mg/kg/die) or vehicle (Et-OH) alone for 7 days before until 14 days after the AAA induction with elastase | Resveratrol counteracted the CD62L-monocyte subset expansion, CD143 monocyte expression and circulating levels of MMP-9 activity and TNFα associated with AAA induction. Resveratrol markedly attenuated AAA expansion, vessel wall macrophage infiltration and MMP-9, VEGF and TNFα expression, compared with AAA from Et-OH group. Conclusions: Resveratrol limited the monocyte-dependent inflammatory response, macrophage differentiation and aortic lumen enlargement in elastase-induced AAA. | [124] | |
| Animal study Fibrillin-1 hypomorphic mice; Fbn1mgR/mgR receiving resveratrol (105 mg/kg/day mixed in food) | The addition of resveratrol to food significantly reduced mortality of Fbn1mgR/mgR mice compared to mice on regular chow. Resveratrol-fed mice had considerably decreased plasma levels of active TGF-β1 and higher levels of total reversible oxidations of proteins compared to wild-type mice. Conclusions: Resveratrol may diminish the incidence and mortality of aortic aneurysm via reduction of oxidative post-translational modifications of SirT-1 in VSM cells and inhibition of excessive TGF-β1 in Fbn1mgR/mgR mice. | [121] | |
| Targeted therapies inducing HO-1: heme, rosuvastatin | Porcine pancreatic elastase (PPE) model of AAA induction in HO-1 heterozygous (HO-1+/−, HO-1 Het) mice (heme). Murine AAA model (Ang II-ApoE−/−) (resveratrol) | Deficiency in HO-1 leads to augmented AAA development. Peritoneal macrophages from HO-1+/− mice showed increased gene expression of MCP-1, TNF-alpha, IL-1-beta and IL-6, and decreased expression of anti-inflammatory cytokines (IL-10 and TGF-β). Treatment with heme returned AAA progression in HO-1 Het mice to a wild-type profile. Low doses of rosuvastatin can induce HO-1 expression in aortic tissue and suppress AAA progression. Conclusions: Pleiotropic statin effects might be beneficial in AAA, possibly through the upregulation of HO-1. | [128] |
| n-3 PUFA docosahexaenoic acid (DHA) | Cells obtained from men with small AAA and age-matched male controls incubated with DHA for 1 h before exposure to 0.1 µg/mL LPS for 24 h | DHA supplementation decreased the concentration of TNF-α and IL-6 in macrophage supernatants. DHA increased glutathione peroxidase activity and HO-1 mRNA expression. | [130] |
| Lipoic acid Losartan | MFS mgΔloxPneo mouse model vs. WT mice: untreated, treated with losartan and treated with lipoic acid | MFS animals treated with lipoic acid showed markedly reduced ROS production and lower ERK1/2 phosphorylation. Aortic dilation and elastic fiber breakdown were unaltered. Absence of focal inhomogeneous regions in MFS animals treated with lipoic acid. Losartan reduced aortic dilation and elastic fiber breakdown despite no change in ROS generation. | [132] |
| Hibiscus sabdariffa Linne | HSL infusion in MFS patients | Treatment significantly decreased ECSOD (p = 0.03), EGPx (p = 0.04), GST (p = 0.03), GSH (p = 0.01) and TAC and ascorbic acid (p = 0.02). GSSG-R activity (p = 0.04) and LPO (p = 0.02) were increased in MFS patients vs. patients receiving the HSL. Conclusions: Infusion of HSL allows an increase in antioxidant capacity of both the enzymatic and nonenzymatic systems, in the plasma of the MSF patients. | [137] |
| Losartan | MFS mice lacking ATR1a expression | MFS/ATR1a-null mice showed unabated aortic root enlargement and remained fully responsive to losartan. Losartan’s anti-remodeling properties may be ATR- independent. Losartan can activate the endothelial function in mice and patients. In vitro, losartan can increase endothelial NO release in the absence of AngII and correct MFS NO levels in vivo. Conclusions: Increased protective endothelial function, rather than ATR1 inhibition or BP lowering, may be beneficial in preventing aortic root disease in MFS. | [159] |
| Sub-BP-lowering dose valsartan Hypotensive dose of losartan | Patients with MFS | Valsartan attenuated MFS aortic root widening by 75.9%. A similar effect was seen in the case of a hypotensive dose of losartan (79.4%). Medial thickening, elastic fiber fragmentation and phospho-ERK signaling were inhibited to a similar degree with both treatments. Valsartan and losartan decreased vascular contractility ex vivo in a NO-sensitive fashion. Conclusions: Artic root stability can be achieved with valsartan in absence of BP-lowering effects. | [160] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rysz, J.; Gluba-Brzózka, A.; Rokicki, R.; Franczyk, B. Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome. Biomedicines 2021, 9, 1171. https://doi.org/10.3390/biomedicines9091171
Rysz J, Gluba-Brzózka A, Rokicki R, Franczyk B. Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome. Biomedicines. 2021; 9(9):1171. https://doi.org/10.3390/biomedicines9091171
Chicago/Turabian StyleRysz, Jacek, Anna Gluba-Brzózka, Robert Rokicki, and Beata Franczyk. 2021. "Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome" Biomedicines 9, no. 9: 1171. https://doi.org/10.3390/biomedicines9091171
APA StyleRysz, J., Gluba-Brzózka, A., Rokicki, R., & Franczyk, B. (2021). Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome. Biomedicines, 9(9), 1171. https://doi.org/10.3390/biomedicines9091171







