Characterization of a Compound Heterozygous SLC2A9 Mutation That Causes Hypouricemia
Abstract
:1. Introduction
2. Methods
2.1. Study Participants
2.2. DNA Preparation and Whole-Exome Sequencing
2.3. WES Variant Filtering Analysis
2.4. Direct Sanger Sequencing
2.5. In Silico Analysis of Novel Missense Variants and Molecular Dynamics
2.5.1. In Silico Prediction
2.5.2. Molecular Dynamics
2.5.3. Molecular/Functional Studies
Generation of SLC2A9b Expression Vectors
Site-Directed Mutagenesis for the Met126Val Mutant
In Vitro Transcription
SLC2A9b Expression in X. laevis Oocytes
Western Blot Analysis
Uric Acid Uptake Assay
Confocal Microscopy
3. Results
3.1. Demographics
3.1.1. Identification of Novel Variants in SLC2A9b by Whole-Exome Sequencing
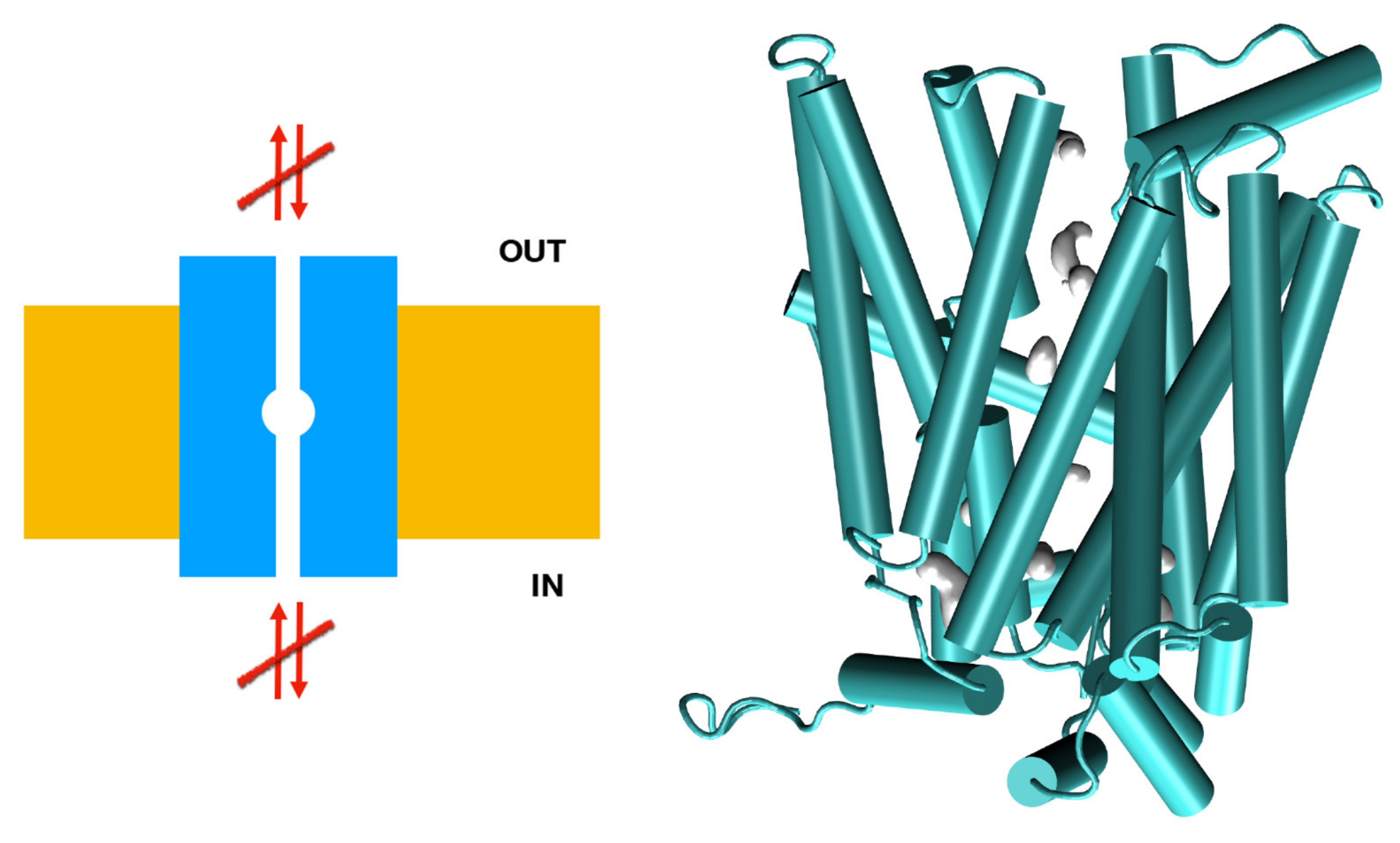
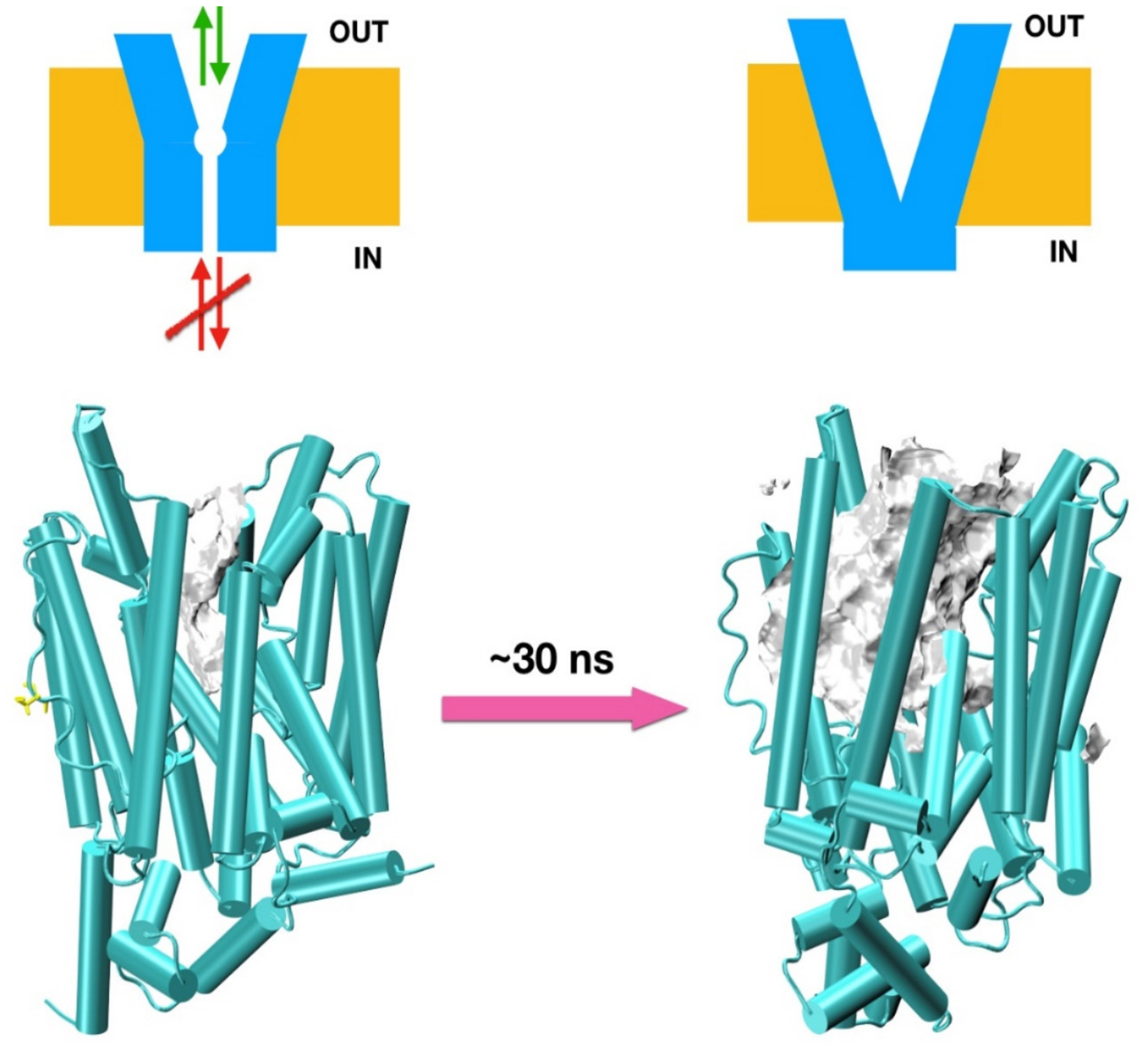
3.1.2. In Silico and Molecular Dynamics Prediction of SLC2A9b
3.1.3. Molecular Dynamics Prediction of SLC2A9b and Its Affinity for Uric Acid
3.2. Molecular Analysis
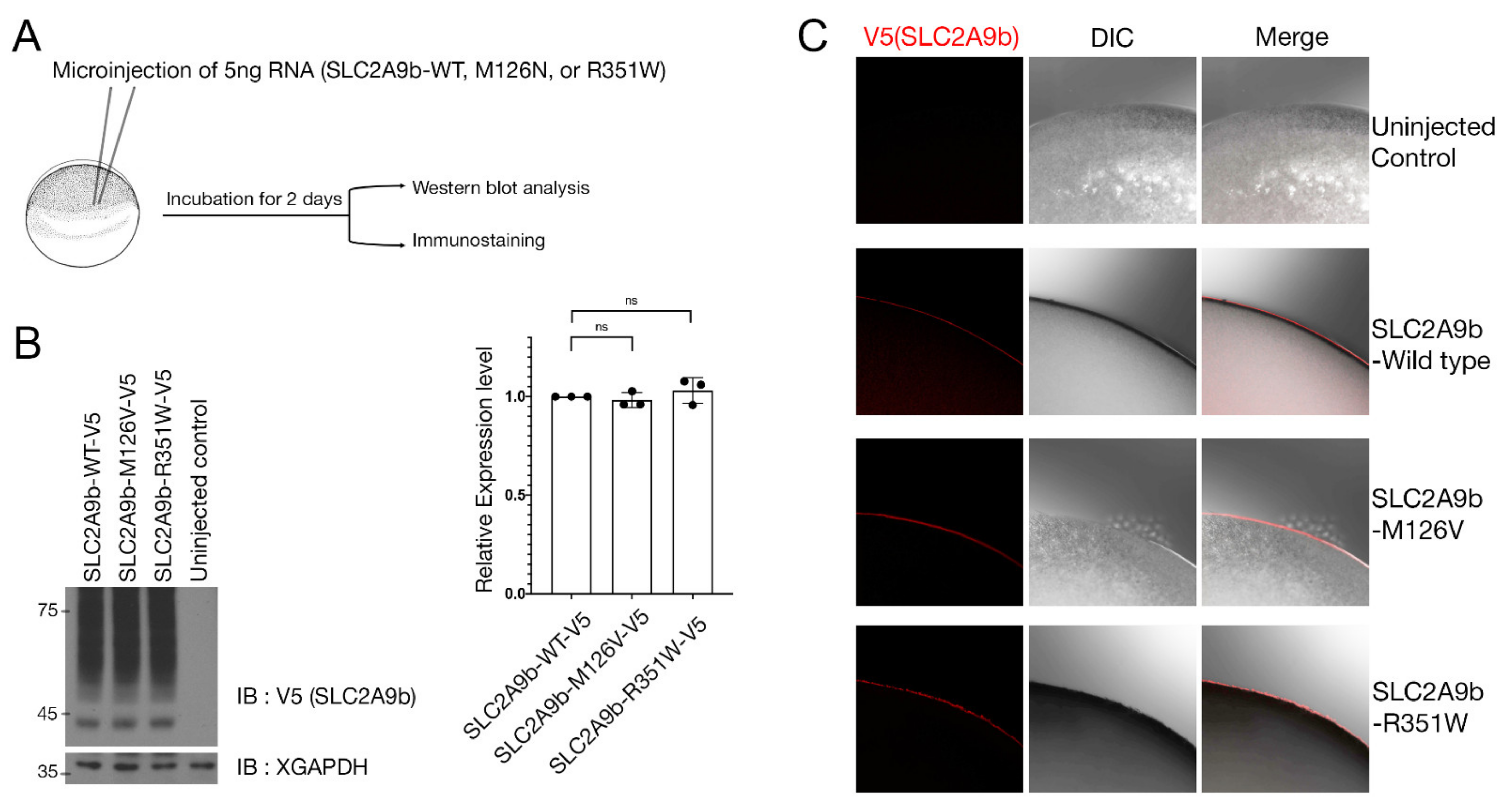
3.2.1. SLC2A9b-p.Met126Val Expression Analysis in X. laevis Oocytes
3.2.2. Urate Transport Activity of SLC2A9b-p.Met126Val in Xenopus Oocytes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cho, S.K.; Chang, Y.; Kim, I.; Ryu, S. U-Shaped Association Between Serum Uric Acid Level and Risk of Mortality: A Cohort Study. Arthritis Rheumatol. 2018, 70, 1122–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sebesta, I.; Stiburkova, B.; Bartl, J.; Ichida, K.; Hosoyamada, M.; Taylor, J.; Marinaki, A. Diagnostic tests for primary renal hypouricemia. Nucleosides Nucleic Acids 2011, 30, 1112–1116. [Google Scholar] [CrossRef]
- Cheong, H.I.; Kang, J.H.; Lee, J.H.; Ha, I.S.; Kim, S.; Komoda, F.; Sekine, T.; Igarashi, T.; Choi, Y. Mutational analysis of idiopathic renal hypouricemia in Korea. Pediatric Nephrol. 2005, 20, 886–890. [Google Scholar] [CrossRef] [PubMed]
- Ichida, K.; Hosoyamada, M.; Hisatome, I.; Enomoto, A.; Hikita, M.; Endou, H.; Hosoya, T. Clinical and molecular analysis of patients with renal hypouricemia in Japan-influence of URAT1 gene on urinary urate excretion. J. Am. Soc. Nephrol. 2004, 15, 164–173. [Google Scholar] [CrossRef]
- Cho, S.K.; Kim, B.; Myung, W.; Chang, Y.; Ryu, S.; Kim, H.N.; Kim, H.L.; Kuo, P.H.; Winkler, C.A.; Won, H.H. Polygenic analysis of the effect of common and low-frequency genetic variants on serum uric acid levels in Korean individuals. Sci. Rep. 2020, 10, 9179. [Google Scholar] [CrossRef] [PubMed]
- Kawasoe, S.; Ide, K.; Usui, T.; Kubozono, T.; Yoshifuku, S.; Miyahara, H.; Maenohara, S.; Ohishi, M.; Kawakami, K. Distribution and Characteristics of Hypouricemia within the Japanese General Population: A Cross-Sectional Study. Medicina 2019, 55, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enomoto, A.; Kimura, H.; Chairoungdua, A.; Shigeta, Y.; Jutabha, P.; Cha, S.H.; Hosoyamada, M.; Takeda, M.; Sekine, T.; Igarashi, T.; et al. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 2002, 417, 447–452. [Google Scholar] [CrossRef]
- Tin, A.; Li, Y.; Brody, J.A.; Nutile, T.; Chu, A.Y.; Huffman, J.E.; Yang, Q.; Chen, M.H.; Robinson-Cohen, C.; Mace, A.; et al. Large-scale whole-exome sequencing association studies identify rare functional variants influencing serum urate levels. Nat. Commun. 2018, 9, 4228. [Google Scholar] [CrossRef] [Green Version]
- Stiburkova, B.; Taylor, J.; Marinaki, A.M.; Sebesta, I. Acute kidney injury in two children caused by renal hypouricaemia type 2. Pediatric Nephrol. 2012, 27, 1411–1415. [Google Scholar] [CrossRef]
- Dinour, D.; Bahn, A.; Ganon, L.; Ron, R.; Geifman Holtzman, O.; Knecht, A.; Gafter, U.; Rachamimov, R.; Sela, B.; Burckhardt, G.; et al. URAT1 mutations cause renal hypouricemia type 1 in Iraqi Jews. Nephrol. Dial. Transplant. 2011, 26, 2175–2181. [Google Scholar] [CrossRef] [Green Version]
- Tasic, V.; Hynes, A.M.; Kitamura, K.; Cheong, H.I.; Lozanovski, V.J.; Gucev, Z.; Jutabha, P.; Anzai, N.; Sayer, J.A. Clinical and functional characterization of URAT1 variants. PLoS ONE 2011, 6, e28641. [Google Scholar] [CrossRef] [PubMed]
- Stiburkova, B.; Sebesta, I.; Ichida, K.; Nakamura, M.; Hulkova, H.; Krylov, V.; Kryspinova, L.; Jahnova, H. Novel allelic variants and evidence for a prevalent mutation in URAT1 causing renal hypouricemia: Biochemical, genetics and functional analysis. Eur. J. Hum. Genet. 2013, 21, 1067–1073. [Google Scholar] [CrossRef] [Green Version]
- Bhasin, B.; Stiburkova, B.; De Castro-Pretelt, M.; Beck, N.; Bodurtha, J.N.; Atta, M.G. Hereditary renal hypouricemia: A new role for allopurinol? Am. J. Med. 2014, 127, e3–e4. [Google Scholar] [CrossRef] [PubMed]
- Cha, D.H.; Gee, H.Y.; Cachau, R.; Choi, J.M.; Park, D.; Jee, S.H.; Ryu, S.; Kim, K.K.; Won, H.H.; Limou, S.; et al. Contribution of SLC22A12 on hypouricemia and its clinical significance for screening purposes. Sci. Rep. 2019, 9, 14360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, S.K.; Winkler, C.A.; Lee, S.J.; Chang, Y.; Ryu, S. The Prevalence of Hyperuricemia Sharply Increases from the Late Menopausal Transition Stage in Middle-Aged Women. J. Clin. Med. 2019, 8, 296. [Google Scholar] [CrossRef] [Green Version]
- Merriman, T.R. An update on the genetic architecture of hyperuricemia and gout. Arthritis Res. Ther. 2015, 17, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, S.K.; Kim, S.; Chung, J.; Jee, S.H. Discovery of URAT1 SNPs and association between serum uric acid levels and URAT1. BMJ Open 2015, 5, e009360. [Google Scholar] [CrossRef] [Green Version]
- MacArthur, D.G.; Manolio, T.A.; Dimmock, D.P.; Rehm, H.L.; Shendure, J.; Abecasis, G.R.; Adams, D.R.; Altman, R.B.; Antonarakis, S.E.; Ashley, E.A.; et al. Guidelines for investigating causality of sequence variants in human disease. Nature 2014, 508, 469–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Muzny, D.M.; Reid, J.G.; Bainbridge, M.N.; Willis, A.; Ward, P.A.; Braxton, A.; Beuten, J.; Xia, F.; Niu, Z.; et al. Clinical whole-exome sequencing for the diagnosis of mendelian disorders. N. Engl. J. Med. 2013, 369, 1502–1511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sperling, O. Hereditary renal hypouricemia. Mol. Genet. Metab. 2006, 89, 14–18. [Google Scholar] [CrossRef]
- Kim, Y.; Han, B.G. Cohort Profile: The Korean Genome and Epidemiology Study (KoGES) Consortium. Int. J. Epidemiol. 2017, 46, 1350. [Google Scholar] [CrossRef]
- Adzhubei, I.A.; Schmidt, S.; Peshkin, L.; Ramensky, V.E.; Gerasimova, A.; Bork, P.; Kondrashov, A.S.; Sunyaev, S.R. A method and server for predicting damaging missense mutations. Nat. Methods 2010, 7, 248–249. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Henikoff, S.; Ng, P.C. Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat. Protoc. 2009, 4, 1073–1081. [Google Scholar] [CrossRef]
- Gonzalez-Perez, A.; Lopez-Bigas, N. Improving the assessment of the outcome of nonsynonymous SNVs with a consensus deleteriousness score, Condel. Am. J. Hum. Genet. 2011, 88, 440–449. [Google Scholar] [CrossRef] [Green Version]
- Schwarz, J.M.; Cooper, D.N.; Schuelke, M.; Seelow, D. MutationTaster2: Mutation prediction for the deep-sequencing age. Nat. Methods 2014, 11, 361–362. [Google Scholar] [CrossRef]
- Yokoyama, S.; Cai, Y.; Murata, M.; Tomita, T.; Yoneda, M.; Xu, L.; Pilon, A.L.; Cachau, R.E.; Kimura, S. A novel pathway of LPS uptake through syndecan-1 leading to pyroptotic cell death. eLife 2018, 7, e37854. [Google Scholar] [CrossRef] [PubMed]
- Cachau, R.E.; Erickson, J.W.; Villar, H.O. Novel procedure for structure refinement in homology modeling and its application to the human class Mu glutathione S-transferases. Protein Eng. 1994, 7, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.C.; Stone, J.E.; Vandivort, K.L.; Armstrong, T.G.; Wozniak, J.M.; Wilde, M.; Schulten, K. Petascale Tcl with NAMD, VMD, and Swift/T. In Proceedings of the 2014 First Workshop for High Performance Technical Computing in Dynamic Languages, New Orleans, LA, USA, 17 November 2014; pp. 6–17. [Google Scholar]
- Clémençon, B.; Lüscher, B.P.; Fine, M.; Baumann, M.U.; Surbek, D.V.; Bonny, O.; Hediger, M.A. Expression, purification, and structural insights for the human uric acid transporter, GLUT9, using the Xenopus laevis oocytes system. PLoS ONE 2014, 9, e108852. [Google Scholar] [CrossRef] [Green Version]
- Takanaga, H.; Chaudhuri, B.; Frommer, W.B. GLUT1 and GLUT9 as major contributors to glucose influx in HepG2 cells identified by a high sensitivity intramolecular FRET glucose sensor. Biochim. Biophys. Acta 2008, 1778, 1091–1099. [Google Scholar] [CrossRef] [Green Version]
- Augustin, R.; Carayannopoulos, M.O.; Dowd, L.O.; Phay, J.E.; Moley, J.F.; Moley, K.H. Identification and characterization of human glucose transporter-like protein-9 (GLUT9): Alternative splicing alters trafficking. J. Biol. Chem. 2004, 279, 16229–16236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandal, A.K.; Mercado, A.; Foster, A.; Zandi-Nejad, K.; Mount, D.B. Uricosuric targets of tranilast. Pharm. Res. Perspect. 2017, 5, e00291. [Google Scholar] [CrossRef]
- Anzai, N.; Ichida, K.; Jutabha, P.; Kimura, T.; Babu, E.; Jin, C.J.; Srivastava, S.; Kitamura, K.; Hisatome, I.; Endou, H.; et al. Plasma urate level is directly regulated by a voltage-driven urate efflux transporter URATv1 (SLC2A9) in humans. J. Biol. Chem. 2008, 283, 26834–26838. [Google Scholar] [CrossRef] [Green Version]
- Matsuo, H.; Chiba, T.; Nagamori, S.; Nakayama, A.; Domoto, H.; Phetdee, K.; Wiriyasermkul, P.; Kikuchi, Y.; Oda, T.; Nishiyama, J.; et al. Mutations in glucose transporter 9 gene SLC2A9 cause renal hypouricemia. Am. J. Hum. Genet. 2008, 83, 744–751. [Google Scholar] [CrossRef] [Green Version]
- Dinour, D.; Gray, N.K.; Campbell, S.; Shu, X.; Sawyer, L.; Richardson, W.; Rechavi, G.; Amariglio, N.; Ganon, L.; Sela, B.A.; et al. Homozygous SLC2A9 mutations cause severe renal hypouricemia. J. Am. Soc. Nephrol. 2010, 21, 64–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz, A.; Gautschi, I.; Schild, L.; Bonny, O. Human Mutations in SLC2A9 (Glut9) Affect Transport Capacity for Urate. Front. Physiol. 2018, 9, 476. [Google Scholar] [CrossRef] [PubMed]
- Kottgen, A.; Albrecht, E.; Teumer, A.; Vitart, V.; Krumsiek, J.; Hundertmark, C.; Pistis, G.; Ruggiero, D.; O’Seaghdha, C.M.; Haller, T.; et al. Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat. Genet. 2013, 45, 145–154. [Google Scholar] [CrossRef] [Green Version]
- Sull, J.W.; Park, E.J.; Lee, M.; Jee, S.H. Effects of SLC2A9 variants on uric acid levels in a Korean population. Rheumatol. Int. 2013, 33, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Vitart, V.; Rudan, I.; Hayward, C.; Gray, N.K.; Floyd, J.; Palmer, C.N.; Knott, S.A.; Kolcic, I.; Polasek, O.; Graessler, J.; et al. SLC2A9 is a newly identified urate transporter influencing serum urate concentration, urate excretion and gout. Nat. Genet. 2008, 40, 437–442. [Google Scholar] [CrossRef]
- Meng, Q.; Yue, J.; Shang, M.; Shan, Q.; Qi, J.; Mao, Z.; Li, J.; Zhang, F.; Wang, B.; Zhao, T.; et al. Correlation of GLUT9 Polymorphisms with Gout Risk. Medicine 2015, 94, e1742. [Google Scholar] [CrossRef]
- Dehghan, A.; Köttgen, A.; Yang, Q.; Hwang, S.; Kao, W.L.; Rivadeneira, F.; Boerwinkle, E.; Levy, D.; Hofman, A.; Astor, B.C.; et al. Association of three genetic loci with uric acid concentration and risk of gout: A genome-wide association study. Lancet 2008, 372, 1953–1961. [Google Scholar] [CrossRef] [Green Version]
- Wei, W.H.; Guo, Y.; Kindt, A.S.; Merriman, T.R.; Semple, C.A.; Wang, K.; Haley, C.S. Abundant local interactions in the 4p16.1 region suggest functional mechanisms underlying SLC2A9 associations with human serum uric acid. Hum. Mol. Genet. 2014, 23, 5061–5068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caulfield, M.J.; Munroe, P.B.; O’Neill, D.; Witkowska, K.; Charchar, F.J.; Doblado, M.; Evans, S.; Eyheramendy, S.; Onipinla, A.; Howard, P.; et al. SLC2A9 is a high-capacity urate transporter in humans. PLoS Med. 2008, 5, e197. [Google Scholar] [CrossRef] [PubMed]
- Kohroki, J.; Nishiyama, T.; Nakamura, T.; Masuho, Y. ASB proteins interact with Cullin5 and Rbx2 to form E3 ubiquitin ligase complexes. FEBS Lett. 2005, 579, 6796–6802. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Characteristics | Unexplained Group | SLC2A9 Compound Heterozygote |
|---|---|---|
| n = 6 | n = 1 | |
| Age (years) | 43 ± 12 | 40 |
| BMI † (kg/m2) | 25.1 ± 2.9 | 23.8 |
| Waist circumference, cm | 81 ± 5 | 72 |
| Blood pressure, mmHg | ||
| Systolic | 110 ± 3 | 124 |
| Diastolic | 71 ± 12 | 64 |
| Smoking status | ||
| Never a smoker, no. (%) | 1 (16.67) | 1 (100) |
| Ever a smoker, no. (%) | 5 (83.33) | 0 (0) |
| Alcohol consumption | ||
| Never a drinker, no. (%) | 2 (33.33) | 1 (100) |
| Ever a drinker, no. (%) | 4 (66.67) | 0 (0) |
| Uric acid, mg/dL | 0.78 ± 0.52 | 0.80 |
| Total cholesterol, mg/dL | 214 ± 34 | 174 |
| Triglycerides, mg/dL | 169 ± 69 | 178 |
| Fasting glucose, mg/dL | 86 ± 14 | 92 |
| LDL cholesterol, mg/dL | 116 ± 22 | 90.4 |
| HDL cholesterol, mg/dL | 64 ± 18 | 48 |
| Creatinine, mg/dL | 0.80 ± 0.25 | 0.70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, J.; Cachau, R.; David, V.A.; Thompson, M.; Jung, W.; Jee, S.-H.; Daar, I.O.; Winkler, C.A.; Cho, S.-K. Characterization of a Compound Heterozygous SLC2A9 Mutation That Causes Hypouricemia. Biomedicines 2021, 9, 1172. https://doi.org/10.3390/biomedicines9091172
Yoon J, Cachau R, David VA, Thompson M, Jung W, Jee S-H, Daar IO, Winkler CA, Cho S-K. Characterization of a Compound Heterozygous SLC2A9 Mutation That Causes Hypouricemia. Biomedicines. 2021; 9(9):1172. https://doi.org/10.3390/biomedicines9091172
Chicago/Turabian StyleYoon, Jaeho, Raul Cachau, Victor A. David, Mary Thompson, Wooram Jung, Sun-Ha Jee, Ira O. Daar, Cheryl A. Winkler, and Sung-Kweon Cho. 2021. "Characterization of a Compound Heterozygous SLC2A9 Mutation That Causes Hypouricemia" Biomedicines 9, no. 9: 1172. https://doi.org/10.3390/biomedicines9091172
APA StyleYoon, J., Cachau, R., David, V. A., Thompson, M., Jung, W., Jee, S.-H., Daar, I. O., Winkler, C. A., & Cho, S.-K. (2021). Characterization of a Compound Heterozygous SLC2A9 Mutation That Causes Hypouricemia. Biomedicines, 9(9), 1172. https://doi.org/10.3390/biomedicines9091172






