A Systematic Review of Therapeutic Approaches Used in Experimental Models of Interstitial Cystitis/Bladder Pain Syndrome
Abstract
1. Introduction
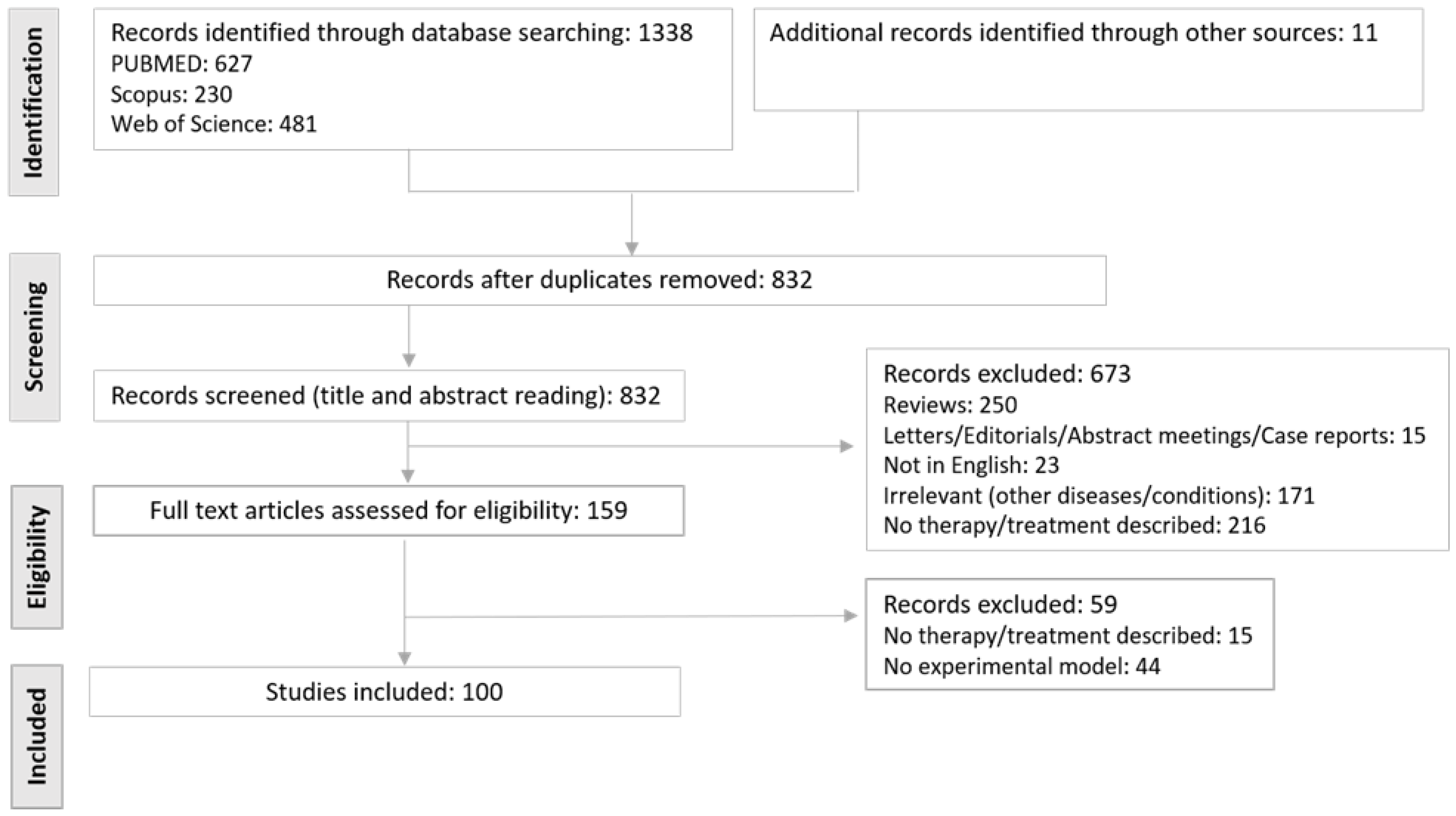
2. Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria and Data Extraction
2.3. Methodological Quality and Risk of Bias
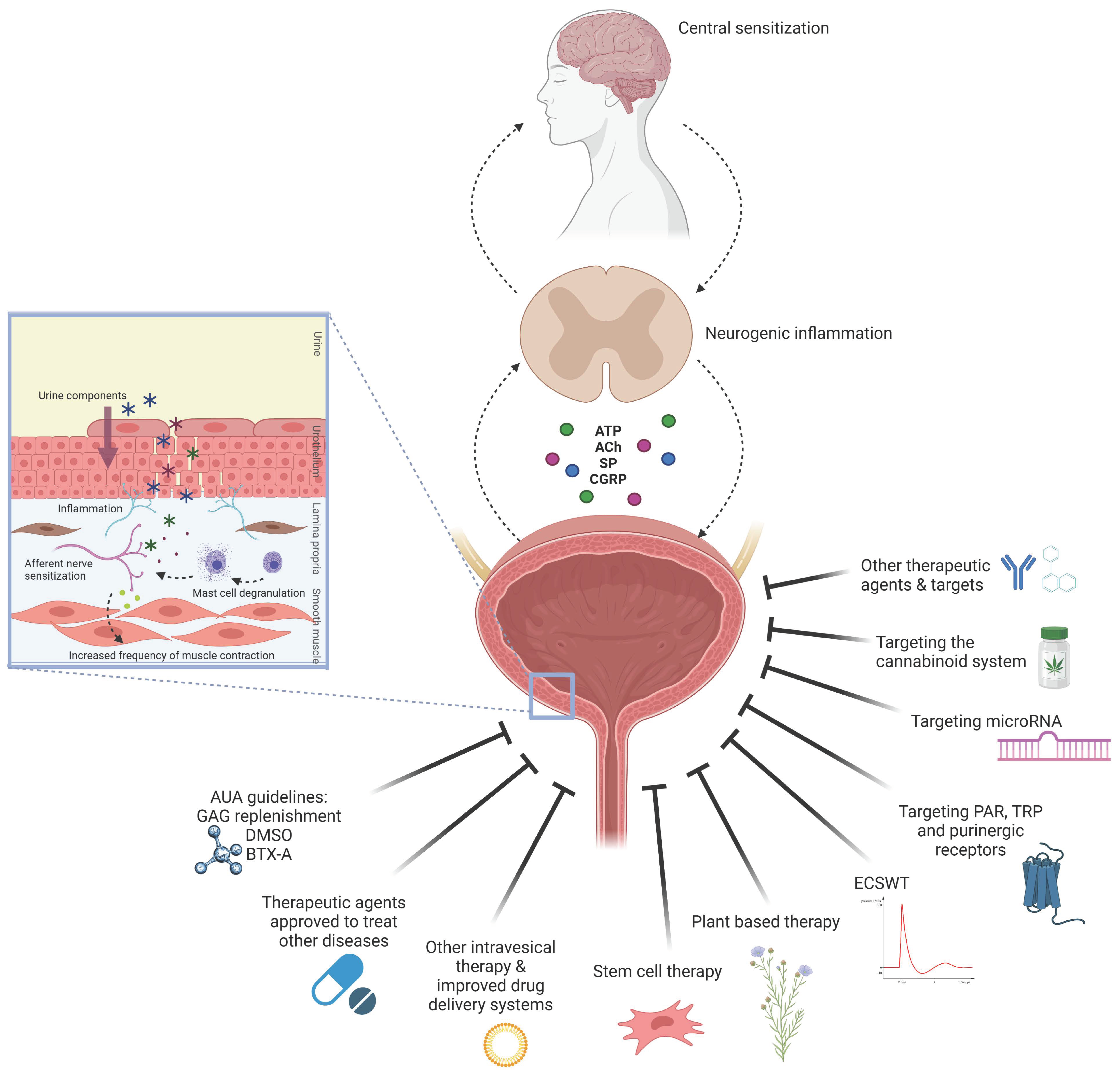
3. Results and Discussion
3.1. Experimental Models of IC/BPS
3.1.1. In Vitro and Ex Vivo Models
3.1.2. In Vivo (Animal) Models
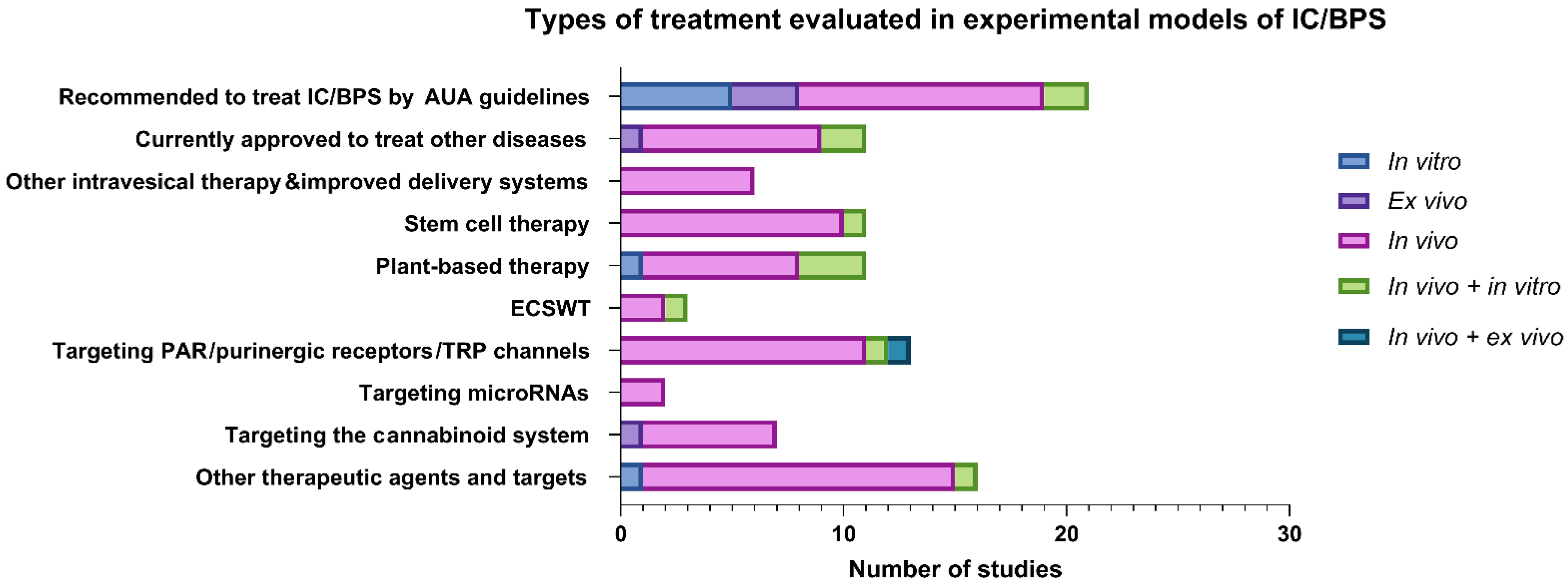
3.2. Types of Treatment Evaluated in Experimental Models
3.2.1. Therapeutic Agents Recommended by AUA Guidelines for Treatment of IC/BPS
Glycosaminoglycan Replenishment Therapy
Dimethyl Sulfoxide (DMSO)
Botulinum Toxin A
3.2.2. Therapeutic Agents Currently Approved to Treat Other Diseases
3.2.3. Other Intravesical Therapies and Improved Drug Delivery Systems
3.2.4. Novel Emerging Therapeutic Options and Targets
Stem-Cell Therapy
Plant-Based Therapy
Extracorporeal Shock Wave Therapy
Targeting Protease-Activated Receptors
Targeting Purinergic Receptors
Targeting Transient Receptor Potential Channels
Targeting microRNA
Activation of the Cannabinoid System
3.3. Methodological Quality and Risk of Bias
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| A2 | adenosine receptor |
| Ab | antibody |
| ACh | acetylcholine |
| ACTH | adrenocorticotropic hormone |
| AJA | ajulemic acid |
| APF | antiproliferative factor |
| ASC | apoptosis-associated speck-like protein |
| A7R5 | smooth muscle cell line |
| AYPGKF-NH2 | PAR4 agonist |
| BCP | beta-caryophyllene |
| BTX-A | botulinum toxin A |
| CAT | catalase |
| CB | cannabinoid receptor |
| CBX | carbenexolone |
| CD | cluster of differentiation |
| CGRP | calcitonin gene-related peptide |
| CRH | corticotropin-releasing hormone |
| CS | chondroitin sulfate |
| CYP | cyclophosphamide |
| DMSO | dimethyl sulfoxide |
| DRG | dorsal root ganglia |
| ECSWT | extracorporeal shock wave therapy |
| EGCG | epigallocatechin gallate |
| eNOS | endothelial nitric oxide synthase |
| EP | prostaglandin E2 receptor subtype |
| ERK | extracellular signal-regulated kinase |
| FcεRIα | high-affinity IgE receptor |
| FTLK | transcriptional factors FOXA1, TP63, MYCL, and KLF4 |
| GAG | glycosaminoglycan |
| GM-0111 | modified GAG |
| GSDMD | gasdermin D |
| GSH | glutathione |
| HA | hyaluronic acid |
| H-BLAK | primary human bladder cell line |
| h-ESC | human embryonic stem cells |
| HMGB1 | high mobility group box 1 |
| HO-1 | heme oxygenase-1 |
| H2O2 | hydrogen peroxide |
| HTB4, HRB2 | human urothelial cells |
| ICAM | intercellular adhesion molecule |
| IC/BPS | interstitial cystitis/bladder pain syndrome |
| ICI | intercontraction interval |
| IFN-γ | interferon gamma |
| IκBα | inhibitor of NF-κB |
| IL | interleukin |
| iNOS | inducible nitric oxide synthase |
| JAK | janus kinase |
| KC | keratinocytes-derived chemokine |
| LDH | lactate dehydrogenase |
| LL37 | antimicrobial peptide |
| LPS | lipopolysaccharide |
| MAPK | mitogen-activated protein kinase |
| MCP-1 | monocyte chemoattractant protein-1 |
| MDA | malondialdehyde |
| MIF | macrophage migration inhibitory factor |
| miR | microRNA |
| MMP9 | matrix metalloproteinase 9 |
| M-MSC | multipotent mesenchymal stem cells |
| MPO | myeloperoxidase |
| MSC | mesenchymal stem cells |
| NF-κB | nuclear factor kappa B |
| NGF | nerve growth factor |
| NK1R | neurokinin 1 receptor |
| NLRP3 | NLR family pyrin domain-containing 3 |
| NMDAR | N-methyl-d-aspartate receptor |
| NOX | NAPDH oxidase |
| NQO-1 | NADPH quinine oxidoreductase |
| NRK-52E | renal tubular epithelial cell line |
| NVC | non-voiding contraction |
| OVX | ovariectomized |
| P1, P2 | purinoceptors |
| P2X | purinergic receptors |
| p38 | mitogen-activated protein kinase |
| p65 | NF-κB subunit |
| p-AKT | protein kinase B, phosphorylated |
| PAR | proteinase-activated receptor |
| PGE2 | prostaglandin 2 |
| PMN | polymorphonuclear cells |
| p-mTOR | mechanistic target of rapamycin, phosphorylated |
| POMC | pro-opiomelanocortin |
| PPAR | peroxisome proliferator-activated receptor |
| PPS | pentosan polysulfate sodium |
| PS | protamine sulfate |
| PTX3 | pentraxin 3 |
| RANTES | chemokine ligand 5 |
| RhoA | Ras homolog gene family, member A |
| rhsTM | recombinant human soluble thrombomodulin |
| ROCK | Rho-associated protein kinase; R |
| OS | reactive oxygen species; |
| RT112 | 3D human bladder epithelium preparation |
| SAGE | semi-synthetic glycosaminoglycan ethers |
| SC | stem cells |
| SD rats | Sprague-Dawley rats |
| sGC | soluble guanylyl cyclase |
| MAPK | mitogen-activated protein kinase |
| MCP-1 | monocyte chemoattractant protein-1 |
| MDA | malondialdehyde |
| SMAD | proteins for signal transduction of the transforming growth factor beta superfamily |
| TRPV | transient receptor potential channel, vanilloid subgroup |
| UPK | uroplakin |
| VEGF | vascular endothelial growth factor |
| ZO-1 | tight junction protein 1 |
References
- Homma, Y.; Ueda, T.; Tomoe, H.; Lin, A.T.; Kuo, H.C.; Lee, M.H.; Oh, S.J.; Kim, J.C.; Lee, K.S. Clinical guidelines for interstitial cystitis and hypersensitive bladder updated in 2015. Int. J. Urol. 2016, 23, 542–549. [Google Scholar] [CrossRef]
- Patnaik, S.S.; Laganà, A.S.; Vitale, S.G.; Butticè, S.; Noventa, M.; Gizzo, S.; Valenti, G.; Rapisarda, A.M.C.; La Rosa, V.L.; Magno, C.; et al. Etiology, pathophysiology and biomarkers of interstitial cystitis/painful bladder syndrome. Arch. Gynecol. Obstet. 2017, 295, 1341–1359. [Google Scholar] [CrossRef] [PubMed]
- Hanno, P.; Lin, A.; Nordling, J.; Nyberg, L.; van Ophoven, A.; Ueda, T.; Wein, A. Bladder Pain Syndrome Committee of the International Consultation on Incontinence. Neurourol. Urodyn. 2010, 29, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Clemens, J.Q.; Mullins, C.; Ackerman, A.L.; Bavendam, T.; van Bokhoven, A.; Ellingson, B.M.; Harte, S.E.; Kutch, J.J.; Lai, H.H.; Martucci, K.T.; et al. Urologic chronic pelvic pain syndrome: Insights from the MAPP Research Network. Nat. Rev. Urol. 2019, 16, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Berry, S.H.; Elliott, M.N.; Suttorp, M.; Bogart, L.M.; Stoto, M.A.; Eggers, P.; Nyberg, L.; Clemens, J.Q. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J. Urol. 2011, 186, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Konkle, K.S.; Berry, S.H.; Elliott, M.N.; Hilton, L.; Suttorp, M.J.; Clauw, D.J.; Clemens, J.Q. Comparison of an interstitial cystitis/bladder pain syndrome clinical cohort with symptomatic community women from the RAND Interstitial Cystitis Epidemiology study. J. Urol. 2012, 187, 508–512. [Google Scholar] [CrossRef]
- Anger, J.T.; Zabihi, N.; Clemens, J.Q.; Payne, C.K.; Saigal, C.S.; Rodriguez, L.V. Treatment choice, duration, and cost in patients with interstitial cystitis and painful bladder syndrome. Int. Urogynecol. J. 2011, 22, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, Y.; Hanno, P. Phenotyping of interstitial cystitis/bladder pain syndrome. Int. J. Urol. 2019, 26 (Suppl. 1), 17–19. [Google Scholar] [CrossRef] [PubMed]
- Warren, J.W.; Howard, F.M.; Cross, R.K.; Good, J.L.; Weissman, M.M.; Wesselmann, U.; Langenberg, P.; Greenberg, P.; Clauw, D.J. Antecedent nonbladder syndromes in case-control study of interstitial cystitis/painful bladder syndrome. Urology 2009, 73, 52–57. [Google Scholar] [CrossRef]
- Doiron, R.C.; Tolls, V.; Irvine-Bird, K.; Kelly, K.L.; Nickel, J.C. Clinical Phenotyping Does Not Differentiate Hunner Lesion Subtype of Interstitial Cystitis/Bladder Pain Syndrome: A Relook at the Role of Cystoscopy. J. Urol. 2016, 196, 1136–1140. [Google Scholar] [CrossRef] [PubMed]
- Birder, L.A. Pathophysiology of interstitial cystitis. Int. J. Urol. 2019, 26 (Suppl. 1), 12–15. [Google Scholar] [CrossRef]
- Lin, Z.; Hu, H.; Liu, B.; Chen, Y.; Tao, Y.; Zhou, X.; Li, M. Biomaterial-assisted drug delivery for interstitial cystitis/bladder pain syndrome treatment. J. Mater. Chem. B. 2021, 9, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Grundy, L.; Caldwell, A.; Brierley, S.M. Mechanisms Underlying Overactive Bladder and Interstitial Cystitis/Painful Bladder Syndrome. Front. Neurosci. 2018, 12, 931. [Google Scholar] [CrossRef]
- Hurst, R.E. Structure, function, and pathology of proteoglycans and glycosaminoglycans in the urinary tract. World J. Urol. 1994, 12, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Keay, S. Cell signaling in interstitial cystitis/painful bladder syndrome. Cell Signal. 2008, 20, 2174–2179. [Google Scholar] [CrossRef] [PubMed]
- Slobodov, G.; Feloney, M.; Gran, C.; Kyker, K.D.; Hurst, R.E.; Culkin, D.J. Abnormal expression of molecular markers for bladder impermeability and differentiation in the urothelium of patients with interstitial cystitis. J. Urol. 2004, 171, 1554–1558. [Google Scholar] [CrossRef]
- Liu, H.T.; Shie, J.H.; Chen, S.H.; Wang, Y.S.; Kuo, H.C. Differences in mast cell infiltration, E-cadherin, and zonula occludens-1 expression between patients with overactive bladder and interstitial cystitis/bladder pain syndrome. Urology 2012, 80, 225.e13–225.e18. [Google Scholar] [CrossRef] [PubMed]
- Parsons, C.L. The role of a leaky epithelium and potassium in the generation of bladder symptoms in interstitial cystitis/overactive bladder, urethral syndrome, prostatitis and gynaecological chronic pelvic pain. BJU Int. 2011, 107, 370–375. [Google Scholar] [CrossRef]
- Winder, M.; Tobin, G.; Zupančič, D.; Romih, R. Signalling molecules in the urothelium. Biomed. Res. Int. 2014, 2014, 297295. [Google Scholar] [CrossRef]
- Liu, H.T.; Tyagi, P.; Chancellor, M.B.; Kuo, H.C. Urinary nerve growth factor level is increased in patients with interstitial cystitis/bladder pain syndrome and decreased in responders to treatment. BJU Int. 2009, 104, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Searl, T.J.; Yaggie, R.; Schaeffer, A.J.; Klumpp, D.J. A MAPP Network study: Overexpression of tumor necrosis factor-α in mouse urothelium mimics interstitial cystitis. Am. J. Physiol. Renal. Physiol. 2018, 315, F36–F44. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.H.; Jhang, J.F.; Hsu, Y.H.; Ho, H.C.; Wu, Y.H.; Kuo, H.C. Urine cytokines as biomarkers for diagnosing interstitial cystitis/bladder pain syndrome and mapping its clinical characteristics. Am. J. Physiol. Renal. Physiol. 2020, 318, F1391–F1399. [Google Scholar] [CrossRef]
- Jiang, Y.H.; Jhang, J.F.; Hsu, Y.H.; Ho, H.C.; Wu, Y.H.; Kuo, H.C. Urine biomarkers in ESSIC type 2 interstitial cystitis/bladder pain syndrome and overactive bladder with developing a novel diagnostic algorithm. Sci. Rep. 2021, 11, 914. [Google Scholar] [CrossRef] [PubMed]
- Hanno, P.M.; Erickson, D.; Moldwin, R.; Faraday, M.M. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: AUA guideline amendment. J. Urol. 2015, 193, 1545–1553. [Google Scholar] [CrossRef]
- Hooijmans, C.R.; Ritskes-Hoitinga, M. Progress in using systematic reviews of animal studies to improve translational research. PLoS. Med. 2013, 10, e1001482. [Google Scholar] [CrossRef] [PubMed]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS. Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef]
- Hauser, P.J.; Dozmorov, M.G.; Bane, B.L.; Slobodov, G.; Culkin, D.J.; Hurst, R.E. Abnormal expression of differentiation related proteins and proteoglycan core proteins in the urothelium of patients with interstitial cystitis. J. Urol. 2008, 179, 764–769. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kullmann, F.A.; Daugherty, S.L.; de Groat, W.C.; Birder, L.A. Bladder smooth muscle strip contractility as a method to evaluate lower urinary tract pharmacology. J. Vis. Exp. 2014, e51807. [Google Scholar] [CrossRef]
- Birder, L.; Andersson, K.E. Animal Modelling of Interstitial Cystitis/Bladder Pain Syndrome. Int. Neurourol. J. 2018, 22, S3-9. [Google Scholar] [CrossRef]
- Wyndaele, J.J.J.; Riedl, C.; Taneja, R.; Lovasz, S.; Ueda, T.; Cervigni, M. GAG replenishment therapy for bladder pain syndrome/interstitial cystitis. Neurourol. Urodyn. 2019, 38, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Rozenberg, B.B.; Janssen, D.A.W.; Jansen, C.F.J.; Schalken, J.A.; Heesakkers, J. Improving the barrier function of damaged cultured urothelium using chondroitin sulfate. Neurourol. Urodyn. 2020, 39, 558–564. [Google Scholar] [CrossRef]
- Rooney, P.; Srivastava, A.; Watson, L.; Quinlan, L.R.; Pandit, A. Hyaluronic acid decreases IL-6 and IL-8 secretion and permeability in an inflammatory model of interstitial cystitis. Acta. Biomater. 2015, 19, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Stellavato, A.; Pirozzi, A.V.A.; Diana, P.; Reale, S.; Vassallo, V.; Fusco, A.; Donnarumma, G.; De Rosa, M.; Schiraldi, C. Hyaluronic acid and chondroitin sulfate, alone or in combination, efficiently counteract induced bladder cell damage and inflammation. PLoS ONE 2019, 14, e0218475. [Google Scholar] [CrossRef] [PubMed]
- Rooney, P.; Ryan, C.; McDermott, B.J.; Dev, K.; Pandit, A.; Quinlan, L.R. Effect of Glycosaminoglycan Replacement on Markers of Interstitial Cystitis In Vitro. Front. Pharmacol. 2020, 11, 575043. [Google Scholar] [CrossRef]
- Rooney, P.R.; Kannala, V.K.; Kotla, N.G.; Benito, A.; Dupin, D.; Loinaz, I.; Quinlan, L.R.; Rochev, Y.; Pandit, A. A high molecular weight hyaluronic acid biphasic dispersion as potential therapeutics for interstitial cystitis. J. Biomed. Mater. Res. B. Appl. Biomater. 2021, 109, 864–876. [Google Scholar] [CrossRef]
- Yeh, C.H.; Chiang, H.S.; Chien, C.T. Hyaluronic acid ameliorates bladder hyperactivity via the inhibition of H2O2-enhanced purinergic and muscarinic signaling in the rat. Neurourol. Urodyn. 2010, 29, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Hauser, P.J.; Buethe, D.A.; Califano, J.; Sofinowski, T.M.; Culkin, D.J.; Hurst, R.E. Restoring barrier function to acid damaged bladder by intravesical chondroitin sulfate. J. Urol. 2009, 182, 2477–2482. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Engles, C.D.; Hauser, P.J.; Abdullah, S.N.; Culkin, D.J.; Hurst, R.E. Intravesical chondroitin sulfate inhibits recruitment of inflammatory cells in an acute acid damage "leaky bladder" model of cystitis. Urology 2012, 79, 483.e13–483.e17. [Google Scholar] [CrossRef][Green Version]
- Rajasekaran, M.; Stein, P.; Parsons, C.L. Toxic factors in human urine that injure urothelium. Int. J. Urol. 2006, 13, 409–414. [Google Scholar] [CrossRef]
- Hurst, R.E.; Zebrowski, R. Identification of proteoglycans present at high density on bovine and human bladder luminal surface. J. Urol. 1994, 152, 1641–1645. [Google Scholar] [CrossRef]
- Greenwood-Van Meerveld, B.; Mohammadi, E.; Latorre, R.; Truitt, E.R., 3rd; Jay, G.D.; Sullivan, B.D.; Schmidt, T.A.; Smith, N.; Saunders, D.; Ziegler, J.; et al. Preclinical Animal Studies of Intravesical Recombinant Human Proteoglycan 4 as a Novel Potential Therapy for Diseases Resulting From Increased Bladder Permeability. Urology 2018, 116, 230.e231–230.e237. [Google Scholar] [CrossRef] [PubMed]
- Towner, R.A.; Greenwood-Van Meerveld, B.; Mohammadi, E.; Saunders, D.; Smith, N.; Sant, G.R.; Shain, H.C.; Jozefiak, T.H.; Hurst, R.E. SuperGAG biopolymers for treatment of excessive bladder permeability. Pharmacol. Res. Perspect. 2021, 9, e00709. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.M.; Jia, W.; Schults, A.J.; Isaacson, K.J.; Steinhauff, D.; Green, B.; Zachary, B.; Cappello, J.; Ghandehari, H.; Oottamasathien, S. Temperature-responsive silk-elastinlike protein polymer enhancement of intravesical drug delivery of a therapeutic glycosaminoglycan for treatment of interstitial cystitis/painful bladder syndrome. Biomaterials 2019, 217, 119293. [Google Scholar] [CrossRef]
- Oottamasathien, S.; Jia, W.J.; McCoard, L.; Slack, S.; Zhang, J.X.; Skardal, A.; Job, K.; Kennedy, T.P.; Dull, R.O.; Prestwich, G.D. A Murine Model of Inflammatory Bladder Disease: Cathelicidin Peptide Induced Bladder Inflammation and Treatment With Sulfated Polysaccharides. J. Urol. 2011, 186, 1684–1692. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.Y.; Savage, J.R.; Zhang, J.X.; Jia, W.J.; Oottamasathien, S.; Prestwich, G.D. Prevention of Anti-microbial Peptide LL-37-Induced Apoptosis and ATP Release in the Urinary Bladder by a Modified Glycosaminoglycan. PLoS ONE 2013, 8, e77854. [Google Scholar] [CrossRef]
- Rawls, W.F.; Cox, L.; Rovner, E.S. Dimethyl sulfoxide (DMSO) as intravesical therapy for interstitial cystitis/bladder pain syndrome: A review. Neurourol. Urodyn. 2017, 36, 1677–1684. [Google Scholar] [CrossRef]
- Melchior, D.; Packer, C.S.; Johnson, T.C.; Kaefer, M. Dimethyl sulfoxide: Does it change the functional properties of the bladder wall? J. Urol. 2003, 170, 253–258. [Google Scholar] [CrossRef]
- Kim, R.; Liu, W.; Chen, X.; Kreder, K.J.; Luo, Y. Intravesical dimethyl sulfoxide inhibits acute and chronic bladder inflammation in transgenic experimental autoimmune cystitis models. J. Biomed. Biotechnol. 2011, 2011, 937061. [Google Scholar] [CrossRef]
- Soler, R.; Bruschini, H.; Truzzi, J.C.; Martins, J.R.; Camara, N.O.; Alves, M.T.; Leite, K.R.; Nader, H.B.; Srougi, M.; Ortiz, V. Urinary glycosaminoglycans excretion and the effect of dimethyl sulfoxide in an experimental model of non-bacterial cystitis. Int. Braz. J. Urol. 2008, 34, 503–511; discussion 511. [Google Scholar] [CrossRef]
- Rössberger, J.; Fall, M.; Peeker, R. Critical appraisal of dimethyl sulfoxide treatment for interstitial cystitis: Discomfort, side-effects and treatment outcome. Scand. J. Urol. Nephrol. 2005, 39, 73–77. [Google Scholar] [CrossRef]
- Erbguth, F.J. Historical notes on botulism, Clostridium botulinum, botulinum toxin, and the idea of the therapeutic use of the toxin. Mov. Disord. 2004, 19 (Suppl. 8), S2–S6. [Google Scholar] [CrossRef] [PubMed]
- Reitz, A.; Stohrer, M.; Kramer, G.; Del Popolo, G.; Chartier-Kastler, E.; Pannek, J.; Burgdorfer, H.; Gocking, K.; Madersbacher, H.; Schumacher, S.; et al. European experience of 200 cases treated with botulinum-A toxin injections into the detrusor muscle for urinary incontinence due to neurogenic detrusor overactivity. Eur. Urol. 2004, 45, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Kuo, H.C. Clinical application of intravesical botulinum toxin type A for overactive bladder and interstitial cystitis. Investig Clin. Urol. 2020, 61, S33–S42. [Google Scholar] [CrossRef]
- Dan Spinu, A.; Gabriel Bratu, O.; Cristina Diaconu, C.; Maria Alexandra Stanescu, A.; Bungau, S.; Fratila, O.; Bohiltea, R.; Liviu Dorel Mischianu, D. Botulinum toxin in low urinary tract disorders—Over 30 years of practice. Exp. Ther. Med. 2020, 20, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Rapp, D.E.; Turk, K.W.; Bales, G.T.; Cook, S.P. Botulinum toxin type a inhibits calcitonin gene-related peptide release from isolated rat bladder. J. Urol. 2006, 175, 1138–1142. [Google Scholar] [CrossRef]
- Lucioni, A.; Bales, G.T.; Lotan, T.L.; McGehee, D.S.; Cook, S.P.; Rapp, D.E. Botulinum toxin type A inhibits sensory neuropeptide release in rat bladder models of acute injury and chronic inflammation. BJU Int. 2008, 101, 366–370. [Google Scholar] [CrossRef]
- Smith, C.P.; Vemulakonda, V.M.; Kiss, S.; Boone, T.B.; Somogyi, G.T. Enhanced ATP release from rat bladder urothelium during chronic bladder inflammation: Effect of botulinum toxin A. Neurochem. Int. 2005, 47, 291–297. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Yoshimura, N.; Huang, C.C.; Wu, M.; Chiang, P.H.; Chancellor, M.B. Intravesical botulinum toxin A administration inhibits COX-2 and EP4 expression and suppresses bladder hyperactivity in cyclophosphamide-induced cystitis in rats. Eur. Urol. 2009, 56, 159–166. [Google Scholar] [CrossRef]
- Bicer, F.; Altuntas, C.Z.; Izgi, K.; Ozer, A.; Kavran, M.; Tuohy, V.K.; Daneshgari, F. Chronic pelvic allodynia is mediated by CCL2 through mast cells in an experimental autoimmune cystitis model. Am. J. Physiol. Renal Physiol. 2015, 308, F103–F113. [Google Scholar] [CrossRef]
- Grundy, L.; Caldwell, A.; Garcia Caraballo, S.; Erickson, A.; Schober, G.; Castro, J.; Harrington, A.M.; Brierley, S.M. Histamine induces peripheral and central hypersensitivity to bladder distension via the histamine H(1) receptor and TRPV1. Am. J. Physiol. Renal Physiol. 2020, 318, F298–F314. [Google Scholar] [CrossRef]
- Çetinel, S.; Çanıllıoğlu, Y.E.; Çikler, E.; Sener, G.; Ercan, F. Leukotriene D4 receptor antagonist montelukast alleviates protamine sulphate-induced changes in rat urinary bladder. BJU Int. 2011, 107, 1320–1325. [Google Scholar] [CrossRef]
- Zhang, X.L.; Gao, S.; Tanaka, M.; Zhang, Z.; Huang, Y.R.; Mitsui, T.; Kamiyama, M.; Koizumi, S.; Fan, J.L.; Takeda, M.; et al. Carbenoxolone inhibits TRPV4 channel-initiated oxidative urothelial injury and ameliorates cyclophosphamide-induced bladder dysfunction. J. Cell. Mol. Med. 2017, 21, 1791–1802. [Google Scholar] [CrossRef] [PubMed]
- Funahashi, Y.; Yoshida, M.; Yamamoto, T.; Majima, T.; Takai, S.; Gotoh, M. Intravesical application of rebamipide promotes urothelial healing in a rat cystitis model. J. Urol. 2014, 192, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Ichihara, K.; Aizawa, N.; Akiyama, Y.; Kamei, J.; Masumori, N.; Andersson, K.E.; Homma, Y.; Igawa, Y. Toll-like receptor 7 is overexpressed in the bladder of Hunner-type interstitial cystitis, and its activation in the mouse bladder can induce cystitis and bladder pain. Pain 2017, 158, 1538–1545. [Google Scholar] [CrossRef] [PubMed]
- Boucher, W.; Stern, J.M.; Kotsinyan, V.; Kempuraj, D.; Papaliodis, D.; Cohen, M.S.; Theoharides, T.C. Intravesical nanocrystalline silver decreases experimental bladder inflammation. J. Urol. 2008, 179, 1598–1602. [Google Scholar] [CrossRef]
- Boudieu, L.; Mountadem, S.; Lashermes, A.; Meleine, M.; Ulmann, L.; Rassendren, F.; Aissouni, Y.; Sion, B.; Carvalho, F.A.; Ardid, D. Blocking α(2)δ-1 Subunit Reduces Bladder Hypersensitivity and Inflammation in a Cystitis Mouse Model by Decreasing NF-kB Pathway Activation. Front. Pharmacol. 2019, 10, 133. [Google Scholar] [CrossRef]
- Yoshizumi, M.; Watanabe, C.; Mizoguchi, H. Gabapentin reduces painful bladder hypersensitivity in rats with lipopolysaccharide-induced chronic cystitis. Pharmacol. Res. Perspect. 2021, 9, e00697. [Google Scholar] [CrossRef]
- Holschneider, D.P.; Wang, Z.; Chang, H.Y.; Zhang, R.; Gao, Y.L.; Guo, Y.M.; Mao, J.; Rodriguez, L.V. Ceftriaxone inhibits stress-induced bladder hyperalgesia and alters cerebral micturition and nociceptive circuits in the rat: A multidisciplinary approach to the study of urologic chronic pelvic pain syndrome research network study. Neurourol. Urodyn. 2020, 39, 1628–1643. [Google Scholar] [CrossRef]
- Liu, B.K.; Jin, X.W.; Lu, H.Z.; Zhang, X.; Zhao, Z.H.; Shao, Y. The Effects of Neurokinin-1 Receptor Antagonist in an Experimental Autoimmune Cystitis Model Resembling Bladder Pain Syndrome/Interstitial Cystitis. Inflammation 2019, 42, 246–254. [Google Scholar] [CrossRef]
- Fraser, M.O.; Chuang, Y.C.; Tyagi, P.; Yokoyama, T.; Yoshimura, N.; Huang, L.; De Groat, W.C.; Chancellor, M.B. Intravesical liposome administration--a novel treatment for hyperactive bladder in the rat. Urology 2003, 61, 656–663. [Google Scholar] [CrossRef]
- Tyagi, P.; Chancellor, M.; Yoshimura, N.; Huang, L. Activity of different phospholipids in attenuating hyperactivity in bladder irritation. BJU Int. 2008, 101, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Hsieh, V.C.; Yoshimura, N.; Kaufman, J.; Chancellor, M.B. Instillation of liposomes vs dimethyl sulphoxide or pentosan polysulphate for reducing bladder hyperactivity. BJU Int. 2009, 104, 1689–1692. [Google Scholar] [CrossRef]
- Lin, T.S.; Zhao, X.Z.; Zhang, Y.F.; Lian, H.B.; Zhuang, J.L.; Zhang, Q.; Chen, W.; Wang, W.; Liu, G.X.; Guo, S.H.; et al. Floating Hydrogel with Self-Generating Micro-Bubbles for Intravesical Instillation. Materials 2016, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Rappaport, Y.H.; Zisman, A.; Jeshurun-Gutshtat, M.; Gerassi, T.; Hakim, G.; Vinshtok, Y.; Stav, K. Safety and Feasibility of Intravesical Instillation of Botulinum Toxin-A in Hydrogel-based Slow-release Delivery System in Patients With Interstitial Cystitis-Bladder Pain Syndrome: A Pilot Study. Urology 2018, 114, 60–65. [Google Scholar] [CrossRef]
- Lee, H.; Cima, M.J. An intravesical device for the sustained delivery of lidocaine to the bladder. J. Control. Release 2011, 149, 133–139. [Google Scholar] [CrossRef]
- Xu, X.; Goyanes, A.; Trenfield, S.J.; Diaz-Gomez, L.; Alvarez-Lorenzo, C.; Gaisford, S.; Basit, A.W. Stereolithography (SLA) 3D printing of a bladder device for intravesical drug delivery. Mater. Sci. Eng. C. Mater. Biol. Appl. 2021, 120, 111773. [Google Scholar] [CrossRef] [PubMed]
- Lupo, N.; Jalil, A.; Nazir, I.; Gust, R.; Bernkop-Schnürch, A. In vitro evaluation of intravesical mucoadhesive self-emulsifying drug delivery systems. Int. J Pharm. 2019, 564, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Kolawole, O.M.; Lau, W.M.; Khutoryanskiy, V.V. Methacrylated chitosan as a polymer with enhanced mucoadhesive properties for transmucosal drug delivery. Int. J. Pharm. 2018, 550, 123–129. [Google Scholar] [CrossRef]
- Ali, M.S.; Metwally, A.A.; Fahmy, R.H.; Osman, R. Chitosan-coated nanodiamonds: Mucoadhesive platform for intravesical delivery of doxorubicin. Carbohydr. Polym. 2020, 245, 116528. [Google Scholar] [CrossRef]
- Li, G.; He, S.; Schätzlein, A.G.; Weiss, R.M.; Martin, D.T.; Uchegbu, I.F. Achieving highly efficient gene transfer to the bladder by increasing the molecular weight of polymer-based nanoparticles. J. Control. Release 2021, 332, 210–224. [Google Scholar] [CrossRef]
- Dayem, A.A.; Kim, K.; Lee, S.B.; Kim, A.; Cho, S.G. Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives. J. Clin. Med. 2020, 9, 766. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Lim, J.; Yu, H.Y.; Park, J.; Chun, J.Y.; Jeong, J.; Heo, J.; Kang, H.; Kim, Y.; Cho, Y.M.; et al. Mesenchymal Stem Cell Therapy Alleviates Interstitial Cystitis by Activating Wnt Signaling Pathway. Stem. Cells 2015, 24, 1648–1657. [Google Scholar] [CrossRef]
- Hirose, Y.; Yamamoto, T.; Nakashima, M.; Funahashi, Y.; Matsukawa, Y.; Yamaguchi, M.; Kawabata, S.; Gotoh, M. Injection of Dental Pulp Stem Cells Promotes Healing of Damaged Bladder Tissue in a Rat Model of Chemically Induced Cystitis. Cell Transplant. 2016, 25, 425–436. [Google Scholar] [CrossRef]
- Furuta, A.; Yamamoto, T.; Igarashi, T.; Suzuki, Y.; Egawa, S.; Yoshimura, N. Bladder wall injection of mesenchymal stem cells ameliorates bladder inflammation, overactivity, and nociception in a chemically induced interstitial cystitis-like rat model. Int. Urogynecol. J. 2018, 29, 1615–1622. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Chun, S.Y.; Lee, E.H.; Chung, J.W.; Lee, J.N.; Ha, Y.S.; Choi, J.Y.; Song, P.H.; Kwon, T.G.; Han, M.H.; et al. Efficacy of combination therapy with pentosan polysulfate sodium and adipose tissue-derived stem cells for the management of interstitial cystitis in a rat model. Stem. Cell. Res. 2020, 45, 101801. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, S.; Liu, G.; Shi, Y.; Wu, R.; Yang, B.; He, T.; Fan, Y.; Lu, X.; Zhou, X.; Liu, H.; et al. Multipotential differentiation of human urine-derived stem cells: Potential for therapeutic applications in urology. Stem. Cells. 2013, 31, 1840–1856. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Luo, H.; Dong, X.; Liu, Q.; Wu, C.; Zhang, T.; Hu, X.; Zhang, Y.; Song, B.; Li, L. Therapeutic effect of urine-derived stem cells for protamine/lipopolysaccharide-induced interstitial cystitis in a rat model. Stem. Cell. Res. Ther. 2017, 8, 107. [Google Scholar] [CrossRef]
- Chung, J.W.; Chun, S.Y.; Lee, E.H.; Ha, Y.S.; Lee, J.N.; Song, P.H.; Yoo, E.S.; Kwon, T.G.; Chung, S.K.; Kim, B.S. Verification of mesenchymal stem cell injection therapy for interstitial cystitis in a rat model. PLoS ONE 2019, 14, e0226390. [Google Scholar] [CrossRef]
- Rusu, E.; Necula, L.; Neagu, A.; Alecu, M.; Stan, C.; Albulescu, R.; Tanase, C. Current status of stem cell therapy: Opportunities and limitations. Turk. J. Biol. 2016, 40, 955–967. [Google Scholar] [CrossRef]
- Kim, A.; Yu, H.Y.; Lim, J.; Ryu, C.M.; Kim, Y.H.; Heo, J.; Han, J.Y.; Lee, S.; Bae, Y.S.; Kim, J.Y.; et al. Improved efficacy and in vivo cellular properties of human embryonic stem cell derivative in a preclinical model of bladder pain syndrome. Sci. Rep. 2017, 7, 8872. [Google Scholar] [CrossRef]
- Lee, S.W.; Ryu, C.M.; Shin, J.H.; Choi, D.; Kim, A.; Yu, H.Y.; Han, J.Y.; Lee, H.Y.; Lim, J.; Kim, Y.H.; et al. The Therapeutic Effect of Human Embryonic Stem Cell-Derived Multipotent Mesenchymal Stem Cells on Chemical-Induced Cystitis in Rats. Int. Neurourol. J. 2018, 22, S34–S45. [Google Scholar] [CrossRef]
- Ryu, C.M.; Yu, H.Y.; Lee, H.Y.; Shin, J.H.; Lee, S.; Ju, H.; Paulson, B.; Lee, S.; Kim, S.; Lim, J.; et al. Longitudinal intravital imaging of transplanted mesenchymal stem cells elucidates their functional integration and therapeutic potency in an animal model of interstitial cystitis/bladder pain syndrome. Theranostics 2018, 8, 5610–5624. [Google Scholar] [CrossRef]
- Huang, P.; He, Z.; Ji, S.; Sun, H.; Xiang, D.; Liu, C.; Hu, Y.; Wang, X.; Hui, L. Induction of functional hepatocyte-like cells from mouse fibroblasts by defined factors. Nature 2011, 475, 386–389. [Google Scholar] [CrossRef]
- Inoue, Y.; Kishida, T.; Kotani, S.I.; Akiyoshi, M.; Taga, H.; Seki, M.; Ukimura, O.; Mazda, O. Direct conversion of fibroblasts into urothelial cells that may be recruited to regenerating mucosa of injured urinary bladder. Sci. Rep. 2019, 9, 13850. [Google Scholar] [CrossRef]
- Wang, X.; Fan, L.; Yin, H.; Zhou, Y.; Tang, X.; Fei, X.; Tang, H.; Peng, J.; Ren, X.; Xue, Y.; et al. Protective effect of Aster tataricus extract on NLRP3-mediated pyroptosis of bladder urothelial cells . J. Cell Mol. Med. 2020, 24, 13336–13345. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yin, H.; Fan, L.; Zhou, Y.Q.; Tang, X.L.; Fei, X.J.; Tang, H.L.; Peng, J.; Zhang, J.J.; Xue, Y.; et al. Shionone alleviates NLRP3 inflammasome mediated pyroptosis in interstitial cystitis injury. Int. Immunopharmacol. 2021, 90, 107132. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Lee, W.T.; Liang, C.C.; Lo, T.S.; Hsieh, W.C.; Lin, Y.H. Beneficial effect of Bletilla striata extract solution on zymosan-induced interstitial cystitis in rat. Neurourol. Urodyn. 2021, 40, 763–770. [Google Scholar] [CrossRef]
- Nassir, A.M.; Ibrahim, I.A.A.; Afify, M.A.; ElSawy, N.A.; Imam, M.T.; Shaheen, M.H.; Basyuni, M.A.; Bader, A.; Azhar, R.A.; Shahzad, N. Olea europaea subsp. Cuspidata and Juniperus procera Hydroalcoholic Leaves’ Extracts Modulate Stress Hormones in Stress-Induced Cystitis in Rats. Urol. Sci. 2019, 30, 151–156. [Google Scholar] [CrossRef]
- Li, W.; Yang, F.; Zhan, H.; Liu, B.; Cai, J.; Luo, Y.; Zhou, X. Houttuynia cordata Extract Ameliorates Bladder Damage and Improves Bladder Symptoms via Anti-Inflammatory Effect in Rats with Interstitial Cystitis. Evid. Based. Complement. Alternat. Med. 2020, 2020, 9026901. [Google Scholar] [CrossRef] [PubMed]
- Bazi, T.; Hajj-Hussein, I.A.; Awwad, J.; Shams, A.; Hijaz, M.; Jurjus, A. A modulating effect of epigallocatechin gallate (EGCG), a tea catechin, on the bladder of rats exposed to water avoidance stress. Neurourol. Urodyn. 2013, 32, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Xu, Y.F.; Feng, Y.; Yang, F.Q.; Luo, J.; Zhai, W.; Che, J.P.; Wang, G.C.; Zheng, J.H. Epigallocatechin gallate attenuates interstitial cystitis in human bladder urothelium cells by modulating purinergic receptors. J. Surg. Res. 2013, 183, 397–404. [Google Scholar] [CrossRef]
- Ostardo, E.; Impellizzeri, D.; Cervigni, M.; Porru, D.; Sommariva, M.; Cordaro, M.; Siracusa, R.; Fusco, R.; Gugliandolo, E.; Crupi, R.; et al. Adelmidrol plus sodium hyaluronate in IC/BPS or conditions associated to chronic urothelial inflammation. A translational study. Pharmacol. Res. 2018, 134, 16–30. [Google Scholar] [CrossRef]
- Luo, J.; Yang, C.; Luo, X.; Yang, Y.; Li, J.; Song, B.; Zhao, J.; Li, L. Chlorogenic acid attenuates cyclophosphamide-induced rat interstitial cystitis. Life Sci. 2020, 254, 117590. [Google Scholar] [CrossRef] [PubMed]
- Shih, H.J.; Chang, C.Y.; Lai, C.H.; Huang, C.J. Therapeutic effect of modulating the NLRP3-regulated transforming growth factor-β signaling pathway on interstitial cystitis/bladder pain syndrome. Biomed. Pharmacother. 2021, 138, 111522. [Google Scholar] [CrossRef]
- d′Agostino, M.C.; Craig, K.; Tibalt, E.; Respizzi, S. Shock wave as biological therapeutic tool: From mechanical stimulation to recovery and healing, through mechanotransduction. Int. J. Surg. 2015, 24, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J. An overview of shock wave therapy in musculoskeletal disorders. Chang. Gung. Med. J. 2003, 26, 220–232. [Google Scholar] [PubMed]
- Chen, Y.T.; Yang, C.C.; Sun, C.K.; Chiang, H.J.; Chen, Y.L.; Sung, P.H.; Zhen, Y.Y.; Huang, T.H.; Chang, C.L.; Chen, H.H.; et al. Extracorporeal shock wave therapy ameliorates cyclophosphamide-induced rat acute interstitial cystitis though inhibiting infammation and oxidative stress-in vitro and in vivo experiment studies. Am. J. Transl. Res. 2014, 6, 631–648. [Google Scholar]
- Wang, H.J.; Lee, W.C.; Tyagi, P.; Huang, C.C.; Chuang, Y.C. Effects of low energy shock wave therapy on inflammatory moleculars, bladder pain, and bladder function in a rat cystitis model. Neurourol. Urodyn. 2017, 36, 1440–1447. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Z.; Peng, J.; Xin, Z.; Li, M.; Yang, B.; Fang, D.; Tang, Y.; Guo, Y. Treatment with low-energy shock wave alleviates pain in an animal model of uroplakin 3A-induced autoimmune interstitial cystitis/painful bladder syndrome. Investig. Clin. Urol. 2019, 60, 359–366. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Huang, T.L.; Tyagi, P.; Huang, C.C. Urodynamic and Immunohistochemical Evaluation of Intravesical Botulinum Toxin A Delivery Using Low Energy Shock Waves. J. Urol. 2016, 196, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Nageib, M.; Zahran, M.H.; El-Hefnawy, A.S.; Barakat, N.; Awadalla, A.; Aamer, H.G.; Khater, S.; Shokeir, A.A. Low energy shock wave-delivered intravesical botulinum neurotoxin-A potentiates antioxidant genes and inhibits proinflammatory cytokines in rat model of overactive bladder. Neurourol. Urodyn. 2020, 39, 2447–2454. [Google Scholar] [CrossRef] [PubMed]
- Rayegani, S.M.; Razzaghi, M.R.; Raeissadat, S.A.; Allameh, F.; Eliaspour, D.; Abedi, A.R.; Javadi, A.; Rahavian, A.H. Extracorporeal Shockwave Therapy Combined with Drug Therapy in Chronic Pelvic Pain Syndrome: A Randomized Clinical Trial. Urol. J. 2020, 17, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Skaudickas, D.; Telksnys, T.; Veikutis, V.; Aniulis, P.; Jievaltas, M. Extracorporeal shock wave therapy for the treatment of chronic pelvic pain syndrome. Open Med. 2020, 15, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.C.; Meng, E.; Chancellor, M.; Kuo, H.C. Pain reduction realized with extracorporeal shock wave therapy for the treatment of symptoms associated with interstitial cystitis/bladder pain syndrome-A prospective, multicenter, randomized, double-blind, placebo-controlled study. Neurourol. Urodyn. 2020, 39, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Saban, R.; D′Andrea, M.R.; Andrade-Gordon, P.; Derian, C.K.; Dozmorov, I.; Ihnat, M.A.; Hurst, R.E.; Davis, C.A.; Simpson, C.; Saban, M.R. Mandatory role of proteinase-activated receptor 1 in experimental bladder inflammation. BMC. Physiol. 2007, 7, 4. [Google Scholar] [CrossRef]
- Monjotin, N.; Gillespie, J.; Farrié, M.; Le Grand, B.; Junquero, D.; Vergnolle, N. F16357, a novel protease-activated receptor 1 antagonist, improves urodynamic parameters in a rat model of interstitial cystitis. Br. J. Pharmacol. 2016, 2224–2236. [Google Scholar] [CrossRef] [PubMed]
- Vera, P.L.; Wolfe, T.E.; Braley, A.E.; Meyer-Siegler, K.L. Thrombin Induces Macrophage Migration Inhibitory Factor Release and Upregulation in Urothelium: A Possible Contribution to Bladder Inflammation. PLoS ONE 2010, 5, e15904. [Google Scholar] [CrossRef]
- Kouzoukas, D.E.; Meyer-Siegler, K.L.; Ma, F.; Westlund, K.N.; Hunt, D.E.; Vera, P.L. Macrophage Migration Inhibitory Factor Mediates PAR-Induced Bladder Pain. PLoS ONE 2015, 10, e0127628. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Kouzoukas, D.E.; Meyer-Siegler, K.L.; Hunt, D.E.; Leng, L.; Bucala, R.; Vera, P.L. MIF mediates bladder pain, not inflammation, in cyclophosphamide cystitis. Cytokine X. 2019, 1, 100003. [Google Scholar] [CrossRef]
- Sims, G.P.; Rowe, D.C.; Rietdijk, S.T.; Herbst, R.; Coyle, A.J. HMGB1 and RAGE in inflammation and cancer. Annu. Rev. Immunol. 2010, 28, 367–388. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, J.; Yamaguchi, K.; Ishikura, H.; Tsubota, M.; Sekiguchi, F.; Seki, Y.; Tsujiuchi, T.; Murai, A.; Umemura, T.; Kawabata, A. Bladder pain relief by HMGB1 neutralization and soluble thrombomodulin in mice with cyclophosphamide-induced cystitis. Neuropharmacology 2014, 79, 112–118. [Google Scholar] [CrossRef]
- Irie, Y.; Tsubota, M.; Maeda, M.; Hiramoto, S.; Sekiguchi, F.; Ishikura, H.; Wake, H.; Nishibori, M.; Kawabata, A. HMGB1 and its membrane receptors as therapeutic targets in an intravesical substance P-induced bladder pain syndrome mouse model. J. Pharmacol. Sci. 2020, 143, 112–116. [Google Scholar] [CrossRef]
- Kouzoukas, D.E.; Ma, F.; Meyer-Siegler, K.L.; Westlund, K.N.; Hunt, D.E.; Vera, P.L. Protease-Activated Receptor 4 Induces Bladder Pain through High Mobility Group Box-1. PLoS ONE 2016, 11, e0152055. [Google Scholar] [CrossRef]
- Hiramoto, S.; Tsubota, M.; Yamaguchi, K.; Okazaki, K.; Sakaegi, A.; Toriyama, Y.; Tanaka, J.; Sekiguchi, F.; Ishikura, H.; Wake, H.; et al. Cystitis-Related Bladder Pain Involves ATP-Dependent HMGB1 Release from Macrophages and Its Downstream H(2)S/Ca(v)3.2 Signaling in Mice. Cells 2020, 9, 1748. [Google Scholar] [CrossRef]
- Martins, J.P.; Silva, R.B.; Coutinho-Silva, R.; Takiya, C.M.; Battastini, A.M.; Morrone, F.B.; Campos, M.M. The role of P2X7 purinergic receptors in inflammatory and nociceptive changes accompanying cyclophosphamide-induced haemorrhagic cystitis in mice. Br. J. Pharmacol. 2012, 165, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Aronsson, P.; Johnsson, M.; Vesela, R.; Winder, M.; Tobin, G. Adenosine receptor antagonism suppresses functional and histological inflammatory changes in the rat urinary bladder. Auton. Neurosci. 2012, 171, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Beckel, J.M.; Daugherty, S.L.; Tyagi, P.; Wolf-Johnston, A.S.; Birder, L.A.; Mitchell, C.H.; de Groat, W.C. Pannexin 1 channels mediate the release of ATP into the lumen of the rat urinary bladder. J. Physiol. 2015, 593, 1857–1871. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, H.; Lu, Q.; Liu, X.; Fan, Y.; Zhu, J.; Sun, B.; Zhao, J.; Dong, X.; Li, L. Suppression of adenosine A(2a) receptors alleviates bladder overactivity and hyperalgesia in cyclophosphamide-induced cystitis by inhibiting TRPV1. Biochem. Pharmacol. 2021, 183, 114340. [Google Scholar] [CrossRef] [PubMed]
- Ko, I.G.; Jin, J.J.; Hwang, L.; Kim, S.H.; Kim, C.J.; Won, K.Y.; Na, Y.G.; Kim, K.H.; Kim, S.J. Adenosine A(2A) Receptor Agonist Polydeoxyribonucleotide Alleviates Interstitial Cystitis-Induced Voiding Dysfunction by Suppressing Inflammation and Apoptosis in Rats. J. Inflamm. Res. 2021, 14, 367–378. [Google Scholar] [CrossRef]
- Kim, S.-E.; Ko, I.-G.; Jin, J.-J.; Hwang, L.; Kim, C.-J.; Kim, S.-H.; Han, J.-H.; Jeon, J.W. Polydeoxyribonucleotide Exerts Therapeutic Effect by Increasing VEGF and Inhibiting Inflammatory Cytokines in Ischemic Colitis Rats. BioMed. Res. Int. 2020, 2020, 2169083. [Google Scholar] [CrossRef]
- Ko, I.-G.; Kim, S.-E.; Jin, J.-J.; Hwang, L.; Ji, E.-S.; Kim, C.-J.; Han, J.-H.; Hong, I.T.; Kwak, M.S.; Yoon, J.Y.; et al. Combination therapy with polydeoxyribonucleotide and proton pump inhibitor enhances therapeutic effectiveness for gastric ulcer in rats. Life Sci. 2018, 203, 12–19. [Google Scholar] [CrossRef]
- Rho, J.H.; Ko, I.G.; Jin, J.J.; Hwang, L.; Kim, S.H.; Chung, J.Y.; Hwang, T.J.; Han, J.H. Polydeoxyribonucleotide Ameliorates Inflammation and Apoptosis in Achilles Tendon-Injury Rats. Int. Neurourol. J. 2020, 24, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Franken, J.; Uvin, P.; De Ridder, D.; Voets, T. TRP channels in lower urinary tract dysfunction. Br. J. Pharmacol. 2014, 171, 2537–2551. [Google Scholar] [CrossRef] [PubMed]
- Deruyver, Y.; Voets, T.; De Ridder, D.; Everaerts, W. Transient receptor potential channel modulators as pharmacological treatments for lower urinary tract symptoms (LUTS): Myth or reality? BJU Int. 2015, 115, 686–697. [Google Scholar] [CrossRef]
- Merrill, L.; Girard, B.M.; May, V.; Vizzard, M.A. Transcriptional and Translational Plasticity in Rodent Urinary Bladder TRP Channels with Urinary Bladder Inflammation, Bladder Dysfunction, or Postnatal Maturation. J. Mol. Neurosci. 2012, 48, 744–756. [Google Scholar] [CrossRef] [PubMed]
- Merrill, L.; Vizzard, M.A. Intravesical TRPV4 blockade reduces repeated variate stress-induced bladder dysfunction by increasing bladder capacity and decreasing voiding frequency in male rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R471–R480. [Google Scholar] [CrossRef]
- Kawasaki, S.; Soga, M.; Sakurai, Y.; Nanchi, I.; Yamamoto, M.; Imai, S.; Takahashi, T.; Tsuno, N.; Asaki, T.; Morioka, Y.; et al. Selective blockade of transient receptor potential vanilloid 4 reduces cyclophosphamide-induced bladder pain in mice. Eur. J. Pharmacol. 2021, 174040. [Google Scholar] [CrossRef] [PubMed]
- DeBerry, J.J.; Saloman, J.L.; Dragoo, B.K.; Albers, K.M.; Davis, B.M. Artemin Immunotherapy Is Effective in Preventing and Reversing Cystitis-Induced Bladder Hyperalgesia via TRPA1 Regulation. J. Pain 2015, 16, 628–636. [Google Scholar] [CrossRef]
- Braicu, C.; Cojocneanu-Petric, R.; Chira, S.; Truta, A.; Floares, A.; Petrut, B.; Achimas-Cadariu, P.; Berindan-Neagoe, I. Clinical and pathological implications of miRNA in bladder cancer. Int. J. Nanomed. 2015, 10, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Gheinani, A.H.; Kiss, B.; Moltzahn, F.; Keller, I.; Bruggmann, R.; Rehrauer, H.; Fournier, C.A.; Burkhard, F.C.; Monastyrskaya, K. Characterization of miRNA-regulated networks, hubs of signaling, and biomarkers in obstruction-induced bladder dysfunction. JCI Insight 2017, 2, e89560. [Google Scholar] [CrossRef][Green Version]
- Fırat, E.; Aybek, Z.; Akgün, Ş.; Küçüker, K.; Akça, H.; Aybek, H. Exploring biomarkers in the overactive bladder: Alterations in miRNA levels of a panel of genes in patients with OAB. Neurourol. Urodyn. 2019, 38, 1571–1578. [Google Scholar] [CrossRef]
- Gheinani, A.H.; Burkhard, F.C.; Monastyrskaya, K. Deciphering microRNA code in pain and inflammation: Lessons from bladder pain syndrome. Cell Mol. Life Sci. 2013, 70, 3773–3789. [Google Scholar] [CrossRef] [PubMed]
- Arai, T.; Fuse, M.; Goto, Y.; Kaga, K.; Kurozumi, A.; Yamada, Y.; Sugawara, S.; Okato, A.; Ichikawa, T.; Yamanishi, T.; et al. Molecular pathogenesis of interstitial cystitis based on microRNA expression signature: miR-320 family-regulated molecular pathways and targets. J. Hum. Genet. 2018, 63, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.J.; Cao, J.Y.; Jin, Z.; Hu, W.G.; Wu, R.H.; Tian, L.H.; Yang, B.; Wang, J.; Xiao, Y.; Huang, C.B. Inhibition of microRNA-132 attenuates inflammatory response and detrusor fibrosis in rats with interstitial cystitis via the JAK-STAT signaling pathway. J. Cell Biochem. 2019, 120, 9147–9158. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Li, H.; Huo, W. MicroRNA-495 alleviates ulcerative interstitial cystitis via inactivating the JAK-STAT signaling pathway by inhibiting JAK3. Int. Urogynecol. J. 2021, 32, 1253–1263. [Google Scholar] [CrossRef]
- Hedlund, P.; Gratzke, C. The endocannabinoid system—A target for the treatment of LUTS? Nat. Rev. Urol. 2016, 13, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, V.; Philips, B.J.; Su, R.; Smaldone, M.C.; Erickson, V.L.; Chancellor, M.B.; Yoshimura, N.; Tyagi, P. Differential expression of functional cannabinoid receptors in human bladder detrusor and urothelium. J. Urol. 2009, 181, 1932–1938. [Google Scholar] [CrossRef]
- Hayn, M.H.; Ballesteros, I.; de Miguel, F.; Coyle, C.H.; Tyagi, S.; Yoshimura, N.; Chancellor, M.B.; Tyagi, P. Functional and immunohistochemical characterization of CB1 and CB2 receptors in rat bladder. Urology 2008, 72, 1174–1178. [Google Scholar] [CrossRef]
- Tambaro, S.; Casu, M.A.; Mastinu, A.; Lazzari, P. Evaluation of selective cannabinoid CB(1) and CB(2) receptor agonists in a mouse model of lipopolysaccharide-induced interstitial cystitis. Eur. J. Pharmacol. 2014, 729, 67–74. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Wang, P.; Bjorling, D.E. Activation of cannabinoid receptor 2 inhibits experimental cystitis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R846–R853. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Wang, P.; Bjorling, D.E. Treatment with a cannabinoid receptor 2 agonist decreases severity of established cystitis. J. Urol. 2014, 191, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wu, Z.; Liu, Y.; Chen, L.; Zhao, H.; Guo, H.; Zhu, K.; Wang, W.; Chen, S.; Zhou, N.; et al. Cannabinoid receptor 2 activation decreases severity of cyclophosphamide-induced cystitis via regulating autophagy. Neurourol. Urodyn. 2020, 39, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.; Arora, N.; Burkovskiy, I.; Xia, Y.F.; Chinnadurai, A.; Westhofen, R.; Hagn, G.; Cox, A.; Kelly, M.; Zhou, J.; et al. Experimental Cannabinoid 2 Receptor Activation by Phyto-Derived and Synthetic Cannabinoid Ligands in LPS-Induced Interstitial Cystitis in Mice. Molecules 2019, 24, 4239. [Google Scholar] [CrossRef]
- Pessina, F.; Capasso, R.; Borrelli, F.; Aveta, T.; Buono, L.; Valacchi, G.; Fiorenzani, P.; Di Marzo, V.; Orlando, P.; Izzo, A.A. Protective effect of palmitoylethanolamide in a rat model of cystitis. J. Urol. 2015, 193, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2018, 11, 487. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuret, T.; Peskar, D.; Erman, A.; Veranič, P. A Systematic Review of Therapeutic Approaches Used in Experimental Models of Interstitial Cystitis/Bladder Pain Syndrome. Biomedicines 2021, 9, 865. https://doi.org/10.3390/biomedicines9080865
Kuret T, Peskar D, Erman A, Veranič P. A Systematic Review of Therapeutic Approaches Used in Experimental Models of Interstitial Cystitis/Bladder Pain Syndrome. Biomedicines. 2021; 9(8):865. https://doi.org/10.3390/biomedicines9080865
Chicago/Turabian StyleKuret, Tadeja, Dominika Peskar, Andreja Erman, and Peter Veranič. 2021. "A Systematic Review of Therapeutic Approaches Used in Experimental Models of Interstitial Cystitis/Bladder Pain Syndrome" Biomedicines 9, no. 8: 865. https://doi.org/10.3390/biomedicines9080865
APA StyleKuret, T., Peskar, D., Erman, A., & Veranič, P. (2021). A Systematic Review of Therapeutic Approaches Used in Experimental Models of Interstitial Cystitis/Bladder Pain Syndrome. Biomedicines, 9(8), 865. https://doi.org/10.3390/biomedicines9080865





