Pre-Clinical Insights into the Iron and Breast Cancer Hypothesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Experiments
2.2. Assays and Chemical Analysis
2.3. Statistical Analysis
3. Results and Discussion
3.1. Effect of Iron Status on the Initiation Stage of Mammary Carcinogenesis
3.2. Effect of Iron Status during the Promotion Phase of Mammary Carcinogenesis
3.3. Effect of Deficient Iron Status on the Promotion Phase of Mammary Carcinogenesis
3.4. Examining Other Key Tenants of the Iron Stores Cancer Hypothesis
3.5. Effect of Dietary Iron on Oxidative Indices: Diets Fed from 21 to 90 Days of Age
3.6. Summary and Implications
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sheftel, A.D.; Mason, A.B.; Ponka, P. The long history of iron in the Universe and in health and disease. Biochim. Biophys. Acta Gen. Subj. 2012, 1820, 161–187. [Google Scholar] [CrossRef] [PubMed]
- Clara, C.; Antonella, N.; Laura, S. Iron metabolism and iron disorders revisited in the hepcidin era. Haematologica 2020, 105, 260–272. [Google Scholar] [CrossRef]
- Fung, E.B.; Harmatz, P.; Milet, M.; Ballas, S.K.; De Castro, L.; Hagar, W.; Owen, W.; Olivieri, N.; Smith-Whitley, K.; Darbari, D.; et al. Morbidity and mortality in chronically transfused subjects with thalassemia and sickle cell disease: A report from the multi-center study of iron overload. Am. J. Hematol. 2007, 82, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Kadoglou, N.P.E.; Biddulph, J.P.; Rafnsson, S.B.; Trivella, M.; Nihoyannopoulos, P.; Demakakos, P. The association of ferritin with cardiovascular and all-cause mortality in community-dwellers: The English longitudinal study of ageing. PLoS ONE 2017, 12, e0178994. [Google Scholar] [CrossRef] [PubMed]
- Yiannikourides, A.; Latunde-Dada, G.O. A Short Review of Iron Metabolism and Pathophysiology of Iron Disorders. Medicines 2019, 6, 85. [Google Scholar] [CrossRef] [PubMed]
- Torti, S.V.; Manz, D.H.; Paul, B.T.; Blanchette-Farra, N.; Torti, F.M. Iron and Cancer. Annu. Rev. Nutr. 2018, 38, 97–125. [Google Scholar] [CrossRef]
- Gaur, A.; Collins, H.; Wulaningsih, W.; Holmberg, L.; Garmo, H.; Hammar, N.; Walldius, G.; Jungner, I.; Van Hemelrijck, M. Iron metabolism and risk of cancer in the Swedish AMORIS study. Cancer Causes Control. 2013, 24, 1393–1402. [Google Scholar] [CrossRef]
- Durigova, A.; Lamy, P.J.; Thezenas, S.; Pouderoux, S.; Montels, F.; Romieu, G.; Gutowski, M.; Jacot, W. Anemia and iron biomarkers in patients with early breast cancer. Diagnostic value of hepcidin and soluble transferrin receptor quantification. Clin. Chem. Lab. Med. 2013, 51, 1833–1841. [Google Scholar] [CrossRef] [PubMed]
- Alkhateeb, A.A.; Han, B.; Connor, J.R. Ferritin stimulates breast cancer cells through an iron-independent mechanism and is localized within tumor-associated macrophages. Breast Cancer Res. Treat. 2013, 137, 733–744. [Google Scholar] [CrossRef]
- Nicolini, A.; Ferrari, P.; Fallahi, P.; Antonelli, A. An iron regulatory gene signature in breast cancer: More than a prognostic genetic profile? Future Oncol. 2012, 8, 131–134. [Google Scholar] [CrossRef]
- Durigova, A.; Jacot, W.; Pouderoux, S.; Roques, S.; Montels, F.; Lamy, P.J. Iron metabolism in breast cancer: Knowledge and future. Ann. Biol. Clin. 2012, 70, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Katz, S. Intakes of dietary iron and heme-iron and risk of postmenopausal breast cancer in the National Institutes of Health-AARP Diet and Health Study. Am. J. Clin. Nutr. 2011, 94, 613–614. [Google Scholar] [CrossRef]
- Kabat, G.C.; Cross, A.J.; Park, Y.; Schatzkin, A.; Hollenbeck, A.R.; Rohan, T.E.; Sinha, R. Intakes of dietary iron and heme-iron and risk of postmenopausal breast cancer in the National Institutes of Health-AARP Diet and Health Study. Am. J. Clin. Nutr. 2010, 92, 1478–1483. [Google Scholar] [CrossRef]
- Eckard, J.; Dai, J.; Wu, J.; Jian, J.; Yang, Q.; Chen, H.; Costa, M.; Frenkel, K.; Huang, X. Effects of cellular iron deficiency on the formation of vascular endothelial growth factor and angiogenesis. Iron deficiency and angiogenesis. Cancer Cell Int. 2010, 10, 28. [Google Scholar] [CrossRef]
- Moore, A.B.; Shannon, J.; Chen, C.; Lampe, J.W.; Ray, R.M.; Lewis, S.K.; Lin, M.; Stalsberg, H.; Thomas, D.B. Dietary and stored iron as predictors of breast cancer risk: A nested case-control study in Shanghai. Int. J. Cancer 2009, 125, 1110–1117. [Google Scholar] [CrossRef]
- Ferrucci, L.M.; Cross, A.J.; Graubard, B.I.; Brinton, L.A.; McCarty, C.A.; Ziegler, R.G.; Ma, X.; Mayne, S.T.; Sinha, R. Intake of meat, meat mutagens, and iron and the risk of breast cancer in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Br. J. Cancer 2009, 101, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.J.; Yeon, J.Y.; Sung, C.J.; Kim, H.S.; Sung, M.K. Dietary intake and serum levels of iron in relation to oxidative stress in breast cancer patients. J. Clin. Biochem. Nutr. 2009, 45, 355–360. [Google Scholar] [CrossRef][Green Version]
- Huang, X. Does iron have a role in breast cancer? Lancet Oncol. 2008, 9, 803–807. [Google Scholar] [CrossRef]
- Grant, W.B. An ecological study of cancer mortality rates including indices for dietary iron and zinc. Anticancer Res. 2008, 28, 1955–1963. [Google Scholar]
- Zhang, K.; Ping, L.; Du, T.; Liang, G.; Huang, Y.; Li, Z.; Deng, R.; Tang, J. A Ferroptosis-Related lncRNAs Signature Predicts Prognosis and Immune Microenvironment for Breast Cancer. Front. Mol. Biosci. 2021, 8, 678877. [Google Scholar] [CrossRef]
- Youn, J.; Park, S.; Song, S.; Moon, H.G.; Noh, D.Y.; Jung, S.Y.; Lee, E.; Kim, Z.; Youn, H.J.; Cho, J.; et al. Nutrient intakes from supplement and factors associated with supplement use among breast cancer survivors: A cross-sectional study. Eur. J. Cancer Care 2021, e13447. [Google Scholar] [CrossRef]
- Wu, Z.H.; Tang, Y.; Yu, H.; Li, H.D. The role of ferroptosis in breast cancer patients: A comprehensive analysis. Cell Death Discov. 2021, 7, 93. [Google Scholar] [CrossRef]
- Von Holle, A.; O’Brien, K.M.; Sandler, D.P.; Janicek, R.; Weinberg, C.R. Association Between Serum Iron Biomarkers and Breast Cancer. Cancer Epidemiol. Biomark. Prev. 2021, 30, 422–425. [Google Scholar] [CrossRef]
- Tian, Q.; Zhou, Y.; Zhu, L.; Gao, H.; Yang, J. Development and Validation of a Ferroptosis-Related Gene Signature for Overall Survival Prediction in Lung Adenocarcinoma. Front. Cell Dev. Biol. 2021, 9, 684259. [Google Scholar] [CrossRef]
- Shen, L.; Zhou, Y.; He, H.; Chen, W.; Lenahan, C.; Li, X.; Deng, Y.; Shao, A.; Huang, J. Crosstalk between Macrophages, T Cells, and Iron Metabolism in Tumor Microenvironment. Oxidative Med. Cell Longev. 2021, 2021, 8865791. [Google Scholar] [CrossRef]
- Papadimitriou, N.; Dimou, N.; Gill, D.; Tzoulaki, I.; Murphy, N.; Riboli, E.; Lewis, S.J.; Martin, R.M.; Gunter, M.J.; Tsilidis, K.K. Genetically predicted circulating concentrations of micronutrients and risk of breast cancer: A Mendelian randomization study. Int. J. Cancer 2021, 148, 646–653. [Google Scholar] [CrossRef]
- Mertens, C.; Schnetz, M.; Rehwald, C.; Grein, S.; Elwakeel, E.; Weigert, A.; Brune, B.; Jung, M. Iron-Bound Lipocalin-2 from Tumor-Associated Macrophages Drives Breast Cancer Progression Independent of Ferroportin. Metabolites 2021, 11, 180. [Google Scholar] [CrossRef]
- Liu, K.Y.; Feng, X.L.; Mo, X.F.; Lin, F.Y.; Zhang, X.; Huang, C.Y.; Abulimiti, A.; Li, L.; Zhang, C.X. Iron intake with the risk of breast cancer among Chinese women: A case-control study. Public Health Nutr. 2021, 1–13. [Google Scholar] [CrossRef]
- Li, H.; Li, L.; Xue, C.; Huang, R.; Hu, A.; An, X.; Shi, Y. A Novel Ferroptosis-Related Gene Signature Predicts Overall Survival of Breast Cancer Patients. Biology 2021, 10, 151. [Google Scholar] [CrossRef]
- Huang, Y.; Cao, D.; Chen, Z.; Chen, B.; Li, J.; Guo, J.; Dong, Q.; Liu, L.; Wei, Q. Red and processed meat consumption and cancer outcomes: Umbrella review. Food Chem. 2021, 356, 129697. [Google Scholar] [CrossRef]
- Hou, C.; Hou, Q.; Xie, X.; Wang, H.; Chen, Y.; Lu, T.; Wu, Q.; Liang, Y.; Hu, Y.; Mao, Y. Serum iron status and the risk of breast cancer in the European population: A two-sample Mendelian randomisation study. Genes Nutr. 2021, 16, 9. [Google Scholar] [CrossRef]
- Yuan, S.; Carter, P.; Vithayathil, M.; Kar, S.; Giovannucci, E.; Mason, A.M.; Burgess, S.; Larsson, S.C. Iron Status and Cancer Risk in UK Biobank: A Two-Sample Mendelian Randomization Study. Nutrients 2020, 12, 526. [Google Scholar] [CrossRef]
- Yi, J.; Zhu, J.; Wu, J.; Thompson, C.B.; Jiang, X. Oncogenic activation of PI3K-AKT-mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 31189–31197. [Google Scholar] [CrossRef]
- Sanagoo, A.; Kiani, F.; Saei Gharenaz, M.; Sayehmiri, F.; Koohi, F.; Jouybari, L.; Dousti, M. A systematic review and meta-analysis on the association of serum and tumor tissue iron and risk of breast cancer. Caspian J. Intern. Med. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Lo, J.J.; Park, Y.M.; Sinha, R.; Sandler, D.P. Association between meat consumption and risk of breast cancer: Findings from the Sister Study. Int. J. Cancer 2020, 146, 2156–2165. [Google Scholar] [CrossRef]
- Khan, A.; Singh, P.; Srivastava, A. Iron: Key player in cancer and cell cycle? J. Trace Elem. Med. Biol. 2020, 62, 126582. [Google Scholar] [CrossRef]
- Jerzak, K.J.; Lohmann, A.E.; Ennis, M.; Nemeth, E.; Ganz, T.; Goodwin, P.J. Prognostic associations of plasma hepcidin in women with early breast cancer. Breast Cancer Res. Treat. 2020, 184, 927–935. [Google Scholar] [CrossRef]
- Cheng, M.; Liu, P.; Xu, L.X. Iron promotes breast cancer cell migration via IL-6/JAK2/STAT3 signaling pathways in a paracrine or autocrine IL-6-rich inflammatory environment. J. Inorg. Biochem. 2020, 210, 111159. [Google Scholar] [CrossRef]
- Chang, V.C.; Cotterchio, M.; Bondy, S.J.; Kotsopoulos, J. Iron intake, oxidative stress-related genes and breast cancer risk. Int. J. Cancer 2020, 147, 1354–1373. [Google Scholar] [CrossRef]
- Chang, V.C.; Cotterchio, M.; Khoo, E. Iron intake, body iron status, and risk of breast cancer: A systematic review and meta-analysis. BMC Cancer 2019, 19, 543. [Google Scholar] [CrossRef]
- Scimeca, M.; Bonanno, E. New highlight in breast cancer development: The key role of hepcidin and iron metabolism. Ann. Transl. Med. 2018, 6, S56. [Google Scholar] [CrossRef] [PubMed]
- Quintana Pacheco, D.A.; Sookthai, D.; Graf, M.E.; Schubel, R.; Johnson, T.; Katzke, V.A.; Kaaks, R.; Kuhn, T. Iron status in relation to cancer risk and mortality: Findings from a population-based prospective study. Int. J. Cancer 2018, 143, 561–569. [Google Scholar] [CrossRef]
- Bajbouj, K.; Shafarin, J.; Hamad, M. High-Dose Deferoxamine Treatment Disrupts Intracellular Iron Homeostasis, Reduces Growth, and Induces Apoptosis in Metastatic and Nonmetastatic Breast Cancer Cell Lines. Technol. Cancer Res. Treat. 2018, 17, 1533033818764470. [Google Scholar] [CrossRef]
- Anderson, J.J.; Darwis, N.D.M.; Mackay, D.F.; Celis-Morales, C.A.; Lyall, D.M.; Sattar, N.; Gill, J.M.R.; Pell, J.P. Red and processed meat consumption and breast cancer: UK Biobank cohort study and meta-analysis. Eur. J. Cancer 2018, 90, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, N.; Mahjoub, S.; Haji Hosseini, R.; TaherKhani, M.; Moslemi, D. Alterations in serum levels of trace element in patients with breast cancer before and after chemotherapy. Caspian J. Intern. Med. 2018, 9, 134–139. [Google Scholar] [CrossRef]
- Hooda, N.; Gupta, R.; Gupta, N.R. Prediction of Malignant Breast Cancer Cases using Ensemble Machine Learning: A Case Study of Pesticides Prone Area. IEEE/ACM Trans. Comput. Biol. Bioinform. 2020, 1. [Google Scholar] [CrossRef]
- Singh, M.; Lu, J.; Briggs, S.P.; McGinley, J.N.; Haegele, A.D.; Thompson, H.J. Effect of excess dietary iron on the promotion stage of 1-methyl-1-nitrosourea-induced mammary carcinogenesis: Pathogenetic characteristics and distribution of iron. Carcinogenesis 1994, 15, 1567–1570. [Google Scholar] [CrossRef]
- Young, S.; Hallowes, R.C. Tumours of the mammary gland. IARC Sci. Publ. 1973, 5, 31–73. [Google Scholar]
- Thompson, H.J.; Singh, M.; McGinley, J. Classification of premalignant and malignant lesions developing in the rat mammary gland after injection of sexually immature rats with 1-methyl-1-nitrosourea. J. Mammary Gland Biol. Neoplasia 2000, 5, 201–210. [Google Scholar] [CrossRef]
- Clegg, M.S.; Keen, C.L.; Lonnerdal, B.; Hurley, L.S. Influence of ashing techniques on the analysis of trace elements in animal tissue: I. Wet ashing. Biol. Trace Elem. Res. 1981, 3, 107–115. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Floyd, R.A.; West, M.S.; Eneff, K.L.; Hogsett, W.E.; Tingey, D.T. Hydroxyl free radical mediated formation of 8-hydroxyguanine in isolated DNA. Arch. Biochem. Biophys. 1988, 262, 266–272. [Google Scholar] [CrossRef]
- Haegele, A.D.; Wolfe, P.; Thompson, H.J. X-radiation induces 8-hydroxy-2’-deoxyguanosine formation in vivo in rat mammary gland DNA. Carcinogenesis 1998, 19, 1319–1321. [Google Scholar] [CrossRef][Green Version]
- Snedecor, G.W.; Cochran, W. Statistical Methods, 6th ed.; The Iowa State University Press: Ames, IA, USA, 1967. [Google Scholar]
- Peto, R. Editorial: Guidelines on the analysis of tumour rates and death rates in experimental animals. Br. J. Cancer 1974, 29, 101–105. [Google Scholar] [CrossRef][Green Version]
- García, Y.; Díaz-Castro, J. Advantages and disadvantages of the animal models v. in vitro studies in iron metabolism: A review. Animal 2013, 7, 1651–1658. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; McGinley, J.N.; Thompson, H.J. A comparison of the histopathology of premalignant and malignant mammary gland lesions induced in sexually immature rats with those occurring in the human. Lab. Investig. 2000, 80, 221–231. [Google Scholar] [CrossRef]
- Thompson, H.J.; Singh, M. Rat models of premalignant breast disease. J. Mammary Gland Biol. Neoplasia 2000, 5, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; Kennedy, K.; Witt, M.; Juzefyk, J. Effect of dietary iron deficiency or excess on the induction of mammary carcinogenesis by 1-methyl-1-nitrosourea. Carcinogenesis 1991, 12, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; Zhu, Z.; Jiang, W. Weight control and breast cancer prevention: Are the effects of reduced energy intake equivalent to those of increased energy expenditure? J. Nutr. 2004, 134, 3407S–3411S. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; McGinley, J.N.; Rothhammer, K.; Singh, M. Rapid induction of mammary intraductal proliferations, ductal carcinoma in situ and carcinomas by the injection of sexually immature female rats with 1-methyl-1-nitrosourea. Carcinogenesis 1995, 16, 2407–2411. [Google Scholar] [CrossRef]
- Aksan, A.; Farrag, K.; Aksan, S.; Schroeder, O.; Stein, J. Flipside of the Coin: Iron Deficiency and Colorectal Cancer. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Rusu, I.G.; Suharoschi, R.; Vodnar, D.C.; Pop, C.R.; Socaci, S.A.; Vulturar, R.; Istrati, M.; Morosan, I.; Farcas, A.C.; Kerezsi, A.D.; et al. Iron Supplementation Influence on the Gut Microbiota and Probiotic Intake Effect in Iron Deficiency-A Literature-Based Review. Nutrients 2020, 12, 1993. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, B.; Li, H. Gut Microbiota and Iron: The Crucial Actors in Health and Disease. Pharmaceuticals 2018, 11, 98. [Google Scholar] [CrossRef]
- Kew, M.C. Hepatic iron overload and hepatocellular carcinoma. Liver Cancer 2014, 3, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Jayachandran, A.; Shrestha, R.; Bridle, K.R.; Crawford, D.H.G. Association between hereditary hemochromatosis and hepatocellular carcinoma: A comprehensive review. Hepatoma Res. 2020, 6, 8. [Google Scholar] [CrossRef]
| Dietary Iron (ppm) | Hemoglobin (g/dL) | Hematocrit (%) | Plasma Ferritin (ng/mL) | Liver Iron (µg/g) | Mammary Gland Iron Fat Free (µg/g) |
|---|---|---|---|---|---|
| 6 | 13.1 ± 0.5 | 40.4 ± 0.8 | 25.7 ± 10.6 a | 143 ± 25 a | 96 ± 12 a |
| 35 | 14.6 ± 0.1 | 43.9 ± 1.2 | 89.6 ± 18.2 b | 591 ± 51 b | 131 ± 25 b |
| 350 | 14.6 ± 0.1 | 41.5 ± 2.5 | 204.5 ± 42.4 c | 1006 ± 89 c | 148 ± 3 b |
| Dietary Iron (ppm) | Body Weight at Time of DMBA Administration (g) | Body Weight at End of Study (g) | Final Incidence of Mammary Carcinoma (%) | Final Average Number of Carcinomas per Rat |
|---|---|---|---|---|
| 6 | 171 ± 2 | 282 ± 4 | 86.7 | 4.1 |
| 35 | 173 ± 4 | 288 ± 10 | 89.7 | 3.6 |
| 350 | 172 ± 2 | 286 ± 6 | 90.0 | 4.0 |
| Dietary Iron at Time of Carcinogen Treatment | ||||
|---|---|---|---|---|
| Diagnosis | Comments | Low Fe 6 ppm | Adequate Fe 35 ppm | High Fe 350 ppm |
| Carcinoma | Comedo | 3 | 3 | 2 |
| Cribriform | 3 | 7 | 1 | |
| Highly vascular | 3 | 4 | 7 | |
| Invading muscle | 1 | 6 | 7 | |
| Mucinous | 1 | |||
| Papillary | 8 | 2 | 11 | |
| PDCISC | 8 | 4 | 4 | |
| with FA | 7 | 11 | 11 | |
| Mixed | 91 | 63 | 70 | |
| Total | 124 | 100 | 121 | |
| Ductal carcinoma in situ | Comedo | 1 | ||
| Adenoma | 1 | 2 | 2 | |
| Fibroadenoma | 4 | 7 | 2 | |
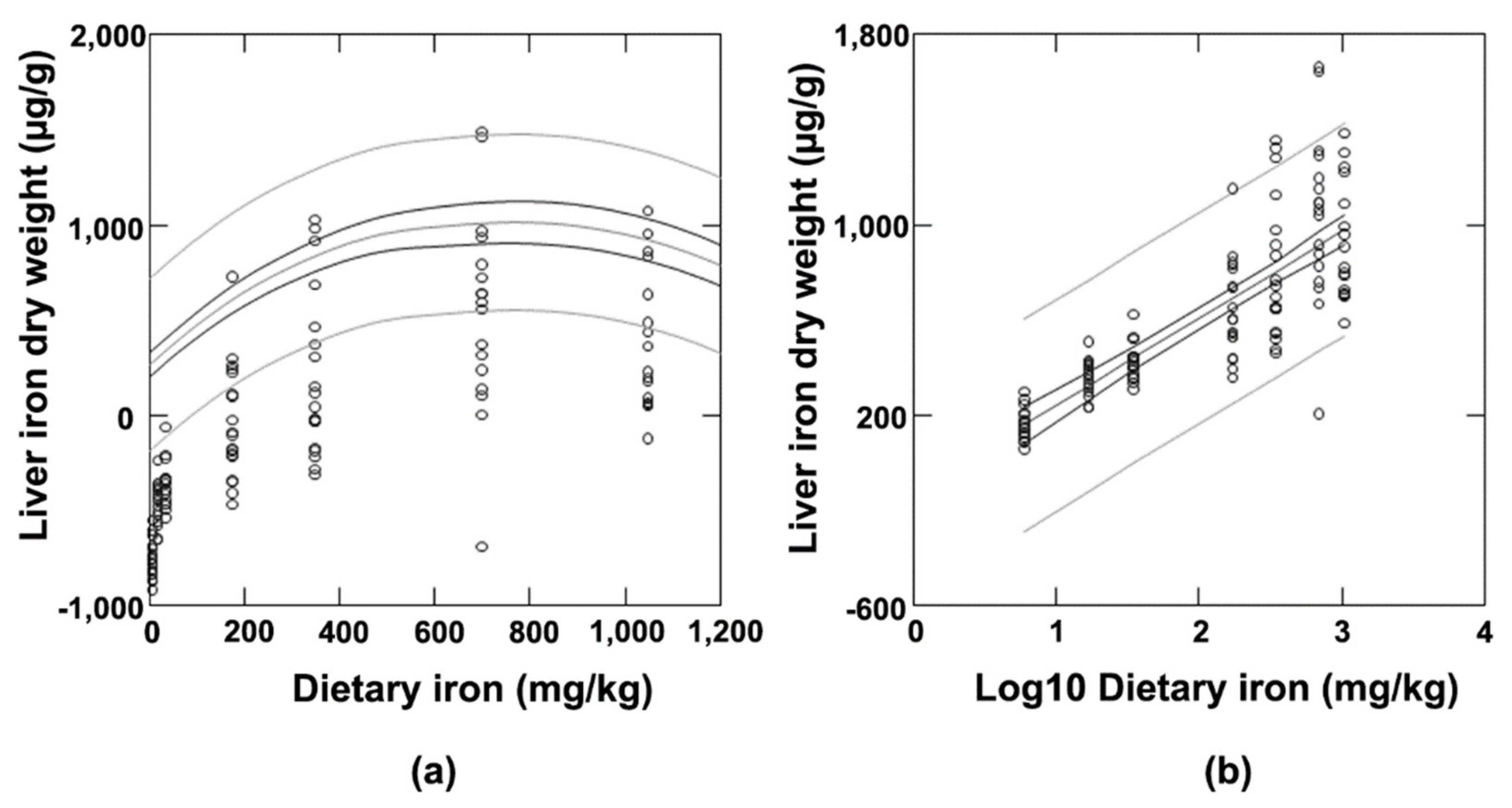
| Dietary Iron (mg/kg) | Liver Iron a Dry wt (µg/g) |
|---|---|
| 6 | 159 ± 68 |
| 17.5 | 356 ± 76 |
| 35 | 421 ± 80 |
| 175 | 641 ± 192 |
| 350 | 796 ± 264 |
| 700 | 1028 ± 344 |
| 1050 | 916 ± 240 |
| Group | Dietary Iron (mg/kg) | Cancer Incidence a (%) | Cancer Multiplicity (Number/Rat) c | Body Weights b (g) |
|---|---|---|---|---|
| 1 | 6 | 58.0 (30) | 0.86 | 339 ± 5 |
| 2 | 17.5 | 70.0 (35) | 1.58 | 342 ± 5 |
| 3 | 35 | 58.0 (30) | 1.04 | 353 ± 6 |
| 4 | 175 | 66.0 (33) | 1.44 | 348 ± 5 |
| 5 | 350 | 80 (40) | 1.49 | 338 ± 6 |
| 6 | 700 | 62.0 (31) | 1.40 | 332 ± 7 |
| 7 | 1050 | 72.0 (36) | 1.44 | 331 ± 5 |
| Group | Final AC Incidence (%) | Final AC Multiplicity (Number/Rat) c | Final Body Weights (g) | Body Length (cm) |
|---|---|---|---|---|
| 35 ppm of Fe Ad Lib (N = 24) | 87.5 a | 3.04 a | 203 ± 3 a | 37.9 ± 0.2 a |
| 35 ppm of Fe Pair-Fed 3 ppm of Fe (N = 24) | 95.8 a | 3.13 a | 189 ± 3 b | 36.2 ± 0.22 b |
| 3 ppm of Fe Ad Lib (N = 24) | 54.2 b | 1.13 b | 177 ± 3 b | 36.0 ± 0.2 b |
| Dietary Iron (ppm) | Final Body Weight (g) | Plasma Ferritin (ng/mL) | Liver MDA (pmol/mg) Protein a | Liver 8-OHdG (Residue/106 dG) a | Urinary Isoprostane F-2 Alpha (ng/mg) Creatinine |
|---|---|---|---|---|---|
| 6 | 242 ± 7 | 25.7 ± 10.6 | 426 ± 29 | 6.6 ± 0.7 | 7.2 ± 0.6 |
| 35 | 248 ± 6 | 89.6 ± 18.2 | 529 ± 23 | 7.8 ± 1.3 | 7.7 ± 0.5 |
| 350 | 248 ± 7 | 204.5 ± 42.4 | 665 ± 46 | 11.3 ± 2.0 | 8.1 ± 0.5 |
| Dietary Iron (ppm) | Mammary Gland Iron (ppm) | Mammary Gland 8-OHdG (Residues/106 dG) | Mammary Tumor Iron (ppm) | Mammary Tumor 8-OHdG (Residues/106 dG) |
|---|---|---|---|---|
| 6 | 48 ± 3 | 6.3 ± 0.4 | 63 ± 22 | 5.5 ± 0.4 |
| 35 | 71 ± 8 | 6.2 ± 0.3 | 75 ± 26 | 6.6 ± 0.6 |
| 350 | 83 ± 7 | 7.0 ± 0.5 | 113 ± 21 | 6.0 ± 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thompson, H.J.; Neil, E.S.; McGinley, J.N. Pre-Clinical Insights into the Iron and Breast Cancer Hypothesis. Biomedicines 2021, 9, 1652. https://doi.org/10.3390/biomedicines9111652
Thompson HJ, Neil ES, McGinley JN. Pre-Clinical Insights into the Iron and Breast Cancer Hypothesis. Biomedicines. 2021; 9(11):1652. https://doi.org/10.3390/biomedicines9111652
Chicago/Turabian StyleThompson, Henry J., Elizabeth S. Neil, and John N. McGinley. 2021. "Pre-Clinical Insights into the Iron and Breast Cancer Hypothesis" Biomedicines 9, no. 11: 1652. https://doi.org/10.3390/biomedicines9111652
APA StyleThompson, H. J., Neil, E. S., & McGinley, J. N. (2021). Pre-Clinical Insights into the Iron and Breast Cancer Hypothesis. Biomedicines, 9(11), 1652. https://doi.org/10.3390/biomedicines9111652







