Arginine and Endothelial Function
Abstract
1. Pleiotropic Effects of Arginine
2. Arginine and NO Production in Physiological Conditions: The Arginine Paradox
3. Impaired NO Production as a Mechanism of Endothelial Dysfunction and Arginine Intervention
4. Arginine Supplementation in Hypertension
5. Arginine Supplementation in Ischemic Heart Disease and Peripheral Artery Disease
6. Arginine Supplementation in Diabetes Mellitus
7. Conclusions and Perspective: Arginine as a Therapeutic Tool
Author Contributions
Funding
Conflicts of Interest
References
- Lopez, M.J.; Mohiuddin, S.S. Biochemistry, Essential Amino Acids. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Luiking, Y.C.; Ten Have, G.A.; Wolfe, R.R.; Deutz, N.E. Arginine de novo and nitric oxide production in disease states. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1177–E1189. [Google Scholar] [CrossRef] [PubMed]
- Agapie, T.; Suseno, S.; Woodward, J.J.; Stoll, S.; Britt, R.D.; Marletta, M.A. NO formation by a catalytically self-sufficient bacterial nitric oxide synthase from Sorangium cellulosum. Proc. Natl. Acad. Sci. USA 2009, 106, 16221–16226. [Google Scholar] [CrossRef] [PubMed]
- Jachymova, M.; Martasek, P.; Panda, S.; Roman, L.J.; Panda, M.; Shea, T.M.; Ishimura, Y.; Kim, J.J. Masters, B.S. Recruitment of governing elements for electron transfer in the nitric oxide synthase family. Proc. Natl. Acad. Sci. USA 2005, 102, 15833–15838. [Google Scholar] [CrossRef] [PubMed]
- Serpe, M.J.; Zhang, X. The Principles, Development and Application of Microelectrodes for the In Vivo Determination of Nitric Oxide. In Electrochemical Methods for Neuroscience; Michael, A.C., Borland, L.M., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2007. [Google Scholar]
- Andrew, P.J.; Mayer, B. Enzymatic function of nitric oxide synthases. Cardiovasc. Res. 1999, 43, 521–531. [Google Scholar] [CrossRef]
- Sax, H.C. Arginine stimulates wound healing and immune function in elderly human beings. JPEN J. Parenter. Enteral. Nutr. 1994, 18, 559–560. [Google Scholar] [CrossRef]
- Barbul, A.; Lazarou, S.A.; Efron, D.T.; Wasserkrug, H.L.; Efron, G. Arginine enhances wound healing and lymphocyte immune responses in humans. Surgery 1990, 108, 331–336. [Google Scholar]
- Durante, W.; Liao, L.; Reyna, S.V.; Peyton, K.J.; Schafer, A.I. Physiological cyclic stretch directs L-arginine transport and metabolism to collagen synthesis in vascular smooth muscle. FASEB J. 2000, 14, 1775–1783. [Google Scholar] [CrossRef]
- Da Silva, R.P.; Nissim, I.; Brosnan, M.E.; Brosnan, J.T. Creatine synthesis: Hepatic metabolism of guanidinoacetate and creatine in the rat in vitro and in vivo. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E256–E261. [Google Scholar] [CrossRef]
- Bai, Y.; Sun, L.; Yang, T.; Sun, K.; Chen, J.; Hui, R. Increase in fasting vascular endothelial function after short-term oral L-arginine is effective when baseline flow-mediated dilation is low: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2009, 89, 77–84. [Google Scholar] [CrossRef]
- Raman, C.S.; Li, H.; Martasek, P.; Kral, V.; Masters, B.S.; Poulos, T.L. Crystal structure of constitutive endothelial nitric oxide synthase: A paradigm for pterin function involving a novel metal center. Cell 1998, 95, 939–950. [Google Scholar] [CrossRef][Green Version]
- Garthwaite, J.; Charles, S.L.; Chess-Williams, R. Endothelium-derived relaxing factor release on activation of NMDA receptors suggests role as intercellular messenger in the brain. Nature 1988, 336, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Bec, N.; Gorren, A.C.; Voelker, C.; Mayer, B.; Lange, R. Reaction of neuronal nitric-oxide synthase with oxygen at low temperature. Evidence for reductive activation of the oxy-ferrous complex by tetrahydrobiopterin. J. Biol. Chem. 1998, 273, 13502–13508. [Google Scholar] [CrossRef]
- Iwai, N.; Hanai, K.; Tooyama, I.; Kitamura, Y.; Kinoshita, M. Regulation of neuronal nitric oxide synthase in rat adrenal medulla. Hypertension 1995, 25, 431–436. [Google Scholar] [CrossRef] [PubMed]
- O’Dell, T.J.; Huang, P.L.; Dawson, T.M.; Dinerman, J.L.; Snyder, S.H.; Kandel, E.R.; Fishman, M.C. Endothelial NOS and the blockade of LTP by NOS inhibitors in mice lacking neuronal NOS. Science 1994, 265, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Geller, D.A.; Lowenstein, C.J.; Shapiro, R.A.; Nussler, A.K.; Di Silvio, M.; Wang, S.C.; Nakayama, D.K.; Simmons, R.L.; Snyder, S.H.; Billiar, T.R. Molecular cloning and expression of inducible nitric oxide synthase from human hepatocytes. Proc. Natl. Acad. Sci. USA 1993, 90, 3491–3495. [Google Scholar] [CrossRef]
- Geller, D.A.; Nussler, A.K.; Di Silvio, M.; Lowenstein, C.J.; Shapiro, R.A.; Wang, S.C.; Simmons, R.L.; Billiar, T.R. Cytokines, endotoxin, and glucocorticoids regulate the expression of inducible nitric oxide synthase in hepatocytes. Proc. Natl. Acad. Sci. USA 1993, 90, 522–526. [Google Scholar] [CrossRef]
- Hokari, A.; Zeniya, M.; Esumi, H. Cloning and functional expression of human inducible nitric oxide synthase (NOS) cDNA from a glioblastoma cell line A-172. J. Biochem. 1994, 116, 575–581. [Google Scholar] [CrossRef]
- Forstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837, 837a–837d. [Google Scholar] [CrossRef]
- Rafikov, R.; Fonseca, F.V.; Kumar, S.; Pardo, D.; Darragh, C.; Elms, S.; Fulton, D.; Black, S.M. eNOS activation and NO function: Structural motifs responsible for the posttranslational control of endothelial nitric oxide synthase activity. J. Endocrinol. 2011, 210, 271–284. [Google Scholar] [CrossRef]
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric oxide synthases: Structure, function and inhibition. Biochem. J. 2001, 357 Pt 3, 593–615. [Google Scholar] [CrossRef]
- Stuehr, D.J. Enzymes of the L-arginine to nitric oxide pathway. J. Nutr. 2004, 134 (Suppl. 10), 2748S–2751S. [Google Scholar] [CrossRef] [PubMed]
- Meulemans, A. Electrochemical detection of nitroso-arginine as an intermediate between N-hydroxy-arginine and citrulline. An in vitro versus in vivo study using microcarbon electrodes in neuronal nitric oxide synthase and mice brain. Neurosci. Lett. 2000, 294, 125–129. [Google Scholar] [CrossRef]
- Tsuboi, T.; Maeda, M.; Hayashi, T. Administration of L-arginine plus L-citrulline or L-citrulline alone successfully retarded endothelial senescence. PLoS ONE 2018, 13, e0192252. [Google Scholar] [CrossRef] [PubMed]
- De Betue, C.T.I.; Garcia Casal, X.C.; van Waardenburg, D.A.; Schexnayder, S.M.; Joosten, K.F.M.; Deutz, N.E.P.; Engelen, M. 24-Hour protein, arginine and citrulline metabolism in fed critically ill children—A stable isotope tracer study. Clin. Nutr. 2017, 36, 876–887. [Google Scholar] [CrossRef]
- Arnett, D.C.; Persechini, A.; Tran, Q.K.; Black, D.J.; Johnson, C.K. Fluorescence quenching studies of structure and dynamics in calmodulin-eNOS complexes. FEBS Lett. 2015, 589, 1173–1178. [Google Scholar] [CrossRef]
- Li, H.; Raman, C.S.; Glaser, C.B.; Blasko, E.; Young, T.A.; Parkinson, J.F.; Whitlow, M.; Poulos, T.L. Crystal structures of zinc-free and -bound heme domain of human inducible nitric-oxide synthase. Implications for dimer stability and comparison with endothelial nitric-oxide synthase. J. Biol. Chem. 1999, 274, 21276–21284. [Google Scholar] [CrossRef]
- Crane, B.R.; Rosenfeld, R.J.; Arvai, A.S.; Ghosh, D.K.; Ghosh, S.; Tainer, J.A.; Stuehr, D.J.; Getzoff, E.D. N-terminal domain swapping and metal ion binding in nitric oxide synthase dimerization. EMBO J. 1999, 18, 6271–6281. [Google Scholar] [CrossRef]
- Daiber, A.; Kroller-Schon, S.; Oelze, M.; Hahad, O.; Li, H.; Schulz, R.; Steven, S.; Munzel, T. Oxidative stress and inflammation contribute to traffic noise-induced vascular and cerebral dysfunction via uncoupling of nitric oxide synthases. Redox Biol. 2020, 34, 101506. [Google Scholar] [CrossRef]
- Jayaram, R.; Goodfellow, N.; Zhang, M.H.; Reilly, S.; Crabtree, M.; De Silva, R.; Sayeed, R.; Casadei, B. Molecular mechanisms of myocardial nitroso-redox imbalance during on-pump cardiac surgery. Lancet 2015, 385 (Suppl. 1), S49. [Google Scholar] [CrossRef]
- Siu, K.L.; Lotz, C.; Ping, P.; Cai, H. Netrin-1 abrogates ischemia/reperfusion-induced cardiac mitochondrial dysfunction via nitric oxide-dependent attenuation of NOX4 activation and recoupling of NOS. J. Mol. Cell Cardiol. 2015, 78, 174–185. [Google Scholar] [CrossRef]
- Gebhart, V.; Reiss, K.; Kollau, A.; Mayer, B.; Gorren, A.C.F. Site and mechanism of uncoupling of nitric-oxide synthase: Uncoupling by monomerization and other misconceptions. Nitric Oxide 2019, 89, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.M.; Huang, A.; Kaley, G.; Sun, D. eNOS uncoupling and endothelial dysfunction in aged vessels. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1829–H1836. [Google Scholar] [CrossRef]
- Musicki, B.; Burnett, A.L. Constitutive NOS uncoupling and NADPH oxidase upregulation in the penis of type 2 diabetic men with erectile dysfunction. Andrology 2017, 5, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Bohmer, A.; Gambaryan, S.; Flentje, M.; Jordan, J.; Tsikas, D. [Ureido-(1)(5)N]citrulline UPLC-MS/MS nitric oxide synthase (NOS) activity assay: Development, validation, and applications to assess NOS uncoupling and human platelets NOS activity. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 965, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Kietadisorn, R.; Juni, R.P.; Moens, A.L. Tackling endothelial dysfunction by modulating NOS uncoupling: New insights into its pathogenesis and therapeutic possibilities. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E481–E495. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Liu, Z.; Lu, H.; Zhang, W.; Mi, Q.; Li, X.; Tang, Y.; Chen, Q.; Ferro, A.; Ji, Y. Pyridoxine inhibits endothelial NOS uncoupling induced by oxidized low-density lipoprotein via the PKCalpha signalling pathway in human umbilical vein endothelial cells. Br. J. Pharmacol. 2012, 165, 754–764. [Google Scholar] [CrossRef]
- Crijns, H.J.; Schotten, U.; Moens, A.L. Is NOS uncoupling the missing link between atrial fibrillation and chronic non-ischaemic cardiomyopathy? Cardiovasc. Res. 2011, 91, 556. [Google Scholar] [CrossRef]
- Moens, A.L.; Leyton-Mange, J.S.; Niu, X.; Yang, R.; Cingolani, O.; Arkenbout, E.K.; Champion, H.C.; Bedja, D.; Gabrielson, K.L.; Chen, J.; et al. Adverse ventricular remodeling and exacerbated NOS uncoupling from pressure-overload in mice lacking the beta3-adrenoreceptor. J. Mol. Cell Cardiol. 2009, 47, 576–585. [Google Scholar] [CrossRef]
- Mollnau, H.; Schulz, E.; Daiber, A.; Baldus, S.; Oelze, M.; August, M.; Wendt, M.; Walter, U.; Geiger, C.; Agrawal, R.; et al. Nebivolol prevents vascular NOS III uncoupling in experimental hyperlipidemia and inhibits NADPH oxidase activity in inflammatory cells. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 615–621. [Google Scholar] [CrossRef]
- Dikalova, A.; Aschner, J.L.; Kaplowitz, M.R.; Cunningham, G.; Summar, M.; Fike, C.D. Combined l-citrulline and tetrahydrobiopterin therapy improves NO signaling and ameliorates chronic hypoxia-induced pulmonary hypertension in newborn pigs. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 318, L762–L772. [Google Scholar] [CrossRef]
- Picciano, A.L.; Crane, B.R. A nitric oxide synthase-like protein from Synechococcus produces NO/NO3 (-) from l-arginine and NADPH in a tetrahydrobiopterin- and Ca(2+)-dependent manner. J. Biol. Chem. 2019, 294, 10708–10719. [Google Scholar] [CrossRef]
- Nagarkoti, S.; Sadaf, S.; Awasthi, D.; Chandra, T.; Jagavelu, K.; Kumar, S.; Dikshit, M. L-Arginine and tetrahydrobiopterin supported nitric oxide production is crucial for the microbicidal activity of neutrophils. Free Radic. Res. 2019, 53, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Latini, A.; de Bortoli da Silva, L.; da Luz Scheffer, D.; Pires, A.C.S.; de Matos, F.J.; Nesi, R.T.; Ghisoni, K.; de Paula Martins, R.; de Oliveira, P.A.; Prediger, R.D.; et al. Tetrahydrobiopterin improves hippocampal nitric oxide-linked long-term memory. Mol. Genet. Metab. 2018, 125, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Chuaiphichai, S.; Rashbrook, V.S.; Hale, A.B.; Trelfa, L.; Patel, J.; McNeill, E.; Lygate, C.A.; Channon, K.M.; Douglas, G. Endothelial Cell Tetrahydrobiopterin Modulates Sensitivity to Ang (Angiotensin) II-Induced Vascular Remodeling, Blood Pressure, and Abdominal Aortic Aneurysm. Hypertension 2018, 72, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Haque, M.M.; Gangoda, M.; Stuehr, D.J. Tetrahydrobiopterin redox cycling in nitric oxide synthase: Evidence supports a through-heme electron delivery. FEBS J. 2016, 283, 4491–4501. [Google Scholar] [CrossRef] [PubMed]
- Stuehr, D.J.; Kwon, N.S.; Nathan, C.F.; Griffith, O.W.; Feldman, P.L.; Wiseman, J. N omega-hydroxy-L-arginine is an intermediate in the biosynthesis of nitric oxide from L-arginine. J. Biol. Chem. 1991, 266, 6259–6263. [Google Scholar]
- Vasquez-Vivar, J.; Kalyanaraman, B.; Martasek, P.; Hogg, N.; Masters, B.S.; Karoui, H.; Tordo, P.; Pritchard, K.A., Jr. Superoxide generation by endothelial nitric oxide synthase: The influence of cofactors. Proc. Natl. Acad. Sci. USA 1998, 95, 9220–9225. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Tsai, A.L.; Berka, V.; Zweier, J.L. Superoxide generation from endothelial nitric-oxide synthase. A Ca2+/calmodulin-dependent and tetrahydrobiopterin regulatory process. J. Biol. Chem. 1998, 273, 25804–25808. [Google Scholar] [CrossRef] [PubMed]
- Landmesser, U.; Dikalov, S.; Price, S.R.; McCann, L.; Fukai, T.; Holland, S.M.; Mitch, W.E.; Harrison, D.G. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J. Clin. Invest. 2003, 111, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.K.; al-Swayeh, O.A.; Chong, N.W.; Evans, R.A.; Gibson, A. L-NG-nitro arginine (L-NOARG), a novel, L-arginine-reversible inhibitor of endothelium-dependent vasodilatation in vitro. Br. J. Pharmacol. 1990, 99, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Gibson, A.; Mirzazadeh, S.; Hobbs, A.J.; Moore, P.K. L-NG-monomethyl arginine and L-NG-nitro arginine inhibit non-adrenergic, non-cholinergic relaxation of the mouse anococcygeus muscle. Br. J. Pharmacol. 1990, 99, 602–606. [Google Scholar] [CrossRef] [PubMed]
- O’Kane, K.P.; Webb, D.J.; Collier, J.G.; Vallance, P.J. Local L-NG-monomethyl-arginine attenuates the vasodilator action of bradykinin in the human forearm. Br. J. Clin. Pharmacol. 1994, 38, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Izumi, Y.; Soma, M.; Watanabe, Y.; Watanabe, M.; Hatano, M.; Sakuma, I.; Yasuda, H. L-NG-monomethyl arginine inhibits the vasodilating effects of low dose of endothelin-3 on rat mesenteric arteries. Biochem. Biophys. Res. Commun. 1990, 167, 739–745. [Google Scholar] [CrossRef]
- Gaw, A.J.; Aberdeen, J.; Humphrey, P.P.; Wadsworth, R.M.; Burnstock, G. Relaxation of sheep cerebral arteries by vasoactive intestinal polypeptide and neurogenic stimulation: Inhibition by L-NG-monomethyl arginine in endothelium-denuded vessels. Br. J. Pharmacol. 1991, 102, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Toda, N.; Minami, Y.; Okamura, T. Inhibitory effects of L-NG-nitro-arginine on the synthesis of EDRF and the cerebroarterial response to vasodilator nerve stimulation. Life Sci. 1990, 47, 345–351. [Google Scholar] [CrossRef]
- Cozzi, M.R.; Guglielmini, G.; Battiston, M.; Momi, S.; Lombardi, E.; Miller, E.C.; De Zanet, D.; Mazzucato, M.; Gresele, P.; De Marco, L. Visualization of nitric oxide production by individual platelets during adhesion in flowing blood. Blood 2015, 125, 697–705. [Google Scholar] [CrossRef]
- Haussinger, D.; Gerok, W.; Sies, H. The effect of urea synthesis on extracellular pH in isolated perfused rat liver. Biochem. J. 1986, 236, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Mavri-Damelin, D.; Eaton, S.; Damelin, L.H.; Rees, M.; Hodgson, H.J.; Selden, C. Ornithine transcarbamylase and arginase I deficiency are responsible for diminished urea cycle function in the human hepatoblastoma cell line HepG2. Int. J. Biochem. Cell Biol. 2007, 39, 555–564. [Google Scholar] [CrossRef]
- Callery, E.M.; Elinson, R.P. Developmental regulation of the urea-cycle enzyme arginase in the direct developing frog Eleutherodactylus coqui. J. Exp. Zool. 1996, 275, 61–66. [Google Scholar] [CrossRef]
- Snellman, K.; Aperia, A.; Broberger, O. Studies of renal urea cycle enzymes. II. Human renal arginase activity and location of the adaptive changes of renal arginase in the protein deprived rat. Scand. J. Clin. Lab. Invest. 1979, 39, 337–342. [Google Scholar] [CrossRef]
- Chan, P.Y.; Cossins, E.A. Regulation of arginase levels by urea and intermediates of the Krebs-Henseleit cycle in Saccharomyces cerevisiae. FEBS Lett. 1972, 19, 335–339. [Google Scholar] [CrossRef]
- Pernow, J.; Jung, C. Arginase as a potential target in the treatment of cardiovascular disease: Reversal of arginine steal? Cardiovasc. Res. 2013, 98, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Pandey, D.; Romer, L.; Berkowitz, D.E. Arginase II: Atherogenesis beyond enzyme activity. J. Am. Heart Assoc. 2013, 2, e000392. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, N.J.; Ward, S.; Piponski, M.; Young, T.W. Purification of human hepatic arginase and its manganese (II)-dependent and pH-dependent interconversion between active and inactive forms: A possible pH-sensing function of the enzyme on the ornithine cycle. Arch. Biochem. Biophys. 1995, 320, 24–34. [Google Scholar] [CrossRef]
- Sumitani, A. Immunological studies of liver arginase in man and various kinds of vertebrates. Part I: Microquantification of arginase enzyme in liver tissue by quantitative immunoelectrophoresis. Part II: Immunological studies of human liver arginase. Hiroshima J. Med. Sci. 1977, 26, 59. [Google Scholar]
- Kim, J.H.; Bugaj, L.J.; Oh, Y.J.; Bivalacqua, T.J.; Ryoo, S.; Soucy, K.G.; Santhanam, L.; Webb, A.; Camara, A.; Sikka, G.; et al. Arginase inhibition restores NOS coupling and reverses endothelial dysfunction and vascular stiffness in old rats. J. Appl. Physiol. (1985) 2009, 107, 1249–1257. [Google Scholar] [CrossRef]
- Romero, M.J.; Platt, D.H.; Tawfik, H.E.; Labazi, M.; El-Remessy, A.B.; Bartoli, M.; Caldwell, R.B.; Caldwell, R.W. Diabetes-induced coronary vascular dysfunction involves increased arginase activity. Circ. Res. 2008, 102, 95–102. [Google Scholar] [CrossRef]
- Mahdi, A.; Kovamees, O.; Pernow, J. Improvement in endothelial function in cardiovascular disease—Is arginase the target? Int. J. Cardiol. 2020, 301, 207–214. [Google Scholar] [CrossRef]
- Wernly, B.; Pernow, J.; Kelm, M.; Jung, C. The role of arginase in the microcirculation in cardiovascular disease. Clin. Hemorheol. Microcirc. 2020, 74, 79–92. [Google Scholar] [CrossRef]
- Chandrasekharan, U.M.; Wang, Z.; Wu, Y.; Wilson Tang, W.H.; Hazen, S.L.; Wang, S.; Elaine Husni, M. Elevated levels of plasma symmetric dimethylarginine and increased arginase activity as potential indicators of cardiovascular comorbidity in rheumatoid arthritis. Arthritis Res. Ther. 2018, 20, 123. [Google Scholar] [CrossRef]
- Yang, Z.; Ming, X.F. Functions of arginase isoforms in macrophage inflammatory responses: Impact on cardiovascular diseases and metabolic disorders. Front. Immunol. 2014, 5, 533. [Google Scholar] [CrossRef] [PubMed]
- Bagnost, T.; Ma, L.; da Silva, R.F.; Rezakhaniha, R.; Houdayer, C.; Stergiopulos, N.; Andre, C.; Guillaume, Y.; Berthelot, A.; Demougeot, C. Cardiovascular effects of arginase inhibition in spontaneously hypertensive rats with fully developed hypertension. Cardiovasc. Res. 2010, 87, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liu, C.; Ming, X.F.; Yang, Z. Inhibition of p38mapk Reduces Adipose Tissue Inflammation in Aging Mediated by Arginase-II. Pharmacology 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Masi, S.; Colucci, R.; Duranti, E.; Nannipieri, M.; Anselmino, M.; Ippolito, C.; Tirotta, E.; Georgiopoulos, G.; Garelli, F.; Nericcio, A.; et al. Aging Modulates the Influence of Arginase on Endothelial Dysfunction in Obesity. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 2474–2483. [Google Scholar] [CrossRef] [PubMed]
- Cecilio, C.A.; Costa, E.H.; Simioni, P.U.; Gabriel, D.L.; Tamashiro, W.M. Aging alters the production of iNOS, arginase and cytokines in murine macrophages. Braz. J. Med. Biol. Res. 2011, 44, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Katusic, Z.S. Mechanisms of endothelial dysfunction induced by aging: Role of arginase I. Circ. Res. 2007, 101, 640–641. [Google Scholar] [CrossRef]
- Sakai, Y.; Masuda, H.; Kihara, K.; Kurosaki, E.; Yamauchi, Y.; Azuma, H. Involvement of increased arginase activity in impaired cavernous relaxation with aging in the rabbit. J. Urol. 2004, 172, 369–373. [Google Scholar] [CrossRef]
- Berkowitz, D.E.; White, R.; Li, D.; Minhas, K.M.; Cernetich, A.; Kim, S.; Burke, S.; Shoukas, A.A.; Nyhan, D.; Champion, H.C.; et al. Arginase reciprocally regulates nitric oxide synthase activity and contributes to endothelial dysfunction in aging blood vessels. Circulation 2003, 108, 2000–2006. [Google Scholar] [CrossRef]
- Cheng, H.; Lu, T.; Wang, J.; Xia, Y.; Chai, X.; Zhang, M.; Yao, Y.; Zhou, N.; Zhou, S.; Chen, X.; et al. HuangqiGuizhiWuwu Decoction Prevents Vascular Dysfunction in Diabetes via Inhibition of Endothelial Arginase 1. Front. Physiol 2020, 11, 201. [Google Scholar] [CrossRef]
- Folley, S.J.; Greenbaum, A.L. Effect of experimental diabetes on tissue arginase levels. J. Endocrinol. 1949, 6. [Google Scholar] [PubMed]
- Yang, J.; Zheng, X.; Mahdi, A.; Zhou, Z.; Tratsiakovich, Y.; Jiao, T.; Kiss, A.; Kovamees, O.; Alvarsson, M.; Catrina, S.B.; et al. Red Blood Cells in Type 2 Diabetes Impair Cardiac Post-Ischemic Recovery Through an Arginase-Dependent Modulation of Nitric Oxide Synthase and Reactive Oxygen Species. JACC Basic Transl. Sci. 2018, 3, 450–463. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Mahdi, A.; Tratsiakovich, Y.; Zahoran, S.; Kovamees, O.; Nordin, F.; Uribe Gonzalez, A.E.; Alvarsson, M.; Ostenson, C.G.; Andersson, D.C.; et al. Erythrocytes From Patients With Type 2 Diabetes Induce Endothelial Dysfunction Via Arginase, I.J. Am. Coll. Cardiol. 2018, 72, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, J.; Qu, D.; Wang, L.; Wong, C.M.; Lau, C.W.; Huang, Y.; Wang, Y.F.; Huang, H.; Xia, Y.; et al. Serum exosomes mediate delivery of arginase 1 as a novel mechanism for endothelial dysfunction in diabetes. Proc. Natl. Acad. Sci. USA 2018, 115, E6927–E6936. [Google Scholar] [CrossRef] [PubMed]
- Shosha, E.; Xu, Z.; Narayanan, S.P.; Lemtalsi, T.; Fouda, A.Y.; Rojas, M.; Xing, J.; Fulton, D.; Caldwell, R.W.; Caldwell, R.B. Mechanisms of Diabetes-Induced Endothelial Cell Senescence: Role of Arginase 1. Int. J. Mol. Sci. 2018, 19, 1215. [Google Scholar] [CrossRef] [PubMed]
- Kovamees, O.; Shemyakin, A.; Checa, A.; Wheelock, C.E.; Lundberg, J.O.; Ostenson, C.G.; Pernow, J. Arginase Inhibition Improves Microvascular Endothelial Function in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2016, 101, 3952–3958. [Google Scholar] [CrossRef]
- Yao, L.; Chandra, S.; Toque, H.A.; Bhatta, A.; Rojas, M.; Caldwell, R.B.; Caldwell, R.W. Prevention of diabetes-induced arginase activation and vascular dysfunction by Rho kinase (ROCK) knockout. Cardiovasc. Res. 2013, 97, 509–519. [Google Scholar] [CrossRef]
- Elms, S.C.; Toque, H.A.; Rojas, M.; Xu, Z.; Caldwell, R.W.; Caldwell, R.B. The role of arginase I in diabetes-induced retinal vascular dysfunction in mouse and rat models of diabetes. Diabetologia 2013, 56, 654–662. [Google Scholar] [CrossRef]
- Shemyakin, A.; Kovamees, O.; Rafnsson, A.; Bohm, F.; Svenarud, P.; Settergren, M.; Jung, C.; Pernow, J. Arginase inhibition improves endothelial function in patients with coronary artery disease and type 2 diabetes mellitus. Circulation 2012, 126, 2943–2950. [Google Scholar] [CrossRef]
- Ren, B.; Van Kampen, E.; Van Berkel, T.J.; Cruickshank, S.M.; Van Eck, M. Hematopoietic arginase 1 deficiency results in decreased leukocytosis and increased foam cell formation but does not affect atherosclerosis. Atherosclerosis 2017, 256, 35–46. [Google Scholar] [CrossRef]
- Teupser, D.; Burkhardt, R.; Wilfert, W.; Haffner, I.; Nebendahl, K.; Thiery, J. Identification of macrophage arginase I as a new candidate gene of atherosclerosis resistance. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 365–371. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Rengarajan, T.; Thangavel, J.; Nishigaki, Y.; Sakthisekaran, D.; Sethi, G.; Nishigaki, I. The vascular endothelium and human diseases. Int. J. Biol. Sci. 2013, 9, 1057–1069. [Google Scholar] [CrossRef] [PubMed]
- Chirinos, J.A.; David, R.; Bralley, J.A.; Zea-Diaz, H.; Munoz-Atahualpa, E.; Corrales-Medina, F.; Cuba-Bustinza, C.; Chirinos-Pacheco, J.; Medina-Lezama, J. Endogenous nitric oxide synthase inhibitors, arterial hemodynamics, and subclinical vascular disease: The Prevencion Study. Hypertension 2008, 52, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Bermejo-Martin, J.F.; Almansa, R.; Torres, A.; Gonzalez-Rivera, M.; Kelvin, D.J. COVID-19 as a cardiovascular disease: The potential role of chronic endothelial dysfunction. Cardiovasc. Res. 2020, 116, e132–e133. [Google Scholar] [CrossRef]
- Daiber, A.; Chlopicki, S. Revisiting pharmacology of oxidative stress and endothelial dysfunction in cardiovascular disease: Evidence for redox-based therapies. Free Radic. Biol. Med. 2020. [Google Scholar] [CrossRef]
- Lima, B.B.; Hammadah, M.; Kim, J.H.; Uphoff, I.; Shah, A.; Levantsevych, O.; Almuwaqqat, Z.; Moazzami, K.; Sullivan, S.; Ward, L.; et al. Association of Transient Endothelial Dysfunction Induced by Mental Stress With Major Adverse Cardiovascular Events in Men and Women With Coronary Artery Disease. JAMA Cardiol. 2019, 4, 988–996. [Google Scholar] [CrossRef]
- Yepuri, G.; Ramasamy, R. Significance and Mechanistic Relevance of SIRT6-Mediated Endothelial Dysfunction in Cardiovascular Disease Progression. Circ. Res. 2019, 124, 1408–1410. [Google Scholar] [CrossRef]
- Daiber, A.; Xia, N.; Steven, S.; Oelze, M.; Hanf, A.; Kroller-Schon, S.; Munzel, T.; Li, H. New Therapeutic Implications of Endothelial Nitric Oxide Synthase (eNOS) Function/Dysfunction in Cardiovascular Disease. Int. J. Mol. Sci. 2019, 20, 187. [Google Scholar] [CrossRef]
- Maruhashi, T.; Soga, J.; Fujimura, N.; Idei, N.; Mikami, S.; Iwamoto, Y.; Iwamoto, A.; Kajikawa, M.; Matsumoto, T.; Oda, N.; et al. Endothelial Dysfunction, Increased Arterial Stiffness, and Cardiovascular Risk Prediction in Patients with Coronary Artery Disease: FMD-J (Flow-Mediated Dilation Japan) Study A. J. Am. Heart Assoc. 2018, 7, e008588. [Google Scholar] [CrossRef]
- Akasaka, T.; Sueta, D.; Tabata, N.; Takashio, S.; Yamamoto, E.; Izumiya, Y.; Tsujita, K.; Kojima, S.; Kaikita, K.; Matsui, K.; et al. Effects of the Mean Amplitude of Glycemic Excursions and Vascular Endothelial Dysfunction on Cardiovascular Events in Nondiabetic Patients With Coronary Artery Disease. J. Am. Heart Assoc. 2017, 6, e004841. [Google Scholar] [CrossRef]
- Chello, M.; Nenna, A. Ethnicity, ABO group, endothelial dysfunction and cardiovascular disease: Multiple connections, multiple implications. Atherosclerosis 2016, 251, 514–515. [Google Scholar] [CrossRef] [PubMed]
- Erqou, S.; Kip, K.E.; Mulukutla, S.R.; Aiyer, A.N.; Reis, S.E. Endothelial Dysfunction and Racial Disparities in Mortality and Adverse Cardiovascular Disease Outcomes. Clin. Cardiol. 2016, 39, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Bodolay, E.; Prohaszka, Z.; Paragh, G.; Csipo, I.; Nagy, G.; Laczik, R.; Demeter, N.; Zold, E.; Nakken, B.; Szegedi, G.; et al. Increased levels of anti-heat-shock protein 60 (anti-Hsp60) indicate endothelial dysfunction, atherosclerosis and cardiovascular diseases in patients with mixed connective tissue disease. Immunol. Res. 2014, 60, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Moody, W.E.; Edwards, N.C.; Madhani, M.; Chue, C.D.; Steeds, R.P.; Ferro, C.J.; Townend, J.N. Endothelial dysfunction and cardiovascular disease in early-stage chronic kidney disease: Cause or association? Atherosclerosis 2012, 223, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Versari, D.; Daghini, E.; Virdis, A.; Ghiadoni, L.; Taddei, S. Endothelial dysfunction as a target for prevention of cardiovascular disease. Diabetes Care 2009, 32 (Suppl. 2), S314–S321. [Google Scholar] [CrossRef] [PubMed]
- Nozaki, T.; Sugiyama, S.; Koga, H.; Sugamura, K.; Ohba, K.; Matsuzawa, Y.; Sumida, H.; Matsui, K.; Jinnouchi, H.; Ogawa, H. Significance of a multiple biomarkers strategy including endothelial dysfunction to improve risk stratification for cardiovascular events in patients at high risk for coronary heart disease. J. Am. Coll. Cardiol. 2009, 54, 601–608. [Google Scholar] [CrossRef]
- Martin, B.J.; Anderson, T.J. Risk prediction in cardiovascular disease: The prognostic significance of endothelial dysfunction. Can. J. Cardiol. 2009, 25 (Suppl. A), 15A–20A. [Google Scholar] [CrossRef]
- Nin, J.W.; Ferreira, I.; Schalkwijk, C.G.; Prins, M.H.; Chaturvedi, N.; Fuller, J.H.; Stehouwer, C.D.; EURODIAB Prospective Complications Study Group. Levels of soluble receptor for AGE are cross-sectionally associated with cardiovascular disease in type 1 diabetes, and this association is partially mediated by endothelial and renal dysfunction and by low-grade inflammation: The Eurodiab Prospective Complications Study. Diabetologia 2009, 52, 705–714. [Google Scholar]
- Friedewald, V.E.; Giles, T.D.; Pool, J.L.; Yancy, C.W.; Roberts, W.C. The Editor’s Roundtable: Endothelial dysfunction in cardiovascular disease. Am. J. Cardiol. 2008, 102, 418–423. [Google Scholar] [CrossRef]
- Rodford, J.L.; Torrens, C.; Siow, R.C.; Mann, G.E.; Hanson, M.A.; Clough, G.F. Endothelial dysfunction and reduced antioxidant protection in an animal model of the developmental origins of cardiovascular disease. J. Physiol. 2008, 586, 4709–4720. [Google Scholar] [CrossRef]
- Xu, Y.; Buikema, H.; van Gilst, W.H.; Henning, R.H. Caveolae and endothelial dysfunction: Filling the caves in cardiovascular disease. Eur. J. Pharmacol. 2008, 585, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Subah Packer, C. Estrogen protection, oxidized LDL, endothelial dysfunction and vasorelaxation in cardiovascular disease: New insights into a complex issue. Cardiovasc. Res. 2007, 73, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Ruef, J.; Marz, W.; Winkelmann, B.R. Markers for endothelial dysfunction, but not markers for oxidative stress correlate with classical risk factors and the severity of coronary artery disease. (A subgroup analysis from the Ludwigshafen Risk and Cardiovascular Health Study). Scand. Cardiovasc. J. 2006, 40, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.; Gluckman, P. Endothelial dysfunction and cardiovascular disease: The role of predictive adaptive responses. Heart 2005, 91, 864–866. [Google Scholar] [CrossRef]
- Brevetti, G.; Silvestro, A.; Schiano, V.; Chiariello, M. Endothelial dysfunction and cardiovascular risk prediction in peripheral arterial disease: Additive value of flow-mediated dilation to ankle-brachial pressure index. Circulation 2003, 108, 2093–2098. [Google Scholar] [CrossRef]
- Gokce, N.; Keaney, J.F., Jr.; Hunter, L.M.; Watkins, M.T.; Nedeljkovic, Z.S.; Menzoian, J.O.; Vita, J.A. Predictive value of noninvasively determined endothelial dysfunction for long-term cardiovascular events in patients with peripheral vascular disease. J. Am. Coll. Cardiol. 2003, 41, 1769–1775. [Google Scholar] [CrossRef]
- Erhardt, L.R. Endothelial dysfunction and cardiovascular disease: The promise of blocking the renin-angiotensin system. Int. J. Clin. Pract. 2003, 57, 211–218. [Google Scholar]
- Sardu, C.; Gambardella, J.; Morelli, M.B.; Wang, X.; Marfella, R.; Santulli, G. Is COVID-19 an Endothelial Disease? Clinical and Basic Evidence. Preprints 2020. [Google Scholar] [CrossRef]
- Sardu, C.; Gambardella, J.; Morelli, M.B.; Wang, X.; Marfella, R.; Santulli, G. Hypertension, Thrombosis, Kidney Failure, and Diabetes: Is COVID-19 an Endothelial Disease? A Comprehensive Evaluation of Clinical and Basic Evidence. J. Clin. Med. 2020, 9, 1417. [Google Scholar] [CrossRef]
- Paules, C.I.; Marston, H.D.; Fauci, A.S. Coronavirus Infections-More Than Just the Common Cold. JAMA 2020, 323, 707–708. [Google Scholar] [CrossRef]
- Hui, D.S.; Azhar, E.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Ippolito, G.; McHugh, T.D.; Memish, Z.A.; Drosten, C.; et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Colmenero, I.; Santonja, C.; Alonso-Riano, M.; Noguera-Morel, L.; Hernandez-Martin, A.; Andina, D.; Wiesner, T.; Rodriguez-Peralto, J.L.; Requena, L.; Torrelo, A. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: Histopathological, immunohistochemical and ultraestructural study of 7 paediatric cases. Br. J. Dermatol. 2020. [Google Scholar] [CrossRef]
- Hanafi, R.; Roger, P.A.; Perin, B.; Kuchcinski, G.; Deleval, N.; Dallery, F.; Michel, D.; Hacein-Bey, L.; Pruvo, J.P.; Outteryck, O.; et al. COVID-19 Neurologic Complication with CNS Vasculitis-Like Pattern. AJNR Am. J. Neuroradiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mosleh, W.; Chen, K.; Pfau, S.E.; Vashist, A. Endotheliitis and Endothelial Dysfunction in Patients with COVID-19: Its Role in Thrombosis and Adverse Outcomes. J. Clin. Med. 2020, 9, 1862. [Google Scholar] [CrossRef] [PubMed]
- Pons, S.; Fodil, S.; Azoulay, E.; Zafrani, L. The vascular endothelium: The cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit. Care 2020, 24, 353. [Google Scholar] [CrossRef]
- Konopka, K.E.; Nguyen, T.; Jentzen, J.M.; Rayes, O.; Schmidt, C.J.; Wilson, A.M.; Farver, C.F.; Myers, J.L. Diffuse Alveolar Damage (DAD) from Coronavirus Disease 2019 Infection is Morphologically Indistinguishable from Other Causes of DAD. Histopathology 2020. [Google Scholar] [CrossRef]
- Benger, M.; Williams, O.; Siddiqui, J.; Sztriha, L. Intracerebral haemorrhage and COVID-19: Clinical characteristics from a case series. Brain Behav. Immun. 2020, 88, 940–944. [Google Scholar] [CrossRef]
- Wang, J.; Saguner, A.M.; An, J.; Ning, Y.; Yan, Y.; Li, G. Dysfunctional Coagulation in COVID-19: From Cell to Bedside. Adv. Ther. 2020, 37, 3033–3039. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Jones, V.G.; Mills, M.; Suarez, D.; Hogan, C.A.; Yeh, D.; Bradley Segal, J.; Nguyen, E.L.; Barsh, G.R.; Maskatia, S.; Mathew, R. COVID-19 and Kawasaki Disease: Novel Virus and Novel Case. Hosp. Pediatr. 2020, 10, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Cyr, A.R.; Huckaby, L.V.; Shiva, S.S.; Zuckerbraun, B.S. Nitric Oxide and Endothelial Dysfunction. Crit. Care Clin. 2020, 36, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, J.; Shaik, N.F.; Yi, B.; Wei, X.; Yang, X.F.; Naik, U.P.; Summer, R.; Yan, G.; Xu, X.; et al. The histone deacetylase inhibitor tubacin mitigates endothelial dysfunction by up-regulating the expression of endothelial nitric oxide synthase. J. Biol. Chem. 2019, 294, 19565–19576. [Google Scholar] [CrossRef] [PubMed]
- Lomeli, O.; Perez-Torres, I.; Marquez, R.; Criales, S.; Mejia, A.M.; Chiney, C.; Hernandez-Lemus, E.; Soto, M.E. The Evaluation of Flow-Mediated Vasodilation in the Brachial Artery Correlates With Endothelial Dysfunction Evaluated by Nitric Oxide Synthase Metabolites in Marfan Syndrome Patients. Front. Physiol. 2018, 9, 965. [Google Scholar] [CrossRef]
- Evans, R.W.; Fernstrom, J.D.; Thompson, J.; Morris, S.M., Jr.; Kuller, L.H. Biochemical responses of healthy subjects during dietary supplementation with L-arginine. J. Nutr. Biochem. 2004, 15, 534–539. [Google Scholar] [CrossRef]
- Alvares, T.S.; Conte-Junior, C.A.; Silva, J.T.; Paschoalin, V.M. Acute L-Arginine supplementation does not increase nitric oxide production in healthy subjects. Nutr. Metab. 2012, 9, 54. [Google Scholar] [CrossRef]
- Meirelles, C.M.; Matsuura, C.; Silva, R.S., Jr.; Guimaraes, F.F.; Gomes, P.S.C. Acute Effects of L-Arginine Supplementation on Oxygen Consumption Kinetics and Muscle Oxyhemoglobin and Deoxyhemoglobin during Treadmill Running in Male Adults. Int. J. Exerc. Sci. 2019, 12, 444–455. [Google Scholar]
- Liu, T.H.; Wu, C.L.; Chiang, C.W.; Lo, Y.W.; Tseng, H.F.; Chang, C.K. No effect of short-term arginine supplementation on nitric oxide production, metabolism and performance in intermittent exercise in athletes. J. Nutr. Biochem. 2009, 20, 462–468. [Google Scholar] [CrossRef]
- Schwedhelm, E.; Maas, R.; Freese, R.; Jung, D.; Lukacs, Z.; Jambrecina, A.; Spickler, W.; Schulze, F.; Boger, R.H. Pharmacokinetic and pharmacodynamic properties of oral L-citrulline and L-arginine: Impact on nitric oxide metabolism. Br. J. Clin. Pharmacol. 2008, 65, 51–59. [Google Scholar] [CrossRef]
- Bredt, D.S.; Snyder, S.H. Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc. Natl. Acad. Sci. USA 1990, 87, 682–685. [Google Scholar] [CrossRef]
- Joyner, M.J.; Casey, D.P. Regulation of increased blood flow (hyperemia) to muscles during exercise: A hierarchy of competing physiological needs. Physiol. Rev. 2015, 95, 549–601. [Google Scholar] [CrossRef] [PubMed]
- Alvares, T.S.; Meirelles, C.M.; Bhambhani, Y.N.; Paschoalin, V.M.; Gomes, P.S. L-Arginine as a potential ergogenic aid in healthy subjects. Sports Med. 2011, 41, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Doutreleau, S.; Rouyer, O.; Di Marco, P.; Lonsdorfer, E.; Richard, R.; Piquard, F.; Geny, B. L-arginine supplementation improves exercise capacity after a heart transplant. Am. J. Clin. Nutr. 2010, 91, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J. Nutritional ergogenic aids and exercise performance. Nutr. Res. Rev. 1999, 12, 255–280. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, I.; Sakuraba, K.; Horiike, T.; Kishi, T.; Yabe, J.; Suzuki, T.; Morita, M.; Nishimura, A.; Suzuki, Y. A combination of oral L-citrulline and L-arginine improved 10-min full-power cycling test performance in male collegiate soccer players: A randomized crossover trial. Eur. J. Appl. Physiol. 2019, 119, 1075–1084. [Google Scholar] [CrossRef]
- Andrade, W.B.; Jacinto, J.L.; da Silva, D.K.; Roveratti, M.C.; Estoche, J.M.; Oliveira, D.B.; Balvedi, M.C.W.; da Silva, R.A.; Aguiar, A.F. l-Arginine supplementation does not improve muscle function during recovery from resistance exercise. Appl. Physiol. Nutr. Metab. 2018, 43, 928–936. [Google Scholar] [CrossRef]
- Dioguardi, F.S. To give or not to give? Lessons from the arginine paradox. J. Nutrigenet. Nutrige. 2011, 4, 90–98. [Google Scholar] [CrossRef]
- Ueda, S.; Petrie, J.R.; Cleland, S.J.; Elliott, H.L.; Connell, J.M. Insulin vasodilatation and the “arginine paradox”. Lancet 1998, 351, 959–960. [Google Scholar] [CrossRef]
- Kurz, S.; Harrison, D.G. Insulin and the arginine paradox. J. Clin. Investig. 1997, 99, 369–370. [Google Scholar] [CrossRef]
- Vallance, P.; Leone, A.; Calver, A.; Collier, J.; Moncada, S. Endogenous dimethylarginine as an inhibitor of nitric oxide synthesis. J. Cardiovasc. Pharmacol. 1992, 20 (Suppl. 12), S60–S62. [Google Scholar] [CrossRef]
- Bartnicki, P.; Kowalczyk, M.; Franczyk-Skora, B.; Baj, Z.; Rysz, J. Evaluation of Endothelial (dys)Function, Left Ventricular Structure and Function in Patients with Chronic Kidney Disease. Curr. Vasc. Pharmacol. 2016, 14, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Bouras, G.; Deftereos, S.; Tousoulis, D.; Giannopoulos, G.; Chatzis, G.; Tsounis, D.; Cleman, M.W.; Stefanadis, C. Asymmetric Dimethylarginine (ADMA): A promising biomarker for cardiovascular disease? Curr. Top. Med. Chem. 2013, 13, 180–200. [Google Scholar] [CrossRef] [PubMed]
- Janes, F.; Cifu, A.; Pessa, M.E.; Domenis, R.; Gigli, G.L.; Sanvilli, N.; Nilo, A.; Garbo, R.; Curcio, F.; Giacomello, R.; et al. ADMA as a possible marker of endothelial damage. A study in young asymptomatic patients with cerebral small vessel disease. Sci. Rep. 2019, 9, 14207. [Google Scholar] [CrossRef] [PubMed]
- Strobel, J.; Muller, F.; Zolk, O.; Endress, B.; Konig, J.; Fromm, M.F.; Maas, R. Transport of asymmetric dimethylarginine (ADMA) by cationic amino acid transporter 2 (CAT2), organic cation transporter 2 (OCT2) and multidrug and toxin extrusion protein 1 (MATE1). Amino Acids 2013, 45, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Closs, E.I.; Basha, F.Z.; Habermeier, A.; Forstermann, U. Interference of L-arginine analogues with L-arginine transport mediated by the y+ carrier hCAT-2B. Nitric Oxide 1997, 1, 65–73. [Google Scholar] [CrossRef]
- Wijnands, K.A.; Hoeksema, M.A.; Meesters, D.M.; van den Akker, N.M.; Molin, D.G.; Briede, J.J.; Ghosh, M.; Kohler, S.E.; van Zandvoort, M.A.; de Winther, M.P.; et al. Arginase-1 deficiency regulates arginine concentrations and NOS2-mediated NO production during endotoxemia. PLoS ONE 2014, 9, e86135. [Google Scholar] [CrossRef]
- Boger, R.H.; Bode-Boger, S.M.; Szuba, A.; Tsao, P.S.; Chan, J.R.; Tangphao, O.; Blaschke, T.F.; Cooke, J.P. Asymmetric dimethylarginine (ADMA): A novel risk factor for endothelial dysfunction: Its role in hypercholesterolemia. Circulation 1998, 98, 1842–1847. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; Teerlink, T.; Van Der Hoven, B.; Siroen, M.P.; Kuik, D.J.; Rauwerda, J.A.; van Leeuwen, P.A. Asymmetrical dimethylarginine (ADMA) in critically ill patients: High plasma ADMA concentration is an independent risk factor of ICU mortality. Clin. Nutr. 2003, 22, 23–30. [Google Scholar] [CrossRef]
- Abedini, S.; Meinitzer, A.; Holme, I.; Marz, W.; Weihrauch, G.; Fellstrom, B.; Jardine, A.; Holdaas, H. Asymmetrical dimethylarginine is associated with renal and cardiovascular outcomes and all-cause mortality in renal transplant recipients. Kidney Int. 2010, 77, 44–50. [Google Scholar] [CrossRef]
- Krzyzanowska, K.; Mittermayer, F.; Wolzt, M.; Schernthaner, G. Asymmetric dimethylarginine predicts cardiovascular events in patients with type 2 diabetes. Diabetes Care 2007, 30, 1834–1839. [Google Scholar] [CrossRef]
- Nanayakkara, P.W.; Teerlink, T.; Stehouwer, C.D.; Allajar, D.; Spijkerman, A.; Schalkwijk, C.; ter Wee, P.M.; van Guldener, C. Plasma asymmetric dimethylarginine (ADMA) concentration is independently associated with carotid intima-media thickness and plasma soluble vascular cell adhesion molecule-1 (sVCAM-1) concentration in patients with mild-to-moderate renal failure. Kidney Int. 2005, 68, 2230–2236. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Bode-Boger, S.; Mallamaci, F.; Benedetto, F.; Tripepi, G.; Malatino, L.; Cataliotti, A.; Bellanuova, I.; Fermo, I.; Frolich, J.; et al. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: A prospective study. Lancet 2001, 358, 2113–2117. [Google Scholar] [CrossRef]
- Abbasi, F.; Asagmi, T.; Cooke, J.P.; Lamendola, C.; McLaughlin, T.; Reaven, G.M.; Stuehlinger, M.; Tsao, P.S. Plasma concentrations of asymmetric dimethylarginine are increased in patients with type 2 diabetes mellitus. Am. J. Cardiol. 2001, 88, 1201–1203. [Google Scholar] [CrossRef]
- Furuki, K.; Adachi, H.; Matsuoka, H.; Enomoto, M.; Satoh, A.; Hino, A.; Hirai, Y.; Imaizumi, T. Plasma levels of asymmetric dimethylarginine (ADMA) are related to intima-media thickness of the carotid artery: An epidemiological study. Atherosclerosis 2007, 191, 206–210. [Google Scholar] [CrossRef]
- Valkonen, V.P.; Paiva, H.; Salonen, J.T.; Lakka, T.A.; Lehtimaki, T.; Laakso, J.; Laaksonen, R. Risk of acute coronary events and serum concentration of asymmetrical dimethylarginine. Lancet 2001, 358, 2127–2128. [Google Scholar] [CrossRef]
- Surdacki, A.; Nowicki, M.; Sandmann, J.; Tsikas, D.; Boeger, R.H.; Bode-Boeger, S.M.; Kruszelnicka-Kwiatkowska, O.; Kokot, F.; Dubiel, J.S.; Froelich, J.C. Reduced urinary excretion of nitric oxide metabolites and increased plasma levels of asymmetric dimethylarginine in men with essential hypertension. J. Cardiovasc. Pharmacol. 1999, 33, 652–658. [Google Scholar] [CrossRef]
- Maas, R.; Xanthakis, V.; Polak, J.F.; Schwedhelm, E.; Sullivan, L.M.; Benndorf, R.; Schulze, F.; Vasan, R.S.; Wolf, P.A.; Boger, R.H.; et al. Association of the endogenous nitric oxide synthase inhibitor ADMA with carotid artery intimal media thickness in the Framingham Heart Study offspring cohort. Stroke 2009, 40, 2715–2719. [Google Scholar] [CrossRef]
- Bai, Y.; Sun, L.; Du, L.; Zhang, T.; Xin, W.; Lan, X.; Du, G. Association of circulating levels of asymmetric dimethylarginine (ADMA) with carotid intima-media thickness: Evidence from 6168 participants. Ageing Res. Rev. 2013, 12, 699–707. [Google Scholar] [CrossRef]
- Sundar, U.M.; Ugusman, A.; Chua, H.K.; Latip, J.; Aminuddin, A. Piper sarmentosum Promotes Endothelial Nitric Oxide Production by Reducing Asymmetric Dimethylarginine in Tumor Necrosis Factor-alpha-Induced Human Umbilical Vein Endothelial Cells. Front. Pharmacol 2019, 10, 1033. [Google Scholar] [CrossRef]
- Arlouskaya, Y.; Sawicka, A.; Glowala, M.; Giebultowicz, J.; Korytowska, N.; Talalaj, M.; Nowicka, G.; Wrzosek, M. Asymmetric Dimethylarginine (ADMA) and Symmetric Dimethylarginine (SDMA) Concentrations in Patients with Obesity and the Risk of Obstructive Sleep Apnea (OSA). J. Clin. Med. 2019, 8, 897. [Google Scholar] [CrossRef]
- Hsu, C.P.; Zhao, J.F.; Lin, S.J.; Shyue, S.K.; Guo, B.C.; Lu, T.M.; Lee, T.S. Asymmetric Dimethylarginine Limits the Efficacy of Simvastatin Activating Endothelial Nitric Oxide Synthase. J. Am. Heart Assoc. 2016, 5, e003327. [Google Scholar] [CrossRef] [PubMed]
- Notsu, Y.; Yano, S.; Shibata, H.; Nagai, A.; Nabika, T. Plasma arginine/ADMA ratio as a sensitive risk marker for atherosclerosis: Shimane CoHRE study. Atherosclerosis 2015, 239, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Boger, R.H.; Endres, H.G.; Schwedhelm, E.; Darius, H.; Atzler, D.; Luneburg, N.; von Stritzky, B.; Maas, R.; Thiem, U.; Benndorf, R.A.; et al. Asymmetric dimethylarginine as an independent risk marker for mortality in ambulatory patients with peripheral arterial disease. J. Intern. Med. 2011, 269, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Pizzarelli, F.; Maas, R.; Dattolo, P.; Tripepi, G.; Michelassi, S.; D’Arrigo, G.; Mieth, M.; Bandinelli, S.; Ferrucci, L.; Zoccali, C. Asymmetric dimethylarginine predicts survival in the elderly. Age 2013, 35, 2465–2475. [Google Scholar] [CrossRef] [PubMed]
- Notsu, Y.; Nabika, T.; Bokura, H.; Suyama, Y.; Kobayashi, S.; Yamaguchi, S.; Masuda, J. Evaluation of asymmetric dimethylarginine and homocysteine in microangiopathy-related cerebral damage. Am. J. Hypertens. 2009, 22, 257–262. [Google Scholar] [CrossRef] [PubMed]
- McDonald, K.K.; Zharikov, S.; Block, E.R.; Kilberg, M.S. A caveolar complex between the cationic amino acid transporter 1 and endothelial nitric-oxide synthase may explain the “arginine paradox”. J. Biol. Chem. 1997, 272, 31213–31216. [Google Scholar] [CrossRef]
- Kim, J.A.; Montagnani, M.; Chandrasekran, S.; Quon, M.J. Role of lipotoxicity in endothelial dysfunction. Heart Fail. Clin. 2012, 8, 589–607. [Google Scholar] [CrossRef]
- Benowitz, N.L. Cigarette smoking and cardiovascular disease: Pathophysiology and implications for treatment. Prog. Cardiovasc. Dis. 2003, 46, 91–111. [Google Scholar] [CrossRef]
- Januzzi, J. Cardiac Biomarkers in Clinical Practice; Jones & Bartlett Learning: Burlington, MA, USA, 2009. [Google Scholar]
- Rohde, D.; Busch, M.; Volkert, A.; Ritterhoff, J.; Katus, H.A.; Peppel, K.; Most, P. Cardiomyocytes, endothelial cells and cardiac fibroblasts: S100A1’s triple action in cardiovascular pathophysiology. Future Cardiol. 2015, 11, 309–321. [Google Scholar] [CrossRef]
- Davies, P.F. Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat. Clin. Pract. Cardiovasc. Med. 2009, 6, 16–26. [Google Scholar] [CrossRef]
- Santulli, G. microRNAs Distinctively Regulate Vascular Smooth Muscle and Endothelial Cells: Functional Implications in Angiogenesis, Atherosclerosis, and In-Stent Restenosis. Adv. Exp. Med. Biol. 2015, 887, 53–77. [Google Scholar] [PubMed]
- Foster, W.; Carruthers, D.; Lip, G.Y.; Blann, A.D. Relationships between endothelial, inflammatory and angiogenesis markers in rheumatoid arthritis: Implications for cardiovascular pathophysiology. Thromb. Res. 2009, 123, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Ciccarelli, M.; Santulli, G.; Campanile, A.; Galasso, G.; Cervero, P.; Altobelli, G.G.; Cimini, V.; Pastore, L.; Piscione, F.; Trimarco, B.; et al. Endothelial alpha1-adrenoceptors regulate neo-angiogenesis. Br. J. Pharmacol. 2008, 153, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Luksha, L.; Agewall, S.; Kublickiene, K. Endothelium-derived hyperpolarizing factor in vascular physiology and cardiovascular disease. Atherosclerosis 2009, 202, 330–344. [Google Scholar] [CrossRef]
- Santulli, G.; Cipolletta, E.; Sorriento, D.; Del Giudice, C.; Anastasio, A.; Monaco, S.; Maione, A.S.; Condorelli, G.; Puca, A.; Trimarco, B.; et al. CaMK4 Gene Deletion Induces Hypertension. J. Am. Heart Assoc. 2012, 1, e001081. [Google Scholar] [CrossRef]
- Deedwania, P.C. Diabetes is a vascular disease: The role of endothelial dysfunction in pathophysiology of cardiovascular disease in diabetes. Cardiol. Clin. 2004, 22, 505–509. [Google Scholar] [CrossRef]
- Sorriento, D.; Santulli, G.; Del Giudice, C.; Anastasio, A.; Trimarco, B.; Iaccarino, G. Endothelial cells are able to synthesize and release catecholamines both in vitro and in vivo. Hypertension 2012, 60, 129–136. [Google Scholar] [CrossRef]
- Li, J.M.; Shah, A.M. Endothelial cell superoxide generation: Regulation and relevance for cardiovascular pathophysiology. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R1014–R1030. [Google Scholar] [CrossRef]
- Ciccarelli, M.; Cipolletta, E.; Santulli, G.; Campanile, A.; Pumiglia, K.; Cervero, P.; Pastore, L.; Astone, D.; Trimarco, B.; Iaccarino, G. Endothelial beta2 adrenergic signaling to AKT: Role of Gi and SRC. Cell Signal. 2007, 19, 1949–1955. [Google Scholar] [CrossRef]
- Sellke, F.W.; Boyle, E.M., Jr.; Verrier, E.D. Endothelial cell injury in cardiovascular surgery: The pathophysiology of vasomotor dysfunction. Ann. Thorac. Surg. 1996, 62, 1222–1228. [Google Scholar] [CrossRef]
- Iaccarino, G.; Ciccarelli, M.; Sorriento, D.; Galasso, G.; Campanile, A.; Santulli, G.; Cipolletta, E.; Cerullo, V.; Cimini, V.; Altobelli, G.G.; et al. Ischemic neoangiogenesis enhanced by beta2-adrenergic receptor overexpression: A novel role for the endothelial adrenergic system. Circ. Res. 2005, 97, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Ogita, H.; Liao, J. Endothelial function and oxidative stress. Endothelium 2004, 11, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Skrypnyk, I.; Maslova, G.; Lymanets, T.; Gusachenko, I. L-arginine is an effective medication for prevention of endothelial dysfunction, a predictor of anthracycline cardiotoxicity in patients with acute leukemia. Exp. Oncol. 2017, 39, 308–311. [Google Scholar] [CrossRef]
- Ignarro, L.J.; Napoli, C. Novel features of nitric oxide, endothelial nitric oxide synthase, and atherosclerosis. Curr. Diab. Rep. 2005, 5, 17–23. [Google Scholar] [CrossRef]
- Nguyen, M.C.; Park, J.T.; Jeon, Y.G.; Jeon, B.H.; Hoe, K.L.; Kim, Y.M.; Lim, H.K.; Ryoo, S. Arginase Inhibition Restores Peroxynitrite-Induced Endothelial Dysfunction via L-Arginine-Dependent Endothelial Nitric Oxide Synthase Phosphorylation. Yonsei Med. J. 2016, 57, 1329–1338. [Google Scholar] [CrossRef]
- Wu, G.; Meininger, C.J. Arginine nutrition and cardiovascular function. J. Nutr. 2000, 130, 2626–2629. [Google Scholar] [CrossRef]
- Scaglia, F.; Brunetti-Pierri, N.; Kleppe, S.; Marini, J.; Carter, S.; Garlick, P.; Jahoor, F.; O’Brien, W.; Lee, B. Clinical consequences of urea cycle enzyme deficiencies and potential links to arginine and nitric oxide metabolism. J. Nutr. 2004, 134 (Suppl. 10), 2775S–2782S. [Google Scholar] [CrossRef]
- Luneburg, N.; Xanthakis, V.; Schwedhelm, E.; Sullivan, L.M.; Maas, R.; Anderssohn, M.; Riederer, U.; Glazer, N.L.; Vasan, R.S.; Boger, R.H. Reference intervals for plasma L-arginine and the L-arginine:asymmetric dimethylarginine ratio in the Framingham Offspring Cohort. J. Nutr. 2011, 141, 2186–2190. [Google Scholar] [CrossRef]
- Contreras, M.T.; Gallardo, M.J.; Betancourt, L.R.; Rada, P.V.; Ceballos, G.A.; Hernandez, L.E.; Hernandez, L.F. Correlation between plasma levels of arginine and citrulline in preterm and full-term neonates: Therapeutical implications. J. Clin. Lab. Anal. 2017, 31, e22134. [Google Scholar] [CrossRef]
- Fernandez Diaz-Rullo, F.; Zamberlan, F.; Mewis, R.E.; Fekete, M.; Broche, L.; Cheyne, L.A.; Dall’Angelo, S.; Duckett, S.B.; Dawson, D.; Zanda, M. Synthesis and hyperpolarisation of eNOS substrates for quantification of NO production by (1)H NMR spectroscopy. Bioorg. Med. Chem. 2017, 25, 2730–2742. [Google Scholar] [CrossRef]
- Schmidt, H.H.; Warner, T.D.; Ishii, K.; Sheng, H.; Murad, F. Insulin secretion from pancreatic B cells caused by L-arginine-derived nitrogen oxides. Science 1992, 255, 721–723. [Google Scholar] [CrossRef] [PubMed]
- Fajans, S.S.; Floyd, J.C., Jr.; Knopf, R.F.; Conn, F.W. Effect of amino acids and proteins on insulin secretion in man. Recent Prog. Horm. Res. 1967, 23, 617–662. [Google Scholar] [PubMed]
- Sener, A.; Best, L.C.; Yates, A.P.; Kadiata, M.M.; Olivares, E.; Louchami, K.; Jijakli, H.; Ladriere, L.; Malaisse, W.J. Stimulus-secretion coupling of arginine-induced insulin release: Comparison between the cationic amino acid and its methyl ester. Endocrine 2000, 13, 329–340. [Google Scholar] [CrossRef]
- Gerich, J.E.; Lorenzi, M.; Schneider, V.; Kwan, C.W.; Karam, J.H.; Guillemin, R.; Forsham, P.H. Inhibition of pancreatic glucagon responses to arginine by somatostatin in normal man and in insulin-dependent diabetics. Diabetes 1974, 23, 876–880. [Google Scholar] [CrossRef]
- Giugliano, D.; Marfella, R.; Verrazzo, G.; Acampora, R.; Coppola, L.; Cozzolino, D.; D’Onofrio, F. The vascular effects of L-Arginine in humans. The role of endogenous insulin. J. Clin. Investig. 1997, 99, 433–438. [Google Scholar] [CrossRef]
- Merimee, T.J.; Rabinowitz, D.; Riggs, L.; Burgess, J.A.; Rimoin, D.L.; McKusick, V.A. Plasma growth hormone after arginine infusion. Clinical experiences. N. Engl. J. Med. 1967, 276, 434–439. [Google Scholar] [CrossRef]
- Fryburg, D.A. NG-monomethyl-L-arginine inhibits the blood flow but not the insulin-like response of forearm muscle to IGF- I: Possible role of nitric oxide in muscle protein synthesis. J. Clin. Investig. 1996, 97, 1319–1328. [Google Scholar] [CrossRef]
- Bode-Boger, S.M.; Boger, R.H.; Loffler, M.; Tsikas, D.; Brabant, G.; Frolich, J.C. L-arginine stimulates NO-dependent vasodilation in healthy humans--effect of somatostatin pretreatment. J. Investig. Med. 1999, 47, 43–50. [Google Scholar]
- Taddei, S.; Virdis, A.; Mattei, P.; Ghiadoni, L.; Gennari, A.; Fasolo, C.B.; Sudano, I.; Salvetti, A. Aging and endothelial function in normotensive subjects and patients with essential hypertension. Circulation 1995, 91, 1981–1987. [Google Scholar] [CrossRef]
- Celermajer, D.S.; Sorensen, K.E.; Gooch, V.M.; Spiegelhalter, D.J.; Miller, O.I.; Sullivan, I.D.; Lloyd, J.K.; Deanfield, J.E. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 1992, 340, 1111–1115. [Google Scholar] [CrossRef]
- Gates, P.E.; Boucher, M.L.; Silver, A.E.; Monahan, K.D.; Seals, D.R. Impaired flow-mediated dilation with age is not explained by L-arginine bioavailability or endothelial asymmetric dimethylarginine protein expression. J. Appl. Physiol. 2007, 102, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Campo, C.; Lahera, V.; Garcia-Robles, R.; Cachofeiro, V.; Alcazar, J.M.; Andres, A.; Rodicio, J.L.; Ruilope, L.M. Aging abolishes the renal response to L-arginine infusion in essential hypertension. Kidney Int. Suppl. 1996, 55, S126–S128. [Google Scholar] [PubMed]
- Blum, A.; Hathaway, L.; Mincemoyer, R.; Schenke, W.H.; Kirby, M.; Csako, G.; Waclawiw, M.A.; Panza, J.A.; Cannon, R.O., III. Effects of oral L-arginine on endothelium-dependent vasodilation and markers of inflammation in healthy postmenopausal women. J. Am. Coll. Cardiol. 2000, 35, 271–276. [Google Scholar] [CrossRef]
- Bode-Boger, S.M.; Muke, J.; Surdacki, A.; Brabant, G.; Boger, R.H.; Frolich, J.C. Oral L-arginine improves endothelial function in healthy individuals older than 70 years. Vasc. Med. 2003, 8, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Y.; St John, P.L.; Kirk, K.A.; Abrahamson, D.R.; Sanders, P.W. Hypertensive nephrosclerosis in the Dahl/Rapp rat. Initial sites of injury and effect of dietary L-arginine supplementation. Lab. Investig. 1993, 68, 174–184. [Google Scholar] [PubMed]
- Zhou, M.S.; Kosaka, H.; Tian, R.X.; Abe, Y.; Chen, Q.H.; Yoneyama, H.; Yamamoto, A.; Zhang, L. L-Arginine improves endothelial function in renal artery of hypertensive Dahl rats. J. Hypertens. 2001, 19, 421–429. [Google Scholar] [CrossRef]
- Fujii, S.; Zhang, L.; Igarashi, J.; Kosaka, H. L-arginine reverses p47phox and gp91phox expression induced by high salt in Dahl rats. Hypertension 2003, 42, 1014–1020. [Google Scholar] [CrossRef]
- Patel, A.; Layne, S.; Watts, D.; Kirchner, K.A. L-arginine administration normalizes pressure natriuresis in hypertensive Dahl rats. Hypertension 1993, 22, 863–869. [Google Scholar] [CrossRef]
- Chen, P.Y.; Sanders, P.W. L-arginine abrogates salt-sensitive hypertension in Dahl/Rapp rats. J. Clin. Investig. 1991, 88, 1559–1567. [Google Scholar] [CrossRef]
- Miyata, N.; Zou, A.P.; Mattson, D.L.; Cowley, A.W., Jr. Renal medullary interstitial infusion of L-arginine prevents hypertension in Dahl salt-sensitive rats. Am. J. Physiol. 1998, 275, R1667–R1673. [Google Scholar] [CrossRef]
- Ozcelikay, A.T.; Tay, A.; Guner, S.; Tasyaran, V.; Yildizoglu-Ari, N.; Dincer, U.D.; Altan, V.M. Reversal effects of L-arginine treatment on blood pressure and vascular responsiveness of streptozotocin-diabetic rats. Pharmacol. Res. 2000, 41, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Tay, A.; Ozcelikay, A.T.; Altan, V.M. Effects of L-arginine on blood pressure and metabolic changes in fructose-hypertensive rats. Am. J. Hypertens. 2002, 15 Pt 1, 72–77. [Google Scholar] [CrossRef]
- Ono, H.; Ono, Y.; Frohlich, E.D. L-arginine reverses severe nephrosclerosis in aged spontaneously hypertensive rats. J. Hypertens. 1999, 17, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Wang, B.; Gao, G.; Cao, W.; Zhang, Y. L-Arginine supplementation improves antioxidant defenses through L-arginine/nitric oxide pathways in exercised rats. J. Appl. Physiol. 2013, 115, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.T.; Yang, S.C.; Chen, K.T.; Huang, C.C.; Lee, N.Y. Protective effects of L-arginine on pulmonary oxidative stress and antioxidant defenses during exhaustive exercise in rats. Acta Pharmacol. Sin. 2005, 26, 992–999. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Tsai, S.C.; Lin, W.T. Potential ergogenic effects of L-arginine against oxidative and inflammatory stress induced by acute exercise in aging rats. Exp. Gerontol. 2008, 43, 571–577. [Google Scholar] [CrossRef] [PubMed]
- de Nigris, F.; Lerman, L.O.; Ignarro, S.W.; Sica, G.; Lerman, A.; Palinski, W.; Ignarro, L.J.; Napoli, C. Beneficial effects of antioxidants and L-arginine on oxidation-sensitive gene expression and endothelial NO synthase activity at sites of disturbed shear stress. Proc. Natl. Acad. Sci. USA 2003, 100, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Sattar, M.Z.; Rathore, H.A.; Hussain, A.I.; Khan, S.A.; Fatima, T.; Afzal, S.; Abdullah, N.A.; Johns, E.J. Antioxidant Activity and Free Radical Scavenging Capacity of L-Arginine and Nahs: A Comparative in Vitro Study. Acta Pol. Pharm. 2015, 72, 245–252. [Google Scholar]
- Wallner, S.; Hermetter, A.; Mayer, B.; Wascher, T.C. The alpha-amino group of L-arginine mediates its antioxidant effect. Eur. J. Clin. Investig. 2001, 31, 98–102. [Google Scholar] [CrossRef]
- Suliburska, J.; Bogdanski, P.; Krejpcio, Z.; Pupek-Musialik, D.; Jablecka, A. The effects of L-arginine, alone and combined with vitamin C, on mineral status in relation to its antidiabetic, anti-inflammatory, and antioxidant properties in male rats on a high-fat diet. Biol. Trace Elem. Res. 2014, 157, 67–74. [Google Scholar] [CrossRef]
- Zheng, P.; Yu, B.; He, J.; Tian, G.; Luo, Y.; Mao, X.; Zhang, K.; Che, L.; Chen, D. Protective effects of dietary arginine supplementation against oxidative stress in weaned piglets. Br. J. Nutr. 2013, 109, 2253–2260. [Google Scholar] [CrossRef] [PubMed]
- Fazelian, S.; Hoseini, M.; Namazi, N.; Heshmati, J.; Sepidar Kish, M.; Mirfatahi, M.; Some Olia, A.S. Effects of L-Arginine Supplementation on Antioxidant Status and Body Composition in Obese Patients with Pre-diabetes: A Randomized Controlled Clinical Trial. Adv. Pharm. Bull. 2014, 4 (Suppl. 1), 449–454. [Google Scholar] [PubMed]
- Wascher, T.C.; Posch, K.; Wallner, S.; Hermetter, A.; Kostner, G.M.; Graier, W.F. Vascular effects of L-arginine: Anything beyond a substrate for the NO-synthase? Biochem Biophys Res. Commun 1997, 234, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Lekakis, J.P.; Papathanassiou, S.; Papaioannou, T.G.; Papamichael, C.M.; Zakopoulos, N.; Kotsis, V.; Dagre, A.G.; Stamatelopoulos, K.; Protogerou, A.; Stamatelopoulos, S.F. Oral L-arginine improves endothelial dysfunction in patients with essential hypertension. Int. J. Cardiol. 2002, 86, 317–323. [Google Scholar] [CrossRef]
- Higashi, Y.; Oshima, T.; Watanabe, M.; Matsuura, H.; Kajiyama, G. Renal response to L-arginine in salt-sensitive patients with essential hypertension. Hypertension 1996, 27 Pt 2, 643–648. [Google Scholar] [CrossRef]
- Campese, V.M.; Amar, M.; Anjali, C.; Medhat, T.; Wurgaft, A. Effect of L-arginine on systemic and renal haemodynamics in salt-sensitive patients with essential hypertension. J. Hum. Hypertens 1997, 11, 527–532. [Google Scholar] [CrossRef]
- Pezza, V.; Bernardini, F.; Pezza, E.; Pezza, B.; Curione, M. Study of supplemental oral l-arginine in hypertensives treated with enalapril + hydrochlorothiazide. Am. J. Hypertens 1998, 11, 1267–1270. [Google Scholar]
- Higashi, Y.; Oshima, T.; Ono, N.; Hiraga, H.; Yoshimura, M.; Watanabe, M.; Matsuura, H.; Kambe, M.; Kajiyama, G. Intravenous administration of L-arginine inhibits angiotensin-converting enzyme in humans. J. Clin. Endocrinol. Metab. 1995, 80, 2198–2202. [Google Scholar]
- De Caterina, R.; Libby, P.; Peng, H.B.; Thannickal, V.J.; Rajavashisth, T.B.; Gimbrone, M.A., Jr.; Shin, W.S.; Liao, J.K. Nitric oxide decreases cytokine-induced endothelial activation. Nitric oxide selectively reduces endothelial expression of adhesion molecules and proinflammatory cytokines. J. Clin. Investig. 1995, 96, 60–68. [Google Scholar] [CrossRef]
- Sperone, A.; Dryden, N.H.; Birdsey, G.M.; Madden, L.; Johns, M.; Evans, P.C.; Mason, J.C.; Haskard, D.O.; Boyle, J.J.; Paleolog, E.M.; et al. The transcription factor Erg inhibits vascular inflammation by repressing NF-kappaB activation and proinflammatory gene expression in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 142–150. [Google Scholar] [CrossRef]
- Gambardella, J.; Santulli, G. Integrating diet and inflammation to calculate cardiovascular risk. Atherosclerosis 2016, 253, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Ley, K. Adhesion molecules and atherogenesis. Acta Physiol. Scand. 2001, 173, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Blum, A.; Miller, H.; Blum, A.; Miller, H. The effects of L-arginine on atherosclerosis and heart disease. Int. J. Cardiovasc. Intervent. 1999, 2, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.R.; Jessup, W.; Hailstones, D.; Celermajer, D.S. L-arginine reduces human monocyte adhesion to vascular endothelium and endothelial expression of cell adhesion molecules. Circulation 1997, 95, 662–668. [Google Scholar] [CrossRef]
- Adams, M.R.; McCredie, R.; Jessup, W.; Robinson, J.; Sullivan, D.; Celermajer, D.S. Oral L-arginine improves endothelium-dependent dilatation and reduces monocyte adhesion to endothelial cells in young men with coronary artery disease. Atherosclerosis 1997, 129, 261–269. [Google Scholar] [CrossRef]
- Ceremuzynski, L.; Chamiec, T.; Herbaczynska-Cedro, K. Effect of supplemental oral L-arginine on exercise capacity in patients with stable angina pectoris. Am. J. Cardiol. 1997, 80, 331–333. [Google Scholar] [CrossRef]
- Lerman, A.; Burnett, J.C., Jr.; Higano, S.T.; McKinley, L.J.; Holmes, D.R., Jr. Long-term L-arginine supplementation improves small-vessel coronary endothelial function in humans. Circulation 1998, 97, 2123–2128. [Google Scholar] [CrossRef]
- Chen, C.; Mattar, S.G.; Lumsden, A.B. Oral administration of L-arginine reduces intimal hyperplasia in balloon-injured rat carotid arteries. J. Surg. Res. 1999, 82, 17–23. [Google Scholar] [CrossRef]
- Yin, W.H.; Chen, J.W.; Tsai, C.; Chiang, M.C.; Young, M.S.; Lin, S.J. L-arginine improves endothelial function and reduces LDL oxidation in patients with stable coronary artery disease. Clin. Nutr. 2005, 24, 988–997. [Google Scholar] [CrossRef]
- Walker, H.A.; McGing, E.; Fisher, I.; Boger, R.H.; Bode-Boger, S.M.; Jackson, G.; Ritter, J.M.; Chowienczyk, P.J. Endothelium-dependent vasodilation is independent of the plasma L-arginine/ADMA ratio in men with stable angina: Lack of effect of oral L-arginine on endothelial function, oxidative stress and exercise performance. J. Am. Coll. Cardiol. 2001, 38, 499–505. [Google Scholar] [CrossRef]
- Padilla, F.; Garcia-Dorado, D.; Agullo, L.; Inserte, J.; Paniagua, A.; Mirabet, S.; Barrabes, J.A.; Ruiz-Meana, M.; Soler-Soler, J. L-Arginine administration prevents reperfusion-induced cardiomyocyte hypercontracture and reduces infarct size in the pig. Cardiovasc. Res. 2000, 46, 412–420. [Google Scholar] [CrossRef][Green Version]
- Rector, T.S.; Bank, A.J.; Mullen, K.A.; Tschumperlin, L.K.; Sih, R.; Pillai, K.; Kubo, S.H. Randomized, double-blind, placebo-controlled study of supplemental oral L-arginine in patients with heart failure. Circulation 1996, 93, 2135–2141. [Google Scholar] [CrossRef] [PubMed]
- Bednarz, B.; Jaxa-Chamiec, T.; Gebalska, J.; Herbaczynska-Cedro, K.; Ceremuzynski, L. L-arginine supplementation prolongs exercise capacity in congestive heart failure. Kardiol. Pol. 2004, 60, 348–353. [Google Scholar] [PubMed]
- Rodrigues-Krause, J.; Krause, M.; Rocha, I.; Umpierre, D.; Fayh, A.P.T. Association of l-Arginine Supplementation with Markers of Endothelial Function in Patients with Cardiovascular or Metabolic Disorders: A Systematic Review and Meta-Analysis. Nutrients 2018, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Hambrecht, R.; Hilbrich, L.; Erbs, S.; Gielen, S.; Fiehn, E.; Schoene, N.; Schuler, G. Correction of endothelial dysfunction in chronic heart failure: Additional effects of exercise training and oral L-arginine supplementation. J. Am. Coll. Cardiol. 2000, 35, 706–713. [Google Scholar] [CrossRef]
- Weyrich, A.S.; Ma, X.L.; Lefer, A.M. The role of L-arginine in ameliorating reperfusion injury after myocardial ischemia in the cat. Circulation 1992, 86, 279–288. [Google Scholar] [CrossRef]
- Huk, I.; Nanobashvili, J.; Orljanski, W.; Neumayer, C.; Punz, A.; Holzaepfel, A.; Fuegl, A.; Mittlboeck, M.; Polterauer, P.; Roth, E. L-arginine treatment in ischemia/reperfusion injury. Cas. Lek. Cesk. 1998, 137, 496–499. [Google Scholar]
- Takeuchi, K.; McGowan, F.X.; Danh, H.C.; Glynn, P.; Simplaceanu, E.; del Nido, P.J. Direct detrimental effects of L-arginine upon ischemia--reperfusion injury to myocardium. J. Mol. Cell Cardiol. 1995, 27, 1405–1414. [Google Scholar] [CrossRef]
- Kubo, S.H.; Rector, T.S.; Bank, A.J.; Williams, R.E.; Heifetz, S.M. Endothelium-dependent vasodilation is attenuated in patients with heart failure. Circulation 1991, 84, 1589–1596. [Google Scholar] [CrossRef]
- Hirooka, Y.; Imaizumi, T.; Tagawa, T.; Shiramoto, M.; Endo, T.; Ando, S.; Takeshita, A. Effects of L-arginine on impaired acetylcholine-induced and ischemic vasodilation of the forearm in patients with heart failure. Circulation 1994, 90, 658–668. [Google Scholar] [CrossRef]
- Doutreleau, S.; Mettauer, B.; Piquard, F.; Rouyer, O.; Schaefer, A.; Lonsdorfer, J.; Geny, B. Chronic L-arginine supplementation enhances endurance exercise tolerance in heart failure patients. Int. J. Sports Med. 2006, 27, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Parnell, M.M.; Holst, D.P.; Kaye, D.M. Augmentation of endothelial function following exercise training is associated with increased L-arginine transport in human heart failure. Clin. Sci. 2005, 109, 523–530. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chin-Dusting, J.P.; Kaye, D.M.; Lefkovits, J.; Wong, J.; Bergin, P.; Jennings, G.L. Dietary supplementation with L-arginine fails to restore endothelial function in forearm resistance arteries of patients with severe heart failure. J. Am. Coll. Cardiol. 1996, 27, 1207–1213. [Google Scholar] [CrossRef]
- Schulman, S.P.; Becker, L.C.; Kass, D.A.; Champion, H.C.; Terrin, M.L.; Forman, S.; Ernst, K.V.; Kelemen, M.D.; Townsend, S.N.; Capriotti, A.; et al. L-arginine therapy in acute myocardial infarction: The Vascular Interaction With Age in Myocardial Infarction (VINTAGE MI) randomized clinical trial. JAMA 2006, 295, 58–64. [Google Scholar] [CrossRef]
- Shu, J.; Santulli, G. Update on peripheral artery disease: Epidemiology and evidence-based facts. Atherosclerosis 2018, 275, 379–381. [Google Scholar] [CrossRef]
- Azab, S.M.; Zamzam, A.; Syed, M.H.; Abdin, R.; Qadura, M.; Britz-McKibbin, P. Serum Metabolic Signatures of Chronic Limb-Threatening Ischemia in Patients with Peripheral Artery Disease. J. Clin. Med. 2020, 9, 1877. [Google Scholar] [CrossRef]
- Misra, S.; Shishehbor, M.H.; Takahashi, E.A.; Aronow, H.D.; Brewster, L.P.; Bunte, M.C.; Kim, E.S.H.; Lindner, J.R.; Rich, K.; American Heart Association Council on Peripheral Vascular Disease; et al. Perfusion Assessment in Critical Limb Ischemia: Principles for Understanding and the Development of Evidence and Evaluation of Devices: A Scientific Statement From the American Heart Association. Circulation 2019, 140, e657–e672. [Google Scholar] [CrossRef]
- Boger, R.H.; Bode-Boger, S.M.; Thiele, W.; Junker, W.; Alexander, K.; Frolich, J.C. Biochemical evidence for impaired nitric oxide synthesis in patients with peripheral arterial occlusive disease. Circulation 1997, 95, 2068–2074. [Google Scholar] [CrossRef]
- Maxwell, A.J.; Cooke, J.P. Cardiovascular effects of L-arginine. Curr. Opin. Nephrol. Hypertens. 1998, 7, 63–70. [Google Scholar] [CrossRef]
- Maxwell, A.J.; Anderson, B.E.; Cooke, J.P. Nutritional therapy for peripheral arterial disease: A double-blind, placebo-controlled, randomized trial of HeartBar. Vasc. Med. 2000, 5, 11–19. [Google Scholar]
- Wilson, A.M.; Harada, R.; Nair, N.; Balasubramanian, N.; Cooke, J.P. L-arginine supplementation in peripheral arterial disease: No benefit and possible harm. Circulation 2007, 116, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Avogaro, A.; Albiero, M.; Menegazzo, L.; de Kreutzenberg, S.; Fadini, G.P. Endothelial dysfunction in diabetes: The role of reparatory mechanisms. Diabetes Care 2011, 34 (Suppl. 2), S285–S290. [Google Scholar] [CrossRef] [PubMed]
- Sena, C.M.; Pereira, A.M.; Seica, R. Endothelial dysfunction—A major mediator of diabetic vascular disease. Biochim. Biophys. Acta 2013, 1832, 2216–2231. [Google Scholar] [CrossRef] [PubMed]
- Bogdanski, P.; Suliburska, J.; Grabanska, K.; Musialik, K.; Cieslewicz, A.; Skoluda, A.; Jablecka, A. Effect of 3-month L-arginine supplementation on insulin resistance and tumor necrosis factor activity in patients with visceral obesity. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 816–823. [Google Scholar]
- Eid, H.M.; Reims, H.; Arnesen, H.; Kjeldsen, S.E.; Lyberg, T.; Seljeflot, I. Decreased levels of asymmetric dimethylarginine during acute hyperinsulinemia. Metabolism 2007, 56, 464–469. [Google Scholar] [CrossRef]
- Sobrevia, L.; Nadal, A.; Yudilevich, D.L.; Mann, G.E. Activation of L-arginine transport (system y+) and nitric oxide synthase by elevated glucose and insulin in human endothelial cells. J. Physiol. 1996, 490 Pt 3, 775–781. [Google Scholar] [CrossRef]
- Mann, G.E.; Yudilevich, D.L.; Sobrevia, L. Regulation of amino acid and glucose transporters in endothelial and smooth muscle cells. Physiol. Rev. 2003, 83, 183–252. [Google Scholar] [CrossRef]
- Lembo, G.; Iaccarino, G.; Vecchione, C.; Barbato, E.; Morisco, C.; Monti, F.; Parrella, L.; Trimarco, B. Insulin enhances endothelial alpha2-adrenergic vasorelaxation by a pertussis toxin mechanism. Hypertension 1997, 30, 1128–1134. [Google Scholar] [CrossRef]
- Zeng, G.; Nystrom, F.H.; Ravichandran, L.V.; Cong, L.N.; Kirby, M.; Mostowski, H.; Quon, M.J. Roles for insulin receptor, PI3-kinase, and Akt in insulin-signaling pathways related to production of nitric oxide in human vascular endothelial cells. Circulation 2000, 101, 1539–1545. [Google Scholar] [CrossRef]
- Vecchione, C.; Aretini, A.; Maffei, A.; Marino, G.; Selvetella, G.; Poulet, R.; Trimarco, V.; Frati, G.; Lembo, G. Cooperation between insulin and leptin in the modulation of vascular tone. Hypertension 2003, 42, 166–170. [Google Scholar] [CrossRef]
- Scherrer, U.; Randin, D.; Vollenweider, P.; Vollenweider, L.; Nicod, P. Nitric oxide release accounts for insulin’s vascular effects in humans. J. Clin. Investig. 1994, 94, 2511–2515. [Google Scholar] [CrossRef] [PubMed]
- Iaccarino, G.; Ciccarelli, M.; Sorriento, D.; Cipolletta, E.; Cerullo, V.; Iovino, G.L.; Paudice, A.; Elia, A.; Santulli, G.; Campanile, A.; et al. AKT participates in endothelial dysfunction in hypertension. Circulation 2004, 109, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Muniyappa, R.; Montagnani, M.; Koh, K.K.; Quon, M.J. Cardiovascular actions of insulin. Endocr. Rev. 2007, 28, 463–491. [Google Scholar] [CrossRef] [PubMed]
- Pieper, G.M.; Siebeneich, W.; Dondlinger, L.A. Short-term oral administration of L-arginine reverses defective endothelium-dependent relaxation and cGMP generation in diabetes. Eur. J. Pharmacol. 1996, 317, 317–320. [Google Scholar] [CrossRef]
- Khaidar, A.; Marx, M.; Lubec, B.; Lubec, G. L-arginine reduces heart collagen accumulation in the diabetic db/db mouse. Circulation 1994, 90, 479–483. [Google Scholar] [CrossRef]
- Lubec, G.; Bartosch, B.; Mallinger, R.; Adamiker, D.; Graef, I.; Frisch, H.; Hoger, H. The effect of substance L on glucose-mediated cross-links of collagen in the diabetic db/db mouse. Nephron 1990, 56, 281–284. [Google Scholar] [CrossRef]
- Lubec, B.; Hayn, M.; Kitzmuller, E.; Vierhapper, H.; Lubec, G. L-Arginine reduces lipid peroxidation in patients with diabetes mellitus. Free Radic. Biol. Med. 1997, 22, 355–357. [Google Scholar] [CrossRef]
- Pieper, G.M. Review of alterations in endothelial nitric oxide production in diabetes: Protective role of arginine on endothelial dysfunction. Hypertension 1998, 31, 1047–1060. [Google Scholar] [CrossRef]
- De Pirro, R.; Tamburrano, G.; Fusco, A.; Lauro, R. Arginine does not influence insulin binding on circulating monocytes. Endokrinologie 1980, 75, 243–246. [Google Scholar]
- Wascher, T.C.; Graier, W.F.; Dittrich, P.; Hussain, M.A.; Bahadori, B.; Wallner, S.; Toplak, H. Effects of low-dose L-arginine on insulin-mediated vasodilatation and insulin sensitivity. Eur. J. Clin. Investig. 1997, 27, 690–695. [Google Scholar] [CrossRef]
- Piatti, P.M.; Monti, L.D.; Valsecchi, G.; Magni, F.; Setola, E.; Marchesi, F.; Galli-Kienle, M.; Pozza, G.; Alberti, K.G. Long-term oral L-arginine administration improves peripheral and hepatic insulin sensitivity in type 2 diabetic patients. Diabetes Care 2001, 24, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N.T.; Tayek, J.A. Oral arginine reduces systemic blood pressure in type 2 diabetes: Its potential role in nitric oxide generation. J. Am. Coll. Nutr. 2002, 21, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.L.; Aoyama, S.; Nagai, R.; Miyoshi, N.; Ohshima, H. Inhibition of L-arginine metabolizing enzymes by L-arginine-derived advanced glycation end products. J. Clin. Biochem. Nutr. 2010, 46, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Fiory, F.; Lombardi, A.; Miele, C.; Giudicelli, J.; Beguinot, F.; Van Obberghen, E. Methylglyoxal impairs insulin signalling and insulin action on glucose-induced insulin secretion in the pancreatic beta cell line INS-1E. Diabetologia 2011, 54, 2941–2952. [Google Scholar] [CrossRef]
- Polykretis, P.; Luchinat, E.; Boscaro, F.; Banci, L. Methylglyoxal interaction with superoxide dismutase 1. Redox Biol. 2020, 30, 101421. [Google Scholar] [CrossRef]
- Martina, V.; Masha, A.; Gigliardi, V.R.; Brocato, L.; Manzato, E.; Berchio, A.; Massarenti, P.; Settanni, F.; Della Casa, L.; Bergamini, S.; et al. Long-term N-acetylcysteine and L-arginine administration reduces endothelial activation and systolic blood pressure in hypertensive patients with type 2 diabetes. Diabetes Care 2008, 31, 940–944. [Google Scholar] [CrossRef]
- Deveaux, A.; Fouillet, H.; Petzke, K.J.; Hermier, D.; Andre, E.; Bunouf, P.; Lantoine-Adam, F.; Benamouzig, R.; Mathe, V.; Huneau, J.F.; et al. A Slow-Compared with a Fast-Release Form of Oral Arginine Increases Its Utilization for Nitric Oxide Synthesis in Overweight Adults with Cardiometabolic Risk Factors in a Randomized Controlled Study. J. Nutr. 2016, 146, 1322–1329. [Google Scholar] [CrossRef][Green Version]
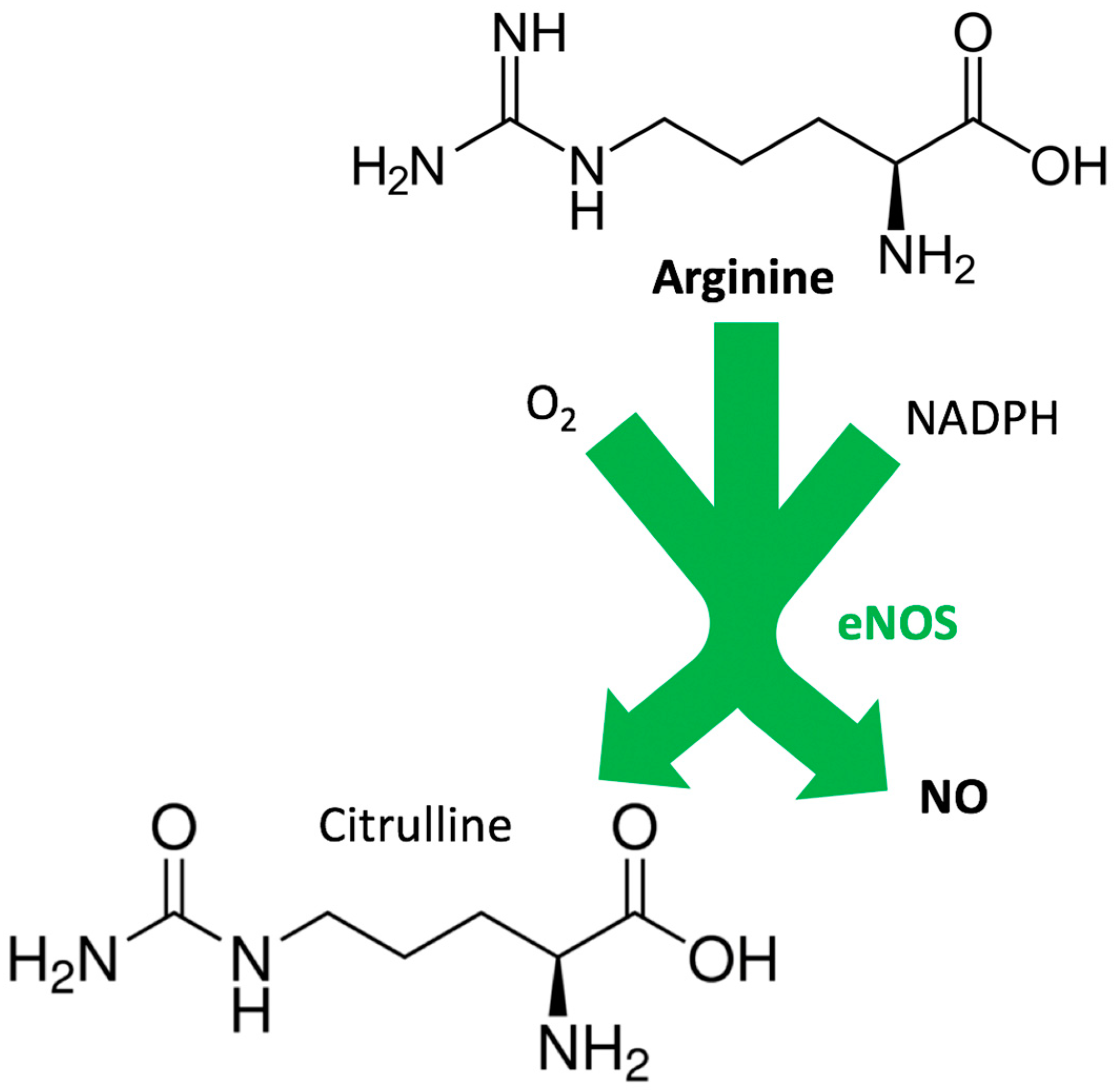

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gambardella, J.; Khondkar, W.; Morelli, M.B.; Wang, X.; Santulli, G.; Trimarco, V. Arginine and Endothelial Function. Biomedicines 2020, 8, 277. https://doi.org/10.3390/biomedicines8080277
Gambardella J, Khondkar W, Morelli MB, Wang X, Santulli G, Trimarco V. Arginine and Endothelial Function. Biomedicines. 2020; 8(8):277. https://doi.org/10.3390/biomedicines8080277
Chicago/Turabian StyleGambardella, Jessica, Wafiq Khondkar, Marco Bruno Morelli, Xujun Wang, Gaetano Santulli, and Valentina Trimarco. 2020. "Arginine and Endothelial Function" Biomedicines 8, no. 8: 277. https://doi.org/10.3390/biomedicines8080277
APA StyleGambardella, J., Khondkar, W., Morelli, M. B., Wang, X., Santulli, G., & Trimarco, V. (2020). Arginine and Endothelial Function. Biomedicines, 8(8), 277. https://doi.org/10.3390/biomedicines8080277