A Two-Dimensional Affinity Capture and Separation Mini-Platform for the Isolation, Enrichment, and Quantification of Biomarkers and Its Potential Use for Liquid Biopsy
Abstract
:1. Introduction
2. Definitions
3. Brief History of Biomarker-Assay Technologies
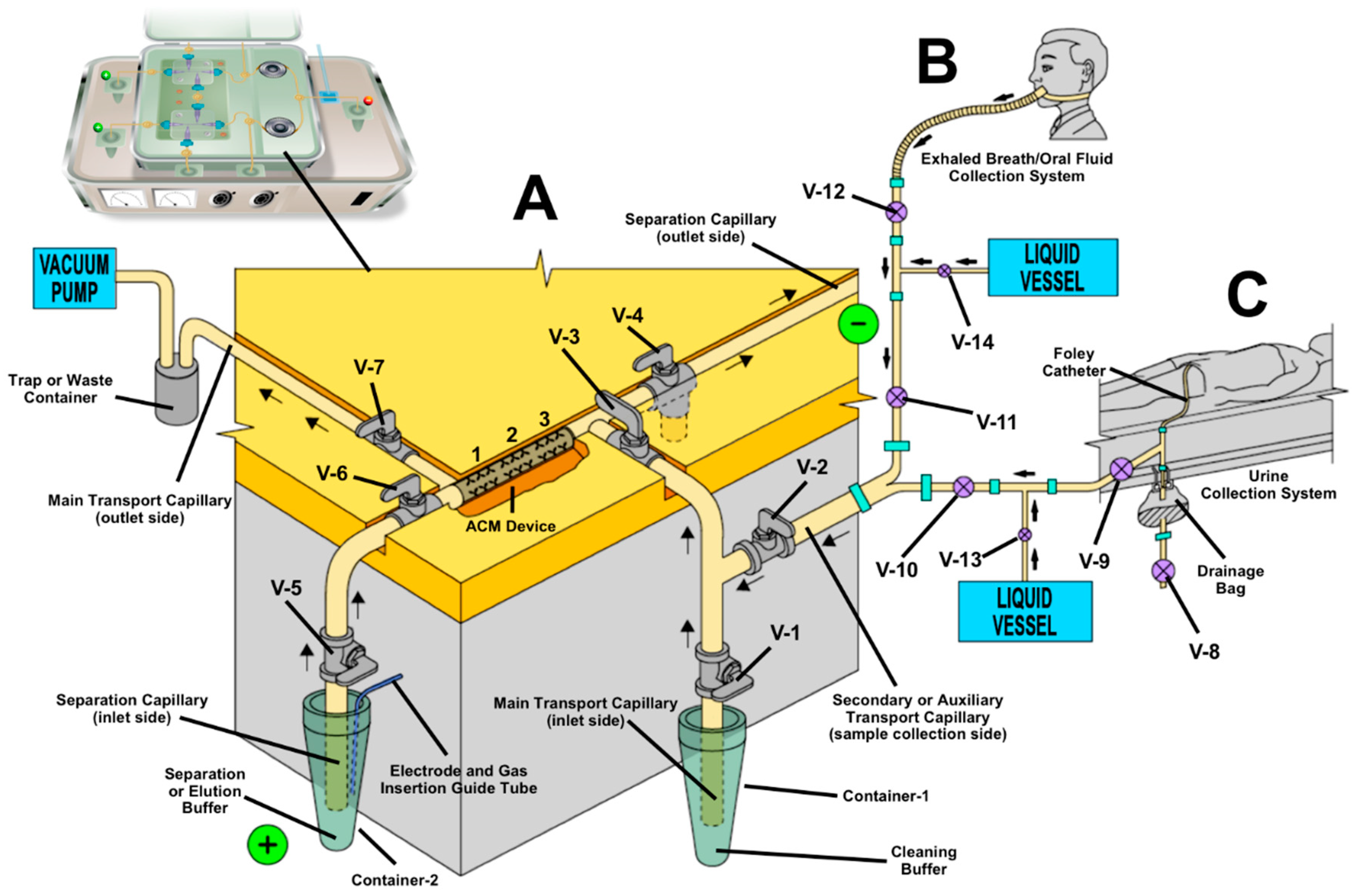
4. Miniaturized Biomarker Analyzers
5. Role of Capillary Electrophoresis in Point-of-Care Diagnostics
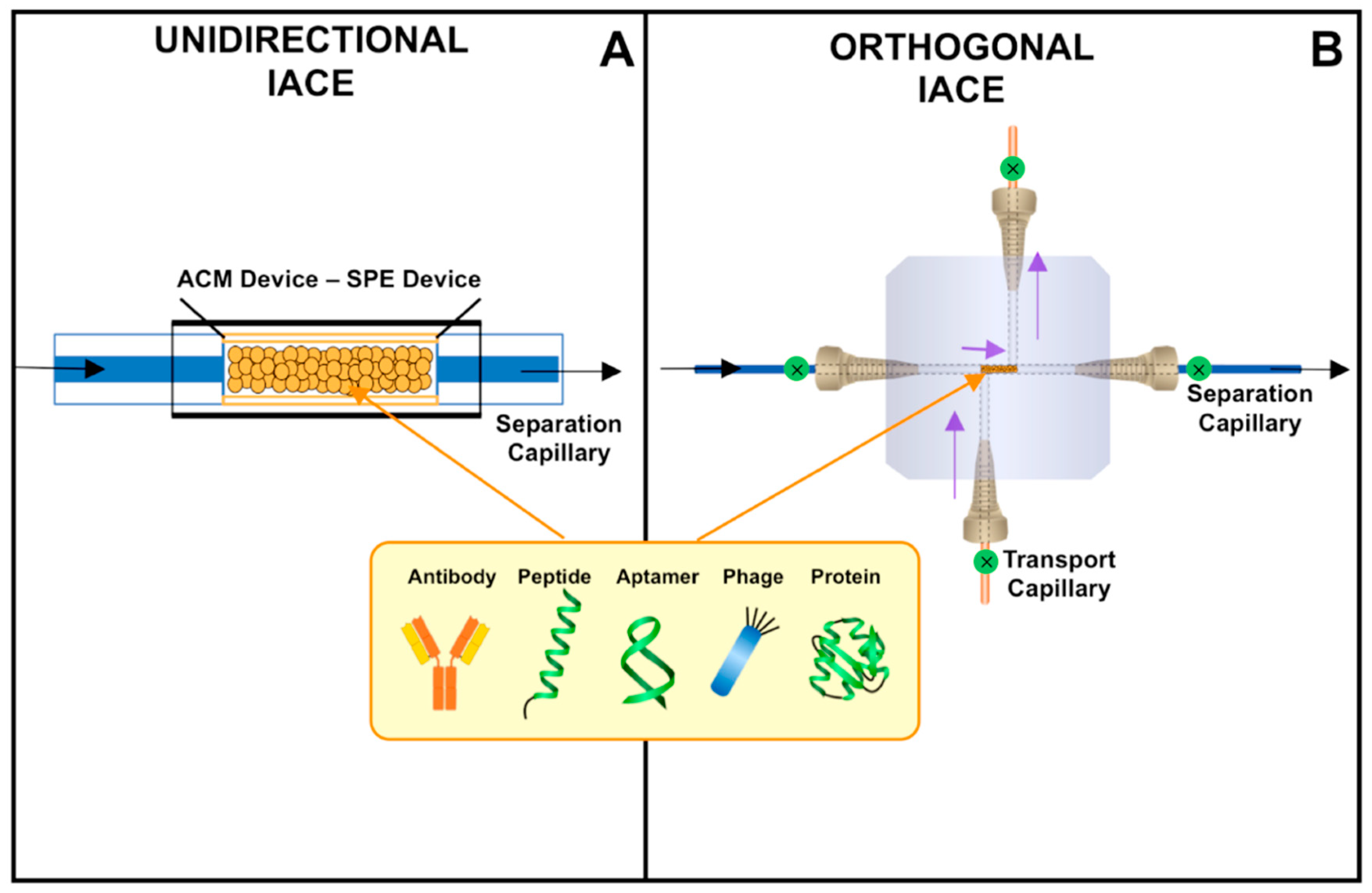
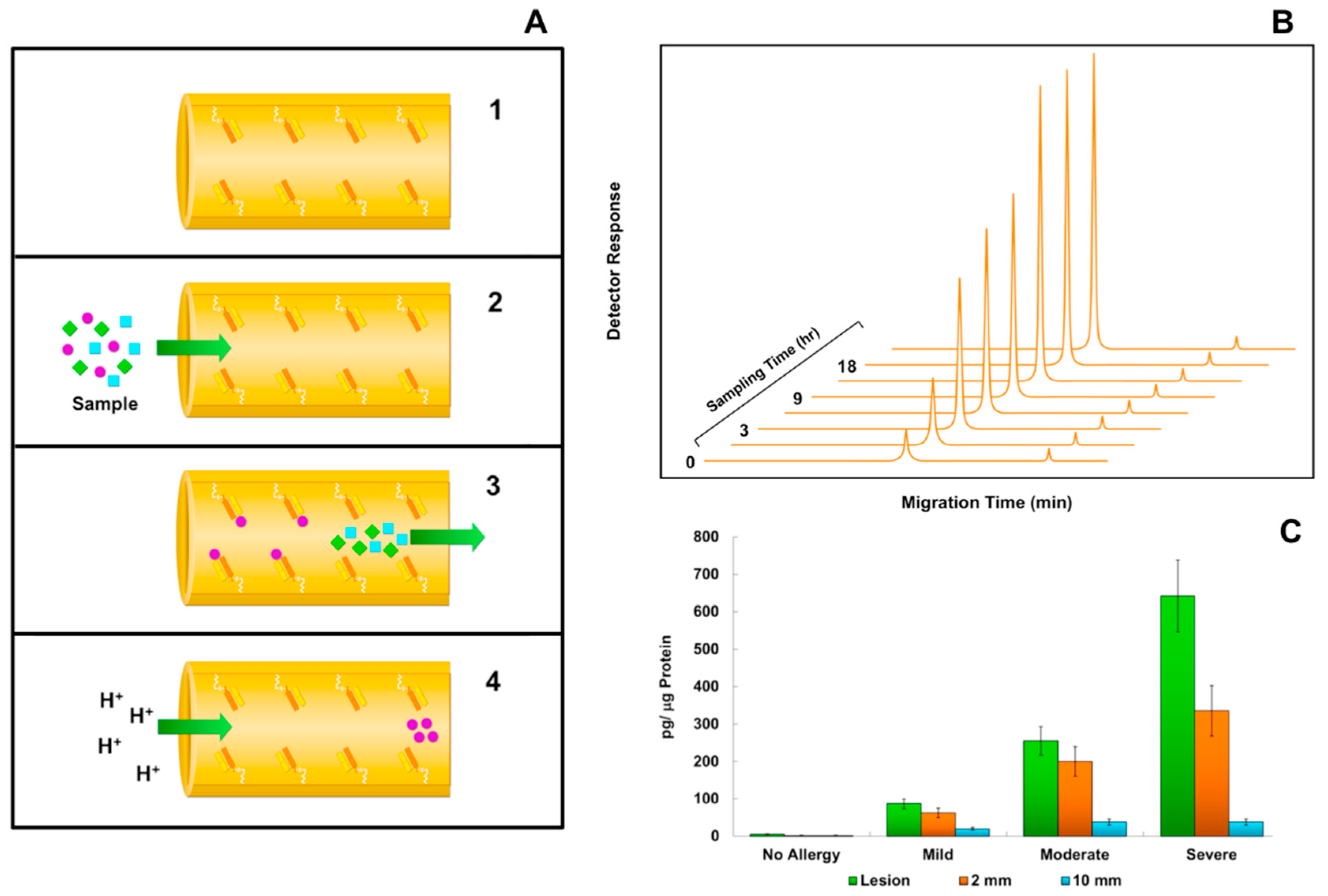
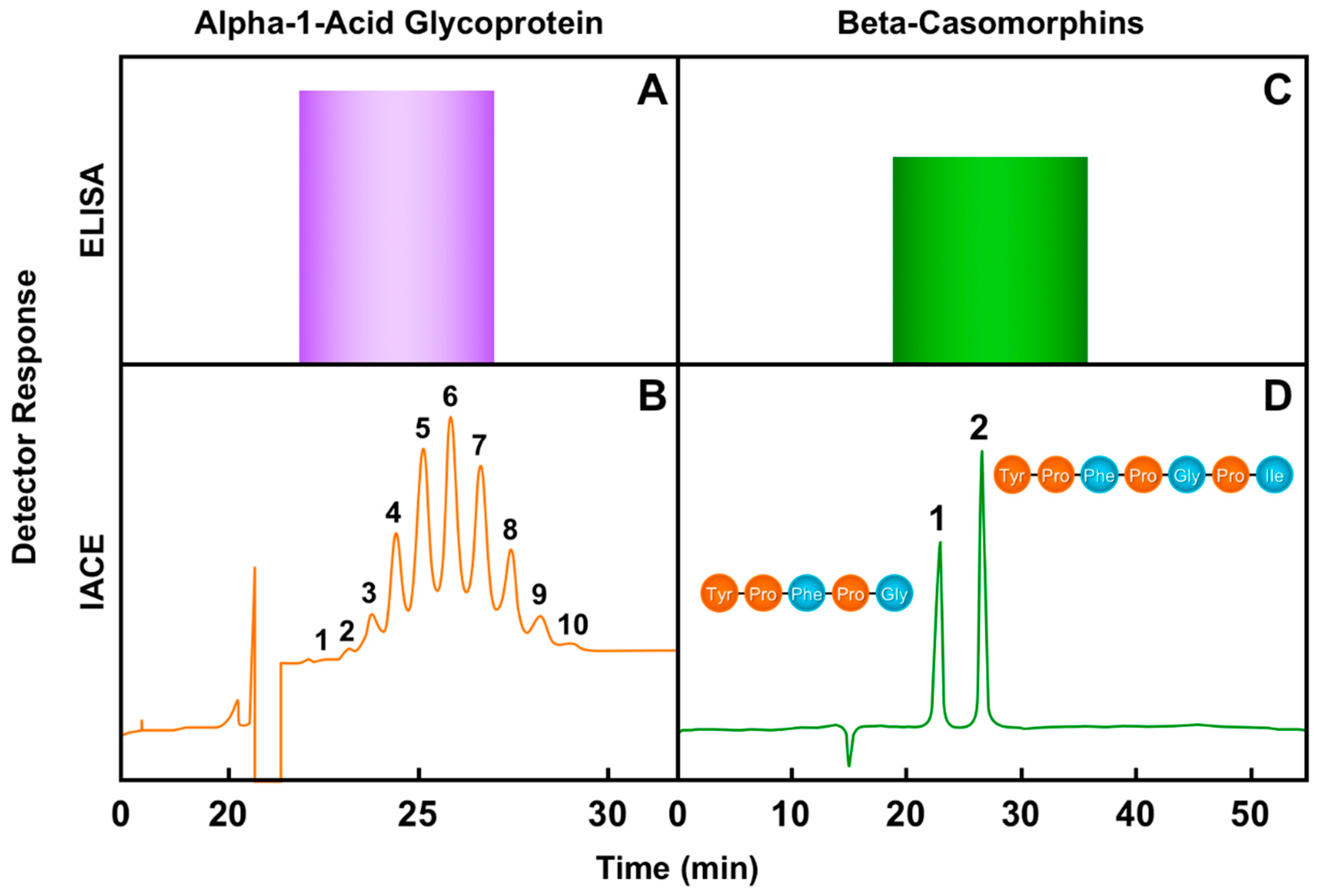
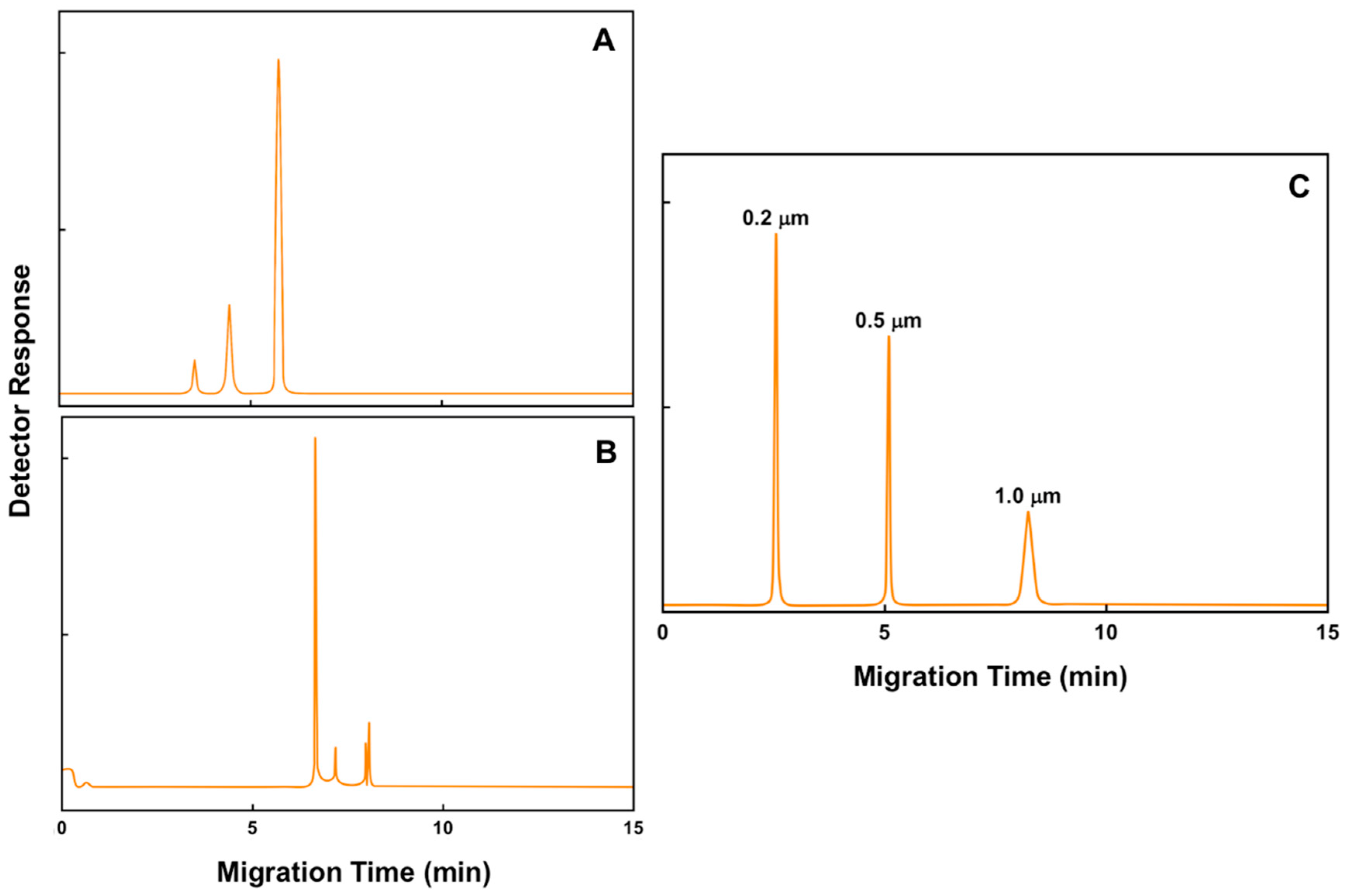
6. Immunoaffinity Capillary Electrophoresis Applications
7. The Potential Use of IACE for Extracellular Vesicle Studies
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FDA-NIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and Other Tools) Resource. Maryland: Food and Drug Administration-National Institutes of Health Biomarker Working Group 2016. Available online: https://www.ncbi.nlm.nih.gov/pubmed/27010052 (accessed on 25 September 2017).
- Mayeux, R. Biomarkers: Potential uses and limitations. NeuroRx 2004, 1, 182–188. [Google Scholar] [CrossRef]
- Diamantis, E.P. How are we going to discover new cancer biomarkers? A proteomic approach for bladder cancer. Clin. Chem. 2004, 50, 793–795. [Google Scholar] [CrossRef] [Green Version]
- Selleck, M.J.; Maheswari, S.; Wall, N.R. Making meaningful clinical use of biomarkers. Biomark. Insights 2017, 12, 1177271917715236. [Google Scholar] [CrossRef] [Green Version]
- Vandamme, D.; Fitzmaurice, W.; Kholodenko, B.; Kolch, W. Systems medicine: Helping us understand the complexity of disease. Q. J. Med. 2013, 106, 891–895. [Google Scholar] [CrossRef] [Green Version]
- Saah, A.J.; Hoover, D.R. “Sensitivity” and “specificity” reconsidered: The meaning of these terms in analytical and diagnostic settings. Ann. Intern. Med. 1997, 126, 91–94. [Google Scholar] [CrossRef]
- Rao, T.N. Validation of analytical methods. In Calibration and Validation of Analytical Methods—A Sampling of Current Approaches, Chapter 7; IntechOpen:-Chemistry, IntechOpen Limited: London, UK, 2018. [Google Scholar] [CrossRef] [Green Version]
- Zerbino, D.D. Biopsy: Its history, current and future outlook. Lik. Sprava 1994, 3–4, 1–9. [Google Scholar]
- Mathai, R.A.; Vidya, R.V.S.; Reddy, B.S.; Thomas, L.; Udupa, K.; Kolesar, J.; Rao, M. Potential utility of liquid biopsy as a diagnostic and prognostic tool for the assessment of solid tumors: Implications in the precision oncology. J. Clin. Med. 2019, 8, 73. [Google Scholar] [CrossRef] [Green Version]
- Soda, N.; Rehm, B.H.A.; Sonar, P.; Nguyen, N.-T.; Shiddiky, M.J.A. Advances liquid biopsy for circulating biomarker detection. J. Mater. Chem. B 2019, 7, 6670. [Google Scholar] [CrossRef] [PubMed]
- Aro, K.; Wei, F.; Wong, D.T.; Tu, M. Saliva liquid biopsy for point-of-care applications. Front. Public Health 2017, 5, 77. [Google Scholar] [CrossRef] [Green Version]
- Morgan, T.M. Liquid biopsy: Where did it come from, what is it, and where is it going? Investig. Clin. Urol. 2019, 60, 139–141. [Google Scholar] [CrossRef]
- Cheng, A.P.; Burnham, P.; Lee, J.R.; Cheng, M.P.; Suthanthiran, M.; Dadhania, D.; De Vlaminck, I. A cell-free DNA metagenomic sequencing assay that integrates the host injury response to infection. Proc. Natl. Acad. Sci. USA 2019, 116, 18738–18744. [Google Scholar] [CrossRef] [Green Version]
- Ihling, C.; Tänzler, D.; Hagemann, S.; Kehlen, A.; Hüttelmaier, S.; Arlt, C.; Sinz, A. Mass spectrometry identification of SARS-Cov-2 proteins from gargle solution samples of COVID-19 patients. J. Proteome Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gosling, J.P. Immunoassays: A Practical Approach; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Yallow, R.S.; Berson, S.A. Immunoassay of endogenous plasma insulin in man. J. Clin. Investig. 1960, 39, 1157–1175. [Google Scholar] [CrossRef] [PubMed]
- Engvall, E.; Perlmann, P. Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 1971, 8, 871–874. [Google Scholar] [CrossRef]
- Kohler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of pre-defined specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Bonwick, G.A.; Smith, C.J. Immunoassays: Their history, development and current place in food science and technology. Int. J. Food Sci. 2004, 39, 817–827. [Google Scholar] [CrossRef]
- Wu, A.H.B. A selected history and future of immunoassay development and applications in clinical chemistry. Clin. Chim. Acta 2006, 369, 119–124. [Google Scholar] [CrossRef]
- Tian, W.; Wang, L.; Lei, H.; Sun, Y.; Xiao, Z. Antibody production and application for immunoassay development of environmental hormones: A review. Chem. Biol. Technol. Agric. 2018, 5, 5. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Zhang, M. Development of immunoassays for the determination of phthalates. Food Agric. Immunol. 2020, 31, 303–316. [Google Scholar] [CrossRef]
- Sajid, M.; Kawde, A.-N.; Daud, M. Designs, formats and applications of lateral flow assay: A literature review. J. Saudi Chem. Soc. 2015, 19, 689–705. [Google Scholar] [CrossRef] [Green Version]
- Cui, X.; Jin, M.; Du, P.; Chen, G.; Zhang, C.; Zhang, Y.; Shao, Y.; Wang, J. Development of immunoassays for multi-residue detection of small molecule compounds. Food Agric. Immunol. 2018, 29, 638–652. [Google Scholar] [CrossRef] [Green Version]
- Guzman, N.A.; Guzman, D.E. An emerging micro-scale immune-analytical diagnostic tool to see the unseen. Holding promise for precision medicine and P4 medicine. J. Chromatogr. B 2016, 1021, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Pinho, A.R.; Fortuna, A.; Falcão, A.; Santos, A.C.; Seiça, R.; Estevens, C.; Veiga, F.; Ribeiro, A.J. Comparison of ELISA and HPLC-MS methods for the determination of exenatide in biological and biotechnology-based formulation matrices. J. Pharm. Anal. 2019, 9, 143–155. [Google Scholar] [CrossRef]
- Bowerbank, S.L.; Carlin, M.G.; Dean, J.R. A direct comparison of liquid chromatography-mass spectrometry with clinical routine testing immunoassay methods for the detection and quantification of thyroid hormones in blood serum. Anal. Bioanal. Chem. 2019, 411, 2839–2853. [Google Scholar] [CrossRef] [Green Version]
- Le Goff, C.; Farre-Segura, J.; Stojkovic, V.; Dufour, P.; Peeters, S.; Courtois, J.; Nizet, A.; De Vos, N.; Cavalier, E. The pathway through LC-MS method development: In-house or ready-to-use kit-based methods? Clin. Chem. Lab. Med. 2020. [Google Scholar] [CrossRef] [Green Version]
- Kočová-Vlčková, H.; Pilařová, V.; Svobodová, P.; Plíšek, J.; Švec, F.; Nováková, L. Current state of bioanalytical chromatography in clinical analysis. Analyst 2018, 143, 1305–1325. [Google Scholar] [CrossRef]
- Kormaz, K.S.; Butuner, B.D.; Roggenbuck, D. Detection of 8-OHdG as a diagnostic biomarker. J. Lab. Precis. Med. 2018, 3, 95. [Google Scholar] [CrossRef]
- Beyene, A.M.; Du, X.; Schrunk, D.E.; Ensley, S.; Rumbeiha, W.K. High-performance liquid chromatography and enzyme-linked immunosorbent assay techniques for detection and quantification of aflatoxin B1 in feed samples: A comparative study. BMC Res. Notes 2019, 12, 492. [Google Scholar] [CrossRef]
- Broassaud, J.; Leban, M.; Corcuff, J.B.; Boux de Casson, F.; Leloupp, A.G.; Masson, D.; Moal, V.; Bach-Ngohou, K. LC-MSMS assays of urinary cortisol, a comparison between four in-house assays. Clin. Chem. Lab. Med. 2018, 56, 1109–1116. [Google Scholar] [CrossRef]
- Chen, K.-M.; Calcagnotto, A.; Zhu, J.; Sun, Y.-W.; El-Bayoumy, K.; Richie, J.P., Jr. Comparison of an HPLC-MS/MS method with multiple commercial ELISA kits on the determination of levels of 8-oxo-7,8-dihydro-2′-deoxyguanosine in human urine. J. New Dev. Chem. 2018, 2, 1–13. [Google Scholar] [CrossRef]
- Neubert, H.; Shuford, C.M.; Olah, T.V.; Garofolo, F.; Schultz, G.A.; Jones, B.R.; Amaravadi, L.; Laterza, O.F.; Xu, K.; Ackermann, B.L. Protein biomarker quantification by immunoaffinity liquid chromatography-tandem mass spectrometry: Current state and future vision. Clin. Chem. 2020, 66, 282–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berger, D. A brief history of medical diagnosis and the birth of the clinical laboratory. Med. Lab. Obs. 1999, 31, 28–30. [Google Scholar]
- Moore, R.E. An historical perspective on the clinical diagnostic laboratory. In Molecular Diagnostics: For the Clinical Laboratories, 2nd ed.; Coleman, W.B., Tsongalis, G.J., Eds.; Humana Press: Totowa, NJ, USA, 2006; pp. 3–10. [Google Scholar]
- St. John, A.; Price, C.P. Existing and emerging technologies for point-of-care testing. Clin. Biochem. Rev. 2014, 35, 155–167. [Google Scholar] [PubMed]
- Rebel, A.; Rice, M.A.; Fahy, B.G. The accuracy of point-of-care glucose measurements. J. Diabetes Sci. Technol. 2012, 6, 396–411. [Google Scholar] [CrossRef] [Green Version]
- Havele, S.A.; Pfoh, E.R.; Yan, C.; Misra-Hebert, A.D.; Le, P.; Rothberg, M.B. Physicians’views of self-monitoring of blood glucose in patients with Type 2 diabetes not on insulin. Ann. Fam. Med. 2018, 16, 349–352. [Google Scholar] [CrossRef]
- Sharma, S.; Zapatero-Rodríguez, J.; Estrela, P.; O’Kennedy, R. Point-of-care diagnostics in low resource settings: Present status and future role of microfluidics. Biosensors 2015, 5, 577–601. [Google Scholar] [CrossRef] [Green Version]
- Abel, G. Current status and future prospects of point-of-care testing around the globe. Expert Rev. Mol. Diagnost. 2015, 15, 853–855. [Google Scholar] [CrossRef] [Green Version]
- Jordan, B.; Mitchell, C.; Anderson, A.; Farkas, N.; Batrla, R. The clinical and health economic value of clinical laboratory diagnostics. E. J. Intern. Feder. Clin. Chem. 2015, 26, 47–62. [Google Scholar]
- Cheng, J.; Kricka, J.J.; Sheldon, E.L.; Wilding, P. Sample preparation in microstructures devices. In Microsystem Technology in Chemistry and Life Science; Springer: Berlin/Heidelberg, Germany, 1997; pp. 215–231. [Google Scholar]
- Kricka, L.J. Microchips, microarrays, biochips and nanochips: Personal laboratories for the 21st century. Clin. Chim. Acta 2001, 307, 219–223. [Google Scholar] [CrossRef]
- Hou, C.; Herr, A.E. Clinically relevant advances in on-chip affinity-based electrophoresis and electrochromatography. Electrophoresis 2008, 29, 3306–3319. [Google Scholar] [CrossRef]
- Pan, J.-Z.; Fang, P.; Fang, X.-X.; Hu, T.-T.; Fang, J.; Fang, Q. A low-cost palmtop high-speed capillary electrophoresis bioanalyzer with laser induced fluorescence detection. Sci. Rep. 2018, 1791. [Google Scholar] [CrossRef] [PubMed]
- Guzman, N.A. Capillary Electrophoresis Technology; Marcel Dekker, Inc.: New York, NY, USA, 1993. [Google Scholar]
- Lewis, A.P.; Cranny, A.; Harris, N.R.; Green, N.G.; Wharton, J.A.; Wood, R.J.K.; Stokes, K.R. Review on the development of truly portable and in-situ capillary electrophoresis systems. Meas. Sci. Technol. 2013, 24. [Google Scholar] [CrossRef]
- Yang, Z.; Sweedler, J.V. Applications of capillary electrophoresis for the early diagnosis of cancer. Anal. Bioanal. Chem. 2014, 406, 4013–4031. [Google Scholar] [CrossRef]
- Persson, F.; Rossing, P. Urinary proteomics and precision medicine for chronic kidney disease: Current status and future perspectives. Proteom. Clin. Appl. 2019, 13. [Google Scholar] [CrossRef]
- Ou, X.; Chen, P.; Huang, X.; Li, S.; Liu, B.F. Microfluidic chip electrophoresis for biochemical analysis. J. Sep. Sci. 2020, 43, 258–270. [Google Scholar] [CrossRef]
- Amin, R.; Knowlton, S.; Hart, A.; Yenilmez, B.; Ghaderinezhad, F.; Katebifar, S.; Messina, M.; Khademhosseini, A.; Tasoglu, S. 3-D printed microfluidic devices. Biofabrication 2016, 8. [Google Scholar] [CrossRef]
- Nielsen, A.V.; Beauchamp, M.J.; Nordin, G.P.; Woolley, A.T. 3D printed microfluidics. Annu. Rev. Anal. Chem. 2019, 13. [Google Scholar] [CrossRef]
- Guzman, N.A.; Phillips, T.M. Immunoaffinity CE for proteomics studies. Anal. Chem. 2005, 77, 60A–67A. [Google Scholar] [CrossRef] [Green Version]
- Guzman, N.A.; Blanc, T.; Phillips, T.M. Immunoaffinity capillary electrophoresis as a powerful strategy for the quantification of low-abundance biomarkers, drugs, and metabolites in biological matrices. Electrophoresis 2008, 29, 3259–3278. [Google Scholar] [CrossRef] [Green Version]
- Pagaduan, J.V.; Sahore, V.; Woolley, A.T. Applications of microfluidics and microchip electrophoresis for potential clinical biomarker analysis. Anal. Bioanal. Chem. 2015, 407, 6911–6922. [Google Scholar] [CrossRef] [Green Version]
- Phillips, T.M. Recent advances in CE and microchip-CE in clinical applications: 2014 to mid-2017. Electrophoresis 2018, 39, 126–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phillips, T.M.; Wellner, E.F. Analysis of inflammatory mediators in newborn dried blood spot samples by chip-base immunoaffinity capillary electrophoresis. Methods Mol. Biol. 2019, 1972, 185–198. [Google Scholar]
- Plenis, A.; Oledzka, I.; Kowalski, P.; Miekus, N.; Baczek, T. Recent trends in the quantification of biogenic amines in biofluids as biomarkers of various disorders: A review. J. Clin. Med. 2019, 8, 640. [Google Scholar] [CrossRef] [Green Version]
- Hu, R.; Chen, Y. Biocatalytic amplification of UV signal in capillary electrophoresis of microRNA. Int. J. Mol. Sci. 2020, 21, 51. [Google Scholar] [CrossRef] [Green Version]
- Tomlinson, A.J.; Benson, L.M.; Guzman, N.A.; Naylor, S. Enhancement of concentration limits of detection in capillary electrophoresis: Examples of on-line sample preconcentration, cleanup, and microreactor technology in protein characterization. Tech. Prot. Chem. 1997, 8, 15–24. [Google Scholar]
- Bonneil, E.; Waldron, K.C. On-line solid-phase preconcentration for sensitivity enhancement in capillary electrophoresis. J. Capill. Electrophor. Microchip Technol. 1999, 6, 61–73. [Google Scholar]
- Osbourn, D.M.; Weiss, D.J.; Lunte, C.E. On-line preconcentration methods for capillary electrophoresis. Electrophoresis 2000, 21, 2768–2779. [Google Scholar] [CrossRef] [Green Version]
- Benavente, F.; Vescina, M.C.; Hernández, E.; Sanz-Nebot, V.; Barbosa, J.; Guzman, N.A. Lowering the concentration limits of detection by on-line solid-phase extraction-capillary electrophoresis-electrospray mass spectrometry. J. Chromatogr. A 2007, 1140, 205–212. [Google Scholar] [CrossRef]
- Galievsky, V.A.; Stasheuski, A.S.; Krylov, S.N. “Getting the best sensitivity from on-capillary fluorescence detection in capillary electrophoresis”—A tutorial. Anal. Chim. Acta 2016, 935, 58–81. [Google Scholar] [CrossRef]
- Pont, L.; Pero-Gascon, R.; Gimenez, E.; Sanz-Nebot, V.; Benavente, F. A critical retrospective and prospective review of designs and materials in-line solid-phase extraction capillary electrophoresis. Anal. Chim. Acta 2019, 1079, 1–19. [Google Scholar] [CrossRef]
- Schiff, G.D.; Martin, S.A.; Eidelman, D.H.; Volk, L.A.; Ruan, E.; Cassel, C.; Galanter, W.; Johnson, M.; Jutel, A.; Kroenke, K.; et al. Ten principles for conservative, care-full diagnosis. Ann. Intern. Med. 2018, 169, 643–646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strausbauch, M.A.; Madden, B.J.; Wettstein, P.J.; Landers, J.P. Sensitivity enhancement and second-dimensional information from solid-phase extraction-capillary electrophoresis of entire high-performance liquid chromatography fractions. Electrophoresis 1995, 16, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, F.; Liu, Y. Recent advances in enhancing the sensitivity and resolution of capillary electrophoresis. J. Chromatogr. Sci. 2013, 51, 666–683. [Google Scholar] [CrossRef] [Green Version]
- Glatz, Z. On-capillary derivatization as an approach to enhancing sensitivity in capillary electrophoresis. Electrophoresis 2015, 36, 744–763. [Google Scholar] [CrossRef]
- Ruige, W.; Fung, Y.S. Microfluidic chip-capillary electrophoresis device for the determination of urinary metabolites and proteins. Bioanalysis 2015, 7, 907–922. [Google Scholar] [CrossRef]
- Slampova, A.; Malá, Z.; Gebauer, P. Recent progress of sample stacking in capillary electrophoresis (2016–2018). Electrophoresis 2018, 40, 40–54. [Google Scholar] [CrossRef]
- An, J.; Wang, X.; Ming, M.; Li, J.; Ye, N. Determination of sulfonamides in milk by capillary electrophoresis with PEG@MoS2 as a dispersive solid-phase extraction sorbent. Royal Soc. Open Sci. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Crego, A.L.; Mateos, M.; Nozal, L. Recent contributions for improving sensitivity in chiral CE. Electrophoresis 2018, 39, 67–81. [Google Scholar] [CrossRef]
- Guzman, N.A.; Guzman, D.E. From a central laboratory to the bedside: A point-of-care instrument to monitoring wellness and disease using two-dimensional immunoaffinity capillary electrophoresis technology. Archiv. Biomed. Res. 2018, 1, 1. [Google Scholar]
- Breadmore, M.C.; Grochocki, W.; Kalsoom, U.; Alves, M.; Phung, S.C.; Rokh, M.T.; Cabot, J.M.; Ghiasvand, A.; Li, F.; Shallan, A.I.; et al. Recent advances in enhancing the sensitivity of electrophoresis and electrochromatography in capillaries and microchips (2016–2018). Electrophoresis 2019, 40, 17–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaneta, T. Laser-induced fluorometry for capillary electrophoresis. Chem. Rec. 2019, 19, 452–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomes, F.P.; Yates, J.R., 3rd. Recent trends of capillary electrophoresis-mass spectrometry in proteomics research. Mass Spectrom. Rev. 2019, 38, 445–460. [Google Scholar] [CrossRef]
- Toraño, J.S.; Ramautar, R.; de Jong, G. Advances in capillary electrophoresis for the life science. J. Chromatogr. B 2019, 1118–1119, 116–136. [Google Scholar] [CrossRef] [PubMed]
- Nevidalová, H.; Michalcová, L.; Glatz, Z. Capillary electrophoresis-based approaches for the study of affinity interactions combined with various sensitive and nontraditional detection techniques. Electrophoresis 2019, 40, 625–642. [Google Scholar] [CrossRef] [PubMed]
- Vorobyeva, M.; Vorobjev, P.; Venyaminova, A. Multivalent aptamers: Versatile tools for diagnostic and therapeutic applications. Molecules 2016, 21, 1613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fields, C.; Li, P.; O’Mahony, J.J.; Lee, G.U. Advances in affinity ligand-functionalized nanomaterials for biomagnetic separation. Biotechnol. Bioeng. 2016, 113, 11–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Y.M.; Lin, D.Q.; Yao, S.J. Review on biomimetic affinity chromatography with short peptide ligands and its application to protein purification. J. Chromatogr. A 2018, 1571, 1–15. [Google Scholar] [CrossRef]
- Saeed, A.; Hussain, D.; Saleem, S.; Medhi, S.; Javeed, R.; Jabeen, R.; Najam-Ul-Haq, M. Metal-organics framework-based affinity materials in proteomics. Anal. Bioanal. Chem. 2019, 411, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Dunne, M.; Loessner, M.J. Modified bacteriophage tail fiber proteins for labeling, immobilization, capture, and detection of bacteria. Methods Mol. Biol. 2019, 1918, 67–86. [Google Scholar] [PubMed]
- Matos, M.J.B.; Pina, A.S.; Roque, A.C.A. Rational design of affinity ligands for bioseparation. J. Chromatogr. A 2020. [Google Scholar] [CrossRef] [PubMed]
- Welch, C.J.; Talaga, M.L.; Kadav, P.D.; Edwards, J.L.; Bandyopadhyay, P.; Dam, T.K. A capture and release method based on noncovalent ligand cross-linking and facile filtration for purification of lectins and glycoproteins. J. Biol. Chem. 2020, 295, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Guzman, N.A.; Trebilcock, M.A.; Advis, J.P. The use of a concentration step to collect urinary components separated by capillary electrophoresis and further characterization of collected analytes by mass spectrometry. J. Liquid Chromatogr. 1991, 14, 997–1015. [Google Scholar] [CrossRef]
- Guzman, N.A. Automated Capillary Electrophoresis Apparatus. U.S. Patent No. 5,202,010, 13 April 1993. [Google Scholar]
- Phillips, T.M.; Chmielinska, J.J. Immunoaffinity capillary electrophoretic analysis of cyclosporin in tears. Biomed. Chromatogr. 1994, 8, 242–246. [Google Scholar] [CrossRef]
- Guzman, N.A.; Park, S.S.; Schaufelberger, D.; Hernandez, L.; Paez, X.; Rada, P.; Tomlinson, A.J.; Naylor, S. New approaches in clinical chemistry: On-line analyte concentration and microreaction capillary electrophoresis for the determination of drugs, metabolic intermediates, and biopolymers in biological fluids. J. Chromatogr. B 1997, 697, 37–66. [Google Scholar] [CrossRef]
- Heegaard, N.H.H.; Nilsson, S.; Guzman, N.A. Affinity capillary electrophoresis: Important application areas and some recent developments. J. Chromatogr. B 1998, 715, 29–54. [Google Scholar] [CrossRef]
- Amundsen, L.K.; Sirén, H. Immunoaffinity CE in clinical analysis of body fluids and tissues. Electrophoresis 2007, 28, 99–113. [Google Scholar] [CrossRef]
- Peoples, M.C.; Karnes, H.T. Microfluidic immunoaffinity separations for bioanalysis. J. Chromatogr. B 2008, 866, 14–25. [Google Scholar] [CrossRef]
- Gasilova, N.; Girault, H.H. Bioanalytical methods for food allergy diagnosis, allergen detection and new allergen discovery. Bioanalysis 2015, 7, 1175–1190. [Google Scholar] [CrossRef] [Green Version]
- Sahore, V.; Sonker, M.; Nielsen, A.V.; Knob, R.; Kumar, S.; Woolley, A.T. Automated microfluidic devices integrating solid-phase extraction, fluorescent labeling, and microchip electrophoresis for preterm birth biomarker analysis. Anal. Bioanal. Chem. 2018, 410, 933–941. [Google Scholar] [CrossRef]
- Nevidalová, H.; Michalcová, L.; Glatz, Z. Capillary electrophoresis-based immunoassay and aptamer assay: A review. Electrophoresis 2020. [Google Scholar] [CrossRef]
- Zhang, C.; Woolfork, A.G.; Suh, K.; Ovbude, S.; Bi, C.; Elzoeiry, M.; Hage, D.S. Clinical and pharmaceutical applications of affinity ligands in capillary electrophoresis: A review. J. Pharmaceut. Biomed. Anal. 2020, 177. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Attiya, S.; Ocvirk, G.; Lee, W.E.; Harrison, D.J. Red diode laser induced fluorescence detection with a confocal microscope on a microchip for capillary electrophoresis. Biosens. Bioelectr. 2000, 14, 861–869. [Google Scholar] [CrossRef]
- Hodgson, R.J.; Brook, M.A.; Brennan, J.D. Capillary-scale monolithic immunoaffinity columns for immunoextraction with in-line laser induced fluorescence detection. Anal. Chem. 2005, 77, 4404–4412. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.X.; Busnel, J.M.; Peltre, G.; Zhang, X.X.; Girault, H.H. Magnetic beads based immunoaffinity capillary electrophoresis of total serum IgE with laser-induced fluorescence detection. Anal. Chem. 2008, 80, 9583–9588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peoples, M.C.; Phillips, T.M.; Karnes, H.T. Demonstration of a direct capture immunoaffinity separation for C-reactive protein using a capillary-based microfluidic device. J. Pharm. Biomed. Anal. 2008, 48, 376–382. [Google Scholar] [CrossRef]
- Chen, H.X.; Huang, T.; Zhang, X.X. Immunoaffinity extraction of testosterone by antibody immobilized monolithic capillary with on-line laser-induced fluorescence detection. Talanta 2009, 78, 259–264. [Google Scholar] [CrossRef]
- Mazouchi, A.; Dodgson, B.J.; Wegman, D.; Krylov, S.N.; Gradinaru, C.C. Ultrasensitive on-column laser-induced fluorescence in capillary electrophoresis using multiparameter confocal detection. Analyst 2012, 137, 5538–5545. [Google Scholar] [CrossRef]
- Peró-Gascón, R.; Pont, L.; Benavente, F.; Barbosa, J.; Sanz-Nebot, V. Analysis of serum transthyretin by on-line immunoaffinity solid-phase extraction capillary electrophoresis mass spectrometry using magnetic beads. Electrophoresis 2016, 37, 1220–1231. [Google Scholar] [CrossRef] [Green Version]
- Phillips, T.M.; Wellner, E.; McMohan, S.; Kalish, H. Measurement of inflammatory chemokines in micro-dissected tissue biopsy samples by chip-based immunoaffinity capillary electrophoresis. Methods Mol. Biol. 2016, 1466, 121–136. [Google Scholar]
- Han, M.; Pearson, J.T.; Wang, Y.; Winters, D.; Soto, M.; Rock, D.A.; Rock, B.M. Immunoaffinity capture couples with capillary electrophoresis—Mass spectrometry to study therapeutic protein stability in vivo. Anal. Biochem. 2017, 539, 118–126. [Google Scholar] [CrossRef]
- Weaver, M.T.; Lynch, K.B.; Zhu, Z.; Chen, H.; Lu, J.J.; Pu, Q.; Liu, S. Confocal laser-induced fluorescence detector for narrow capillary system with yoctomole limit of detection. Talanta 2017, 165, 240–244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guzman, N.A.; Trebilcock, M.A.; Advis, J.P. Separation of urinary constituents by capillary electrophoresis and further characterization of collected analytes by mass spectrometry. In Proceedings of the First Annual Conference on Capillary Electrophoresis, Frederick, MD, USA, 15–16 October 1990. [Google Scholar]
- Guzman, N.A.; Trebilcock, M.A.; Advis, J.P. Capillary electrophoresis for the analytical separation and semi-preparative collection of monoclonal antibodies. Anal. Chem. Acta 1991, 249, 247–255. [Google Scholar] [CrossRef]
- Guzman, N.A.; Trebilcock, M.A.; Advis, J.P. Affinity capillary electrophoresis: Two semi-preparative approaches to concentrate samples on the capillary column and to recover microgram quantities of material. In Proceedings of the 42nd Pittsburgh Conference & Exposition on Analytical Chemistry and Applied Spectroscopy, Chicago, IL, USA, 4–7 March 1991; p. 160. [Google Scholar]
- Guzman, N.A. Fabrication of an analyte concentrator-reaction chamber containing immobilized S. aureus V8 protease: On-column proteolytic digestion of nanogram quantities of substrate using capillary electrophoresis. In Proceedings of the Sixth Annual Symposium of the Protein Society, San Diego, CA, USA, 25–29 July 1992. [Google Scholar]
- Nashabeh, W.; El Rassi, Z. Enzymophoresis of nucleic acids by tandem capillary enzyme reactor-capillary zone electrophoresis. J. Chromatogr. A 1992, 596, 251–264. [Google Scholar] [CrossRef]
- Guzman, N.A. Determination of prolyl 4-hydroxylase beta-subunit at the zeptomole level using capillary electrophoresis. In Proceedings of the Fourth International Conference on the Molecular Biology and Pathology of Matrix, Philadelphia, PA, USA, 10–13 June 1992. [Google Scholar]
- Guzman, N.A. On-line peptide mapping of antibodies by capillary electrophoresis. In Proceedings of the Third International Conference on Human Antibodies and Hybridomas, San Antonio, TX, USA, 10–13 October 1993. [Google Scholar]
- Cole, L.J.; Kennedy, R.T. Selective preconcentration for capillary zone electrophoresis using protein G immunoaffinity capillary electrophoresis. Electrophoresis 1995, 16, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Guzman, N.A. Consecutive protein digestion and peptide derivatization employing an on-line analyte concentrator to map proteins using capillary electrophoresis. In CRC Series in Analytical Biotechnology; Righetti, P.G., Ed.; CRC Press, Inc.: Boca Raton, FL, USA, 1996; Chapter 4; pp. 101–121. [Google Scholar]
- Villegas, L.; Pero-Gascon, R.; Benavente, F.; Barbosa, J.; Sanz-Nebot, V. On-line protein digestion by immobilized enzyme microreactor capillary electrophoresis-mass spectrometry. Talanta 2019, 199, 116–123. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, Y.Z.; Zhou, D.D.; Yang, F.Q.; Li, D.Q. On-line immobilized microreactor for evaluating inhibitory activity of phenolic acids by capillary electrophoresis and molecular docking. Food Chem. 2020, 310. [Google Scholar] [CrossRef]
- Guzman, N.A. Disease Detection System and Method. U.S. Patent No. 9,146,234, 29 September 2015. [Google Scholar]
- Guzman, N.A. Disease Detection System and Method. U.S. Patent No. 10,408,789, 10 September 2019. [Google Scholar]
- Guzman, N.A. Method and System for Simultaneous Determination of Multiple Measurable Biomarkers during the Development of a Communicable Disease. U.S. Patent Application No. 2020/16/894,316, 5 June 2020. [Google Scholar]
- Johansson, T.; Petersson, M.; Johansson, J.; Nilsson, S. Real-time imaging through optical fiber array-assisted laser-induced fluorescence of capillary electrophoretic enantiomer separations. Anal. Chem. 1999, 71, 4190–4197. [Google Scholar] [CrossRef]
- Phillips, T.M.; Wellner, E.F. Chip-based immunoaffinity capillary electrophoresis: Application to the measurement of brain-derived neurotrophic factor in skin biopsies. Electrophoresis 2009, 30, 2307–2312. [Google Scholar] [CrossRef] [Green Version]
- Bathina, S.; Das, U.N. Brain-derived neurotrophic factor and its clinical implications. Arch. Med. Sci. 2015, 11, 1164–1178. [Google Scholar] [CrossRef]
- Giacobbo, B.L.; Doorduin, J.; Klein, H.C.; Dierckx, R.A.J.O.; Bromberg, E.; de Vries, E.F.J. Brain-derived neurotropic factor in brain disorders: Focus on neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312. [Google Scholar] [CrossRef] [Green Version]
- Morales-Cid, G.; Diez-Masa, J.C.; de Frutos, M. On-line immunoaffinity capillary electrophoresis based on magnetic beads for the determination of alpha-1-acid glycoprotein isoforms profile to facilitate its use as biomarker. Anal. Chim. Acta 2013, 773, 89–96. [Google Scholar] [CrossRef]
- Zhang, C.; Hage, D.S. Glycoform analysis of alpha-1-acid glycoprotein by capillary electrophoresis. J. Chromatogr. A 2016, 1475, 102–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sumanth, M.S.; Jacob, S.P.; Abhilasha, K.V.; Manne, B.K.; Basrur, V.; Lehoux, S.; Campbell, R.A.; Yost, C.C.; Rhondina, M.T.; McIntyre, T.M.; et al. Differential glycosylation of alpha-1-acid glycoprotein (AGP-I) contributes to its functional diversity. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- De Noni, I.; Fitzgerald, R.J.; Korhonen, H.J.T.; Le Roux, Y.; Livesey, C.T.; Thorsdottir, I.; Tomé, D.; Witkamp, R. Review on the potential health impact of ß-casomorphins and related peptides. EFSA Sci. Rep. 2009, 231, 1–107. [Google Scholar]
- Ul Haq, M.R.; Kapila, R.; Shandilya, U.K.; Kapila, S. Impact of milk derived ß-casomorphins on physiological functions and trends in research: A review. Inter. J. Food Propert. 2014, 17, 1726–1741. [Google Scholar] [CrossRef] [Green Version]
- Sahin, Ö.; Boztepe, S.; Aytekin, I. A1 and A2 bovine milk, the risk of beta-casomorphin-7 and its possible effects on human health: (I) A1 and A2 milk and the risk of beta-casomorphin-7. Selcuk J. Agric. Food. Sci. 2018, 32, 632–639. [Google Scholar]
- Clemens, R. A1/A2 milk and ß-casomorphins: The resurgence of controversy. Food Technol. 2018, 72, 12. [Google Scholar]
- Küllenberg de Gaudry, D.; Lohner, S.; Schmucker, C.; Kapp, P.; Motschall, E.; Hörrlein, S.; Roger, C.; Meerpohl, J.J. Milk A1 ß-casein and health related outcomes in humans: A systematic review. Nutr. Rev. 2019, 77, 278–306. [Google Scholar] [CrossRef]
- Summer, A.; Di Frangia, F.; Ajmone, M.P.; De Noni, I.; Malacarne, M. Ocurrence, biological properties and other potential effects on human health of ß-casomorphin 7: Current knowledge and concerns. Crit. Rev. Food Sci. Nutr. 2020, 1–19. [Google Scholar] [CrossRef]
- Zhang, F.; Chen, J.Y. A method for identifying discriminative isoform-specific peptides for clinical proteomics application. BMC Gen. 2016, 17 (Suppl. 7), 522. [Google Scholar] [CrossRef] [Green Version]
- Slim, F.A.; Ouellette, G.; Ennour-Idrissi, K.; Jacob, S.; Diorio, C.; Durocher, F. An isoform of AIF1 involved in breast cancer. Cancer Cell Int. 2018, 18, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malaney, P.; Uversky, V.N.; Davé, V. PTEN proteoforms in biology and disease. Cell. Mol. Life Sci. 2017, 74, 2783–2794. [Google Scholar] [CrossRef]
- Aebersold, R.; Agar, J.N.; Amster, I.J.; Baker, M.S.; Bertozzi, C.R.; Boja, E.S.; Costello, C.E.; Cravatt, B.J.; Fenselau, C.; Garcia, B.A.; et al. How many human proteoforms are there? Nat. Chem. Biol. 2018, 14, 206–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogaert, A.; Fernandez, E.; Gevaert, K. N-terminal proteoforms in human disease. Trends Biochem. Sci. 2020, 45, 308–320. [Google Scholar] [CrossRef] [Green Version]
- Lorentzian, A.; Uzozie, A.; Lange, P.F. Origins and clinical relevance of proteoforms in pediatric malignancies. Exp. Rev. Prot. 2019, 16, 185–200. [Google Scholar] [CrossRef] [Green Version]
- Durnez, L.; Van Bortel, W.; Denis, L.; Roelants, P.; Veracx, A.; Trung, H.D.; Sochantha, T.; Coosemans, M. False positive circumsporozoite protein ELISA: A challenge for the estimation of the entomological inoculation rate of malaria and for vector incrimination. Malar. J. 2011, 10, 195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwok, J.; Yang, M.; Wu, J.K.; Carter, C.J.; Jackson, S. High false positive rate of an ELISA screen for the detection of anti-factor VIII antibodies in congenital hemophilia A. Blood 2013, 122, 3586. [Google Scholar] [CrossRef]
- Terato, K.; Do, C.T.; Cutler, D.; Waritani, T.; Shionoya, H. Preventing intense false positive and negative reactions attributed to the principle of ELISA to re-investigate antibody studies in autoimmune diseases. J. Immunol. Meth. 2014, 407, 15–25. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Lei, Y.; Lu, X.; Wang, G.; Du, Q.; Guo, X.; Xing, Y.; Zhang, G.; Wang, D. Urea-mediated dissociation alleviate the false-positive Treponema pallidum-specific antibodies detected by ELISA. PLoS ONE 2019, 14, e0212893. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.H.; Tzioufas, A.G.; Notkins, A.L. Properties and function of polyreactive antibodies and polyreactive antigen-binding B cells. J. Autoimmun. 2007, 29, 219–228. [Google Scholar] [CrossRef] [Green Version]
- Dimitrov, J.D.; Planchais, C.; Roumenina, L.T.; Vassilev, T.L.; Kaveri, S.V.; Lacroix-Desmazes, S. Antibody polyreactivity in health and disease: Status variabilis. J. Immunol. 2013, 191, 993–999. [Google Scholar] [CrossRef] [Green Version]
- Palma, J.; Tokarz-Deptula, B.; Deptula, J.; Deptula, W. Natural antibodies—Facts known and unknown. Cent. Eur. J. Immunol. 2018, 43, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Notkins, A.L. Polyreactivity of antibody molecules. Trends Immunol. 2004, 25, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.; Ward, G. Interferences in immunoassay. Clin. Biochem. Rev. 2004, 25, 105–120. [Google Scholar] [PubMed]
- Nishi, N.; Takasu, M.; Kojima, M.; Hachisu, T.; Wakabayashi, K.; Iwasawa, A.; Maeda, S.; Ohba, Y.; Kitagawa, H. Presence of anti-insulin natural autoantibodies in healthy cats and its interference with immunoassay for serum insulin concentrations. Domest. Anim. Endocrinol. 2010, 38, 138–145. [Google Scholar] [CrossRef]
- Fortgens, P.H.; Omans, F. Cardiac troponin T quantitative assay failure as a result of antibody interference. Afr. J. Lab. Med. 2013, 2, 23. [Google Scholar] [CrossRef]
- Warade, J. Retrospective approach to evaluate interferences in immunoassay. EJIFCC 2017, 28, 224–232. [Google Scholar]
- Guzman, N.A. Electrophoresis Apparatus Having Staggered Passage Configuration. U.S. Patent No. 7,329,388, 12 February 2008. [Google Scholar]
- Guzman, N.A.; Phillips, T.M. Immunoaffinity capillary electrophoresis: A new versatile tool for the determining protein biomarkers in inflammatory processes. Electrophoresis 2011, 32, 1565–1578. [Google Scholar] [CrossRef]
- Hassanzadeh-Ghassabeh, G.; Devoogdt, N.; De Pauw, P.; Vincke, C.; Muyldermans, S. Nanobodies and their potential applications. Nanomedicine 2013, 8, 1013–1026. [Google Scholar] [CrossRef] [Green Version]
- Chanier, T.; Chames, P. Nanobody engineering: Toward next generation immunotherapies and immunoimaging of cancer. Antibodies 2019, 8, 13. [Google Scholar] [CrossRef] [Green Version]
- Jovcevska, I.; Muyldermans, S. The therapeutic potential of nanobodies. BioDrugs 2020, 34, 11–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nimjee, S.M.; White, R.R.; Becker, R.C.; Sullenger, B.A. Aptamers as therapeutics. Ann. Rev. Pharmacol. Toxicol. 2017, 57, 61–79. [Google Scholar] [CrossRef] [PubMed]
- Dunn, M.R.; Jimenez, R.M.; Chaput, J.C. Analysis of aptamer discovery and technology. Nat. Rev. Chem. 2017, 1, 76. [Google Scholar] [CrossRef]
- Adachi, T.; Nakamura, Y. Aptamers: A review of their chemical properties and modifications for therapeutic application. Molecules 2019, 24, 4229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, Y.; Tateno, H.; Ogawa, H.; Yamamoto, K.; Hirabayashi, J. Comprehensive list of lectins: Origins, natures, and carbohydrate specificities. Methods Mol. Biol. 2014, 1200, 555–577. [Google Scholar]
- Hirabayashi, J.; Arai, R. Lectin engineering: The possible and the actual. Interface Focus 2019, 9, 20180068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Warner, E.; Parikh, N.D.; Lubman, D.M. Glycoprotein markers of hepatocellular carcinoma-mass spectrometry based approaches. Mass Spectrom. Rev. 2019, 38, 265–290. [Google Scholar] [CrossRef]
- Lastovickova, M.; Strouhalova, D.; Bobalova, J. Use of lectin-based affinity techniques in breast cancer glycoproteomics: A review. J. Proteome Res. 2020. [Google Scholar] [CrossRef]
- Goda, T.; Higashi, D.; Matsumoto, A.; Hoshi, T.; Sawaguchi, T.; Miyahara, Y. Dual aptamer-immobilized surfaces for improved affinity through multiple target binding in potentiometric thrombin sensing. Biosens. Bioelectron. 2015, 73, 174–180. [Google Scholar] [CrossRef] [Green Version]
- Phillips, T.M.; Wellner, E. Detection of cerebral spinal fluid-associated chemokines in birth traumatized premature babies by chip-based immunoaffinity CE. Electrophoresis 2013, 34, 1530–1538. [Google Scholar] [CrossRef] [Green Version]
- Guzman, N.A. Integrated Modular Unit including an Analyte Concentrator Microreactor Device Connected to a Cartridge-Cassette. U.S. Patent No. 9,696,299, 4 July 2017. [Google Scholar]
- Gregus, M.; Foret, F.; Kuban, P. Single-breath analysis using a novel simple sampler and capillary electrophoresis with contactless conductometric detection. Electrophoresis 2014, 36, 526–533. [Google Scholar] [CrossRef]
- Van der Schee, M.; Pinheiro, H.; Gaude, E. Breath biopsy for early detection and precision medicine in cancer. Ecancermedicalscience 2018, 12, ed84. [Google Scholar] [CrossRef] [Green Version]
- Fabian, P.; Brain, J.; Houseman, E.A.; Gern, J.; Milton, D.K. Origin of exhaled breath particles from healthy and human rhinovirus-infected subjected. J. Aerosol Med. Pulm Drug Deliv. 2011, 24, 137–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawal, O.; Ahmed, W.M.; Nijsen, T.M.E.; Goodacre, R.; Fowler, S.J. Exhaled breath analysis: A review of ‘breath-taking’ methods off-line analysis. Metabolomics 2017, 13, 110. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Pal, M. Review–Non-invasive monitoring of human heath by exhaled breath analysis: A comprehensive review. J. Electrochem. Soc. 2020, 167, 37562. [Google Scholar] [CrossRef]
- Miekisch, W.; Schubert, J.K.; Noeldge-Schomburg, G.F.E. Diagnostic potential of breath analysis—Focus on volatile organic compounds. Clin. Chim. Acta 2004, 347, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Patra, A.; Kutty, V.K.; Venkatesan, T. Critical review of volatile organic compound analysis in breath and in vitro cell culture for detection of lung cancer. Metabolites 2019, 9, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashoul, D.; Haick, H. Sensors for detecting pulmonary diseases from exhaled breath. Eur. Resp. Rev. 2019, 28, 190011. [Google Scholar] [CrossRef] [Green Version]
- Bourouiba, L. Turbulent gas clouds and respiratory pathogen emissions. Potential implications for reducing transmission of COVID-19. J. Am. Med. Assoc. 2020, 323, 1837–1838. [Google Scholar]
- Aliberti, S.; Morlacchi, L.C.; Faverio, P.; Fernandez-Botran, R.; Cosentini, R.; Mantero, M.; Peyrani, P.; Ramirez, J.; Bordon, J.; Blasi, F. Serum and exhaled breath condensate inflammatory cytokines in community-acquired pneumonia: A prospective cohort study. Pneumonia 2016, 8. [Google Scholar] [CrossRef] [Green Version]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Nicolle, L.E. Catheter associated urinary tract infections. Antimicrob. Resist. Infect. Control. 2014, 3, 23. [Google Scholar] [PubMed] [Green Version]
- Gadalla, A.A.H.; Friberg, I.M.; Kift-Morgan, A.; Zhang, J.; Eberl, M.; Topley, N.; Weeks, I.; Cuff, S.; Wooton, M.; Gal, M.; et al. Identification of clinical and urine biomarkers for uncomplicated urinary tract infection using machine learning algorithms. Sci. Rep. 2019, 19694. [Google Scholar] [CrossRef] [PubMed]
- Lagier, J.-C.; Edouard, S.; Pagnier, I.; Mediannikov, O.; Drancourt, M.; Raoult, D. Current and past strategies for bacterial culture in clinical microbiology. Clin. Microbiol. Rev. 2015, 28, 208–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Street, J.M.; Koritzinsky, E.H.; Glispie, D.M.; Star, R.A.; Yuen, P.S. Urine exosomes: An emerging trove of biomarkers. Adv. Clin. Chem. 2017, 78, 103–122. [Google Scholar] [PubMed]
- Ashkenazi-Hoffnung, L.; Oved, K.; Navon, R.; Friedman, T.; Boico, O.; Paz, M.; Kronenfeld, G.; Etshtein, L.; Cohen, A.; Gottlieb, T.M.; et al. A host-protein signature is superior to other biomarkers for differentiating between bacterial and viral disease in patients with respiratory infection and fever without source: A prospective observational study. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 1361–1371. [Google Scholar] [CrossRef] [Green Version]
- Binnicker, M.J. Emergence of a novel coronavirus disease (COVID-19) and the importance of diagnostic testing: Why partnership between clinical laboratories, public health agencies, and industry is essential to control the outbreak. Clin. Chem. 2020, 66, 664–666. [Google Scholar] [CrossRef]
- Murthy, S.; Gomersall, C.D.; Fowler, R.A. Care for critically ill patients with COVID-19. Jama 2020, 323, 499–1500. [Google Scholar] [CrossRef] [Green Version]
- Vembadi, A.; Menachery, A.; Qasaimeh, M.A. Cell cytometry: Review and perspective on biotechnological advances. Front. Bioengineer. Biotechnol. 2019, 7, 147. [Google Scholar] [CrossRef]
- Hu, P.; Zhang, W.; Xin, H.; Deng, G. Single cell isolation and analysis. Front. Cell Dev. Biol. 2016, 4, 116. [Google Scholar] [CrossRef] [Green Version]
- Lippé, R. Flow virometry: A powerful tool to functionally characterize viruses. J. Virol. 2018, 92, e01765-17. [Google Scholar] [CrossRef] [Green Version]
- Pospichalova, V.; Svoboda, J.; Dave, Z.; Kotrbova, A.; Kaiser, K.; Klemova, D.; Ilkovics, L.; Hampl, A.; Crha, I.; Jandakova, E.; et al. Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer. J. Extracel. Ves. 2015, 4, 25530. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; He, M.; Jiang, S.C. Detection of infectious adenoviruses in environmental waters by fluorescence-activated cell sorting assay. Appl. Environ. Microbiol. 2010, 76, 1442–1448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khalill, J.Y.; Langlois, T.; Andreani, J.; Sorraing, J.-M.; Raoult, D.; Camoin, L.; La Scola, B. Flow cytometry sorting to separate viable giant viruses from amoeba co-cultures supernatants. Front. Cell. Infect. Microbiol. 2017, 6, 202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ewing, A.G. Microcolumn separations of single nerve cell components. J. Neurosci. Meth. 1993, 48, 215–224. [Google Scholar] [CrossRef]
- Cecala, C.; Sweedler, J.V. Sampling techniques for single-cell electrophoresis. Analyst 2012, 137, 2922–2929. [Google Scholar] [CrossRef] [Green Version]
- Allen, P.B.; Doepker, B.R.; Chiu, D.T. High-throughput capillary-electrophoresis analysis of the contents of single mitochondria. Anal. Chem. 2009, 81, 3784–3791. [Google Scholar] [CrossRef] [Green Version]
- Kostal, V.; Fonslow, B.R.; Arriaga, E.A.; Bowser, M.T. Fast determination electrophoretic mobility using micro-free flow electrophoresis. Anal. Chem. 2009, 81, 9267–9273. [Google Scholar]
- Saeedi, S.; Israel, S.; Nagy, C.; Turecki, G. The emerging role of exosomes in mental disorders. Transl. psychiatry 2019, 9, 122. [Google Scholar] [CrossRef]
- Zaborowski, M.J.; Balaj, L.; Breakefield, X.O.; Lai, C.P. Extracellular vesicles: Composition, biological relevance, and methods of study. Bioscience 2015, 65, 783–797. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Higginbotham, J.N.; Jeppesen, D.K.; Yang, Y.-P.; Li, W.; McKinley, E.T.; Graves-Deal, R.; Ping, J.; Britain, C.M.; Dorsett, K.A.; et al. Transfer of functional cargo in Exomeres. Cell Rep. 2019, 27, 940–954.e6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nature Rev. Mol. Cell. Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Margolis, L.; Sadovsky, Y. The biology of extracellular vesicles: The known unkowns. PLoS Biol. 2019, 17, e3000363. [Google Scholar] [CrossRef]
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaa6977. [Google Scholar] [CrossRef]
- Kremser, L.; Konecsni, T.; Blaas, D.; Kenndler, E. Fluorescence labeling of human rhinovirus capsid and analysis by capillary electrophoresis. Anal. Chem. 2004, 76, 4175–4181. [Google Scholar] [CrossRef]
- Desai, M.J.; Armstrong, D.W. Separation, identification, and characterization of microorganisms by capillary electrophoresis. Microbiol. Mol. Biol. Rev. 2003, 67, 38–51. [Google Scholar] [CrossRef] [Green Version]
- Riley, K.R.; Liu, S.; Yu, G.; Libby, K.; Cubicciotti, R.; Colyer, C.L. Using capillary electrophoresis to characterize polymeric particles. J. Chromatogr. A 2016, 1463, 169–175. [Google Scholar] [CrossRef]
- Bäck, A.T.; Lundkvist, A. Dengue viruses—An overview. Infect. Ecol. Epidemiol. 2013, 3, 19839. [Google Scholar] [CrossRef]
- Raut, R.; Corbett, K.S.; Tennekoon, R.N.; Premawansa, S.; Wijewickrama, A.; Premawansa, G.; Mieczkowski, P.; Ruckert, C.; Ebel, G.D.; De Silva, A.D.; et al. Dengue type 1 viruses circulating in humans are highly infectious and poorly neutralized by human antibodies. Proc. Natl. Acad. Sci. USA 2019, 116, 227–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention. Human Coronaviruses. 2020. Available online: https://www.cdc.gov/coronavirus/types.html (accessed on 15 February 2020).
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 16, 69. [Google Scholar] [CrossRef] [Green Version]
- Rey, F.A.; Lok, S.-M. Common features of enveloped viruses and implications for immunogen design for next-generation vaccines. Cell 2018, 172, 1319–1334. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.P.; Kermode, A.G.; Lucas, R.M.; Carroll, W.M.; Nolan, D.; Hart, P.H. Circulating immune cells in multiple sclerosis. Clin. Exp. Immunol. 2016, 187, 193–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.-L.; Shan, S.; Xiong, M.; Xia, X.-H.; Xu, J.-J.; Chen, H.-Y. On-chip selective capture of cancer cells and ultrasensitive fluorescence detection of surviving mRNA in a single living cell. Lab Chip 2013, 13, 3868–3875. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Carrión, C.; Moliner-Martínez, Y.; Simonet, B.M.; Valcárcel, M. Capillary electrophoresis method for the characterization and separation of CdSe quantum dots. Anal. Chem. 2011, 83, 2807–2813. [Google Scholar] [CrossRef]
- Piotrowska, M.; Ciura, K.; Zalewska, M.; Dawid, M.; Correia, B.; Sawicka, P.; Lewczuk, B.; Kasprzyk, J.; Sola, L.; Piekoszewski, W.; et al. Capillary electrophoresis of bacterial extracellular vesicles: A proof of concept. J. Chromatogr. A 2020, 1621, 461047. [Google Scholar] [CrossRef]
- Contreras-Naranjo, J.C.; Wu, H.-J.; Ugaz, V.M. Microfluidics for exosome isolation and analysis: Enabling liquid biopsy for personalized medicine. Lab Chip 2017, 17, 3558–3577. [Google Scholar] [CrossRef]
- Bruce, T.F.; Slonecki, T.J.; Wang, L.; Huang, S.; Powell, R.R.; Marcus, R.K. Exosome isolation and purification via hydrophobic interaction chromatography using polyester, capillary-channeled polymer fiber phase. Electrophoresis 2018, 40, 571–581. [Google Scholar] [CrossRef]
- Willms, E.; Johansson, H.J.; Mäger, I.; Lee, Y.; Blomberg, K.E.M.; Sadik, M.; Alaerg, A.; Smith, C.I.E.; Lehtiö, J.; El Andaloussi, S.; et al. Cells release subpopulations of exosomes with distinct molecular and biological properties. Sci. Rep. 2016, 6, 22519. [Google Scholar] [CrossRef]
- Eldh, M.; Lötvall, J.; Malmhäll, C.; Ekström, K. Importance of RNA isolation methods for analysis of exosomal RNA: Evaluation of different methods. Mol. Immunol. 2012, 50, 278–286. [Google Scholar] [CrossRef]
- Cheung, L.S.; Sahloul, S.; Orozaliev, A.; Song, Y.-A. Rapid detection and trapping of extracellular vesicles by electrokinetic concentration for liquid biopsy on chip. Micromachines 2018, 9, 306. [Google Scholar] [CrossRef] [Green Version]
- Kato, K.; Kobayashi, M.; Hanamura, N.; Akagi, T.; Kosaka, N.; Ochiya, T.; Ichiki, T. Electrokinetic evaluation of individual exosomes by on-chip microcapillary electrophoresis with laser dark-field microscopy. Jpn. J. Appl. Phys. 2013, 52, 06GK10. [Google Scholar] [CrossRef]
- Grigor’eva, A.E.; Tamkovich, S.N.; Eremina, A.V.; Tupikin, A.E.; Kabilov, M.R.; Chernykh, V.V.; Vlassov, V.V.; Laktionov, P.P.; Ryabchikova, E.I. Characteristics of exosomes and microparticles discovered in human tears. Biomed. Khimiia 2016, 62, 99–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hisey, C.L.; Dorayappan, K.D.P.; Cohn, D.E.; Selvendiran, K.; Hansford, D.J. Microfluidic affinity separation chip for selective capture and release of label-free ovarian cancer exosomes. Lab Chip 2018, 18, 3144–3153. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.G.; Buller, B.; Chopp, M. Exosomes—Beyond stems cells for restorative therapy in stroke and neurological injury. Nat. Rev. Neurol. 2019, 15, 193–203. [Google Scholar] [CrossRef]
- Li, X.; Corbett, A.L.; Taatizadeh, E.; Tasmin, N.; Little, J.P.; Garnis, C.; Daugaard, M.; Guns, E.; Hoorfar, M.; Li, I.T.S. Challenges and opportunities in exosome research—Perspectives from biology, engineering, and cancer therapy. APL Bioeng. 2019, 3, 1153. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Li, C.; Zhang, L.; Wu, M.; Cao, K.; Jiang, F.; Chen, D.; Li, N.; Li, W. The significance of exosomes in the development and treatment of hepatocellular carcinoma. Mol. Cancer 2020, 19, 1. [Google Scholar] [CrossRef]
- Szatanek, R.; Baj-Krzyworzeka, M.; Zimoch, J.; Lekka, M.; Siedlar, M.; Baran, J. The methods of choice for extracellular vesicles (EVs) characterization. Int. J. Mol. Sci. 2017, 18, 1153. [Google Scholar] [CrossRef]
- Hartjes, T.A.; Mytnyk, S.; Jenster, G.W.; van Steijn, V.; van Royen, M.E. Extracellular vesicle quantification and characterization: Common methods and emerging approaches. Bioengineering 2019, 6, 7. [Google Scholar] [CrossRef] [Green Version]
- Ma, C.; Jiang, F.; Ma, Y.; Wang, J.; Li, H.; Zhang, J. Isolation and detection technologies of extracellular vesicles and application on cancer diagnostic. Dose Respon. 2019, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurunathan, S.; Kang, M.-H.; Jeyaraj, M.; Qasim, M.; Kim, J.-H. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells 2019, 8, 307. [Google Scholar] [CrossRef] [Green Version]
- Iliescu, F.S.; Vrtacnik, D.; Neuzil, P.; Iliescu, C. Microfluidic technology for clinical applications of exosomes. Micromachines 2019, 10, 392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koritzinsky, E.H.; Street, J.M.; Star, R.A.; Yuen, P.S.T. Quantification of exosomes. J. Cell Physiol. 2017, 232, 1587–1590. [Google Scholar] [CrossRef]
- Chen, B.-Y.; Sung, C.W.-H.; Chen, C.; Cheng, C.-M.; Lin, D.P.-C.; Huang, C.-T.; Hsu, M.-Y. Advances in exosomes technology. Clin. Chim. Acta 2019, 493, 14–19. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Crow, J.; Roth, M.; Zeng, Y.; Godwin, A.K. Integrated immunoisolation and protein analysis of circulating exosomes using microfluidic technology. Lab Chip 2014, 14, 3773–3780. [Google Scholar] [CrossRef] [Green Version]
- Ko, J.; Carpenter, E.; Issadore, D. Detection and isolation of circulating exosomes and microvesicles for cancer monitoring and diagnostics using micro-/nano-based devices. Analyst 2016, 141, 450–460. [Google Scholar] [CrossRef] [Green Version]
- Akagi, T.; Kato, K.; Kobayashi, M.; Kosaka, N.; Ochiya, T.; Ichiki, T. On-chip immunoelectrophoresis of extracellular vesicles released from human breast cancer cells. PLoS ONE 2015. [Google Scholar] [CrossRef]
- Watkins, N.N.; Irimia, D.; Toner, M.; Bashir, R. On a chip. IEEE Pulse 2011, 2, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Gholizadeh, S.; Draz, M.; Zarghooni, M.; Nezhad, A.S.; Ghavami, S.; Shafiee, H.; Akbari, M. Microfluidic approaches for isolation, detection and characterization of extracellular vesicles: Current status and future directions. Biosens. Bioelectron. 2017, 91, 588–605. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Yao, Y.; Chen, X.; Wu, J.; Gu, T.; Tang, X. Host derived exosomes-pathogens interactions: Potential functions of exosomes in pathogen infection. Biomed. Pharmacother. 2018, 108, 1451–1459. [Google Scholar] [CrossRef] [PubMed]
- Keller, M.D.; Ching, K.L.; Liang, F.-X.; Dhabaria, A.; Tam, K.; Ueberheide, B.M.; Unutmaz, D.; Torres, V.J.; Cadwell, K. Decoy exosomes provide protection against bacterial toxins. Nature 2020, 579, 260–264. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, D.; Zhang, W.; Zhang, H.; Zhang, F.; Chen, L.; Ma, L.; Larcher, L.M.; Chen, S.; Liu, N.; Zhao, Q.; et al. Progress, opportunity, and perspective on exosome-based theranostics. Theranostics 2020, 10, 3684–3707. [Google Scholar] [CrossRef]
- Inamdar, S.; Nitiyanandan, R.; Rege, K. Emerging applications of exosomes in cancer therapeutics and diagnostics. Bioengineer. Transl. Med. 2017, 2, 70–80. [Google Scholar] [CrossRef]
- Madhusoodanan, J. The therapeutic potential of exosomes. Nature 2020, 581, S10–S11. [Google Scholar] [CrossRef]
- Hassanpour, M.; Rezaie, J.; Nouri, M.; Panahi, Y. The role of extracellular vesicles in COVID-19 virus infection. Infect. Genet. Evol. 2020, 85, 104422. [Google Scholar] [CrossRef]
- Ramirez, M.I.; Amorim, M.G.; Gadelha, C.; Milic, I.; Welsh, J.A.; Freitas, V.M.; Nawas, M.; Akbar, N.; Couch, Y.; Makin, L.; et al. Technical challenges of working with extracellular vesicles. Nanoscale 2018, 10, 881–906. [Google Scholar] [CrossRef] [Green Version]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitochaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.; et al. Minimal information of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracel. Ves. 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Francavilla, A.; Turoczi, S.; Tarallo, S.; Vodicka, P.; Pardini, B.; Naccarati, A. Exosomal microRNAs and other non-coding RNSs as colorectal cancer biomarkers: A review. Mutagenesis 2020, 35, 243–260. [Google Scholar] [CrossRef]
- Silva, M.; Melo, S.A. Non-coding RNAs in exosomes: New players in cancer biology. Curr. Genom. 2015, 16, 295–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dexheimer, P.J.; Cochella, L. MicroRNAs: From mechanism to organism. Front. Cell Dev. Bio. 2020, 8, 409. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.P.; Boon, R.A. Exosomes and non-coding RNA, the healers of the heart? (Editorial). Cardiovasc. Res. 2020, 116, 258–259. [Google Scholar] [PubMed]
- Zeng, B.; Chen, T.; Luo, J.; Xie, M.; Wei, L.; Xi, Q.; Sun, J.; Zhang, Y. Exploration of long non-coding RNAs and circular RNAs in porcine milk exosomes. Front. Genet. 2020, 11, 652. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, A.F.; Lee, E.S. Non-coding RNA: What is functional and what is junk? Front. Genet. 2015, 6, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunasekaran, M.; Bansal, S.; Ravichandran, R.; Sharma, M.; Perincheri, S.; Rodriguez, F.; Hachem, R.; Fisher, C.E.; Limaye, A.P.; Omar, A.; et al. Respiratory viral infection in lung transplantation induces exosomes that trigger chronic rejection. J. Heart Lung Transplant. 2020, 39, 379–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.; Kim, Y.; Mirzaaghasi, A.; Heo, J.; Kim, Y.N.; Shin, J.H.; Kim, S.; Kim, N.H.; Cho, E.S.; Yook, J.I.; et al. Exosomes-based delivery of super-repressor IkBa relieves sepsis-associated organ damage and mortality. Sci. Adv. 2020, 6, eaaz6980. [Google Scholar] [CrossRef] [Green Version]
- Floriano, J.F.; Willis, G.; Catapano, F.; de Lima, P.R.; Reis, F.V.D.S.; Barbosa, A.M.P.; Rudge, M.V.C.; Emanueli, C. Exosomes could offer new options to combat the long-term complications inflicted by gestational diabetes mellitus. Cells 2020, 9, 675. [Google Scholar] [CrossRef] [Green Version]
- Taghikhani, A.; Farzaneh, F.; Sharifzad, F.; Mardpour, S.; Ebrahimi, M.; Hassan, Z.M. Engineered tumor-derived extracellular vesicles: Potentials in cancer immunotheraphy. Front. Immunol. 2020, 11, 221. [Google Scholar] [CrossRef]
- Xing, X.; Han, S.; Cheng, G.; Ni, Y.; Li, Z.; Li, Z. Proteomic analysis of exosomes from adipose-derived mesenchymal stem cells: A novel therapeutic strategy for tissue injury. BioMed Res. Intern. 2020, 6094562. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Y.; Xiao, Y.; Crawford, R.; Mao, X.; Prasadam, I. Extracellular vesicles: Potential role in osteoarthritis regenerative medicine. J. Orthop. Transl. 2020, 21, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xie, Y.; Zhang, A.; Wang, M.; Fang, Z.; Zhang, J. Exosomes: An emerging factor in atherosclerosis. Biomed. Pharmacother. 2019, 115, 108951. [Google Scholar] [CrossRef] [PubMed]
- Pisitkun, T.; Johnstone, R.; Knepper, M.A. Discovery of urinary biomarkers. Mol. Cell. Proteom. 2006, 5, 1760–1771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKierman, J.; Donovan, M.J.; O’Neill, V.; Bentink, S.; Noerholm, M.; Belzer, S.; Skog, J.; Kattan, M.W.; Partin, A.; Andriole, G.; et al. High-grade prostate cancer at initial biopsy. JAMA Oncol. 2016, 2, 882–889. [Google Scholar] [CrossRef] [Green Version]
- Sengupta, V.; Sengupta, S.; Lazo, A.; Woods, P.; Nolan, A.; Bremer, N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020. [Google Scholar] [CrossRef]
- Kwon, Y.; Nukala, S.B.; Srivastava, S.; Miyamoto, H.; Ismail, N.I.; Ong, S.-B.; Lee, W.H.; Ong, S.-G. Exosomes facilitate transmission of SARS-CoV-genome into human induced pluripotent stem cell-derived cardiomyocytes. bioRxiv 2020. [Google Scholar] [CrossRef]
- Munagala, R.; Aqil, F.; Jeyabalan, J.; Gupta, R.C. Bovine milk-derived exosomes for drug delivery. Cancer Lett. 2016, 371, 48–61. [Google Scholar] [CrossRef] [Green Version]
- Mendt, M.; Rezvani, K.; Shpall, E. Mesenchymal stem cell-derived exosomes for clinical use. Bone Marrow Transpl. 2019, 54 (Suppl. 2), 789–792. [Google Scholar] [CrossRef]
- Ivica, A.; Ghayor, C.; Zehnder, M.; Valdec, S.; Weber, F.E. Pulp-derived exosomes in fibrin-based regenerative root filling material. J. Clin. Med. 2020, 9, 491. [Google Scholar] [CrossRef] [Green Version]
- Gluszko, A.; Szczepanski, M.J.; Ludwig, N.; Mirza, S.M.; Olejarz, W. Exosomes in cancer: Circulating immune-related biomarkers. BioMed Res. Int. 2019, 1628029. [Google Scholar] [CrossRef] [PubMed]
- Kuate, S.; Cinati, J.; Doerr, H.W.; Uberla, K. Exosomal vaccines containing the S protein of the SARS coronavirus induce high level of neutralizing antibodies. Virology 2007, 362, 26–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, J.; Meng, J.; Zhu, L.; Peng, Y. Exosomal noncoding RNAs in glioma: Biological functions and potential clinical applications. Mol. Cancer 2020, 19, 66. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, B.; Yang, Y.; Jia, Q.; Qi, Z.; Zhang, A.; Lv, S.; Zhang, J. Exosomes in ischemic heart disease: Novel carriers for bioinformation. Biomed. Pharmacother. 2019, 120, 109451. [Google Scholar] [CrossRef] [PubMed]
- Thongboonkerd, V. Roles for exosomes in various kidney diseases and disorders. Front. Pharmacol. 2020, 10, 1655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Console, L.; Scalise, M.; Indiven, C. Exosomes in inflammation and role as biomarkers. Clin. Chim. Acta 2019, 488, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Feng, Z.; Yuan, G.; Emerson, C.C.; Stewart, P.L.; Ye, F.; Jin, G. Human immunodeficiency virus-associated exosomes promote Kaposi’s sarcoma-associated herpesvirus infection via the epidermal growth factor. J. Virol. 2020, 94, e01782-19. [Google Scholar] [CrossRef] [Green Version]
- Pucci, M.; Taverna, S.; Reclusa, P.; Pinto, J.A.; Durendez, E.; Lewintre, E.J.; Malarani, M.; Zito, G.; Rolfo, C. Exosomes in semen: Opportunities as a new tool in prostate cancer diagnosis. Transl. Cancer Res. 2017, 6 (Suppl. 8), S1331–S1338. [Google Scholar] [CrossRef]
- Cao, X.-Y.; Lu, J.-M.; Zhao, Z.-Q.; Li, M.-C.; Lu, T.; An, X.-S.; Xue, L.-J. MicroRNA biomarkers of Parkinson’s disease in serum exosome-like microvesicles. Neurosci. Lett. 2017, 644, 94–99. [Google Scholar] [CrossRef]
- De la Torre-Gomez, C.; Goreham, R.V.; Bech-Serra, J.J.; Nann, T.; Kussmann, M. “Exosomics”—A review of biophysics, biology and biochemistry of exosomes with a focus on human breast milk. Front. Genet. 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Zhao, Y.; Sun, L.; Sun, X.; Zhao, X.; Sun, X.; Qian, H.; Xu, W.; Zhu, W. Exosomes derived from Akt-modified human umbilical cord mesenchymal stem cells improve cardiac regeneration and promote angiogenesis via activating platelet-derived growth factor D. Stem Cells Transl. Med. 2017, 6, 51–59. [Google Scholar] [CrossRef] [Green Version]
- Hough, K.P.; Deshane, J.S. Exosomes in allergic airway diseases. Curr. Allergy Asthma Rep. 2019, 19, 26. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zheng, T.; Zhang, B. Exosomes in Parkinson’s disease. Neurosci. Bull 2017, 33, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Mesci, P.; Carromeu, C.; McClatchy, D.R.; Schiapparelli, L.; Yates, J.R., III; Muotri, A.R.; Cline, H.T. Exosomes regulate neurogenesis and circuit assembly. Proc. Natl. Acad. Sci. USA 2019, 116, 16086–16094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhuo, C.-J.; Hou, W.-H.; Jiang, D.-G.; Tian, H.-J.; Wang, L.-N.; Jia, F.; Zhou, C.-H.; Zhu, J.-J. Circular RNAs in early brain development and their influence and clinical significance in neuropsychiatric disorders. Neural Regen. Res. 2020, 15, 817–823. [Google Scholar] [CrossRef]
- Cheng, L.; Zhang, K.; Qing, Y.; Li, D.; Cui, M.; Jin, P.; Xu, T. Proteomic and lipidomic analysis of exosomes derived from ovarian cancer cells and ovarian surface epithelial cells. J. Ovarian Res. 2020, 13, 9. [Google Scholar] [CrossRef]
| Source | Clinical Significance | Clinical Studies | Reference |
|---|---|---|---|
| Mesenchymal stromal cells-derived exosomes | Potential to improve neurological injury. | Understanding the effect of exosomes as mediators of the beneficial effects of cell therapy for stroke and traumatic brain injury. | [225] |
| Human hepatocellular carcinoma exosomes | Important role in the diagnosis and therapy for tumors. Screening for biomarkers in early formation of chronic hepatitis and liver cancer. | Elucidation of signal pathways and their involvement in growth, metastasis, and angiogenesis; and of the significance of exosomes in the treatment of hepatocellular carcinoma. | [227] |
| Human serum exosomes | Symptomatic respiratory viral infections after lung transplantation. | Presence of lung self-antigens, 20S proteasome, and viral antigens implies that exosomes trigger chronic rejection. | [255] |
| Optogenetically engineered exosome system (EXPLOR) | Therapeutic carriers to deliver srikB (super-repressor IkB) to a therapeutic target used as sepsis model. | Amelioration of sepsis-induced organ injury and inhibition of secretion of proinflammatory cytokines. | [256] |
| Human placenta exosomes Maternal plasma exosomes | Therapeutic approach for gestational diabetes mellitus. | To reduce the negative impact of gestational diabetes mellitus. | [257] |
| Engineered tumor-derived exosomes | Therapeutic approach as anti-tumor agents. | Potentials usage in cancer immunotherapy. | [258] |
| Mouse adipose- derived mesenchymal stem cell exosomes | Novel therapeutic strategy for tissue injury. | Proteomic analysis of exosomes found more than 1000 protein groups with a number of biological functions, implying such exosomes might be valuable as potential therapeutic targets for tissue repair. | [259] |
| Mesenchymal stem cell-derived exosomes | Potential role in osteoarthritis regenerative medicine. | Elucidation of the inflammatory and multiple pathophysiological processes in the synovium, leading to the degradation of cartilage and bone. Potential role in cartilage repair and osteoarthritis therapy. | [260] |
| Vascular endothelial cells-derived exosomes Vascular smooth muscle cells-derived exosomes Several other cells-derived exosomes | Potential of exosomes in diagnosis, prognosis, and treatment of atherosclerosis. | Understanding of occurrence and development of cardiovascular diseases such as atherosclerosis. | [261] |
| Human urine exosomes | Potential of exosomes in diagnosis of nephropathies. | Characterization of proteolytically derived peptides that are essentially relevant to classify patients with nephropathies, cancers of the urinary tract. | [262] |
| Human urine exosomes | Potential of exosome gene expression assay as noninvasive test for prostate cancer. | Evaluation of a urinary diagnostic assay to help assess whether a prostate biopsy is warranted. | [263] |
| Human bone marrow mesenchymal stem cells-derived exosomes | Potential of exosome as treatment for severe COVID-19. | Understanding of the effect and capacity of exosomes to restore oxygenation, downregulate cytokine storm, and reconstitute immunity. | [264] |
| Human lungs epithelial cells-derived exosomes transduced with selected genes of the SARS-CoV-2 | A new strategy to demonstrate that SARS-CoV-2 RNA-containing exosomes represent an indirect route of entry into cardiomyocytes. | Understanding a potential cardiac dysfunction produced via SARS-CoV-2 RNA containing exosomes, without the need for direct viral infection. | [265] |
| Bovine milk exosomes | An alternative strategy to load hydrophilic and lipophilic small molecules, and chemotherapeutic drugs into exosomes. | Potential drug delivery vehicle or nanocarrier for cancer treatment. | [266] |
| Mesenchymal stem/stromal cell-derived exosomes | May provide considerable advantages over their counterpart live cells, potentially reducing undesirable side effects including infusional toxicities. | Potential use in gene delivery, regenerative medicine, and immunomodulation. | [267] |
| Cultured third-molar pulp cell-derived exosomes | Potential of pulp-derived exosomes in combination with fibrin gel to fill dental hard tissues | Understanding the use exosomes in combination with fibrin gel in clinical translation towards improved cell-free regenerative endodontics. | [268] |
| Tumor-derived exosomes (TEXs) | Potential for circulating immune-related biomarkers, reflecting partially the genetic and molecular contents of the parent cancer cell. | Understanding how TEXs influence boost tumor growth, regulation of tumor neo-angiogenesis, premetastatic niche formation, and therapy resistance. | [269] |
| Human exosomes-based vaccines | Potential use of the S protein of the SARS-CoV, a type I transmembrane glycoprotein, incorporated into exosomes as antigen to be used in vaccines to induce high levels of neutralizing antibodies. | Manufacturing of highly immunogenic SARS-S-based vaccines. Additionally, potential use in inhibiting tumor growth. | [270] |
| Exosomal noncoding RNA in glioma | Potential of exosomes as carriers of bioactive molecules into the brain. Holds great promise in diagnostics and therapy. Noncoding RNAs of exosomes can be modulators of numerous hallmarks of glioma. | Understanding the function of exosomal noncoding RNAs in cell-to-cell communication in the tumor microenvironment, tumor proliferation, invasion, angiogenesis, immune-scape, and treatment resistance. | [271] |
| Human exosomes in cardiovascular diseases | Potential of exosomes as more effective intervention targets on ischemic heart disease. The presence of exosomes in plaque tissue, ischemic heart, and peripheral blood can be potential biomarkers for early diagnosis and prognosis of cardiovascular diseases. | Understanding the participation of exosomes in the evolution of ischemic heart disease, including their role in endothelial dysfunction, lipid deposition, atheromatous plaque formation and rupture, ischemia-reperfusion, and heart failure. | [272] |
| Human urine exosomes | Potential source of biomarkers, pathogenic molecules, and therapeutic biologics in kidney diseases or disorders. | Understanding the role of exosomes in pathogenic mechanisms, biomarker discovery, and therapeutics of various kidney diseases, particularly lupus nephritis, acute kidney injury, diabetic nephropathy, renal fibrosis, kidney transplantation, and renal carcinoma. | [273] |
| Exosomes in inflammatory processes | Potential of exosome’s cargo, such as the novel SLC22A5 transport protein, to serve as useful biomarker of inflammatory processes. | Understanding the significance of exosomes as carriers of inflammatory mediators involved in human pathologies. | [274] |
| Human saliva exosomes from HIV-positive people | Platform to demonstrate that isolated saliva exosomes from HIV-positive individuals promote Kaposi’s sarcoma-associated herpes virus (KSHV) infectivity in human oral epithelial cells. | Understanding how the trans-activation response element (TAR) RNA in HIV-associated exosomes contribute to enhancing KSHV infectivity through the epidermal growth factor receptor (EGFR). | [275] |
| Human semen exosomes | Potential of exosome’s cargo containing molecular fingerprints for a non-invasive diagnosis of prostate cancer. | Understanding the role of semen exosomes in prostate cancer diagnosis, and as possible agents for enhancing the transmission of sexual diseases. | [276] |
| Human serum exosomes | Potential role of some miRNAs extracted from serum exosomes of Parkinson’s disease patients to serve as biomarkers. | Understanding the significance of the expression levels of miR19b, miR24, and miR195 as biomarkers of Parkinson’s disease. | [277] |
| Human milk exosomes | A complementary strategy to deliver more functional insights of human milk. | Providing an enhanced immunological and micronutrient profile of human milk, with significant relevance to breast milk quality and the health of the mother and infant. | [278] |
| Human umbilical cord exosomes | Potential role of exosomes derived from Akt-modified human umbilical cord mesenchymal stem cells as therapy for improving cardiac regeneration. | Understanding the role of why exosomes obtained from Akt-modified umbilical cord mesenchymal stem cells are more effective as therapy in myocardial infarction by promoting angiogenesis via activating platelet-derived growth factor D. | [279] |
| Human bronchoalveolar lavage fluid exosomes | Potential role of miRNA from exosomes with proinflammatory signatures in asthma and in allergic airway diseases. | Understanding the role of miRNA obtained from bronchoalveolar lavage fluid exosomes. Particularly microRNAs miR-24 and miR-27, which can modulate gene programming and promote inflammation. | [280] |
| Human cerebrospinal fluid and plasma exosomes | Potential use of alpha-synuclein and tau proteins obtained from central nervous system-derived exosomes that can efflux into blood can be used as biomarkers of Parkinson’s disease and other neurodegenerative diseases. Exosomes can also serve as carriers of therapeutic substances for diseases of the central nervous system. | Understanding the role of the content of exosomes derived from the central nervous system in Parkinson’s disease. Exosomes can carry and spread toxic alpha-synuclein between cells and induce apoptosis. | [281] |
| Human induced pluripotent stem cell-derived neuronal exosomes | Potential use as a tool to assay the capacity of exosomes to influence neuronal and circuit development. Control exosomes rescue neurodevelopmental defects in a model of Rett syndrome. | Understanding the role of exosomes in the development of neural circuits, the increase in neurogenesis, and the promotion of cell proliferation and neural differentiation. | [282] |
| Blood or cerebrospinal fluid-derived circulating circular exosomal RNAs | Potential use of circulating exosomal circular RNAs (circRNAs) as biomarkers for the early detection and diagnosis of neuropsychiatric disorders. | Understanding the role of closed-loop structure circular RNAs, a novel class of non-coding RNA (ncRNA), in mental diseases. Some studies show that circRNAs possess regulatory potential as “sponges” for target microRNAs (miRNAs) and RNA binding proteins. | [283] |
| Ovarian cancer cells-derived exosomes | Potential use of exosomal proteins and lipids in the early diagnosis of ovarian cancer. | Understanding the role several lipid species and proteins, which significantly differ in cancer derived exosomes when compared to those from ovarian surface epithelial cells. | [284] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzman, N.A.; Guzman, D.E. A Two-Dimensional Affinity Capture and Separation Mini-Platform for the Isolation, Enrichment, and Quantification of Biomarkers and Its Potential Use for Liquid Biopsy. Biomedicines 2020, 8, 255. https://doi.org/10.3390/biomedicines8080255
Guzman NA, Guzman DE. A Two-Dimensional Affinity Capture and Separation Mini-Platform for the Isolation, Enrichment, and Quantification of Biomarkers and Its Potential Use for Liquid Biopsy. Biomedicines. 2020; 8(8):255. https://doi.org/10.3390/biomedicines8080255
Chicago/Turabian StyleGuzman, Norberto A., and Daniel E. Guzman. 2020. "A Two-Dimensional Affinity Capture and Separation Mini-Platform for the Isolation, Enrichment, and Quantification of Biomarkers and Its Potential Use for Liquid Biopsy" Biomedicines 8, no. 8: 255. https://doi.org/10.3390/biomedicines8080255






