Coronavirus Pandemic—Therapy and Vaccines
Abstract
1. Introduction
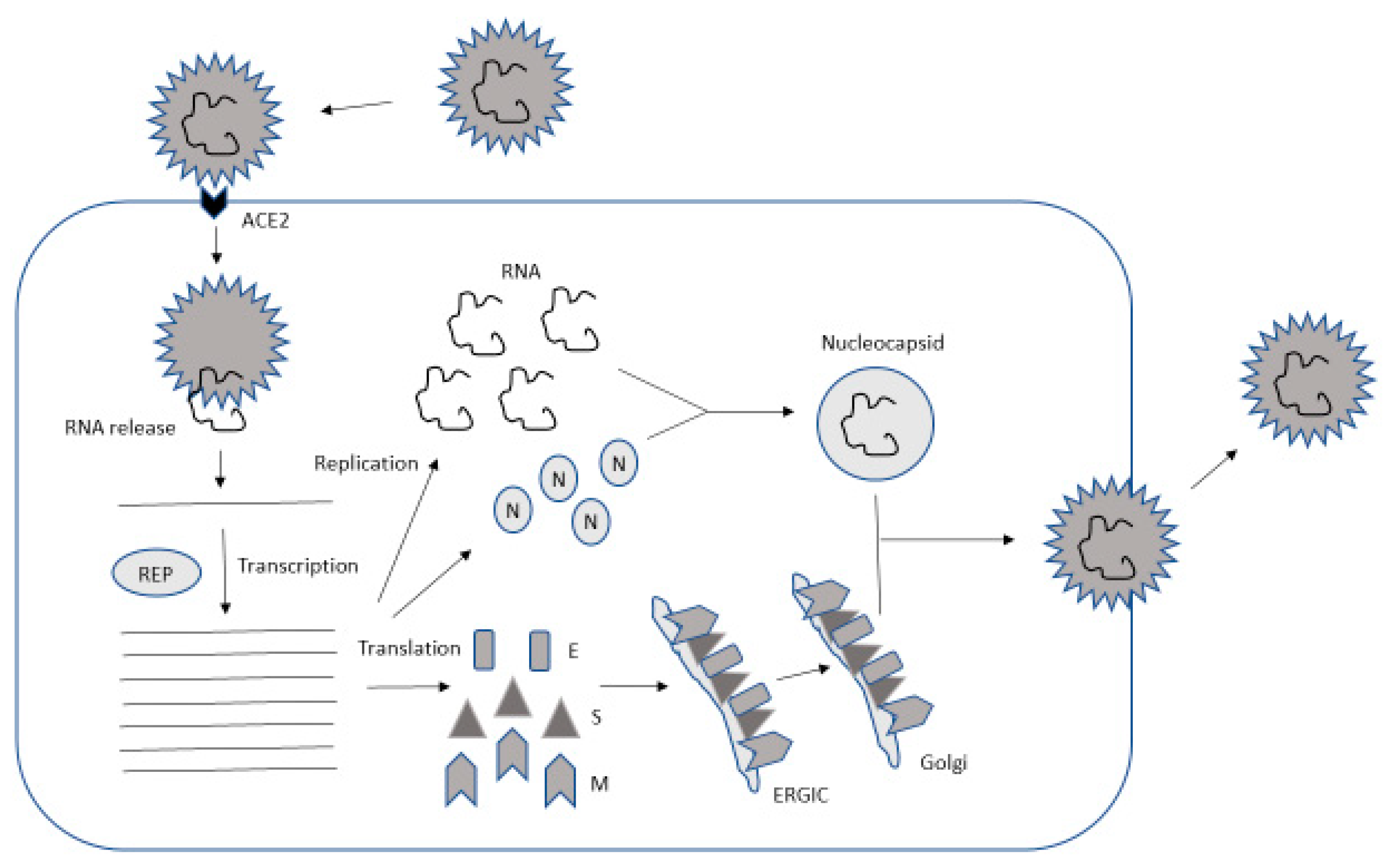
2. Coronavirus Genome and Lifecycle
3. Therapeutic and Prophylactic Options
3.1. Therapeutic Agents
| Therapy | Disease | Effect | Ref. |
|---|---|---|---|
| Lopinavir/Ritonavir | MERS-like in marmosets | Clinical, pathological benefits | [85] |
| MERS | Protocol for clinical trial | [86] | |
| COVID-19 | No difference to standard care | [87] | |
| Nucleoside | SARS-CoV | In vitro anti-SARS-CoV activity | [89] |
| analogues | HoCV-NL63, MERS-CoV | Antiviral activity, but not against SARS-CoV | [90] |
| FIP | Successful treatment of FIP | [91] | |
| Ribavirin | MERS-CoV | Improved survival at 14 days | [92] |
| Remdesivir | HCoV-OC43, HCoV-229E | Potent antiviral activity | [94] |
| MERS-CoV | Potent inhibition | [95] | |
| SARS-CoV-2 | Inhibition in Vero, Huh-7 cells | [96] | |
| SARS-CoV-2 | Reduced viral load in patients | [97] | |
| NA inhibitors | SARS-CoV | No inhibitory effect | [99] |
| Arbidol | SARS-CoV-2 | Positive effect in patients with lopinavir/ritonavir | [102] |
| Hydroxychloroquinine | SARS-CoV-2 | Reduced viral load in 20 COVID-19 patients | [103] |
| Chinese medicines | SARS-CoV-2 | Case studies of recovery from COVID-19 by SHL | [104] |
| SARS-CoV-2 | Successful SJ therapy | [105] | |
| ACE2 | SARS-CoV-2 | nAbs for compassionate therapy | [81] |
| SARS-CoV-2 | TMPRSS2 blocking virus entry | [106] | |
| SARS-CoV-2 | Potent binding of mAbs | [107] | |
| ATR1 blockers | SARS-CoV-2 | Evaluation of existing blockers | [108] |
| mAb | SARS-CoV, SARS-CoV-2 | Neutralization of SARS-CoV-2 | [109] |
3.2. Gene Silencing
3.3. Vaccine Development
| Vaccine/Vector | Disease | Effect | Ref. |
|---|---|---|---|
| Live attenuated | BCoV | High Ab titers against BCoV in calves | [131] |
| BCoV | Approved nasal vaccine in calves | [132] | |
| BCoV vaccine | ECoV | Increased Ab titers against ECoV | [133] |
| Plasmid DNA | SARS-CoV | Humoral and cellular immune responses in mice | [141] |
| DNA-CTE/PRE | SARS-CoV | nAbs, protection against SARS-CoV in mice | [142] |
| DNA/PEI NPs | SARS-CoV | NPs induced humoral and cellular responses in mice after intranasal administration | [143] |
| TCoV | Humoral response, partial TCoV protection | [144] | |
| DNA + protein | TGEV | Oral administration induced mucosal and cellular immune responses in mice | [145] |
| Lactobacillus acidophilus TGEV | MERS-CoV | nAbs, protection of mice against MERS-CoV | [146] |
| CHO/S377-588 | SARS-CoV | Overexpression of S protein in plants | [147] |
| Tobacco/lettuce | SARS-CoV | IgA Abs in mice fed with tomato-derived S | [148] |
| Tomato/tobacco | SARS-CoV | Humoral and cellular immune responses | [149] |
| Tobacco/suppressor | SARS-CoV N protein | ||
| p19 TBSV | PEDV | Immune response in mice and piglets | [150] |
| Ad-LTB-COE | MERS-CoV | Reduced viral excretion and viral RNA in dromedary camels | [151] |
| MVA-MERS-CoV S | MERS-CoV | Identification of T cell-responding epitope | [152] |
| MVA-MERS-CoV N | SARS-CoV | Strong nAbs response in mice | [153] |
| RV-SARS-CoV N/S | SARS-CoV | Protection against SARS-CoV in mice | [154] |
| VEE-SARS-CoV S | SARS-CoV | Protection also in aged mice | [155] |
4. Conclusions and Future Aspects
Funding
Conflicts of Interest
References
- De Groot, R.J.; Baker, S.C.; Baric, R.; Enjuanes, L.; Gorbalenya, A.E.; Holmes, K.V.; Perlman, S.; Poon, L.; Rottier, P.J.; Talbot, P.J.; et al. Family Coronaviridae. In Ninth Report of the International Committee on Taxonomy of Viruses; International Committee on Taxonomy of Viruses, International Union of Microbiological Societies. Virology Division; King, A.M., Lefkowitz, E., Adams, M.J., Carstens, E.B., Eds.; Elsevier: Oxford, UK, 2011; pp. 806–828. ISBN 978-0-12-384684-6. [Google Scholar]
- Fehr, A.R.; Perlman, S. Coronaviruses: An Overview of Their Replication and Pathogenesis. Methods Mol. Biol. 2015, 1282, 1–23. [Google Scholar] [PubMed]
- Doyle, L.P.; Hutchings, L.M. A transmissible gastroenteritis in pigs. J. Am. Vet. Med. Assoc. 1946, 108, 257–259. [Google Scholar] [PubMed]
- Lin, C.M.; Saif, L.J.; Marthaler, D.; Wang, Q. Evolution, antigenicity and pathogenicity of porcine epidemic diarrhea virus. Virus Res. 2016, 226, 20–39. [Google Scholar] [CrossRef] [PubMed]
- Mora-Díaz, J.C.; Piñeyro, P.E.; Houston, E.; Zimmerman, J.; Giménez-Lirola, L.G. Porcine Hemagglutinating Encephalomyelitis Virus: A Review. Front. Vet. Sci. 2019, 6, 53. [Google Scholar] [CrossRef]
- Amer, H.M. Bovine-like coronaviruses in domestic and wild ruminants. Anim. Health Res. Rev. 2018, 19, 113–124. [Google Scholar] [CrossRef]
- Perlman, S.; Netland, J. Coronaviruses post-SARS: Update on replication and pathogenesis. Nat. Rev. Microbiol. 2009, 7, 439–450. [Google Scholar] [CrossRef]
- Funk, C.J.; Manzer, R.; Miura, T.A.; Groshong, S.D.; Ito, Y.; Travanty, E.A.; Leete, J.; Holmes, K.V.; Mason, R.J. Rat respiratory coronavirus infection: Replication in airway and alveolar epithelial cells and the innate immune response. J. Gen. Virol. 2009, 90, 2956–2964. [Google Scholar] [CrossRef]
- Cavanagh, D. Coronavirus avian infectious bronchitis virus. Vet. Res. 2007, 38, 281–297. [Google Scholar] [CrossRef]
- Wolfe, L.G.; Griesemer, R.A. Feline infectious peritonitis. Pathol. Vet. 1966, 3, 255–270. [Google Scholar] [CrossRef]
- Jaimes, J.A.; Whittaker, G.A. Feline coronavirus: Insights into viral pathogenesis based on the spike protein structure and function. Virology 2018, 517, 108–121. [Google Scholar] [CrossRef]
- Izumi, T. Sarcoidosis in various organs. Clin. Dermatol. 1986, 4, 150–153. [Google Scholar] [CrossRef]
- Mihindukulasuriya, K.A.; Wu, G.; St Leger, J.; Nordhausen, R.W.; Wang, D. Identification of a novel coronavirus from a beluga whale by using a panviral microarray. J. Virol. 2008, 82, 5084–5088. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Zhang, Y.; Xu, L.; Yang, W.; Yang, F.; Feng, Y.; Xia, L.; Zhou, J.; Zhen, W.; Feng, Y.; et al. Identification of diverse alphacoronaviruses and genomic characterization of a novel severe acute respiratory syndrome-like coronavirus from bats in china. J. Virol. 2014, 88, 7070–7082. [Google Scholar] [CrossRef] [PubMed]
- Menachery, V.D.; Yont, B.L., Jr.; Debbink, K.; Agnihothram, S.; Gralinski, L.E.; Plante, J.A.; Graham, R.L.; Scobey, T.; Ge, X.Y.; Donaldson, E.F.; et al. A SARS-like cluster of bat coronaviruses shows potential for human emergence. Nat. Med. 2015, 21, 1508–1513. [Google Scholar] [CrossRef]
- Wege, H.; Siddell, S.G.; ter Meulen, V. The biology and pathogenesis of coronaviruses. Curr. Top Microbiol. Immunol. 1982, 99, 165–200. [Google Scholar]
- Lampert, P.W.; Sims, J.K.; Kniazeff, A.J. Mechanism of demyelination in JHM virus encephalomyelitis. Acta Neuropathol. 1973, 24, 76–85. [Google Scholar] [CrossRef]
- Bradburne, A.F.; Bynoe, M.L.; Tyrell, D.A.J. Effects of a “new” human respiratory virus in volunteers. Br. Med. J. 1967, 3, 767–769. [Google Scholar] [CrossRef]
- van der Hoek, L.; Sure, K.; Ihorst, G.; Stang, A.; Pyrc, K.; Jebbink, M.F.; Petersen, G.; Forster, J.; Berkhout, B.; Uberla, K. Croup is associated with the novel coronavirus NL63. PLoS Med. 2005, 2, e240. [Google Scholar] [CrossRef]
- Hamre, D.; Procknow, J.J. A new virus isolated from the human respiratory tract. Proc. Soc. Exp. Biol. Med. 1966, 121, 190–193. [Google Scholar] [CrossRef]
- Anderson, R.M.; Fraser, C.; Ghani, A.C.; Donelly, C.A.; Riley, S.; Ferguson, N.M.; Leung, G.M.; Lam, T.H.; Hedley, A.J. Epidemiology, transmission dynamics and control of SARS: The 2002–2003 epidemic. Philos. Trans. R Soc. London B Biol. Sci. 2004, 359, 1091–1105. [Google Scholar] [CrossRef]
- Cherry, J.D. The chronology of the 2002–2003 SARS mini pandemic. Paediatr. Resp. Rev. 2004, 5, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Aleanizi, F.S.; Mohmed, N.; Alqahtani, F.Y.; El Hadi Mohamed, R.A. Outbreak of Middle east respiratory syndrome coronavirus in Saudi Arabia: A retrospective study. BMC Infect. Dis. 2017, 17, 23. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Zheng, B.J.; He, Y.Q.; Liu, X.L.; Zhuang, Z.X.; Cheung, C.L.; Luo, S.W.; Li, P.H.; Zhang, L.J.; Guan, Y.J.; et al. Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China. Science 2003, 302, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Woo, P.C.; Li, K.S.; Huang, Y.; Tsoi, H.W.; Wong, B.H.; Wong, S.S.; Leung, S.Y.; Chan, K.H.; Yuen, K.Y. Severe acute respiratory syndrome coronavirus-like virus in Chinese horseshoe bats. Proc. Natl. Acad. Sci. USA 2005, 102, 14040–14045. [Google Scholar] [CrossRef]
- Li, W.; Shi, Z.; Yu, M.; Ren, W.; Smith, C.; Epstein, J.H.; Wang, H.; Crameri, G.; Hu, Z.; Zhang, H.; et al. Bats are natural reservoirs of SARS-like coronaviruses. Science 2005, 310, 676–679. [Google Scholar] [CrossRef]
- Ge, X.Y.; Li, J.L.; Yang, X.L.; Chmura, A.A.; Zhu, G.; Epstein, J.H.; Mazet, J.K.; Hu, B.; Zhang, W.; Peng, C.; et al. Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor. Nature 2013, 503, 535–538. [Google Scholar] [CrossRef]
- Peiris, J.S.; Yuen, K.Y.; Osterhaus, A.D.; Stohr, K. The severe acute respiratory syndrome. N. Engl. J. Med. 2003, 349, 2431–2441. [Google Scholar] [CrossRef]
- Zaki, A.M.; van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef]
- van Boheemen, S.; de Graaf, M.; Lauber, C.; Bestebroer, T.M.; Raj, V.S.; Zaki, A.M.; Osterhaus, A.D.; Haagmans, B.L.; Gorbalenya, A.E.; Snijder, E.J.; et al. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. mBio 2012, 3, e00473-12. [Google Scholar] [CrossRef]
- Chibo, D.; Birch, C. Analysis of human coronavirus 229E spike and nucleoprotein genes demonstrates genetic drift between chronologically distinct strains. J. Gen. Virol. 2006, 87, 1203–1208. [Google Scholar] [CrossRef]
- Vijgen, L.; Keyaerts, E.; Lemey, P.; Moes, E.; Li, S.; Vandamme, A.M.; Van Ranst, M. Circulation of genetically distinct contemporary human coronavirus OC43 strains. Virology 2005, 337, 85–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meyer, B.; Muller, M.A.; Corman, V.M.; Reusken, C.B.; Ritz, D.; Godeke, G.J.; Lattwein, E.; Kallies, S.; Siemens, A.; van Beek, J.; et al. Antibodies against MERS coronavirus in dromedary camels, United Arab Emirates, 2003 and 2013. Emerg. Infect. Dis. 2014, 20, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Eckerle, I.; Corman, V.M.; Muller, M.A.; Lenk, M.; Ulrich, R.G.; Drosten, C. Replicative Capacity of MERS Coronavirus in Livestock Cell Lines. Emerg. Infect. Dis. 2014, 20, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Azhar, E.I.; El-Kafrawy, S.A.; Farraj, S.A.; Hassan, A.M.; Al-Saeed, M.S.; Hashem, A.M.; Madani, T.A. Evidence for Camel-to-Human Transmission of MERS Coronavirus. N. Engl. J. Med. 2014, 370, 2499–2505. [Google Scholar] [CrossRef]
- Raj, V.S.; Mou, H.; Smits, S.L.; Dekkers, D.H.; Muller, M.A.; Dijkman, R.; Muth, D.; Demmers, J.A.; Zaki, A.; Fouchier, R.A.; et al. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature 2013, 495, 251–254. [Google Scholar] [CrossRef]
- Zhao, J.; Li, K.; Wohlford-Lenane, C.; Agnihothram, S.S.; Fett, C.; Zhao, J.; Gale, M.J., Jr.; Baric, R.S.; Enjuanes, L.; Gallagher, T.; et al. Rapid generation of a mouse model for Middle East respiratory syndrome. Proc. Natl. Acad. Sci. USA 2014, 111, 4970–4975. [Google Scholar] [CrossRef]
- Kaplan, E.H. Containing 2019-nCoV (Wuhan) coronavirus. Health Care Manag. Sci. 2020. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, F.; Wang, R.; Guan, K.; Jiang, T.; Xu, G.; Sun, J.; Chang, C. The deadly coronavirus; The 2003 SARS epidemic and the 2020 novel coronavirus epidemic in China. J. Autoimmun. 2020. [Google Scholar] [CrossRef]
- Global Health Policy. Available online: https://www.google.com/covid19-map (accessed on 28 April 2020).
- Han, Q.; Lin, Q.; Jin, S.; You, L. Coronavirus 2019-nCoV: A brief perspective from the front line. J. Infect. 2020. [Google Scholar] [CrossRef]
- Kahn, J.; McIntosh, K. History and recent advances in coronavirus discovery. Ped. Infect. Dis. J. 2005, 24, s223–s227. [Google Scholar] [CrossRef]
- Zhao, L.; Jha, B.K.; Wu, A.; Elliott, R.; Ziebuhr, J.; Gorbalenya, A.E.; Silverman, R.H.; Weiss, S.R. Antagonism of the interferon-induced OAS-RNase L pathway by murine coronavirus ns2 protein is required for virus replication and liver pathology. Cell. Host Microbe 2012, 11, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Barcena, M.; Oostergetel, G.T.; Bartelink, W.; Faas, F.G.; Verkleij, A.; Rottier, P.J.; Koster, A.J.; Bosch, B.J. Cryo-electron tomography of mouse hepatitis virus: Insights into the structure of the coronavirion. Proc. Natl. Acad. Sci. USA 2009, 106, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Neuman, B.W.; Adair, B.D.; Yoshioka, C.; Quispe, J.D.; Orca, G.; Kuhn, P.; Milligan, R.A.; Yeager, M.; Buchmeier, M.J. Supramolecular architecture of severe acute respiratory syndrome coronavirus revealed by electron cryomicroscopy. J. Virol. 2006, 80, 7918–7928. [Google Scholar] [CrossRef] [PubMed]
- de Groot, R.J.; Luytjes, W.; Horzinek, M.C.; van der Zeijst, B.A.; Spaan, W.J.; Lenstra, J.A. Evidence for a coiled-coil structure in the spike proteins of coronaviruses. J. Mol. Biol. 1987, 196, 963–966. [Google Scholar] [CrossRef]
- Stohlman, S.A.; Baric, R.S.; Nelson, G.N.; Soe, L.H.; Welter, L.M.; Deans, R.J. Specific interaction between coronavirus leader RNA and nucleocapsid protein. J. Virol. 1988, 62, 4288–4295. [Google Scholar] [CrossRef] [PubMed]
- Molenkamp, R.; Spaan, W.J. Identification of a specific interaction between the coronavirus mouse hepatitis virus A59 nucleocapsid protein and packaging signal. Virology 1997, 239, 78–86. [Google Scholar] [CrossRef]
- Kuo, L.; Masters, P.S. Functional analysis of the murine coronavirus genomic RNA packaging signal. J. Virol. 2013, 87, 5182–5192. [Google Scholar] [CrossRef]
- Neuman, B.W.; Kiss, G.; Kunding, A.H.; Bhella, D.; Baksh, M.F.; Connelly, S.; Droese, B.; Klaus, J.P.; Makino, S.; Sawicki, S.G.; et al. A structural analysis of M protein in coronavirus assembly and morphology. J. Struct. Biol. 2011, 174, 11–22. [Google Scholar] [CrossRef]
- Armstrong, J.; Niemann, H.; Smeekens, S.; Rottier, P.; Warren, G. Sequence and topology of a model intracellular membrane protein, E1 glycoprotein, from a coronavirus. Nature 1984, 308, 751–752. [Google Scholar] [CrossRef]
- Godet, M.; L’Haridon, R.; Vautherot, J.F.; Laude, H. TGEV corona virus ORF4 encodes a membrane protein that is incorporated into virions. Virology 1992, 188, 666–675. [Google Scholar] [CrossRef]
- Nieto-Torres, J.L.; Dediego, M.L.; Verdia-Baguena, C.; Jimenez-Guardeno, J.M.; Regla-Nava, J.A.; Fernandez-Delgado, R.; Castano-Rodriguez, C.; Alcaraz, A.; Torres, J.; Aguilella, V.M.; et al. Severe acute respiratory syndrome coronavirus envelope protein ion channel activity promotes virus fitness and pathogenesis. PLoS Pathogens 2014, 10, 1004077. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R.; Knobler, R.L.; Powell, H.; Buchmeier, M.J. Monoclonal antibodies to murine hepatitis virus-4 (strain JHM) define the viral glycoprotein responsible for attachment and cell-cell fusion. Virology 1982, 119, 358–371. [Google Scholar] [CrossRef]
- Abraham, S.; Kienzle, T.E.; Lapps, W.; Brian, D.A. Deduced sequence of the bovine coronavirus spike protein and identification of the internal proteolytic cleavage site. Virology 1990, 176, 296–301. [Google Scholar] [CrossRef]
- Luytjes, W.; Sturman, L.S.; Bredenbeek, P.J.; Charite, J.; van der Zeijst, B.A.; Horzinek, M.C.; Spaan, W.J. Primary structure of the glycoprotein E2 of coronavirus MHV-A59 and identification of the trypsin cleavage site. Virology 1987, 161, 479–487. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020. [Google Scholar] [CrossRef] [PubMed]
- Klausegger, A.; Strobl, B.; Regl, G.; Kaser, A.; Luytjes, W.; Vlasak, R. Identification of a coronavirus hemagglutinin-esterase with a substrate specificity different from those of influenza C virus and bovine coronavirus. J. Virol. 1999, 73, 3737–3743. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, L.A.; Wierda, C.M.; van der Meer, F.J.; Herrewegh, A.A.; Horzinek, M.C.; Egberink, H.F.; de Groot, R.J. Hemagglutinin-esterase, a novel structural protein of torovirus. J. Virol. 1997, 71, 5277–5286. [Google Scholar] [CrossRef]
- Kazi, L.; Lissenberg, A.; Watson, R.; de Groot, R.J.; Weiss, S.R. Expression of hemagglutinin esterase protein from recombinant mouse hepatitis virus enhances neurovirulence. J. Virol. 2005, 79, 15064–15073. [Google Scholar] [CrossRef]
- Kubo, H.; Yamada, Y.K.; Taguchi, F. Localization of neutralizing epitopes and the receptor-binding site within the amino-terminal 330 amino acids of the murine coronavirus spike protein. J. Virol. 1994, 68, 5403–5410. [Google Scholar] [CrossRef]
- Cheng, P.K.; Wong, D.A.; Tong, L.K.; Ip, S.M.; Lo, A.C.; Lau, C.S.; Yeung, E.Y.; Lim, W.W. Viral shedding patterns of coronavirus in patients with probable severe acute respiratory syndrome. Lancet 2004, 363, 1699–1700. [Google Scholar] [CrossRef]
- Nedellec, P.; Dveksler, G.S.; Daniels, E.; Turbide, C.; Chow, B.; Basile, A.A.; Holmes, K.V.; Beauchemin, N. Bgp2, a new member of the carcinoembryonic antigen-related gene family, encodes an alternative receptor for mouse hepatitis viruses. J. Virol. 1994, 68, 4525–4537. [Google Scholar] [CrossRef] [PubMed]
- van Doremalen, N.; Miazgowicz, K.L.; Milne-Price, S.; Bushmaker, T.; Robertson, S.; Scott, D.; Kinne, J.; McLellan, J.S.; Zhu, J.; Munster, V.J. Host species restriction of Middle East respiratory syndrome coronavirus through its receptor dipeptidyl peptidase 4. J. Virol. 2014, 88, 9220–9232. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.; Rottier, P.J. The coronavirus spike protein is a class I virus fusion protein: Structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef]
- Belouzard, S.; Chu, V.C.; Whittaker, G.R. Activation of the SARS coronavirus spike protein via sequential proteolytic cleavage at two distinct sites. Proc. Natl. Acad. Sci. USA 2009, 106, 5871–5876. [Google Scholar] [CrossRef]
- Baranov, P.V.; Henderson, C.M.; Anderson, C.B.; Gesteland, R.F.; Atkins, J.F.; Howard, M.T. Programmed ribosomal frameshifting in decoding the SARS-CoV genome. Virology 2005, 332, 498–510. [Google Scholar] [CrossRef]
- Brierley, I.; Digard, P.; Inglis, S.C. Characterization of an efficient coronavirus ribosomal frameshifting signal: Requirement for an RNA pseudoknot. Cell 1989, 57, 537–547. [Google Scholar] [CrossRef]
- Brown, C.G.; Nixon, K.S.; Senanayake, S.D.; Brian, D.A. An RNA stem-loop within the bovine coronavirus nsp1 coding region is a cis-acting element in defective interfering RNA replication. J. Virol. 2007, 81, 7716–7724. [Google Scholar] [CrossRef]
- Guan, B.J.; Wu, H.Y.; Brian, D.A. An optimal cis-replication stem-loop IV in the 5’ untranslated region of the mouse coronavirus genome extends 16 nucleotides into open reading frame 1. J. Virol. 2011, 85, 5593–5605. [Google Scholar] [CrossRef]
- Liu, P.; Li, L.; Keane, S.C.; Yang, D.; Leibowitz, J.L.; Giedroc, D.P. Mouse hepatitis virus stem-loop 2 adopts a Uynmg (U)a-like tetraloop structure that is highly functionally tolerant of base substitutions. J. Virol. 2009, 83, 12084–12093. [Google Scholar] [CrossRef]
- Madhugiri, R.; Fricke, M.; Marz, M.; Ziebuhr, J. Coronavirus cis-Acting RNA Elements. Adv. Virus Res. 2016, 96, 127–163. [Google Scholar] [PubMed]
- Raman, S.; Bouma, P.; Williams, G.D.; Brian, D.A. Stem-loop III in the 5’ untranslated region is a cisacting element in bovine coronavirus defective interfering RNA replication. J. Virol. 2003, 77, 6720–6730. [Google Scholar] [CrossRef] [PubMed]
- Sethna, P.B.; Hofmann, M.A.; Brian, D.A. Minus-strand copies of replicating coronavirus mRNAs contain antileaders. J. Virol. 1991, 65, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, S.G.; Sawicki, D.L.; Siddell, S.G. A contemporary view of coronavirus transcription. J. Virol. 2007, 81, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Krijnse-Locker, J.; Ericsson, M.; Rottier, P.J.M.; Griffiths, G. Characterization of the budding compartment of mouse hepatitis virus: Evidence that transport from the RER to the golgi complex requires only one vesicular transport step. J. Cell Biol. 1994, 124, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Tooze, J.; Tooze, S.; Warren, G. Replication of coronavirus MHV-A59 in sac- cells: Determination of the first site of budding of progeny virions. Eur. J. Cell Biol. 1984, 33, 281–293. [Google Scholar]
- de Haan, C.A.; Rottier, P.J. Molecular interactions in the assembly of coronaviruses. Adv. Virus Res. 2005, 64, 165–230. [Google Scholar]
- Lu, H. Drug treatment options for the 2019-new coronavirus (2019.nCoV). Biosci. Trends. 2020. [Google Scholar] [CrossRef]
- Kruse, R.L. Therapeutic strategies in an outbreak scenario to treat the novel coronavirus originating in Wuhan, China. F1000Res 2020, 9, 72. [Google Scholar] [CrossRef]
- Wu, C.J.; Chan, Y.L. Antiviral applications of RNAi for coronavirus. Exp. Opin. Investig. Drugs 2006, 15, 89–97. [Google Scholar] [CrossRef]
- Enjuanes, L.; Zuniga, S.; Castano-Rodriguez, C.; Gutierrez-Alvarez, J.; Canton, J.; Sola, I. Molecular Basis of Coronavirus Virulence and Vaccine Development. Adv. Virus Res. 2016, 95, 245–286. [Google Scholar]
- Croxtall, J.D.; Perry, C.M. Lopinavir/Ritonavir: A review of its use in the management of HIV-1 infection. Drugs 2010, 70, 1885–1915. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.; Yao, Y.; Yeung, M.L.; Deng, W.; Bao, L.; Jia, L.; Li, F.; Xiao, C.; Gao, H.; Yu, P.; et al. Treatment with Lopinavir/Ritonavir or Interferon-β1b Improves Outcome of MERS-CoV Infection in a Nonhuman Primate Model of Common Marmoset. J. Infect. Dis. 2015, 212, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Alothman, A.; Balkhy, H.H.; Al-Dawood, A.; Al Johani, S.; Al Harbi, S.; Kojan, S.; Al Jeraisy, M.; Deeb, A.M.; Assiri, A.M. Treatment of Middle East Respiratory Syndrome with a combination of lopinavir-ritonavir and interferon-β1b (MIRACLE) trial: Study protocol for a randomized controlled trial. Trials 2018, 19, 81. [Google Scholar] [CrossRef]
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruang, L.; Song, B.; Cai, Y.; Wei, M.; et al. A trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Prujisssers, A.J.; Denison, M.R. Nucleoside analogues for the treatment of coronavirus infections. Curr. Opin. Virol. 2019, 35, 57–62. [Google Scholar] [CrossRef]
- Chu, C.K.; Gadthula, S.; Chen, X.; Choo, H.; Olgen, S.; Barnard, D.L.; Sidwell, R.W. Antiviral activity of nucleoside analogues against SARS-coronavirus (SARS-CoV). Antivir. Chem. Chemother. 2006, 17, 285–289. [Google Scholar] [CrossRef]
- Peters, H.L.; Jochmans, D.; de Wilde, A.H.; Posthuma, C.C.; Snijder, E.J.; Neyts, J.; Seley-Radtke, K.L. Design, synthesis and evaluation of a series of acyclic fleximer nucleoside analogues with anti-coronavirus activity. Bioorg. Med. Chem. Lett. 2015, 25, 2923–2926. [Google Scholar] [CrossRef]
- Murphy, B.G.; Perron, M.; Murakami, E.; Bauer, K.; Park, Y.; Eckstrand, C.; Liepnieks, M.; Pedersen, N.C. The nucleoside analog GS-441524 strongly inhibits feline infectious peritonitis (FIP) virus in tissue culture and in experimental cat infection studies. Vet. Microbiol. 2018, 219, 226–233. [Google Scholar] [CrossRef]
- Omrani, A.S.; Saad, M.M.; Baig, K.; Bahloul, A.; Abdul-Matin, M.; Alaidaroos, A.Y.; Almakhlafi, G.A.; Albarrak, M.M.; Memish, Z.A.; Albarrak, A.M. Ribavirin and interferon alpha-2a for severe Middle East respiratory syndrome coronavirus infection: A retrospective cohort study. Lancet Infect. Dis. 2014, 14, 1090–1095. [Google Scholar] [CrossRef]
- Lo, M.K.; Jordan, R.; Arvey, A.; Sudhamsu, J.; Shrivastava-Ranjan, P.; Hotard, A.L.; Flint, M.; McMullan, L.K.; Siegel, D.; Clarke, M.O.; et al. GS-5734 and its parent nucleoside analog inhibit Filo-, Pneumo and Paramyxoviruses. Sci. Rep. 2017, 7, 43395. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Won, J.J.; Graham, R.L.; Dinnon, K.H., 3rd; Sims, A.C.; Feng, J.Y.; Cihlar, T.; Denison, M.R.; Baric, R.S.; Sheahan, T.P. Broad spectrum antiviral remdesivir inhibits human endemic and zoonotic deltacoronaviruses with a highly divergent RNA dependent RNA polymerase. Antiviral Res. 2019, 169, 104541. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.J.; Tchesnokov, E.P.; Feng, J.Y.; Porter, D.P.; Gotte, M. The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus. J. Biol. Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.-C.; Rolain, J.-M.; Lee, N.-Y.; Chen, P.L.; Huang, C.-T.; Lee, P.-I.; Hsueh, P.-R. Arguments in favour of remdesivir for treating SARS-CoV-2 infections. Int. J. Antimicrob. Agents 2020. [Google Scholar] [CrossRef] [PubMed]
- Laborda, P.; Wang, S.Y.; Voglmeir, J. Influenza Neuraminidase Inhibitors: Synthetic Approaches, Derivatives and Biological Activity. Molecules 2016, 21, 1513. [Google Scholar] [CrossRef]
- Tan, E.L.C.; Eong Ooi, E.; Lin, C.-Y.; Cheng Tan, H.; Ling, A.E.; Lim, B.; Stanton, L.W. Inhibition of SARS Coronavirus Infection In Vitro with Clinically Approved Drugs. Emerg. Inf. Dis. 2004, 10, 581–586. [Google Scholar] [CrossRef]
- Haviemik, J.; Stefanik, M.; Foitikova, M.; Kali, S.; Tordo, N.; Rudolf, I.; Hubalek, Z.; Eyer, L.; Ruzek, D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses 2018, 10, 184. [Google Scholar] [CrossRef]
- Fink, S.L.; Voitech, L.; Wagoner, J.; Slivinski, N.S.J.; Jackson, K.J.; Wang, R.; Khadka, S.; Luthra, P.; Basler, C.F.; Polyak, S.J. The Antiviral Drug Arbidol Inhibits Zika Virus. Sci. Rep. 2018, 8, 8989. [Google Scholar] [CrossRef]
- Deng, L.; Li, C.; Zeng, Q.; Liu, X.; Li, X.; Zhang, H.; Hong, Z.; Xia, J. Arbidol combined with LPV/r versus LPV/r alone against Corona Virus Disease 2019: A retrospective cohort study. J. Infect. 2020. [Google Scholar] [CrossRef]
- Gautret, P.; Lagier, J.-C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, E.V.; et al. Hydroxychloroquine and azothromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents 2020. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Zhou, L.; Zhou, M.; Zhao, J.; Wang, D.W. Combination of western medicine and Chinese traditional patent medicine in treating a family case of COVID-19 in Wuhan. Front. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, X.; Lu, Y.; Chen, F.; Zhang, W. Clinical characteristics and therapeutic procedure for four cases with 2019 novel coronavirus pneumonia receiving combined Chinese and Western medicine treatment. Biosci. Trends 2020, 14, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Li, C.; Huang, A.; Xia, S.; Lu, S.; Shi, Z.; Lu, L.; Jiang, S.; Yang, Z.; Wu, Y.; et al. Potent binding of novel 2019 coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody. Emerg. Mircobes. Infect. 2020, 9, 382–385. [Google Scholar] [CrossRef] [PubMed]
- Gurwitz, D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev. Res. 2020. [Google Scholar] [CrossRef]
- Wang, C.; Li, W.; Drabek, D.; Okba, N.M.A.; van Haperen, R.; Osterhaus, A.D.; van Kupperveld, F.; Haagmans, B.L.; Grosveld, F.; Bosch, B.-J. A human monoclonal antibody blocking SARS-CoV infection. bioRxiv 2020. preprint. [Google Scholar] [CrossRef]
- Vincent, M.J.; Bergeron, E.; Benjannet, S.; Erickson, B.R.; Rollin, P.E.; Ksiazek, T.G.; Seidah, G.S.; Nichol, S.T. Chlkoroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005, 2, 69. [Google Scholar] [CrossRef]
- Colson, P.; Rolain, J.-M.; Raoult, D. Chloroquine for the 2019 novel coronavirus SARS-CoV-2. Int. J. Antimicrob. Agents 2020, 55, 105923. [Google Scholar] [CrossRef]
- Zhou, D.; Dai, S.M.; Tong, Q. COVID-19: A recommendation to examine the effect of hydrochloroquine in preventing infection and progression. J. Antimicrob. Chemother. 2020, 114. [Google Scholar] [CrossRef]
- Di Pierro, F.; Bertuccioli, A.; Cavecchia, I. Possible therapeutic role of a highly standardized mixture of active compounds derived from cultured Lentinula edodes mycelia (AHCC) in patients infected with 2019 novel coronavirus. Minerva. Gastoenterol. Dietol. 2020. [Google Scholar] [CrossRef]
- Ding, Y.; Zeng, L.; Li, R.; Chen, Q.; Zhou, B.; Chen, Q.; Cheng, P.L.; Yutao, W.; Zheng, J.; Yang, Z. The Chinese prescription lianhuaqingwen capsule exerts anti-influenza activity through the inhibition of viral propagation and impacts immune function. BMC Complement Altern. Med. 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Moen, M.D.; Wagstaff, A.J. Losartan: A review of its use in stroke risk reduction in patients with hypertension and left ventricular hypertrophy. Drugs 2005, 65, 2657–2674. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V.; The, C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complimentary to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Lundstrom, K. Micro-RNA in disease and gene therapy. Curr. Drug Discov. Technol. 2011, 8, 76–86. [Google Scholar] [CrossRef]
- Shi, Y. Mammalian RNAi for the masses. Trends Genet. 2003, 19, 9–12. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, T.; Fu, L.; Yu, C.; Li, Y.; Xu, X.; Wang, Y.; Ning, H.; Zhang, S.; Chen, W.; et al. Silencing SARS-CoV Spike protein expression in cultured cells by RNA interference. FEBS Lett. 2004, 560, 141–146. [Google Scholar] [CrossRef]
- Zheng, B.J.; Guan, Y.; Tang, Q.; Du, C.; Xie, F.Y.; He, M.L.; Chan, K.W.; Wong, K.L.; Lader, E.; Woodle, M.C.; et al. Prophylactic and therapeutic effects of small interfering RNA targeting SARS-coronavirus. Antivir. Res. 2004, 9, 365–374. [Google Scholar]
- Chang, Z.; Hu, J. Can siRNA conquer SARS? Gene Ther. 2006, 13, 871–872. [Google Scholar] [CrossRef]
- Lu, C.Y.; Huang, H.-Y.; Yang, T.H.; Chang, L.Y.; Lee, C.Y.; Huang, L.M. siRNA silencing of angiotensin-converting enzyme 2 reduced severe acute respiratory syndrome-associated coronavirus replications in Vero cells. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 709–715. [Google Scholar] [CrossRef][Green Version]
- Millet, J.K.; Nal, B. Investigation of the functional roles of host cell proteins involved in coronavirus infection using highly specific and scalable RNA interference (RNAi) approach. Methods Mol. Biol. 2015, 1282, 231–240. [Google Scholar] [PubMed]
- Nur, S.M.; Hasan, M.A.; Amin, M.A.; Hossain, M.; Sharmin, T. Design of Potential RNAi (miRNA and siRNA) Molecules for Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Gene Silencing by Computational Method. Interdiscip. Sci. 2015, 7, 257–265. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, P.; Sheehy, P.A.; Norris, J.M. Combination siRNA therapy against feline coronavirus can delay the emergence of antiviral resistance in vitro. Vet. Microbiol. 2015, 176, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.Y.; Li, Y.; Liu, B.J.; Wang, J.; Yuan, G.F.; Chen, S.J.; Zuo, Y.Z.; Fan, J.H. Short hairpin RNAs targeting M and N genes reduce replication of porcine deltacoronavirus in ST cells. Virus Genes 2019, 55, 795–801. [Google Scholar] [CrossRef]
- Li, K.; Li, H.; Bi, Z.; Song, D.; Zhang, F.; Lei, D.; Luo, S.; Li, Z.; Gong, W.; Huang, D.; et al. Significant inhibition of re-emerged and emerging swine enteric coronavirus in vitro using the multiple shRNA expression vector. Antiviral. Res. 2019, 166, 11–18. [Google Scholar] [CrossRef]
- Sohrab, S.S.; El-Kafrawy, S.A.; Mirza, Z.; Kamal, M.A.; Azhar, E.L. Design and Delivery of Therapeutic siRNAs: Application to MERS-Coronavirus. Curr. Pharm. Des. 2018, 24, 62–77. [Google Scholar] [CrossRef]
- Delrue, I.; Verzele, D.; Madder, A.; Nauwynck, H.J. Inactivated virus vaccines: From chemistry to prophylaxis: Merits, risks and challenges. Expert Rev. Vaccines 2012, 11, 695–719. [Google Scholar] [CrossRef]
- Deng, M.P.; Hu, Z.H.; Wang, H.L.; Deng, F. Developments of subunit and VLP vaccines against influenza A virus. Virol. Sin. 2012, 27, 145–153. [Google Scholar] [CrossRef]
- Decaro, N.; Campolo, M.; Mari, V.; Desario, C.; Colaianni, M.L.; Di Trani, L.; Cordioli, P.; Buonavoglia, C. A candidate modified-live bovine coronavirus vaccine: Safety and immunogenicity evaluation. New Microbiol. 2009, 32, 109–132. [Google Scholar]
- Bovilis® Coronavirus. Available online: https://www.merck-animal-health-usa.com/product/cattle/Bovilis-Coronavirus/1 (accessed on 25 March 2020).
- Nemoto, M.; Kanno, T.; Bannai, H.; Tsujimura, K.; Yamanaka, T.; Kokado, H. Antibody response to equine coronavirus in horses inoculated with a bovine coronavirus vaccine. Vet. Med. Sci. 2017, 79, 1889–1891. [Google Scholar] [CrossRef]
- Ibrahim, H.S.; Kafi, S.K. A Computational Vaccine Design Approach for MESR-CoV Infections. Methods Mol. Biol. 2020, 2131, 39–145. [Google Scholar] [PubMed]
- Battacharaya, M.; Sharma, A.R.; Patra, P.; Ghosh, P.; Sharma, G.; Patra, B.C.; Lee, S.S.; Chakraborty, C. Development of epitope-based peptide vaccine against novel coronavirus 2019 (SARS-CoV-2): Immunoinformatics approach. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tahir, U.I.; Qamar, M.; Saleem, S.; Ashfaq, U.A.; Bari, A.; Anwar, F.; Alqatani, S. Epitope-based peptide vaccine design and target site depiction against Middle East Respiratory Syndrome Coronavirus: An immune-informatics study. J. Transl. Med. 2019, 17, 362. [Google Scholar] [CrossRef]
- Srivastava, S.; Kamthania, M.; Singh, S.; Saxena, A.K.; Sharma, N. Structural basis of development of multi-epitope vaccine against Middle East respiratory syndrome using in silico approach. Infect. Drug Resist. 2018, 11, 2377–2391. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Rosen, O.; Wang, L.; Turner, H.L.; Stevens, L.J.; Corbett, K.S.; Bowman, C.A.; Pallesen, J.; Shi, W.; Zhang, Y.; et al. Structural Definition of a Neutralization-Sensitive Epitope on the MERS-CoV S1-NTD. Cell Rep. 2019, 28, 3395–3405. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Tortorici, M.A.; Frenz, B.; Snjider, J.; Li, W.; Rey, F.A.; DiMaio, F.; Bosch, B.J.; Veesler, D. Glycan shield and epitope masking of a coronavirus spike protein observed by cryo-electron microscopy. Nat. Struct. Mol. Biol. 2016, 23, 899–905. [Google Scholar] [CrossRef]
- Ahmed, S.F.; Quadeer, A.A.; McKay, M.R. Preliminary Identification of Potential Vaccine Targets for the COVID-19 Coronavirus (SARS-CoV-2) Based on SARS-CoV Immunological Studies. Viruses 2020, 12, 254. [Google Scholar] [CrossRef]
- Zakhartchouk, A.N.; Viswanathan, N.; Moshynskyy, I.; Petric, M.; Babiuk, L.A. Optimization of a DNA vaccine against SARS. DNA Cell Biol. 2007, 26, 721–726. [Google Scholar] [CrossRef]
- Callendret, B.; Lorin, V.; Charneau, P.; Marianneau, P.; Contamin, H.; Betton, J.M.; van der Werf, S.; Escriou, N. Heterologous viral RNA export elements improve expression of severe acute respiratory syndrome (SARS) coronavirus spike protein and protective efficacy of DNA vaccines against SARS. Virology 2007, 363, 288–302. [Google Scholar] [CrossRef]
- Shim, B.S.; Park, S.M.; Quan, J.S.; Jere, D.; Chu, H.; Song, M.K.; Kim, D.W.; Jang, Y.S.; Yang, M.S.; Han, S.H.; et al. Intranasal immunization with plasmid DNA encoding spike protein of SARS-coronavirus/polyethylenimine nanoparticles elicits antigen-specific humoral and cellular immune responses. BMC Immunol. 2010, 11, 65. [Google Scholar] [CrossRef]
- Chen, Y.N.; Wu, C.C.; Yeo, Y.; Xu, P.; Lin, T.L. A DNA prime-protein boost vaccination strategy targeting turkey coronavirus spike protein fragment containing neutralizing epitope against infectious challenge. Vet. Immunol. Immunopathol. 2013, 152, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Tian, Y.; Zhang, Y.; Zhang, M.; Tian, F.; Ma, J.; Jiang, S. Efficacy and immunogenicity of a live L. acidophilus expressing SAD epitope of transmissible gastroenteritis virus as an oral vaccine. Acta Virol. 2019, 63, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Nyon, M.P.; Du, L.; Tseng, C.K.; Seid, C.A.; Pollet, J.; Naceanceno, K.S.; Agrawal, A.; Algaissi, A.; Peng, B.H.; Tai, W.; et al. Engineering a stable CHO cell line for the expression of a MERS-coronavirus vaccine antigen. Vaccine 2018, 36, 1853–1862. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Y.; Ramalingam, S.; Chye, M.L. Accumulation of recombinant SARS-CoV spike protein in cytosol and chloroplasts indicate potential for development of plant-derived oral vaccines. Exp. Biol. Med. 2006, 231, 1346–1352. [Google Scholar] [CrossRef]
- Pogrebnyak, N.; Golovkin, M.; Andrianov, V.; Spitsin, S.; Smirnov, Y.; Egolf, R.; Koprowski, H. Severe acute respiratory syndrome (SARS) S protein production in plants: Development of recombinant vaccine. Proc. Natl. Acad. Sci. USA 2005, 102, 9062–9067. [Google Scholar] [CrossRef]
- Zheng, N.; Xia, R.; Yang, C.; Yin, B.; Li, Y.; Duan, C.; Liang, L.; Guo, H.; Xie, Q. Boosted expression of the SARS-CoV nucleocapsid protein in tobacco and its immunogenicity in mice. Vaccine 2009, 27, 5001–5007. [Google Scholar] [CrossRef]
- Do, V.T.; Jang, J.; Park, J.; Dao, H.T.; Kim, K.; Hahn, T.W. Recombinant adenovirus carrying a core neutralizing epitope of porcine epidemic diarrhea virus and heat-labile enterotoxin B of Escherichia coli as a mucosal vaccine. Arch. Virol. 2020, 165, 609–618. [Google Scholar] [CrossRef]
- Haagmans, B.L.; van den Brand, J.M.; Raj, V.S.; Volz, A.; Wohlsein, P.; Smits, S.L.; Schipper, D.; Bestebroer, T.M.; Okba, N.; Fux, R.; et al. An orthopoxvirus-based vaccine reduces virus excretion after MERS-CoV infection in dromedary camels. Science 2016, 351, 77–81. [Google Scholar] [CrossRef]
- Veit, S.; Jany, S.; Fux, R.; Sutter, G.; Volz, A. CD8+ T Cells Responding to the Middle East Respiratory Syndrome Coronavirus Nucleocapsid Delivered by Vaccinia Virus MVA in Mice. Viruses 2018, 10, 718. [Google Scholar] [CrossRef]
- Faber, M.; Lamirande, E.W.; Roberts, A.; Rice, A.B.; Koprowski, H.; Dietschold, B.; Schnell, M.J. A single imunization with a rhabdovirus-based vector expressing severe acute respiratory syndrome coronavirus (SARS-CoV) S protein results in the production of high levels of SARS-CoV-neutralizing antibodies. J. Gen. Virol. 2005, 86, 1435–1440. [Google Scholar] [CrossRef]
- Deming, D.; Sheahan, T.; Heise, M.; Yount, B.; Davis, N.; Sims, A.; Suthar, M.; Harkema, J.; Whitmore, A.; Pickles, R.; et al. Vaccine efficacy in senescent mice challenged with recombinant SARS-CoV bearing epidemic and zoonotic spike variants. PLoS Med. 2006, 3, e525. [Google Scholar] [CrossRef] [PubMed]
- Sheahan, T.; Whitmore, A.; Long, K.; Ferris, M.; Rockx, B.; Funkhouser, B.; Donaldson, E.; Gralinski, L.; Collier, M.; Heise, M.; et al. Successful vaccination strategies that protect aged mice from lethal challenge from influenza virus and heterologous severe acute respiratory syndrome coronavirus. J. Virol. 2011, 85, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.P.; Dalby, B. Protein delivery using VP22. Nat. Biotechnol. 2002, 20, 20. [Google Scholar] [CrossRef] [PubMed]
- Almazan, F.; Galan, C.; Enjuanes, L. Engineering infectious cDNAs of coronavirus as bacterial artificial chromosomes. Methods Mol. Biol. 2008, 454, 275–291. [Google Scholar] [PubMed]
- Shanmugarai, B.; Malla, A.; Phoolcharoen, W. Emergence of Novel Coronavirus 2019-nCoV: Need for Rapid Vaccine and Biologics Development. Pathogens 2020, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K. RNA Viruses as Tools in Gene Therapy and Vaccine Development. Genes 2019, 10, 189. [Google Scholar] [CrossRef]
- Lundstrom, K. Plasmid DNA-based Alphavirus Vaccines. Vaccines 2019, 7, 29. [Google Scholar] [CrossRef]
- Francesco, N. Is antibody-dependent enhancement playing a role in COVID-19 pathogenesis? Swiss. Med. Wkly. 2020, 150, w20249. [Google Scholar]
- Tetro, J.A. Is COVID-19 receiving ADE from other coronaviruses? Microbes. Inf. 2020, 22, 72–73. [Google Scholar] [CrossRef]
- Wan, Y.; Shang, J.; Sun, S.; Tan, W.; Chen, J.; Geng, Q.; He, L.; Chen, Y.; Wu, J.; Shi, Z.; et al. Molecular Mechanism for Antibody-Dependent Enhancement of Coronavirus Entry. J. Virol. 2020, 94, e02015-19. [Google Scholar] [CrossRef]
- Chen, K.; Kolls, J.K. T cell-mediated host immune defenses in the lung. Annu. Rev. Immunol. 2013, 31, 605–633. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.J. Pathogenesis of COVID-19 from a cell biologic perspective. Eur. Respir. J. 2020, 55, 2000607. [Google Scholar] [CrossRef] [PubMed]
- Bloch, E.M.; Shoham, S.; Casadevall, A.; Sachais, B.S.; Shaz, B.; Winters, J.L.; van Burskirk, C.; Grossman, B.J.; Joyner, M.; Henderson, J.P.; et al. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J. Clin. Investig. 2020, 138745. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Pang, R.; Xue, X.; Bao, J.; Ye, S.; Daj, Y.; Zheng, Y.; Fu, Q.; Hu, Z.; Yi, Y. Anti-SARS-CoV-2 virus antibody levels in convalescent plasma of six donors who have recovered from COVID-19. Aging 2020, 12. [Google Scholar] [CrossRef]
- Shen, C.; Wang, Z.; Zhao, F.; Yang, Y.; Li, J.; Yuan, J.; Wang, F.; Li, D.; Yang, M.; Xing, L.; et al. Treatment of 5 Critically Ill Patients with COVID-19 With Convalescent Plasma. JAMA 2020. [Google Scholar] [CrossRef]
- Grein, J.; Ohmagari, N.; Shin, D.; Diaz, G.; Asperges, E.; Castagna, A.; Feldt, T.; Green, G.; Green, M.L.; Lescure, F.X.; et al. Compassionate Use of Remdesivir for Patients with COVID-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Available online: www.gilead.com/news-and-press/company-statements/gilead-sciences-statement-on-data-from-remdesivir-study-in-patients-with-severe-covid-19-in-china (accessed on 30 April 2020).
- Available online: www.theguardian.com/world/2020/apr/23/high-hopes-drug-for-covid-19-treatment-failed-in-full-trial (accessed on 30 April 2020).
- Ferner, R.E.; Aronson, J.K. Chloroquine and hydroxychloroquine in COVID-19. BMJ 2020, 369, m1432. [Google Scholar] [CrossRef]
- Gao, J.; Tian, Z.; Yang, X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci. Trends 2020, 14, 72–73. [Google Scholar] [CrossRef]
- Chen, Z.; Hu, J.; Zhang, Z.; Jiang, S.; Han, S.; Yan, D.; Zhuang, R.; Hu, B.; Zhang, Z. Efficacy of hydroxychloroquine in patients with COVID-19: Results of a randomized clinical trial. Version 2. medRxiv 2020. [Google Scholar] [CrossRef]
- Yan, D.; Zhang, Z. Therapeutic Effect of Hydroxychloroquine on Novel Coronavirus Pneumonia (COVID-19). Chinese Clinical Trials Registry. Available online: http://www.chictr.org.cn/showproj.aspx?proj=48880 (accessed on 28 April 2020).
- Murphy, M.; Carmichael, A.J. Fatal toxic epidermal necrolysis associated with hydroxychloroquine. Clin. Exp. Dermatol. 2001, 26, 457–458. [Google Scholar] [CrossRef]
- Makin, A.J.; Wendon, J.; Fitt, S.; Portmann, B.C.; Williams, R. Fulminant hepatic failure secondary to hydroxychloroquine. Gut 1994, 35, 569–570. [Google Scholar] [CrossRef] [PubMed]
- Chorin, E.; Dai, M.; Shulman, E.; Wadhwani, L.; Bar-Cohen, R.; Barbhayia, C.; Aizer, A.; Holmes, D.; Bernstein, S.; Spinelli, M.; et al. The QT interval in patients with SARS-CoV-2 infection treated with hydroxychloroquine/azithromycin. medRxiv 2020. [Google Scholar] [CrossRef]
- Li, Y.; Xie, Z.; Lin, W.; Cai, W.; Wen, C.; Guan, Y.; Mo, X.; Wang, J.; Wang, Y.; Peng, P.; et al. An exploratory randomized, controlled study on the efficacy and safety of lopinavir/ritonavir or arbidol treating adult patients hospitalized with mild/moderate COVID-19 (ELACOI). medRxiv 2020. [Google Scholar] [CrossRef]
- Available online: www.who.int/blueprint/priority-diseases/key-action/novel-coronavirus-landscape-ncov.pdf (accessed on 28 April 2020).
- Zhao, H. COVID-19 drives new threats to bats in China. Science 2020, 367, 1436. [Google Scholar]
- Cyranoski, D. Mystery deepens over animal source of coronavirus. Nature 2020, 579, 18–19. [Google Scholar] [CrossRef]

| Virus | Disease | Effect | Ref. |
|---|---|---|---|
| TGEV | Gastroenteritis in pigs | High morbidity, mortality | [3] |
| PEDV | Gastroenteritis in pigs | High morbidity, mortality | [4] |
| PHEV | Enteric infection | Diarrhea, encephalitis | [5] |
| BCoV | Respiratory tract infection | Significant loss in cattle industry | [6,7] |
| RCoV | Respiratory tract infection | Model system | [8] |
| IBV | Respiratory infections, renal disease | Significant losses in the chicken industry | [7,9] |
| FCoV | Respiratory tract infection | Mild or asymptomatic | [10] |
| FIPV | Infectious peritonitis | Lethal FIP | [11] |
| SW1 | Respiratory, acute liver | SW1 found in deceased whale | [13] |
| Bat CoV | Respiratory tract infection | Potential threat of epidemics | [14] |
| SHC014-CoV | Risk of epidemic | Chimeric CoV presents risk | [15] |
| MHV | Respiratory, enteric, neurological infections | Mouse model for human disease | [16] |
| A59, JHMV | Chronic demyelination | Mouse model for MS | [17] |
| α HCoV-229E | Respiratory infection | 15%–30% of annual common cold | [18] |
| α HCoV-NL63 | Respiratory infection | Also associated with croup | [18,19] |
| β HcoV-OC43 | Respiratory infection | 15%–30% of annual common cold | [20] |
| β HcoV-HKU1 | Respiratory infection | 15%–30% of annual common cold | [20] |
| SARS-CoV | SARS | Epidemic with 774 deaths | [21,22] |
| MERS-CoV | MERS | Epidemic with 333 deaths | [23] |
| Therapy | Disease | Effect | Ref. |
|---|---|---|---|
| siRNAs | SARS-CoV | Inhibition of replication in Vero E6 cells | [119] |
| SARS-CoV | Prophylactic/therapeutic effects in FRhK4 cells | [120] | |
| SARS-CoV | Suppression of SARS symptoms in primates | [121] | |
| SARS-CoV | Reduced infection in ACE2-silenced cells | [122] | |
| siRNAs, miRNAs | SARS-CoV | Knockdown of ezrin | [123] |
| MERS-CoV | Design of siRNAs, miRNAs for MERS control | [124] | |
| siRNAs | FIPV | siRNA combination delays resistance | [125] |
| shRNAs | PDCoV | Reduced titers and viral RNA in ST cells | [126] |
| PEDV | Inhibition of viral RNA and replication | [127] | |
| SADS-CoV | Inhibition of viral RNA and replication | [127] | |
| PDCoV | Inhibition of viral RNA and replication | [127] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lundstrom, K. Coronavirus Pandemic—Therapy and Vaccines. Biomedicines 2020, 8, 109. https://doi.org/10.3390/biomedicines8050109
Lundstrom K. Coronavirus Pandemic—Therapy and Vaccines. Biomedicines. 2020; 8(5):109. https://doi.org/10.3390/biomedicines8050109
Chicago/Turabian StyleLundstrom, Kenneth. 2020. "Coronavirus Pandemic—Therapy and Vaccines" Biomedicines 8, no. 5: 109. https://doi.org/10.3390/biomedicines8050109
APA StyleLundstrom, K. (2020). Coronavirus Pandemic—Therapy and Vaccines. Biomedicines, 8(5), 109. https://doi.org/10.3390/biomedicines8050109





