Antioxidant, Antifungal Activities of Ethnobotanical Ficus hirta Vahl. and Analysis of Main Constituents by HPLC-MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Plant Materials and Reagents
2.2. HFF Extracts Preparation
2.3. Estimation of Total Phenolic Contents
2.4. Estimation of Total Flavonoid Contents
2.5. In Vitro Evaluation of Antioxidant Activity
2.5.1. DPPH assay
2.5.2. ABTS Assay
2.5.3. Ferric Reducing Antioxidant Potential (FRAP) Assay
2.6. Antifungal Activity of HFF Extracts
2.6.1. Fungi Strains and Growth Condition
2.6.2. In vitro Antifungal Assay
2.6.3. In Vivo Antifungal Assay on Mycelial Growth of P. Italicum
2.7. HPLC-QTOF-MS Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Extract Yields, TPC, and TFC of HFF Extracts
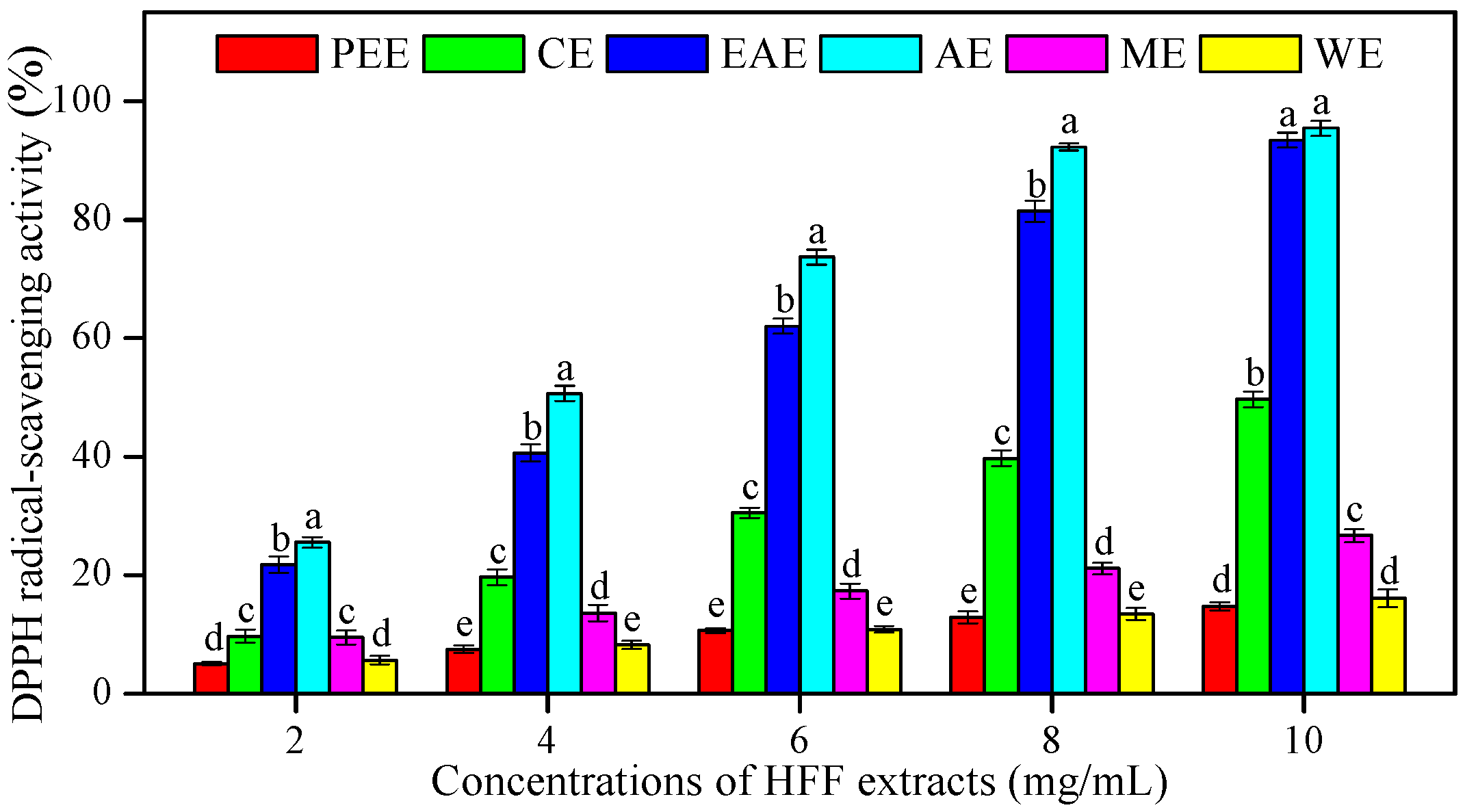
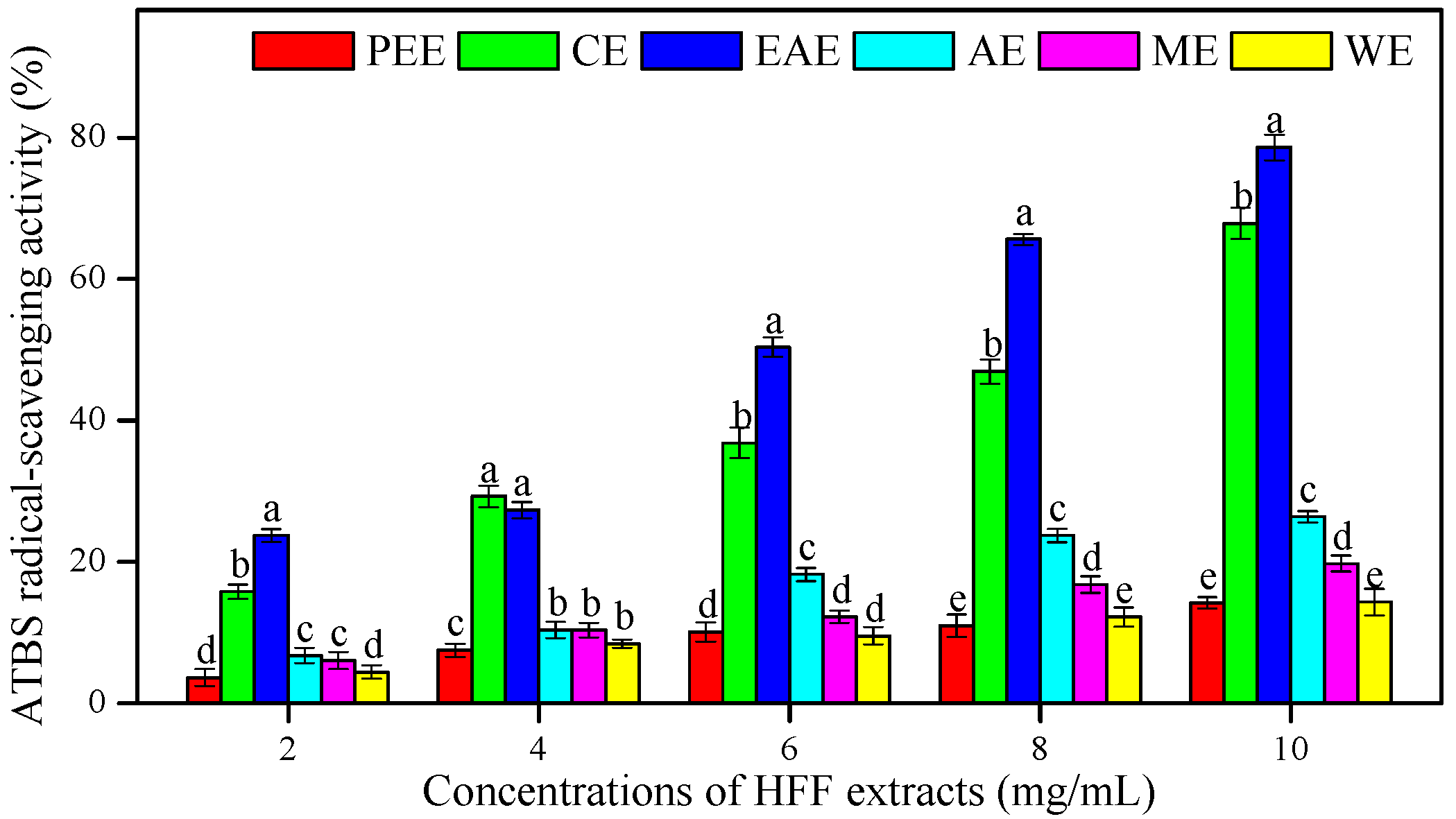
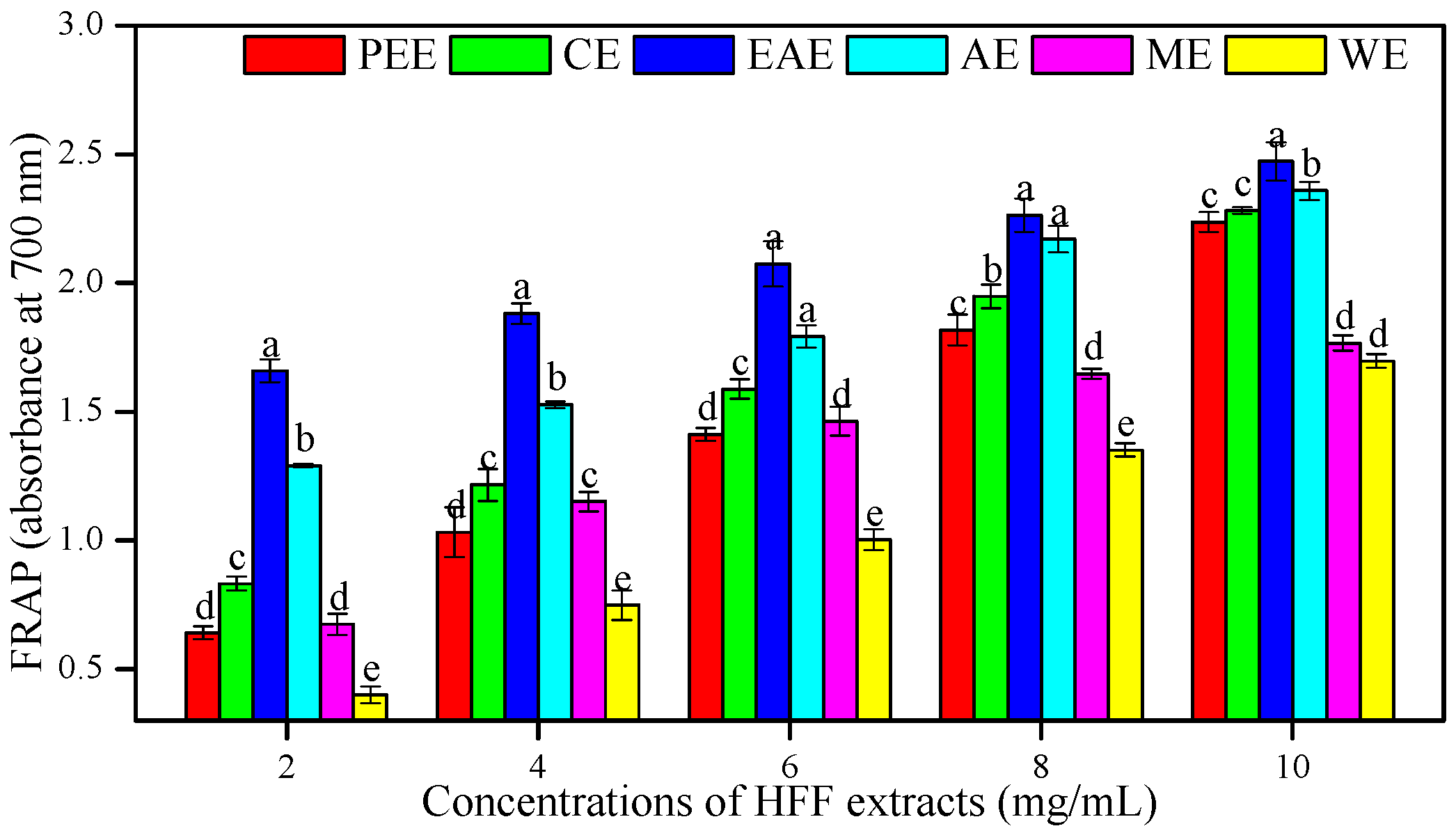
3.2. In Vitro Antioxidant Activities of HFF Extracts
3.3. In Vitro Antifungal Activities of HFF Extracts
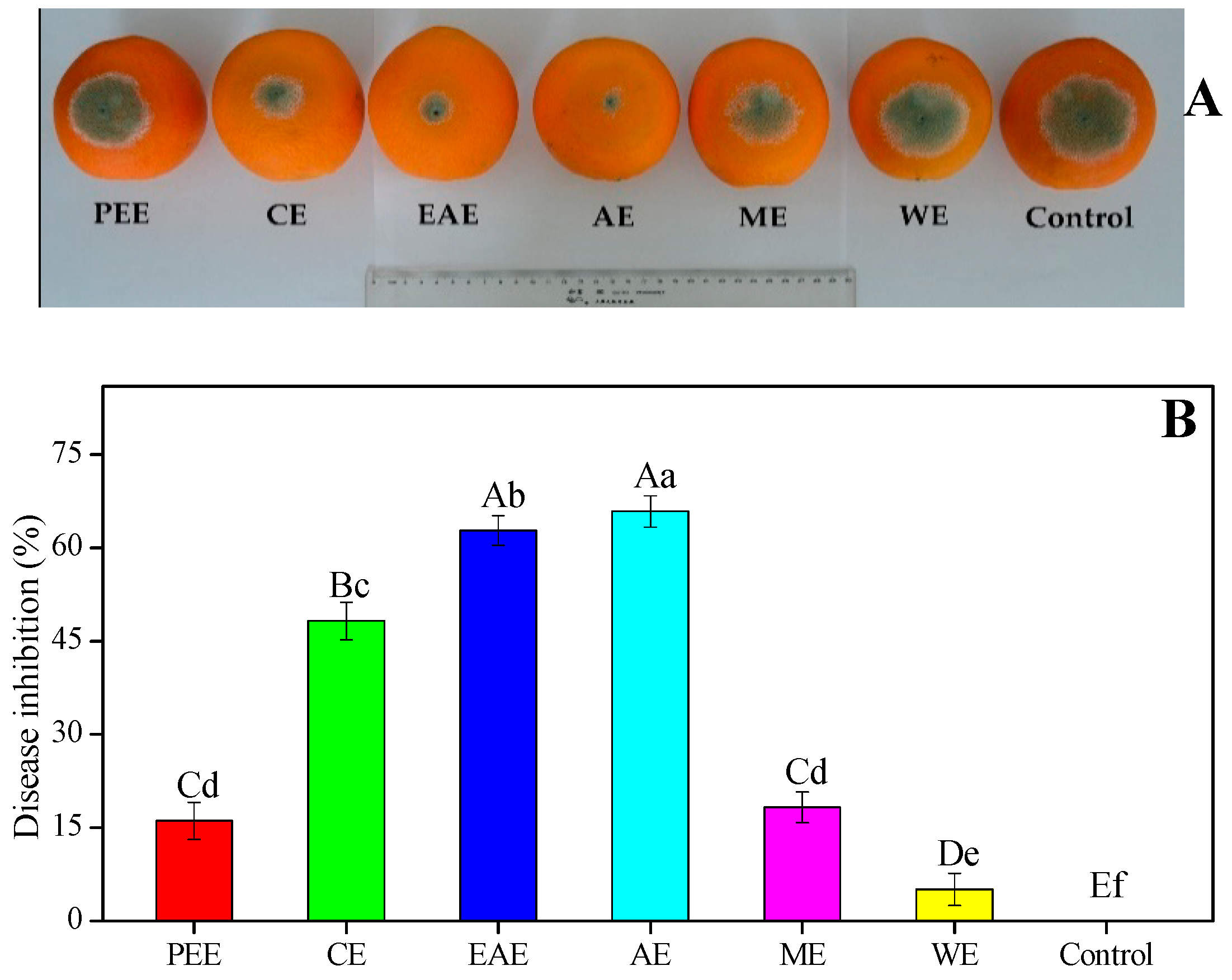
3.4. In Vivo Antifungal Activity of HFF Extracts on Blue Mold Development on Inoculated Fruit
3.5. HPLC-QTOF-MS Analysis of HFF Extracts
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, C.; Cai, N.; Chen, J.; Peng, X.; Wan, C. Chitosan-Based Coating Enriched with Hairy Fig (Ficus hirta Vahl.) Fruit Extract for “Newhall” Navel Orange Preservation. Coatings 2018, 8, 445. [Google Scholar] [CrossRef]
- Cheng, J.; Yi, X.; Wang, Y.; Huang, X.; He, X. Phenolics from the roots of hairy fig (Ficus hirta Vahl.) exert prominent anti-inflammatory activity. J. Funct. Foods 2017, 31, 79–88. [Google Scholar] [CrossRef]
- Yi, T.; Chen, Q.L.; He, X.C.; So, S.W.; Lo, Y.L.; Fan, L.L.; Xu, J.; Tang, Y.; Zhang, J.Y.; Zhao, Z.Z.; et al. Chemical quantification and antioxidant assay of four active components in Ficus hirta root using UPLC-PAD-MS fingerprinting combined with cluster analysis. Chem. Cent. J. 2013, 7, 1752–1760. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Yi, X.; Chen, H.; Wang, Y.; He, X. Anti-inflammatory phenylpropanoids and phenolics from Ficus hirta Vahl. Fitoterapia 2017, 121, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Ya, J.; Zhang, X.Q.; Wang, Y.; Zhang, Q.W.; Chen, J.X.; Ye, W.C. Two new phenolic compounds from the roots of Ficus hirta. Nat. Prod. Res. 2010, 24, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.W.; Liu, X.Z.; Lv, Z.C.; Peng, Y.-H. Effects of Ficus hirta Vahl. (Wuzhimaotao) extracts on growth inhibition of HeLa cells. Exp. Toxicol. Pathol. 2012, 64, 743–749. [Google Scholar] [CrossRef]
- Chen, C.; Peng, X.; Zeng, R.; Chen, M.; Wan, C.; Chen, J. Ficus hirta fruits extract incorporated into an alginate-based edible coating for Nanfeng mandarin preservation. Sci. Hortic. 2016, 202, 41–48. [Google Scholar] [CrossRef]
- Wan, C.; Han, J.; Chen, C.; Yao, L.; Chen, J.; Yuan, T. Monosubstituted Benzene Derivatives from Fruits of Ficus hirta and Their Antifungal Activity against Phytopathogen Penicillium italicum. J. Agric. Food Chem. 2016, 64, 5621–5624. [Google Scholar] [CrossRef]
- Chen, Q.; Ye, S.X. Antibacterial activity of Ficus hirta roots by chromotest microassay. Anhui Agric. Sci. 2012, 40, 8452–8454. [Google Scholar] [CrossRef]
- Chen, C.; Wan, C.; Peng, X.; Chen, Y.; Chen, M.; Chen, J. Optimization of Antifungal Extracts from Ficus hirta Fruits Using Response Surface Methodology and Antifungal Activity Tests. Molecules 2015, 20, 19647–19659. [Google Scholar] [CrossRef]
- Wan, C.; Chen, C.; Li, M.; Yang, Y.; Chen, M.; Chen, J. Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits. Plants 2017, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.D.; Thanh, T.N.; Duc, T.D.; Hoang, S.N.; Thi, T.T.; Thi, H.A.N.; Duc, Q.T. A New Furanocoumarin Glycoside from the Roots of Ficus hirta. Lett. Org. Chem. 2018, 15, 1007–1011. [Google Scholar] [CrossRef]
- Chen, C.; Cai, N.; Chen, J.; Wan, C. UHPLC-Q-TOF/MS-Based Metabolomics Approach Reveals the Antifungal Potential of Pinocembroside against Citrus Green Mold Phytopathogen. Plants 2020, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zheng, K.; Mei, W.; Wang, Y.; Yu, C.; Yu, B.; Deng, S.; Hu, J. Anti-inflammatory and proresolution activities of bergapten isolated from the roots of Ficus hirta in an in vivo zebrafish model. Biochem. Biophys. Res. Commun. 2018, 496, 763–769. [Google Scholar] [CrossRef]
- Lv, Z.C.; Chen, K.; Peng, Y.H. Isolation and identification of chemical constituents with anti-inflammatory activity from the roots of Ficus hirta Vahl. Chin. J. Trop. Crop. 2017, 38, 1134–1137. [Google Scholar] [CrossRef]
- Wan, C.; Yuan, T.; Cirello, A.L.; Seeram, N.P. Antioxidant and α-glucosidase inhibitory phenolics isolated from highbush blueberry flowers. Food Chem. 2012, 135, 1929–1937. [Google Scholar] [CrossRef]
- Tan, J.B.L.; Yap, W.J.; Tan, S.Y.; Lim, Y.Y.; Lee, S.M. Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family. Antioxidants 2014, 3, 758–769. [Google Scholar] [CrossRef]
- Su, B.L.; Zeng, R.; Chen, J.Y.; Chen, C.Y.; Guo, J.H.; Huang, C.G. Antioxidant and antimicrobial properties of various solvent extracts from Impatiens balsamina L. stems. J. Food Sci. 2012, 77, C614–C619. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Barry, A.L.; Coyle, M.B.; Thornsberry, C.; Gerlach, E.; Hawkinson, R. Methods of measuring zones of inhibition with the Bauer-Kirby disk susceptibility test. J. Clin. Microbiol. 1979, 10, 885–889. [Google Scholar] [CrossRef]
- Chen, C.; Nie, Z.; Wan, C.; Chen, J. Preservation of Xinyu Tangerines with an Edible Coating Using Ficus hirta Vahl. Fruits Extract-Incorporated Chitosan. Biomolecules 2019, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Karimkhani, M.M.; Salarbashi, D.; Sanjari Sefidy, S.; Mohammadzadeh, A. Effect of extraction solvents on lipid peroxidation, antioxidant, antibacterial and antifungal activities of Berberis orthobotrys Bienerat ex C.K. Schneider. J. Food Meas. Charact. 2019, 13, 357–367. [Google Scholar] [CrossRef]
- Liu, S.C.; Lin, J.T.; Wang, C.K.; Chen, H.Y.; Yang, D.J. Antioxidant properties of various solvent extracts from lychee (Litchi chinenesis Sonn.) flowers. Food Chem. 2009, 114, 577–581. [Google Scholar] [CrossRef]
- Lim, S.; Choi, A.H.; Kwon, M.; Joung, E.J.; Shin, T.; Lee, S.G.; Kim, N.G.; Kim, H.R. Evaluation of antioxidant activities of various solvent extract from Sargassum serratifolium and its major antioxidant components. Food Chem. 2019, 278, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Kuang, W.; Liu, Z.W.; Zhang, C.; Li, B.L. Studies on the Antioxidant Activities of Ficus hirta Vahl. Guangdong Chem. 2015, 42, 42–43. [Google Scholar]
- Kasangana, P.B.; Haddad, P.S.; Stevanovic, T. Study of Polyphenol Content and Antioxidant Capacity of Myrianthus Arboreus (Cecropiaceae) Root Bark Extracts. Antioxidants 2015, 4, 410–426. [Google Scholar] [CrossRef]
- Mabrouki, H.; Duarte, C.M.M.; Akretche, D.E. Estimation of Total Phenolic Contents and In Vitro Antioxidant and Antimicrobial Activities of Various Solvent Extracts of Melissa officinalis L. Arabian J. Sci. Eng. 2018, 43, 3349–3357. [Google Scholar] [CrossRef]
- Park, C.H.; Yeo, H.J.; Baskar, T.B.; Park, Y.E.; Park, J.S.; Lee, S.Y.; Park, S.U. In Vitro Antioxidant and Antimicrobial Properties of Flower, Leaf, and Stem Extracts of Korean Mint. Antioxidants 2019, 8, 75. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, D.; Yu, C.; Yao, L.; Chen, Z.; Tao, Y.; Cao, W. Phytochemical Analysis, Antioxidant and Analgesic Activities of Incarvillea compacta Maxim from the Tibetan Plateau. Molecules 2019, 24, 1692. [Google Scholar] [CrossRef]
- Wang, L.; Jin, J.; Liu, X.; Wang, Y.; Liu, Y.; Zhao, Y.; Xing, F. Effect of Cinnamaldehyde on Morphological Alterations of Aspergillus ochraceus and Expression of Key Genes Involved in Ochratoxin A Biosynthesis. Toxins 2018, 10, 340. [Google Scholar] [CrossRef]
- Belgacem, I.; Pangallo, S.; Abdelfattah, A.; Romeo, F.V.; Cacciola, S.O.; Li Destri Nicosia, M.G.; Ballistreri, G.; Schena, L. Transcriptomic Analysis of Orange Fruit Treated with Pomegranate Peel Extract (PGE). Plants 2019, 8, 101. [Google Scholar] [CrossRef] [PubMed]
- Palou, L.; Ali, A.; Fallik, E.; Romanazzi, G. GRAS, plant- and animal-derived compounds as alternatives to conventional fungicides for the control of postharvest diseases of fresh horticultural produce. Postharvest Biol. Technol. 2016, 122, 41–52. [Google Scholar] [CrossRef]
- Karim, H.; Boubaker, H.; Askarne, L.; Cherifi, K.; Lakhtar, H.; Msanda, F.; Boudyach, E.H.; Ait Ben Aoumar, A. Use of Cistus aqueous extracts as botanical fungicides in the control of Citrus sour rot. Microb. Pathog. 2017, 104, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Askarne, L.; Talibi, I.; Boubaker, H.; Boudyach, E.; Msanda, F.; Saadi, B.; Ait Ben Aoumar, A. Use of Moroccan medicinal plant extracts as botanical fungicide against citrus blue mould. Lett. Appl. Microbiol. 2013, 56, 37–43. [Google Scholar] [CrossRef]
- Regnier, T.; Combrinck, S.; Veldman, W.; Du Plooy, W. Application of essential oils as multi-target fungicides for the control of Geotrichum citri-aurantii and other postharvest pathogens of citrus. Ind. Crop. Prod. 2014, 61, 151–159. [Google Scholar] [CrossRef]
- Chen, C.; Qi, W.; Peng, X.; Chen, J.; Wan, C. Inhibitory Effect of 7-Demethoxytylophorine on Penicillium italicum and its Possible Mechanism. Microorganisms 2019, 7, 36. [Google Scholar] [CrossRef]
- Ortega-Ramirez, L.A.; Rodriguez-Garcia, I.; Leyva, J.M.; Cruz-Valenzuela, M.R.; Silva-Espinoza, B.A.; Gonzalez-Aguilar, G.A.; Siddiqui, W.; Ayala-Zavala, J.F. Potential of medicinal plants as antimicrobial and antioxidant agents in food industry: A hypothesis. J. Food Sci. 2014, 79, 129–137. [Google Scholar] [CrossRef]
- Karim, H.; Boubaker, H.; Askarne, L.; Talibi, I.; Msanda, F.; Boudyach, E.; Saadi, B.; Aoumar, A.A.B. Antifungal properties of organic extracts of eight Cistus L. species against postharvest citrus sour rot. Lett. Appl. Microbiol. 2016, 62, 16–22. [Google Scholar] [CrossRef]
- Scorzoni, L.; Sangalli-Leite, F.; de Lacorte Singulani, J.; de Paula e Silva, A.C.A.; Costa-Orlandi, C.B.; Fusco-Almeida, A.M.; Mendes-Giannini, M.J.S. Searching new antifungals: The use of in vitro and in vivo methods for evaluation of natural compounds. J. Microbiol. Methods 2016, 123, 68–78. [Google Scholar] [CrossRef]
| Extract Solvents | Polarity | Yield (%) | TPC (mg GAE/g dw) | TFC (mg RE/g dw) |
|---|---|---|---|---|
| Petroleum ether | 0.01 | 1.29 ± 0.09 e | 17.75 ± 0.52 e | 26.69 ± 1.93 d |
| Chloroform | 2.60 | 1.77 ± 0.12 d | 33.36 ± 1.16 b | 102.56 ± 6.48 b |
| Ethyl acetate | 4.30 | 1.71 ± 0.08 d | 84.03 ± 2.29 a | 81.74 ± 4.04 c |
| Acetone | 5.40 | 2.79 ± 0.21 c | 85.25 ± 1.72 a | 144.22 ± 8.46 a |
| Methanol | 6.60 | 3.59 ± 0.29 b | 30.68 ± 0.66 c | 24.07 ± 1.28 d |
| Water | 10.20 | 8.34 ± 0.32 a | 28.87 ± 0.88 d | 15.80 ± 0.59 e |
| Extract Solvents | Regression Equations | R2 | IC50 (mg/mL) |
|---|---|---|---|
| Petroleum ether | y = 2.4799x + 2.7274 | 0.9922 | 19.06 |
| Chloroform | y = 9.6033x + 1.4601 | 0.9983 | 5.06 |
| Ethyl acetate | y = 17.019x + 7.1932 | 0.9792 | 2.52 |
| Acetone | y = 22.323x + 4.8447 | 0.9917 | 2.02 |
| Methanol | y = 4.6913x + 5.0789 | 0.9878 | 9.58 |
| Water | y = 2.8148x + 2.4214 | 0.9792 | 16.90 |
| Ascorbic acid | y = 378.49x + 0.2104 | 0.9991 | 0.13 |
| Index | Correlation R2 | ||
|---|---|---|---|
| TPC | TFC | ||
| Antioxidants | DPPH scavenging activity | 0.82 * | 0.84 * |
| ABTS scavenging activity | 0.67 | 0.83 * | |
| FRAP assay | 0.92 ** | 0.70 | |
| Antifungals | P. italicum | 0.74 | 0.87 * |
| P. digitatum | 0.80 | 0.86 * | |
| A. niger | 0.78 | 0.78 | |
| A. oryzae | 0.72 | 0.88 * | |
| S. cerevisiae | 0.68 | 0.82 * | |
| C. utilis | 0.84 * | 0.77 | |
| Extract Solvents | Regression Equations | R2 | IC50 (mg/mL) |
|---|---|---|---|
| Petroleum ether | y = 1.9809x + 3.1291 | 0.9801 | 23.69 |
| Chloroform | y = 12.189x + 2.7832 | 0.9674 | 3.87 |
| Ethyl acetate | y = 14.121x + 6.7744 | 0.9655 | 3.06 |
| Acetone | y = 5.2587x + 1.3240 | 0.9760 | 9.26 |
| Methanol | y = 3.3853x + 2.8794 | 0.9885 | 13.92 |
| Water | y = 2.3497x + 2.7319 | 0.9732 | 20.12 |
| Ascorbic acid | y = 296.09x + 1.6043 | 0.9993 | 0.16 |
| No. | Name | Molecular Formula | Exact Mass | RT (min) | M + H | M − H | Extracts | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PEE | CE | EAE | AE | ME | WE | |||||||
| 1 | methyl-1,2,3,4-tetrahydro-β-carboline-3-carboxylic acid | C13H14N2O2 | 230.1055 | 0.73 | 231.1128 | 229.0975 | + | + | + | |||
| 2 | methyl-1-methyl-1,2,3,4-tetrahydro-β-carboline-3-carboxylate | C14H16N2O2 | 244.1212 | 1.60 | 245.1285 | + | + | + | + | |||
| 3 | dihydrophaseic acid | C15H22O5 | 282.1467 | 0.82 | 281.1388 | + | + | + | + | + | + | |
| 4 | vomifoliol | C13H20O3 | 224.1412 | 1.17 | 225.1505 | + | + | + | + | + | ||
| 5 | dehydrovomifoliol | C13H18O3 | 222.1256 | 1.77 | 223.1329 | + | + | + | + | + | ||
| 6 | pubinernoid A | C11H16O3 | 196.1099 | 1.47 | 197.1177 | + | + | + | + | + | + | |
| 7 | 2-phenylethyl-O-β d-glucoside | C14H20O6 | 284.1260 | 1.15 | 285.1329 | + | + | + | + | |||
| 8 | 1-O-trans-cinnamoyl-β-D-glucopyranosyl-(1→6)-β-d-glucopyranoside | C21H28O12 | 472.1581 | 4.56 | 473.1656 | + | + | + | + | + | + | |
| 9 | 4-O-benzoyl-quinic acid | C14H16O7 | 296.0896 | 0.99 | 297.0970 | 295.0817 | + | + | + | + | + | |
| 10 | 3-O-benzoyl-quinic acid | C14H16O7 | 296.0896 | 0.73 | 297.0973 | 295.0818 | + | + | + | + | + | |
| 11 | benzyl-β-d-glucopyranoside | C13H18O6 | 270.1103 | 0.60 | 269.1027 | + | + | + | ||||
| 12 | (2S) 2-O-benzoyl-butanedioic acid-4-methyl ester | C12H12O6 | 252.0634 | 3.77 | 253.0712 | + | ||||||
| 13 | pinocembrin-7-O-β-d-glucoside | C21H22O9 | 418.1264 | 3.59 | 417.1189 | + | + | + | + | |||
| 14 | naringenin-7-O-β-d-glucoside | C21H22O10 | 434.1213 | 2.09 | 433.1126 | + | + | + | ||||
| 15 | eriodictyol-7-O-β-d-glucoside | C21H22O11 | 450.1162 | 1.23 | 449.1084 | + | + | + | ||||
| 16 | luteolin | C15H10O6 | 286.0477 | 3.92 | 285.0399 | + | + | + | ||||
| 17 | apigenin | C15H10O5 | 270.0528 | 3.94 | 271.0614 | + | + | + | ||||
| 18 | umbelliferone | C9H6O3 | 162.0317 | 3.72 | 163.0389 | + | ||||||
| Fungi | Inhibition Zone (mm) | ||||||
|---|---|---|---|---|---|---|---|
| PEE | CE | EAE | AE | ME | WE | Natamycin | |
| P. italicum | 27.50 ± 0.58 c | 37.50 ± 1.29 b | 41.00 ± 0.82 a | 41.75 ± 0.96 a | 28.00 ± 0.80 c | 15.50 ± 1.00 d | 27.20 ± 1.50 c |
| P. digitatum | 20.00 ± 0.82 d | 24.25 ± 1.89 c | 28.00 ± 2.16 b | 31.50 ± 1.29 a | 21.50 ± 1.29 d | 12.25 ± 0.96 e | 24.70 ± 0.96 c |
| A. niger | 17.75 ± 0.96 e | 25.00 ± 1.41 c | 37.25 ± 1.26 a | 33.25 ± 1.26 b | 17.50 ± 0.58 e | — | 27.00 ± 0.82 d |
| A. oryzae | 14.50 ± 1.29 d | 20.00 ± 0.82 c | 24.00 ± 1.15 b | 28.00 ± 1.41 a | — | — | 24.20 ± 0.50 b |
| S. cerevisiae | 19.75 ± 0.50 d | 23.50 ± 1.00 c | 27.75 ± 0.96 b | 31.50 ± 1.29 a | 14.25 ± 0.96 e | — | 24.50 ± 1.00 c |
| C. utilis | — | 14.50 ± 1.29 c | 22.00 ± 0.82 a | 19.50 ± 1.00 b | 12.50 ± 1.00 d | — | 22.75 ± 1.50 a |
| Fungi Strains | MIC a | ||||||
|---|---|---|---|---|---|---|---|
| PEE | CE | EAE | AE | ME | WE | Natamycin b | |
| P. italicum | 1000 | 250 | 125 | 125 | 500 | 2000 | 2 |
| P. digitatum | 1000 | 500 | 500 | 250 | 1000 | NT | 2 |
| A. niger | 2000 | 1000 | 500 | 500 | 2000 | NT | 2 |
| A. oryzae | 1000 | 500 | 500 | 250 | NT | NT | 2 |
| S. cerevisiae | 1000 | 1000 | 1000 | 500 | 2000 | NT | 4 |
| C. utilis | NT | 2000 | 1000 | 1000 | 2000 | NT | 4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.; Peng, X.; Chen, J.; Wan, C. Antioxidant, Antifungal Activities of Ethnobotanical Ficus hirta Vahl. and Analysis of Main Constituents by HPLC-MS. Biomedicines 2020, 8, 15. https://doi.org/10.3390/biomedicines8010015
Chen C, Peng X, Chen J, Wan C. Antioxidant, Antifungal Activities of Ethnobotanical Ficus hirta Vahl. and Analysis of Main Constituents by HPLC-MS. Biomedicines. 2020; 8(1):15. https://doi.org/10.3390/biomedicines8010015
Chicago/Turabian StyleChen, Chuying, Xuan Peng, Jinyin Chen, and Chunpeng Wan. 2020. "Antioxidant, Antifungal Activities of Ethnobotanical Ficus hirta Vahl. and Analysis of Main Constituents by HPLC-MS" Biomedicines 8, no. 1: 15. https://doi.org/10.3390/biomedicines8010015
APA StyleChen, C., Peng, X., Chen, J., & Wan, C. (2020). Antioxidant, Antifungal Activities of Ethnobotanical Ficus hirta Vahl. and Analysis of Main Constituents by HPLC-MS. Biomedicines, 8(1), 15. https://doi.org/10.3390/biomedicines8010015






