Zimmermann-Laband-1 Syndrome: Clinical, Histological, and Proteomic Findings of a 3-Year-Old Patient with Hereditary Gingival Fibromatosis
Abstract
1. Introduction
2. Experimental Section
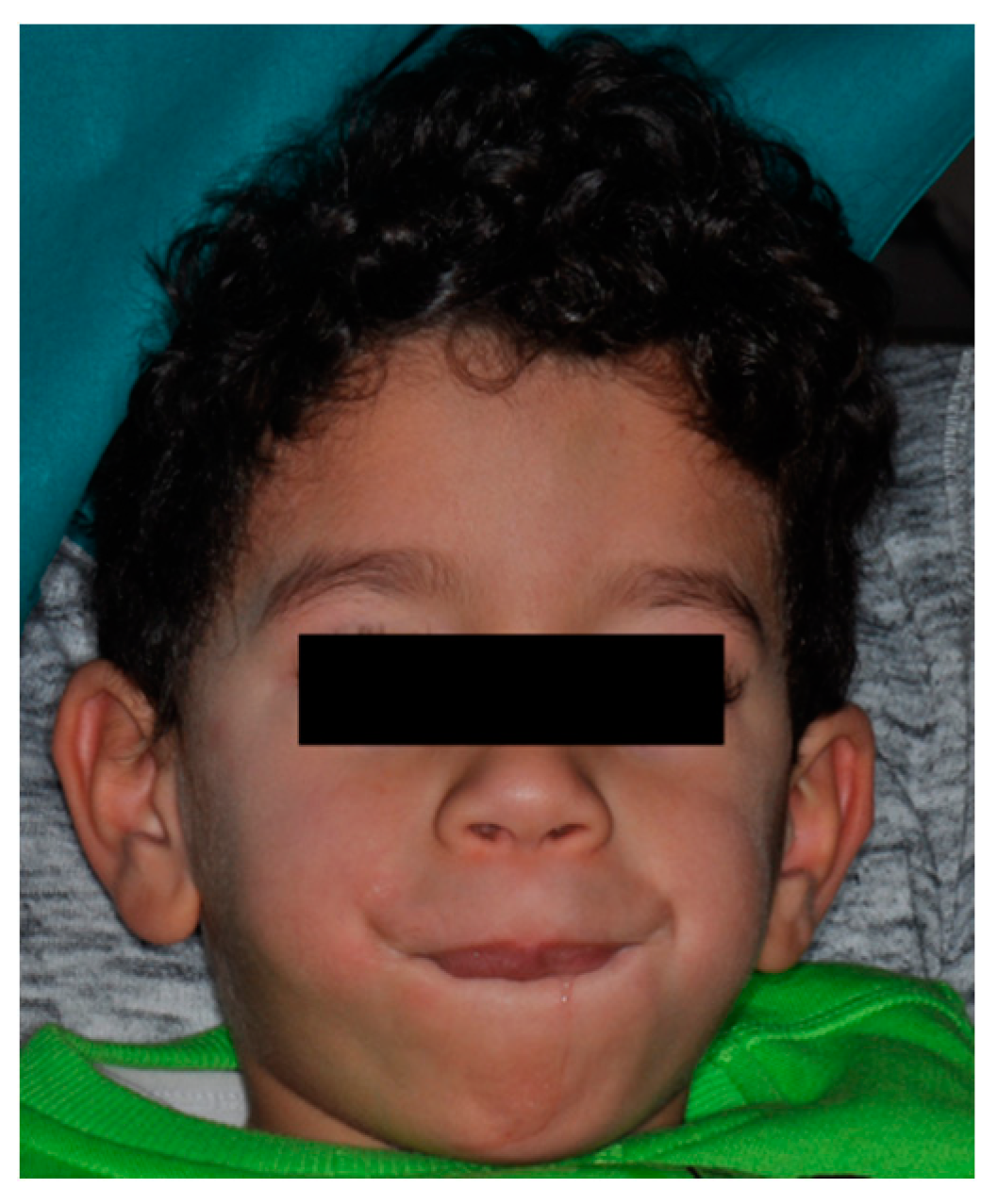
2.1. Patient Information
2.2. Medical, Family, and Psychosocial History
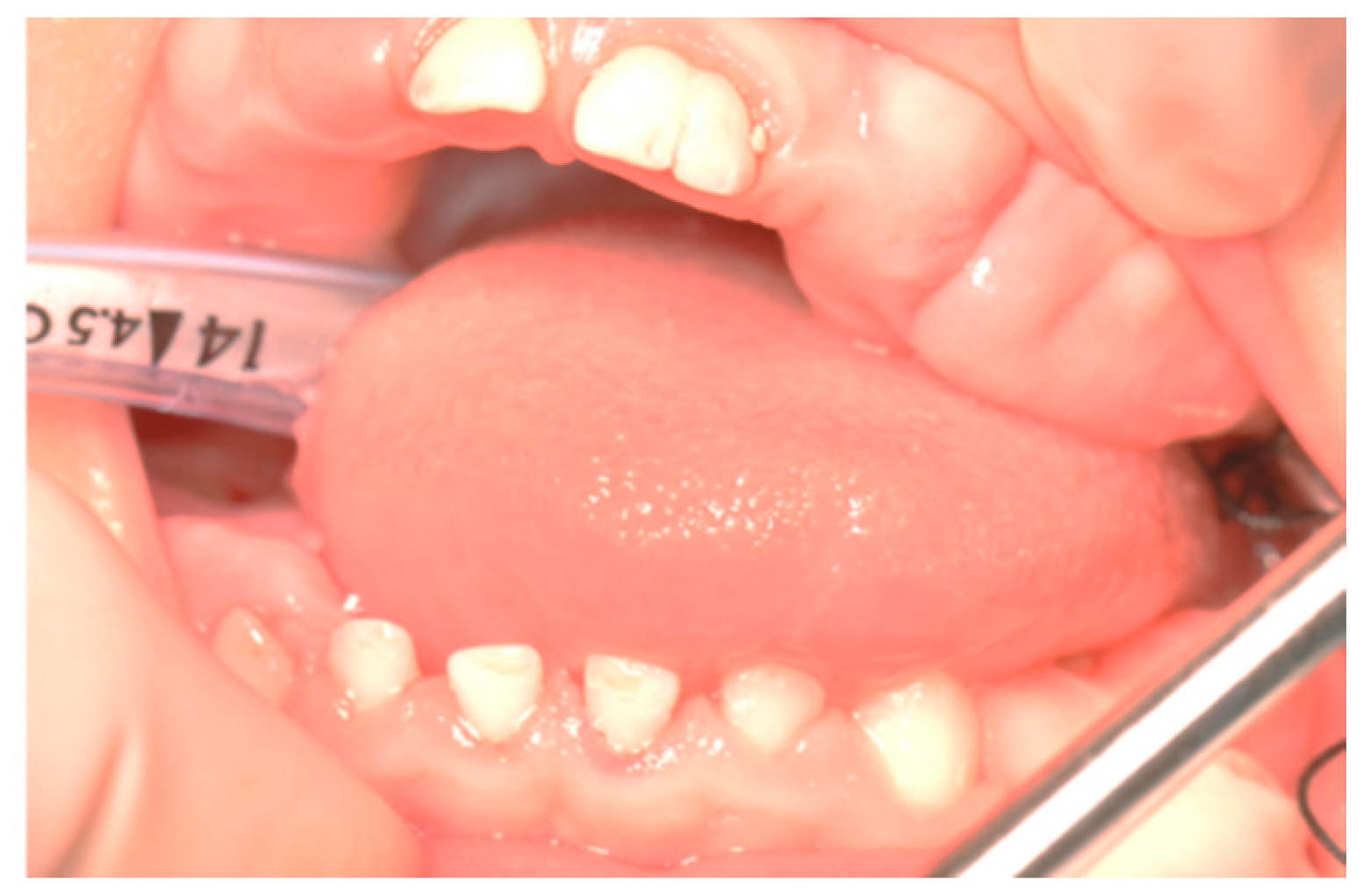
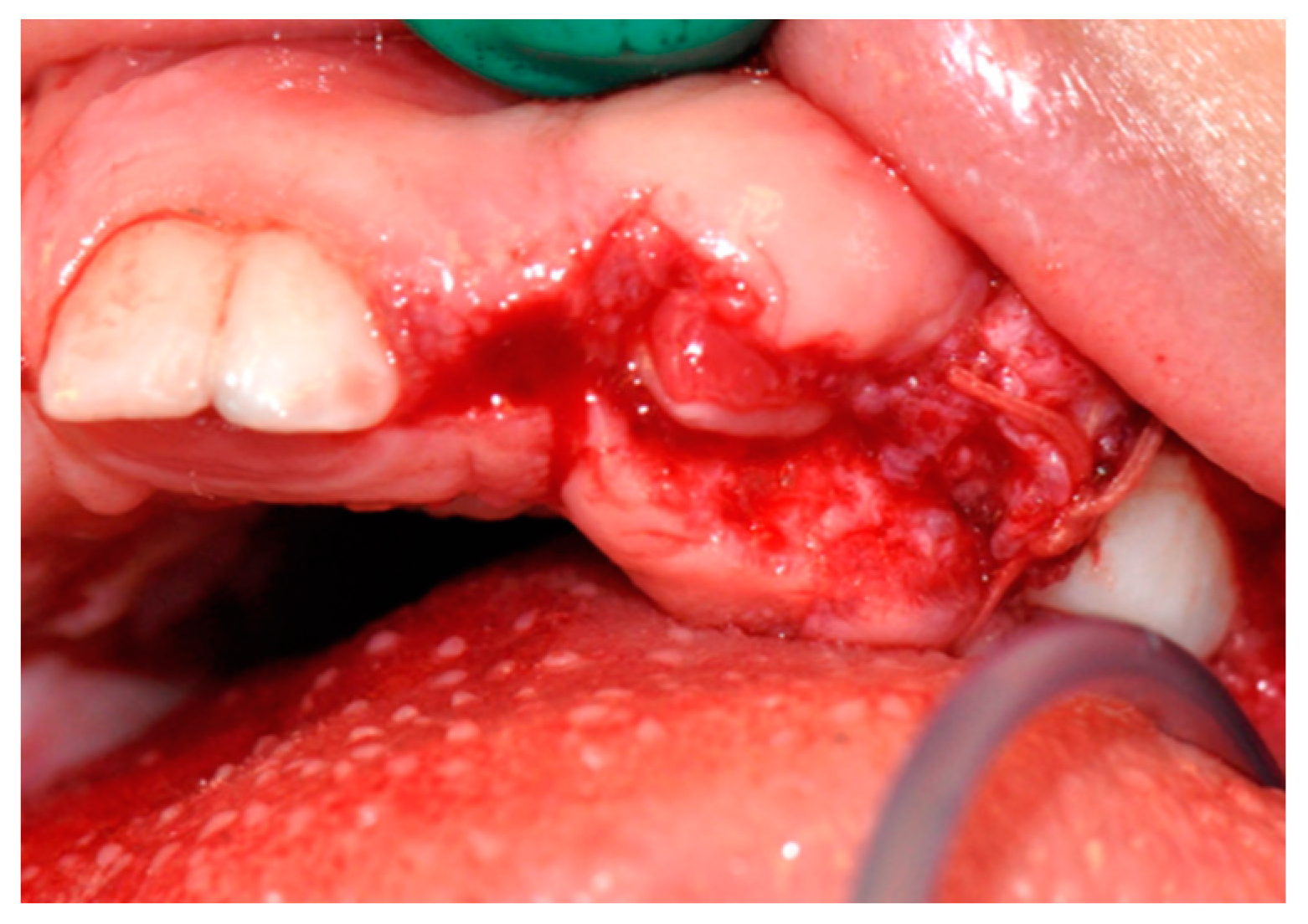
2.3. Therapeutic Intervention
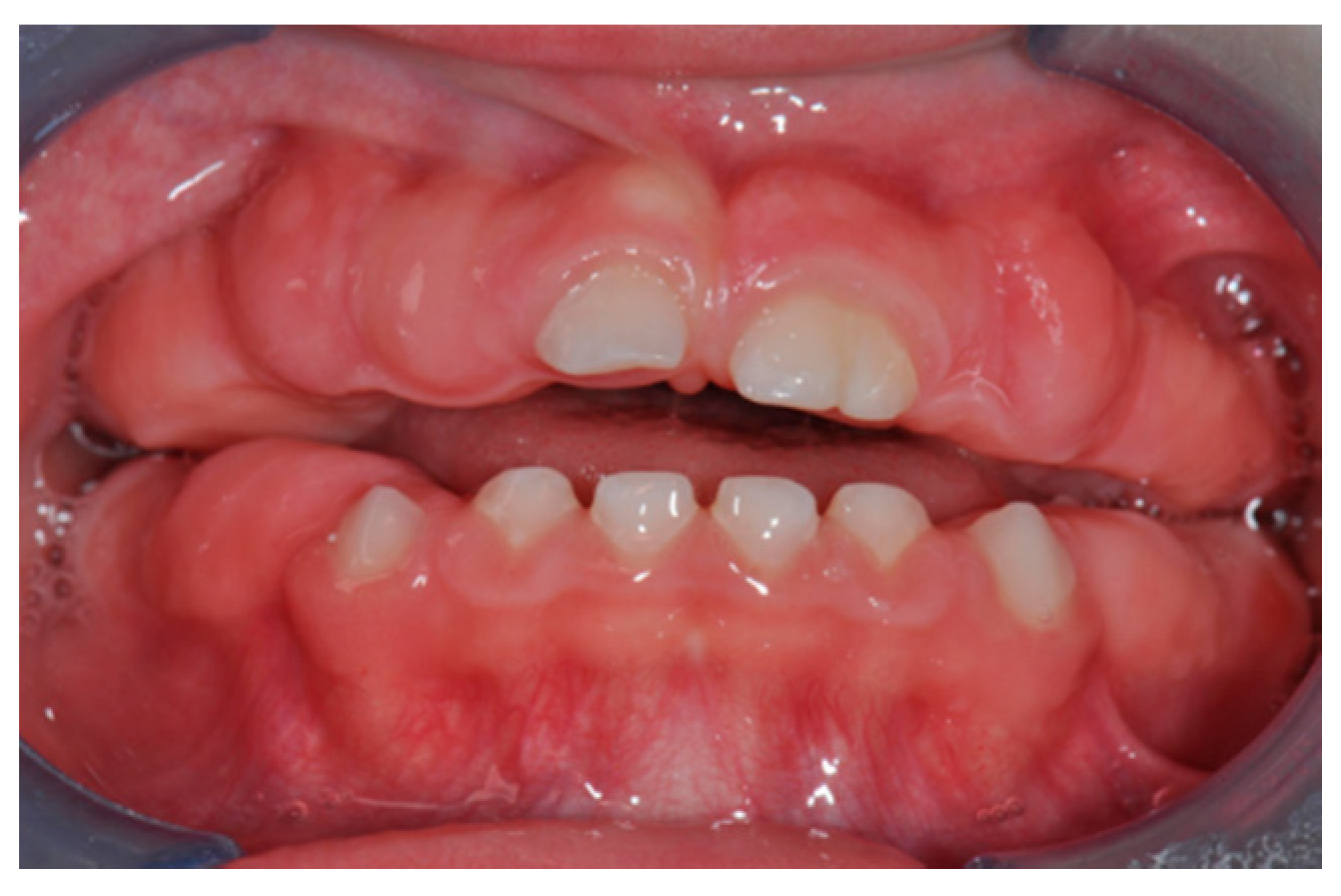
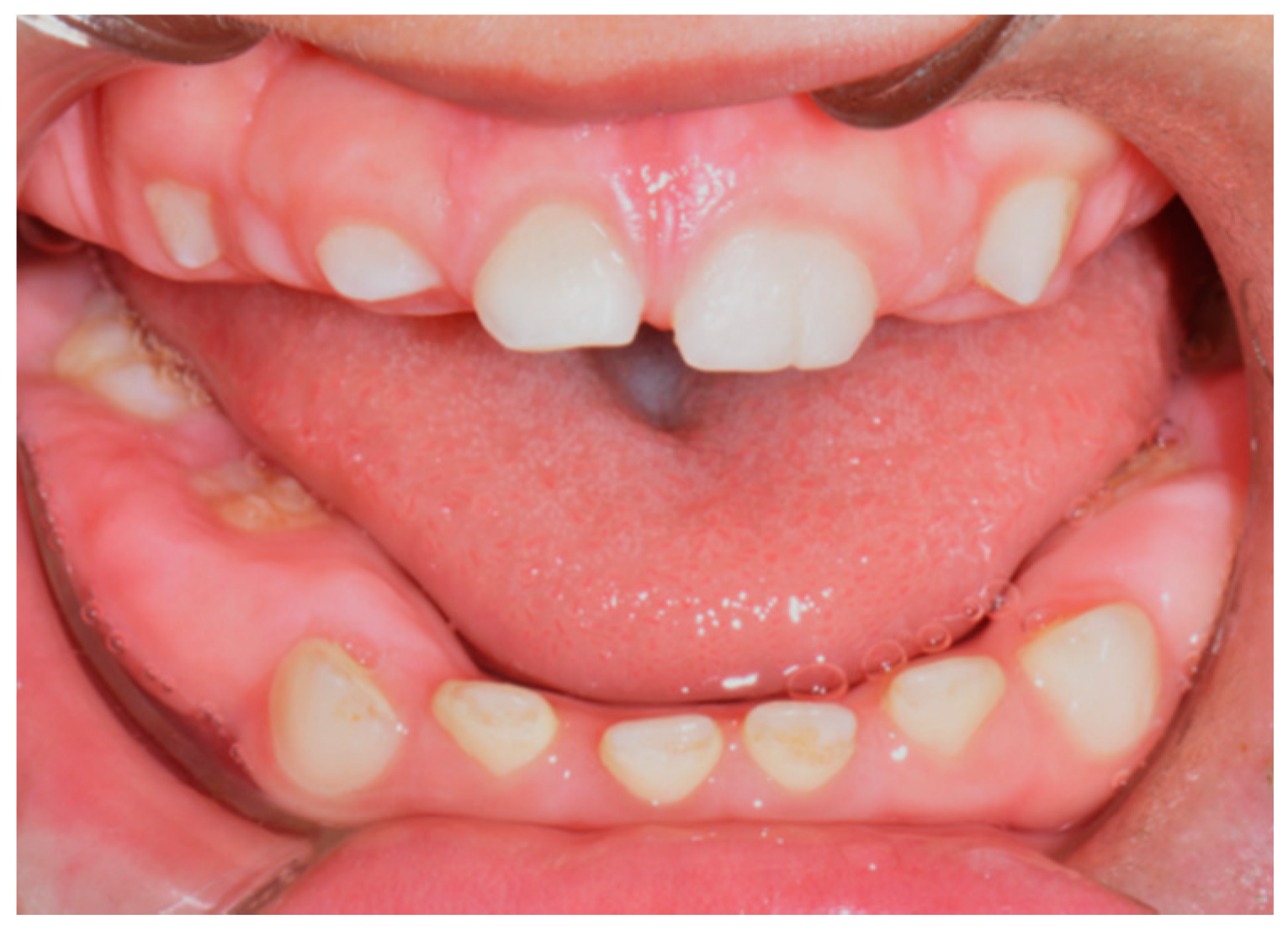
3. Results
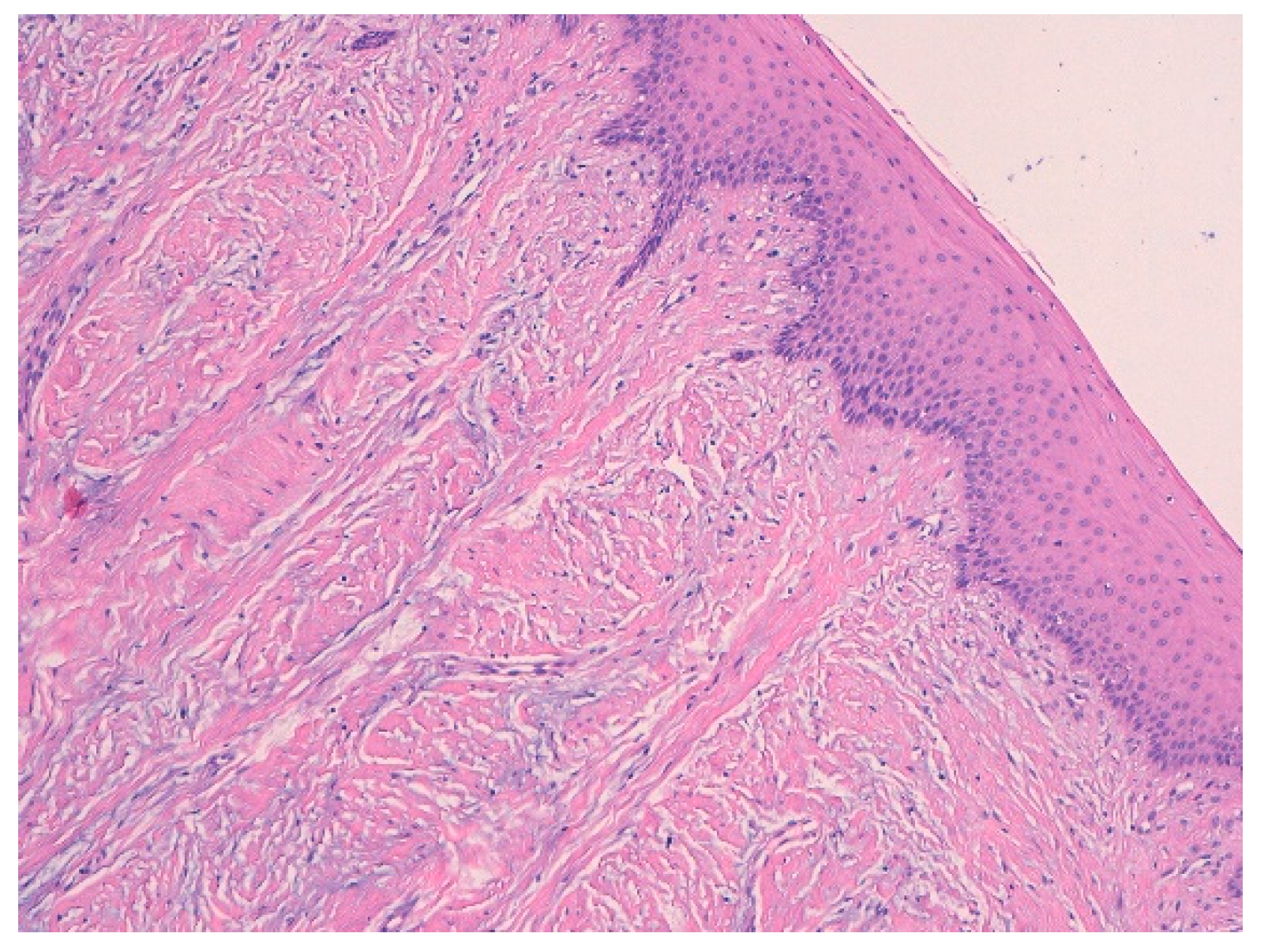
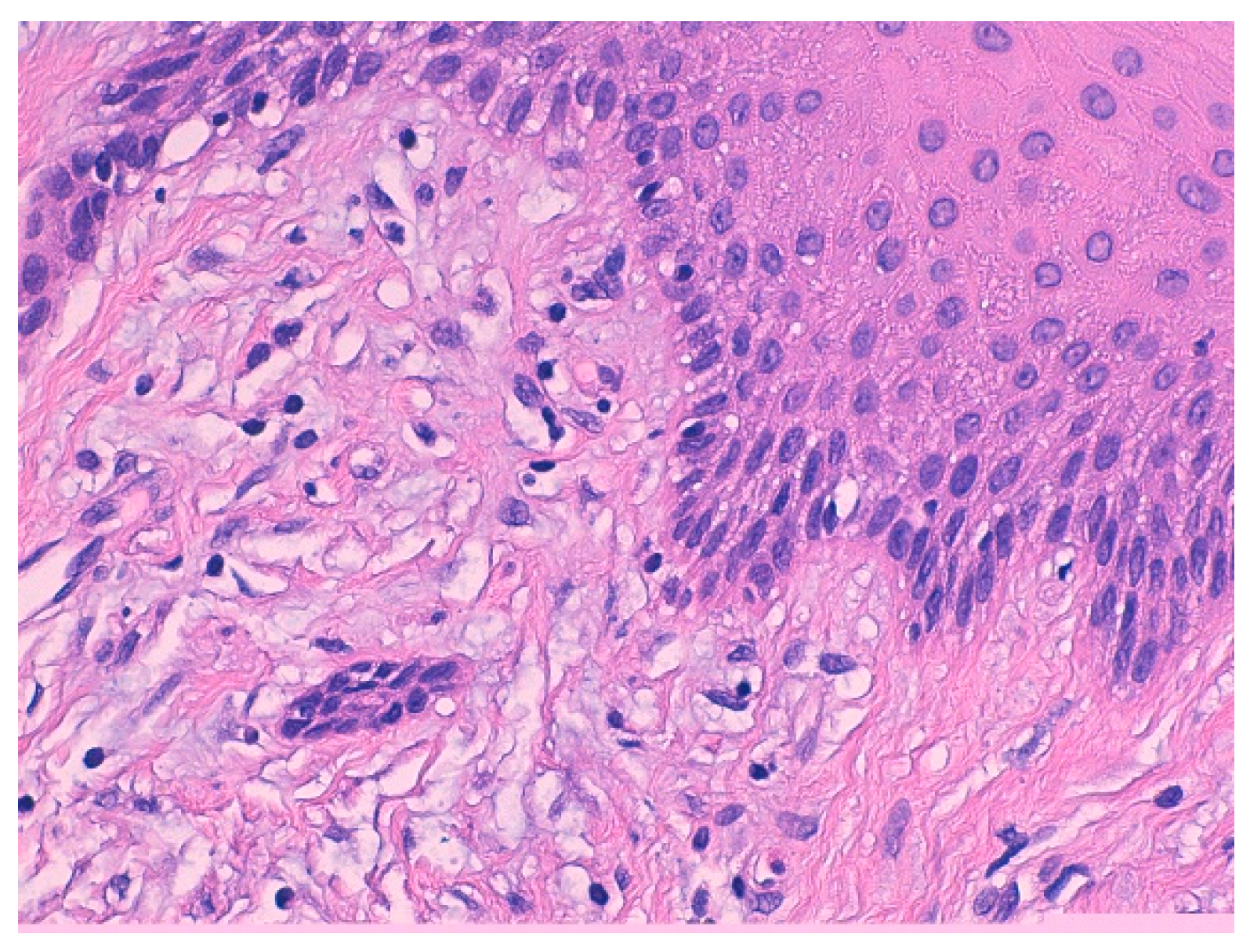
3.1. Histological Characteristics
3.2. Analysis of Salivary Biomarkers
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Alminana-Pastor, P.J.; Buitrago-Vera, P.J.; Alpiste-Illueca, F.M.; Catala-Pizarro, M. Hereditary gingival fibromatosis: Characteristics and treatment approach. J. Clin. Exp. Dent. 2017, 9, e599–e602. [Google Scholar] [CrossRef] [PubMed]
- Jorgenson, R.J.; Cocker, M.E. Variation in the Inheritance and Expression of Gingival Fibromatosis. J. Periodontol. 1974, 45, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Patini, R.; Staderini, E.; Lajolo, C.; Lopetuso, L.; Mohammed, H.; Rimondini, L.; Rocchetti, V.; Franceschi, F.; Cordaro, M.; Gallenzi, P. Relationship between oral microbiota and periodontal disease: A systematic review. Eur. Rev. Med Pharmacol. Sci. 2018, 22, 5775–5788. [Google Scholar] [CrossRef] [PubMed]
- Gagnier, J.J.; Kienle, G.; Altman, D.G.; Moher, D.; Sox, H.; Riley, D.; CARE Group. The CARE Guidelines: Consensus-based Clinical Case Reporting Guideline Development. Glob. Adv. Health Med. 2013, 2, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.; Varoni, E.M.; Cochis, A.; Cordaro, M.; Gallenzi, P.; Patini, R.; Staderini, E.; Lajolo, C.; Rimondini, L.; Rocchetti, V. Oral Dysbiosis in Pancreatic Cancer and Liver Cirrhosis: A Review of the Literature. Biomedicines 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Locker, D.; Liddell, A.; Shapiro, D. Diagnostic categories of dental anxiety: A population-based study. Behav. Res. Ther. 1999, 37, 25–37. [Google Scholar] [CrossRef]
- Cabras, T.; Pisano, E.; Montaldo, C.; Giuca, M.R.; Iavarone, F.; Zampino, G.; Castagnola, M.; Messana, I. Significant modifications of the salivary proteome potentially associated with complications of Down syndrome revealed by top-down proteomics. Mol. Cell Proteomics 2013, 12, 1844–1852. [Google Scholar] [CrossRef] [PubMed]
- Patini, R.; Staderini, E.; Gallenzi, P. Multidisciplinary surgical management of Cowden syndrome: Report of a case. J. Clin. Exp. Dent. 2016, 8, e472–e474. [Google Scholar] [CrossRef] [PubMed]
- Staderini, E.; Guglielmi, F.; Cordaro, M.; Gallenzi, P. Ossifying epulis in pseudohypo-parathyroidism: A case-based therapeutic approach. Eur. J. Paediatr. Dent. 2018, 19, 218–220. [Google Scholar] [CrossRef] [PubMed]
- Staderini, E.; Patini, R.; De Luca, M.; Gallenzi, P. Three-dimensional stereophotogrammetric analysis of nasolabial soft tissue effects of rapid maxillary expansion: A systematic review of clinical trials. Acta Otorhinolaryngol. Ital. 2018, 38, 399–408. [Google Scholar] [CrossRef] [PubMed]
| Classification of Hereditary Gingival Fibromatosis |
|---|
| 1. Hereditary gingival fibromatosis |
| 2. Gingival fibromatosis with craniofacial dysmorphism |
| 3. Gingival fibromatosis with progressive deafness |
| 4. Gingival fibromatosis/ hypertrichosis syndrome |
| 5. Ramon syndrome |
| 6. Zimmermann-Laband syndrome |
| 7. Infantile systemic hyalinosis |
| 8. Juvenile hyaline fibromatosis |
| 9. Oculodental syndrome, Rutherfurd type |
| 10. Amelogenesis imperfecta/nephrocalcinosis syndrome |
| 11. Amelogenesis imperfecta/gingival fibromatosis syndrome |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guglielmi, F.; Staderini, E.; Iavarone, F.; Di Tonno, L.; Gallenzi, P. Zimmermann-Laband-1 Syndrome: Clinical, Histological, and Proteomic Findings of a 3-Year-Old Patient with Hereditary Gingival Fibromatosis. Biomedicines 2019, 7, 48. https://doi.org/10.3390/biomedicines7030048
Guglielmi F, Staderini E, Iavarone F, Di Tonno L, Gallenzi P. Zimmermann-Laband-1 Syndrome: Clinical, Histological, and Proteomic Findings of a 3-Year-Old Patient with Hereditary Gingival Fibromatosis. Biomedicines. 2019; 7(3):48. https://doi.org/10.3390/biomedicines7030048
Chicago/Turabian StyleGuglielmi, Federica, Edoardo Staderini, Federica Iavarone, Laura Di Tonno, and Patrizia Gallenzi. 2019. "Zimmermann-Laband-1 Syndrome: Clinical, Histological, and Proteomic Findings of a 3-Year-Old Patient with Hereditary Gingival Fibromatosis" Biomedicines 7, no. 3: 48. https://doi.org/10.3390/biomedicines7030048
APA StyleGuglielmi, F., Staderini, E., Iavarone, F., Di Tonno, L., & Gallenzi, P. (2019). Zimmermann-Laband-1 Syndrome: Clinical, Histological, and Proteomic Findings of a 3-Year-Old Patient with Hereditary Gingival Fibromatosis. Biomedicines, 7(3), 48. https://doi.org/10.3390/biomedicines7030048







