Periodontitis Stage III–IV, Grade C and Correlated Factors: A Histomorphometric Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethical Approval
2.2. Patients and Collection of the Samples
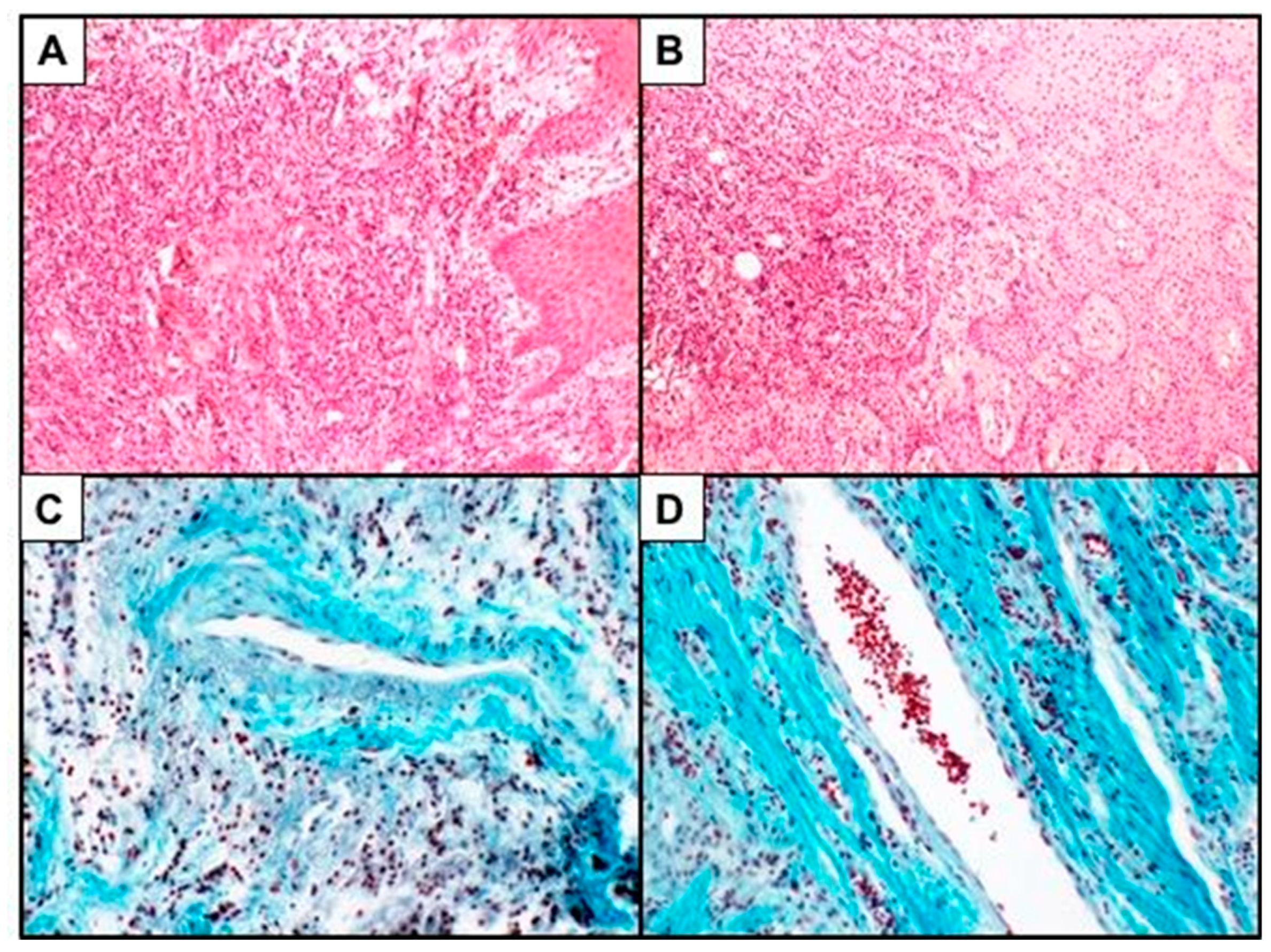
2.3. Histomorphometric Analysis
2.4. Statistical Analysis
3. Results
4. Discussion
4.1. Gender
4.2. Age
4.3. Smoking
4.4. Pocket Probing Depth
4.5. Plaque
4.6. Pus
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Meyle, J.; Chapple, I. Molecular aspects of the pathogenesis of periodontitis. Periodontol. 2000 2015, 69, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Clin. Periodont. 2018, 45, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Caton, J.C.; Armitage, G.; Berglundh, T.; Chapple, I.L.C.; Jepsen, S.; Kornman, K.S.; Mealey, B.L.; Papapanou, P.N.; Sanz, M.; Tonetti, M. A new classification scheme for periodontal and peri-implant diseases and conditions—Introduction and key changes from the 1999 classification. J. Clin. Periodont. 2018, 45, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cornelini, R.; Artese, L.; Rubini, C.; Fioroni, M.; Ferrero, G.; Santinelli, A.; Piattelli, A. Vascular endothelial growth factor and microvessel density around healthy and failing dental implants. Int. J. Oral Maxillofac. Implants 2001, 16, 389–393. [Google Scholar] [PubMed]
- Kasprzak, A.; Surdacka, A.; Tomczak, M.; Konkol, M. Role of high endothelial postcapillary venules and selected adhesion molecules in periodontal diseases: A review. J. Periodontal. Res. 2013, 48, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Borsani, E.; Salgarello, S.; Mensi, M.; Boninsegna, R.; Stacchiotti, A.; Rezzani, R.; Sapelli, P.; Bianchi, R.; Rodella, L.F. Histochemical and immunohistochemical evaluation of gingival collagen and metalloproteinases in peri-implantitis. Acta Histochem. 2005, 107, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Buffoli, B.; Dalessandri, M.; Favero, G.; Mensi, M.; Dalessandri, D.; di Rosario, F.; Stacchi, C.; Rezzani, R.; Salgarello, S.; Rodella, L.F. AQP1 expression in human gingiva and its correlation with periodontal and peri-implant tissue alterations. Acta Histochem. 2014, 116, 898–904. [Google Scholar] [CrossRef]
- Page, R.C.; Schroeder, H.E. Pathogenesis of inflammatory periodontal disease. A summary of current work. Lab. Investig. 1976, 34, 235–249. [Google Scholar]
- Younes, R.; Ghorra, C.; Khalife, S.; Igondjo-Tchen-Changotade, S.; Yousfi, M.; Willig, C.; Senni, K.; Godeau, G.; Naaman, N. Pertinent cell population to characterize periodontal disease. Tissue Cell 2009, 41, 141–150. [Google Scholar] [CrossRef]
- Vladau, M.; Cimpean, A.M.; Balica, R.A.; Jitariu, A.A.; Popovici, R.A.; Raica, M. VEGF/VEGFR2 Axis in Periodontal Disease Progression and Angiogenesis: Basic Approach for a New Therapeutic Strategy. In Vivo 2016, 30, 53–60. [Google Scholar] [PubMed]
- Know, V.; Caton, J.C. Commentary: Prognosis revisited a system for assigning periodontal prognosis. J. Periodontol. 2007, 78, 2063–2071. [Google Scholar]
- Preshaw, P.M.; Henne, K.; Taylor, J.J.; Valentine, R.A.; Conrads, G. Age-related changes in immune function (immune senescence) in caries and periodontal diseases: A systematic review. J. Clin. Periodontol. 2017, 44, S153–S177. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Bäumer, A.; Pritsch, M.; Cosgarea, R.; el Sayed, N.; Kim, T.S.; Eickholz, P.; Pretzl, B. Prognostic value of the periodontal risk assessment in patients with aggressive periodontitis. J. Clin. Periodontol. 2012, 39, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, C.; Leyland, A.H.; Wennström, J.L. Factors influencing the outcome of non-surgical periodontal treatment: A multilevel approach. J. Clin. Periodontol. 2007, 34, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Lang, N.P.; Tonetti, M.S. Periodontal risk assessment (PRA) for patients in supportive periodontal therapy (SPT). Oral Health Prev. Dent. 2003, 1, 7–16. [Google Scholar] [PubMed]
- Jepsen, S.; Caton, J.G.; Albandar, J.M.; Bissada, N.F.; Bouchard, P.; Cortellini, P.; Demirel, K.; de Sanctis, M.; Ercoli, C.; Fan, J. Periodontal manifestations of systemic diseases and developmental and acquired conditions: Consensus report of workgroup 3 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S237–S248. [Google Scholar] [CrossRef]
- Shin, Y.J.; Choung, H.W.; Lee, J.H.; Rhyu, I.C.; Kim, H.D. Association of periodontitis with Oral Cancer: A Case-Control Study. J. Dent. Res. 2019. [Google Scholar] [CrossRef]
- Malinowski, B.; Węsierska, A.; Zalewska, K.; Sokołowska, M.M.; Bursiewicz, W.; Socha, M.; Ozorowski, M.; Pawlak-Osińska, K.; Wiciński, M. The role of Tannerella forsythia and Porphyromonas gingivalis in pathogenesis of esophageal cancer. Infect. Agent. Cancer 2019. [Google Scholar] [CrossRef]
- Shiau, H.J.; Reynolds, M.A. Sex differences in destructive periodontal disease: Exploring the biologic basis. J. Periodontol. 2010, 81, 1505–1517. [Google Scholar] [CrossRef]
- Prakash, P.; Rath, S.; Mukherjee, M.; Malik, A.; Boruah, D.; Sahoo, N.K.; Dutta, V. Comparative evaluation of the marginal gingival epithelium in smokers and nonsmokers: A histomorphometric and immunohistochemical study. Int. J. Periodontics Restor. Dent. 2014, 4, 781–786. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Calsina, G.; Ramón, J.M.; Echeverría, J.J. Effects of smoking on periodontal tissues. J. Clin. Periodontol. 2002, 29, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Lindhe, J.; Lang, N.P.; Karring, T.; Berglundh, T.; Giannobile, W.V. Clinical Periodontology and Implant Dentistry, 5th ed.; Blackwell: Oxford, UK, 2008. [Google Scholar]
- Sreedevi, M.; Ramesh, A.; Dwarakanath, C. Periodontal status in smokers and nonsmokers: A clinical, microbiological, and histopathological study. Int. J. Dent. 2012, 2012, 571590. [Google Scholar] [CrossRef] [PubMed]
| Mean (sd) | |
|---|---|
| PPD (mm) | 3.46 (1.79) |
| CAL loss (mm) | 4.03 (1.84) |
| Gingival recession (≠ 0) | 2.39 (1.73) |
| % | |
| Gingival recession = 0 | 74.6 |
| Bleeding on probing | 49.0 |
| Mean Percentage of Inflammatory Infiltrates | Mean Percentage of Vascular Area | |
|---|---|---|
| Overall Raw Weighted Mean * | 4.38% | 0.24% |
| Gender Female Male p-value | 6.29% [3.21; 12.30] 2.28% [0.86; 6.04] 0.020 | 0.47% [0.22; 0.93] 0.58% [0.22; 1.54] - |
| Age (median) <51 years old ≥51 years old p-value | 3.21% [1.16; 8.88] 4.46% [2.54; 7.82] - | 0.60% [0.22; 1.65] 0.46% [0.23; 0.86] 0.006 |
| Smoking habit Non-smokers Smokers p-value | 4.56% [2.06; 10.09] 3.14% [1.44; 6.85] 0.048 | 0.55% [0.32; 1.31] 0.41% [0.16; 1.06] 0.008 |
| PPD (median) <9 ≥9 p-value | 3.61% [1.66; 7.85] 3.97% [1.84; 8.58] - | 0.55% [0.28; 1.07] 0.59% [0.18; 1.31] - |
| Plaque Yes No p-value | 3.01% [2.02; 4.49] 4.76% [1.53; 14.77] 0.14 | 0.36% [0.17; 0.77] 0.74% [0.30; 1.77] 0.0005 |
| Pus Yes No p-value | 3.67% [1.29; 10.42] 3.90% [2.00; 7.61] - | 0.51% [0.12; 2.23] 0.53% [0.39; 0.71] - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buffoli, B.; Garzetti, G.; Calza, S.; Scotti, E.; Borsani, E.; Cappa, V.; Rimondini, L.; Mensi, M. Periodontitis Stage III–IV, Grade C and Correlated Factors: A Histomorphometric Study. Biomedicines 2019, 7, 43. https://doi.org/10.3390/biomedicines7020043
Buffoli B, Garzetti G, Calza S, Scotti E, Borsani E, Cappa V, Rimondini L, Mensi M. Periodontitis Stage III–IV, Grade C and Correlated Factors: A Histomorphometric Study. Biomedicines. 2019; 7(2):43. https://doi.org/10.3390/biomedicines7020043
Chicago/Turabian StyleBuffoli, Barbara, Gianluca Garzetti, Stefano Calza, Eleonora Scotti, Elisa Borsani, Veronica Cappa, Lia Rimondini, and Magda Mensi. 2019. "Periodontitis Stage III–IV, Grade C and Correlated Factors: A Histomorphometric Study" Biomedicines 7, no. 2: 43. https://doi.org/10.3390/biomedicines7020043
APA StyleBuffoli, B., Garzetti, G., Calza, S., Scotti, E., Borsani, E., Cappa, V., Rimondini, L., & Mensi, M. (2019). Periodontitis Stage III–IV, Grade C and Correlated Factors: A Histomorphometric Study. Biomedicines, 7(2), 43. https://doi.org/10.3390/biomedicines7020043









