Alcohol Interaction with Cocaine, Methamphetamine, Opioids, Nicotine, Cannabis, and γ-Hydroxybutyric Acid
Abstract
1. Introduction
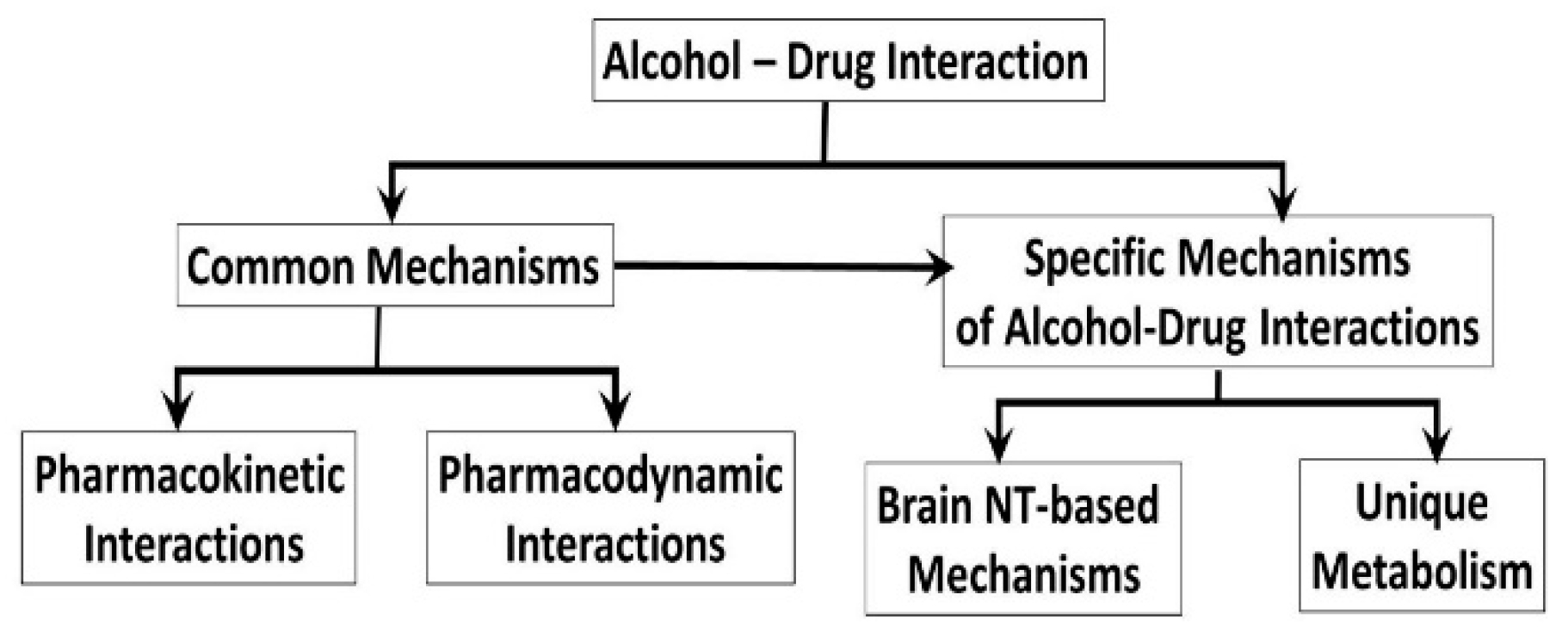
2. Common Mechanisms of the Alcohol-Drug Interactions
2.1. Pharmacokinetic Mechanisms of Alcohol-Drug Interactions
- i.
- ii.
- Acute low dose of alcohol exposure in alcohol-naïve subjects is metabolized to acetaldehyde mostly by ADHs, but acute high-dose or chronic alcohol exposure may be metabolized by both ADH and CYP enzymes listed above. CYP enzymes remain induced in alcohol abstinent subjected chronically exposed to alcohol [11].
- iii.
- In alcohol-naïve subjects using alcohol and another drug, acute dose of alcohol may compete with the drug for the same set of CYP enzymes and inhibit a drug’s metabolism. This may enhance the drug’s availability and ensuing increase in the harmful side effects from the drug [29].
- iv.
- In recently abstinent chronic alcohol drinker, many drug-metabolizing CYPs remain induced, thus decreasing the drug’s availability and diminishing its effects for several weeks after drinking ceased. This suggests that a recently abstinent chronic drinker may need higher doses of medications than those required by nondrinkers to achieve therapeutic levels of certain drugs [30].
- v.
- CYP enzymes activated by chronic alcohol consumption transform some drugs into toxic metabolites that can damage the liver or other organs [11].
2.2. Pharmacodynamics of Alcohol-Drug Interactions
- i.
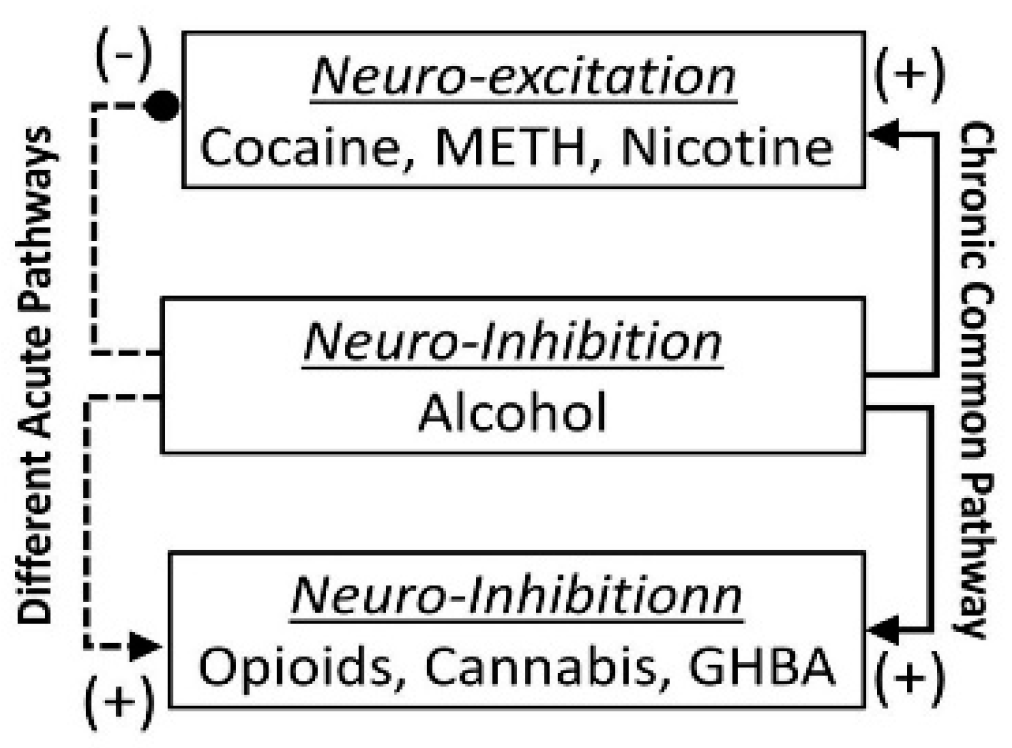
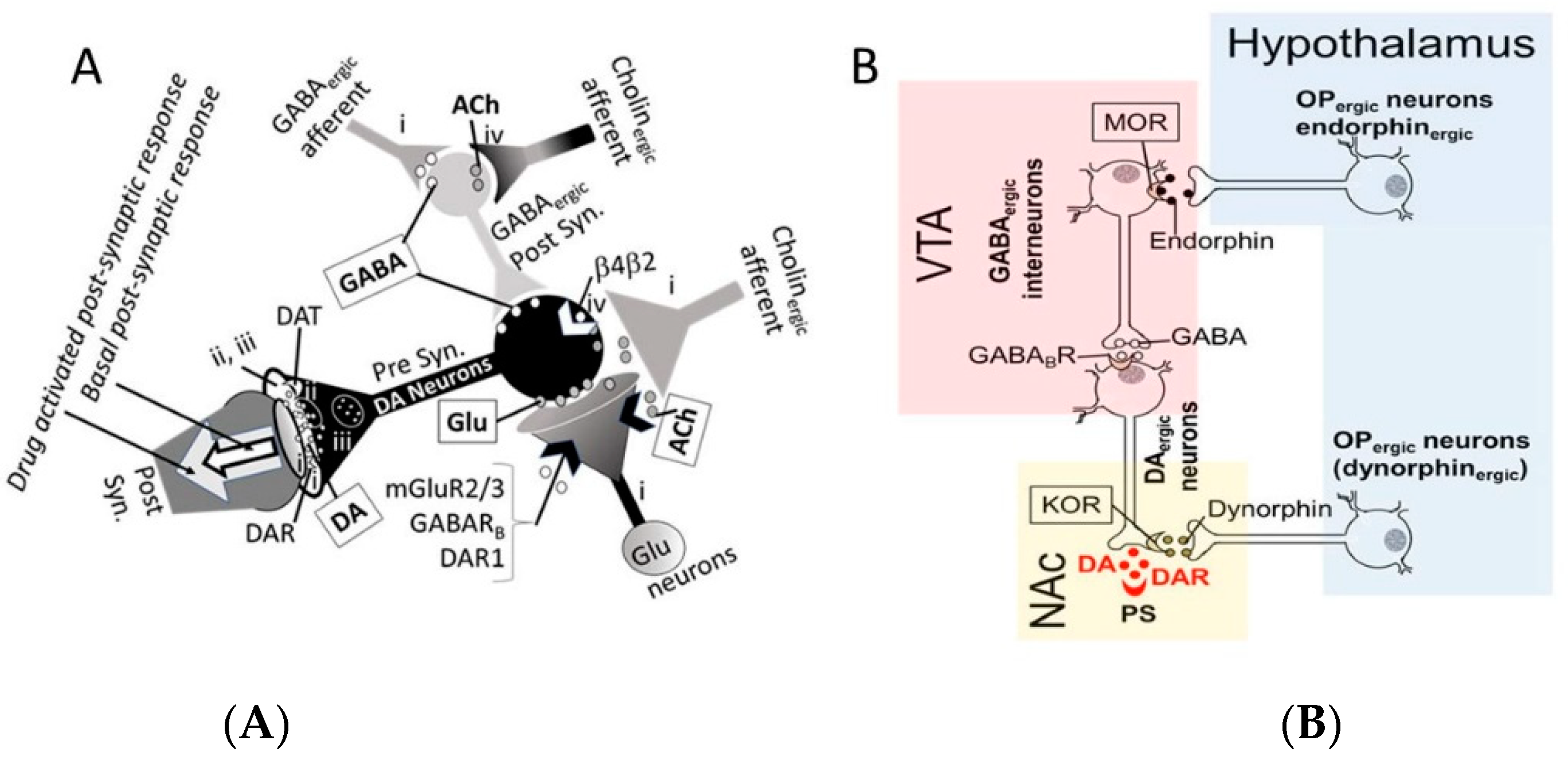
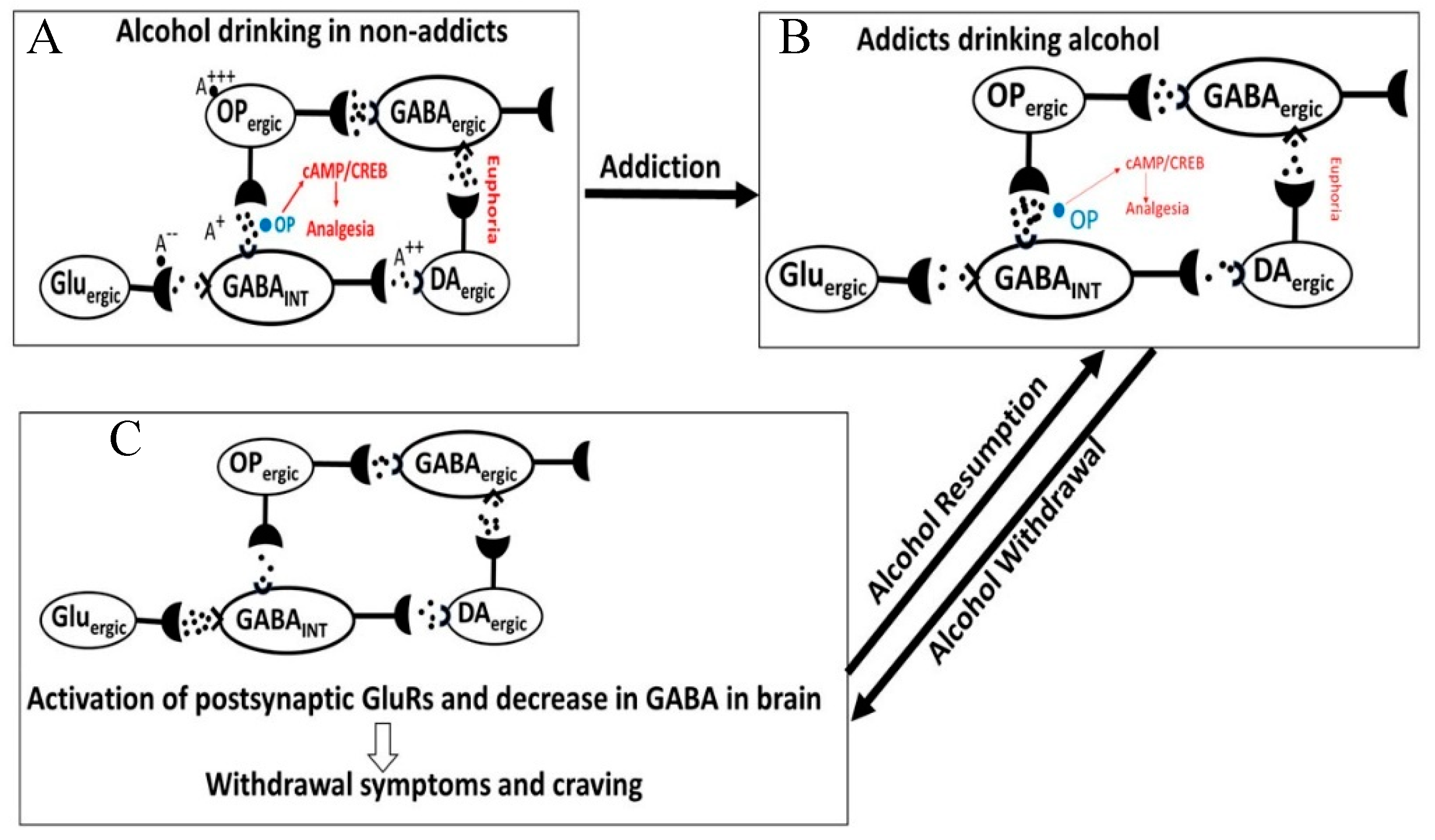
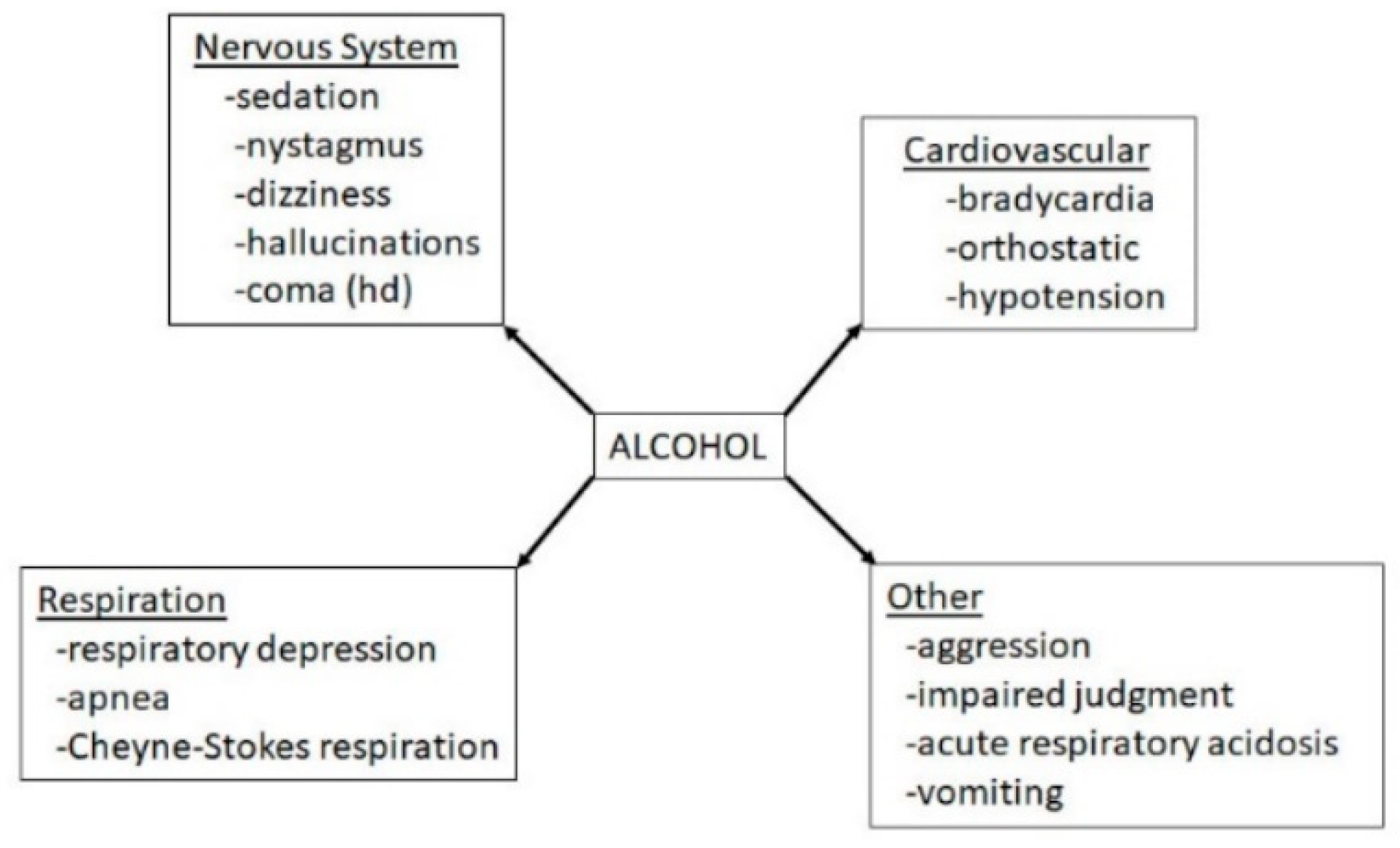
- The acute neuro-inhibitory effects of the alcohol, opioids, cannabis and GHBA are caused via development of inhibitory postsynaptic potential (IPSP). The acute neuro-excitatory effects of cocaine, METH, and nicotine cause development of excitatory postsynaptic potential (EPSP) [11]. Therefore, acute alcohol exposure may attenuate the effects of neuro-stimulatory drugs but augments the effects of neuro-inhibitory drugs (Figure 4A). As an example, alcohol cause neuro-inhibition by inducing Cl− influx into the neurons [47], resulting in development of neural membrane IPSP [48,49] that antagonizes the effects of stimulatory drugs, but additively or synergistically augment the effects of inhibitory drug.
- ii.
- Chronic alcohol and drug exposure results in in development of tolerance and addiction via a common addiction mechanism (Figure 3). Therefore, chronic alcohol exposure may negatively impact addictive effects of both excitatory and inhibitory drugs.
- iii.
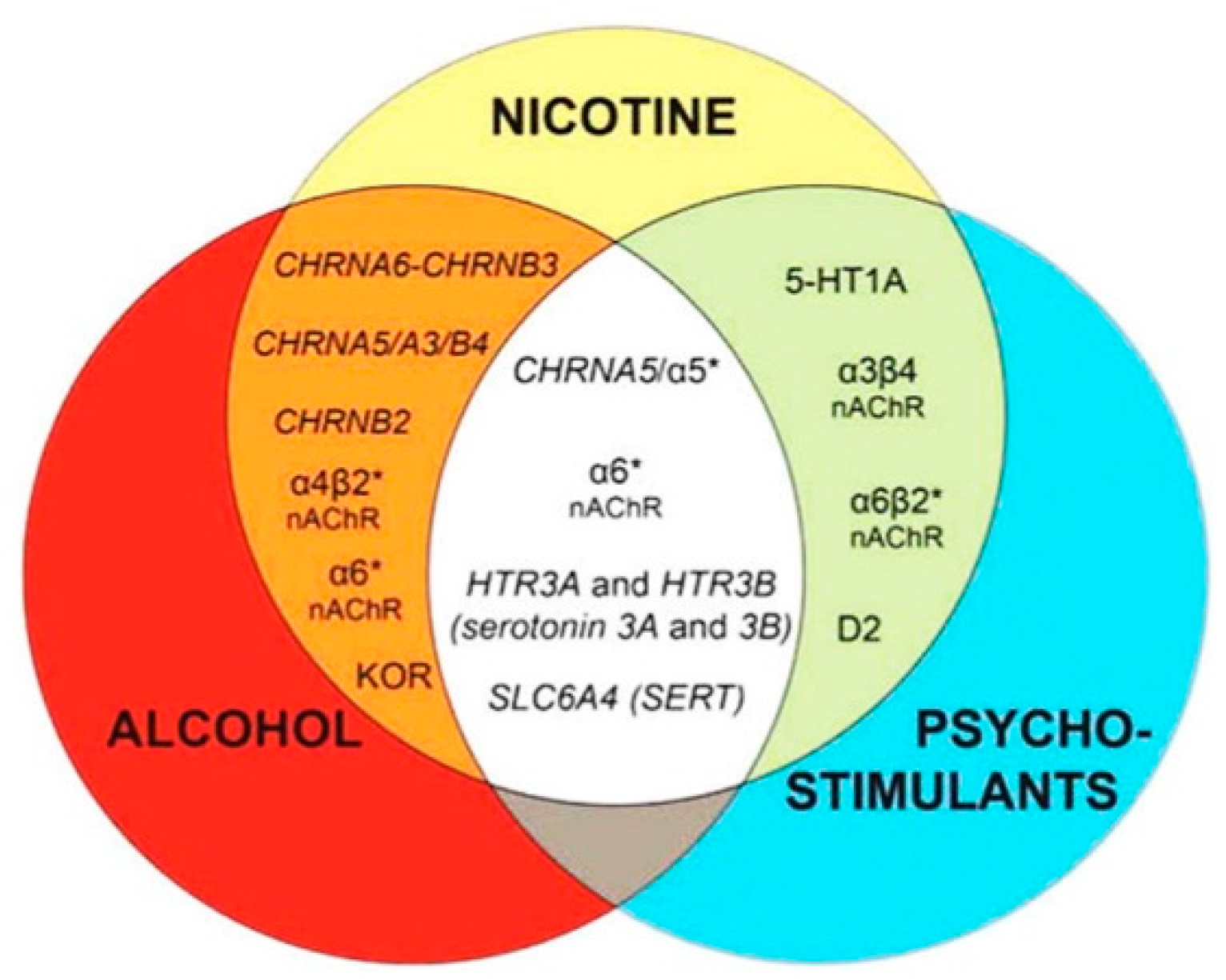
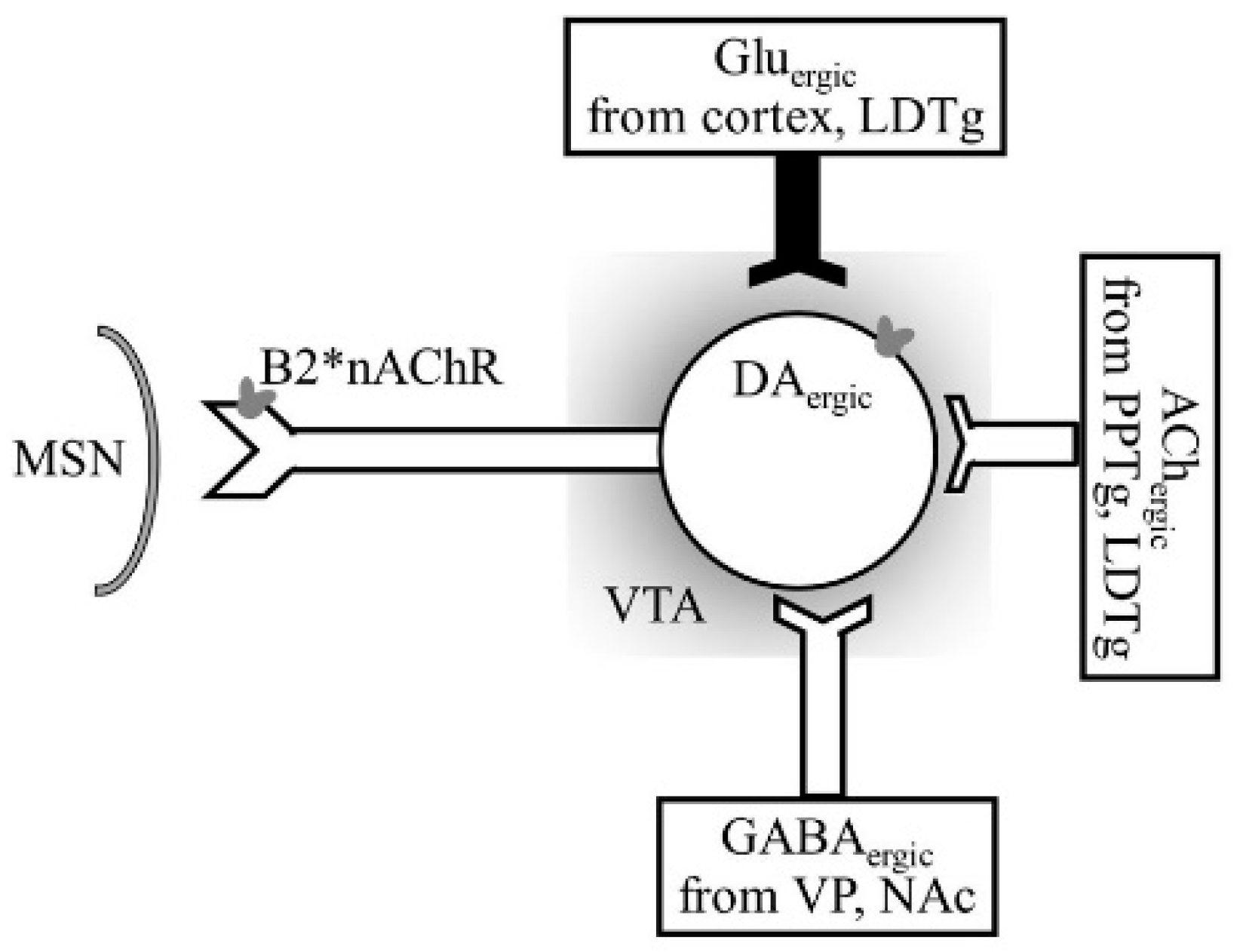
- Figure 4 shows receptor overlap in development of alcohol, nicotine, and psycho-stimulant- (such as cocaine and METH) dependence. The genes listed in Figure 4 have received strong statistical and biological (knockout studies) support for association with multiple substances [50]. The nAChR gene variants such as gene cluster CHRNA5/A3/B4 encoding α3, β4, and α5 nAChR are associated strongly with poly-drug addiction [51,52,53,54]. The possible role of nAChR in alcohol dependence is further validated by the observation that varenicline, a partial agonist at α4β2 nAChRs and a full agonist at the α7 nAChR [55] reduced alcohol craving and total alcohol consumption in patients with alcohol use disorders [56,57].
3. Specific Alcohol-Drug Interactions
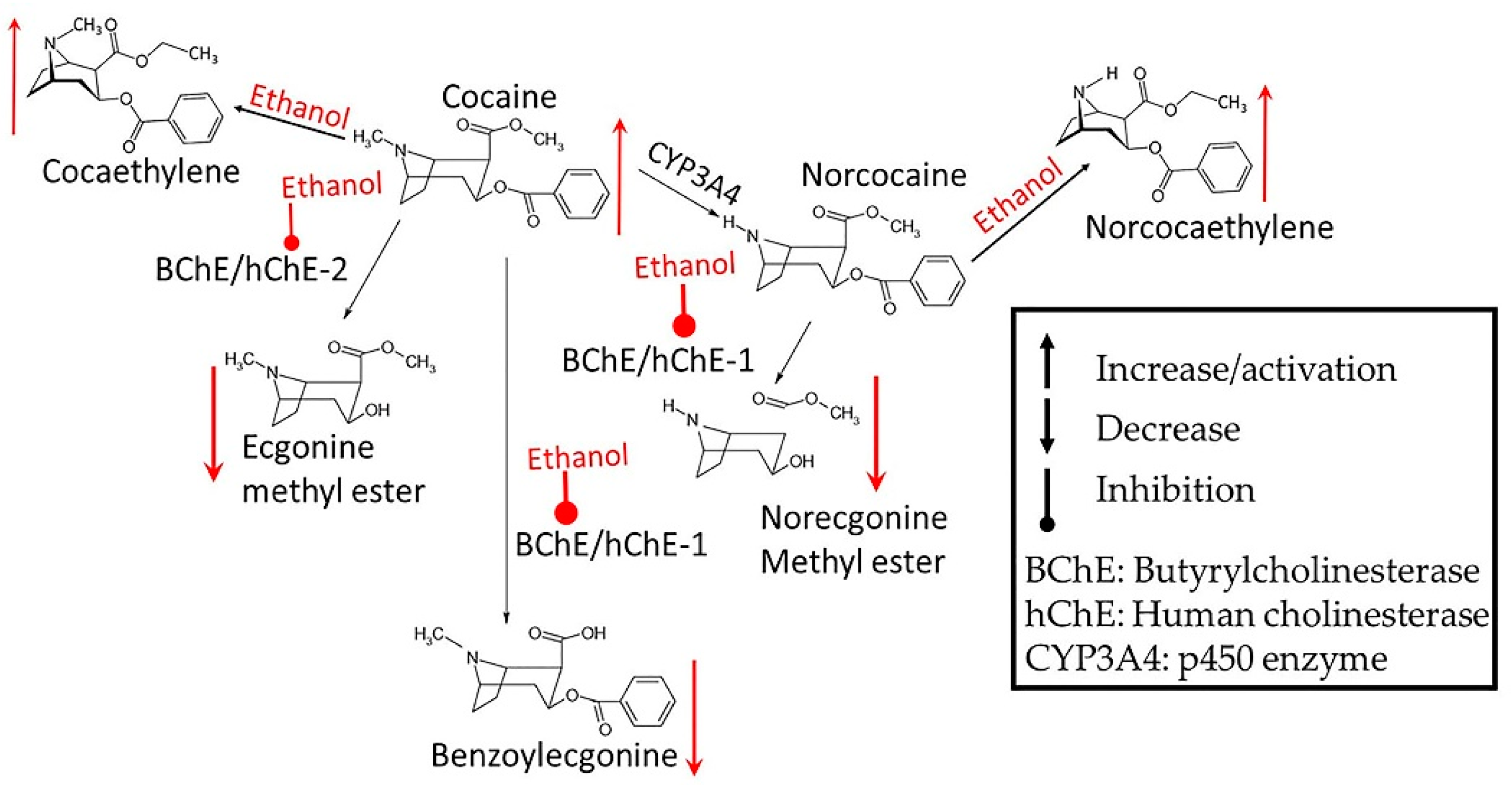
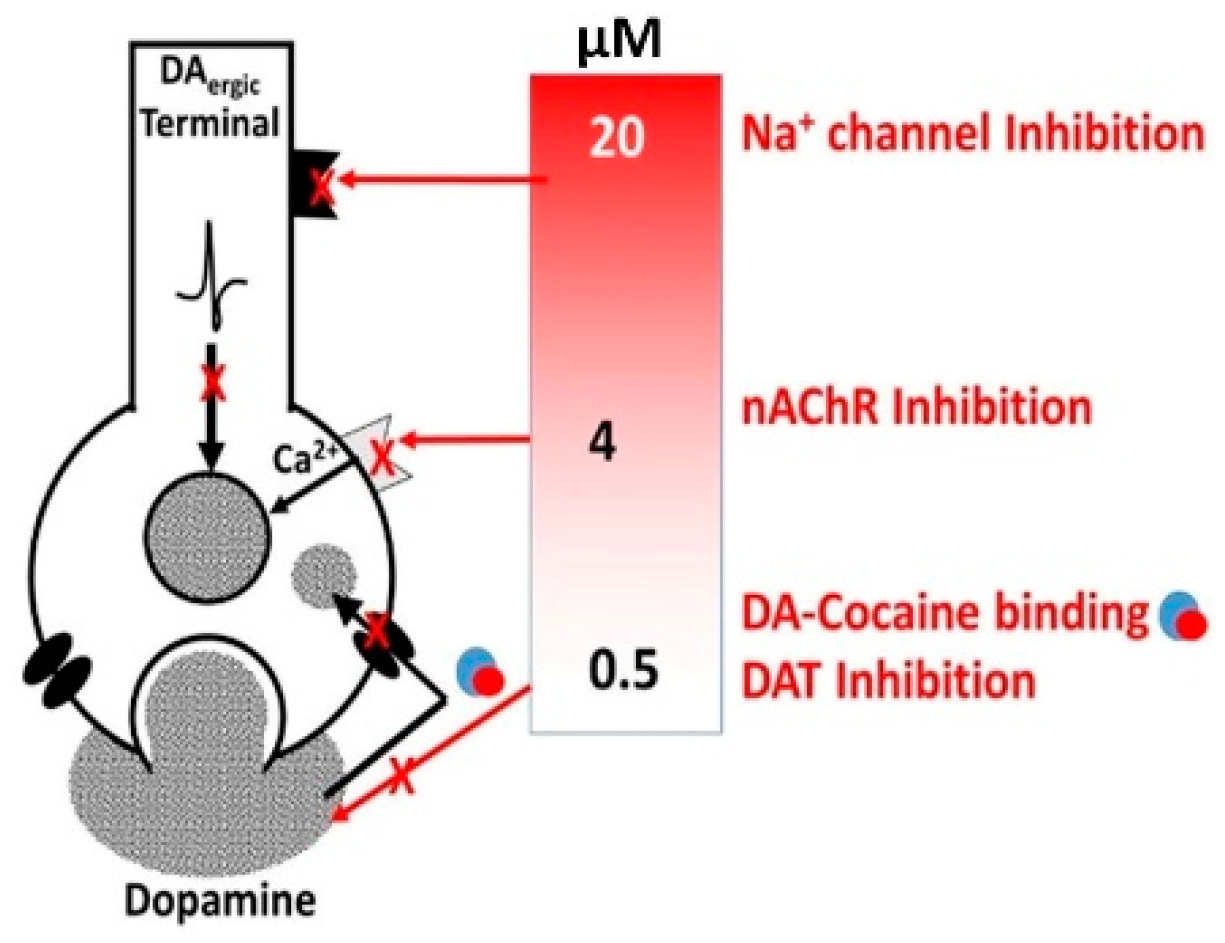
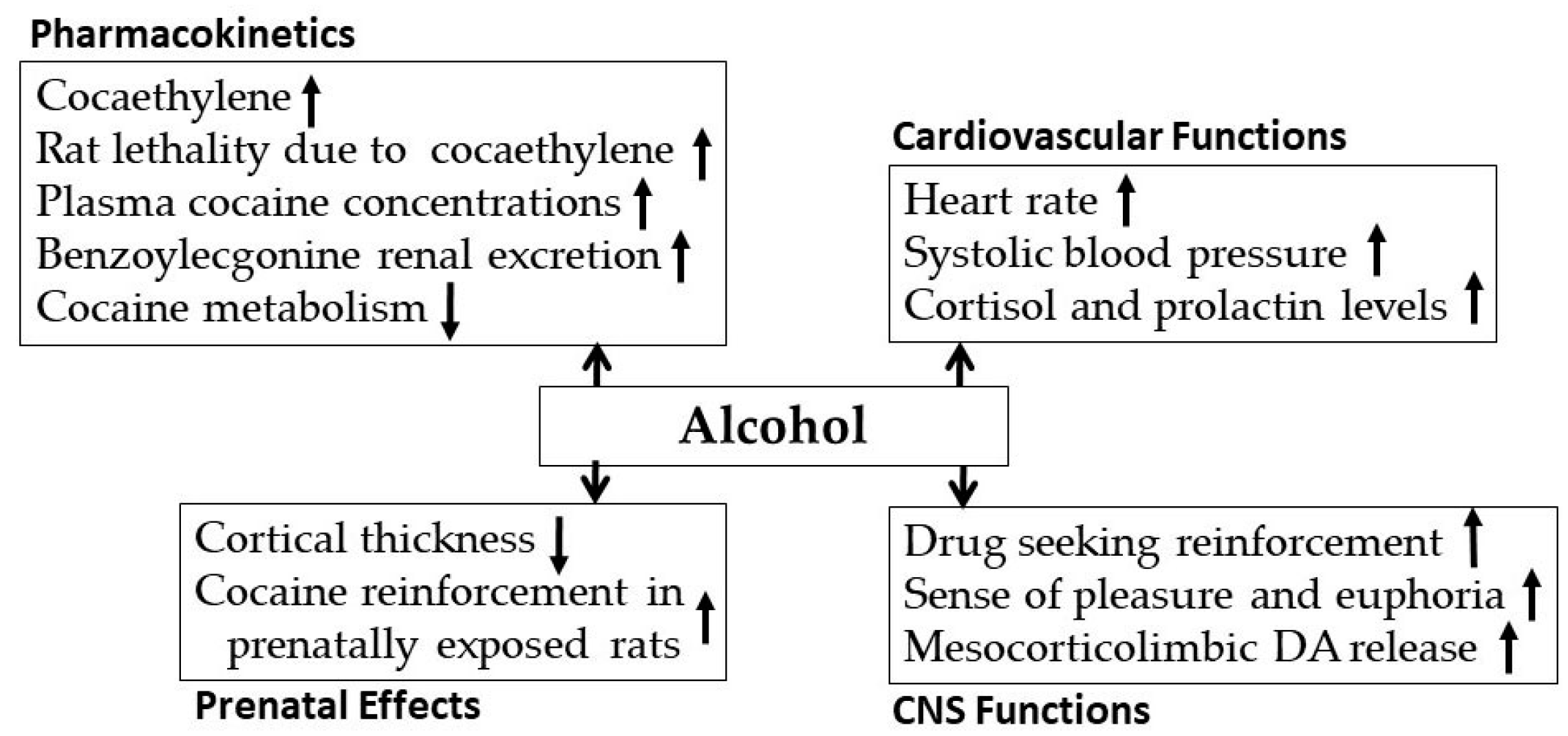
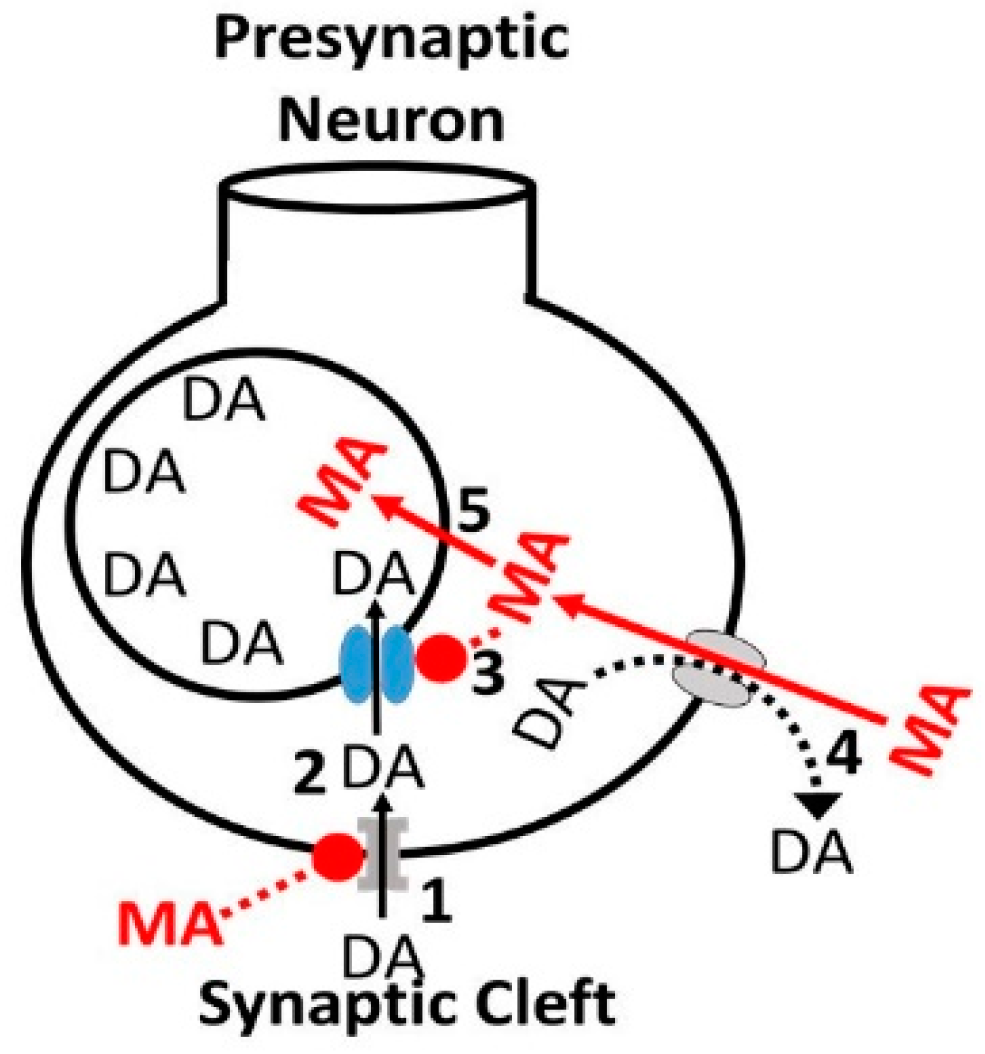
3.1. Alcohol-Cocaine Interaction
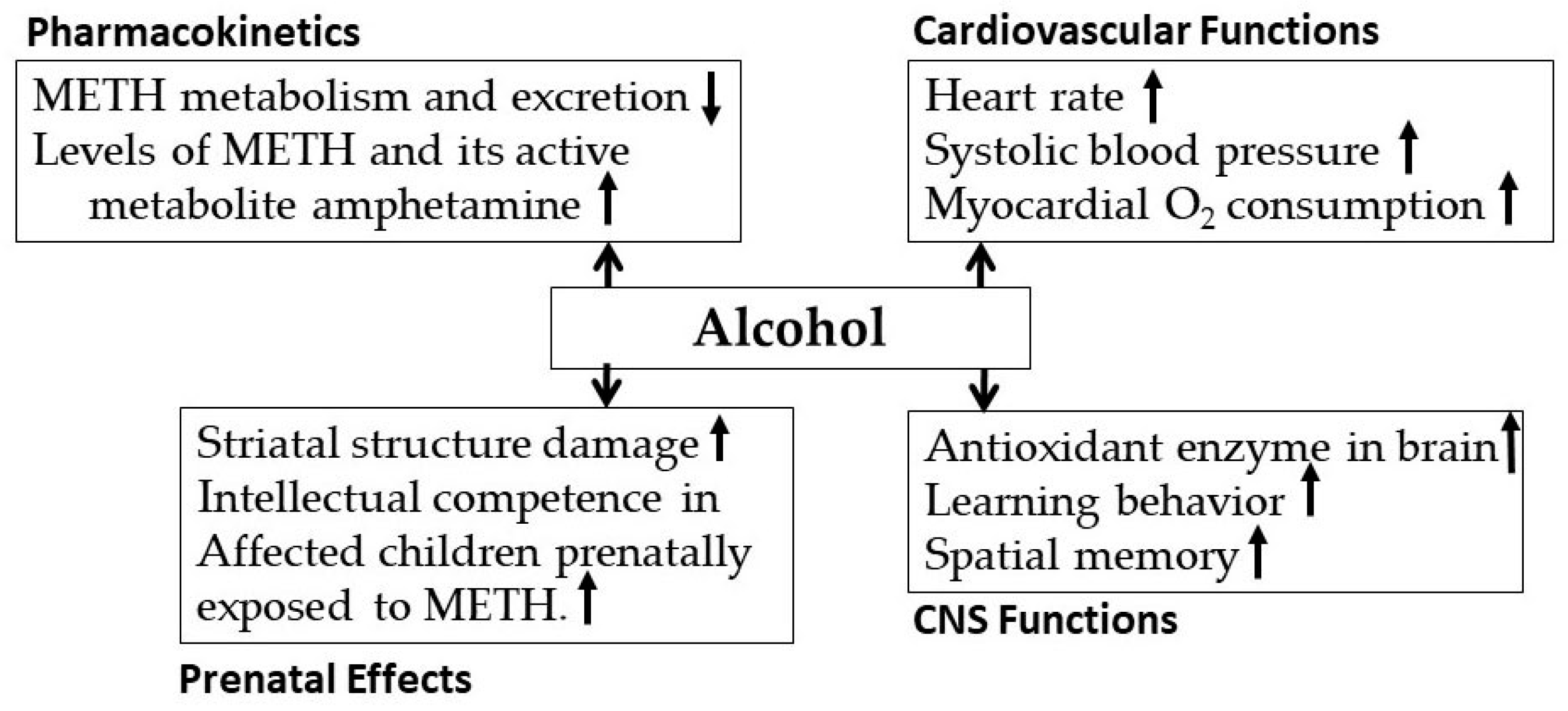
3.2. Alcohol-Methamphetamine Interactions
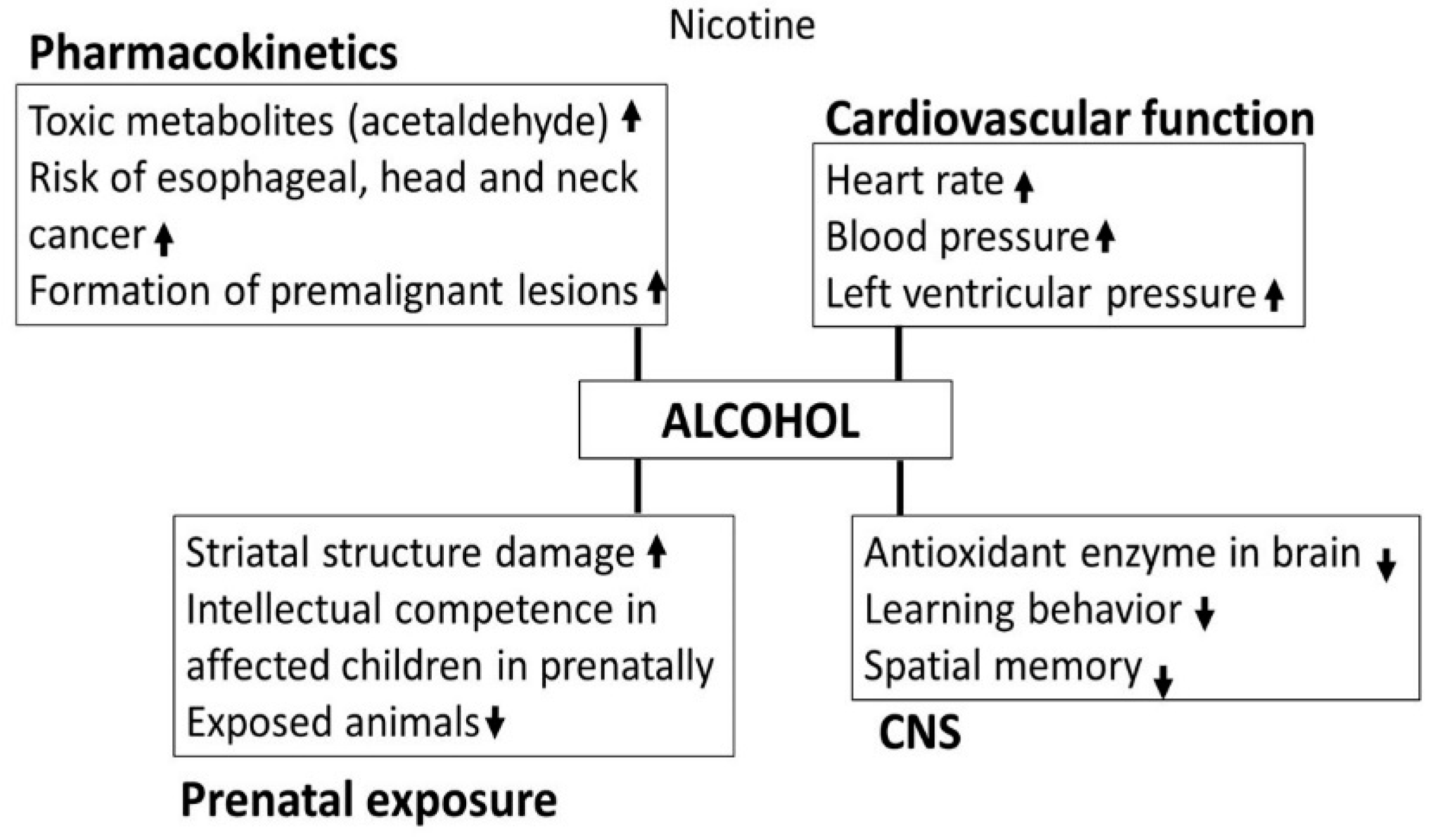
3.3. Nicotine
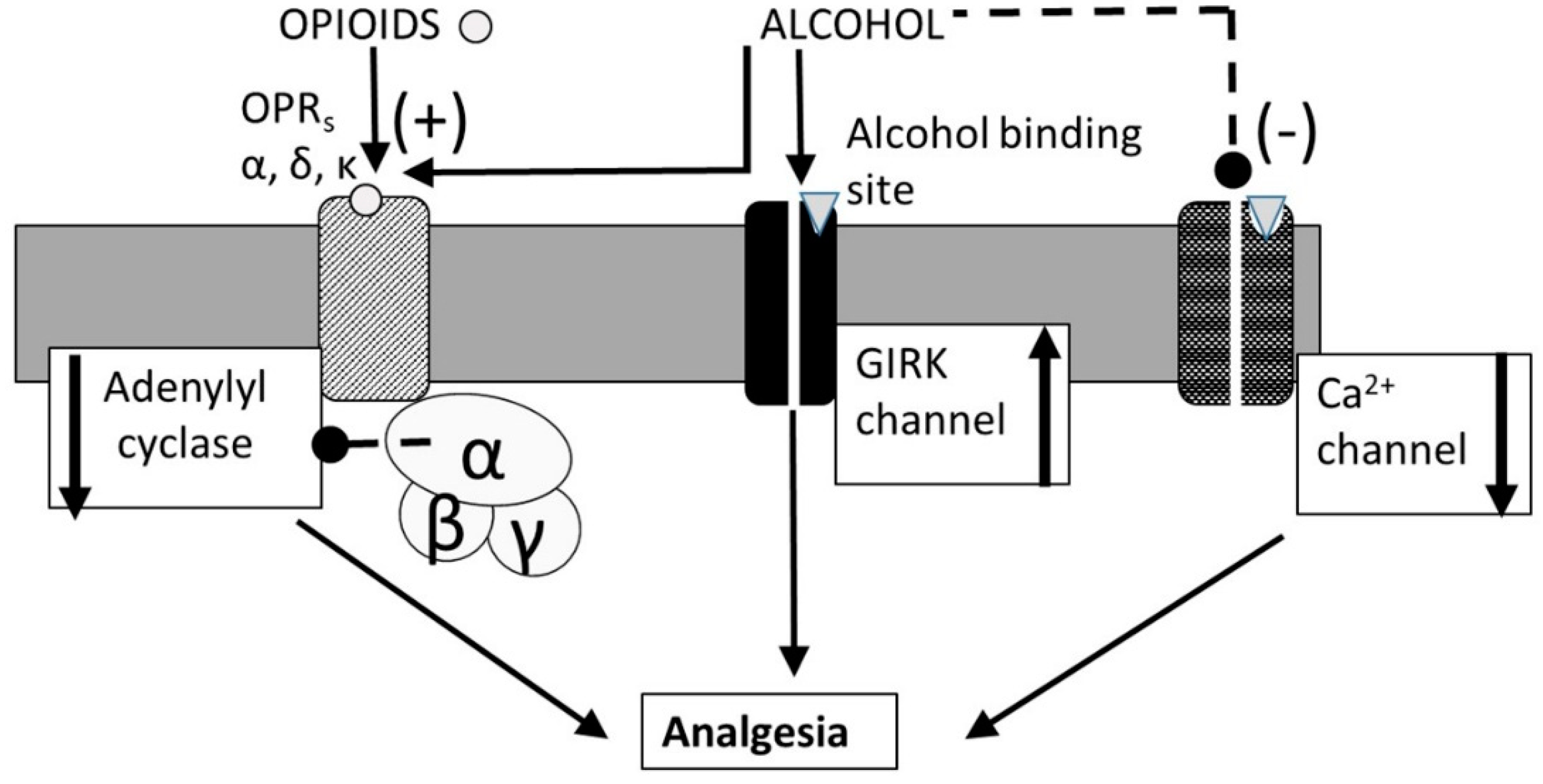
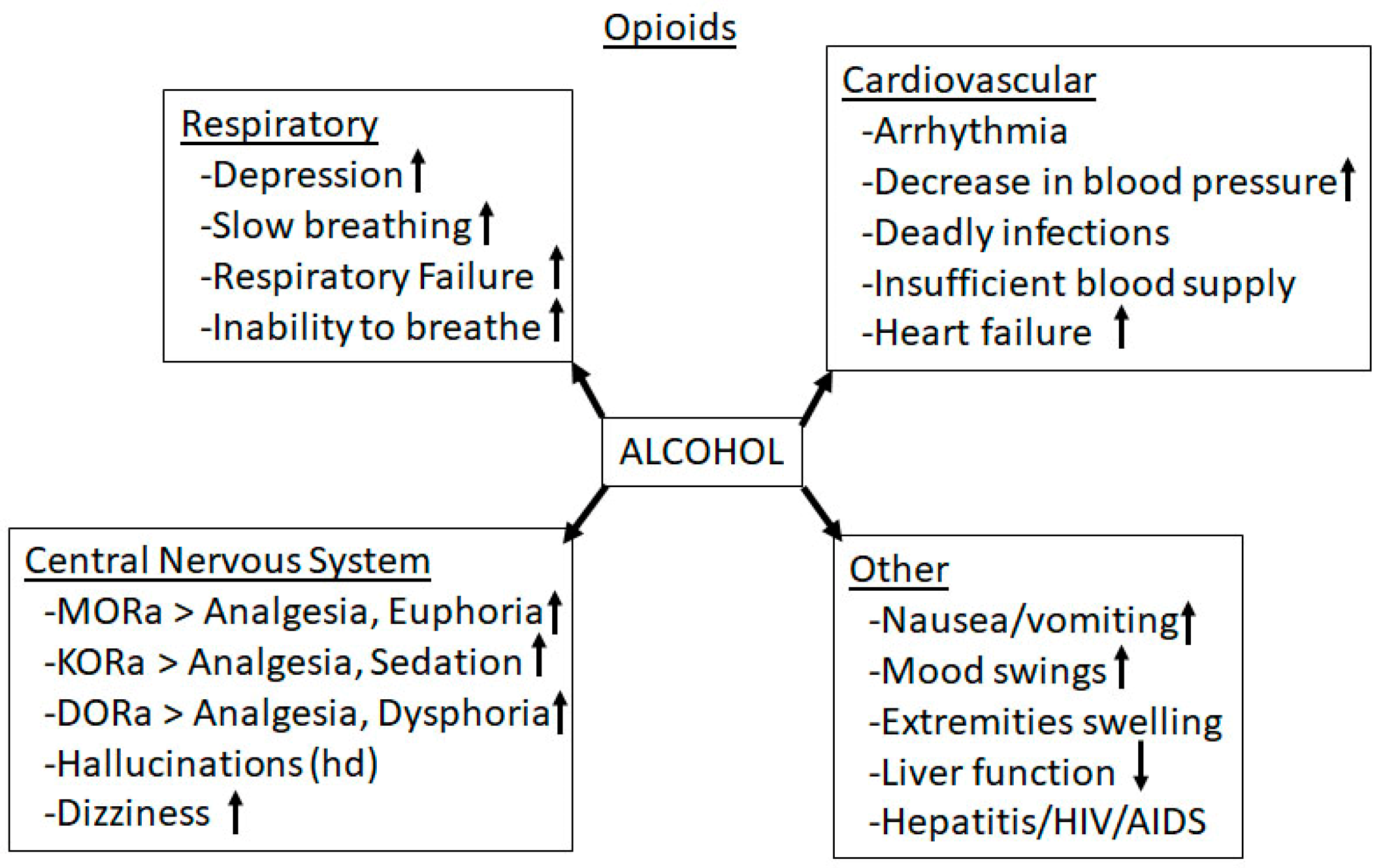
3.4. Alcohol-Opioid Interactions
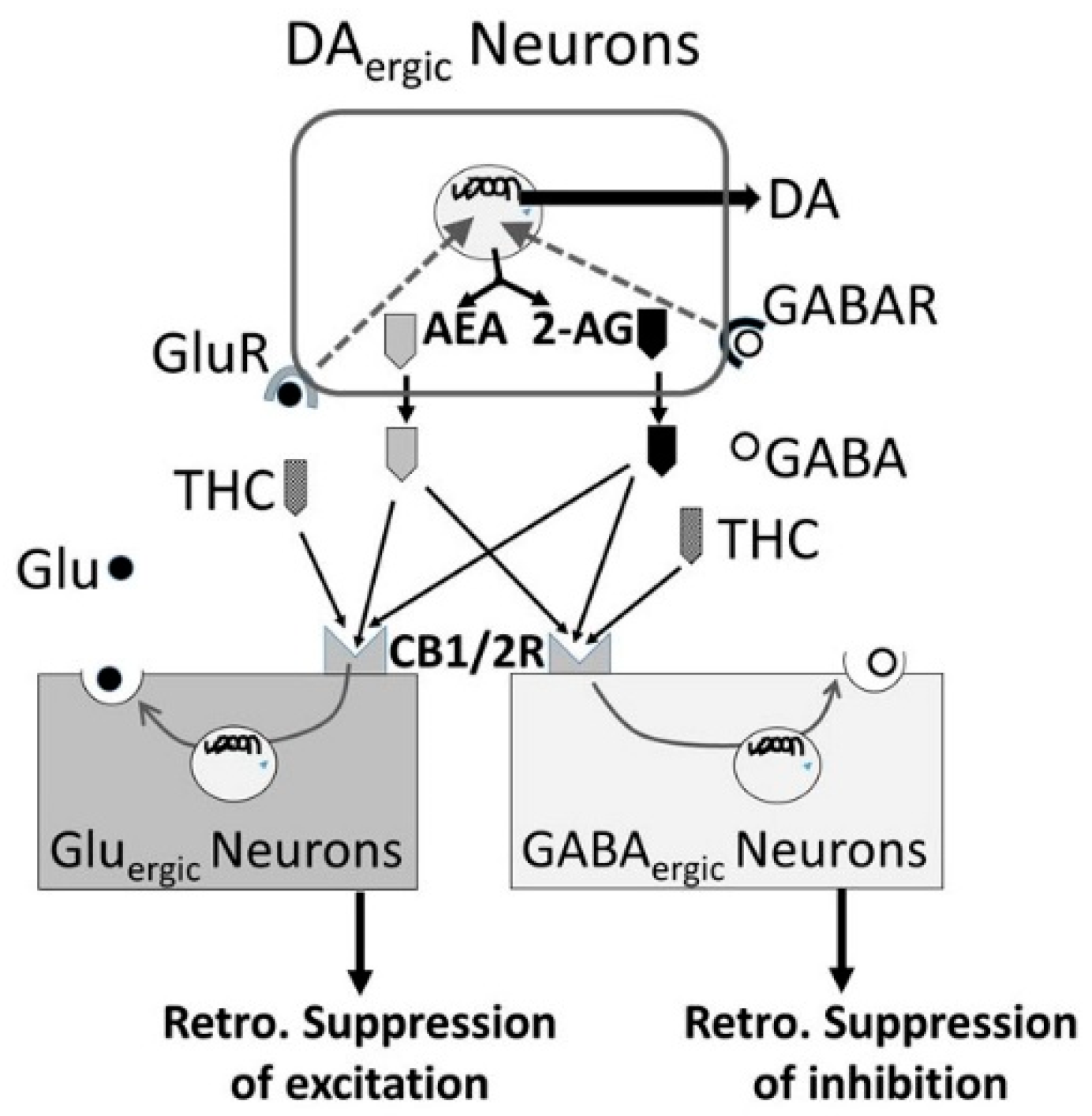
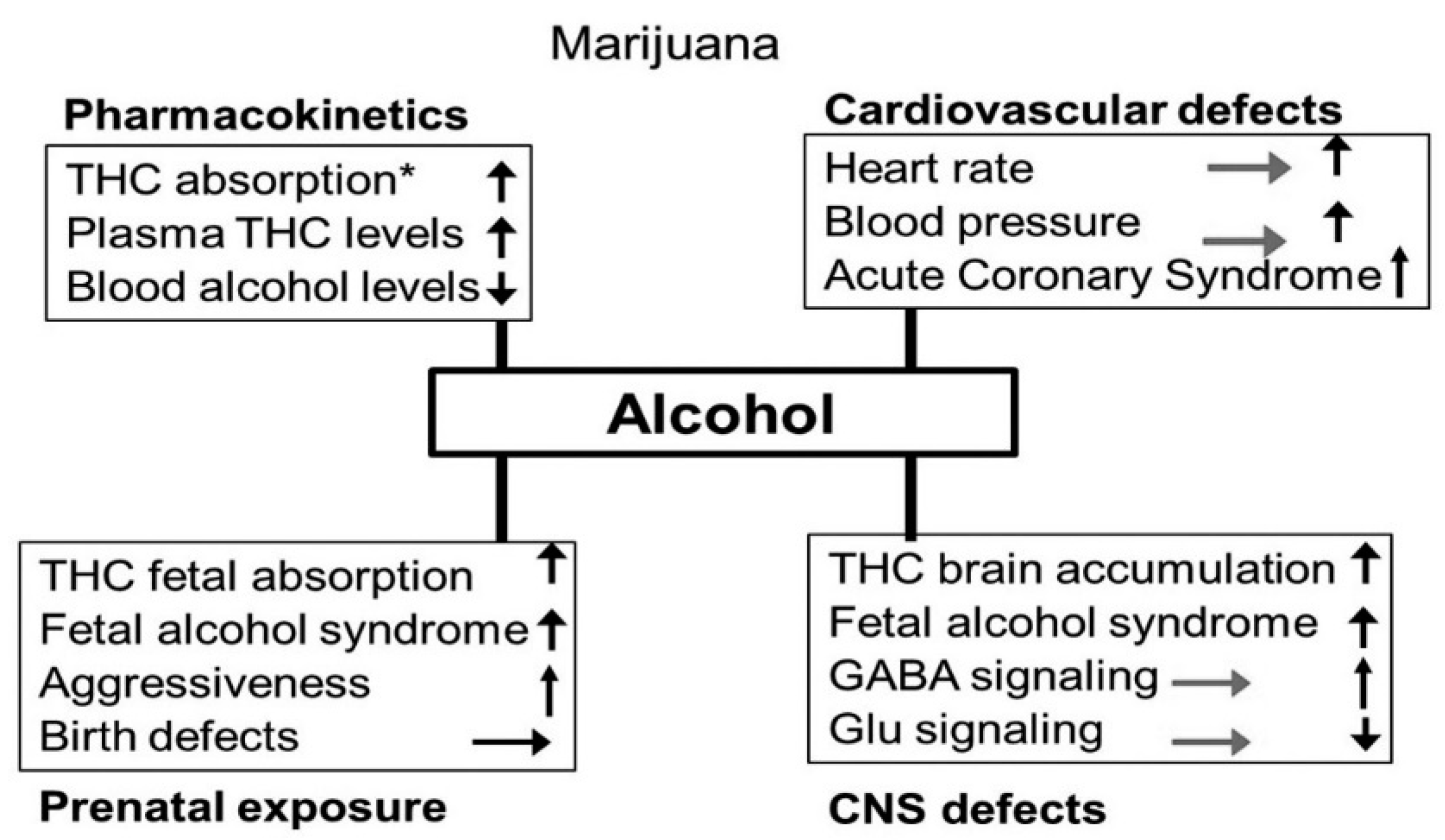
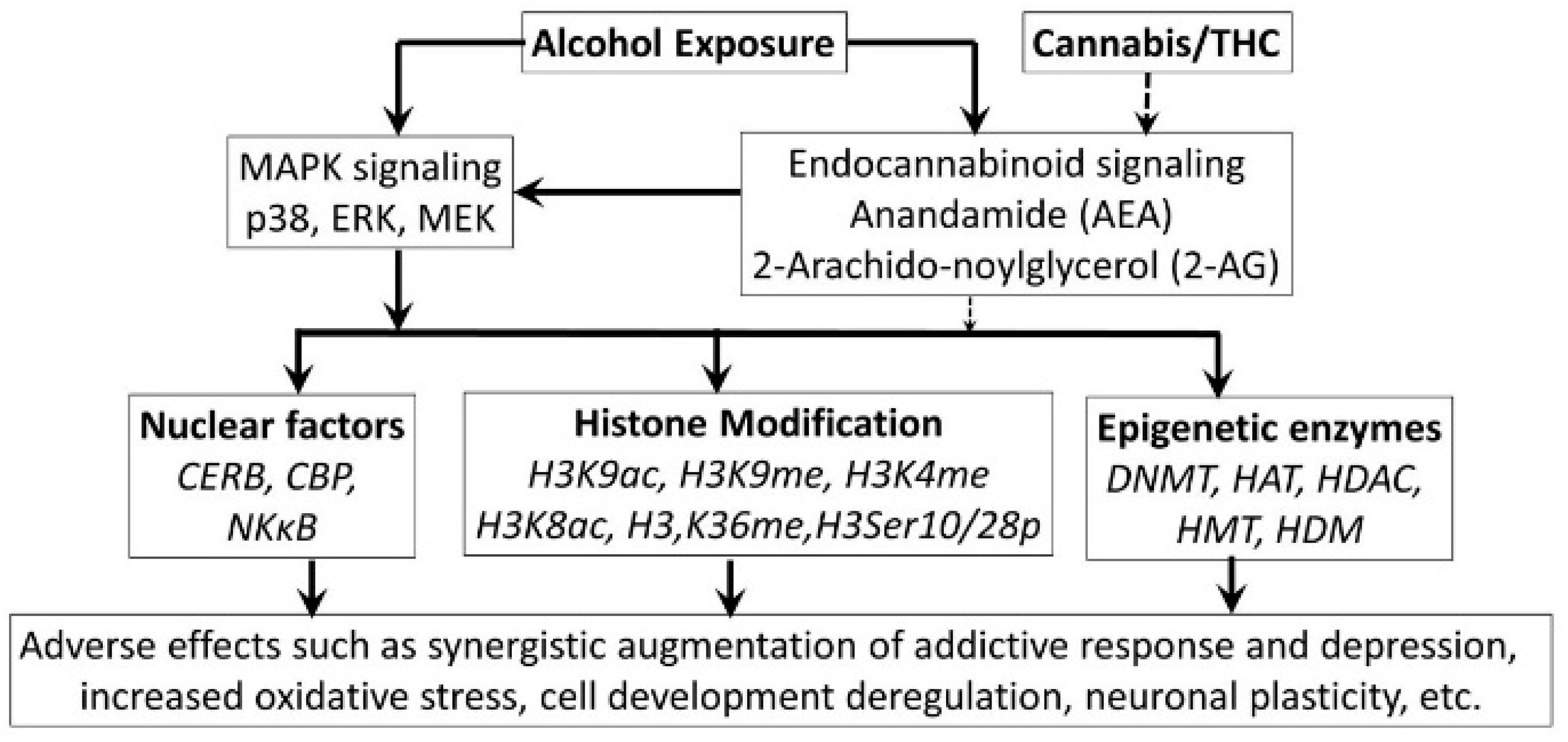
3.5. Alcohol-Cannabis Interactions
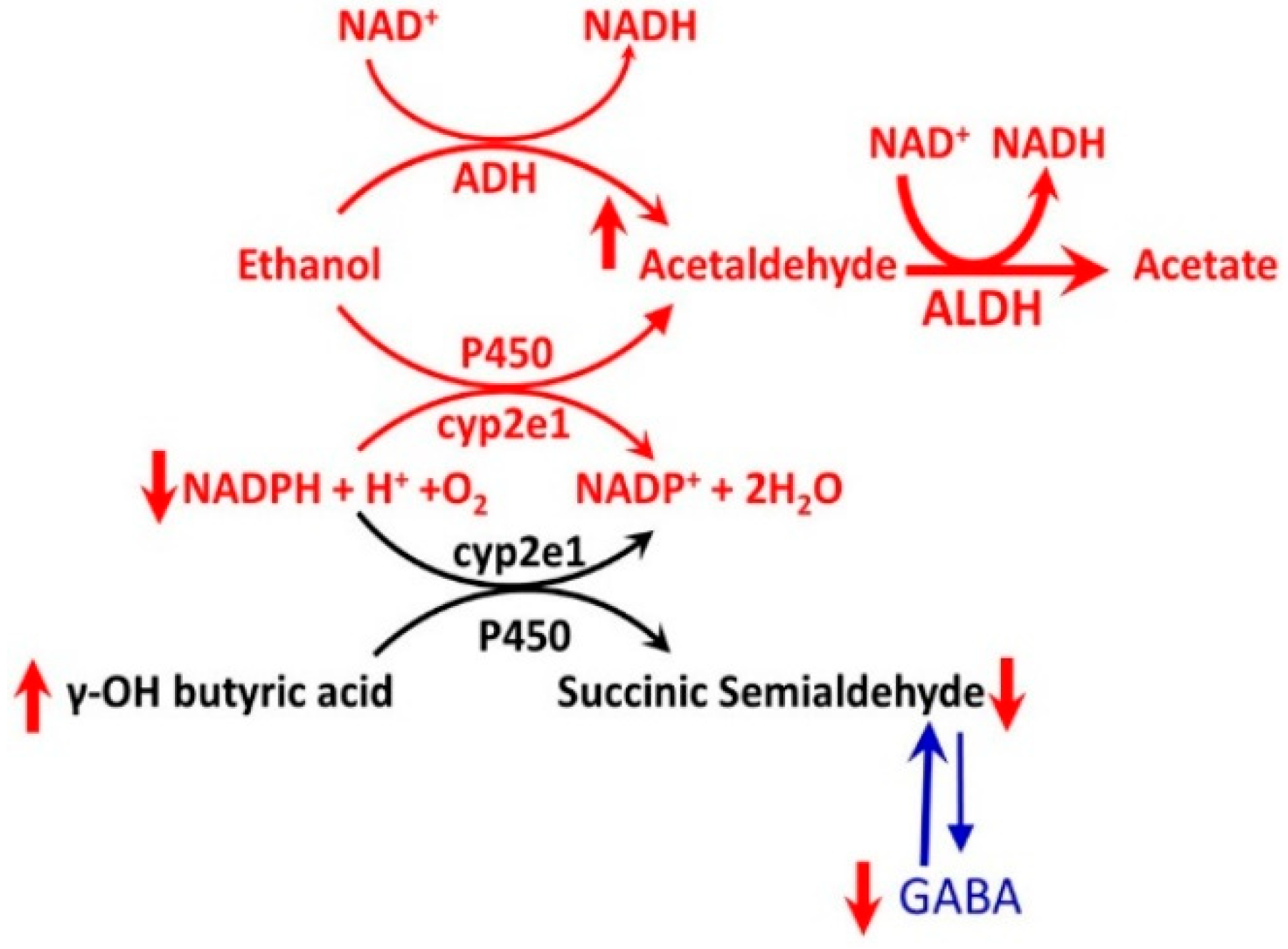
3.6. Alcohol-GHBA Interactions
4. Conclusions
Conflicts of Interest
Abbreviations
| 2-AG | 2-Arachidonoylglycerol |
| 5-HT | Serotonin |
| A(+) | alcohol’s +ve effects |
| A(−) | alcohol’s −ve effects |
| AA | arachidonic acid |
| AC | adenylyl cyclase |
| ACh | Acetylcholine |
| AChergic | ACh releasing neurons |
| ADH | alcohol dehydrogenase |
| ALDH | acetaldehy6de dehydrogenase |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| AUC | area under curve |
| AUD | alcohol use disorder |
| cAMP | cyclic adenosine monophosphate |
| CL | clearance |
| Cmax | maximum concentration |
| COMT | catechol-O-methyl-transferase |
| COX | Cyclooxygenase |
| CYP | cytochrome P450 |
| DA | Dopamine |
| DA | dopamine} |
| DAG | Diacylglycerol |
| DAGL | diacylglycerol lipase |
| DAR1 | dopamine receptor 1 |
| DAT | dopamine transporter |
| DOR | delta OPRs |
| DPDEP | D-Pen2, D-Pen5 enkephalin, DOR agonist |
| EAA | excitatory amino acids |
| eCB | Endocannabinoid |
| eOP | endogenous opioids |
| ERK | extracellular-signal-regulated kinase |
| GABA | γ-aminobutyric acid |
| GABABR | GABA B receptor |
| GABAergic | GABA releasing neurons |
| GHB | γ-hydroxybutyric acid |
| GHBD | GHB dehydrogenase |
| GIRK | G protein-coupled inwardly-rectifying potassium channels |
| Glu | Glutamate |
| Gluergic | glutamate releasing neurons |
| Gly | Glycine |
| IP3 | inositol trisphosphate |
| KOR | kappa OPRs |
| LDTg | laterodorsal tegmental nucleus |
| LPS | Lipopolysaccharide |
| METH | Methamphetamine |
| mGluR2/3 | metabolic Glu receptors 2/3 |
| MOR | mu OPRs |
| NAc | nucleus accumbens |
| nAChR | nicotinic ACh receptor |
| NAPQI | N-acetyl-p-benzoquinone imine |
| NE | Noradrenaline |
| NMDA | N-methyl-d-Aspartate receptors |
| OP | Opioid |
| OPR | opioid receptors |
| OPRM1 | A118G variant of the MOR1 |
| PIP2 | poly inositol diphosphate |
| pptg | pedunculopontine tegmental nucleus |
| SERT | serotonin transporter |
| sOP | synthetic opioids |
| SSA | succinic semialdehyde |
| T | elimination half-life |
| THC | Δ9-Tetrahydrocannabinol |
| tmax | time to Cmax |
| TMU | 1,3,7-trimethyluric acid |
| U50488H | KOR agonist |
| VGSC | voltage gated sodium ion (Na+) channel |
| VGCC | voltage gated calcium ion (Ca2+) channel |
| VTA | ventral tegmental area |
| α7 and α4β2 | nicotinic receptor subtypes |
References
- Peacock, A.; Leung, J.; Larney, S. Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 2018, 113, 1905–1926. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Kaufman, E. The abuse of multiple drugs. I. Definition, classification, and extent of problem. Am. J. Drug Alcohol Abus. 1976, 3, 279–292. [Google Scholar] [CrossRef]
- Karjalainen, K.; Kuussaari, K.; Kataja, K.; Tigerstedt, C.; Hakkarainen, P. Measuring Concurrent Polydrug Use in General Populations: A Critical Assessment. Eur. Addict. Res. 2017, 23, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Pakula, B.; McDonald, S.; Stockwell, Y. Simultaneous use of alcohol and cocaine: A qualitative investigation. J. Subst. Use 2009, 14, 101–112. [Google Scholar] [CrossRef]
- Higgins, S.T.; Sigmon, S.C.; Wong, C.J.; Heil, S.H.; Badger, G.J.; Donham, R.; Dantona, D.L.; Anthony, S. Community reinforcement therapy for cocaine-dependent outpatients. Arch. Gen. Psychiatry 2003, 60, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Budd, R.D. Cocaine Abuse and Violent Death. Am. J. Drug Alcohol Abus. 1989, 15, 375–382. [Google Scholar] [CrossRef]
- Marzuk, P.M.; Tardiff, K.; Leon, A.C.; Stajic, M.; Morgan, E.B.; Mann, J.J. Prevalence of Recent Cocaine Use Among Motor Vehicle Fatalities in New York City. JAMA 1990, 263, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Dani, J.A.; Harris, R.A. Nicotine addiction and comorbidity with alcohol abuse and mental illness. Nat. Neurosci. 2005, 8, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Patrick, M.E.; Schulenberg, J.E.; O’malley, P.M.; Maggs, J.L.; Kloska, D.D.; Johnston, L.D.; Bachman, J.G. Age-Related Changes in Reasons for Using Alcohol and Marijuana from Ages 18 to 30 in a National Sample. Psychol. Addict. Behav. 2011, 25, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Althobaiti, Y.; Sari, Y. Alcohol Interactions with Psychostimulants: An Overview of Animal and Human Studies. J. Addict. Res. Ther. 2016, 7, 281. [Google Scholar] [CrossRef] [PubMed]
- Lemstra, M.; Bennett, N.R.; Neudorf, C.; Kunst, A.; Nannapaneni, U.; Warren, L.M.; Kershaw, T.; Scott, C.R. A meta-analysis of marijuana and alcohol use by socio-economic status in adolescents aged 10–15 years. Can. J. Public Health 2008, 99, 172–177. [Google Scholar] [PubMed]
- Thai, D.; Dyer, J.E.; Benowitz, N.L.; Haller, C.A. GHB and Ethanol Effects and Interactions in Humans. J. Clin. Psychopharmacol. 2006, 26, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Mason, W.A.; Chmelka, M.B.; Howard, B.K.; Thompson, R.W. Comorbid Alcohol and Cannabis Use Disorders Among High-Risk Youth at Intake into Residential Care. J. Adolesc. Health 2012, 53, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Rasch, R.F.; Weisen, C.A.; MacDonald, B.; Wechsberg, W.M.; Perritt, R.; Dennis, M.L. Patterns of HIV risk and alcohol use among African-American crack abusers. Drug Alcohol Depend. 2000, 58, 259–266. [Google Scholar] [CrossRef]
- George, S.; Rogers, R.D.; Duka, T. The acute effect of alcohol on decision making in social drinkers. Psychopharmacology 2005, 182, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.C.; Bang, H.; Kapiga, S.H. The association between HIV infection and alcohol use: A systematic review and meta-analysis of African studies. Sex. Transm. Dis. 2007, 34, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Arasteh, K.; Des Jarlais, D.C.; Perlis, T.E. Alcohol and HIV sexual risk behaviors among injection drug users. Drug Alcohol Depend. 2008, 95, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Van Tieu, H.; Koblin, B.A. HIV, alcohol, and non-injection drug use. Curr. Opin. HIV AIDS 2009, 4, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Stinson, F.S.; Grant, B.F.; Dawson, D.A.; Ruan, W.J.; Huang, B.; Saha, T. Comorbidity between DSM-IV alcohol and specific drug use disorders in the United States: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. Alcohol Res. Health 2006, 29, 94–107. [Google Scholar]
- Levine, B.; Green, D.; Smialek, J. The Role of Ethanol in Heroin Deaths. J. Forensic Sci. 1995, 40, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Hickman, M.; Lingford-Hughes, A.; Bailey, C.; Macleod, J.; Nutt, D.; Henderson, G. Does alcohol increase the risk of overdose death: The need for a translational approach. Addiction 2008, 103, 1060–1062. [Google Scholar] [CrossRef] [PubMed]
- Fraser, A.G. Pharmacokinetic interactions between alcohol and other drugs. Clin. Pharmacokinet. 1997, 33, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Moore, N.; Pollack, C.; Butkerait, P. Adverse drug reactions and drug–drug interactions with over-the-counter NSAIDs. Ther. Clin. Risk Manag. 2015, 11, 1061–1075. [Google Scholar] [PubMed]
- Zakhari, S. Overview: How Is Alcohol Metabolized by the Body? Alcohol Res. Health 2006, 29, 245–255. [Google Scholar] [PubMed]
- Edenberg, H.J. The Genetics of Alcohol Metabolism: Role of Alcohol Dehydrogenase and Aldehyde Dehydrogenase Variants. Alcohol Res. Health 2007, 30, 5–13. [Google Scholar] [PubMed]
- Niemelä, O.; Parkkila, S.; Pasanen, M.; Limuro, Y.; Bradford, B.; Thurman, R.G. Early alcoholic liver injury: Formation of protein adducts with acetaldehyde and lipid peroxidation products, and expression of CYP2E1 and CYP3A. Alcohol. Clin. Exp. Res. 1998, 22, 2118–2124. [Google Scholar] [CrossRef] [PubMed]
- Flockhart, D.A. Drug Interactions: Cytochrome P450 Drug Interaction Table. Indiana University School of Medicine. 2007. Available online: https://drug-interactions.medicine.iu.edu (accessed on 15 November 2018).
- Bibi, Z. Role of P450 in drug interactions. Nutr. Metab. 2008, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.N.; Anderson, G.D. Pharmacokinetic and Pharmacodynamic Drug Interactions with Ethanol (Alcohol). Clin. Pharmacokinet. 2014, 53, 1115–1136. [Google Scholar] [CrossRef] [PubMed]
- Bruce, R.D.; Altice, F.L.; Fried, G.H. Pharmacokinetic drug interactions between drugs of abuse and antiretroviral medications: Implications and management for clinical practice. Exp. Rev. Clin. Pharm. 2008, 1, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.B.; Laizurs, S.C. The Effect of Ethanol on Oral Cocaine Pharmacokinetics Reveals an Unrecognized Class of Ethanol-Mediated Drug Interactions. Drug Metab. Dispos. 2010, 38, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.-J.; Hedaya, M.A. Cocaine and Alcohol Interactions in the Rat: Effect on Cocaine Pharmacokinetics and Pharmacodynamics. J. Pharm. Sci. 1999, 88, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Shi, X.; Wang, L.; Gosnell, B.A.; Chen, C. Characterization of Differential Cocaine Metabolism in Mouse and Rat through Metabolomics-Guided Metabolite Profiling. Drug Metab. Dispos. 2013, 41, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Patrick, K.S.; Straughn, A.B.; Minhinnett, R.R.; Yeatts, A.E.; DeVane, C.L.; Malcolm, R.; Janis, G.C.; Markowitx, J.S. Influence of ethanol and gender on methylphenidate pharmacokinetics and pharmacodynamics. Clin. Pharmacol. Ther. 2007, 81, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, Y.; Zhang, Y.; Liu, M. Effects of Ethanol on the Toxicokinetics of Methamphetamine in Rabbits. Iran. J. Pharm. Res. 2014, 13, 329–336. [Google Scholar] [PubMed]
- Adir, J.; Wildfeur, W.; Miller, R.P. Effect of Ethanol Pretreatment on the Pharmacokinetics of Nicotine in Rats. J. Pharmacol. Exp. Ther. 1980, 212, 274–279. [Google Scholar] [PubMed]
- Rose, J.E.; Brauer, L.H.; Behm, F.M. Psychopharmacological interactions between nicotine and ethanol. Nicotine Tobacco Res. 2004, 6, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, C.S.; Miksys, S.; Palmour, R.M.; Tyndale, R.F. Differential Effects of Nicotine Treatment and Ethanol Self-Administration on CYP2A6, CYP2B6 and Nicotine Pharmacokinetics in African Green Monkeys. J. Pharm. Exp. Ther. 2012, 343, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Toennes, S.W.; Schneider, K.; Kauert, G.F.; Wunder, C.; Moeller, M.R.; Theunissen, E.L.; Ramaekers, J.G. Influence of ethanol on cannabinoid pharmacokinetic parameters in chronic users. Anal. Bioanal. Chem. 2011, 400, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Toennes, S.W.; Schneider, K.; Wunder, C.; Kauert, G.F.; Moeller, M.R.; Theunissen, E.L.; Ramaekers, J.C. Influence of Ethanol on the Pharmacokinetic Properties of Δ9-Tetrahydrocannabinol in Oral Fluid. J. Anal. Toxicol. 2013, 37, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Hartman, R.L.; Brown, T.L.; Milavetz, G.; Spurgin, A.; Gorelick, D.A.; Gaffney, G.; Huestis, M.A. Controlled cannabis vaporizer administration: Blood and plasma cannabinoids with and without alcohol. Clin. Chem. 2015, 61, 850–869. [Google Scholar] [CrossRef] [PubMed]
- Lukas, S.E.; Oroczo, S. Ethanol increases plasma Delta (9)-tetrahydrocannabinol (THC) levels and subjective effects after marihuana smoking in human volunteers. Drug Alcohol Depend. 2001, 64, 143–149. [Google Scholar] [CrossRef]
- Dorian, P.; Sellers, E.M.; Reed, K.L. Amitriptyline and ethanol: Pharmacokinetic and pharmacodynamic interaction. Eur. J. Clin. Pharmacol. 1983, 25, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.J.; McLean, A.J. Clinical pharmacokinetic and pharmacodynamic considerations in patients with liver disease. Clin. Pharmacokinet. 1995, 29, 370–391. [Google Scholar] [CrossRef] [PubMed]
- Hoyovadillo, C.; Mandema, J.W.; Danhof, M. Pharmacodynamic interaction between midazolam and a low dose of ethanol in vivo. Life Sci. 1995, 57, 325–333. [Google Scholar] [CrossRef]
- Allan, A.M.; Harris, R.A. Acute and chronic ethanol treatments alter GABA receptor-operated chloride channels. Pharmacol. Biochem. Behav. 1987, 27, 665–670. [Google Scholar] [CrossRef]
- Wan, F.J.; Berton, F.; Madamba, S.G.; Francesconi, W.; Siggins, G.R. Low ethanol concentrations enhance GABAergic inhibitory postsynaptic potentials in hippocampal pyramidal neurons only after block of GABAB receptors. Proc. Natl. Acad. Sci. USA 1996, 93, 5049–5054. [Google Scholar] [CrossRef] [PubMed]
- Roberto, M.; Madamba, S.G.; Moore, S.D.; Tallent, M.K.; Siggins, G.R. Ethanol increases GABAergic transmission at both pre- and postsynaptic sites in rat central amygdala neurons. Proc. Natl. Acad. Sci. USA 2003, 100, 2053–2058. [Google Scholar] [CrossRef] [PubMed]
- Cross, S.J.; Lotfipour, S.; Leslie, F.M. Mechanisms and genetic factors underlying co-use of nicotine and alcohol or other drugs of abuse. Am. J. Drug Abus. 2017, 43, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Spitz, M.; Amos, C.I.; Dong, Q.; Lin, J.; Wu, X. The CHRNA5-A3 region on chromosome 15q24-25.1 is a risk factor both for nicotine dependence and for lung cancer. J. Natl. Cancer Inst. 2008, 100, 1552–1556. [Google Scholar] [CrossRef] [PubMed]
- Bierut, L.J. Nicotine dependence and genetic variation in the nicotinic receptors. Drug Alcohol Depend. 2009, 104, S64–S69. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Grucza, R.; Cruchaga, C.; Hinrichs, A.L.; Bertelsen, S.; Budde, J.P.; Fox, L.; Goldstein, E.; Reyes, O.; Saccone, N.; et al. Genetic variation in the CHRNA5 gene affects mRNA levels and is associated with risk for alcohol dependence. Mol. Psychiatry 2009, 14, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Li, M.D.; Xu, Q.; Lou, X.Y.; Payne, T.J.; Niu, T.; Ma, J.Z. Association and interaction analysis of variants in CHRNA5/CHRNA3/CHRNB4 gene cluster with nicotine dependence in African and European Americans. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2010, 153B, 745–756. [Google Scholar] [PubMed]
- Mihalak, K.B.; Carroll, F.I.; Luetje, C.W. Varenicline is a partial agonist at alpha4beta2 and a full agonist at alpha7 neuronal nicotinic receptors. Mol. Pharmacol. 2006, 70, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Erwin, B.L.; Slaton, R.M. Varenicline in the treatment of alcohol use disorders. Ann. Pharmacother. 2014, 48, 1445–1455. [Google Scholar] [CrossRef] [PubMed]
- Narahashi, T.; Aistrup, G.L.; Marszalec, W.; Nagata, K. Neuronal nicotinic acetylcholine receptors: A new target site of ethanol. Neurochem. Int. 1999, 35, 131–141. [Google Scholar] [CrossRef]
- Li, M.D.; Burmeister, M. New insights into the genetics of addiction. Nature Rev. Genet. 2009, 10, 225–231. [Google Scholar] [CrossRef]
- Hearn, W.L.; Flynn, D.D.; Hime, G.W.; Rose, S.; Cofino, J.C.; Mantero-Atienza, E.; Wetli, C.V.; Mash, D.C. Cocaethylene: A Unique Cocaine Metabolite Displays High Affinity for the Dopamine Transporter. J. Neurochem. 1991, 56, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.E.; Chen, M.; Stamatakis, A.M. Levetiracetam Has Opposite Effects on Alcohol- and Cocaine-Related Behaviors in C57BL/6J Mice. Neuropsychopharmacology 2013, 38, 1322–1333. [Google Scholar] [CrossRef] [PubMed]
- McBride, W.J.; Murphy, J.M.; Ikemoto, S. Localization of brain reinforcement mechanisms: Intracranial self-administration and intracranial place-conditioning studies. Behav. Brain Res. 1999, 101, 129–152. [Google Scholar] [CrossRef]
- Wise, R.A. Ventral tegmental glutamate: A role in stress-, cue-, and cocaine-induced reinstatement of cocaine-seeking. Neuropharmacology 2009, 56 (Suppl. 1), S174–S176. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, H.; Morrisett, R.A. Ethanol action on dopaminergic neurons in the ventral tegmental area: Interaction with intrinsic ion channels and neurotransmitter inputs. Int. Rev. Neurobiol. 2009, 91, 235–288. [Google Scholar]
- Stuber, G.D.; Hopf, F.W.; Tye, K.M. Neuroplastic alterations in the limbic system following cocaine or alcohol exposure. Curr. Top. Behav. Neurosci. 2010, 3, 3–27. [Google Scholar] [PubMed]
- Singh, A.K. Critical Review of Alcohol, Alcoholism and the Withdrawal Symptoms I. Mechanisms of Addiction and the Withdrawal Syndrome. Arch. Addict. Rehab. 2017, 1, 11–30. [Google Scholar]
- Roberto, M.; Varodavan, F. Synaptic Targets: Chronic Alcohol Actions. Neuropharmacology 2017, 122, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, L.K.; Mark, G.P. Comparison of Systemic and Local Methamphetamine Treatment on Acetylcholine and Dopamine Levels in the Ventral Tegmental Area in the Mouse. Neuroscience 2008, 156, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Pomerleau, O.F.; Rosecrans, J. Neuroregulatory effects of nicotine. Psychoneuroendocrinology 1989, 14, 407–423. [Google Scholar] [CrossRef]
- Barik, J.; Wonnacott, S. Molecular and Cellular Mechanisms of Action of Nicotine in the CNS. In Nicotine Psychopharmacology. Handbook of Experimental Pharmacology; Henningfield, J.E., London, E.D., Pogun, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 192. [Google Scholar]
- Qi, Z.; Tretter, F.; Voit, E.O. A heuristic model of alcohol dependence. PLoS ONE 2014, 9, e92221. [Google Scholar] [CrossRef] [PubMed]
- Richardson, B.D.; Rossi, D.J. Recreational concentrations of alcohol enhance synaptic inhibition of cerebellar unipolar brush cells via pre- and postsynaptic mechanisms. J. Neurophysiol. 2017, 118, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Mark, F.; Zoltan, N. Variations on an inhibitory theme: Phasic and tonic activation of GABAA receptors. Nat. Rev. Neurosci. 2005, 6, 215–229. [Google Scholar]
- Roberto, M.; Madamba, S.G.; Stouffer, D.G.; Parsons, L.H.; Siggins, G.R. Increased GABA release in the central amygdala of ethanol-dependent rats. J. Neurosci. 2004, 24, 10159–10166. [Google Scholar] [CrossRef] [PubMed]
- Brousse, G.; Arnaud, B.; Vorspan, F.; Richard, D.; Dissard, A.; Dubois, M.; Pic, D.; Geneste, J.; Xavier, L.; Authier, N. Alteration of Glutamate/GABA Balance During Acute Alcohol Withdrawal in Emergency Department: A Prospective Analysis. Alcohol Alcohol. 2012, 47, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, N. Neurotransmitters in alcoholism: A review of neurobiological and genetic studies. Indian J. Hum. Genet. 2014, 20, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Buck, K.J.; Harris, R.A. Benzodiazepine agonist and inverse agonist actions on GABAA receptor-operated chloride channels. II. Chronic effects of ethanol. J. Pharmacol. Exp. Ther. 1990, 253, 713–719. [Google Scholar]
- Costin, B.N.; Miles, M.F. Molecular and neurologic responses to chronic alcohol use. Handb. Clin. Neurol. 2014, 125, 157–171. [Google Scholar] [PubMed]
- Freed, J.W.; Michaelis, E.K. Glutamic acid and ethanol dependence. Pharm. Biochem. Behav. 1978, 8, 509–514. [Google Scholar] [CrossRef]
- Acevedo-Rodriguez, A.; Zhang, L.; Zhou, F. Cocaine inhibition of nicotinic acetylcholine receptors influences dopamine release. Front. Synaptic Neurosci. 2014, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.; Block, E.; Hu, F.; Cobanoglu, M.C.; Sorkin, A.; Bahar, I. Insights into the modulation of dopamine transporter function by amphetamine, orphenadrine, and cocaine binding. Front. Neurol. 2015, 6, 134. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Grace, A.A. Dopaminergic modulation of limbic and cortical drive of nucleus accumbens in goal-directed behavior. Nat. Neurosci. 2005, 8, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.N.; Ware, C.D.; Boone, E.M. Ethanol Modulates Cocaine-Induced Behavioral Change in Inbred Mice. Pharm. Biochem. Behav. 1998, 59, 567–575. [Google Scholar] [CrossRef]
- O’Malley, P.; Johnston, L.D. Epidemiology of Alcohol and Other Drug Use among American College Students. J. Stud. Alcohol 2002, S14, 23–39. [Google Scholar] [CrossRef]
- Lewis, M.J.; June, H.L. Synergistic effects of ethanol and cocaine on brain simulation reward. J. Exp. Anal. Behav. 1994, 61, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Busse, G.D.; Lawrence, E.T.; Riley, A.L. The modulation of cocaine-induced conditioned place preferences by alcohol: Effects of cocaine dose. Prog. Neuro-Psychopharm. Biol. Psychiatry 2004, 28, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Knackstedt, L.A.; Ben-Shahar, O.; Ettenberg, A. Alcohol consumption is preferred to water in rats pre-treated with intravenous cocaine. Pharm. Biochem. Behav. 2006, 85, 81–286. [Google Scholar] [CrossRef] [PubMed]
- Katner, S.N.; Oster, S.M.; Ding, Z.M.; Deehan, G.A., Jr.; Toalston, J.E.; Hauser, S.R.; McBride, W.A.; Rodd, Z.A. Alcohol-preferring (P) rats are more sensitive than Wistar rats to the reinforcing effects of cocaine self-administered directly into the nucleus accumbens shell. Pharm. Biochem. Behav. 2011, 99, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Nurnberger, J.I., Jr.; Wiegand, R.; Bucholz, K. A family study of alcohol dependence: Co-aggregation of multiple disorders in relatives of alcohol-dependent probands. Arch. Gen. Psychiatry 2004, 61, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Hearn, W.L.; Rose, S.; Wagner, D.C. Cocaethylene is more potent than cocaine in mediating lethality. Pharmacol. Biochem. Behav. 1991, 39, 531–533. [Google Scholar] [CrossRef]
- Bradberry, C.W.; Nobiletti, J.B.; Elsworth, J.D. Cocaine and Cocaethylene: Microdialysis Comparison of Brain Drug Levels and Effects on Dopamine and Serotonin. J. Neurochem. 1993, 60, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Raven, M.A.; Necessary, B.D.; Danluck, D.A.; Ettenberg, A. Comparison of the reinforcing and anxiogenic effects of intravenous cocaine and cocaethylene. Exp. Clin. Psychopharmacol. 2000, 8, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Hart, C.; Jatlow, P.; Sevarino, K.; McCance-Katz, E.F. Comparison of intravenous cocaethylene and cocaine in humans. Psychopharmacology 2000, 149, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Shannak, K.; Kish, S.J.; Levey, A.I.; Bergeron, C.; Deck, J.; Kalasinsky, K.; Ang, L.; Peretti, F.; Adams, V.I. Striatal dopamine, dopamine transporter, and vesicular monoamine transporter in chronic cocaine users. Ann. Neurol. 1996, 40, 428–439. [Google Scholar] [CrossRef]
- Andrews, P. Cocaethylene Toxicity. J. Addict. Dis. 1997, 16, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Álvarez, T.; Racamonde, I.; González-Mariño, I.; Borsotti, A.; Rodil, R.; Rodríguez, I.; Zuccato, E.; Quintana, J.B.; Castiglioni, S. Alcohol and cocaine co-consumption in two European cities assessed by wastewater analysis. Sci. Total Environ. 2015, 536, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.S.; Everhart, E.T.; Mendelson, J.; Jones, R.T. The pharmacology of cocaethylene in humans following cocaine and ethanol administration. Drug Alcohol Depend. 2003, 72, 169–182. [Google Scholar] [CrossRef]
- Herbst, E.D.; Harris, D.S.; Everhart, E.T.; Mendelson, J.J.; Jones, P.; Reese, T. Cocaethylene formation following ethanol and cocaine administration by different routes. Exp. Clin. Psychopharmacol. 2011, 19, 95–104. [Google Scholar] [CrossRef] [PubMed]
- McCance-Katz, E.F.; Kosten, T.R.; Jatlow, P. Concurrent use of cocaine and alcohol is more potent and potentially more toxic than use of either alone—A multiple-dose study. Biol. Psychiatry 1998, 44, 250–259. [Google Scholar] [CrossRef]
- Farré, M.; De La Torre, R.; González, M.L.; Teran, M.T.; Roset, P.N.; Menoyo, E.; Cami, J. Cocaine and alcohol interactions in humans: Neuroendocrine effects and cocaethylene metabolism. J. Pharm. Exp. Ther. 1997, 283, 164–176. [Google Scholar]
- Gottschalk, P.C.; Kosten, T.R. Cerebral perfusion defects in combined cocaine and alcohol dependence. Drug Alcohol Depend. 2002, 68, 95–104. [Google Scholar] [CrossRef]
- Robinson, J.E.; Heaton, R.K.; O’Malley, S.S. Neuropsychological functioning in cocaine abusers with and without alcohol dependence. J. Int. Neuropsychol. Soc. 1999, 5, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Kosten, T.R.; Cheeves, C.; Palumbo, J.; Kosten, T.R.; Cheeves, C.; Palumbo, J.; Seibyl, J.P.; Price, L.P.; Woods, S.W. Regional cerebral blood flow during acute and chronic abstinence from combined cocaine–alcohol abuse. Drug Alcohol Depend. 1998, 50, 187–195. [Google Scholar] [CrossRef]
- Bolla, K.I.; Funderburk, F.R.; Cadet, J.L. Differential effects of cocaine and cocaine alcohol on neurocognitive performance. Neurology 2000, 54, 2285–2292. [Google Scholar] [CrossRef] [PubMed]
- Verdejo-García, A.; Pérez-García, M. Profile of executive deficits in cocaine and heroin polysubstance users: Common and differential effects on separate executive components. Psychopharmacology 2007, 190, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Dackis, C.A.; O’Brien, C.P. Cocaine dependence: A disease of the brain’s reward centers. J. Subst. Abus. Treat. 2001, 21, 111–117. [Google Scholar] [CrossRef]
- Koob, G.F.; Bloom, F.E. Cellular and molecular mechanisms of drug dependence. Science 1988, 242, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Rompre, P.P. Brain dopamine and reward. Annu. Rev. Psychol. 1989, 40, 191–225. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Sanna, P.P.; Bloom, F.E. Neuroscience of addiction. Neuron 1998, 21, 467–476. [Google Scholar] [CrossRef]
- Lindholm, S.; Rosin, A.; Dahlin, I.; Georgieva, J.; Franck, J. Ethanol administration potentiates cocaine-induced dopamine levels in the rat nucleus accumbens. Br. Res. 2001, 915, 176–184. [Google Scholar] [CrossRef]
- Gautam, P.; Warner, T.D.; Kan, E.C.; Sowell, E.R. Executive function and cortical thickness in youths prenatally exposed to cocaine, alcohol and tobacco. Dev. Cogn. Neurosci. 2015, 16, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Makris, N.; Gasic, G.P.; Kennedy, D.N.; Hodge, S.M.; Kaiser, J.R.; JooLee, M.; WooKin, B.; Blood, A.J.; Evins, E.; Seidman, L. Cortical Thickness Abnormalities in Cocaine Addiction—A Reflection of Both Drug Use and a Pre-existing Disposition to Drug Abuse? Neuron 2008, 60, 174–188. [Google Scholar] [CrossRef] [PubMed]
- Vearrier, D.; Greenberg, M.I.; Miller, S.N.; Okaneku, T.J.; Haggerty, D.A. Methamphetamine-History, Pathophysiology, Adverse Health Effects, Current Trends, and Hazards Associated with the Clandestine Manufacture of Methamphetamine. Disease-a-Month 2012, 58, 38–89. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, M.F.; Nejtek, V.A.; Khoshbouei, H. Prolonged increase in ser31 tyrosine hydroxylase phosphorylation in substantia nigra following cessation of chronic methamphetamine. Neurotoxicology 2018, 67, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Liu, Y.; Zheng, N. Distribution of Methamphetamine and Its Metabolite Amphetamine in Acute and Subacute Ethanol-Methamphetamine Combination Abuse Model Rats. J. Anal. Toxicol. 2012, 36, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Blaker, A.L.; Yamamoto, B.K. Methamphetamine-Induced Brain Injury and Alcohol Drinking. J. Neuroimmune Pharm. 2018, 13, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Eisch, J.E.; Schmued, L.C.; Marshall, J.F. Characterizing cortical neuron injury with fluoro-jade labeling after a neurotoxic regimen of methamphetamine. Synapse 1998, 30, 329–333. [Google Scholar] [CrossRef]
- Bujars, S.; Roche, D.J.; Lunny, K.; Moallem, N.R.; Couertney, K.E.; Allen, V.; Hartwell, E.; Leventhal, A.; Rohrbuagh, Y. The relationship between methamphetamine and alcohol use in a community sample of methamphetamine users. Drug Alcohol Depend. 2014, 142, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Vaghef, L.; Babri, S. The effect of escalating dose, multiple binge methamphetamine regimen and alcohol combination on spatial memory and oxidative stress markers in rat brain. J. Alcohol Drug Depend. 2014, 2, 159. [Google Scholar]
- Furr, C.D.; Delva, J.; Anthony, J.C. The suspected association between methamphetamine (‘ice’) smoking and frequent episodes of alcohol intoxication: Data from the 1993 National Household Survey on Drug Abuse. Drug Alcohol Depend. 2000, 59, 89–93. [Google Scholar] [CrossRef]
- Shimosato, K. Urinary excretion of p-hydroxylated methamphetamine metabolites in man. II. Effect of alcohol intake on methamphetamine metabolism. Pharm. Biochem. Behav. 1988, 29, 733–740. [Google Scholar] [CrossRef]
- Kirkpatrick, M.G.; Gunderson, E.W.; Levin, F.R.; Foltin, R.W.; Hart, C.L. Acute and residual interactive effects of repeated administrations of oral methamphetamine and alcohol in humans. Psychopharmacology 2012, 219, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, J.; Jones, R.T.; Upton, R.; Jacob, P. Methamphetamine and ethanol interactions in humans. Clin. Pharmacokinet. 1995, 57, 559–568. [Google Scholar] [CrossRef]
- Wells, P.G.; Bhatia, S.; Drake, D.M. Fetal oxidative stress mechanisms of neurodevelopmental deficits and exacerbation by ethanol and methamphetamine. Birth Defects Res. Part C 2016, 108, 108–130. [Google Scholar] [CrossRef] [PubMed]
- Andez-Lopez, C.H.; Farre, M.; Roset, P.N.; Menoyo, E.; Pizarro, N.; Ortuno, J.; Torrens, M.; Cami, J.; de la Torre, R. 3,4-Methylenedioxy-methamphetamine (Ecstasy) and Alcohol Interactions in Humans: Psychomotor Performance, Subjective Effects, and Pharmacokinetics. JPET 2002, 300, 236–244. [Google Scholar] [CrossRef]
- Tolu, S.; Eddine, R.; Marti, F.C. Co-activation of VTA DA and GABA neurons mediates nicotine reinforcement. Mol. Psychiatry 2012, 2012, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Jones, R.T.; Jacob, P., III. Additive cardiovascular effects of nicotine and ethanol. Clin. Pharmacokinet. Ther. 1986, 40, 420–424. [Google Scholar] [CrossRef]
- Perkins, K.A.; Sexton, J.E.; DiMarco, A.; Grobe, J.E.; Scierks, A.; Stiller, R.L. Subjective and cardiovascular responses to nicotine combined with alcohol in male and female smokers. Psychopharmacology 1995, 119, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Aliyu, M.H.; Wilson, R.E.; Zoorob, R.; Brown, K.; Alio, A.P.; Clayton, H.; Salihu, H.M. Prenatal alcohol consumption and fetal growth restriction: Potentiation effect by concomitant smoking. Nicotine Tobacco Res. 2009, 11, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Grant, B.F.; Hasin, D.S.; Chou, S.P.; Stinson, F.S.; Dawson, D.A. Nicotine dependence and psychiatric disorders in the United States: Results from the national epidemiologic survey on alcohol and relatedconditions. Arch. Gen. Psychiatry 2004, 61, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Burling, T.A.; Ziff, D.C. Tobacco smoking: A comparison between alcohol and drug abuse inpatients. Addict. Behav. 1988, 13, 185–190. [Google Scholar] [CrossRef]
- DiFranza, J.R.; Guerrera, M.P. Alcoholism and smoking. J. Stud. Alcohol 1990, 51, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Batel, P.; Pessione, F.; Maitre, C.; Rueff, B. Relationship between alcohol and tobacco dependencies among alcoholics who smoke. Addiction 1995, 90, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Deehan, G.A., Jr.; Hauser, S.R.; Waeiss, R.A.; Toalston, J.E.; Truitt, W.A.; McBride, W.J.; Rodd, Z.A. Co-administration of ethanol and nicotine: The enduring alterations in the rewarding properties of nicotine and glutamate activity within the mesocorticolimbic system of female alcohol-preferring (P) rats. Psychopharmacology 2015, 232, 4293–4302. [Google Scholar] [CrossRef] [PubMed]
- Blomqvist, O.; Ericson, M.; Jörgen, E.A.; Soderpalm, B. Accumbal dopamine overflow after ethanol: Localization of the antagonizing effect of mecamylamine. Eur. J. Pharm. 1997, 334, 149–156. [Google Scholar] [CrossRef]
- Blomqvist, O.; Nissbrandt, H.; Engel, J.A.; Soderpalm, B. The mesolimbic dopamine-activating properties of ethanol are antagonized by mecamylamine. Eur. J. Pharm. 1993, 249, 207–213. [Google Scholar] [CrossRef]
- Truitt, W.A.; Hauser, S.R.; Deehan, G.A., Jr.; Toalston, J.E.; Wildem, J.A.; Brell, R.L.; McBride, W.J.; Rodd, Z.A. Ethanol and nicotine interaction within the posterior ventral tegmental area in male and female alcohol-preferring rats: Evidence of synergy and differential gene activation in the nucleus accumbens shell. Psychopharmacology 2015, 232, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Tolu, S.; Marti, F.; Morel, C.; Perrier, C.; Torquet, N.; Pons, S.; de Beaurepaire, R.; Faure, P. Nicotine enhances alcohol intake and dopaminergic responses through β2* and β4* nicotinic acetylcholine receptors. Sci. Rep. 2017, 7, 45116. [Google Scholar] [CrossRef] [PubMed]
- Lüscher, C.; Malenka, R.C. Drug-evoked synaptic plasticity in addiction: From molecular changes to circuit remodeling. Neuron 2011, 69, 650–663. [Google Scholar] [CrossRef] [PubMed]
- Larraga, A.; Belluzzi, J.D.; Leslie, F.M. Nicotine Increases Alcohol Intake in Adolescent Male Rats. Front. Behav. Neurosci. 2017, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Larsson, A.; Engel, J.A. Neurochemical and behavioral studies on ethanol and nicotine interactions. Neurosci. Biobehav. Rev. 2004, 27, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Vowles, K.E.; McEntee, M.L.; Julnes, P.S.; Frohe, T.; Ney, J.P.; van der Goes, D.N. Rates of opioid misuse, abuse, and addiction in chronic pain: A systematic review and data synthesis. Pain 2015, 156, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Cicero, T.J.; Ellis, M.S.; Surratt, H.L. The changing face of heroin use in the United States: A retrospective analysis of the past 50 years. JAMA Psychiatry 2014, 71, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Stein, C. Opioid Receptors. Annu. Rev. Med. 2016, 67, 433–451. [Google Scholar] [CrossRef] [PubMed]
- William, J. Basic Opioid Pharmacology. Rev. Pain 2016, 1, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Kobayashi, T.; Kumanishi, T.; Yano, R.; Sora, I.; Niki, H. Molecular mechanisms of analgesia induced by opioids and ethanol: Is the GIRK channel one of the keys? Neurosci. Res. 2002, 44, 121–131. [Google Scholar] [CrossRef]
- Janecka, A.; Fichna, J.T. Opioid Receptors and their Ligands. Curr. Top. Med. Chem. 2004, 4, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Herlitze, S.; Hockerm, G.H. Molecular determinants of inactivation and G protein modulation in the intracellular loop connecting domains I and II of the calcium channel α1A subunit. Proc. Natl. Acad. Sci. USA 1997, 94, 1512–1516. [Google Scholar] [CrossRef] [PubMed]
- Public Health Agency of Canada. National Report: Apparent Opioid-Related Deaths in Canada. 2017. Available online: https://www.canada.ca/en/public-health/services/publications/healthy-living/apparent-opioid-related-deaths-report-2016-2017-december.html (accessed on 20 November 2018).
- Food and Drug Administration [FDA]. Labels of Sublimaze (Fentanyl), Duragesic (Fentanyl), Fentora (Fentanyl), Actiq (Fentanyl), Alfenta (Alfentanil), Rapifen (Alfentanil), Sufenta (Sufentanil), Ultiva (Remifentanil), Avinza (Morphine Sulphate), Embeda (Morphine Sulfate and Naltrexone Hydrochloride), Duramorph (Morphine Sulfate Injection), Narcan Nasal Spray (Naloxone Hydrochloride), Evzio (Naloxone Hydrochloride), Naloxone West Ward Pharms Int. (Naloxone Hydrochloride), Entereg (Alvimopan), Vivitrol (Naltrexone for Extended-Release Injectable Suspension). 2018. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (accessed on 20 November 2018).
- Weerts, E.M.; Wand, G.S.; Kuwabara, H.; Munro, C.A.; Dannals, R.F.; Hilton, J.; Frost, J.J.; McCaul, M.E. Positron Emission Tomography Imaging of Mu- and Delta-Opioid Receptor Binding in Alcohol-Dependent and Healthy Control Subjects. Alcohol. Clin. Exp. Res. 2011, 35, 2162–2173. [Google Scholar] [CrossRef] [PubMed]
- Olive, M.F. Pharmacotherapies for Alcoholism: The Old and the New. CNS Neurol. Disord. Drug Targets 2010, 9, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Hermann, D.; Hirth, N.; Reimold, M.; Batra, A.; Smolka, M.N.; Hoffmann, S.; Kiefer, F.; Noori, H.R.; Sommer, W.H.; Reischl, G.; et al. Low μ-Opioid Receptor Status in Alcohol Dependence Identified by Combined Positron Emission Tomography and Post-Mortem Brain Analysis. Neuropsychopharmacology 2017, 42, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Ceccarini, J.; Hompes, T.; Verhaeghen, A.; Casteels, C.; Peuskens, H.; Bormans, G.; Claes, S.; Van Laere, K. Changes in Cerebral CB1 Receptor Availability after Acute and Chronic Alcohol Abuse and Monitored Abstinence. J. Neurosci. 2014, 34, 2822–2831. [Google Scholar] [CrossRef] [PubMed]
- Hillmer, A.T.; Mason, G.F.; Fucito, L.M.; O’malley, S.S.; Cosgrove, K.P. How Imaging Glutamate, γ-Aminobutyric Acid, and Dopamine Can Inform the Clinical Treatment of Alcohol Dependence and Withdrawal. Alcohol. Clin. Exp. Res. 2015, 39, 2268–2282. [Google Scholar] [CrossRef] [PubMed]
- Laine, T.P.; Ahonen, A.; Rasanen, P.; Tlihonen, J. Dopamine Transporter Availability and Depressive Symptoms during Alcohol Withdrawal. Psychiatry Res. 1999, 90, 153–157. [Google Scholar] [CrossRef]
- Polettini, A.; Poloni, V.; Groppi, A.; Stramesi, C.; Vignali, C.; Politi, L.; Montagna, M. The role of cocaine in heroin-related deaths. Hypothesis on the interaction between heroin and cocaine. Forensic Sci. Int. 2005, 4, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Thaulow, C.H.; Hoiseth, G.; Andersen, J.M.; Handal, M.; Morland, J. Pharmacokinetic interactions between ethanol and heroin: A study on post-mortem cases. Forensic Sci. Int. 2014, 242, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Ikeda, K.; Kojima, H.; Niki, H.; Yano, R.; Yoshioka, T.; Kumanishi, T. Ethanol opens G-protein-activated inwardly rectifying K+ channels. Nat. Neurosci. 1999, 2, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Blednov, Y.A.; Stoffel, M.; Chang, S.R.; Harris, R.A. Potassium Channels as Targets for Ethanol: Studies of G-Protein-Coupled Inwardly Rectifying Potassium Channel 2 (GIRK2) Null Mutant Mice. JPET 2001, 298, 521–530. [Google Scholar]
- Blednov, Y.A.; Stoffel, M.; Alva, H.; Harris, R.A. A pervasive mechanism for analgesia: Activation of GIRK2 channels. Proc. Natl. Acad. Sci. USA 2003, 100, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Kunkel, M.T.; Peralta, E.G. Identification of domains conferring G protein regulation on inward rectifier potassium channels. Cell 1995, 83, 443–449. [Google Scholar] [CrossRef]
- Nelson, L.R.; Taylor, A.N.; Lewis, J.W.; Branch, B.J.; Liebalind, C.J. Opioid but not nonopioid stress-induced analgesia is enhanced following prenatal exposure to ethanol. Psychopharmacology 1985, 85, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Kranzler, H.R.; Modesto-Lowe, V.; Nuwayser, E.S. Sustained-Release Naltrexone for Alcoholism Treatment: A Preliminary Study. Alcohol. Clin. Exp. Res. 1998, 22, 1074–1079. [Google Scholar] [CrossRef]
- Zhang, H.; Luo, X.; Kranzler, H.R.; Lappalainen, J.; Yang, B.-Z.; Krupitsky, E.; Zvartau, E.; Gelernter, J. Association between two µ-opioid receptor gene (OPRM1) haplotype blocks and drug or alcohol dependence. Hum. Mol. Genet. 2006, 15, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.Y.; Yang, L.-C.; Lu, H.-F.; Ko, J.Y.; Wang, C.H.; Lin, S.H.; Lee, T.H.; Concejero, A.; Hsu, C.J. Association of μ-opioid receptor gene polymorphism (A118G) with variations in morphine consumption for analgesia after total knee arthroplasty. Acta Anaesthesiol. Scand. 2006, 50, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Schellekens, A.F.; Franke, B.; Ellenbroek, B.; Cools, A.; de Jong, C.A.; Buitelaar, J.K.; Verkes, R.J. Reduced Dopamine Receptor Sensitivity as an Intermediate Phenotype in Alcohol Dependence and the Role of the COMT Val158Met and DRD2 Taq1A Genotypes. Arch. Gen. Psychiatry 2012, 69, 339–348. [Google Scholar] [PubMed]
- Hendershot, C.S.; Lindgren, K.P.; Liang, T.; Hutchison, K.E. COMT and ALDH2 polymorphisms moderate associations of implicit drinking motives with alcohol use. Addict. Biol. 2012, 17, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Drehera, J.C.; Kohn, P.; Kolachana, B.; Weinberger, D.R.; Berman, K.F. Variation in dopamine genes influences responsivity of the human reward system. Proc. Natl. Acad. Sci. USA 2009, 106, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Zubieta, J.K.; Mary, M.; Heitzeg, M.H. COMT val158met Genotype Affects µ-Opioid Neurotransmitter Responses to a Pain Stressor. Science 2003, 299, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Thompson, T.; Oram, C.; Correll, C.U.; Bueller, J.A.; Xu, K.; Xu, Y.; Koeppe, R.A.; Stohler, C.S.; Goldman, D. Analgesic Effects of Alcohol: A Systematic Review and Meta-Analysis of Controlled Experimental Studies in Healthy Participants. J. Pain 2017, 18, 5499–54510. [Google Scholar] [CrossRef] [PubMed]
- Bass, M.B.; Friedman, H.J.; Lester, D. Antagonism of naloxone hyperalgesia by ethanol. Life Sci. 1978, 22, 1939–1946. [Google Scholar] [CrossRef]
- Xiong, M.; Shiwalkar, N.; Reddy, K.; Shin, P.; Bekker, A. Neurobiology of Propofol Addiction and Supportive Evidence: What Is the New Development? Brain Sci. 2018, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-Y.; Mao, X.; Wei, L. Genes and (Common) Pathways Underlying Drug Addiction. PLoS Comput. Biol. 2008, 4, e2. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J. Is there a common molecular pathway for addiction? Nat. Neurosci. 2005, 8, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Gianoulakis, C. Endogenous Opioids and Addiction to Alcohol and other Drugs of Abuse. Curr. Top. Med. Chem. 2004, 4, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Oswald, L.M.; Wand, G.S. Receptor Knockout Mice. Opioids and alcoholism. Physiol. Behav. 2004, 81, 339–358. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, F.; Wu, Y.; Tuke, J. Alcohol-induced sedation and synergistic interactions between alcohol and morphine: A key mechanistic role for Toll-Like Receptors and MyD88-dependent signaling. Brain Behav. Immun. 2015, 45, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, F.; Hutchinson, M. Are the effects of alcohol on the CNS influenced by Toll-like receptor signaling? Expert Rev. Clin. Immunol. 2012, 8, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Wachtel, S.R.; ElSohly, M.A.; Ross, S.A.; Ambre, J.; de Wit, H. Comparison of the subjective effects of Delta(9)-tetrahydrocannabinol and marijuana in humans. Psychopharmacology. 2002, 161, 331–339. [Google Scholar] [PubMed]
- Felder, C.C.; Veluz, J.S.; Williams, H.L.; Briley, E.M.; Matsuda, L.A. Cannabinoid agonists stimulate both receptor- and non-receptor-mediated signal transduction pathways in cells transfected with and expressing cannabinoid receptor clones. Mol. Pharmacol. 1992, 42, 838–845. [Google Scholar] [PubMed]
- Blankman., J.L.; Simon, G.M.; Cravatt, B.F. A Comprehensive Profile of Brain Enzymes that Hydrolyze the Endocannabinoid 2-Arachidonoylglycerol. Chem. Biol. 2007, 14, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Castillo, P.E.; Younts, T.J.; Chavez, A.E.; Hashimotodani, Y. Endocannabinoid signaling and synaptic function. Neuron 2012, 76, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, M.A.P.; Ashok, A.H.; Volkow, N.D.; Howes, O.D. The effects of Δ9-tetrahydrocannabinol on the dopamine system. Nature 2016, 539, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Parsons, L.H.; Hurd, Y.L. Endocannabinoid signaling in reward and addiction. Nat. Neurosci. 2015, 16, 579–594. [Google Scholar] [CrossRef] [PubMed]
- Iversen, L. Cannabis and the brain. Brain 2003, 126, 1252–1270. [Google Scholar] [CrossRef] [PubMed]
- Liguori, A.; Gatto, C.P.; Jarrett, D.B. Separate and combined effects of marijuana and alcohol on mood, equilibrium and simulated driving. Psychopharmacology 2002, 163, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Ramaekers, J.G.; Kuypers, K.P. Acute effects of 3, 4-methylenedioxymethamphetamine (MDMA) on behavioral measures of impulsivity: Alone and in combination with alcohol. Neuropsychopharmacology 2006, 31, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.S.; Kaczynski, N.A.; Maisto, S.A.; Tarter, R.E. Polydrug use in adolescent drinkers with and without DSM-IV alcohol abuse and dependence. Alcohol. Clin. Exp. Res. 1996, 20, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Agosti, V.; Edward, N.; Frances, L. Rates of psychiatric comorbidity among U.S. residents with lifetime cannabis dependence. Am. J. Drug Alcohol Abus. 2002, 28, 643–652. [Google Scholar] [CrossRef]
- Terry-McElrath, Y.M.; O’Malley, O.M.; Johnston, L.D. Alcohol and Marijuana Use Patterns Associated With Unsafe Driving Among U.S. High School Seniors: High Use Frequency, Concurrent Use, and Simultaneous Use. J. Stud. Alcohol Drugs 2014, 31, 1048–1055. [Google Scholar] [CrossRef]
- Giedd, J.N.; Snell, J.W.; Lange, N.; Rajapakse, J.C.; Casey, B.J.; Kozuch, P.L.; Vaituzis, A.C.; Vauss, Y.C.; Hamburger, S.D.; Kaysen, D.; et al. Quantitative magnetic resonance imaging of human brain development: Ages 4–18. Cereb Cortex 1996, 6, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Sowell, E.R.; Trauner, D.A.; Gamst, A.; Jerngan, T.L. Development of cortical and subcortical brain structures in childhood and adolescence: A structural MRI study. Dev Med. Child Neurol. 2002, 44, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Eichenbaum, H. The hippocampus and mechanisms of declarative memory. Behav. Br. Res. 1999, 103, 123–133. [Google Scholar] [CrossRef]
- Aloi, J.; Blair, K.S.; Crum, K.I.; Meffert, H.; White, S.F.; Tyler, P.M.; Thornton, L.C.; Mobley, A.M.; Killanin, A.D.; Adams, K.O.; et al. Adolescents show differential dysfunctions related to Alcohol and Cannabis Use Disorder severity in emotion and executive attention neuro-circuitries. Neuroimage Clin. 2018, 19, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Oleson, E.B.; Cheer, J.F. A Brain on Cannabinoids: The Role of Dopamine Release in Reward Seeking. Cold Spring Harb. Perspect. Med. 2012, 2, a012229. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Paredes, W.; Li, J.; Lowinson, J.; Gardner, E.L. D9-Tetrahydrocannabinol produces naloxoneblockable enhancement of presynaptic basal dopamine efflux in nucleus accumbens of conscious, freely-moving rats as measured by intracerebral microdialysis. Psychopharmacology 1990, 102, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Cheer, J.F.; Wassum, K.M.; Sombers, L.A.; Heien, M.L.A.V.; Ariansen, J.L.; Aragona, B.J.; Phillips, P.E.M.; Wightman, R.M. Phasic dopamine release evoked by abused substances requires cannabinoid receptor activation. J. Neurosci. 2007, 27, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Hungund, B.L.; Basavarajappa, B.S.; Vadasz, C.; Kunos, G.; Rodriguez de Fonseca, F.; Colombo, G.; Serra, S.; Parsons, L.; Koob, G.F. Ethanol, endocannabinoids, and the cannabinoidergic signaling system. Alcohol. Clin. Exp. Res. 2002, 26, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Cohen, C.; Perrault, G.; Voltz, C.; Steinberg, R.; Soubrie, P. SR141716, a central cannabinoid (CB1) receptor antagonist, blocks the motivational and dopamine-releasing effects of nicotine in rats. Behav. Pharm. 2002, 13, 451–463. [Google Scholar] [CrossRef]
- Guillot, C.R.; Blumenthal, H.; Zvolensky, J.; Schmidt, N.B. Anxiety sensitivity components in relation to alcohol and cannabis use, motives, and problems in treatment-seeking cigarette smokers. Addict. Behav. 2018, 82, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Szutorisz, H.; Hurd, Y.L. Epigenetic Effects of Cannabis Exposure. Biol. Psychiatry 2016, 79, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Parira, T.; Laverde, A.; Agudelo, M. Epigenetic Interactions between Alcohol and Cannabinergic Effects: Focus on Histone Modification and DNA Methylation. J. Alcohol Drug Depend. 2017, 5, 259. [Google Scholar] [CrossRef] [PubMed]
- Dobs, Y.E.; Ali, M.M. The epigenetic modulation of alcohol/ethanol and cannabis exposure/co-exposure during different stages. Open Biol. 2019, 9, 180115. [Google Scholar] [CrossRef]
- Hungund, B.L.; Szakall, I.; Adam, A.; Basavarajappa, B.S.; Vadasz, C. Cannabinoid CB1 receptor knockout mice exhibit markedly reduced voluntary alcohol consumption and lack alcohol-induced dopamine release in the nucleus accumbens. J. Neurochem. 2003, 84, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Subbanna, S.; Nagre, N.N.; Umapathy, N.S.; Pace, B.S.; Basavarajappa, B.S. Ethanol Exposure Induces Neonatal Neurodegeneration by Enhancing CB1R Exon1 Histone H4K8 Acetylation and Up-regulating CB1R Function causing Neurobehavioral Abnormalities in Adult Mice. Int. J. Neuropsychopharmacol. 2015, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nagre, N.N. CB1-receptor knockout neonatal mice are protected against ethanol?induced impairments of DNMT1, DNMT3A, and DNA methylation. J. Neurochem. 2015, 132, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Abel, T.; Zukin, R.S. Epigenetic targets of HDAC inhibition in neurodegenerative and psychiatric disorders. Curr. Opin. Pharmacol. 2008, 8, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Renthal, W.; Nestler, E.J. Chromatin regulation in drug addiction and depression. Dialogues Clin. Neurosci. 2009, 11, 257–268. [Google Scholar] [PubMed]
- Maitre, M. The γ-hydroxybutyrate signaling system in brain: Organization and functional implications. Prog. Neurobiol. 1997, 51, 337–361. [Google Scholar] [CrossRef]
- Cash, C.D. Gamma hydroxybutyrate: An overview of the pros and cons for it being an neurotransmitter and/or a useful therapeutic agent. Neurosci. Biobehav. Rev. 1994, 18, 291–304. [Google Scholar] [CrossRef]
- Wong, C.G.T.; Chan, K.F.Y.; Gibson, K.M.; Snead, O.C. γ-Hydroxybutyric. Toxicol. Rev. 2004, 23, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Cooper, F.J.; Logan, B.K. GHB and driving impairment. J. Forensic Sci. 2001, 46, 919–923. [Google Scholar] [CrossRef]
- Mattila, M.J.; Palva, E.; Seppälä, T.; Ostrovskaya, R.U. Actions and interactions with alcohol of drugs on psychomotor skills: Comparison of diazepam and gamma-hydroxybutyric acid. Arch. Int. Pharmacodyn. 1978, 234, 236–246. [Google Scholar] [PubMed]
- Nicholson, K.L.; Balster, R.L. GHB: A new and novel drug of abuse. Drug Alcohol Depend. 2001, 63, 1–22. [Google Scholar] [CrossRef]
- McCabe, E.R.; Layne, E.C.; Sayler, D.F. Synergy of ethanol and a natural soporific—Gamma hydroxybutyrate. Science 1971, 171, 404–406. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, T.; Kaye, L.; Plosay, J.J. Gamma-Hydroxybutyrate (GHB): A Newer Drug of Abuse. Am. Fam. Physician 2000, 62, 2478–2482. [Google Scholar] [PubMed]
- Ropero-Miller, J.D.; Goldberger, B.A. Recreational drugs. Current trends in the 90s. Clin. Lab Med. 1998, 18, 727–746. [Google Scholar] [CrossRef]
- Li, J.; Stokes, S.A.; Woeckener, A. A tale of novel intoxication: A review of the effects of gamma-hydroxybutyric acid with recommendations for management. Ann. Emerg. Med. 1998, 31, 729–736. [Google Scholar] [CrossRef]
- Galloway, G.P.; Frederick, S.L.; Staggers, F.E.; Gonzales, M.; Stalcup, S.A.; Smith, D.E. Gamma-hydroxybutyrate: An emerging drug of abuse that causes physical dependence. Addiction 1997, 92, 89–96. [Google Scholar] [CrossRef] [PubMed]
 action potential.
action potential.
 action potential.
action potential.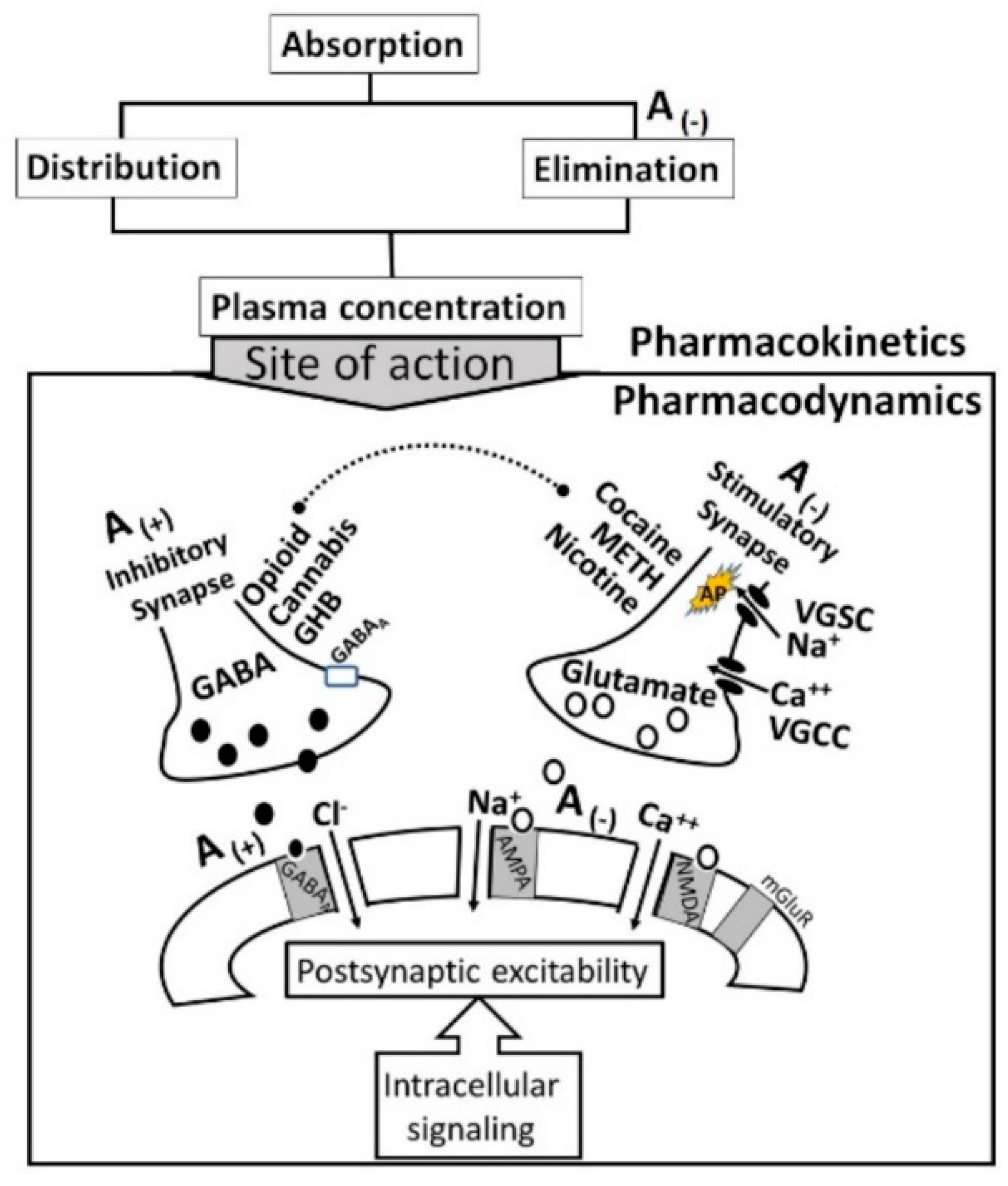
| Indices | Oral Cocaine | Oral Cocaine + Alcohol | Intravenous Cocaine | Intravenous Cocaine + Alcohol |
|---|---|---|---|---|
| AUC0–α (mg·min/L) | 15.0 ± 4.7 *× | 58.0 ± 10 | 83.1 ± 4.7 × | 110.3 ± 22.5 |
| CL (L/min) | 5.6 ± 1.8 *× | 1.6 ± 0.35 | 1.0 ± 1.8 × | 0.74 ± 0.2 |
| Cmax (ng/mL) | 116.0 ± 98 *× | 331.0 ± 131 | 2677 ± 98 | 2885 ± 702 |
| Tmax (min) | 83.6 ± 46 | 99.8 ± 32.5 | ||
| T1/2 (min) | 85.2 ± 6.6 | 84.2 ± 9.1 | 75.0 ± 6.6 *× | 84.0 ± 8.2 |
| F | 0.2 ± 0.05 × | 0.7 ± 0.17 | ||
| CE Cmax (ng/mL) | ND | 30.9 ± 7.3 | ND | ND |
| BE AUC0–α (mg·min/L) | 172.0 ± 46 *× | 410.0 ± 82 | 375.0 ± 46 | 407.0 ± 110 |
| BE/cocaine AUC0–α | 11.9 ± 3 *× | 7.1 ± 1.5 | 4.9 ± 3 | 3.7 ± 0.6 |
| Pharmacodynamic Parameters | Cocaine (ip) a + Normal Saline | Cocaine (ip) a + Alcohol (po) |
|---|---|---|
| A. Neurochemical | ||
| Emax (% of baseline) | 850 ± 200 | 1550 ± 640 |
| EC50 (ng/mL) | 3400 ± 580 | 2000 ± 650 |
| N | 1.23 ± 0.17 | 2.31 ± 0.29 b |
| B. Cardiovascular | ||
| kin (% of baseline/min) | 23.8 ± 5.1 | 36.0 ± 13.0 |
| Kout (min−1) | 0.218 ± 0.047 | 0.31 ± 0.11 |
| Imax | 0.304 ± 0.033 | 0.307 ± 0.035 |
| IC50 (mg/mL) | 6700 ± 2100 | 5600 ± 710 |
| Rmax (% of baseline) | 146 ± 6.9 | 148 ± 8.9 |
| N | 3.0 ± 1.5 | 3.6 ± 1.9 |
| NTs | Glu | GABA | 5-HT | DA | NA | ACh |
|---|---|---|---|---|---|---|
| Glu | + | + | + | - | + | |
| GABA | - | - | - | - | - | |
| 5-HT | + | - | + | + | - | |
| DA | - | - | - | + | - | |
| NA | - | - | - | + | - | |
| ACh | + | + | + | + | + |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, A.K. Alcohol Interaction with Cocaine, Methamphetamine, Opioids, Nicotine, Cannabis, and γ-Hydroxybutyric Acid. Biomedicines 2019, 7, 16. https://doi.org/10.3390/biomedicines7010016
Singh AK. Alcohol Interaction with Cocaine, Methamphetamine, Opioids, Nicotine, Cannabis, and γ-Hydroxybutyric Acid. Biomedicines. 2019; 7(1):16. https://doi.org/10.3390/biomedicines7010016
Chicago/Turabian StyleSingh, Ashok K. 2019. "Alcohol Interaction with Cocaine, Methamphetamine, Opioids, Nicotine, Cannabis, and γ-Hydroxybutyric Acid" Biomedicines 7, no. 1: 16. https://doi.org/10.3390/biomedicines7010016
APA StyleSingh, A. K. (2019). Alcohol Interaction with Cocaine, Methamphetamine, Opioids, Nicotine, Cannabis, and γ-Hydroxybutyric Acid. Biomedicines, 7(1), 16. https://doi.org/10.3390/biomedicines7010016




