Assessment of Diffuse Myocardial Fibrosis and Myocardial Oedema in Sepsis Survivors Using Cardiovascular Magnetic Resonance: Correlation with Left Ventricular Systolic Function
Abstract
1. Introduction
2. Materials and Methods
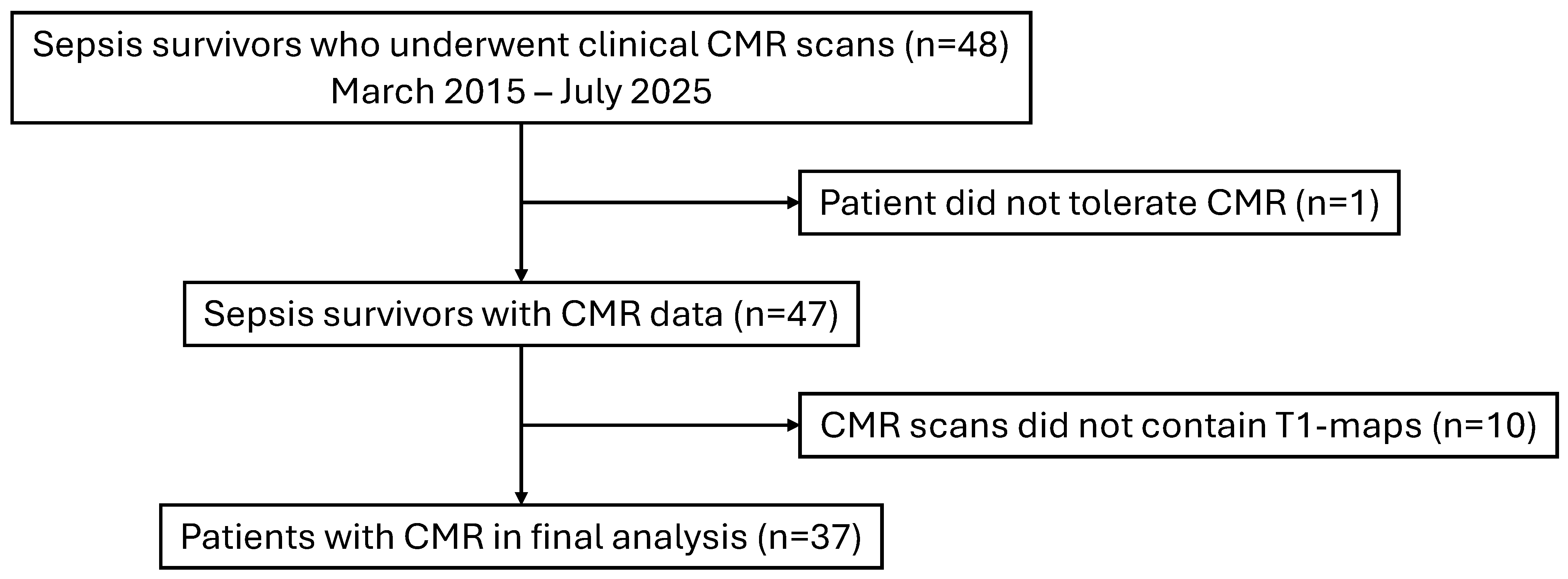
2.1. Study Subjects
2.2. Ethical Approval Statement
2.3. Data Collection
2.4. Cardiovascular Magnetic Resonance (CMR)
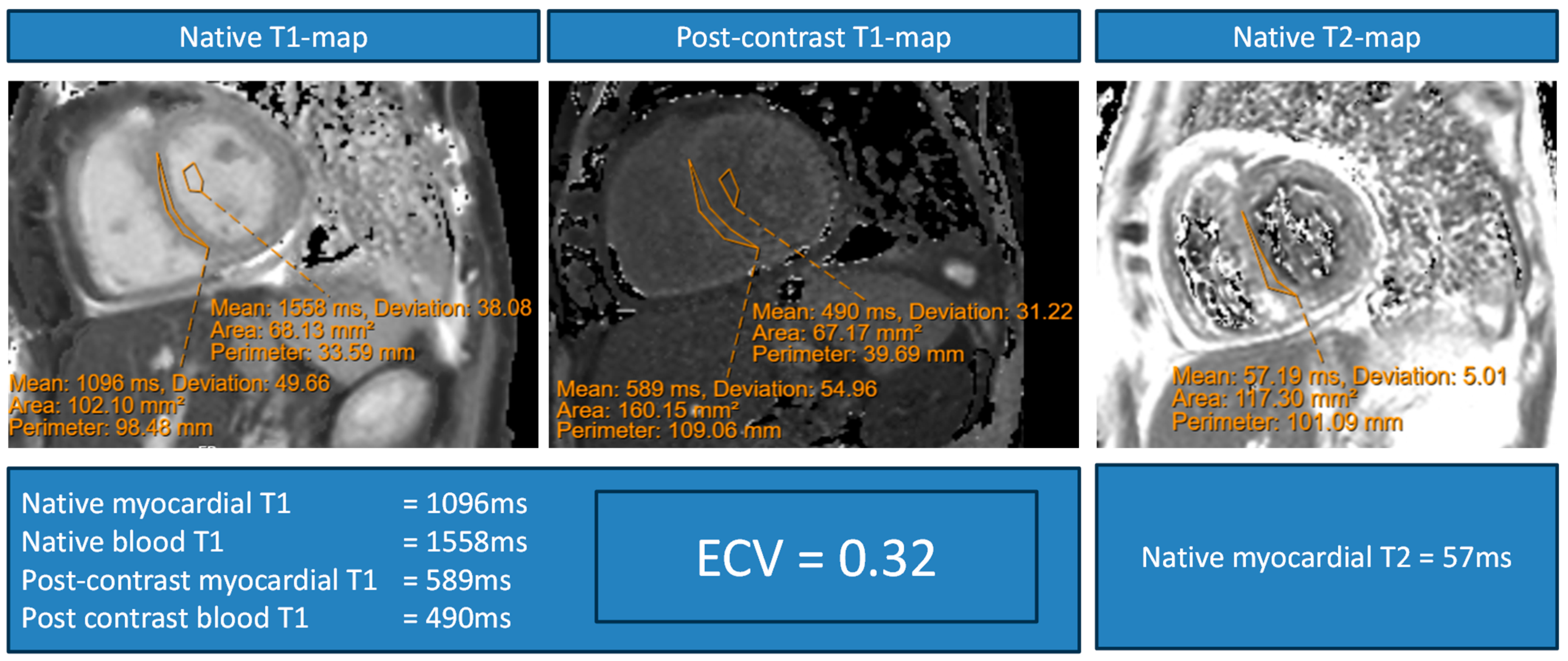
2.5. CMR Image Analysis
2.6. Statistical Analysis
3. Results
3.1. Patient Demographics Data
3.2. CMR Data of Study Patients
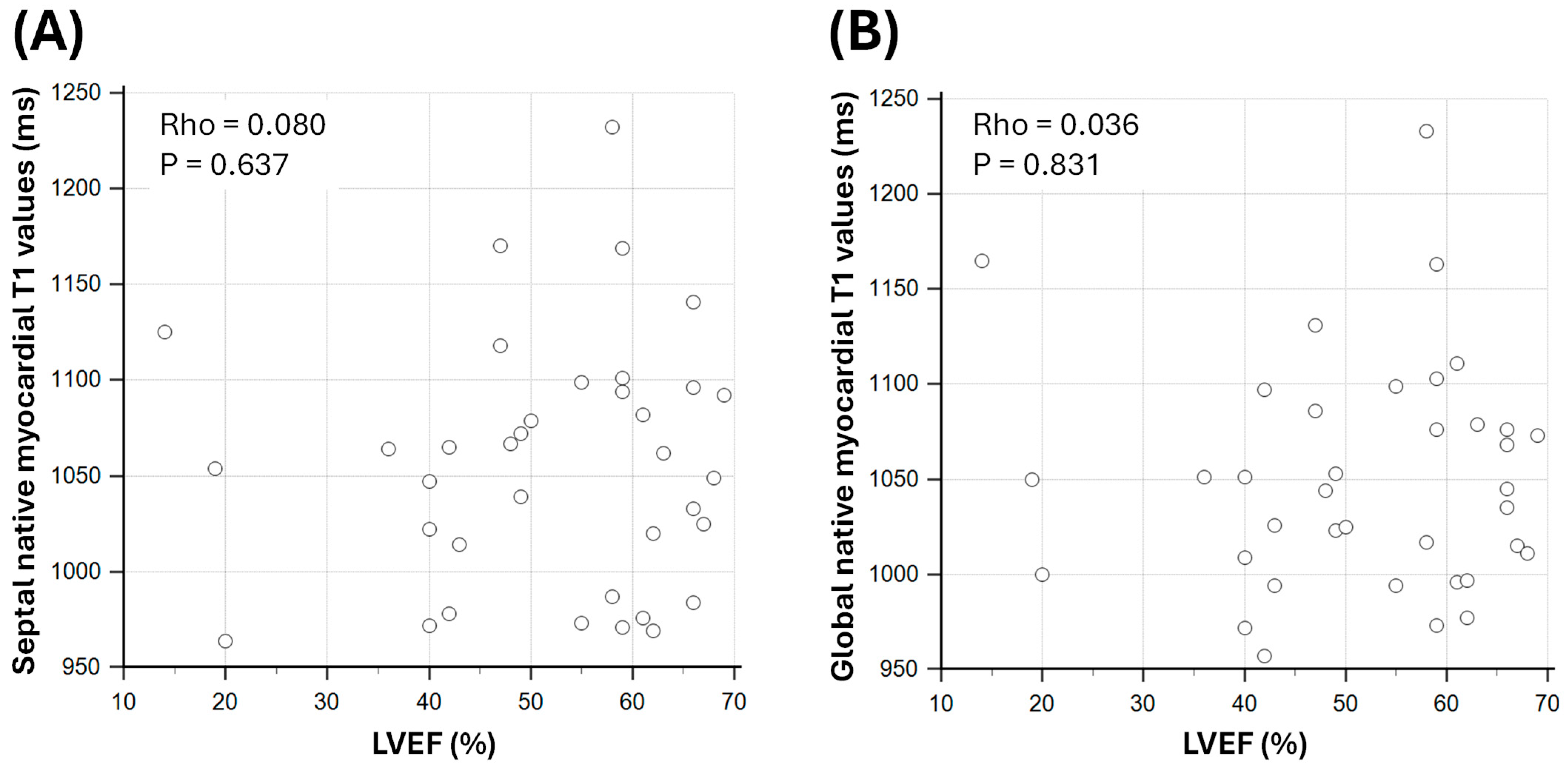
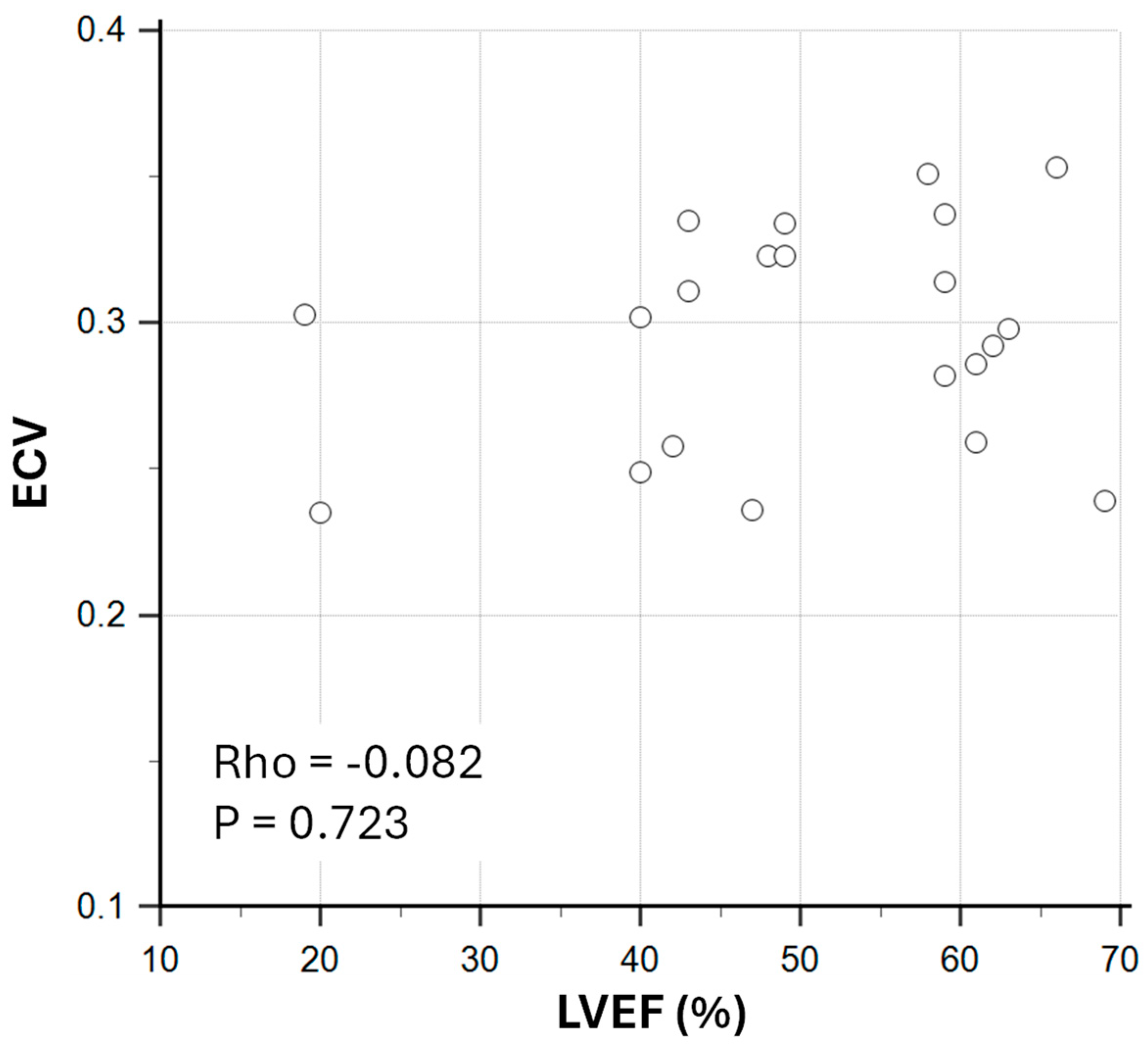
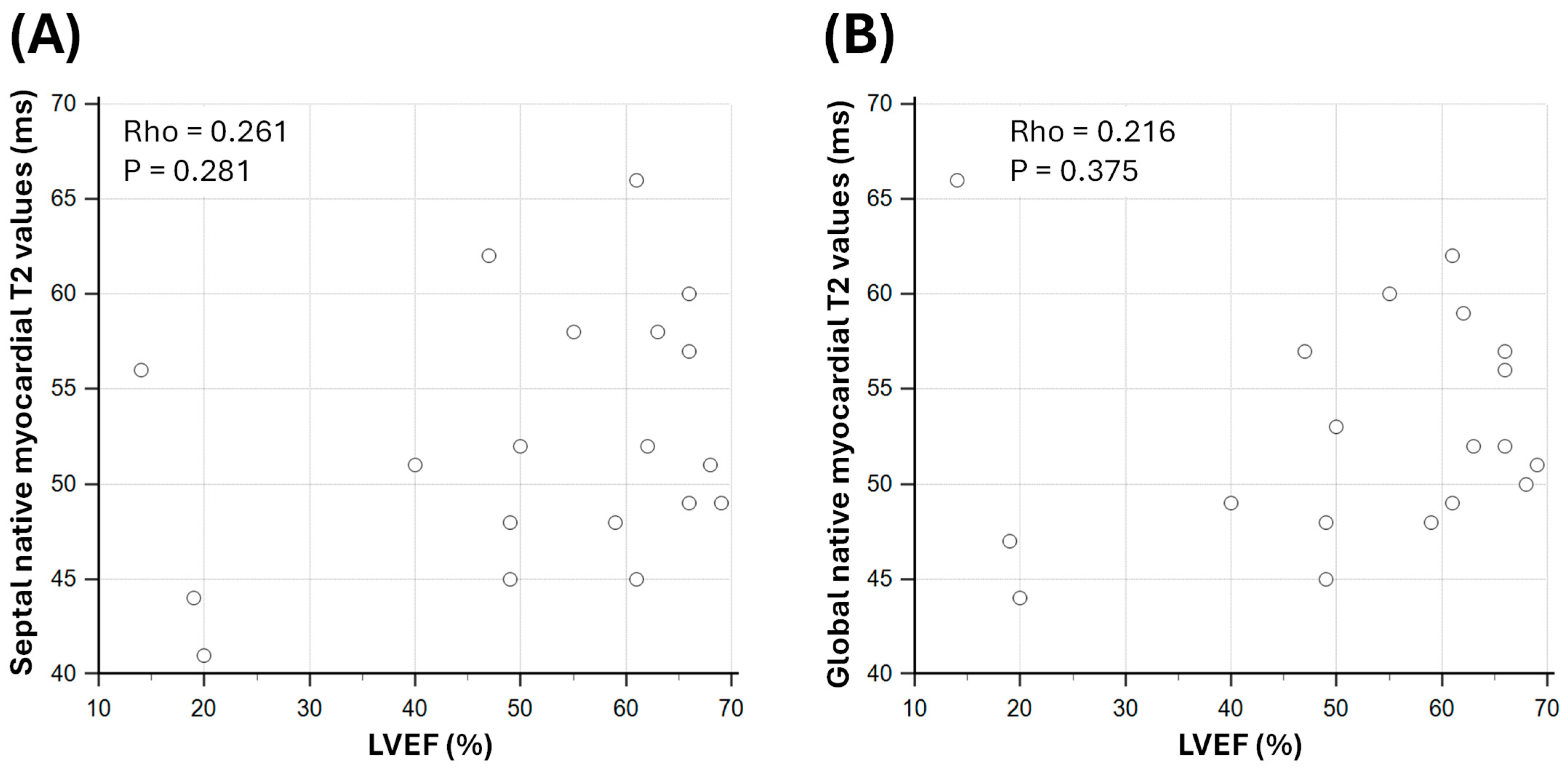
3.3. Relationship Between Myocardial T1 and T2 Parameters with LV Systolic Function
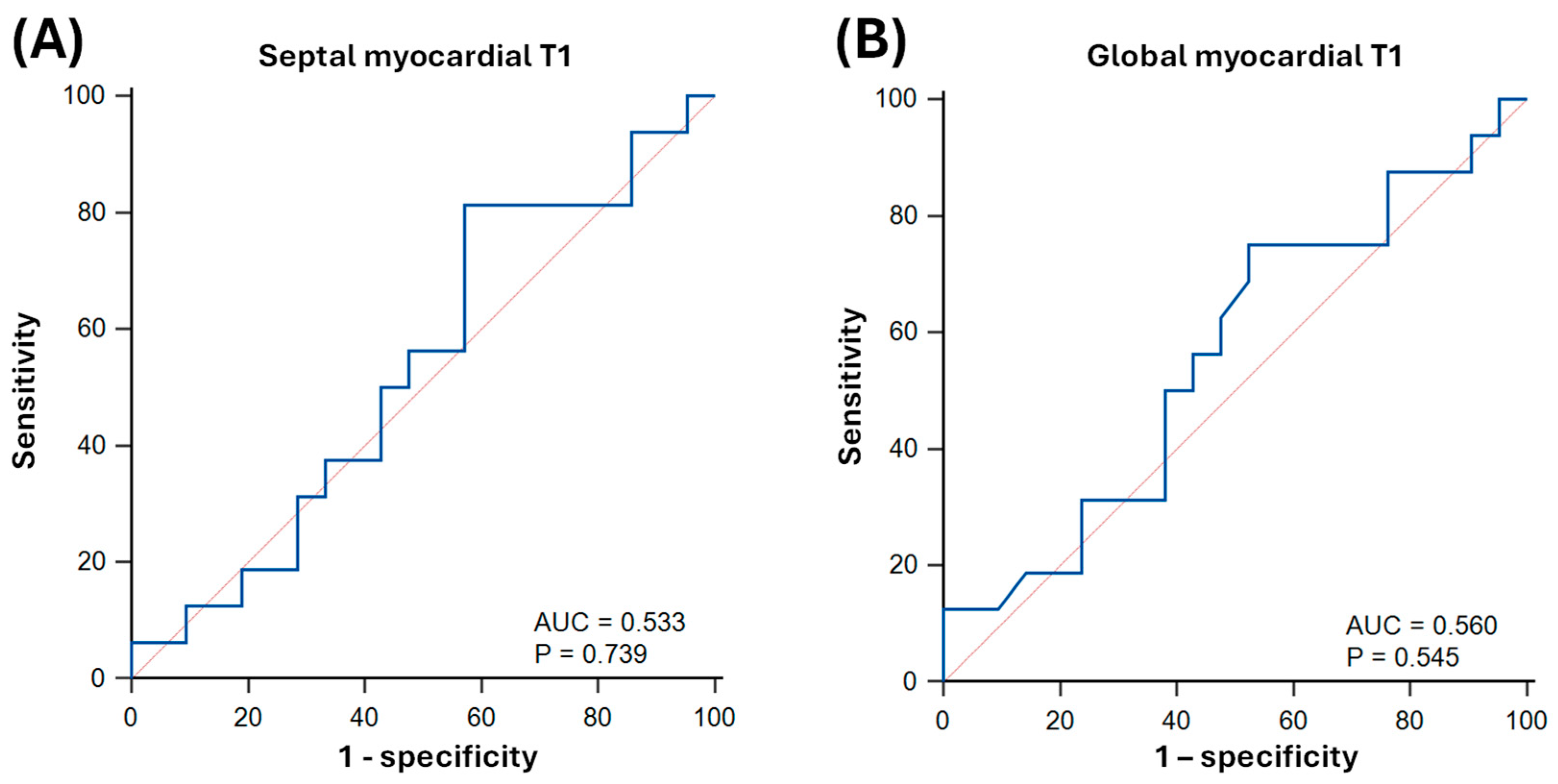
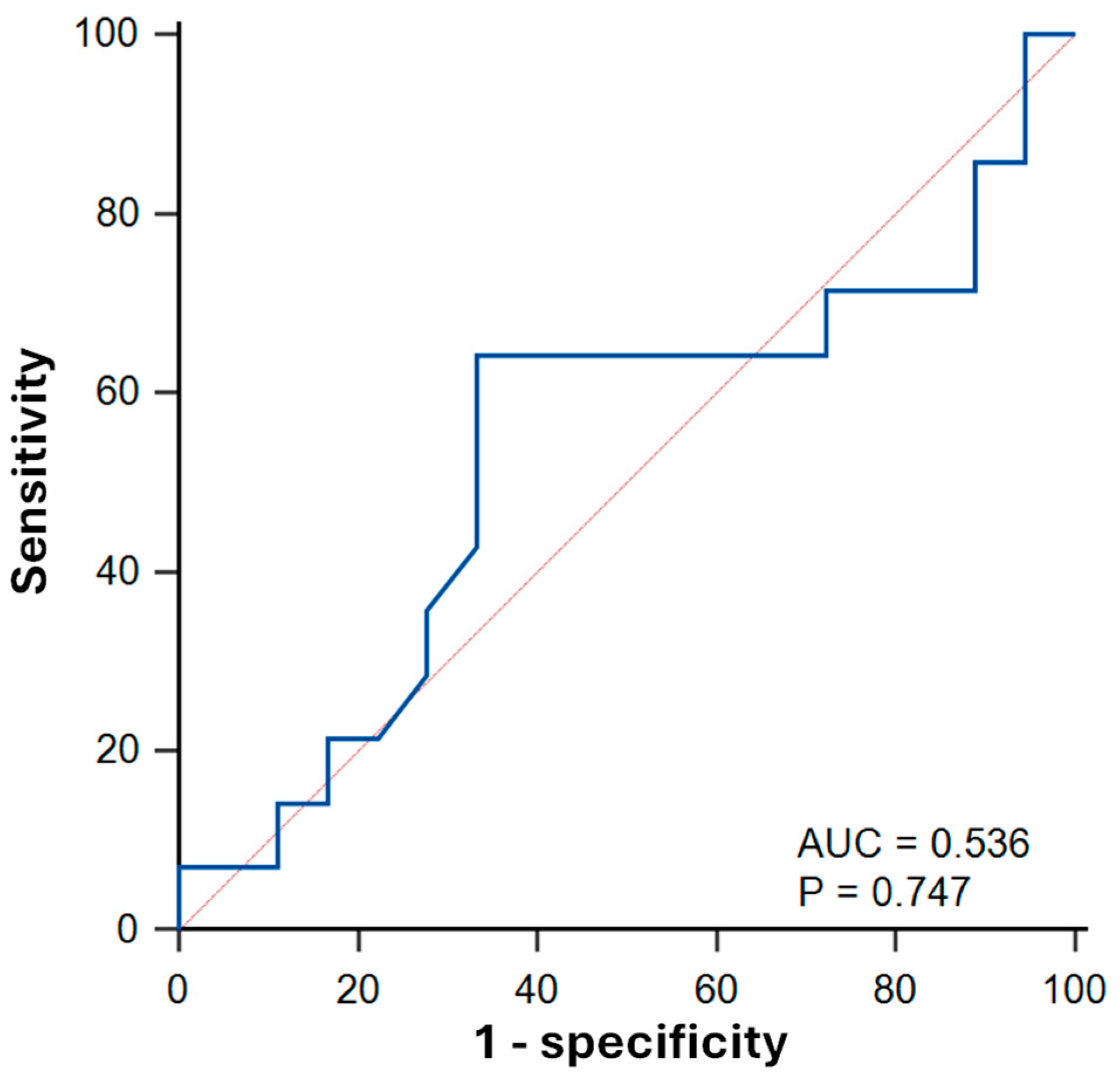
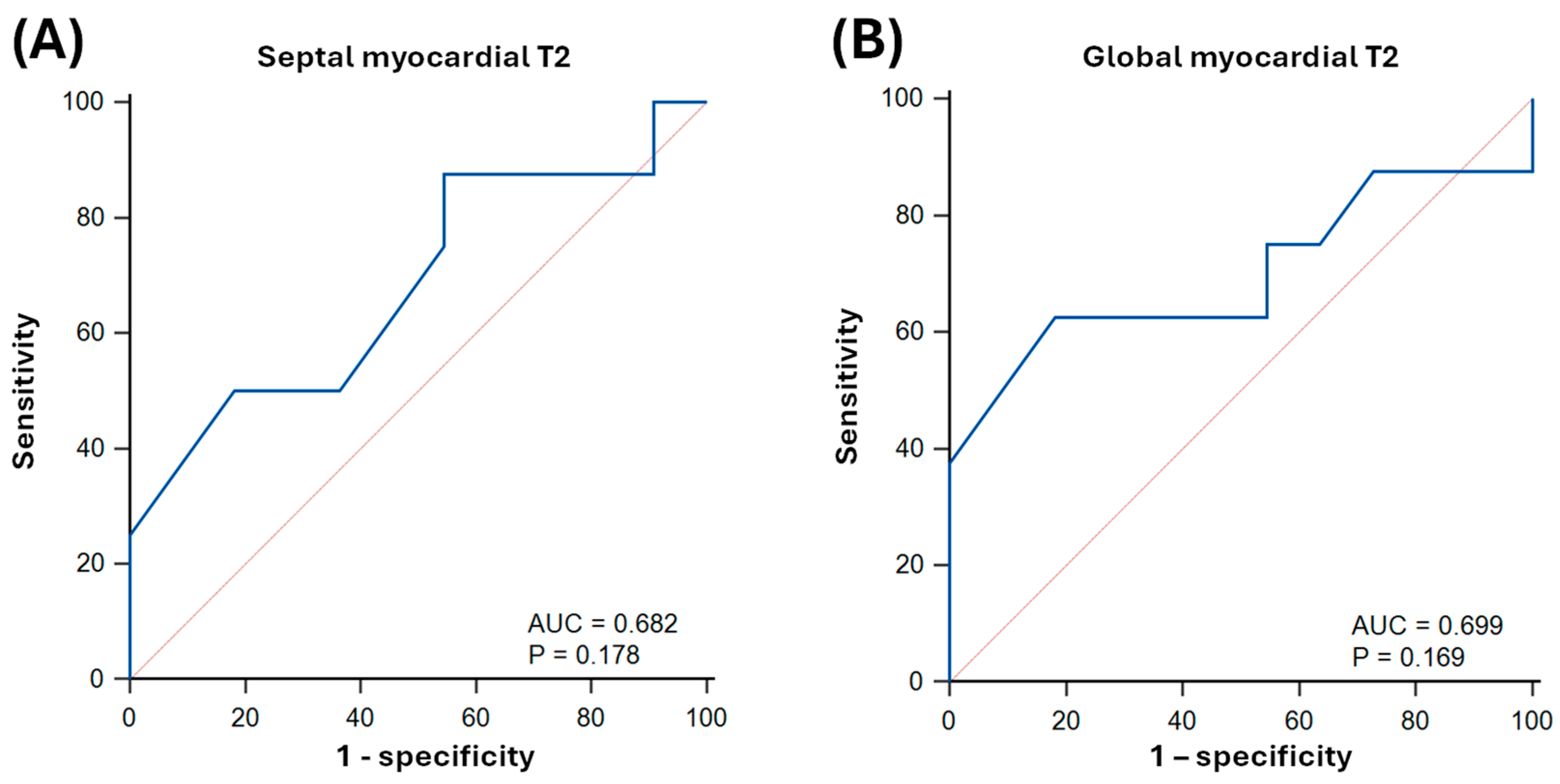
3.4. Predictive Value of Parametric Mapping Methods for LV Dysfunction in Sepsis Survivors
4. Discussion
4.1. Diffuse Myocardial Fibrosis in Sepsis Survivors
4.2. Myocardial Oedema in Sepsis Survivors
4.3. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: Analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef]
- La Via, L.; Sangiorgio, G.; Stefani, S.; Marino, A.; Nunnari, G.; Cocuzza, S.; La Mantia, I.; Cacopardo, B.; Stracquadanio, S.; Spampinato, S.; et al. The Global Burden of Sepsis and Septic Shock. Epidemiologia 2024, 5, 456–478. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-M.; Lee, M.-C.; Toh, H.S.; Chang, W.-T.; Chen, S.-Y.; Kuo, F.-H.; Tang, H.-J.; Hua, Y.-M.; Wei, D.; Melgarejo, J.; et al. Association of sepsis-induced cardiomyopathy and mortality: A systematic review and meta-analysis. Ann. Intensive Care 2022, 12, 112. [Google Scholar] [CrossRef]
- Hiraiwa, H.; Kasugai, D.; Okumura, T.; Murohara, T. Clinical implications of septic cardiomyopathy: A narrative review. Medicine 2024, 103, e37940. [Google Scholar] [CrossRef]
- Kakihana, Y.; Ito, T.; Nakahara, M.; Yamaguchi, K.; Yasuda, T. Sepsis-induced myocardial dysfunction: Pathophysiology and management. J. Intensive Care 2016, 4, 22. [Google Scholar] [CrossRef]
- Liang, Y.W.; Zhu, Y.F.; Zhang, R.; Zhang, M.; Ye, X.L.; Wei, J.R. Incidence, prognosis, and risk factors of sepsis-induced cardiomyopathy. World J. Clin. Cases 2021, 9, 9452–9468. [Google Scholar] [CrossRef]
- Muehlberg, F.; Blaszczyk, E.; Will, K.; Wilczek, S.; Brederlau, J.; Schulz-Menger, J. Characterization of critically ill patients with septic shock and sepsis-associated cardiomyopathy using cardiovascular MRI. ESC Heart Fail. 2022, 9, 2147–2156. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, Y.; Crouser, E.D.; Raman, S.V. Nonischemic myocardial changes detected by cardiac magnetic resonance in critical care patients with sepsis. Am. J. Respir. Crit. Care Med. 2013, 188, 1037–1039. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.M.; Shelhamer, J.H.; Bacharach, S.L.; Green, M.V.; Natanson, C.; Frederick, T.M.; Damske, B.A.; Parrillo, J.E. Profound but reversible myocardial depression in patients with septic shock. Ann. Intern. Med. 1984, 100, 483–490. [Google Scholar] [CrossRef]
- Parker, M.M.; Suffredini, A.F.; Natanson, C.; Ognibene, F.P.; Shelhamer, J.H.; Parrillo, J.E. Responses of left ventricular function in survivors and nonsurvivors of septic shock. J. Crit. Care 1989, 4, 19–25. [Google Scholar] [CrossRef]
- Malomo, S.; Oswald, T.; Stephenson, E.; Yip, A.; Alway, T.; Hadjivassilev, S.; Coombs, S.; Ellery, S.; Lee, J.; James, R.; et al. Characterisation of Post-Sepsis Cardiomyopathy Using Cardiovascular Magnetic Resonance. Diagnostics 2025, 15, 997. [Google Scholar] [CrossRef] [PubMed]
- Oswald, T.; Malomo, S.; Alway, T.; Hadjivassilev, S.; Coombs, S.; Ellery, S.; Lee, J.; Phillips, C.; Philips, B.; James, R.; et al. Guideline-Directed Medical Therapy in Sepsis Survivors with Left Ventricular Systolic Dysfunction: An Observational Study. J. Clin. Med. 2025, 14, 3253. [Google Scholar] [CrossRef]
- Jentzer, J.C.; Lawler, P.R.; Van Houten, H.K.; Yao, X.; Kashani, K.B.; Dunlay, S.M. Cardiovascular Events Among Survivors of Sepsis Hospitalization: A Retrospective Cohort Analysis. J. Am. Heart Assoc. 2023, 12, e027813. [Google Scholar] [CrossRef]
- Hollenberg, S.M. Sepsis-Associated Cardiomyopathy: Long-Term Prognosis, Management, and Guideline-Directed Medical Therapy. Curr. Cardiol. Rep. 2025, 27, 5. [Google Scholar] [CrossRef]
- Hundley, W.G.; Bluemke, D.A.; Bogaert, J.; Flamm, S.D.; Fontana, M.; Friedrich, M.G.; Grosse-Wortmann, L.; Karamitsos, T.D.; Kramer, C.M.; Kwong, R.Y.; et al. Society for Cardiovascular Magnetic Resonance (SCMR) guidelines for reporting cardiovascular magnetic resonance examinations. J. Cardiovasc. Magn. Reson. 2022, 24, 29. [Google Scholar] [CrossRef]
- Kim, R.J.; Wu, E.; Rafael, A.; Chen, E.L.; Parker, M.A.; Simonetti, O.; Klocke, F.J.; Bonow, R.O.; Judd, R.M. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. N. Engl. J. Med. 2000, 343, 1445–1453. [Google Scholar] [CrossRef]
- Aquaro, G.D.; De Gori, C.; Faggioni, L.; Parisella, M.L.; Cioni, D.; Lencioni, R.; Neri, E. Diagnostic and prognostic role of late gadolinium enhancement in cardiomyopathies. Eur. Heart J. Suppl. 2023, 25, C130–C136. [Google Scholar] [CrossRef]
- Meier, C.; Eisenblätter, M.; Gielen, S. Myocardial Late Gadolinium Enhancement (LGE) in Cardiac Magnetic Resonance Imaging (CMR)-An Important Risk Marker for Cardiac Disease. J. Cardiovasc. Dev. Dis. 2024, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Fent, G.J.; Garg, P.; Foley, J.R.J.; Swoboda, P.P.; Dobson, L.E.; Erhayiem, B.; Treibel, T.A.; Moon, J.C.; Greenwood, J.P.; Plein, S. Synthetic Myocardial Extracellular Volume Fraction. JACC Cardiovasc. Imaging 2017, 10, 1402–1404. [Google Scholar] [CrossRef]
- Messroghli, D.R.; Moon, J.C.; Ferreira, V.M.; Grosse-Wortmann, L.; He, T.; Kellman, P.; Mascherbauer, J.; Nezafat, R.; Salerno, M.; Schelbert, E.B.; et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J. Cardiovasc. Magn. Reson. 2016, 19, 75. [Google Scholar] [CrossRef] [PubMed]
- Messroghli, D.R.; Walters, K.; Plein, S.; Sparrow, P.; Friedrich, M.G.; Ridgway, J.P.; Sivananthan, M.U. Myocardial T1 mapping: Application to patients with acute and chronic myocardial infarction. Magn. Reson. Med. 2007, 58, 34–40. [Google Scholar] [CrossRef]
- Malomo, S.; Oswald, T.; Alway, T.; Hadjivassilev, S.; Coombs, S.; Ellery, S.; Lee, J.; Phillips, C.; Philips, B.; James, R.; et al. Characterization of Coronary Artery Disease in Sepsis Survivors. Biomedicines 2025, 13, 1181. [Google Scholar] [CrossRef]
- Nakamori, S.; Alakbarli, J.; Bellm, S.; Motiwala, S.R.; Addae, G.; Manning, W.J.; Nezafat, R. Native T1 value in the remote myocardium is independently associated with left ventricular dysfunction in patients with prior myocardial infarction. J. Magn. Reson. Imaging 2017, 46, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Fouda, S.; Godfrey, R.; Pavitt, C.; Alway, T.; Coombs, S.; Ellery, S.M.; Parish, V.; Silberbauer, J.; Liu, A. Cardiac Sarcoidosis and Inherited Cardiomyopathies: Clinical Masquerade or Overlap? J. Clin. Med. 2025, 14, 1609. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wang, H.P.; Liu, M.X.; Gu, H.; Yuan, X.S.; Biekan, J.; Wang, X.M. Early detection of myocardial fibrosis in cardiomyopathy in the absence of late enhancement: Role of T1 mapping and extracellular volume analysis. Eur. Radiol. 2023, 33, 1982–1991. [Google Scholar] [CrossRef]
- von Stumm, M.; Petersen, J.; Sinn, M.; Holst, T.; Sequeira-Gross, T.M.; Müller, L.; Pausch, J.; Bannas, P.; Adam, G.; Reichenspurner, H.; et al. Correlation of Myocardial Native T1 and Left Ventricular Reverse Remodeling after Valvular Surgery. J. Clin. Med. 2023, 12, 2649. [Google Scholar] [CrossRef] [PubMed]
- Puntmann, V.O.; Peker, E.; Chandrashekhar, Y.; Nagel, E. T1 Mapping in Characterizing Myocardial Disease. Circ. Res. 2016, 119, 277–299. [Google Scholar] [CrossRef]
- Giusca, S.; Steen, H.; Montenbruck, M.; Patel, A.R.; Pieske, B.; Erley, J.; Kelle, S.; Korosoglou, G. Multi-parametric assessment of left ventricular hypertrophy using late gadolinium enhancement, T1 mapping and strain-encoded cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2021, 23, 92. [Google Scholar] [CrossRef]
- Chen, W.; Doeblin, P.; Al-Tabatabaee, S.; Klingel, K.; Tanacli, R.; Jakob Weiß, K.; Stehning, C.; Patel, A.R.; Pieske, B.; Zou, J.; et al. Synthetic Extracellular Volume in Cardiac Magnetic Resonance Without Blood Sampling: A Reliable Tool to Replace Conventional Extracellular Volume. Circ. Cardiovasc. Imaging 2022, 15, e013745. [Google Scholar] [CrossRef] [PubMed]
- Haaf, P.; Garg, P.; Messroghli, D.R.; Broadbent, D.A.; Greenwood, J.P.; Plein, S. Cardiac T1 Mapping and Extracellular Volume (ECV) in clinical practice: A comprehensive review. J. Cardiovasc. Magn. Reson. 2016, 18, 89. [Google Scholar] [CrossRef]
- Yasuda, N.; Kato, S.; Horita, N.; Sekii, R.; Sawamura, S.; Nagase, H.; Utsunomiya, D. Synthetic extracellular volume fraction as an imaging biomarker of the myocardial interstitium without blood sampling: A systematic review and meta-analysis. J. Cardiovasc. Magn. Reson. 2025, 27, 101889. [Google Scholar] [CrossRef]
- Schmittinger, C.A.; Dünser, M.W.; Torgersen, C.; Luckner, G.; Lorenz, I.; Schmid, S.; Joannidis, M.; Moser, P.; Hasibeder, W.R.; Halabi, M.; et al. Histologic pathologies of the myocardium in septic shock: A prospective observational study. Shock 2013, 39, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, W.; Wan, K.; Liang, Y.; Jiang, X.; Wang, J.; Mui, D.; Li, Y.; Tang, S.; Guo, J.; et al. Myocardial Tissue Reverse Remodeling After Guideline-Directed Medical Therapy in Idiopathic Dilated Cardiomyopathy. Circulat. Heart Fail. 2021, 14, e007944. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, A.T.; Gil, K.E.; Varghese, J.; Simonetti, O.P.; Zareba, K.M. T2 mapping in myocardial disease: A comprehensive review. J. Cardiovasc. Magn. Reson. 2022, 24, 33. [Google Scholar] [CrossRef] [PubMed]
- Pedrizzetti, G.; Claus, P.; Kilner, P.J.; Nagel, E. Principles of cardiovascular magnetic resonance feature tracking and echocardiographic speckle tracking for informed clinical use. J. Cardiovasc. Magn. Reson. 2016, 18, 51. [Google Scholar] [CrossRef]
- Oswald, T.; Coombs, S.; Ellery, S.; Liu, A. Now and the Future: Medications Changing the Landscape of Cardiovascular Disease and Heart Failure Management. J. Clin. Med. 2025, 14, 3948. [Google Scholar] [CrossRef]
| Patients (n = 37) | |
|---|---|
| Age, years | 53 ± 16 |
| Male | 20 (57) |
| BMI, kg/m2 | 25 ± 6 |
| Causes of sepsis | |
| Pneumonia | 25 (68) |
| Gastroenterological | 4 (11) |
| Unknown origin | 3 (8) |
| Soft tissue/bacteraemia/abscess | 5 (14) |
| ICU care requirement | 16 (43) |
| Intubation | 5 (14) |
| Vasopressor support | 6 (16) |
| Inotropic support | 5 (14) |
| Symptoms | |
| Dyspnoea | 15 (41) |
| Chest pain | 9 (24) |
| Palpitations | 5 (14) |
| Co-morbidities | |
| Atrial fibrillation | 9 (24) |
| Hypertension | 9 (24) |
| Smoking (Ex-/current) | 6 (16) |
| COPD/Asthma | 6 (16) |
| Diabetes | 5 (14) |
| Hypercholesterolaemia | 3 (8) |
| Chronic kidney disease | 3 (8) |
| Pre-sepsis history of IHD | 2 (5) |
| Stroke/TIA | 1 (3) |
| Pre-sepsis history of heart failure | 0 (0) |
| Medications | |
| ACEi/ARB/ARNI | 20 (54) |
| Beta-blockers | 19 (51) |
| MRA | 19 (51) |
| SGLT2-Inhibitor | 16 (43) |
| Statins | 12 (32) |
| Anticoagulation | 10 (27) |
| Loop diuretics | 8 (22) |
| Anti-platelet agents | 7 (19) |
| Patients (n = 37) | |
|---|---|
| Day from sepsis event to CMR | 72 [25–123] |
| CMR volumes and function | |
| LV EDVi, mL/m2 | 91 [78–107] |
| LV ESVi, mL/m2 | 39 [28–57] |
| LV SVi, mL/m2 | 48 ± 12 |
| LV EF, % | 55 [43–62] |
| LV EF <50% | 16 (42) |
| LV EF <35% | 3 (8) |
| RV EDVi, mL/m2 | 86 ± 23 |
| RV ESVi, mL/m2 | 40 [32–46] |
| RV SVi, mL/m2 | 45 ± 13 |
| RV EF, % | 52 [48–59] |
| LV mass index, g/m2 | 64 [57–73] |
| LGE data | |
| LV LGE present | 19 (51) |
| Subepicardial/Mid-wall | 16 (43) |
| Subendocardial | 3 (8) |
| RV LGE present | 0 (0) |
| Native myocardial T1 values | |
| Septal | 1055 ± 65 |
| Global | 1051 ± 60 |
| Native myocardial T2 values | |
| Septal | 52 ± 7 (n = 19) |
| Global | 53 ± 6 (n = 19) |
| Synthetic ECV | 0.30 ± 0.04 (n = 21) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacobs, E.; Malomo, S.; Oswald, T.; Yip, A.; Alway, T.; Hadjivassilev, S.; Coombs, S.; Ellery, S.; Lee, J.; Phillips, C.; et al. Assessment of Diffuse Myocardial Fibrosis and Myocardial Oedema in Sepsis Survivors Using Cardiovascular Magnetic Resonance: Correlation with Left Ventricular Systolic Function. Biomedicines 2025, 13, 2119. https://doi.org/10.3390/biomedicines13092119
Jacobs E, Malomo S, Oswald T, Yip A, Alway T, Hadjivassilev S, Coombs S, Ellery S, Lee J, Phillips C, et al. Assessment of Diffuse Myocardial Fibrosis and Myocardial Oedema in Sepsis Survivors Using Cardiovascular Magnetic Resonance: Correlation with Left Ventricular Systolic Function. Biomedicines. 2025; 13(9):2119. https://doi.org/10.3390/biomedicines13092119
Chicago/Turabian StyleJacobs, Ella, Samuel Malomo, Thomas Oswald, Anthony Yip, Thomas Alway, Stanislav Hadjivassilev, Steven Coombs, Susan Ellery, Joon Lee, Claire Phillips, and et al. 2025. "Assessment of Diffuse Myocardial Fibrosis and Myocardial Oedema in Sepsis Survivors Using Cardiovascular Magnetic Resonance: Correlation with Left Ventricular Systolic Function" Biomedicines 13, no. 9: 2119. https://doi.org/10.3390/biomedicines13092119
APA StyleJacobs, E., Malomo, S., Oswald, T., Yip, A., Alway, T., Hadjivassilev, S., Coombs, S., Ellery, S., Lee, J., Phillips, C., Philips, B., Hildick-Smith, D., Parish, V., & Liu, A. (2025). Assessment of Diffuse Myocardial Fibrosis and Myocardial Oedema in Sepsis Survivors Using Cardiovascular Magnetic Resonance: Correlation with Left Ventricular Systolic Function. Biomedicines, 13(9), 2119. https://doi.org/10.3390/biomedicines13092119






