Artificial Intelligence-Driven Neuromodulation in Neurodegenerative Disease: Precision in Chaos, Learning in Loss
Abstract
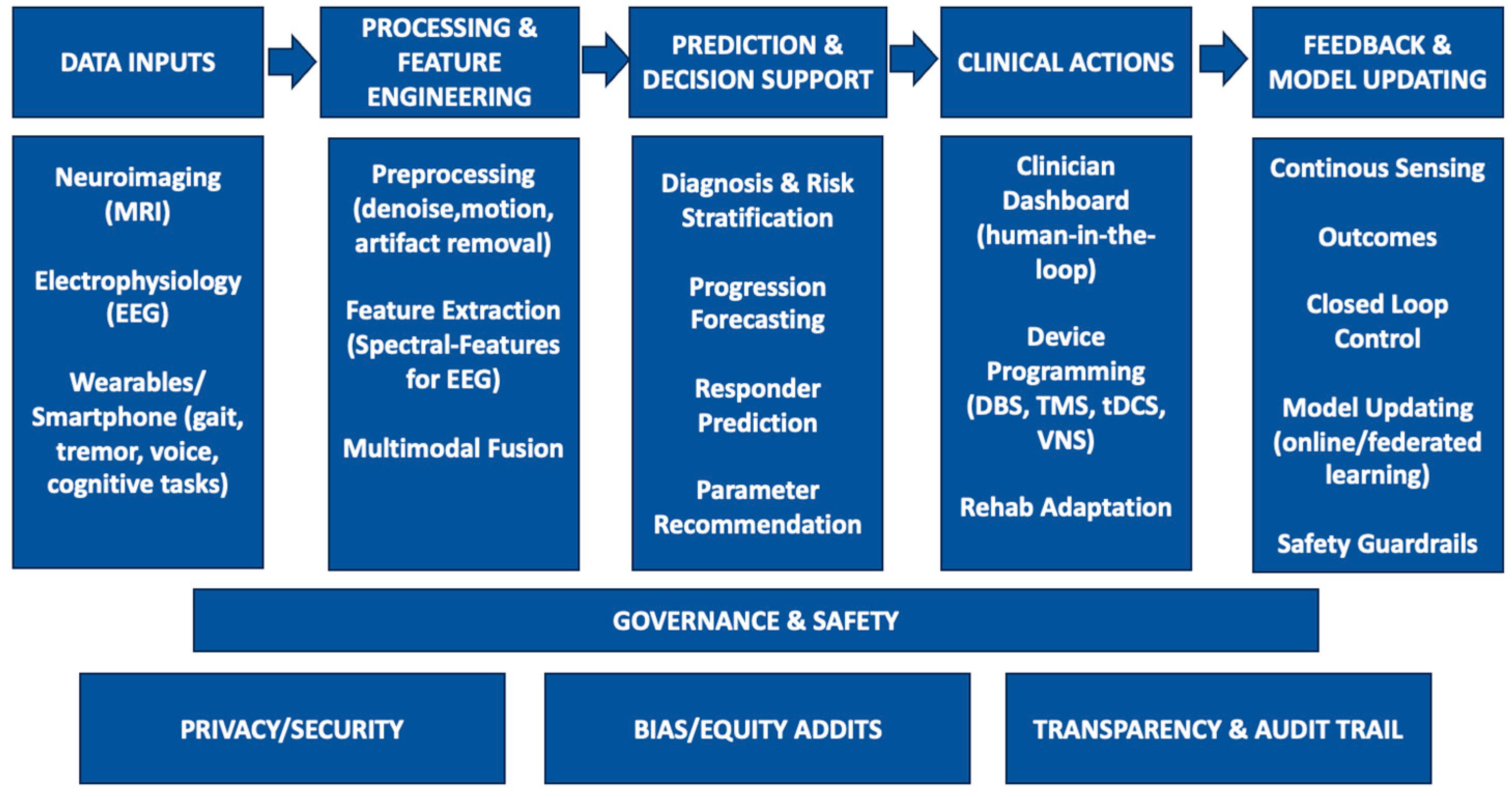
1. Introduction
2. Neuromodulation in Neurodegenerative Disease: Foundations and Clinical Innovations
2.1. Deep Brain Stimulation: Circuit-Based Therapies for Complex Neurological Symptoms
2.2. Transcranial Magnetic Stimulation: Expanding Clinical Horizons in Neurorehabilitation
2.3. Transcranial Direct Current Stimulation in Practice: Pathways to Clinical Impact
2.4. Vagus Nerve Stimulation: Bridging Central and Peripheral Modulation
2.5. Limitations and Challenges of Conventional Neuromodulation
3. The Role of Artificial Intelligence in Neuroscience and Rehabilitation
3.1. State-of-the-Art AI Methodologies in Neurorehabilitation
3.2. Applications of AI in Diagnosis, Patient Monitoring, and Outcome Prediction
3.3. Advantages and Limitations of AI Compared to Traditional Analytics in Neurorehabilitation
4. A New Clinical Paradigm: AI-Integrated, Adaptive Neuromodulation in PD, AD, and MS
4.1. Adaptive and Closed-Loop Neuromodulation Systems
4.2. AI-Driven Optimization of Neuromodulation Parameters
4.3. AI for Predictive Modeling and Biomarker Identification
4.4. Future Perspectives: The Convergence of AI, Neuroimaging, and Smart Biomaterials for Next-Generation Neuromodulation
5. Translational and Clinical Implications of AI-Enhanced Neuromodulation in AD, PD, and MS
5.1. Patient-Level Personalization and Connected Care
5.2. Standards, Ethics, and Health System Adoption
6. Discussion: Critical Reflections on the Path to Personalized Neurorehabilitation in Neurodegenerative Disorders
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gao, H.M.; Hong, J.S. Why neurodegenerative diseases are progressive: Uncontrolled inflammation drives disease progression. Trends Immunol. 2008, 29, 357–365. [Google Scholar] [CrossRef]
- Doroszkiewicz, J.; Winkel, I.; Mroczko, B. Comparative analysis of neuroinflammatory pathways in Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis: Insights into similarities and distinctions. Front. Neurosci. 2025, 19, 1579511. [Google Scholar] [CrossRef]
- Hussain, R.; Zubair, H.; Pursell, S.; Shahab, M. Neurodegenerative Diseases: Regenerative Mechanisms and Novel Therapeutic Approaches. Brain Sci. 2018, 8, 177. [Google Scholar] [CrossRef]
- Batista, P.; Pereira, A. Quality of life in patients with neurodegenerative diseases. J. Neurol. Neurosci. 2016, 7, 74. [Google Scholar] [CrossRef]
- Han, T.; Xu, Y.; Sun, L.; Hashimoto, M.; Wei, J. Microglial response to aging and neuroinflammation in the development of neurodegenerative diseases. Neural Regen. Res. 2024, 19, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- von Bernhardi, R.; Eugenín-von Bernhardi, L.; Eugenín, J. Microglial cell dysregulation in brain aging and neurodegeneration. Front. Aging Neurosci. 2015, 7, 124. [Google Scholar] [CrossRef] [PubMed]
- Daulatzai, M. Early stages of pathogenesis in memory impairment during normal senescence and Alzheimer’s disease. J. Alzheimers Dis. 2010, 20, 355–367. [Google Scholar] [CrossRef]
- World Health Organization. Over 1 in 3 People Affected by Neurological Conditions, the Leading Cause of Illness and Disability Worldwide. Geneva: WHO; 2024. Available online: https://www.who.int/news/item/14-03-2024-over-1-in-3-people-affected-by-neurological-conditions--the-leading-cause-of-illness-and-disability-worldwide (accessed on 28 August 2025).
- GBD 2021 Nervous System Disorders Collaborators. Global, regional, and national burden of disorders affecting the nervous system, 1990–2021: A systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024, 23, 344–381. [Google Scholar] [CrossRef]
- Guo, X.; Liu, X.; Lin, J.; Huang, Z.; Lin, S.; Zhang, M.; Xu, Z.; Lin, X. Global, regional, and national burden of four major neurological diseases in women from 1990 to 2021. Front. Public Health 2025, 13, 1561216. [Google Scholar] [CrossRef]
- Chen, D.; Zhao, Z.; Zhang, S.; Chen, S.; Wu, X.; Shi, J.; Liu, N.; Pan, C.; Tang, Y.; Meng, C.; et al. Evolving Therapeutic Landscape of Intracerebral Hemorrhage: Emerging Cutting-Edge Advancements in Surgical Robots, Regenerative Medicine, and Neurorehabilitation Techniques. Transl. Stroke Res. 2025, 16, 975–989. [Google Scholar] [CrossRef]
- Davidson, B.; Bhattacharya, A.; Sarica, C.; Darmani, G.; Raies, N.; Chen, R.; Lozano, A.M. Neuromodulation techniques—From non-invasive brain stimulation to deep brain stimulation. Neurotherapeutics 2024, 21, e00330. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Shang, L.; Hu, D.; Huang, J.; Wang, Y.; Li, M.; Song, Y.; Yang, Q.; Luo, Y.; Wang, Y.; et al. Neuromodulation techniques for modulating cognitive function: Enhancing stimulation precision and intervention effects. Neural Regen. Res. 2026, 21, 491–501. [Google Scholar] [CrossRef]
- Nardone, R.; Höller, Y.; Leis, S.; Höller, P.; Thon, N.; Thomschewski, A.; Golaszewski, S.; Brigo, F.; Trinka, E. Invasive and non-invasive brain stimulation for treatment of neuropathic pain in patients with spinal cord injury: A review. J. Spinal Cord Med. 2014, 37, 19–31. [Google Scholar] [CrossRef]
- Calderone, A.; Cardile, D.; Gangemi, A.; De Luca, R.; Quartarone, A.; Corallo, F.; Calabrò, R.S. Traumatic Brain Injury and Neuromodulation Techniques in Rehabilitation: A Scoping Review. Biomedicines 2024, 12, 438. [Google Scholar] [CrossRef]
- Danilov, Y.; Kaczmarek, K.; Skinner, K.; Tyler, M. Cranial Nerve Noninvasive Neuromodulation: New Approach to Neurorehabilitation. In Brain Neurotrauma: Molecular, Neuropsychological, and Rehabilitation Aspects; Kobeissy, F.H., Ed.; Chapter 44; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2015. Available online: https://www.ncbi.nlm.nih.gov/books/NBK299239/ (accessed on 28 August 2025).
- Johnson, M.D.; Lim, H.H.; Netoff, T.I.; Connolly, A.T.; Johnson, N.; Roy, A.; Holt, A.; Lim, K.O.; Carey, J.R.; Vitek, J.L.; et al. Neuromodulation for brain disorders: Challenges and opportunities. IEEE Trans. Biomed. Eng. 2013, 60, 610–624. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, X.; Cao, X.; Huang, C.; Liu, E.; Qian, S.; Liu, X.; Wu, Y.; Dong, F.; Qiu, C.-W.; et al. Artificial intelligence: A powerful paradigm for scientific research. Innovation 2021, 2, 100179. [Google Scholar] [CrossRef]
- Rasa, A.R. Artificial Intelligence and Its Revolutionary Role in Physical and Mental Rehabilitation: A Review of Recent Advancements. Biomed Res. Int. 2024, 2024, 9554590. [Google Scholar] [CrossRef]
- Calderone, A.; Latella, D.; Bonanno, M.; Quartarone, A.; Mojdehdehbaher, S.; Celesti, A.; Calabrò, R.S. Towards Transforming Neurorehabilitation: The Impact of Artificial Intelligence on Diagnosis and Treatment of Neurological Disorders. Biomedicines 2024, 12, 2415. [Google Scholar] [CrossRef] [PubMed]
- Gutman, B.; Shmilovitch, A.H.; Aran, D.; Shelly, S. Twenty-five years of AI in neurology: The journey of predictive medicine and biological breakthroughs. JMIR Neurotechnology 2024, 3, e59556. [Google Scholar] [CrossRef]
- Dhankhar, S.; Mujwar, S.; Garg, N.; Chauhan, S.; Saini, M.; Sharma, P.; Kumar, S.; Sharma, S.K.; Kamal, M.A.; Rani, N. Artificial Intelligence in The Management of Neurodegenerative Disorders. CNS Neurol. Disord. Drug Targets 2024, 23, 931–940. [Google Scholar] [CrossRef]
- Krishna, V.; Fasano, A. Neuromodulation: Update on current practice and future developments. Neurotherapeutics 2024, 21, e00371. [Google Scholar] [CrossRef]
- Lozano, A.M.; Lipsman, N.; Bergman, H.; Brown, P.; Chabardes, S.; Chang, J.W.; Matthews, K.; McIntyre, C.C.; Schlaepfer, T.E.; Schulder, M.; et al. Deep brain stimulation: Current challenges and future directions. Nat. Rev. Neurol. 2019, 15, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Dallapiazza, R.F.; De Vloo, P.; Fomenko, A.; Lee, D.J.; Hamani, C.; Munhoz, R.P.; Hodaie, M.; Lozano, A.M.; Fasano, A.; Kalia, S.K. Considerations for patient and target selection in deep brain stimulation surgery for Parkinson’s disease. In Parkinson’s Disease: Pathogenesis and Clinical Aspects; Stoker, T.B., Greenland, J.C., Eds.; Chapter 8; Codon Publications: Brisbane, Australia, 2018. Available online: https://www.ncbi.nlm.nih.gov/books/NBK536714/ (accessed on 28 August 2025).
- Mirza, S.; Yazdani, U.; Dewey, R., III; Patel, N.; Dewey, R.B., Jr.; Miocinovic, S.; Chitnis, S. Comparison of Globus Pallidus Interna and Subthalamic Nucleus in Deep Brain Stimulation for Parkinson Disease: An Institutional Experience and Review. Park. Dis. 2017, 2017, 3410820. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.; Ansari, A.; Kajita, Y.; Yamada, Y.; Kawase, T.; Kato, Y. Staged Deep Brain Stimulation of Ventral Intermediate Nucleus of the Thalamus for Suppression of Essential Tremors. Asian J. Neurosurg. 2019, 14, 1030–1032. [Google Scholar] [CrossRef]
- Willems, Y.R.; van der Gaag, N.A.; Kho, K.H.; Tveiten, Ø.V.; Krüger, M.T.; Jakobs, M.; ESSFN Young Functional Neurosurgeons taskforce. Choice of Implantable Pulse Generators for Deep Brain Stimulation: An Overview of Clinical Practice. Stereotact. Funct. Neurosurg. 2023, 101, 135–145. [Google Scholar] [CrossRef]
- Hariz, M.; Blomstedt, P. Deep brain stimulation for Parkinson’s disease. J. Intern. Med. 2022, 292, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Deuschl, G.; Schade-Brittinger, C.; Krack, P.; Volkmann, J.; Schäfer, H.; Bötzel, K.; Daniels, C.; Deutschländer, A.; Dillmann, U.; Eisner, W.; et al. A randomized trial of deep-brain stimulation for Parkinson’s disease. N. Engl. J. Med. 2006, 355, 896–908. [Google Scholar] [CrossRef]
- Burchiel, K.J.; Anderson, V.C.; Favre, J.; Hammerstad, J.P. Comparison of pallidal and subthalamic nucleus deep brain stimulation for advanced Parkinson’s disease: Results of a randomized, blinded pilot study. Neurosurgery 1999, 45, 1375–1382, discussion 1382–1384. [Google Scholar] [CrossRef]
- Weaver, F.M.; Follett, K.; Stern, M.; Hur, K.; Harris, C.; Marks, W.J., Jr.; Rothlind, J.; Sagher, O.; Reda, D.; Moy, C.S.; et al. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: A randomized controlled trial. JAMA 2009, 301, 63–73. [Google Scholar] [CrossRef]
- Nogueira, L.O.; Corso, A.M.S.; Dallé, L.D.C.; Antunes, V.L.J.; Fernandes, M.M.; Rabelo, I.S.M.; Della Coletta, M.V.; da Silva, C.C.; Barcelos, L.B.; Ferraz, H.B.; et al. Long-Term Quality of Life Trend after Subthalamic Stimulation for Parkinson’s Disease: An Updated Systematic Review and Meta-Analysis. Mov. Disord. Clin. Pract. 2025, 12, 577–587. [Google Scholar] [CrossRef]
- Petersen, J.J.; Kamp, C.B.; Faltermeier, P.; Juul, S.; Løkkegaard, A.; Gluud, C.; Jakobsen, J.C. Deep brain stimulation for Parkinson’s disease: Systematic review with meta-analysis and trial sequential analysis. BMJ Med. 2024, 3, e000705. [Google Scholar] [CrossRef]
- Brontë-Stewart, H. Deep brain stimulation. Neurol. Clin. Pract. 2012, 2, 67–71. [Google Scholar] [CrossRef]
- Kuhn, J.; Gründler, T.O.J.; Lenartz, D.; Sturm, V.; Klosterkötter, J.; Huff, W. Deep brain stimulation for psychiatric disorders. Dtsch. Arztebl. Int. 2010, 107, 105–113. [Google Scholar] [CrossRef]
- Martinez-Ramirez, D.; Jimenez-Shahed, J.; Leckman, J.F.; Porta, M.; Servello, D.; Meng, F.G.; Kuhn, J.; Huys, D.; Baldermann, J.C.; Foltynie, T.; et al. Efficacy and Safety of Deep Brain Stimulation in Tourette Syndrome: The International Tourette Syndrome Deep Brain Stimulation Public Database and Registry. JAMA Neurol. 2018, 75, 353–359. [Google Scholar] [CrossRef]
- Davidson, B.; Milosevic, L.; Kondrataviciute, L.; Kalia, L.V.; Kalia, S.K. Neuroscience fundamentals relevant to neuromodulation: Neurobiology of deep brain stimulation in Parkinson’s disease. Neurotherapeutics 2024, 21, e00348. [Google Scholar] [CrossRef]
- Shukla, A.W.; Bange, M.; Muthuraman, M. Patient, target, device, and program selection for DBS in Parkinson’s disease: Advancing toward precision care. npj Park. Dis. 2025, 11, 195. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, T.; Zhang, C.C.; Zeljic, K.; Zhan, S.; Sun, B.M.; Li, D.Y. The safety issues and hardware-related complications of deep brain stimulation therapy: A single-center retrospective analysis of 478 patients with Parkinson’s disease. Clin. Interv. Aging 2017, 12, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Fenoy, A.J.; Simpson, R.K., Jr. Risks of common complications in deep brain stimulation surgery: Management and avoidance. J. Neurosurg. 2014, 120, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Eldaief, M.C.; Dickerson, B.C.; Camprodon, J.A. Transcranial Magnetic Stimulation for the Neurological Patient: Scientific Principles and Applications. Semin. Neurol. 2022, 42, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.L.; Mak, M.K.Y. Effect of repetitive transcranial magnetic stimulation on physical function and motor signs in Parkinson’s disease: A systematic review and meta-analysis. Brain Stimul. 2016, 9, 475–487. [Google Scholar] [CrossRef]
- Conte, D.; Roman, A.; Beorchia, Y.; Pinzini, C.; Castriotta, L.; Valente, M. The effects of transcranial magnetic stimulation in motor symptoms of Parkinson’s disease: An overview of systematic reviews with meta-analysis. Neurol. Sci. 2025, 46, 3405–3418. [Google Scholar] [CrossRef]
- Chou, Y.H.; Hickey, P.T.; Sundman, M.; Song, A.W.; Chen, N.K. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson disease: A systematic review and meta-analysis. JAMA Neurol. 2015, 72, 432–440. [Google Scholar] [CrossRef]
- Dell’Osso, B.; Camuri, G.; Castellano, F.; Vecchi, V.; Benedetti, M.; Bortolussi, S.; Altamura, A.C. Meta-Review of Metanalytic Studies with Repetitive Transcranial Magnetic Stimulation (rTMS) for the Treatment of Major Depression. Clin. Pract. Epidemiol. Ment. Health 2011, 7, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Guo, Z.; McClure, M.A.; He, L.; Mu, Q. Effect of rTMS on Parkinson’s cognitive function: A systematic review and meta-analysis. BMC Neurol. 2020, 20, 377. [Google Scholar] [CrossRef]
- Zhou, L.; Guo, Z.; Xing, G.; Peng, H.; Cai, M.; Chen, H.; McClure, M.A.; He, L.; Xiong, L.; He, B.; et al. Antidepressant effects of repetitive transcranial magnetic stimulation over prefrontal cortex of Parkinson’s disease patients with depression: A meta-analysis. Front. Psychiatry 2019, 9, 769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhu, L.; Li, Y.; Yu, H.; Wang, T.; Chu, X. Therapeutic potential and mechanisms of repetitive transcranial magnetic stimulation in Alzheimer’s disease: A literature review. Eur. J. Med. Res. 2025, 30, 233. [Google Scholar] [CrossRef] [PubMed]
- Esposito, S.; Trojsi, F.; Cirillo, G.; de Stefano, M.; Di Nardo, F.; Siciliano, M.; Caiazzo, G.; Ippolito, D.; Ricciardi, D.; Buonanno, D.; et al. Repetitive Transcranial Magnetic Stimulation (rTMS) of Dorsolateral Prefrontal Cortex May Influence Semantic Fluency and Functional Connectivity in Fronto-Parietal Network in Mild Cognitive Impairment (MCI). Biomedicines 2022, 10, 994. [Google Scholar] [CrossRef]
- Georgopoulou, E.N.; Nousia, A.; Martzoukou, M.; Dimitriou, N.K.; Liampas, I.; Messinis, L.; Nasios, G. Efficacy of rTMS Combined with Cognitive and Language Training in People Living with Alzheimer’s Disease: A Systematic Review. Brain Sci. 2024, 14, 891. [Google Scholar] [CrossRef]
- Gaede, G.; Tiede, M.; Lorenz, I.; Brandt, A.U.; Pfueller, C.; Dörr, J.; Bellmann-Strobl, J.; Piper, S.K.; Roth, Y.; Zangen, A.; et al. Safety and preliminary efficacy of deep transcranial magnetic stimulation in MS-related fatigue. Neurol. Neuroimmunol. Neuroinflamm. 2017, 5, e423. [Google Scholar] [CrossRef]
- Makowiecki, K.; Stevens, N.; Cullen, C.L.; Zarghami, A.; Nguyen, P.T.; Johnson, L.; Rodger, J.; Hinder, M.R.; Barnett, M.; Young, K.M.; et al. Safety of low-intensity repetitive transcranial magneTic brAin stimUlation foR people living with mUltiple Sclerosis (TAURUS): Study protocol for a randomized controlled trial. Trials 2022, 23, 626. [Google Scholar] [CrossRef]
- Sy, A.; Thebault, S.; Aviv, R.I.; Auriat, A.M. An Overview of Transcranial Magnetic Stimulation and Its Application in Multiple Sclerosis. Appl. Sci. 2023, 13, 12679. [Google Scholar] [CrossRef]
- Zhou, X.; Li, K.; Chen, S.; Zhou, W.; Li, J.; Huang, Q.; Xu, T.; Gao, Z.; Wang, D.; Zhao, S.; et al. Clinical application of transcranial magnetic stimulation in multiple sclerosis. Front. Immunol. 2022, 13, 902658. [Google Scholar] [CrossRef]
- van den Heuvel, O.A.; Oberman, L.M. Current State of the Art of Transcranial Magnetic Stimulation in Psychiatry: Innovations and Challenges for the Future. Biol. Psychiatry 2024, 95, 485–487. [Google Scholar] [CrossRef]
- Fitzsimmons, S.M.D.D.; Oostra, E.; Postma, T.S.; van der Werf, Y.D.; van den Heuvel, O.A. Repetitive Transcranial Magnetic Stimulation—Induced Neuroplasticity and the Treatment of Psychiatric Disorders: State of the Evidence and Future Opportunities. Biol. Psychiatry 2024, 95, 592–600. [Google Scholar] [CrossRef]
- Jannati, A.; Oberman, L.M.; Rotenberg, A.; Pascual-Leone, A. Assessing the Mechanisms of Brain Plasticity by Transcranial Magnetic Stimulation. Neuropsychopharmacology 2023, 48, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, A.; Mancuso, M.; Sveva, V.; Maccarrone, D.; Todisco, A.; Motolese, F.; Santoro, F.; Pilato, F.; Spampinato, D.A.; Rocchi, L.; et al. Using TMS-EEG to Assess the Effects of Neuromodulation Techniques: A Narrative Review. Front. Hum. Neurosci. 2023, 17, 1247104. [Google Scholar] [CrossRef] [PubMed]
- Tăuƫan, A.-M.; Casula, E.P.; Pellicciari, M.C.; Borghi, I.; Maiella, M.; Bonni, S.; Minei, M.; Assogna, M.; Palmisano, A.; Smeralda, C.; et al. TMS-EEG Perturbation Biomarkers for Alzheimer’s Disease Patients Classification. Sci. Rep. 2023, 13, 7667. [Google Scholar] [CrossRef]
- Lu, Q.Q.; Zhu, P.A.; Li, Z.L.; Holmes, C.; Zhong, Y.; Liu, H.; Bao, X.; Xie, J.Y. Efficacy of Repetitive Transcranial Magnetic Stimulation over the Supplementary Motor Area on Motor Function in Parkinson’s Disease: A Meta-Analysis. Am. J. Phys. Med. Rehabil. 2025, 104, 318–324. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, W.; Zang, W. Repetitive Transcranial Magnetic Stimulation Improves Cognition, Depression, and Walking Ability in Patients with Parkinson’s Disease: A Meta-Analysis. BMC Neurol. 2024, 24, 490. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Antal, A.; Bestmann, S.; Bikson, M.; Brewer, C.; Brockmöller, J.; Carpenter, L.L.; Cincotta, M.; Chen, R.; Daskalakis, J.D.; et al. Safety and Recommendations for TMS Use in Healthy Subjects and Patient Populations, with Updates on Training, Ethical and Regulatory Issues: Expert Guidelines. Clin. Neurophysiol. 2021, 132, 269–306. [Google Scholar] [CrossRef]
- Kim, W.S.; Paik, N.J. Safety Review for Clinical Application of Repetitive Transcranial Magnetic Stimulation. Brain Neurorehabil. 2021, 14, e6. [Google Scholar] [CrossRef]
- Dalhuisen, I.; Bui, K.; Kleijburg, A.; van Oostrom, I.; Spijker, J.; van Exel, E.; van Mierlo, H.; de Waardt, D.; Arns, M.; Tendolkar, I.; et al. Cost-Effectiveness of rTMS as a Next Step in Antidepressant Non-Responders: A Randomized Comparison with Current Antidepressant Treatment Approaches. Acta Psychiatr. Scand. 2025, 151, 613–624. [Google Scholar] [CrossRef]
- ACOUSTIM Investigators Group; Bulteau, S.; Network, H.; Laurin, A.; Volteau, C.; Dert, C.; Lagalice, L.; Schirr-Bonnans, S.; Bukowski, N.; Guitteny, M.; et al. Cost-Utility Analysis of Curative and Maintenance Repetitive Transcranial Magnetic Stimulation (rTMS) for Treatment-Resistant Unipolar Depression: A Randomized Controlled Trial Protocol. Trials 2020, 21, 312. [Google Scholar] [CrossRef]
- Brunoni, A.R.; Nitsche, M.A.; Bolognini, N.; Bikson, M.; Wagner, T.; Merabet, L.; Edwards, D.J.; Valero-Cabre, A.; Rotenberg, A.; Pascual-Leone, A.; et al. Clinical research with transcranial direct current stimulation (tDCS): Challenges and future directions. Brain Stimul. 2012, 5, 175–195. [Google Scholar] [CrossRef] [PubMed]
- Chmiel, J.; Kurpas, D.; Stępień-Słodkowska, M. The Potential of Transcranial Direct Current Stimulation (tDCS) in Improving Quality of Life in Patients with Multiple Sclerosis: A Review and Discussion of Mechanisms of Action. J. Clin. Med. 2025, 14, 373. [Google Scholar] [CrossRef]
- Zhao, H.; Qiao, L.; Fan, D.; Zhang, S.; Turel, O.; Li, Y.; Li, J.; Xue, G.; Chen, A.; He, Q. Modulation of Brain Activity with Noninvasive Transcranial Direct Current Stimulation (tDCS): Clinical Applications and Safety Concerns. Front. Psychol. 2017, 8, 685. [Google Scholar] [CrossRef]
- Pilloni, G.; Charvet, L.E.; Bikson, M.; Palekar, N.; Kim, M.J. Potential of Transcranial Direct Current Stimulation in Alzheimer’s Disease: Optimizing Trials Toward Clinical Use. J. Clin. Neurol. 2022, 18, 391–400. [Google Scholar] [CrossRef]
- Sandrini, M.; Brambilla, M.; Manenti, R.; Rosini, S.; Cohen, L.G.; Cotelli, M. Noninvasive stimulation of prefrontal cortex strengthens existing episodic memories and reduces forgetting in the elderly. Front. Aging Neurosci. 2014, 6, 289. [Google Scholar] [CrossRef]
- Javadi, A.H.; Cheng, P.; Walsh, V. Short duration transcranial direct current stimulation (tDCS) modulates verbal memory. Brain Stimul. 2012, 5, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, P.C.; Fong, K.N.K.; Brown, T. Transcranial direct current stimulation as an adjunct to cognitive training for older adults with mild cognitive impairment: A randomized controlled trial. Ann. Phys. Rehabil. Med. 2021, 64, 101536. [Google Scholar] [CrossRef] [PubMed]
- Burton, C.Z.; Garnett, E.O.; Capellari, E.; Chang, S.E.; Tso, I.F.; Hampstead, B.M.; Taylor, S.F. Combined Cognitive Training and Transcranial Direct Current Stimulation in Neuropsychiatric Disorders: A Systematic Review and Meta-analysis. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2023, 8, 151–161. [Google Scholar] [CrossRef]
- Filmer, H.L.; Lyons, M.; Mattingley, J.B.; Dux, P.E. Anodal tDCS applied during multitasking training leads to transferable performance gains. Sci. Rep. 2017, 7, 12988. [Google Scholar] [CrossRef] [PubMed]
- Wörsching, J.; Padberg, F.; Ertl-Wagner, B.; Kumpf, U.; Kirsch, B.; Keeser, D. Imaging transcranial direct current stimulation (tDCS) of the prefrontal cortex—Correlation or causality in stimulation-mediated effects? Neurosci. Biobehav. Rev. 2016, 69, 333–356. [Google Scholar] [CrossRef] [PubMed]
- Bidaki, R.; Mirhosseini, H.; Zare, N. Impact of transcranial direct current stimulation on patients with Parkinson’s disease: A report of 2 cases. Hosp. Pract. Res. 2021, 6, 173–176. [Google Scholar] [CrossRef]
- Saiote, C.; Goldschmidt, T.; Timäus, C.; Steenwijk, M.D.; Opitz, A.; Antal, A.; Paulus, W.; Nitsche, M.A. Impact of transcranial direct current stimulation on fatigue in multiple sclerosis. Restor. Neurol. Neurosci. 2014, 32, 423–436. [Google Scholar] [CrossRef]
- Chalah, M.A.; Riachi, N.; Ahdab, R.; Mhalla, A.; Abdellaoui, M.; Créange, A.; Lefaucheur, J.-P.; Ayache, S.S. Effects of left DLPFC versus right PPC tDCS on multiple sclerosis fatigue. J. Neurol. Sci. 2017, 372, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Zakibakhsh, N.; Basharpoor, S.; Ghalyanchi Langroodi, H.; Narimani, M.; Nitsche, M.A.; Salehinejad, M.A. Repeated prefrontal tDCS for improving mental health and cognitive deficits in multiple sclerosis: A randomized, double-blind, parallel-group study. J. Transl. Med. 2024, 22, 843. [Google Scholar] [CrossRef]
- Sanches, C.; Stengel, C.; Godard, J.; Mertz, J.; Teichmann, M.; Migliaccio, R.; Valero-Cabré, A. Past, present, and future of non-invasive brain stimulation approaches to treat cognitive impairment in neurodegenerative diseases: Time for a comprehensive critical review. Front. Aging Neurosci. 2021, 12, 578339. [Google Scholar] [CrossRef]
- Yamada, Y.; Sumiyoshi, T. Neurobiological Mechanisms of Transcranial Direct Current Stimulation for Psychiatric Disorders: Neurophysiological, Chemical, and Anatomical Considerations. Front. Hum. Neurosci. 2021, 15, 631838. [Google Scholar] [CrossRef]
- Cancel, L.M.; Silas, D.; Bikson, M.; Tarbell, J.M. Direct Current Stimulation Modulates Gene Expression in Isolated Astrocytes with Implications for Glia-Mediated Plasticity. Sci. Rep. 2022, 12, 17964. [Google Scholar] [CrossRef]
- Qi, S.; Cao, L.; Wang, Q.; Sheng, Y.; Yu, J.; Liang, Z. The Physiological Mechanisms of Transcranial Direct Current Stimulation to Enhance Motor Performance: A Narrative Review. Biology 2024, 13, 790. [Google Scholar] [CrossRef]
- Oishi, R.; Takeda, I.; Ode, Y.; Okada, Y.; Kato, D.; Nakashima, H.; Imagama, S.; Wake, H. Neuromodulation with Transcranial Direct Current Stimulation Contributes to Motor Function Recovery via Microglia in Spinal Cord Injury. Sci. Rep. 2024, 14, 18031. [Google Scholar] [CrossRef]
- Hou, Y.; Liu, F.; Su, G.; Tu, S.; Lyu, Z. Systematic Review and Meta-Analysis of Transcranial Direct Current Stimulation (tDCS) for Global Cognition in Mild Cognitive Impairment and Alzheimer’s Disease. Geriatr. Nurs. 2024, 59, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Duan, Z.; Zhang, C. Transcranial Direct Current Stimulation for Parkinson’s Disease: Systematic Review and Meta-Analysis of Motor and Cognitive Effects. npj Park. Dis. 2024, 10, 214. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.X.D.; Mai, P.T.; Chang, Y.-J.; Hsieh, T.-H. Effects of Transcranial Direct Current Stimulation Alone and in Combination with Rehabilitation Therapies on Gait and Balance among Individuals with Parkinson’s Disease: A Systematic Review and Meta-Analysis. J. Neuroeng. Rehabil. 2024, 21, 27. [Google Scholar] [CrossRef]
- Charvet, L.; Goldberg, J.D.; Li, X.; Best, P.; Lustberg, M.; Shaw, M.; Zhovtis, L.; Gutman, J.; Datta, A.; Bikson, M.; et al. Home-Based Transcranial Direct Current Stimulation Paired with Cognitive Training to Reduce Fatigue in Multiple Sclerosis. Sci. Rep. 2025, 15, 4551. [Google Scholar] [CrossRef] [PubMed]
- Charvet, L.E.; Dobbs, B.; Shaw, M.T.; Bikson, M.; Datta, A.; Krupp, L.B. Remotely Supervised Transcranial Direct Current Stimulation for the Treatment of Fatigue in Multiple Sclerosis: Results from a Randomized, Sham-Controlled Trial. Mult. Scler. 2018, 24, 1760–1769. [Google Scholar] [CrossRef]
- Matsumoto, H.; Ugawa, Y. Adverse Events of tDCS and tACS: A Review. Clin. Neurophysiol. Pract. 2016, 2, 19–25. [Google Scholar] [CrossRef]
- Chhabra, H.; Bose, A.; Shivakumar, V.; Agarwal, S.M.; Sreeraj, V.S.; Shenoy, S.; Hazari, N.; Dinakaran, D.; Parlikar, R.; Koparde, V.; et al. Tolerance of Transcranial Direct Current Stimulation in Psychiatric Disorders: An Analysis of 2000+ Sessions. Psychiatry Res. 2020, 284, 112744. [Google Scholar] [CrossRef]
- Charvet, L.; George, A.; Charlson, E.; Lustberg, M.; Vogel-Eyny, A.; Eilam-Stock, T.; Cho, H.; Best, P.; Fernandez, L.; Datta, A.; et al. Home-Administered Transcranial Direct Current Stimulation Is a Feasible Intervention for Depression: An Observational Cohort Study. Front. Psychiatry 2023, 14, 1199773. [Google Scholar] [CrossRef]
- Kocahasan, M.; Stockbridge, M.D.; Stilling, J.; Utianski, R.L.; Sebastian, R.; Keser, Z. Remotely Supervised Transcranial Direct Current Stimulation in Post-Stroke Recovery: A Scoping Review. Medicina 2025, 61, 627. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, K. Mechanism and Applications of Vagus Nerve Stimulation. Curr. Issues Mol. Biol. 2025, 47, 122. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Dong, J.; Wang, Y.; Liu, Y. Exploring the potential of vagus nerve stimulation in treating brain diseases: A review of immunologic benefits and neuroprotective efficacy. Eur. J. Med. Res. 2023, 28, 444. [Google Scholar] [CrossRef]
- Shan, J.; Li, Z.; Ji, M.; Zhang, M.; Zhang, C.; Zhu, Y.; Feng, Z. Transcutaneous vagus nerve stimulation for Parkinson’s disease: A systematic review and meta-analysis. Front. Aging Neurosci. 2025, 16, 1498176. [Google Scholar] [CrossRef]
- Morris, R.; Yarnall, A.J.; Hunter, H.; Taylor, J.P.; Baker, M.R.; Rochester, L. Noninvasive vagus nerve stimulation to target gait impairment in Parkinson’s disease. Mov. Disord. 2019, 34, 918–919. [Google Scholar] [CrossRef]
- Mondal, B.; Choudhury, S.; Simon, B.; Baker, M.R.; Kumar, H. Noninvasive vagus nerve stimulation improves gait and reduces freezing of gait in Parkinson’s disease. Mov. Disord. 2019, 34, 917–918. [Google Scholar] [CrossRef]
- Hays, S.A.; Rennaker, R.L.; Kilgard, M.P. Targeting plasticity with vagus nerve stimulation to treat neurological disease. Prog. Brain Res. 2013, 207, 275–299. [Google Scholar] [CrossRef]
- Kamoga, R.; Godfrey, Z.; Rukundo, G.Z.; Nakidde, G. Effects of vagus nerve stimulation in Alzheimer’s disease: A systematic review and meta-analysis of clinical studies. J. Alzheimers Dis. Park. 2023, 13, 1000571. [Google Scholar]
- Austelle, C.W.; Cox, S.S.; Wills, K.E.; Badran, B.W. Vagus nerve stimulation (VNS): Recent advances and future directions. Clin. Auton. Res. 2024, 34, 529–547. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, C.; Le, L.H.D.; Gunasekaran, M.; Tracey, K.J.; Chernoff, D.; Levine, Y.A. Electrical stimulation of the vagus nerve ameliorates inflammation and disease activity in a rat EAE model of multiple sclerosis. Proc. Natl. Acad. Sci. USA 2024, 121, e2322577121. [Google Scholar] [CrossRef] [PubMed]
- Ben-Menachem, E.; Revesz, D.; Simon, B.J.; Silberstein, S. Surgically implanted and non-invasive vagus nerve stimulation: A review of efficacy, safety and tolerability. Eur. J. Neurol. 2015, 22, 1260–1268. [Google Scholar] [CrossRef]
- Forum on Neuroscience and Nervous System Disorders, Board on Health Sciences Policy, Institute of Medicine, The National Academies of Sciences, Engineering, and Medicine. 2. Overview of gaps, challenges, and potential opportunities. In Non-Invasive Neuromodulation of the Central Nervous System: Opportunities and Challenges: Workshop Summary; National Academies Press: Washington, DC, USA, 2015. Available online: https://www.ncbi.nlm.nih.gov/sites/books/NBK332919/ (accessed on 28 August 2025).
- Wang, W.; Li, R.; Li, C.; Liang, Q.; Gao, X. Advances in VNS Efficiency and Mechanisms of Action on Cognitive Functions. Front. Physiol. 2024, 15, 1452490. [Google Scholar] [CrossRef] [PubMed]
- Parpura, V.; Silva, G.A.; Tass, P.A.; Bennet, K.E.; Meyyappan, M.; Koehne, J.; Lee, K.H.; Andrews, R.J. Neuromodulation: Selected approaches and challenges. J. Neurochem. 2013, 124, 436–453. [Google Scholar] [CrossRef] [PubMed]
- Evancho, A.; Tyler, W.J.; McGregor, K. A review of combined neuromodulation and physical therapy interventions for enhanced neurorehabilitation. Front. Hum. Neurosci. 2023, 17, 1151218. [Google Scholar] [CrossRef] [PubMed]
- Neumann, W.J.; Köhler, R.M.; Kühn, A.A. A practical guide to invasive neurophysiology in patients with deep brain stimulation. Clin. Neurophysiol. 2022, 140, 171–180. [Google Scholar] [CrossRef]
- Sun, W.; Wu, Q.; Gao, L.; Zheng, Z.; Xiang, H.; Yang, K.; Yu, B.; Yao, J. Advancements in Transcranial Magnetic Stimulation Research and the Path to Precision. Neuropsychiatr. Dis. Treat. 2023, 19, 1841–1851. [Google Scholar] [CrossRef]
- Onciul, R.; Tataru, C.-I.; Dumitru, A.V.; Crivoi, C.; Serban, M.; Covache-Busuioc, R.-A.; Radoi, M.P.; Toader, C. Artificial Intelligence and Neuroscience: Transformative Synergies in Brain Research and Clinical Applications. J. Clin. Med. 2025, 14, 550. [Google Scholar] [CrossRef]
- Shivadekar, S. Artificial Intelligence for Cognitive Systems: Deep Learning, Neuro-symbolic Integration, and Human-Centric Intelligence; Deep Science Publishing: Fresno, CA, USA, 2025. [Google Scholar] [CrossRef]
- Sarker, I.H. Machine learning: Algorithms, real-world applications and research directions. SN Comput. Sci. 2021, 2, 160. [Google Scholar] [CrossRef]
- Oh, J.; Capezzuto, L.; Kriara, L.; Schjodt-Eriksen, J.; van Beek, J.; Bernasconi, C.; Montalban, X.; Butzkueven, H.; Kappos, L.; Giovannoni, G.; et al. Use of Smartphone-Based Remote Assessments of Multiple Sclerosis in Floodlight Open, a Global, Prospective, Open-Access Study. Sci. Rep. 2024, 14, 122. [Google Scholar] [CrossRef]
- Ko, H.; Park, N.; Kwon, S. AI-Based Dementia Risk Prediction Using Voice Digital Biomarkers: A Web-Based Platform. Alzheimers Dement 2025, 20, e088519. [Google Scholar] [CrossRef]
- Cay, G.; Pfeifer, V.A.; Lee, M.; Rouzi, M.D.; Nunes, A.S.; El-Refaei, N.; Momin, A.S.; Atique, M.M.U.; Mehl, M.R.; Vaziri, A.; et al. Harnessing Speech-Derived Digital Biomarkers to Detect and Quantify Cognitive Decline Severity in Older Adults. Gerontology 2024, 70, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Lefkovitz, I.; Walsh, S.; Blank, L.J.; Jetté, N.; Kummer, B.R. Direct Clinical Applications of Natural Language Processing in Common Neurological Disorders: Scoping Review. JMIR Neurotechnol. 2024, 3, e51822. [Google Scholar] [CrossRef]
- Taye, M.M. Understanding of Machine Learning with Deep Learning: Architectures, Workflow, Applications and Future Directions. Computers. 2023, 12, 91. [Google Scholar] [CrossRef]
- Mienye, I.D.; Swart, T.G.; Obaido, G.; Jordan, M.; Ilono, P. Deep Convolutional Neural Networks in Medical Image Analysis: A Review. Information 2025, 16, 195. [Google Scholar] [CrossRef]
- Muksimova, S.; Umirzakova, S.; Iskhakova, N.; Khaitov, A.; Cho, Y. Advanced convolutional neural network with attention mechanism for Alzheimer’s disease classification using MRI. Comput. Biol. Med. 2025, 190, 110095. [Google Scholar] [CrossRef] [PubMed]
- Tawhid, M.N.A.; Siuly, S.; Kabir, E.; Li, Y. Advancing Alzheimer’s disease detection: A novel convolutional neural network based framework leveraging EEG data and segment length analysis. Brain Inform. 2025, 12, 13. [Google Scholar] [CrossRef]
- Mao, S.; Sejdic, E. A review of recurrent neural network-based methods in computational physiology. IEEE Trans. Neural Netw. Learn. Syst. 2023, 34, 6983–7003. [Google Scholar] [CrossRef] [PubMed]
- Sadeghzadehyazdi, N.; Batabyal, T.; Acton, S. Modeling spatiotemporal patterns of gait anomaly with a CNN-LSTM deep neural network. Expert. Syst. Appl. 2021, 185, 115582. [Google Scholar] [CrossRef]
- Rad, E.M.; Mazinani, S.M.; Zendehbad, S.A. Diagnosis of Alzheimer’s disease using non-linear features of ERP signals through a hybrid attention-based CNN-LSTM model. Comput. Methods Programs Biomed. Update 2025, 7, 100192. [Google Scholar] [CrossRef]
- Seccia, R.; Romano, S.; Salvetti, M.; Crisanti, A.; Palagi, L.; Grassi, F. Machine learning use for prognostic purposes in multiple sclerosis. Life 2021, 11, 122. [Google Scholar] [CrossRef] [PubMed]
- Svoboda, E.; Bořil, T.; Rusz, J.; Tykalová, T.; Horáková, D.; Guttmann, C.R.G.; Blagoev, K.B.; Hatabu, H.; Valtchinov, V.I. Assessing clinical utility of machine learning and artificial intelligence approaches to analyze speech recordings in multiple sclerosis: A pilot study. Comput. Biol. Med. 2022, 148, 105853. [Google Scholar] [CrossRef]
- Hossain, M.; Mahabub, S.; Masum, A.; Jahan, I. Natural language processing (NLP) in analyzing electronic health records for better decision making. J. Comput. Sci. Technol. Stud. 2024, 6, 216–228. [Google Scholar] [CrossRef]
- Umesh, C.; Mahendra, M.; Bej, S.; Wolkenhauer, O.; Wolfien, M. Challenges and applications in generative AI for clinical tabular data in physiology. Pflugers Arch. 2025, 477, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Kamble, G.U.; Patil, C.S.; Alman, V.V.; Kundale, S.S.; Kim, J.H. Neuromorphic computing: Cutting-edge advances and future directions. In Artificial Intelligence; IntechOpen: London, UK, 2024. [Google Scholar] [CrossRef]
- Uhumuavbi, I. An adaptive conceptualisation of artificial intelligence and the law, regulation and ethics. Laws 2025, 14, 19. [Google Scholar] [CrossRef]
- Syaifullah, A.H.; Shiino, A.; Kitahara, H.; Ito, R.; Ishida, M.; Tanigaki, K. Machine Learning for Diagnosis of AD and Prediction of MCI Progression From Brain MRI Using Brain Anatomical Analysis Using Diffeomorphic Deformation. Front. Neurol. 2021, 11, 576029. [Google Scholar] [CrossRef] [PubMed]
- Salomon, A.; Gazit, E.; Ginis, P.; Urazalinov, B.; Takoi, H.; Yamaguchi, T.; Goda, S.; Lander, D.; Lacombe, J.; Sinha, A.K.; et al. A Machine Learning Contest Enhances Automated Freezing of Gait Detection and Reveals Time-of-Day Effects. Nat. Commun. 2024, 15, 4853. [Google Scholar] [CrossRef]
- Chang, B.; Geng, Z.; Mei, J.; Wang, Z.; Chen, P.; Jiang, Y.; Niu, C. Application of Multimodal Deep Learning and Multi-Instance Learning Fusion Techniques in Predicting STN-DBS Outcomes for Parkinson’s Disease Patients. Neurotherapeutics 2024, 21, e00471. [Google Scholar] [CrossRef]
- Wang, L.X.; Wang, Y.Z.; Han, C.G.; Zhao, L.; He, L.; Li, J. Revolutionizing early Alzheimer’s disease and mild cognitive impairment diagnosis: A deep learning MRI meta-analysis. Arq. Neuropsiquiatr. 2024, 82, s00441788657. [Google Scholar] [CrossRef]
- Garcia Santa Cruz, B.; Husch, A.; Hertel, F. Machine learning models for diagnosis and prognosis of Parkinson’s disease using brain imaging: General overview, main challenges, and future directions. Front. Aging Neurosci. 2023, 15, 1216163. [Google Scholar] [CrossRef]
- Valerio, J.E.; Aguirre Vera, G.J.; Fernandez Gomez, M.P.; Zumaeta, J.; Alvarez-Pinzon, A.M. AI-Driven Advances in Parkinson’s Disease Neurosurgery: Enhancing Patient Selection, Trial Efficiency, and Therapeutic Outcomes. Brain Sci. 2025, 15, 494. [Google Scholar] [CrossRef]
- Sigcha, L.; Borzì, L.; Amato, F.; Rechichi, I.; Ramos-Romero, C.; Cárdenas, A.; Gascó, L.; Olmo, G. Deep learning and wearable sensors for the diagnosis and monitoring of Parkinson’s disease: A systematic review. Expert Syst Appl. 2023, 229, 120541. [Google Scholar] [CrossRef]
- Falet, J.R.; Nobile, S.; Szpindel, A.; Barile, B.; Kumar, A.; Durso-Finley, J.; Arbel, T.; Arnold, D.L. The role of AI for MRI-analysis in multiple sclerosis-A brief overview. Front. Artif. Intell. 2025, 8, 1478068. [Google Scholar] [CrossRef]
- Nabizadeh, F.; Masrouri, S.; Ramezannezhad, E.; Ghaderi, A.; Sharafi, A.M.; Soraneh, S.; Moghadasi, A.N. Artificial intelligence in the diagnosis of multiple sclerosis: A systematic review. Mult. Scler. Relat. Disord. 2022, 59, 103673. [Google Scholar] [CrossRef]
- Kitaoka, Y.; Uchihashi, T.; Kawata, S.; Nishiura, A.; Yamamoto, T.; Hiraoka, S.-I.; Yokota, Y.; Isomura, E.T.; Kogo, M.; Tanaka, S.; et al. Role and potential of artificial intelligence in biomarker discovery and development of treatment strategies for amyotrophic lateral sclerosis. Int. J. Mol. Sci. 2025, 26, 4346. [Google Scholar] [CrossRef]
- Saba, L.; Maindarkar, M.; Johri, A.M.; Mantella, L.; Laird, J.R.; Khanna, N.N.; Paraskevas, K.I.; Ruzsa, Z.; Kalra, M.K.; Fernandes, J.F.E.; et al. UltraAIGenomics: Artificial Intelligence-Based Cardiovascular Disease Risk Assessment by Fusion of Ultrasound-Based Radiomics and Genomics Features for Preventive, Personalized and Precision Medicine: A Narrative Review. Rev. Cardiovasc. Med. 2024, 25, 184. [Google Scholar] [CrossRef] [PubMed]
- Shajari, S.; Kuruvinashetti, K.; Komeili, A.; Sundararaj, U. The Emergence of AI-Based Wearable Sensors for Digital Health Technology: A Review. Sensors 2023, 23, 9498. [Google Scholar] [CrossRef] [PubMed]
- Malaguti, M.C.; Longo, C.; Moroni, M.; Ragni, F.; Bovo, S.; Chierici, M.; Gios, L.; Avanzino, L.; Marchese, R.; Di Biasio, F.; et al. Machine Learning Predicts Risk of Falls in Parkison’s Disease Patients in a Multicenter Observational Study. Eur. J. Neurol. 2025, 32, e70118. [Google Scholar] [CrossRef]
- Franco, A.; Russo, M.; Amboni, M.; Ponsiglione, A.M.; Di Filippo, F.; Romano, M.; Amato, F.; Ricciardi, C. The role of deep learning and gait analysis in Parkinson’s disease: A systematic review. Sensors 2024, 24, 5957. [Google Scholar] [CrossRef]
- Dini, M.; Comi, G.; Leocani, L. Digital remote monitoring of people with multiple sclerosis. Front. Immunol. 2025, 16, 1514813. [Google Scholar] [CrossRef]
- Chen, Y.; Ou, Z.; Zhou, D.; Wu, X. Advancements and challenges of artificial intelligence-assisted electroencephalography in epilepsy management. J. Clin. Med. 2025, 14, 4270. [Google Scholar] [CrossRef] [PubMed]
- Perez, K.; Wisniewski, D.; Ari, A.; Lee, K.; Lieneck, C.; Ramamonjiarivelo, Z. Investigation into Application of AI and Telemedicine in Rural Communities: A Systematic Literature Review. Healthcare 2025, 13, 324. [Google Scholar] [CrossRef]
- Galmarini, C.M.; Lucius, M. Artificial intelligence: A disruptive tool for a smarter medicine. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 7462–7474. [Google Scholar] [CrossRef]
- Chakraborty, C.; Bhattacharya, M.; Pal, S.; Lee, S.-S. From machine learning to deep learning: Advances of the recent data-driven paradigm shift in medicine and healthcare. Curr. Res. Biotechnol. 2024, 7, 100164. [Google Scholar] [CrossRef]
- Basem, J.; Mani, R.; Sun, S.; Gilotra, K.; Dianati-Maleki, N.; Dashti, R. Clinical applications of artificial intelligence and machine learning in neurocardiology: A comprehensive review. Front. Cardiovasc. Med. 2025, 12, 1525966. [Google Scholar] [CrossRef]
- Moulaei, K.; Afshari, L.; Moulaei, R.; Sabet, B.; Mousavi, S.M.; Afrash, M.R. Explainable artificial intelligence for stroke prediction through comparison of deep learning and machine learning models. Sci. Rep. 2024, 14, 31392. [Google Scholar] [CrossRef] [PubMed]
- Houshi, S.; Khodakarami, Z.; Shaygannejad, A.; Khosravi, F.; Shaygannejad, V. AI-powered disease progression prediction in multiple sclerosis using magnetic resonance imaging: A systematic review and meta-analysis. Neurol. Res. 2025, 47, 1–15. [Google Scholar] [CrossRef]
- Vélez-Guerrero, M.A.; Callejas-Cuervo, M.; Mazzoleni, S. Artificial Intelligence-Based Wearable Robotic Exoskeletons for Upper Limb Rehabilitation: A Review. Sensors 2021, 21, 2146. [Google Scholar] [CrossRef]
- Abedi, A.; Colella, T.J.F.; Pakosh, M.; Khan, S.S. Artificial intelligence-driven virtual rehabilitation for people living in the community: A scoping review. npj Digit. Med. 2024, 7, 25. [Google Scholar] [CrossRef]
- Ueda, D.; Kakinuma, T.; Fujita, S.; Kamagata, K.; Fushimi, Y.; Ito, R.; Matsui, Y.; Nozaki, T.; Nakaura, T.; Fujima, N.; et al. Fairness of artificial intelligence in healthcare: Review and recommendations. Jpn. J. Radiol. 2024, 42, 3–15. [Google Scholar] [CrossRef]
- Rinderknecht, M.D.; Zanon, M.; Boonstra, T.A.; Angelini, L.; Stanev, D.; Chan, G.G.; Bunn, L.; Dondelinger, F.; Hosking, R.; Freeman, J.; et al. An Observational Study to Assess Validity and Reliability of Smartphone Sensor-Based Gait and Balance Assessments in Multiple Sclerosis: Floodlight GaitLab Protocol. Digit. Health 2023, 9, 20552076231205284. [Google Scholar] [CrossRef]
- Spagnolo, F.; Depeursinge, A.; Schädelin, S.; Akbulut, A.; Müller, H.; Barakovic, M.; Melie-Garcia, L.; Bach Cuadra, M.; Granziera, C. How Far MS Lesion Detection and Segmentation Are Integrated into the Clinical Workflow? A Systematic Review. NeuroImage Clin. 2023, 39, 103491. [Google Scholar] [CrossRef] [PubMed]
- Chudzik, A.; Śledzianowski, A.; Przybyszewski, A.W. Machine Learning and Digital Biomarkers Can Detect Early Stages of Neurodegenerative Diseases. Sensors 2024, 24, 1572. [Google Scholar] [CrossRef]
- Israni, S.; Bader, G.D.; Baranzini, S.E.; Capra, J.A.; Sirota, M.; Theodoris, C.V.; Ye, C.J. Precision medicine: Crossing the biomedical scales with AI. J. Precis. Med. Health Dis. 2025, 3, 100010. [Google Scholar] [CrossRef]
- Lanotte, F.; O’Brien, M.K.; Jayaraman, A. AI in Rehabilitation Medicine: Opportunities and Challenges. Ann. Rehabil. Med. 2023, 47, 444–458. [Google Scholar] [CrossRef] [PubMed]
- Calabrò, R.S.; Mojdehdehbaher, S. AI-driven telerehabilitation: Benefits and challenges of a transformative healthcare approach. AI 2025, 6, 62. [Google Scholar] [CrossRef]
- Pavuluri, S.; Sangal, R.; Sather, J.; Taylor, R.A. Balancing act: The complex role of artificial intelligence in addressing burnout and healthcare workforce dynamics. BMJ Health Care Inform. 2024, 31, e101120. [Google Scholar] [CrossRef]
- Obuchi, S.P.; Kojima, M.; Suzuki, H.; Garbalosa, J.C.; Imamura, K.; Ihara, K.; Hirano, H.; Sasai, H.; Fujiwara, Y.; Kawai, H. Artificial intelligence detection of cognitive impairment in older adults during walking. Alzheimers Dement 2024, 16, e70012. [Google Scholar] [CrossRef] [PubMed]
- Tsvetanov, F. Integrating AI technologies into remote monitoring patient systems. Eng. Proc. 2024, 70, 54. [Google Scholar] [CrossRef]
- Chen, P.; Wu, L.; Wang, L. AI fairness in data management and analytics: A review on challenges, methodologies and applications. Appl. Sci. 2023, 13, 10258. [Google Scholar] [CrossRef]
- Fehr, J.; Citro, B.; Malpani, R.; Lippert, C.; Madai, V.I. A trustworthy AI reality-check: The lack of transparency of artificial intelligence products in healthcare. Front. Digit. Health 2024, 6, 1267290. [Google Scholar] [CrossRef]
- Zahavi, I.; Ben Shitrit, I.; Einav, S. Using augmented intelligence to improve long term outcomes. Curr. Opin. Crit. Care 2024, 30, 523–531. [Google Scholar] [CrossRef]
- Mennella, C.; Maniscalco, U.; De Pietro, G.; Esposito, M. Ethical and regulatory challenges of AI technologies in healthcare: A narrative review. Heliyon 2024, 10, e26297. [Google Scholar] [CrossRef]
- Stanslaski, S.; Summers, R.L.S.; Tonder, L.; Tan, Y.; Case, M.; Raike, R.S.; Morelli, N.; Herrington, T.M.; Beudel, M.; Ostrem, J.L.; et al. Sensing Data and Methodology from the Adaptive DBS Algorithm for Personalized Therapy in Parkinson’s Disease (ADAPT-PD) Clinical Trial. npj Park. Dis. 2024, 10, 174. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, G.; Shi, L.; Zhang, C.; Wu, B.; Yang, A.; Meng, F.; Jiang, Y.; Zhang, J. Closed-Loop Adaptive Deep Brain Stimulation in Parkinson’s Disease: Procedures to Achieve It and Future Perspectives. J. Park. Dis. 2023, 13, 453–471. [Google Scholar] [CrossRef]
- Rosenbacke, R.; Melhus, Å.; McKee, M.; Stuckler, D. How Explainable Artificial Intelligence Can Increase or Decrease Clinicians’ Trust in AI Applications in Health Care: Systematic Review. JMIR AI 2024, 3, e53207. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Faustino, B.L.; Jagatheesaperumal, S.K.; Rolim, F.d.P.S.; de Albuquerque, V.H.C. Reinforcement Learning-Based Adaptive Deep Brain Stimulation Computational Model for the Treatment of Tremor in Parkinson’s Disease. Expert. Syst. Appl. 2025, 267, 126154. [Google Scholar] [CrossRef]
- Pozzi, N.G.; Isaias, I.U. Adaptive deep brain stimulation: Retuning Parkinson’s disease. Handb. Clin. Neurol. 2022, 184, 273–284. [Google Scholar] [CrossRef]
- Little, S.; Pogosyan, A.; Neal, S.; Zavala, B.; Zrinzo, L.; Hariz, M.; Foltynie, T.; Limousin, P.; Ashkan, K.; FitzGerald, J.; et al. Adaptive deep brain stimulation in advanced Parkinson disease. Ann. Neurol. 2013, 74, 449–457. [Google Scholar] [CrossRef]
- Velisar, A.; Syrkin-Nikolau, J.; Blumenfeld, Z.; Trager, M.H.; Afzal, M.F.; Prabhakar, V.; Bronte-Stewart, H. Dual threshold neural closed loop deep brain stimulation in Parkinson disease patients. Brain Stimul. 2019, 12, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Gilron, R.; Little, S.; Perrone, R.; Wilt, R.; de Hemptinne, C.; Yaroshinsky, M.S.; Racine, C.A.; Wang, S.S.; Ostrem, J.L.; Larson, P.S.; et al. Long-term wireless streaming of neural recordings for circuit discovery and adaptive stimulation in individuals with Parkinson’s disease. Nat. Biotechnol. 2021, 39, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Swann, N.C.; de Hemptinne, C.; Thompson, M.C.; Miocinovic, S.; Miller, A.M.; Gilron, R.; Ostrem, J.L.; Chizeck, H.J.; Starr, P.A. Adaptive deep brain stimulation for Parkinson’s disease using motor cortex sensing. J. Neural Eng. 2018, 15, 046006. [Google Scholar] [CrossRef]
- Ansó, J.; Benjaber, M.; Parks, B.; Parker, S.; Oehrn, C.R.; Petrucci, M.; Gilron, R.; Little, S.; Wilt, R.; Bronte-Stewart, H.; et al. Concurrent Stimulation and Sensing in Bi-Directional Brain Interfaces: A Multi-Site Translational Experience. J. Neural Eng. 2022, 19, 026025. [Google Scholar] [CrossRef] [PubMed]
- Cummins, D.D.; Kochanski, R.B.; Gilron, R.; Swann, N.C.; Little, S.; Hammer, L.H.; Starr, P.A. Chronic Sensing of Subthalamic Local Field Potentials: Comparison of First and Second Generation Implantable Bidirectional Systems within a Single Subject. Front. Neurosci. 2021, 15, 725797. [Google Scholar] [CrossRef] [PubMed]
- Gkintoni, E.; Vassilopoulos, S.P.; Nikolaou, G.; Vantarakis, A. Neurotechnological Approaches to Cognitive Rehabilitation in Mild Cognitive Impairment: A Systematic Review of Neuromodulation, EEG, Virtual Reality, and Emerging AI Applications. Brain Sci. 2025, 15, 582. [Google Scholar] [CrossRef] [PubMed]
- Aviles, M.; Sánchez-Reyes, L.M.; Álvarez-Alvarado, J.M.; Rodríguez-Reséndiz, J. Machine and deep learning trends in EEG-based detection and diagnosis of Alzheimer’s disease: A systematic review. Eng 2024, 5, 1464–1484. [Google Scholar] [CrossRef]
- Caravati, E.; Barbeni, F.; Chiarion, G.; Raggi, M.; Mesin, L. Closed-Loop Transcranial Electrical Neurostimulation for Sustained Attention Enhancement: A Pilot Study towards Personalized Intervention Strategies. Bioengineering 2024, 11, 467. [Google Scholar] [CrossRef]
- Tao, M.; Zhang, S.; Han, Y.; Li, C.; Wei, Q.; Chen, D.; Zhao, Q.; Yang, J.; Liu, R.; Fang, J.; et al. Efficacy of transcranial direct current stimulation on postoperative delirium in elderly patients undergoing lower limb major arthroplasty: A randomized controlled trial. Brain Stimul. 2023, 16, 88–96. [Google Scholar] [CrossRef]
- Jacques, F.H.; Apedaile, B.E.; Danis, I.; Sikati-Foko, V.; Lecompte, M.; Fortin, J. Motor Evoked Potential-A Pilot Study Looking at Reliability and Clinical Correlations in Multiple Sclerosis. J. Clin. Neurophysiol. 2024, 41, 357–364. [Google Scholar] [CrossRef]
- Rudroff, T. Digital Biomarkers and AI for Remote Monitoring of Fatigue Progression in Neurological Disorders: Bridging Mechanisms to Clinical Applications. Brain Sci. 2025, 15, 533. [Google Scholar] [CrossRef]
- Tecchio, F.; Bertoli, M.; Sbragia, E.; Stara, S.; Pasqualetti, P.; L’Abbate, T.; Croce, P.; Pizzichino, A.; Cancelli, A.; Armonaite, K.; et al. Fatigue relief in multiple sclerosis by personalized neuromodulation: A multicenter pilot study [FaremusGE]. Mult. Scler. Relat. Disord. 2025, 94, 106276. [Google Scholar] [CrossRef]
- Naji, Y.; Mahdaoui, M.; Klevor, R.; Kissani, N. Artificial Intelligence and Multiple Sclerosis: Up-to-Date Review. Cureus 2023, 15, e45412. [Google Scholar] [CrossRef]
- Soleimani, G.; Nitsche, M.A.; Bergmann, T.O.; Towhidkhah, F.; Violante, I.R.; Lorenz, R.; Kuplicki, R.; Tsuchiyagaito, A.; Mulyana, B.; Mayeli, A.; et al. Closing the Loop between Brain and Electrical Stimulation: Towards Precision Neuromodulation Treatments. Transl. Psychiatry 2023, 13, 279. [Google Scholar] [CrossRef]
- Ramachandran, A. Neuromorphic Computing: The Next Frontier in Brain-Inspired AI, Scalable Architectures, and Intelligent Systems. 2025. Available online: https://www.researchgate.net/publication/388876273_Neuromorphic_Computing_The_Next_Frontier_in_Brain-Inspired_AI_Scalable_Architectures_and_Intelligent_Systems (accessed on 28 August 2025).
- Muir, D.R.; Sheik, S. The Road to Commercial Success for Neuromorphic Technologies. Nat. Commun. 2025, 16, 3586. [Google Scholar] [CrossRef]
- Teo, Z.L.; Jin, L.; Li, S.; Miao, D.; Zhang, X.; Ng, W.Y.; Tan, T.F.; Lee, D.M.; Chua, K.J.; Heng, J.; et al. Federated Machine Learning in Healthcare: A Systematic Review on Clinical Applications and Technical Architecture. Cell Rep. Med. 2024, 5, 101419. [Google Scholar] [CrossRef] [PubMed]
- Cole, E.R.; Miocinovic, S. Are we ready for automated deep brain stimulation programming? Parkinsonism Relat. Disord. 2025, 134, 107347. [Google Scholar] [CrossRef]
- Eid, M.M.; Chinnaperumal, S.; Raju, S.K.; Kannan, S.; Alharbi, A.H.; Natarajan, S.; Khafaga, D.S.; Tawfeek, S.M. Machine learning-powered lead-free piezoelectric nanoparticle-based deep brain stimulation: A paradigm shift in Parkinson’s disease diagnosis and evaluation. AIP Adv. 2024, 14, 035040. [Google Scholar] [CrossRef]
- Boutet, A.; Madhavan, R.; Elias, G.J.B.; Joel, S.E.; Gramer, R.; Ranjan, M.; Paramanandam, V.; Xu, D.; Germann, J.; Loh, A.; et al. Predicting optimal deep brain stimulation parameters for Parkinson’s disease using functional MRI and machine learning. Nat. Commun. 2021, 12, 3043. [Google Scholar] [CrossRef]
- Neudorfer, C.; Butenko, K.; Oxenford, S.; Rajamani, N.; Achtzehn, J.; Goede, L.; Hollunder, B.; Ríos, A.S.; Hart, L.; Tasserie, J.; et al. Lead-DBS v3.0: Mapping Deep Brain Stimulation Effects to Local Anatomy and Global Networks. Neuroimage 2023, 268, 119862. [Google Scholar] [CrossRef]
- Van Hoornweder, S.; Nuyts, M.; Frieske, J.; Verstraelen, S.; Meesen, R.L.J.; Caulfield, K.A. Outcome Measures for Electric Field Modeling in tES and TMS: A Systematic Review and Large-Scale Modeling Study. Neuroimage 2023, 281, 120379. [Google Scholar] [CrossRef] [PubMed]
- Sellers, K.K.; Gilron, R.; Anso, J.; Louie, K.H.; Shirvalkar, P.R.; Chang, E.F.; Little, S.J.; Starr, P.A. Analysis-rcs-data: Open-Source Toolbox for the Ingestion, Time-Alignment, and Visualization of Sense and Stimulation Data from the Medtronic Summit RC+S System. Front. Hum. Neurosci. 2021, 15, 714256. [Google Scholar] [CrossRef]
- AlSharabi, K.; Bin Salamah, Y.; Aljalal, M.; Abdurraqeeb, A.M.; Alturki, F.A. EEG-based clinical decision support system for Alzheimer’s disorders diagnosis using EMD and deep learning techniques. Front. Hum. Neurosci. 2023, 17, 1190203. [Google Scholar] [CrossRef]
- Yperman, J.; Becker, T.; Valkenborg, D.; Popescu, V.; Hellings, N.; Van Wijmeersch, B.; Peeters, L.M. Machine learning analysis of motor evoked potential time series to predict disability progression in multiple sclerosis. BMC Neurol. 2020, 20, 72. [Google Scholar] [CrossRef] [PubMed]
- Watts, J.; Khojandi, A.; Shylo, O.; Ramdhani, R.A. Machine learning’s application in deep brain stimulation for Parkinson’s disease: A review. Brain Sci. 2020, 10, 809. [Google Scholar] [CrossRef]
- Stam, M.J.; de Neeling, M.G.J.; Keulen, B.J.; Hubers, D.; de Bie, R.M.A.; Schuurman, R.; Buijink, A.W.G.; van Wijk, B.C.M.; Beudel, M. AI-DBS study: Protocol for a longitudinal prospective observational cohort study of patients with Parkinson’s disease for the development of neuronal fingerprints using artificial intelligence. BMJ Open 2025, 15, e091563. [Google Scholar] [CrossRef] [PubMed]
- Winchester, L.M.; Harshfield, E.L.; Shi, L.; Badhwar, A.; Khleifat, A.A.; Clarke, N.; Dehsarvi, A.; Lengyel, I.; Lourida, I.; Madan, C.R.; et al. Artificial intelligence for biomarker discovery in Alzheimer’s disease and dementia. Alzheimers Dement. 2023, 19, 5860–5871. [Google Scholar] [CrossRef]
- Andrade, S.M.; da Silva-Sauer, L.; de Carvalho, C.D.; Medeiros de Araújo, E.L.; de Oliveira Lima, E.; Fernandes, F.M.L.; Moreira, K.L.A.F.; Camilo, M.E.; dos Santos Andrade, L.M.; Borges, D.T.; et al. Identifying biomarkers for tDCS treatment response in Alzheimer’s disease patients: A machine learning approach using resting-state EEG classification. Front. Hum. Neurosci. 2023, 17, 1234168. [Google Scholar] [CrossRef]
- Khattap, M.G.; Abd Elaziz, M.; Hassan, H.G.E.M.A.; Elgarayhi, A.; Sallah, M. AI-based model for automatic identification of multiple sclerosis based on enhanced sea-horse optimizer and MRI scans. Sci. Rep. 2024, 14, 12104. [Google Scholar] [CrossRef] [PubMed]
- Yousef, H.; Malagurski Tortei, B.; Castiglione, F. Predicting multiple sclerosis disease progression and outcomes with machine learning and MRI-based biomarkers: A review. J. Neurol. 2024, 271, 6543–6572. [Google Scholar] [CrossRef]
- Al-Iedani, O.; Lea, S.; Alshehri, A.; Maltby, V.E.; Saugbjerg, B.; Ramadan, S.; Lea, R.; Lechner-Scott, J. Multi-modal neuroimaging signatures predict cognitive decline in multiple sclerosis: A 5-year longitudinal study. Mult. Scler. Relat. Disord. 2024, 81, 105379. [Google Scholar] [CrossRef]
- Rieke, N.; Hancox, J.; Li, W.; Milletarì, F.; Roth, H.R.; Albarqouni, S.; Bakas, S.; Galtier, M.N.; Landman, B.A.; Maier-Hein, K.; et al. The Future of Digital Health with Federated Learning. npj Digit. Med. 2020, 3, 119. [Google Scholar] [CrossRef]
- Hassija, V.; Chamola, V.; Mahapatra, A.; Singal, A.; Goel, D.; Huang, K.; Scardapane, S.; Spinelli, I.; Mahmud, M.; Hussain, A. Interpreting black-box models: A review on explainable artificial intelligence. Cogn. Comput. 2024, 16, 45–74. [Google Scholar] [CrossRef]
- Ianculescu, M.; Băjenaru, L.; Vasilevschi, A.-M.; Gheorghe-Moisii, M.; Gheorghe, C.-G. Advancing neurodegenerative disease management: Technical, ethical, and regulatory insights from the NeuroPredict platform. Future Internet 2025, 17, 320. [Google Scholar] [CrossRef]
- Berkani, M.R.A.; Chouchane, A.; Himeur, Y.; Ouamane, A.; Miniaoui, S.; Atalla, S.; Mansoor, W.; Al-Ahmad, H. Advances in federated learning: Applications and challenges in smart building environments and beyond. Computers 2025, 14, 124. [Google Scholar] [CrossRef]
- Cao, D.; Ding, J. Recent advances in regenerative biomaterials. Regen Biomater. 2022, 9, rbac098. [Google Scholar] [CrossRef]
- Jawarkar, S.G.; Pillai, M.; Chavan, P.; Sengupta, P. Next-generation smart biomaterials for storage and extraction of bioanalytical samples: Current standing and path forward. Microchem. J. 2024, 207, 111846. [Google Scholar] [CrossRef]
- Merk, T.; Peterson, V.; Köhler, R.; Haufe, S.; Richardson, R.M.; Neumann, W.J. Machine learning based brain signal decoding for intelligent adaptive deep brain stimulation. Exp. Neurol. 2022, 351, 113993. [Google Scholar] [CrossRef]
- Wischnewski, M.; Shirinpour, S.; Alekseichuk, I.; Lapid, M.I.; Nahas, Z.; Lim, K.O.; Croarkin, P.E.; Opitz, A. Real-time TMS-EEG for brain state-controlled research and precision treatment: A narrative review and guide. J. Neural Eng. 2024, 21, 061001. [Google Scholar] [CrossRef] [PubMed]
- Pini, L.; Pizzini, F.B.; Boscolo-Galazzo, I.; Ferrari, C.; Galluzzi, S.; Cotelli, M.; Gobbi, E.; Cattaneo, A.; Cotelli, M.S.; Geroldi, C.; et al. Brain network modulation in Alzheimer’s and frontotemporal dementia with transcranial electrical stimulation. Neurobiol. Aging 2022, 111, 24–34. [Google Scholar] [CrossRef]
- Praet, J.; Anderhalten, L.; Comi, G.; Horakova, D.; Ziemssen, T.; Vermersch, P.; Lukas, C.; van Leemput, K.; Steppe, M.; Aguilera, C.; et al. A future of AI-driven personalized care for people with multiple sclerosis. Front. Immunol. 2024, 15, 1446748. [Google Scholar] [CrossRef] [PubMed]
- Montori, V.M.; Ruissen, M.M.; Hargraves, I.G.; Brito, J.P.; Kunneman, M. Shared Decision-Making as a Method of Care. BMJ Evid. Based Med. 2023, 28, 213–217. [Google Scholar] [CrossRef]
- Vasey, B.; Nagendran, M.; Campbell, B.; Clifton, D.A.; Collins, G.S.; Denaxas, S.; Denniston, A.K.; Faes, L.; Geerts, B.; Ibrahim, M.; et al. Reporting Guideline for the Early-Stage Clinical Evaluation of Decision Support Systems Driven by Artificial Intelligence: DECIDE-AI. Nat. Med. 2022, 28, 924–933. [Google Scholar] [CrossRef]
- Chaturvedi, U.; Baghel Chauhan, S.; Singh, I. The impact of artificial intelligence on remote healthcare: Enhancing patient engagement, connectivity, and overcoming challenges. Intell Pharm. 2025, in press. [Google Scholar] [CrossRef]
- Bekbolatova, M.; Mayer, J.; Ong, C.W.; Toma, M. Transformative Potential of AI in Healthcare: Definitions, Applications, and Navigating the Ethical Landscape and Public Perspectives. Healthcare 2024, 12, 125. [Google Scholar] [CrossRef]
- Ardito, V.; Cappellaro, G.; Compagni, A.; Petracca, F.; Preti, L.M. Adoption of artificial intelligence applications in clinical practice: Insights from a Survey of Healthcare Organizations in Lombardy, Italy. Digit. Health 2025, 11, 20552076251355680. [Google Scholar] [CrossRef] [PubMed]
- Vidalis, T. Artificial Intelligence in Biomedicine: A Legal Insight. BioTech 2021, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Junaid, S.B.; Imam, A.A.; Balogun, A.O.; De Silva, L.C.; Surakat, Y.A.; Kumar, G.; Abdulkarim, M.; Shuaibu, A.N.; Garba, A.; Sahalu, Y.; et al. Recent Advancements in Emerging Technologies for Healthcare Management Systems: A Survey. Healthcare 2022, 10, 1940. [Google Scholar] [CrossRef] [PubMed]
- Goktas, P.; Grzybowski, A. Shaping the Future of Healthcare: Ethical Clinical Challenges and Pathways to Trustworthy AI. J. Clin. Med. 2025, 14, 1605. [Google Scholar] [CrossRef]
| Neuromodulation Technique | Imaging/Biomarker Guidance | Patient Suitability/Contraindications | Combination with Other Therapies | Unique Adverse Effects in Neurodegenerative Disorders (NDs) | Remote Monitoring/Telemedicine | Typical Session Duration/Frequency |
|---|---|---|---|---|---|---|
| Deep Brain Stimulation (DBS) | This technique utilizes MRI and microelectrode guidance as well as local field potentials for adaptive stimulation to optimize targeting and outcomes [109]. | DBS is generally not suitable for individuals with significant cognitive impairment, severe psychiatric comorbidities, or pronounced frailty. Careful patient selection is especially important in elderly populations [29,39]. | DBS has shown evidence of synergy when used alongside medication and physiotherapy, and it is currently being investigated in combination with cognitive rehabilitation protocols for AD [29,33]. | Patients may experience surgical risks such as infection and hemorrhage, and there is also the potential for neuropsychiatric effects. If the electrode targeting is not optimal, there is a risk of worsened cognition [29,40,41]. | There is a growing possibility for remote adjustment and monitoring of DBS devices, allowing for improved follow-up and patient management [39]. | The procedure requires a multi-hour surgical implantation, followed by regular programming sessions and long-term device management [29,32]. |
| Transcranial Magnetic Stimulation (TMS) | TMS often relies on MRI navigation, and in some studies, EEG or cognitive biomarkers are employed to guide stimulation protocols for enhanced efficacy [56]. | TMS is contraindicated in individuals with epilepsy, metal implants, or unstable medical conditions, and should be used with caution in advanced dementia [42,56]. | It Is frequently combined with cognitive training in AD and with physical rehabilitation in PD and MS. There is also potential synergy with pharmacological treatments [43,44,51,52]. | The most common adverse effects include transient headache and discomfort. There is a rare risk of seizure, and agitation may occur in individuals with severe dementia [42,56]. | Currently, TMS is primarily clinic-based. Home or remote use is limited but represents an ongoing area of research [56]. | Typical treatment consists of sessions lasting twenty to forty minutes, administered daily or several times per week for a series of weeks. Maintenance protocols may be used for chronic NDs [42]. |
| Transcranial Direct Current Stimulation (tDCS) | Some clinical trials utilize EEG or cognitive biomarkers, and there is emerging use of artificial intelligence for the optimization of stimulation parameters [76]. | tDCS is generally well tolerated, but caution is advised in patients with epilepsy or unstable cardiac conditions. Data on its use in severe dementia are limited [67,81]. | It is commonly combined with cognitive or physical training and there is strong evidence supporting additional benefits in AD, mild cognitive impairment, and PD. The technique is also feasible for use in the home setting [73,74,81]. | Adverse effects are usually mild, including skin irritation or tingling. In rare cases, confusion may develop in elderly patients with cognitive impairment [67]. | There is high potential for mobile or home-based tDCS systems, with development of apps and cloud data integration for supervision and monitoring [67,81]. | Sessions typically last twenty to thirty minutes, are performed daily or several times per week, and are often continued for several weeks or months [67,70]. |
| Vagus Nerve Stimulation (VNS) | Cardiac monitoring is used to trigger stimulation in some cases, and there is exploratory research on using EEG and neurochemical markers to guide therapy [95,96]. | Surgical VNS should be avoided in patients with severe cardiopulmonary disease or coagulopathy. Non-invasive VNS is currently under evaluation for broader patient populations [104,105]. | Early evidence suggests synergy with task-specific rehabilitation with pharmacotherapy for PD. There is also some investigation into combining VNS with cognitive training [20,93,100,106,108]. | Adverse effects may include hoarseness, cough, and vocal changes in patients with implanted devices, as well as surgical risks. Rarely, patients may experience bradycardia or dyspnea [104,105]. | Non-invasive VNS devices are increasingly compatible with remote use, and remote patient monitoring is under active investigation [104]. | Implanted VNS requires regular surgical check-ups, whereas non-invasive VNS may involve multiple daily sessions that can be performed at home [105]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calderone, A.; Latella, D.; La Fauci, E.; Puleo, R.; Sergi, A.; De Francesco, M.; Mauro, M.; Foti, A.; Salemi, L.; Calabrò, R.S. Artificial Intelligence-Driven Neuromodulation in Neurodegenerative Disease: Precision in Chaos, Learning in Loss. Biomedicines 2025, 13, 2118. https://doi.org/10.3390/biomedicines13092118
Calderone A, Latella D, La Fauci E, Puleo R, Sergi A, De Francesco M, Mauro M, Foti A, Salemi L, Calabrò RS. Artificial Intelligence-Driven Neuromodulation in Neurodegenerative Disease: Precision in Chaos, Learning in Loss. Biomedicines. 2025; 13(9):2118. https://doi.org/10.3390/biomedicines13092118
Chicago/Turabian StyleCalderone, Andrea, Desirèe Latella, Elvira La Fauci, Roberta Puleo, Arturo Sergi, Mariachiara De Francesco, Maria Mauro, Angela Foti, Leda Salemi, and Rocco Salvatore Calabrò. 2025. "Artificial Intelligence-Driven Neuromodulation in Neurodegenerative Disease: Precision in Chaos, Learning in Loss" Biomedicines 13, no. 9: 2118. https://doi.org/10.3390/biomedicines13092118
APA StyleCalderone, A., Latella, D., La Fauci, E., Puleo, R., Sergi, A., De Francesco, M., Mauro, M., Foti, A., Salemi, L., & Calabrò, R. S. (2025). Artificial Intelligence-Driven Neuromodulation in Neurodegenerative Disease: Precision in Chaos, Learning in Loss. Biomedicines, 13(9), 2118. https://doi.org/10.3390/biomedicines13092118








