A MEMS µ-Preconcentrator Employing a Carbon Molecular Sieve Membrane for Highly Volatile Organic Compound Sampling
Abstract
1. Introduction
2. Materials and Methods
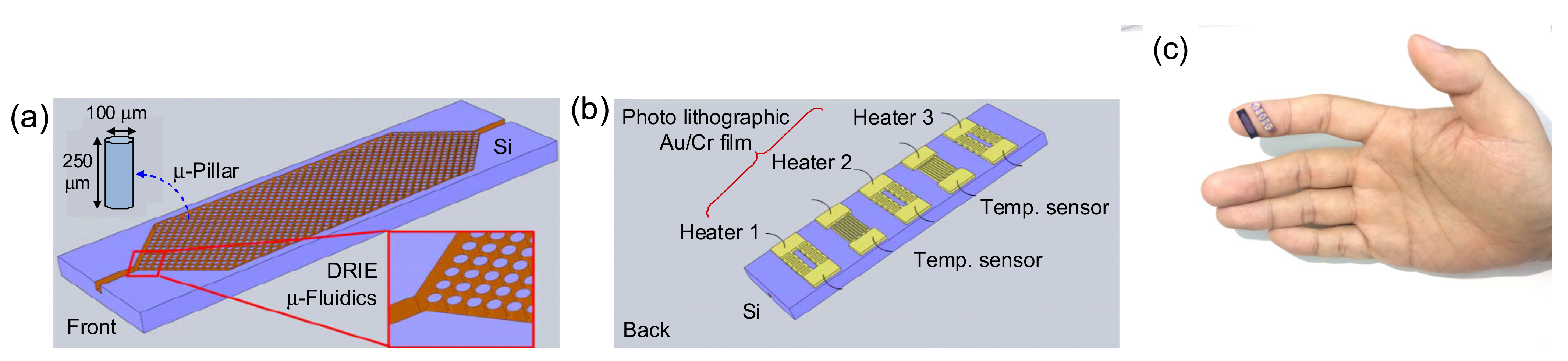
2.1. µ-Preconcentrator Design and Fabrication
2.2. In-Situ Grown of Carbon Molecular Sieve Membrane
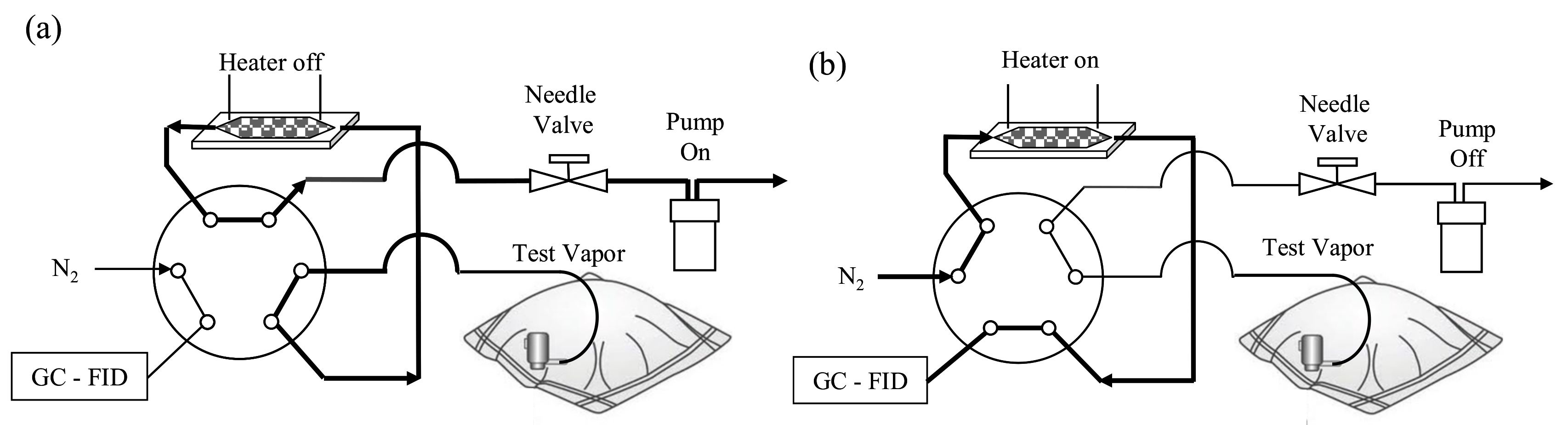
2.3. Preconcentration Test Setup
3. Results
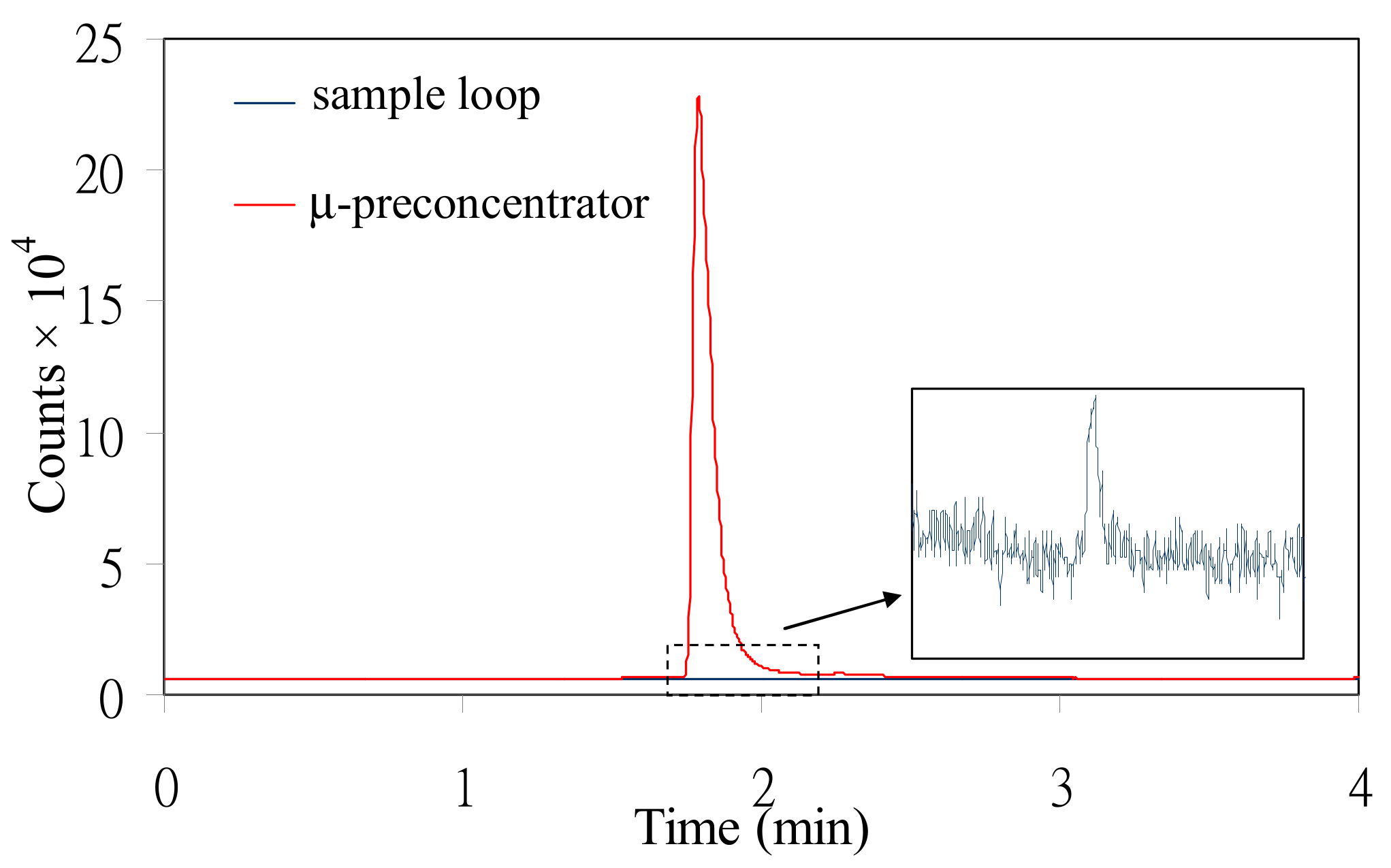
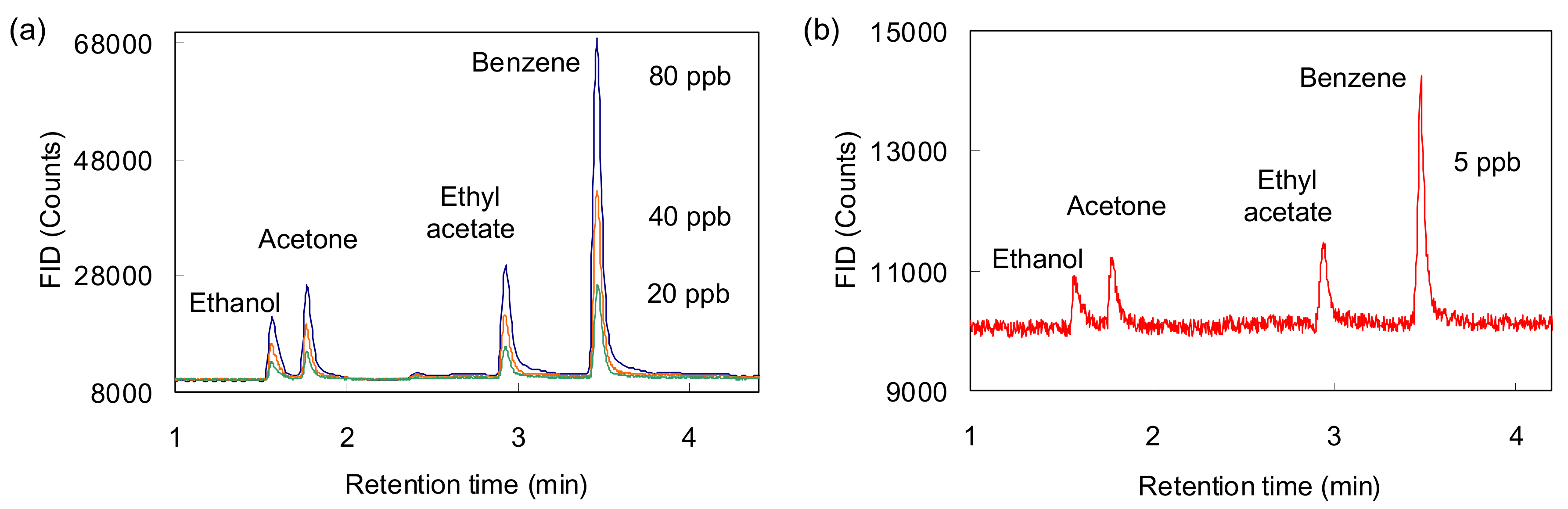
3.1. Initial Peak Amplification Test
3.2. Desorption Residual Test
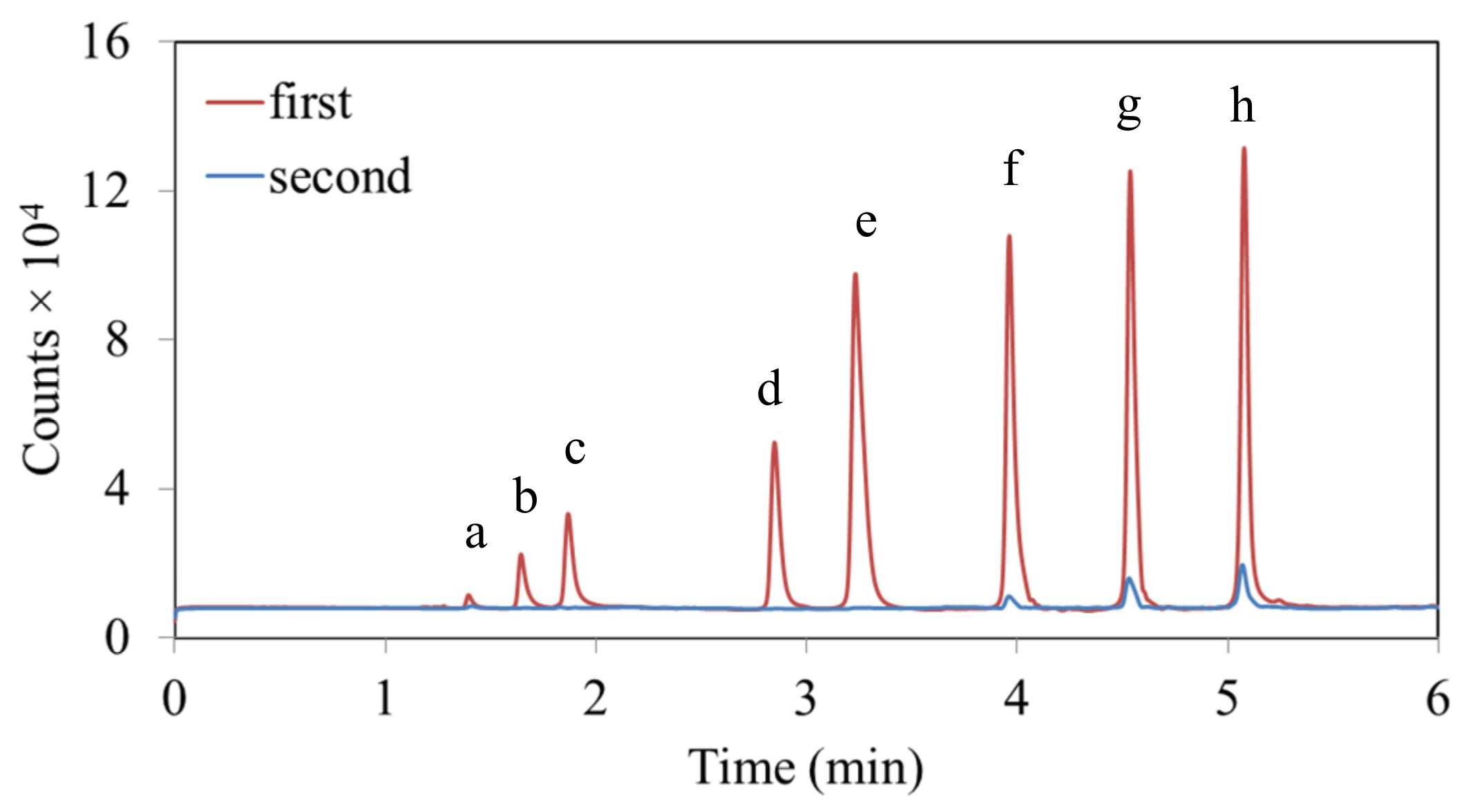
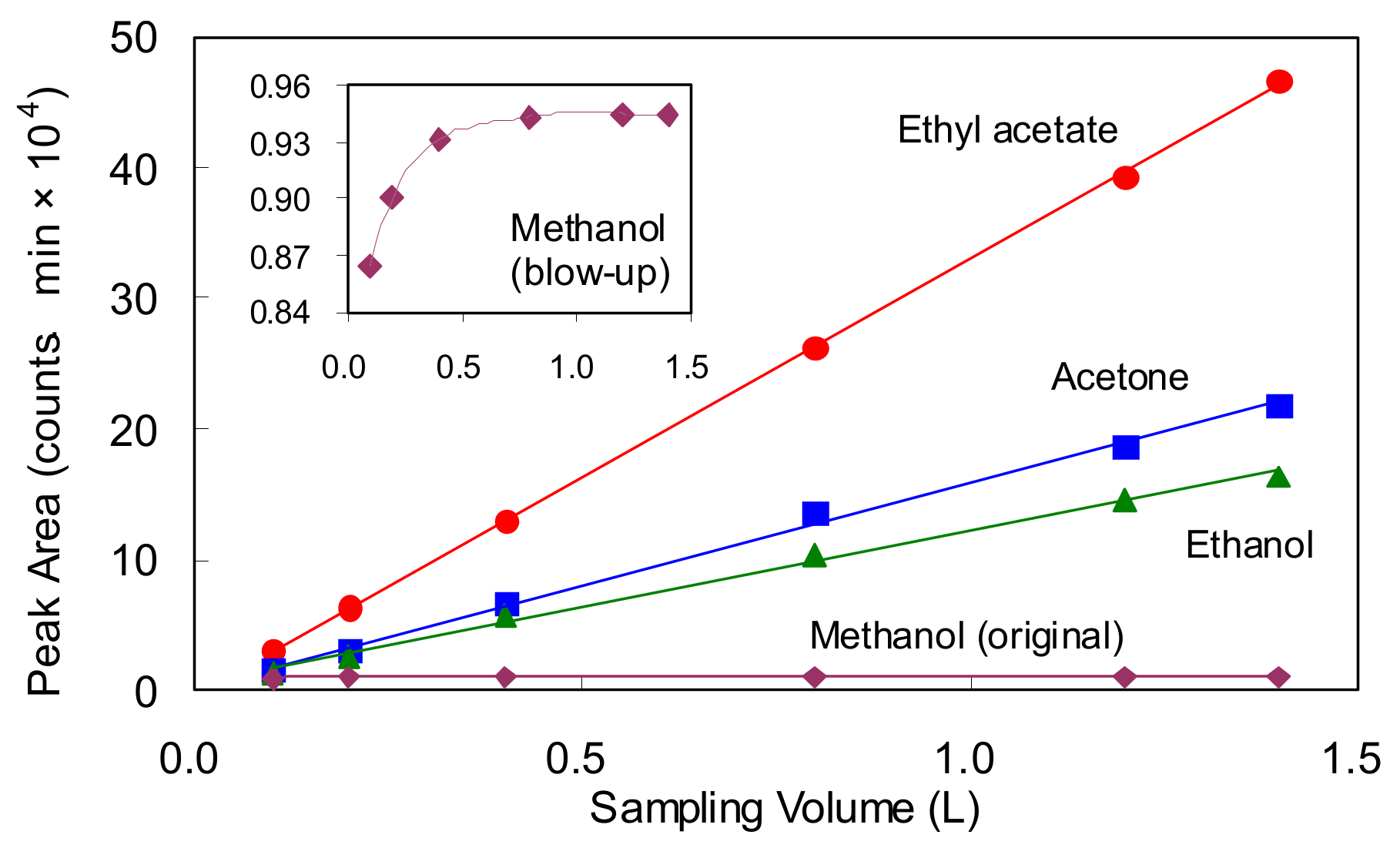
3.3. Capacity Limits for High Volatiles
3.4. Concentration Tests with Constant Sample Volume
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valcarcel, M.; Cardenas, S.; Simonet, B.M.; Moliner-Martinez, Y.; Lucena, R. Carbon nanostructures as sorbent materials in analytical processes. Trends Anal. Chem. 2008, 27, 34–43. [Google Scholar] [CrossRef]
- Duran, C.; Ozdes, D.; Gundogdu, A.; Imamoglu, M.; Senturk, H.B. Tea-industry waste activated carbon, as a novel adsorbent, for separation, preconcentration and speciation of chromium. Anal. Chim. Acta 2011, 688, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Gallego, E.; Roca, F.J.; Perales, J.F.; Guardino, X. Comparative study of the adsorption performance of a multi-sorbent bed (Carbotrap, Carbopack X, Carboxen 569) and a Tenax TA adsorbent tube for the analysis of volatile organic compounds (VOCs). Talanta 2010, 81, 916–924. [Google Scholar] [CrossRef]
- Liaud, C.; Nguyen, N.T.; Nasreddine, R.; Le Calvé, S. Experimental performances study of a transportable GC-PID and two thermo-desorption based methods coupled to FID and MS detection to assess BTEX exposure at sub-ppb level in air. Talanta 2014, 127, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Ras-Mallorquí, M.R.; Marcé-Recasens, R.M.; Borrull-Ballarín, F. Determination of volatile organic compounds in urban and industrial air from Tarragona by thermal desorption and gas chromatography–mass spectrometry. Talanta 2007, 72, 941–950. [Google Scholar] [CrossRef]
- Lama, L.; Ferronato, C.; Fine, L.; Jaber, F.; Chovelon, J.M. Evaluation of adsorbents for volatile methyl siloxanes sampling based on the determination of their breakthrough volume. Talanta 2013, 115, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Garbacz, M.; Malec, A.; Duda-Saternus, S.; Suchorab, Z.; Guz, L.; Łagód, G. Methods for Early Detection of Microbiological Infestation of Buildings Based on Gas Sensor Technologies. Chemosensors 2020, 8, 7. [Google Scholar] [CrossRef]
- Serrano, G.; Sukaew, T.; Zellers, E.T. Hybrid preconcentrator/focuser module for determinations of explosive marker compounds with a micro-scale gas chromatograph. J. Chromatogr. A 2013, 1279, 76–85. [Google Scholar] [CrossRef]
- Lu, C.-J.; Zellers, E.T. A dual-adsorbent preconcentrator for a portable indoor-VOC microsensor system. Anal. Chem. 2001, 73, 3449–3457. [Google Scholar] [CrossRef]
- Lu, C.-J.; Zellers, E.T. Multi-adsorbent preconcentration/focusing module for portable-GC/microsensor-array analysis of complex vapor mixtures. Analyst 2002, 127, 1061–1068. [Google Scholar]
- Terry, S.C.; Jerman, J.H.; Angell, J.B. A gas chromatographic air analyzer fabricated on a silicon wafer. IEEE Trans. Electron Dev. 1979, 26, 1880–1886. [Google Scholar] [CrossRef]
- Tian, W.-C.; Pang, S.W.; Lu, C.-J.; Zellers, E.T. Microfabricated preconcentrator-focuser for a microscale gas chromatograph. J. Microelectromech. Syst. 2003, 12, 264–272. [Google Scholar] [CrossRef]
- Tian, W.-C.; Chan, H.K.L.; Pang, S.W.; Lu, C.-J.; Zellers, E.T. Multiple-stage microfabricated preconcentrator-focuser for micro gas chromatography system. J. Microelectromech. Syst. 2005, 14, 498–507. [Google Scholar] [CrossRef]
- Lu, C.-J.; Steinecker, W.H.; Tian, W.-C.; Oborny, M.C.; Nichols, J.M.; Agah, M.; Potkay, J.A.; Chan, H.K.L.; Driscoll, J.; Sacks, R.D.; et al. First-generation hybrid MEMS gas chromatograph. Lab Chip 2005, 5, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.-X.; Hung, T.-Y.; Jian, R.-S.; Lu, C.-J. A multidimensional micro gas chromatograph employing a parallel separation multi-column chip and stop-flow μGC×μGCs configuration. Lab Chip 2013, 13, 1333–1341. [Google Scholar] [CrossRef]
- Voiculescu, I.; Zaghloul, M.; Narasimhan, N. Microfabricated chemical preconcentrators for gas-phase microanalytical detection systems. Trends Anal. Chem. 2008, 27, 327–345. [Google Scholar] [CrossRef]
- Ruiz, A.M.; Gracia, I.; Sabate, N.; Ivanov, P.; Sanchez, A.; Duch, M.; Gerboles, M.; Moreno, A.; Cane, C. Membrane-suspended microgrid as a gas preconcentrator for chromatographic applications. Sens. Actuat. A 2007, 135, 192–196. [Google Scholar] [CrossRef]
- Tian, W.-C.; Wu, T.H.; Lu, C.-J.; Chen, W.R.; Sheen, H.J. A novel and simple- fabricated micropreconcentrator for micro gas chromatography. J. Micromech. Microeng. 2012, 22, 065014. [Google Scholar] [CrossRef]
- McCartney, M.M.; Zrodnikov, Y.; Fung, A.G.; LeVasseur, M.K.; Pedersen, J.M.; Zamuruyev, K.O.; Aksenov, A.A.; Kenyon, N.J.; Davis, C.E. An Easy to Manufacture Micro Gas Preconcentrator for Chemical Sensing Applications. ACS Sens. 2017, 2, 1167–1174. [Google Scholar] [CrossRef]
- Mohsen, Y.; Lahlou, H.; Sanchez, J.-B.; Berger, F.; Bezverkhyy, I.; Weber, G.; Bellat, J.-P. Development of a micro-analytical prototype for selective trace detection of orthonitrotoluene. Microchem. J. 2014, 114, 48–52. [Google Scholar] [CrossRef]
- James, F.; Breuil, P.; Pijolat, C.; Camara, M.; Briand, D.; Bart, A.; Cozic, R. Development of a MEMS preconcentrator for micro-gas chromatography analyses. Procedia Eng. 2014, 87, 500–503. [Google Scholar] [CrossRef]
- Lahlou, H.; Vilanova, X.; Correig, X. Gas phase micro-preconcentrators for benzene monitoring: A review. Sens. Actuat. B 2013, 176, 198–210. [Google Scholar] [CrossRef]
- Janssen, S.; Tessmann, T.; Lang, W. High sensitive and selective ethylene measurement by using a large-capacity-on-chip preconcentrator device. Sens. Actuat. B 2014, 197, 405–413. [Google Scholar] [CrossRef]
- Ivanov, P.; Blanco, F.; Gracia, I.; Sabate, N.; Ruiz, A.M.; Vilanova, X.; Correig, X.; Fonseca, L.; Figueras, E.; Santander, J.; et al. Improvement of the gas sensor response via silicon μ-preconcentrator. Sens. Actuat. B 2007, 127, 288–294. [Google Scholar] [CrossRef]
- Dow, A.B.A.; Sklorz, A.; Lang, W. A microfluidic preconcentrator for enhanced monitoring of ethylene gas. Sens. Actuat. A 2011, 167, 226–230. [Google Scholar] [CrossRef]
- Camara, M.; Breuil, P.; Briand, D.; de Rooij, N.F.; Pijolat, C. A micro gas preconcentrator with improved performance for pollution monitoring and explosives detection. Anal. Chim. Acta 2011, 688, 175–182. [Google Scholar] [CrossRef]
- Slimani, S.; Bultel, E.; Cubizolle, T.; Herrier, C.; Rousselle, T.; Livache, T. Opto-Electronic Nose Coupled to a Silicon Micro Pre-Concentrator Device for Selective Sensing of Flavored Waters. Chemosensors 2020, 8, 60. [Google Scholar] [CrossRef]
- Wong, M.-Y.; Cheng, W.-R.; Liu, M.-H.; Tian, W.-C.; Lu, C.-J. A preconcentrator chip employing μ-SPME array coated with in-situ-synthesized carbon adsorbent film for VOCs analysis. Talanta 2012, 101, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Vasilescu, A.; Fanjul-Bolado, P.; Titoiu, A.-M.; Porumb, R.; Epure, P. Progress in Electrochemical (Bio)Sensors for Monitoring Wine Production. Chemosensors 2019, 7, 66. [Google Scholar] [CrossRef]
- Jian, R.-S.; Wang, T.-Y.; Song, L.-Y.; Kuo, C.-Y.; Tian, W.-C.; Lo, E.-W.; Lu, C.-J. Field investigations and dynamic measurements of process activity induced VOCs inside a semiconductor cleanroom. Build. Environ. 2015, 94, 287–295. [Google Scholar] [CrossRef]
- Centeno, T.A.; Fuertes, A.B. Carbon molecular sieve gas separation membranes based on poly(vinylidene chloride-co-vinyl chloride). Carbon 2000, 38, 1067–1073. [Google Scholar] [CrossRef]
- Sachan, A.; Castro, M.; Choudhary, V.; Feller, J.-F. Influence of Water Molecules on the Detection of Volatile Organic Compounds (VOC) Cancer Biomarkers by Nanocomposite Quantum Resistive Vapor Sensors vQRS. Chemosensors 2018, 6, 64. [Google Scholar] [CrossRef]
- Huang, R.-X.; Lu, C.-J.; Tian, W.C. The shell structure effect on the vapor selectivity of monolayer-protected gold nanoparticle sensors. Chemosensors 2014, 2, 85–96. [Google Scholar] [CrossRef]
- Zhang, M.; Shi, J.; Liao, C.; Tian, Q.; Wang, C.; Chen, S.; Zang, L. Perylene Imide-Based Optical Chemosensors for Vapor Detection. Chemosensors 2021, 9, 1. [Google Scholar] [CrossRef]
- Wang, C.-C.; Lin, P.-Y.; Lu, C.-J.; Liu, M.-H. Rapid determination of volatile organics using a nanoporous zinc oxide microsphere-coated quartz crystal microbalance. Instrument. Sci. Tech. 2017, 45, 639–649. [Google Scholar] [CrossRef]
- Rianjanu, A.; Hasanah, S.A.; Nugroho, D.B.; Kusumaatmaja, A.; Roto, R.; Triyana, K. Polyvinyl Acetate Film-Based Quartz Crystal Microbalance for the Detection of Benzene, Toluene, and Xylene Vapors in Air. Chemosensors 2019, 7, 20. [Google Scholar] [CrossRef]
- Rodriguez-Cuevas, A.; Lara-Ibeas, I.; Leprince, A.; Wolf, M.; Le Calve, S. Easy-to-manufacture micro gas preconcentrator integrated in a portable GC for enhanced trace detection of BTEX. Sens. Actuat. B Chem. 2020, 324, 128690. [Google Scholar] [CrossRef]
- Lara-Ibeas, I.; Rodriguez-Cuevas, A.; Andrikopoulou, C.; Person, V.; Baldas, L.; Colin, S.; Le Calve, S. Sub-ppb Level Detection of BTEX Gaseous Mixtures with a Compact Prototype GC Equipped with a Preconcentration Unit. Micromachines 2019, 10, 187. [Google Scholar] [CrossRef] [PubMed]

| Target VOCs | Device Material | Adsorbent Material a/mass | Outer Dimensions l × w × h (mm3) | Sample Vol. (mL) | Desorp. Temp. (°C) | Ref. |
|---|---|---|---|---|---|---|
| Ethanol, <C7 | Si-Gl | CMSM/1.4 mg | 14 × 4 × 1.6 | 1400 | 320 | This work |
| BTEX | Al | C-B/5.0 mg | 14 × 12 × 3.2 | 20 | 330 | [37] |
| BTEX | Al | Basolite/5.8 mg | 40 × 40 × 12 | 80 | 150 | [38] |
| Toluene, ≥C6 | Si-Gl | Am-CF/2.0 mg | 14 × 4 × 1.6 | 1000 | 320 | [28] |
| Flavors, ≥C9 | Si-Gl | Tenax/7.0 mg | 21 × 8 × ≤ 1.6 b | 80 | 200 | [27] |
| Benzene | Si-Gl | SWCNTs/1.0 mg | 30 × 15 × ≤ 1.6 b | 835 | 280 | [26] |
| Ethylene | Gl-Si-Gl | C-1000/6.6 mg | 30 × 15 × ≤ 2.0 b | 200 | 300 | [23,25] |
| Benzene | Si-Gl | C-B/0.39 mg | 3 × 3 × ≤ 1.6 b | 6000 | 250 | [24] |
| Toluene, CHCl3 | Si-Gl | C-NP/-- | 85 × 20 × ≤ 1.6 b | 833 | 230 | [21] |
| Orthonitrotoluene | Si-Gl | Zeolite/2.25 mg | 10 × 5 × 0.4 (cavity) | 500 | 230 | [20] |
| d-Limonene, >C6 | Gl-Gl | Tenax/-- | 25.4 × 25.4 × 1.4 | 180 | 260 | [19] |
| Benzene, xylene | Si-Gl | Am-CF/0.3 mg | 13 × 8 × ≤ 1.6 b | 35 | 300 | [18] |
| IPA → d-Limonene | Si-Gl | 3-stage/3.2 mg | 12 × 4 × ≤ 1.6 b | 250 | 250 | [13] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, H.-Y.; Cheng, W.-R.; Wu, T.-H.; Sheen, H.-J.; Wang, C.-C.; Lu, C.-J. A MEMS µ-Preconcentrator Employing a Carbon Molecular Sieve Membrane for Highly Volatile Organic Compound Sampling. Chemosensors 2021, 9, 104. https://doi.org/10.3390/chemosensors9050104
Kuo H-Y, Cheng W-R, Wu T-H, Sheen H-J, Wang C-C, Lu C-J. A MEMS µ-Preconcentrator Employing a Carbon Molecular Sieve Membrane for Highly Volatile Organic Compound Sampling. Chemosensors. 2021; 9(5):104. https://doi.org/10.3390/chemosensors9050104
Chicago/Turabian StyleKuo, Hung-Yen, Wei-Riu Cheng, Tzu-Heng Wu, Horn-Jiunn Sheen, Chih-Chia Wang, and Chia-Jung Lu. 2021. "A MEMS µ-Preconcentrator Employing a Carbon Molecular Sieve Membrane for Highly Volatile Organic Compound Sampling" Chemosensors 9, no. 5: 104. https://doi.org/10.3390/chemosensors9050104
APA StyleKuo, H.-Y., Cheng, W.-R., Wu, T.-H., Sheen, H.-J., Wang, C.-C., & Lu, C.-J. (2021). A MEMS µ-Preconcentrator Employing a Carbon Molecular Sieve Membrane for Highly Volatile Organic Compound Sampling. Chemosensors, 9(5), 104. https://doi.org/10.3390/chemosensors9050104






