Electrochemical Study of Enzymatic Glucose Sensors Biocatalyst: Thermal Degradation after Long-Term Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
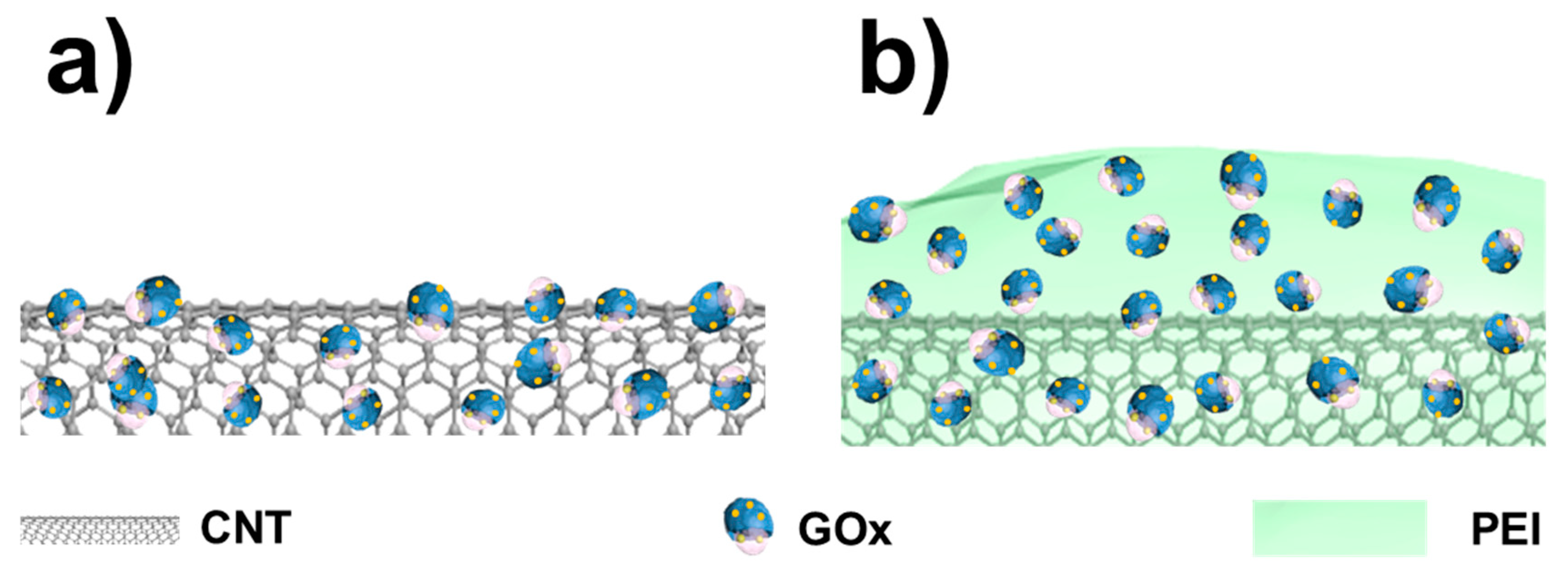
2.2. Biocatalyst Fabrication
2.3. Electrochemical Measurement
3. Results and Discussion
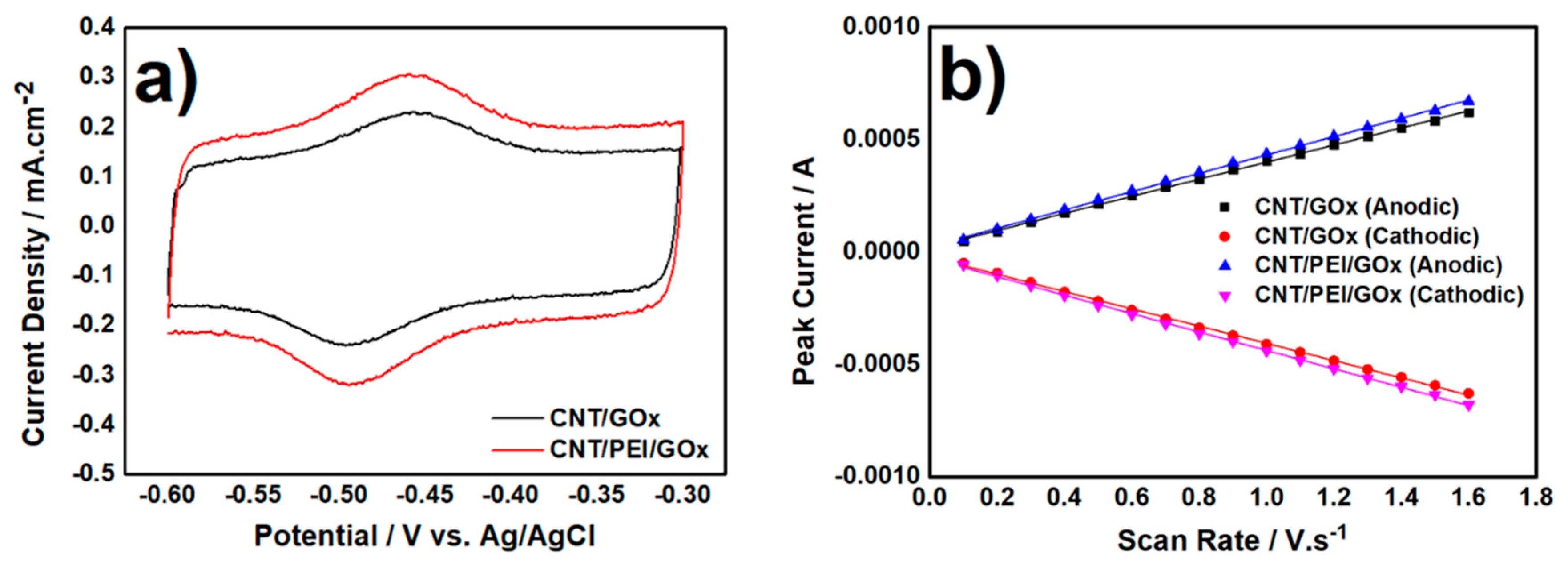
3.1. Electrochemical Characterization of Biocatalyst at Day-0
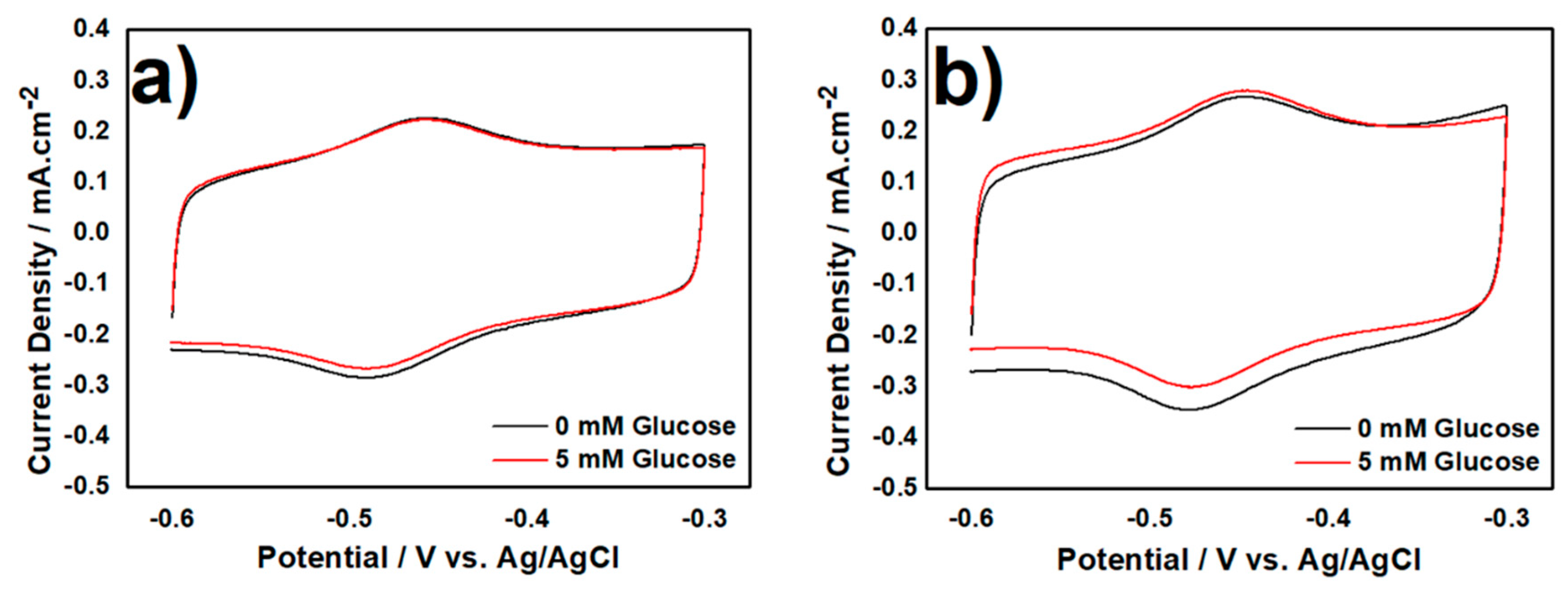
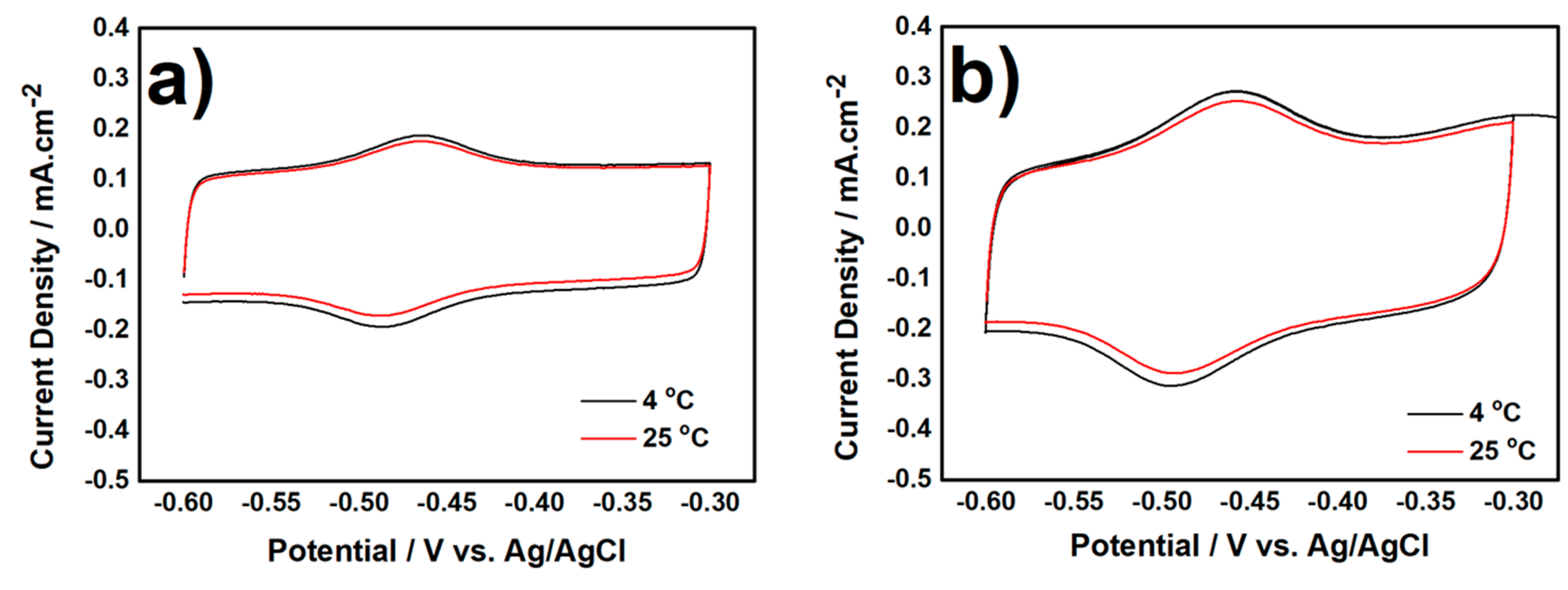
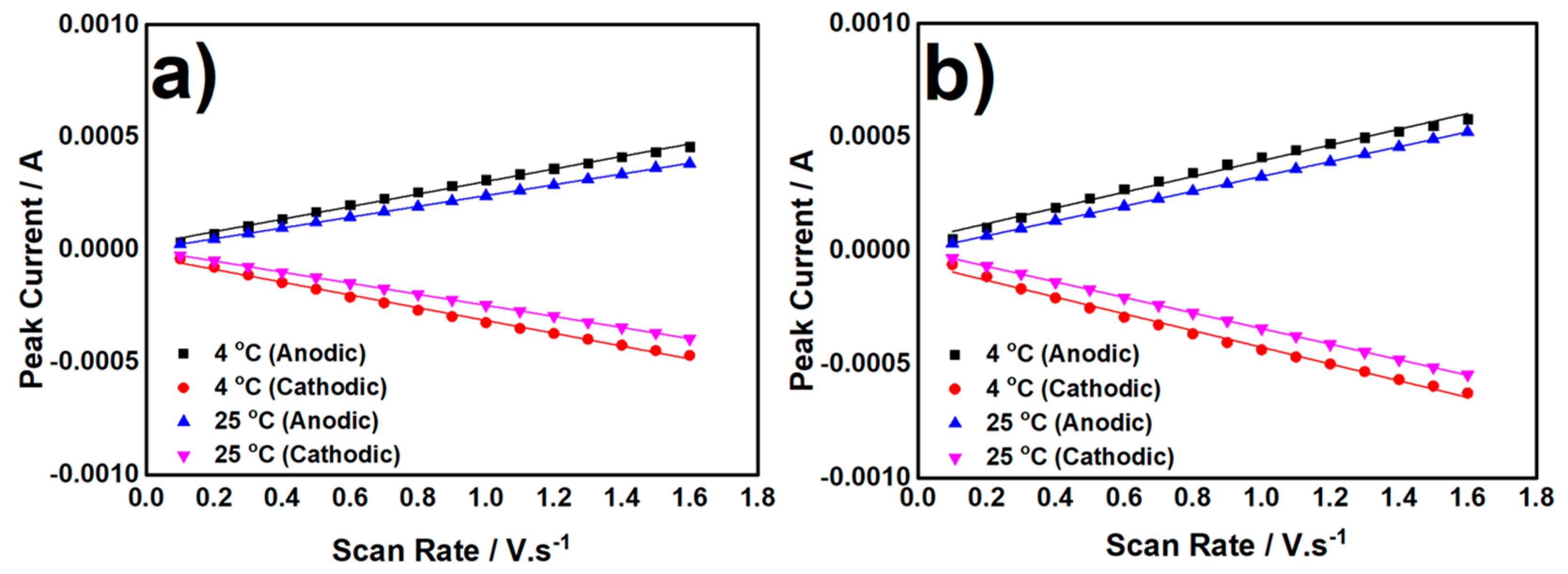
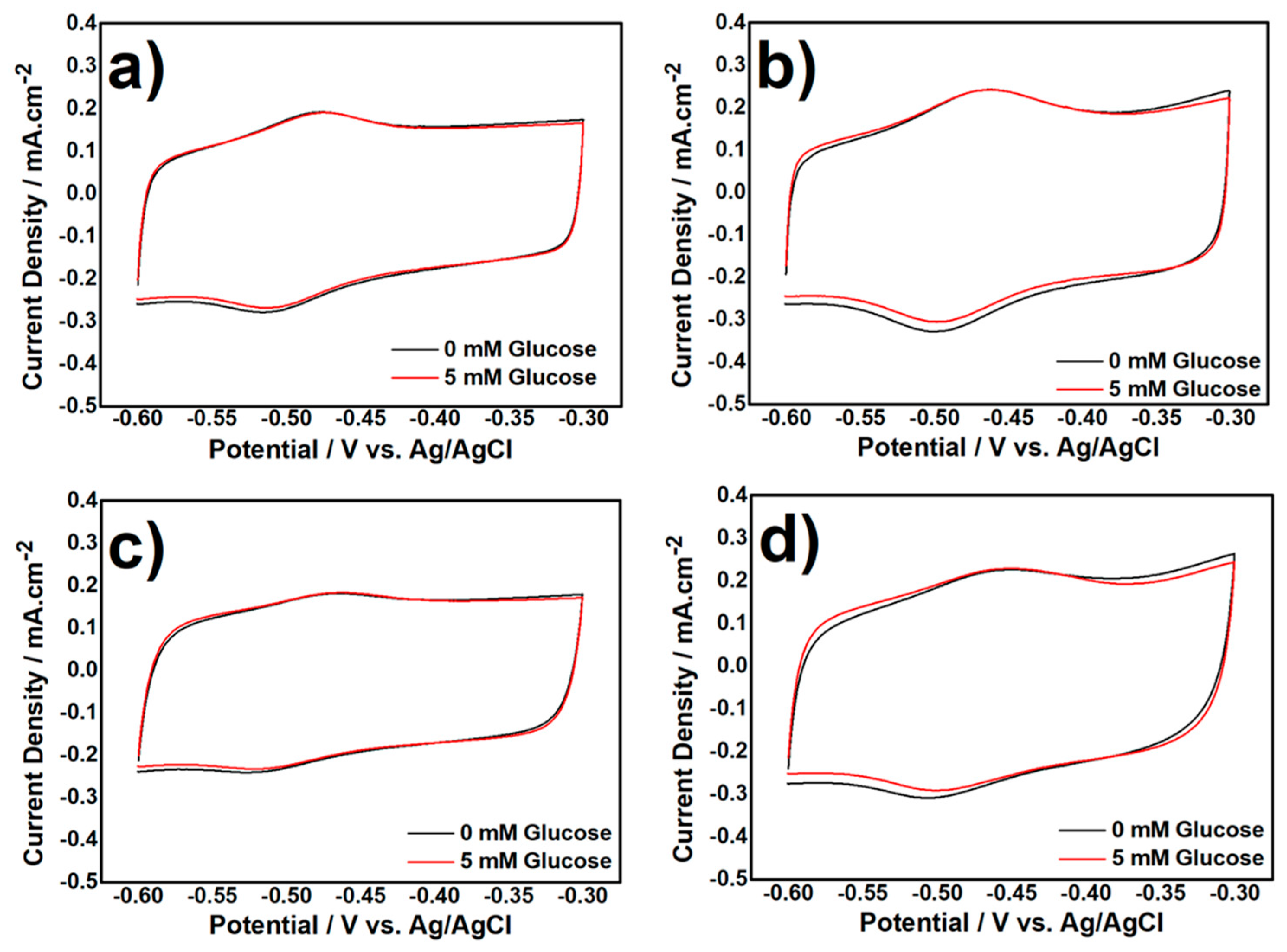
3.2. Electrochemical Characterization of Biocatalyst at Day-120
3.2.1. CV measurement in Anaerobic Condition
3.2.2. CV Measurement in Aerobic Condition
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, N.; Chen, X.; Ren, T.; Zhang, P.; Yang, D. Carbon nanotube based biosensors. Sens. Actuators B Chem. 2015, 207, 690–715. [Google Scholar] [CrossRef]
- Yu, M.-F.; Files, B.S.; Arepalli, S.; Ruoff, R.S. Tensile Loading of Ropes of Single Wall Carbon Nanotubes and their Mechanical Properties. Phys. Rev. Lett. 2000, 84, 5552–5555. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Li, W.; Pan, Z.; Chang, B.; Sun, L. Mechanical and physical properties on carbon nanotube. J. Phys. Chem. Solids 2000, 61, 1153–1158. [Google Scholar] [CrossRef]
- Elliott, J.A.; Sandler, J.K.W.; Windle, A.H.; Young, R.J.; Shaffer, M.S.P. Collapse of Single-Wall Carbon Nanotubes is Diameter Dependent. Phys. Rev. Lett. 2004, 92, 095501. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.J.; Green, M.L.H.; Allen, O.; Hill, H.; Leung, Y.C.; Sadler, P.J.; Sloan, J.; Xavier, A.V.; Chi Tsang, S. The immobilisation of proteins in carbon nanotubes. Inorg. Chim. Acta 1998, 272, 261–266. [Google Scholar] [CrossRef]
- Guo, Z.; Sadler, P.J.; Tsang, S.C. Immobilization and Visualization of DNA and Proteins on Carbon Nanotubes. Adv. Mater. 1998, 10, 701–703. [Google Scholar] [CrossRef]
- Kang, X.; Mai, Z.; Zou, X.; Cai, P.; Mo, J. A novel glucose biosensor based on immobilization of glucose oxidase in chitosan on a glassy carbon electrode modified with gold–platinum alloy nanoparticles/multiwall carbon nanotubes. Anal. Biochem. 2007, 369, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Wei, J.; Lin, J.; Li, Q.; KuaYou, J. A glucose biosensor based on electrodeposition of palladium nanoparticles and glucose oxidase onto Nafion-solubilized carbon nanotube electrode. Biosens. Bioelectron. 2005, 20, 2341–2346. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Xu, J.-J.; Chen, H.-Y. A novel glucose biosensor based on the nanoscaled cobalt phthalocyanine–glucose oxidase biocomposite. Biosens. Bioelectron. 2005, 20, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Galaev, I.Y.; Mattiasson, B. Polymer displacement/shielding in protein chromatography. J. Chromatogr. B Biomed. Sci. Appl. 2000, 741, 103–113. [Google Scholar] [CrossRef]
- Bahulekar, R.; Ayyangar, N.R.; Ponrathnam, S. Polyethyleneimine in immobilization of biocatalysts. Enzym. Microb. Technol. 1991, 13, 858–868. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernandez-Lafuente, R. Amination of enzymes to improve biocatalyst performance: Coupling genetic modification and physicochemical tools. RSC Adv. 2014, 4, 38350–38374. [Google Scholar] [CrossRef]
- Virgen-Ortíz, J.J.; Peirce, S.; Tacias-Pascacio, V.G.; Cortes-Corberan, V.; Marzocchella, A.; Russo, M.E.; Fernandez-Lafuente, R. Reuse of anion exchangers as supports for enzyme immobilization: Reinforcement of the enzyme-support multiinteraction after enzyme inactivation. Process Biochem. 2016, 51, 1391–1396. [Google Scholar] [CrossRef]
- Virgen-Ortíz, J.; Pedrero, S.; Fernandez-Lopez, L.; Lopez-Carrobles, N.; Gorines, B.; Otero, C.; Fernandez-Lafuente, R. Desorption of Lipases Immobilized on Octyl-Agarose Beads and Coated with Ionic Polymers after Thermal Inactivation. Stronger Adsorption of Polymers/Unfolded Protein Composites. Molecules 2017, 22, 91. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.; Ahn, Y.; Christwardana, M.; Kim, H.; Kwon, Y. Development of a glucose oxidase-based biocatalyst adopting both physical entrapment and crosslinking, and its use in biofuel cells. Nanoscale 2016, 8, 9201–9210. [Google Scholar] [CrossRef] [PubMed]
- Christwardana, M.; Chung, Y.; Kwon, Y. Co-immobilization of glucose oxidase and catalase for enhancing the performance of a membraneless glucose biofuel cell operated under physiological conditions. Nanoscale 2017, 9, 1993–2002. [Google Scholar] [CrossRef] [PubMed]
- Christwardana, M. Combination of physico-chemical entrapment and crosslinking of low activity laccase-based biocathode on carboxylated carbon nanotube for increasing biofuel cell performance. Enzym. Microb. Technol. 2017, 106, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Christwardana, M.; Chung, Y.; Kwon, Y. Performance Enhancement of Biofuel Cell by Surface Modification of Glucose Oxidase using Ferrocene Carboxylic acid. Trans. Korean Hydrog. New Energy Soc. 2016, 27, 526–532. [Google Scholar] [CrossRef] [Green Version]
- Wooten, M.; Karra, S.; Zhang, M.; Gorski, W. On the Direct Electron Transfer, Sensing, and Enzyme Activity in the Glucose Oxidase/Carbon Nanotubes System. Anal. Chem. 2013, 86, 752–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyun, K.H.; Han, S.W.; Koh, W.-G.; Kwon, Y. Fabrication of biofuel cell containing enzyme catalyst immobilized by layer-by-layer method. J. Power Sources 2015, 286, 197–203. [Google Scholar] [CrossRef]
- Christwardana, M.; Kim, D.-H.; Chung, Y.; Kwon, Y. A hybrid biocatalyst consisting of silver nanoparticle and naphthalenethiol self-assembled monolayer prepared for anchoring glucose oxidase and its use for an enzymatic biofuel cell. Appl. Surf. Sci. 2018, 429, 180–186. [Google Scholar] [CrossRef]
- Christwardana, M.; Chung, Y.; Kwon, Y. A new biocatalyst employing pyrenecarboxaldehyde as an anodic catalyst for enhancing the performance and stability of an enzymatic biofuel cell. NPG Asia Mater. 2017, 9, e386. [Google Scholar] [CrossRef] [Green Version]
- Christwardana, M.; Kwon, Y. Effects of multiple polyaniline layers immobilized on carbon nanotube and glutaraldehyde on performance and stability of biofuel cell. J. Power Sources 2015, 299, 604–610. [Google Scholar] [CrossRef]
- Sharp, M.; Petersson, M.; Edström, K. Preliminary determinations of electron transfer kinetics involving ferrocene covalently attached to a platinum surface. J. Electroanal. Chem. Interfacial Electrochem. 1979, 95, 123–130. [Google Scholar] [CrossRef]
- Bagdžiūnas, G.; Žukauskas, Š.; Ramanavičius, A. Insights into a hole transfer mechanism between glucose oxidase and a p-type organic semiconductor. Biosens. Bioelectron. 2018, 102, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Fotouhi, L.; Hashkavayi, A.B.; Heravi, M.M. Electrochemical behaviour and voltammetric determination of sulphadiazine using a multi-walled carbon nanotube composite film-glassy carbon electrode. J. Exp. Nanosci. 2013, 8, 947–956. [Google Scholar] [CrossRef]
- Terse-Thakoor, T.; Komori, K.; Ramnani, P.; Lee, I.; Mulchandani, A. Electrochemically Functionalized Seamless Three-Dimensional Graphene-Carbon Nanotube Hybrid for Direct Electron Transfer of Glucose Oxidase and Bioelectrocatalysis. Langmuir 2015, 31, 13054–13061. [Google Scholar] [CrossRef] [PubMed]
- Jin-Zhong, X.; Jun-Jie, Z.; Qiang, W.; Zheng, H.; Hong-Yuan, C. Direct Electron Transfer between Glucose Oxidase and Multi-walled Carbon Nanotubes. Chin. J. Chem. 2010, 21, 1088–1091. [Google Scholar] [CrossRef]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. Interfacial Electrochem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Christwardana, M.; Chung, Y.; Kwon, Y. A correlation of results measured by cyclic voltammogram and impedance spectroscopy in glucose oxidase based biocatalysts. Korean J. Chem. Eng. 2017, 34, 3009–3016. [Google Scholar] [CrossRef]
- Chung, Y.; Christwardana, M.; Tannia, D.C.; Kim, K.J.; Kwon, Y. Biocatalyst including porous enzyme cluster composite immobilized by two-step crosslinking and its utilization as enzymatic biofuel cell. J. Power Sources 2017, 360, 172–179. [Google Scholar] [CrossRef]
- Christwardana, M.; Ji, J.; Chung, Y.; Kwon, Y. Highly sensitive glucose biosensor using new glucose oxidase based biocatalyst. Korean J. Chem. Eng. 2017, 34, 2916–2921. [Google Scholar] [CrossRef]
- Appleton, B.; Gibson, T.; Woodward, J.R. High temperature stabilisation of immobilised glucose oxidase: Potential applications in biosensors. Sens. Actuators B Chem. 1997, 43, 65–69. [Google Scholar] [CrossRef]
- Baghayeri, M.; Veisi, H.; Ghanei-Motlagh, M. Amperometric glucose biosensor based on immobilization of glucose oxidase on a magnetic glassy carbon electrode modified with a novel magnetic nanocomposite. Sens. Actuators B Chem. 2017, 249, 321–330. [Google Scholar] [CrossRef]
- Salimi, A.; Sharifi, E.; Noorbakhsh, A.; Soltanian, S. Immobilization of glucose oxidase on electrodeposited nickel oxide nanoparticles: Direct electron transfer and electrocatalytic activity. Biosens. Bioelectron. 2007, 22, 3146–3153. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.-Y.; Hou, S.-H.; Yin, F.; Zhao, Z.-X.; Wang, Y.-Y.; Wang, X.-S.; Chen, Q. Amperometric glucose biosensor based on multilayer films via layer-by-layer self-assembly of multi-wall carbon nanotubes, gold nanoparticles and glucose oxidase on the Pt electrode. Biosens. Bioelectron. 2007, 22, 2854–2860. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, X.; Zhang, X.; Qin, X.; Zhao, Z.; Miao, Z.; Huang, N.; Chen, Q. A novel glucose biosensor based on the immobilization of glucose oxidase onto gold nanoparticles-modified Pb nanowires. Biosens. Bioelectron. 2009, 25, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.-H.; Xu, Q.; Zhou, M.; Zhu, J.-J. In situ immobilization of glucose oxidase in chitosan–gold nanoparticle hybrid film on Prussian Blue modified electrode for high-sensitivity glucose detection. Electrochem. Commun. 2006, 8, 1468–1474. [Google Scholar] [CrossRef]
- Liu, G.; Lin, Y. Amperometric glucose biosensor based on self-assembling glucose oxidase on carbon nanotubes. Electrochem. Commun. 2006, 8, 251–256. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; Imani, S.; Nuñez-Flores, R.; Kumar, R.; Wang, C.; Mohan, A.M.V.; Wang, J.; Mercier, P.P. Re-usable electrochemical glucose sensors integrated into a smartphone platform. Biosens. Bioelectron. 2018, 101, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Bandodkar, A.J.; Jia, W.; Ramírez, J.; Wang, J. Biocompatible Enzymatic Roller Pens for Direct Writing of Biocatalytic Materials: “Do-it-Yourself” Electrochemical Biosensors. Adv. Healthc. Mater. 2015, 4, 1215–1224. [Google Scholar] [CrossRef] [PubMed]
- Frattini, D.; Accardo, G.; Ferone, C.; Cioffi, R. Fabrication and characterization of graphite-cement composites for microbial fuel cells applications. Mater. Res. Bull. 2017, 88, 188–199. [Google Scholar] [CrossRef]
- Christwardana, M.; Frattini, D.; Accardo, G.; Yoon, S.P.; Kwon, Y. Early-stage performance evaluation of flowing microbial fuel cells using chemically treated carbon felt and yeast biocatalyst. Appl. Energy 2018, 222, 369–382. [Google Scholar] [CrossRef]
| Catalyst Structure | ||||
|---|---|---|---|---|
| CNT/GOx | 5.23 | 5.30 | 5.25 | 8.5 ± 0.2 |
| CNT/PEI/GOx | 5.61 | 5.64 | 5.62 | 10.1 ± 0.4 |
| Catalyst Structure | Storage Condition (°C) | ||||
|---|---|---|---|---|---|
| CNT/GOx | 4 | 3.84 | 3.91 | 3.87 | 5.1 ± 0.2 |
| 25 | 2.69 | 2.76 | 2.73 | 2.8 ± 0.4 | |
| CNT/PEI/GOx | 4 | 4.76 | 5.10 | 4.91 | 7.5 ± 0.5 |
| 25 | 3.66 | 3.84 | 3.75 | 6.6 ± 0.3 |
| Catalyst Structure | Maintained Storage Stability 2 (%) | Storage Time (Days) | Reference |
|---|---|---|---|
| GOx/Ag@MWCNT-IL-Fe3O4/MGCE | 79.0 | 28 | [34] |
| GOx-NiO | 95.0 | 10 | [35] |
| GOD/GNp/MWCNTs/Pt | 92.0 | 30 | [36] |
| GNPs/Pb NWs | 80.7 | 70 | [37] |
| GOD-chitosan-AuNP-PB/GCE | 70.0 | 14 | [38] |
| PDDA/GOx/PDDA/CNT/GC | 90.0 | 28 | [39] |
| CNT/PEI/GOx | 66.7 | 120 | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christwardana, M.; Frattini, D. Electrochemical Study of Enzymatic Glucose Sensors Biocatalyst: Thermal Degradation after Long-Term Storage. Chemosensors 2018, 6, 53. https://doi.org/10.3390/chemosensors6040053
Christwardana M, Frattini D. Electrochemical Study of Enzymatic Glucose Sensors Biocatalyst: Thermal Degradation after Long-Term Storage. Chemosensors. 2018; 6(4):53. https://doi.org/10.3390/chemosensors6040053
Chicago/Turabian StyleChristwardana, Marcelinus, and Domenico Frattini. 2018. "Electrochemical Study of Enzymatic Glucose Sensors Biocatalyst: Thermal Degradation after Long-Term Storage" Chemosensors 6, no. 4: 53. https://doi.org/10.3390/chemosensors6040053
APA StyleChristwardana, M., & Frattini, D. (2018). Electrochemical Study of Enzymatic Glucose Sensors Biocatalyst: Thermal Degradation after Long-Term Storage. Chemosensors, 6(4), 53. https://doi.org/10.3390/chemosensors6040053






