Recent Trends in Field-Effect Transistors-Based Immunosensors
Abstract
:1. Introduction to Immunosensors
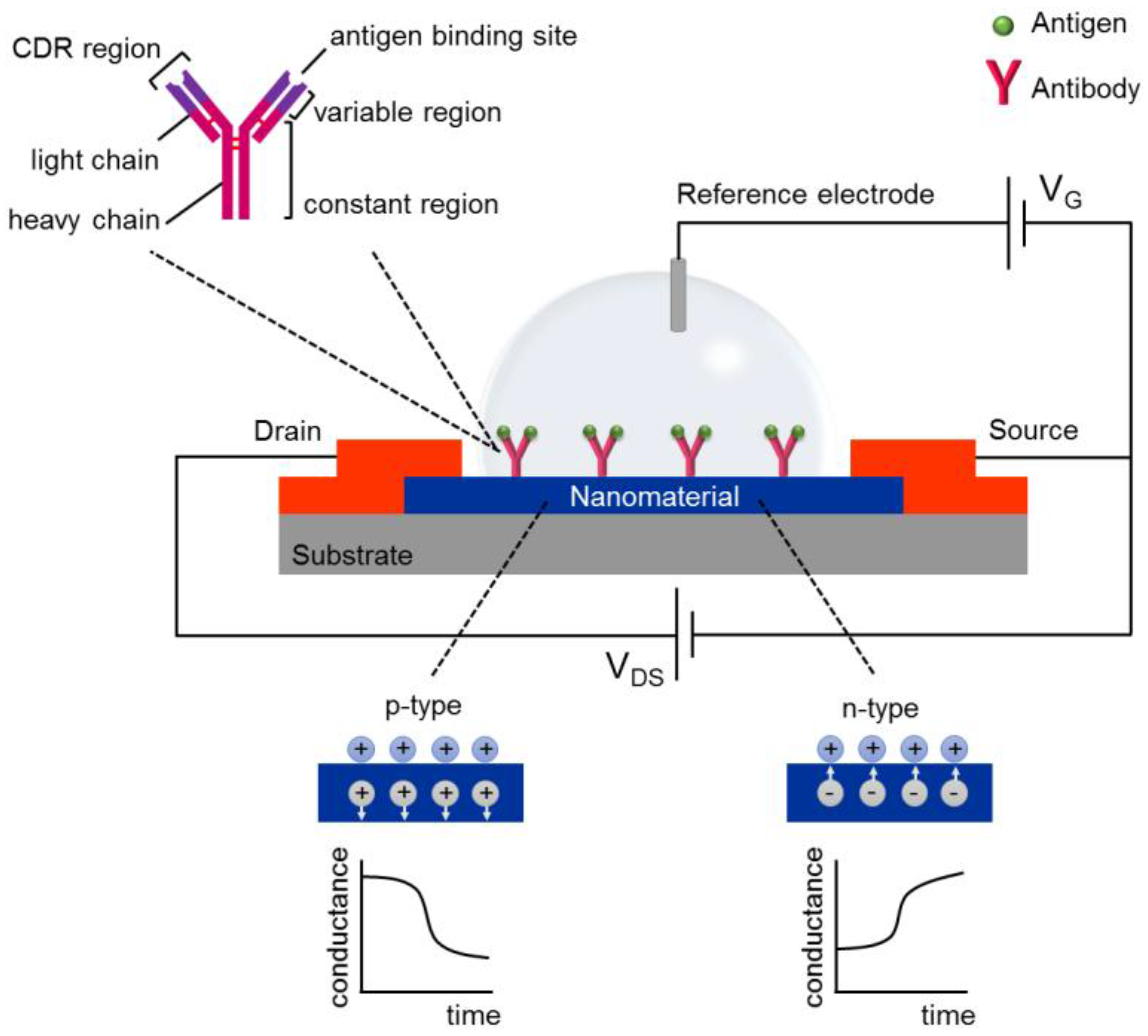
2. FET-Based Immunosensors
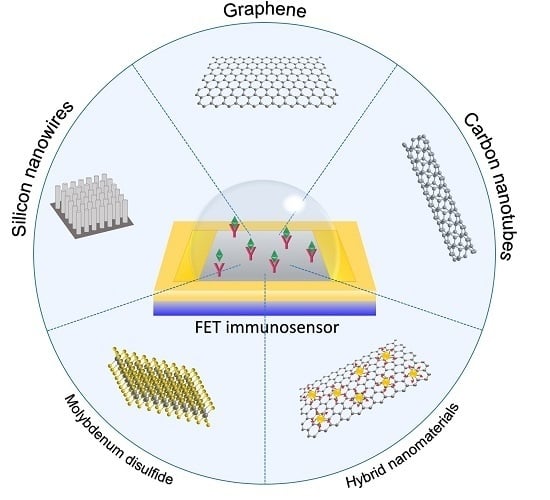
3. Trends in FET-Based Immunosensors Using Nanomaterials as Sensing Platforms
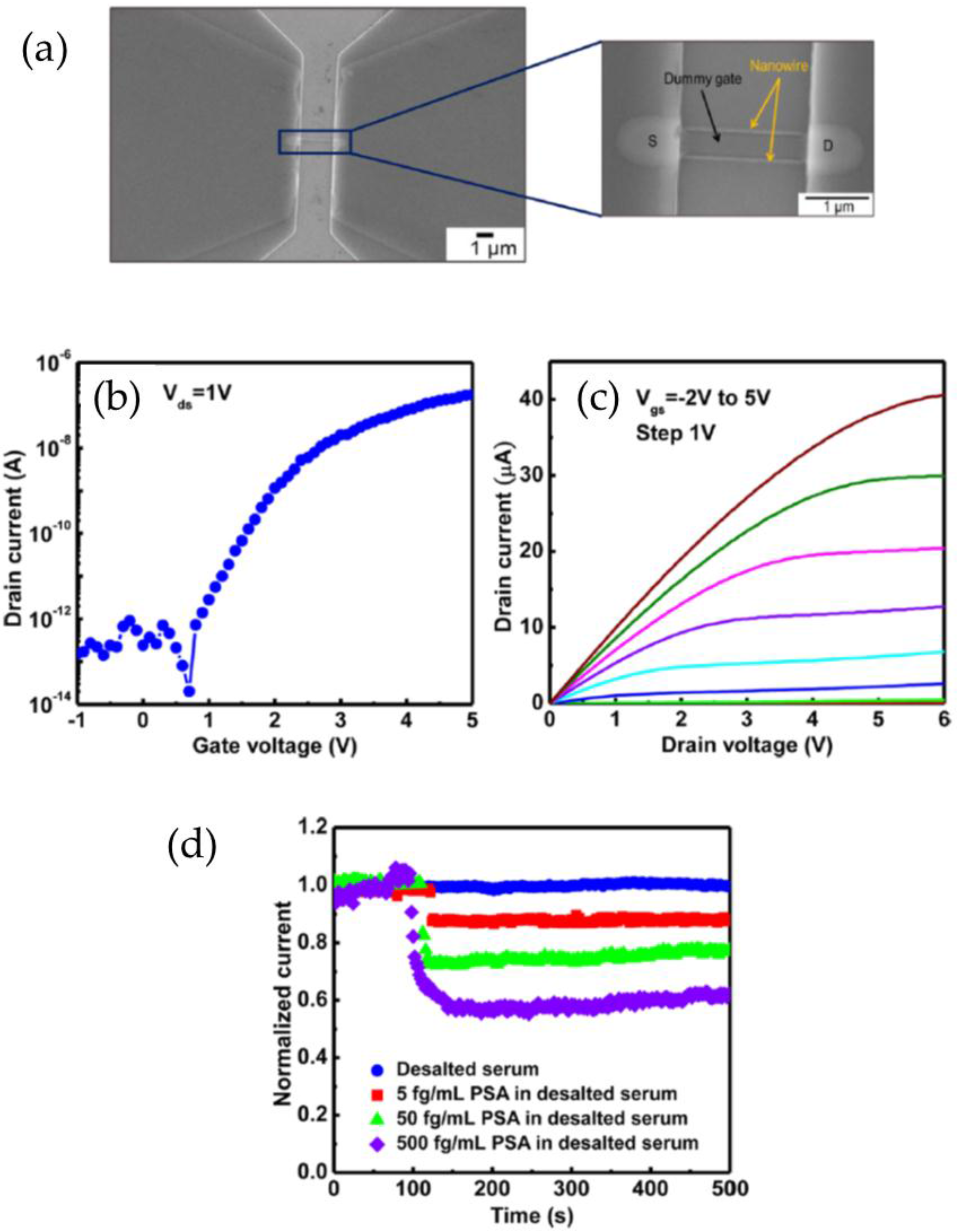
3.1. Silicon Nanowires
3.2. Carbon Nanotubes
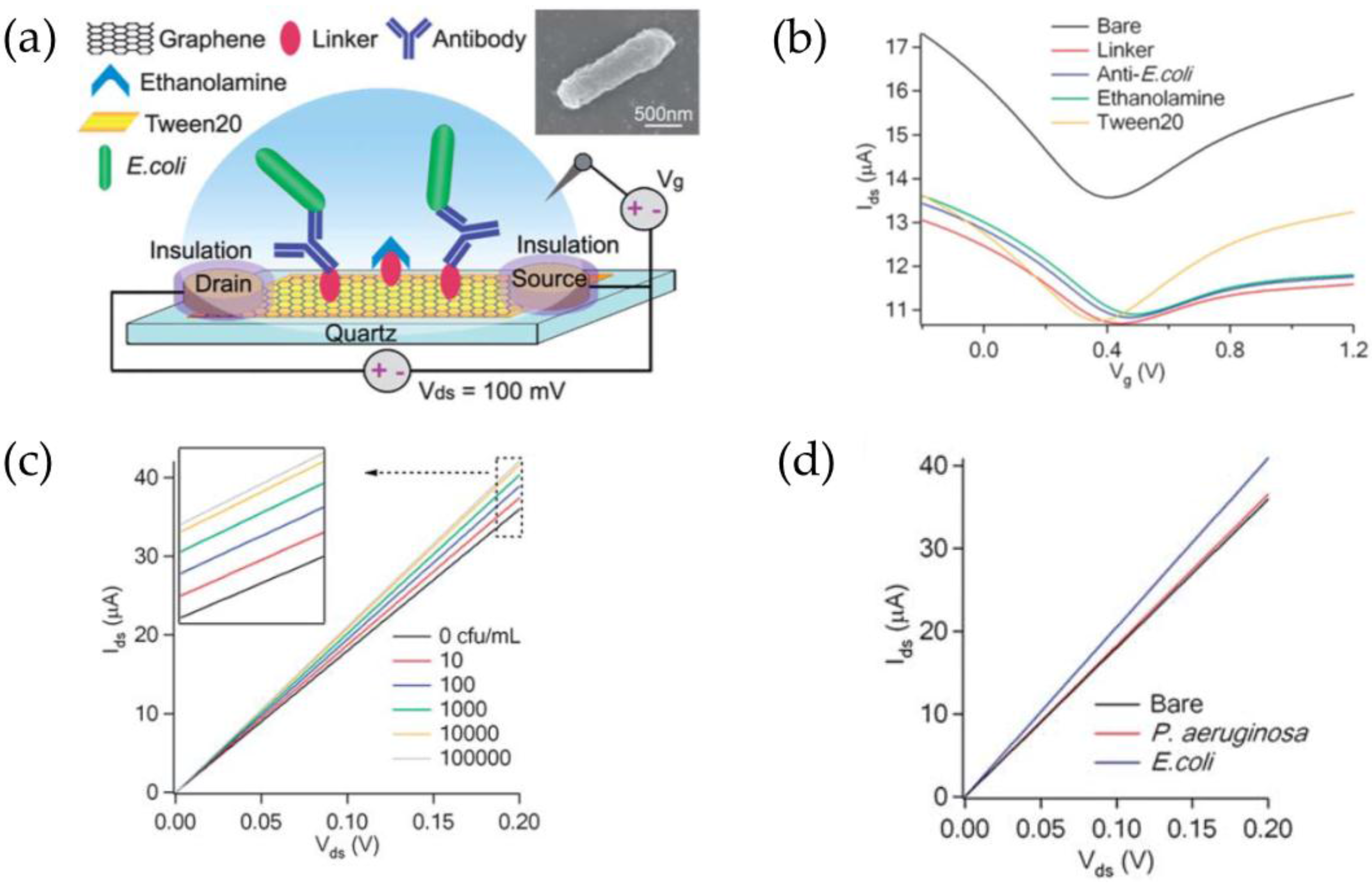
3.3. Graphene
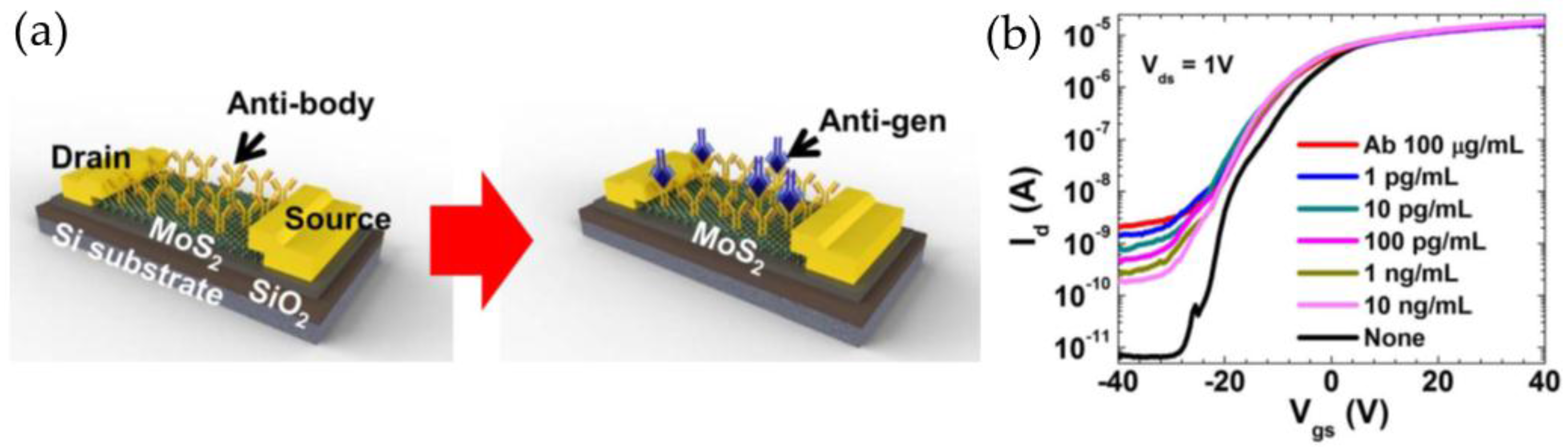
3.4. Molybdenum Disulfide
3.5. Titanium Dioxide
3.6. Zinc Oxide
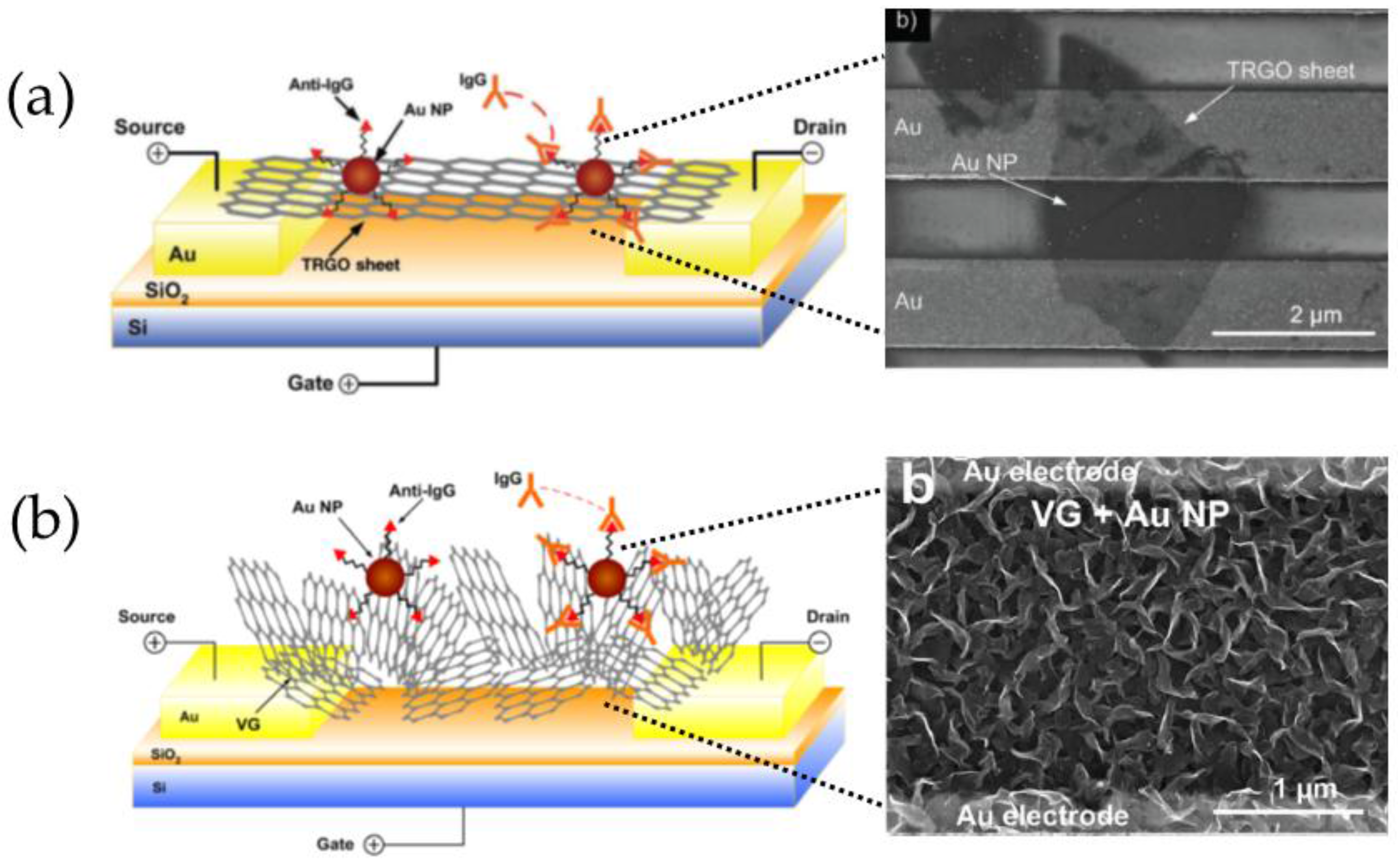
3.7. Hybrid Nanomaterials
3.8. Other Nanomaterials/Other Reports of Interest
4. Aptamers Instead of Antibodies
5. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
Abbreviations
| λD | Debye length |
| AFB1 | aflatoxin |
| AFP | α-fetoprotein |
| APTES | 3-aminopropyltriethoxysilane |
| BSA | bovine serum albumin |
| CA 19-9 | carbohydrate antigen 19-9 |
| CDR | complementarity determining regions |
| CEA | carcinoembryonic antigen |
| CeOx | cerium oxide |
| CPPyNP | carboxylated polypirrole nanoparticles |
| CRP | C-reactive protein |
| cTnI | cardiac troponin I |
| CVD | chemical vapor deposition |
| CYFRA21-1 | cytokeratin 19 fragments |
| DHEA-S | dehydroepiandrosterone sulfate |
| EDC | 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide |
| EGFR | epidermal growth factor receptor |
| ELISA | enzyme-linked immunosorbent assay |
| Fab | antigen-binding fragments |
| FET | field-effect transistor |
| HER2 | human epidermal growth factor receptor 2 |
| HBsAg | hepatitis B marker |
| HIV | human immunodeficiency virus |
| InP | indium phosphide |
| IDS | drain-source current |
| IFN-γ | interferon-gamma |
| IgE | immunoglobulin E |
| IgG | immunoglobulin G |
| IL-6 | interleukin-6 |
| IL-8 | interleukin-8 |
| ISFET | ion-sensitive field effect transistor |
| LOD | limit of detection |
| MCRL | microcystin-LR |
| MoS2 | molybdenum disulfide |
| MWCNT | multi-walled carbon nanotubes |
| NHS | N-hydroxysuccinimide |
| NSE | neuron-specific enolase |
| OPN | osteopontin |
| P3HT | poly-3-hexyl thiophene |
| PBASE | 1-pyrene butanoic acid succinimidyl ester |
| PBS | phosphate-buffered saline |
| PDMS | polydimethylsiloxane |
| PoC | point-of-care |
| PSA | prostate specific antigen |
| PSA-ACT | Prostate specific antigen/α1-antichymotrypsin |
| RIA | radioimmunoassay |
| RGO | reduced graphene oxide |
| SAM | self-assembled monolayers |
| SELEX | selection evolution of ligands by exponential enrichment |
| SEM | scanning electron microscopy |
| SiNG | silicon nanogratings |
| SiNW | silicon nanowire |
| SWCNT | single-walled carbon nanotubes |
| TiO2 | titanium dioxide |
| TNF-α | tumor necrosis factor α |
| TNT | 2,4,6-trinitrotoluene |
| VDS | drain-source voltage |
| VEGF | vascular endothelial growth factor |
| VG | gate voltage |
| ZnO | zinc oxide |
References
- Freire, R.S.; Pessoa, C.A.; Mello, L.D.; Kubota, L.T. Direct electron transfer: An approach for electrochemical biosensors with higher selectivity and sensitivity. J. Braz. Chem. Soc. 2003, 14, 230–243. [Google Scholar] [CrossRef]
- Thevenot, D.R.; Toth, K.; Durst, R.A.; Wilson, G.S. Electrochemical biosensors: Recommended definitions and classification. Biosens. Bioelectron. 2001, 16, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Luppa, P.B.; Sokoll, L.J.; Chan, D.W. Immunosensors—Principles and applications to clinical chemistry. Clin. Chim. Acta 2001, 314, 1–26. [Google Scholar] [CrossRef]
- Conroy, P.J.; Hearty, S.; Leonard, P.; O’Kennedy, R.J. Antibody production, design and use for biosensor-based applications. Semin. Cell Dev. Biol. 2009, 20, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Viguier, C.; Lynam, C.; O’Kennedy, R. Trends and perspectives in immunosensors. In Antibodies Applications and New Developments; 2012; pp. 184–208. Available online: http://ebooks.benthamscience.com/book/9781608052646/ (accessed on 20 October 2016).
- Hock, B. Antibodies for immunosensors—A review. Anal. Chim. Acta 1997, 347, 177–186. [Google Scholar] [CrossRef]
- Wang, J. Electrochemical biosensors: Towards point-of-care cancer diagnostics. Biosens. Bioelectron. 2006, 21, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Luo, Y.N.; Zhu, C.Z.; Li, H.; Du, D.; Lin, Y.H. Recent advances in electrochemical biosensors based on graphene two-dimensional nanomaterials. Biosens. Bioelectron. 2016, 76, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.L.; Davis, J.J. Electrical biosensors and the label free detection of protein disease biomarkers. Chem. Soc. Rev. 2013, 42, 5944–5962. [Google Scholar] [CrossRef] [PubMed]
- Porstmann, T.; Kiessig, S.T. Enzyme-immunoassay techniques—An overview. J. Immunol. Methods 1992, 150, 5–21. [Google Scholar] [CrossRef]
- Lee, T.M.H. Over-the-counter biosensors: Past, present, and future. Sensors 2008, 8, 5535–5559. [Google Scholar] [CrossRef]
- Ivnitski, D.; Abdel-Hamid, I.; Atanasov, P.; Wilkins, E. Biosensors for detection of pathogenic bacteria. Biosens. Bioelectron. 1999, 14, 599–624. [Google Scholar] [CrossRef]
- Yalow, R.S.; Berson, S.A. Immunoassay of endogenous plasma insulin in man. J. Clin. Investig. 1960, 39, 1157–1175. [Google Scholar] [CrossRef] [PubMed]
- Holford, T.R.J.; Davis, F.; Higson, S.P.J. Recent trends in antibody based sensors. Biosens. Bioelectron. 2012, 34, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Centi, S.; Laschi, S.; Mascini, M. Strategies for electrochemical detection in immunochemistry. Bioanalysis 2009, 1, 1271–1291. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.P.; Gee, S.; Hammock, B.D. Immunochemical techniques for environmental-analysis. 1. Immunosensors. Trends Anal. Chem. 1995, 14, 341–350. [Google Scholar] [CrossRef]
- Zachariah, E.S.; Gopalakrishnakone, P.; Neuzil, P. Immunologically sensitive field-effect transistors. In Encyclopedia of Medical Devices and Instrumentation; John Wiley & Sons: New York, NY, USA, 2006. [Google Scholar]
- Poghossian, A.; Schoning, M.J. Label-free sensing of biomolecules with field-effect devices for clinical applications. Electroanal 2014, 26, 1197–1213. [Google Scholar] [CrossRef]
- Schoning, M.J.; Poghossian, A. Bio feds (field-effect devices): State-of-the-art and new directions. Electroanal 2006, 18, 1893–1900. [Google Scholar] [CrossRef]
- Bergveld, P. Development, operation, and application of ion-sensitive field-effect transistor as a tool for electrophysiology. IEEE Trans. Biomed. Eng. 1972, 19, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Bergveld, P. A critical-evaluation of direct electrical protein-detection methods. Biosens. Bioelectron. 1991, 6, 55–72. [Google Scholar] [CrossRef]
- Bergveld, P. The future of biosensors. Sens. Actuat. A Phys. 1996, 56, 65–73. [Google Scholar] [CrossRef]
- Schasfoort, R.B.M.; Bergveld, P.; Kooyman, R.P.H.; Greve, J. Possibilities and limitations of direct detection of protein charges by means of an immunological field-effect transistor. Anal. Chim. Acta 1990, 238, 323–329. [Google Scholar] [CrossRef]
- Yin, P.T.; Shah, S.; Chhowalla, M.; Lee, K.B. Design, synthesis, and characterization of graphene-nanoparticle hybrid materials for bioapplications. Chem. Rev. 2015, 115, 2483–2531. [Google Scholar] [CrossRef] [PubMed]
- Ohno, Y.; Maehashi, K.; Yamashiro, Y.; Matsumoto, K. Electrolyte-gated graphene field-effect transistors for detecting ph protein adsorption. Nano Lett. 2009, 9, 3318–3322. [Google Scholar] [CrossRef] [PubMed]
- Ang, P.K.; Chen, W.; Wee, A.T.S.; Loh, K.P. Solution-gated epitaxial graphene as ph sensor. J. Am. Chem. Soc. 2008, 130, 14392–14393. [Google Scholar] [CrossRef] [PubMed]
- Di Bartolomeo, A.; Rinzan, M.; Boyd, A.K.; Yang, Y.; Guadagno, L.; Giubileo, F.; Barbara, P. Electrical properties and memory effects of field-effect transistors from networks of single-and double-walled carbon nanotubes. Nanotechnology 2010, 21, 115204. [Google Scholar] [CrossRef] [PubMed]
- Cramer, T.; Campana, A.; Leonardi, F.; Casalini, S.; Kyndiah, A.; Murgia, M.; Biscarini, F. Water-gated organic field effect transistors—opportunities for biochemical sensing and extracellular signal transduction. J. Mater. Chem. B 2013, 1, 3728–3741. [Google Scholar] [CrossRef]
- Patolsky, F.; Zheng, G.F.; Lieber, C.M. Nanowire-based biosensors. Anal. Chem. 2006, 78, 4260–4269. [Google Scholar] [CrossRef] [PubMed]
- Schenk, J.F. Theory, Design, and Biomedical Applications of Solid State Chemical Sensors; Cheung, P.W., Ed.; CRC Press: West Palm Beach, FL, USA, 1978; p. 296. [Google Scholar]
- Yin, P.T.; Kim, T.H.; Choi, J.W.; Lee, K.B. Prospects for graphene-nanoparticle-based hybrid sensors. Phys. Chem. Chem. Phys. 2013, 15, 12785–12799. [Google Scholar] [CrossRef] [PubMed]
- Pumera, M. Graphene in biosensing. Mater. Today 2011, 14, 308–315. [Google Scholar] [CrossRef]
- Ramnani, P.; Saucedo, N.M.; Mulchandani, A. Carbon nanomaterial-based electrochemical biosensors for label-free sensing of environmental pollutants. Chemosphere 2016, 143, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Nehra, A.; Singh, K.P. Current trends in nanomaterial embedded field effect transistor-based biosensor. Biosens. Bioelectron. 2015, 74, 731–743. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Yan, F. Organic thin-film transistors for chemical and biological sensing. Adv. Mater. 2012, 24, 34–51. [Google Scholar] [CrossRef] [PubMed]
- He, R.X.; Lin, P.; Liu, Z.K.; Zhu, H.W.; Zhao, X.Z.; Chan, H.L.W.; Yan, F. Solution-gated graphene field effect transistors integrated in microfluidic systems and used for flow velocity detection. Nano Lett. 2012, 12, 1404–1409. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Chang, Y.; Sawtelle, S.D.; Wipf, M.; Duan, X.X.; Reed, M.A. Silicon nanowire field-effect transistors-a versatile class of potentiometric nanobiosensors. IEEE Access 2015, 3, 287–302. [Google Scholar] [CrossRef]
- Debye, P. Reaction rates in ionic solutions. J. Electrochem. Soc. 1942, 82, 265–272. [Google Scholar] [CrossRef]
- Russel, W.B.; Saville, D.A.; Schowalter, W.R. Colloidal Dispersions; Cambridge University Press: Cambridge, UK, 1989. [Google Scholar]
- Elnathan, R.; Kwiat, M.; Pevzner, A.; Engel, Y.; Burstein, L.; Khatchtourints, A.; Lichtenstein, A.; Kantaev, R.; Patolsky, F. Biorecognition layer engineering: Overcoming screening limitations of nanowire-based fet devices. Nano Lett. 2012, 12, 5245–5254. [Google Scholar] [CrossRef] [PubMed]
- Stern, E.; Wagner, R.; Sigworth, F.J.; Breaker, R.; Fahmy, T.M.; Reed, M.A. Importance of the debye screening length on nanowire field effect transistor sensors. Nano Lett. 2007, 7, 3405–3409. [Google Scholar] [CrossRef] [PubMed]
- Casal, P.; Wen, X.J.; Gupta, S.; Nicholson, T.; Wang, Y.J.; Theiss, A.; Bhushan, B.; Brillson, L.; Lu, W.; Lee, S.C. Immunofet feasibility in physiological salt environments. Philos. Trans. R. Soc. A 2012, 370, 2474–2488. [Google Scholar] [CrossRef] [PubMed]
- Schoning, M.J.; Poghossian, A. Recent advances in biologically sensitive field-effect transistors (biofets). Analyst 2002, 127, 1137–1151. [Google Scholar] [CrossRef] [PubMed]
- Ansari, A.A.; Alhoshan, M.; Alsalhi, M.S.; Aldwayyan, A.S. Prospects of nanotechnology in clinical immunodiagnostics. Sensors 2010, 10, 6535–6581. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.Z.; Yang, G.H.; Li, H.; Du, D.; Lin, Y.H. Electrochemical sensors and biosensors based on nanomaterials and nanostructures. Anal. Chem. 2015, 87, 230–249. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.H.; Li, C.M. Nanomaterial-based advanced immunoassays. Wires Nanomed. Nanobiotechnol. 2011, 3, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Gao, A.R.; Dai, P.F.; Mao, H.J.; Zuo, X.L.; Fan, C.H.; Wang, Y.L.; Li, T. Ultrasensitive detection of dual cancer biomarkers with integrated cmos-compatible nanowire arrays. Anal. Chem. 2015, 87, 11203–11208. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Wu, C.S.; Chuang, C.K.; Pang, S.T.; Pan, T.M.; Yang, Y.S.; Ko, F.H. Real-time and label-free detection of the prostate-specific antigen in human serum by a polycrystalline silicon nanowire field-effect transistor biosensor. Anal. Chem. 2013, 85, 7912–7918. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.J.; Sohn, I.Y.; Jung, J.H.; Yoon, O.J.; Lee, N.E.; Park, J.S. Reduced graphene oxide field-effect transistor for label-free femtomolar protein detection. Biosens. Bioelectron. 2013, 41, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Dak, P.; Lee, Y.; Park, H.; Choi, W.; Alam, M.A.; Kim, S. Two-dimensional layered mos2 biosensors enable highly sensitive detection of biomolecules. Sci. Rep. 2014, 4, 7352. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Y.; Wong, J.I.; Palacios, T.; Kong, J.; Yang, H.Y. Functionalized mos2 nanosheet-based field-effect biosensor for label-free sensitive detection of cancer marker proteins in solution. Small 2014, 10, 1101–1105. [Google Scholar] [CrossRef] [PubMed]
- Myung, S.; Solanki, A.; Kim, C.; Park, J.; Kim, K.S.; Lee, K.B. Graphene-encapsulated nanoparticle-based biosensor for the selective detection of cancer biomarkers. Adv. Mater. 2011, 23, 2221–2225. [Google Scholar] [CrossRef] [PubMed]
- Reyes, P.I.; Ku, C.J.; Duan, Z.Q.; Lu, Y.C.; Solanki, A.; Lee, K.B. Zno thin film transistor immunosensor with high sensitivity and selectivity. Appl. Phys. Lett. 2011, 98, 173702. [Google Scholar] [CrossRef]
- Zhu, K.Y.; Zhang, Y.; Li, Z.Y.; Zhou, F.; Feng, K.; Dou, H.Q.; Wang, T. Simultaneous detection of alpha-fetoprotein and carcinoembryonic antigen based on si nanowire field-effect transistors. Sensors 2015, 15, 19225–19236. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.L.; Chen, R.M.; Xu, L.; Ning, Y.; Xie, S.G.; Zhang, G.J. Silicon nanowire biosensor for highly sensitive and multiplexed detection of oral squamous cell carcinoma biomarkers in saliva. Anal. Sci. 2015, 31, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Sohn, I.Y.; Kim, D.J.; Kim, B.Y.; Fang, M.; Lee, N.E. Enhancement of protein detection performance in field-effect transistors with polymer residue-free graphene channel. Carbon 2013, 62, 312–321. [Google Scholar] [CrossRef]
- Chen, H.C.; Chen, Y.T.; Tsai, R.Y.; Chen, M.C.; Chen, S.L.; Xiao, M.C.; Chen, C.L.; Hua, M.Y. A sensitive and selective magnetic graphene composite-modified polycrystalline-silicon nanowire field-effect transistor for bladder cancer diagnosis. Biosens. Bioelectron. 2015, 66, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Justino, C.I.L.; Freitas, A.C.; Amaral, J.P.; Rocha-Santos, T.A.P.; CardosoC, S.; Duarte, A.C. Disposable immunosensors for c-reactive protein based on carbon nanotubes field effect transistors. Talanta 2013, 108, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Park, C.; Kwon, D.; Kim, D.; Meyyappan, M.; Jeon, S.; Lee, J.S. Silicon nanowire biosensors for detection of cardiac troponin i (ctni) with high sensitivity. Biosens. Bioelectron. 2016, 77, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Kong, T.; Su, R.G.; Zhang, B.B.; Zhang, Q.; Cheng, G.S. Cmos-compatible, label-free silicon-nanowire biosensors to detect cardiac troponin i for acute myocardial infarction diagnosis. Biosens. Bioelectron. 2012, 34, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Dai, P.F.; Gao, A.R.; Valiaho, J.; Kallio, P.; Wang, Y.L.; Li, T. Label-free and rapid electrical detection of htsh with cmos-compatible silicon nanowire transistor arrays. ACS Appl. Mater. Interfaces 2014, 6, 20378–20384. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xu, J.-Q.; Li, T.; Huang, L.; Pang, D.-W.; Ning, Y.; Huang, W.-H.; Zhang, Z.; Zhang, G.-J. Photocatalysis-induced renewable field-effect transistor for protein detection. Anal. Chem. 2016, 88, 4048–4054. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Yoo, G.; Chang, Y.W.; Kim, H.J.; Jose, J.; Kim, E.; Pyun, J.C.; Yoo, K.H. A carbon nanotube metal semiconductor field effect transistor-based biosensor for detection of amyloid-beta in human serum. Biosens. Bioelectron. 2013, 50, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Regonda, S.; Tian, R.H.; Gao, J.M.; Greene, S.; Ding, J.H.; Hu, W. Silicon multi-nanochannel fets to improve device uniformity/stability and femtomolar detection of insulin in serum. Biosens. Bioelectron. 2013, 45, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.N.; Bian, C.; Tong, J.H.; Sun, J.Z.; Zhang, H.; Xia, S.H. Fet immunosensor for hemoglobin a1c using a gold nanofilm grown by a seed-mediated technique and covered with mixed self-assembled monolayers. Microchim. Acta 2012, 176, 65–72. [Google Scholar] [CrossRef]
- Lerner, M.B.; Goldsmith, B.R.; McMillon, R.; Dailey, J.; Pillai, S.; Singh, S.R.; Johnson, A.T.C. A carbon nanotube immunosensor for salmonella. Aip Adv. 2011, 1. [Google Scholar] [CrossRef]
- Starodub, N.F.; Ogorodnijchuk, J.O. Immune biosensor based on the isfets for express determination of Salmonella typhimurium. Electroanal 2012, 24, 600–606. [Google Scholar] [CrossRef]
- Chang, J.B.; Mao, S.; Zhang, Y.; Cui, S.M.; Zhou, G.H.; Wu, X.G.; Yang, C.H.; Chen, J.H. Ultrasonic-assisted self-assembly of monolayer graphene oxide for rapid detection of Escherichia coli bacteria. Nanoscale 2013, 5, 3620–3626. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aljaro, C.; Cella, L.N.; Shirale, D.J.; Park, M.; Munoz, F.J.; Yates, M.V.; Mulchandani, A. Carbon nanotubes-based chemiresistive biosensors for detection of microorganisms. Biosens. Bioelectron. 2010, 26, 1437–1441. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.X.; Dong, X.C.; Liu, Y.X.; Li, L.J.; Chen, P. Graphene-based biosensors for detection of bacteria and their metabolic activities. J. Mater. Chem. 2011, 21, 12358–12362. [Google Scholar] [CrossRef]
- Yamada, K.; Kim, C.T.; Kim, J.H.; Chung, J.H.; Lee, H.G.; Jun, S. Single walled carbon nanotube-based junction biosensor for detection of Escherichia coli. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Kim, Y.H.; Cheon, D.S.; Seo, T.S. Micropatterned reduced graphene oxide based field-effect transistor for real-time virus detection. Sens. Actuat. B Chem. 2013, 186, 252–257. [Google Scholar] [CrossRef]
- Wong, J.I.; Wang, L.; Shi, Y.M.; Palacios, T.; Kong, J.; Dong, X.C.; Yang, H.Y. Real-time, sensitive electrical detection of cryptosporidium parvum oocysts based on chemical vapor deposition-grown graphene. Appl. Phys. Lett. 2014, 104, 063705. [Google Scholar] [CrossRef]
- Belkhamssa, N.; Justino, C.I.L.; Santos, P.S.M.; Cardoso, S.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T.; Ksibi, M. Label-free disposable immunosensor for detection of atrazine. Talanta 2016, 146, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Wijaya, I.P.M.; Nie, T.J.; Gandhi, S.; Boro, R.; Palaniappan, A.; Hau, G.W.; Rodriguez, I.; Suri, C.R.; Mhaisalkar, S.G. Femtomolar detection of 2,4-dichlorophenoxyacetic acid herbicides via competitive immunoassays using microfluidic based carbon nanotube liquid gated transistor. Lab Chip 2010, 10, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Cella, L.N.; Chen, W.F.; Myung, N.V.; Mulchandani, A. Carbon nanotubes-based chemiresistive immunosensor for small molecules: Detection of nitroaromatic explosives. Biosens. Bioelectron. 2010, 26, 1297–1301. [Google Scholar] [CrossRef] [PubMed]
- Moreau, A.L.D.; Janissen, R.; Santos, C.A.; Peroni, L.A.; Stach-Machado, D.R.; de Souza, A.A.; de Souza, A.P.; Cotta, M.A. Highly-sensitive and label-free indium phosphide biosensor for early phytopathogen diagnosis. Biosens. Bioelectron. 2012, 36, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Chiang, P.L.; Chou, T.C.; Wu, T.H.; Li, C.C.; Liao, C.D.; Lin, J.Y.; Tsai, M.H.; Tsai, C.C.; Sun, C.J.; Wang, C.H.; et al. Nanowire transistor-based ultrasensitive virus detection with reversible surface functionalization. Chem. Asian J. 2012, 7, 2073–2079. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, Y.D.; Pleshakova, T.O.; Kozlov, A.F.; Malsagova, K.A.; Krohin, N.V.; Shumyantseva, V.V.; Shumov, I.D.; Popov, V.P.; Naumova, O.V.; Fomin, B.I.; et al. Soi nanowire for the high-sensitive detection of hbsag and alpha-fetoprotein. Lab Chip 2012, 12, 5104–5111. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Hong, S.; Singh, R.; Jang, J. Single-walled carbon nanotube based transparent immunosensor for detection of a prostate cancer biomarker osteopontin. Anal. Chim. Acta 2015, 869, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Choo, T.K.; Huang, J.F.; Wang, Y.; Liu, Y.J.; Platt, M.; Palaniappan, A.; Liedberg, B.; Tok, A.I.Y. Label-free electronic detection of interleukin-6 using horizontally aligned carbon nanotubes. Mater. Des. 2016, 90, 852–857. [Google Scholar] [CrossRef]
- Tlili, C.; Myung, N.V.; Shetty, V.; Mulchandani, A. Label-free, chemiresistor immunosensor for stress biomarker cortisol in saliva. Biosens. Bioelectron. 2011, 26, 4382–4386. [Google Scholar] [CrossRef] [PubMed]
- Lerner, M.B.; Dailey, J.; Goldsmith, B.R.; Brisson, D.; Johnson, A.T.C. Detecting lyme disease using antibody-functionalized single-walled carbon nanotube transistors. Biosens. Bioelectron. 2013, 45, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Lerner, M.B.; D'Souza, J.; Pazina, T.; Dailey, J.; Goldsmith, B.R.; Robinson, M.K.; Johnson, A.T.C. Hybrids of a genetically engineered antibody and a carbon nanotube transistor for detection of prostate cancer biomarkers. ACS Nano 2012, 6, 5143–5149. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.H.; Kumar, V.; Moyano, D.R.; Wen, S.H.; Parashar, V.; Hsiao, S.H.; Srivastava, A.; Saxena, P.S.; Huang, K.P.; Chang, C.C.; et al. High-performance and high-sensitivity applications of graphene transistors with self-assembled monolayers. Biosens. Bioelectron. 2016, 77, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.S.; Lee, S.H.; Park, S.J.; An, J.H.; Song, H.S.; Kim, T.; Oh, J.H.; Bae, J.; Yoon, H.; Park, T.H.; et al. Large-scale graphene micropattern nano-biohybrids: High-performance transducers for fet-type flexible fluidic HIV immunoassays. Adv. Mater. 2013, 25, 4177–4185. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.Y.; Sohn, I.Y.; Lee, D.; Han, G.S.; Lee, W.I.; Jung, H.S.; Lee, N.E. Ultrarapid and ultrasensitive electrical detection of proteins in a three-dimensional biosensor with high capture efficiency. Nanoscale 2015, 7, 9844–9851. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Pan, T.M.; Wu, C.S.; Yen, L.C.; Chuang, C.K.; Pang, S.T.; Yang, Y.S.; Ko, F.H. Label-free detection of prostate specific antigen using a silicon nanobelt field-effect transistor. Int. J. Electrochem. Sci. 2012, 7, 4432–4442. [Google Scholar]
- Magliulo, M.; De Tullio, D.; Vikholm-Lundin, I.; Albers, W.; Munter, T.; Manoli, K.; Palazzo, G.; Torsi, L. Label-free c-reactive protein electronic detection with an electrolyte-gated organic field-effect transistor-based immunosensor. Anal. Bioanal. Chem. 2016, 408, 3943–3952. [Google Scholar] [CrossRef] [PubMed]
- Basu, J.; Datta, S.; RoyChaudhuri, C. A graphene field effect capacitive immunosensor for sub-femtomolar food toxin detection. Biosens. Bioelectron. 2015, 68, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Saucedo, N.M.; Ramnani, P.; Mulchandani, A. Label-free electrical immunosensor for highly sensitive and specific detection of microcystin-lr in water samples. Environ. Sci. Technol. 2015, 49, 9256–9263. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.T.; Odom, T.W.; Lieber, C.M. Chemistry and physics in one dimension: Synthesis and properties of nanowires and nanotubes. Acc. Chem. Res. 1999, 32, 435–445. [Google Scholar] [CrossRef]
- Cui, Y.; Duan, X.F.; Hu, J.T.; Lieber, C.M. Doping and electrical transport in silicon nanowires. J. Phys. Chem. B 2000, 104, 5213–5216. [Google Scholar] [CrossRef]
- Hasan, M.; Huq, M.F.; Mahmood, Z.H. A review on electronic and optical properties of silicon nanowire and its different growth techniques. Springerplus 2013, 2, 151. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.I.; Li, B.R.; Chen, Y.T. Silicon nanowire field-effect transistor-based biosensors for biomedical diagnosis and cellular recording investigation. Nano Today 2011, 6, 131–154. [Google Scholar] [CrossRef]
- Noor, M.O.; Krull, U.J. Silicon nanowires as field-effect transducers for biosensor development: A review. Anal. Chim. Acta 2014, 825, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Wei, Q.Q.; Park, H.K.; Lieber, C.M. Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species. Science 2001, 293, 1289–1292. [Google Scholar] [CrossRef] [PubMed]
- Puppo, F.; Doucey, M.-A.; Moh, T.S.Y.; Pandraud, G.; Sarro, P.M.; De Micheli, G.; Carrara, S. Femto-molar sensitive field effect transistor biosensors based on silicon nanowires and antibodies. In Proceedings of the 2013 IEEE Sensors Proceedings, Baltimore, MD, USA, 4–6 November 2013.
- Popov, V.N. Carbon nanotubes: Properties and application. Mater. Sci. Eng. R 2004, 43, 61–102. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nature 1993, 363, 603–605. [Google Scholar] [CrossRef]
- Bethune, D.S.; Kiang, C.H.; Devries, M.S.; Gorman, G.; Savoy, R.; Vazquez, J.; Beyers, R. Cobalt-catalyzed growth of carbon nanotubes with single-atomic-layerwalls. Nature 1993, 363, 605–607. [Google Scholar] [CrossRef]
- Sarkar, T.; Gao, Y.N.; Mulchandani, A. Carbon nanotubes-based label-free affinity sensors for environmental monitoring. Appl. Biochem. Biotechnol. 2013, 170, 1011–1025. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Ruoff, R.S. Chemical methods for the production of graphenes. Nat. Nanotechnol. 2009, 4, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.J.; Dong, S.J. Graphene nanosheet: Synthesis, molecular engineering, thin film, hybrids, and energy and analytical applications. Chem. Soc. Rev. 2011, 40, 2644–2672. [Google Scholar] [CrossRef] [PubMed]
- Di Bartolomeo, A. Graphene schottky diodes: An experimental review of the rectifying graphene/semiconductor heterojunction. Phys. Rep. 2016, 606, 1–58. [Google Scholar] [CrossRef]
- Mas-Balleste, R.; Gomez-Navarro, C.; Gomez-Herrero, J.; Zamora, F. 2D materials: To graphene and beyond. Nanoscale 2011, 3, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Partoens, B.; Peeters, F.M. From graphene to graphite: Electronic structure around the k point. Phys. Rev. B 2006, 74, 075404. [Google Scholar] [CrossRef]
- Schwierz, F. Graphene transistors. Nat. Nanotechnol. 2010, 5, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Di Bartolomeo, A.; Giubileo, F.; Santandrea, S.; Romeo, F.; Citro, R.; Schroeder, T.; Lupina, G. Charge transfer and partial pinning at the contacts as the origin of a double dip in the transfer characteristics of graphene-based field-effect transistors. Nanotechnology 2011, 22, 275702. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.X.; Dong, X.C.; Chen, P. Biological and chemical sensors based on graphene materials. Chem. Soc. Rev. 2012, 41, 2283–2307. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Ohno, Y.; Maehashi, K.; Inoue, K.; Matsumoto, K. Immunosensors based on graphene field-effect transistors fabricated using antigen-binding fragment. Jpn. J. Appl. Phys. 2012, 51, 06FD08. [Google Scholar] [CrossRef]
- Tong, X.; Ashalley, E.; Lin, F.; Li, H.D.; Wang, Z.M.M. Advances in mos2-based field effect transistors (fets). Nano-Micro Lett. 2015, 7, 203–218. [Google Scholar] [CrossRef]
- Sarkar, D.; Liu, W.; Xie, X.J.; Anselmo, A.C.; Mitragotri, S.; Banerjee, K. Mos2 field-effect transistor for next-generation label-free biosensors. ACS Nano 2014, 8, 3992–4003. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.; Xie, X.J.; Kang, J.H.; Zhang, H.J.; Liu, W.; Navarrete, J.; Moskovits, M.; Banerjee, K. Functionalization of transition metal dichalcogenides with metallic nanoparticles: Implications for doping and gas-sensing. Nano Lett. 2015, 15, 2852–2862. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zhou, B.X. Titanium dioxide nanomaterials for sensor applications. Chem. Rev. 2014, 114, 10131–10176. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.H.; Ruan, C.M.; Kanayeva, D.; Lassiter, K.; Li, Y.B. Tio2 nanowire bundle microelectrode based impedance immunosensor for rapid and sensitive detection of listeria monocytogenes. Nano Lett. 2008, 8, 2625–2631. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.M.; Lin, C.C.; Chang, H.C.; Li, C.M.; Guo, C.X. Tio(2) nanowire fet device: Encapsulation of biomolecules by electro polymerized pyrrole propylic acid. Biosens. Bioelectron. 2011, 26, 2334–2340. [Google Scholar] [CrossRef] [PubMed]
- Arya, S.K.; Saha, S.; Ramirez-Vick, J.E.; Gupta, V.; Bhansali, S.; Singh, S.P. Recent advances in zno nanostructures and thin films for biosensor applications: Review. Anal. Chim. Acta 2012, 737, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Yano, M.; Koike, K.; Mukai, K.; Onaka, T.; Hirofuji, Y.; Ogata, K.; Omatu, S.; Maemoto, T.; Sasa, S. Zinc oxide ion-sensitive field-effect transistors and biosensors. Phys. Status Solidi A 2014, 211, 2098–2104. [Google Scholar] [CrossRef]
- Zhao, Z.W.; Lei, W.; Zhang, X.B.; Wang, B.P.; Jiang, H.L. Zno-based amperometric enzyme biosensors. Sensors 2010, 10, 1216–1231. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Weis, M.; Wei, O.Y.; Taguchi, D.; Manaka, T.; Iwamoto, M. Effects of gold nanoparticles on pentacene organic field-effect transistors. Jpn. J. Appl. Phys. 2011, 50, 041601. [Google Scholar]
- Mao, S.; Lu, G.H.; Yu, K.H.; Chen, J.H. Specific biosensing using carbon nanotubes functionalized with gold nanoparticle-antibody conjugates. Carbon 2010, 48, 479–486. [Google Scholar] [CrossRef]
- Mao, S.; Lu, G.H.; Yu, K.H.; Bo, Z.; Chen, J.H. Specific protein detection using thermally reduced graphene oxide sheet decorated with gold nanoparticle-antibody conjugates. Adv. Mater. 2010, 22, 3521–3526. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Yu, K.H.; Chang, J.B.; Steeber, D.A.; Ocola, L.E.; Chen, J.H. Direct growth of vertically-oriented graphene for field-effect transistor biosensor. Sci. Rep. 2013, 3, 1696. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.S.; Hideshima, S.; Kuroiwa, S.; Nakanishi, T.; Osaka, T. Label-free detection of tumor markers using field effect transistor (fet)-based biosensors for lung cancer diagnosis. Sens. Actuat. B Chem. 2015, 212, 329–334. [Google Scholar] [CrossRef]
- Jayasena, S.D. Aptamers: An emerging class of molecules that rival antibodies in diagnostics. Clin. Chem. 1999, 45, 1628–1650. [Google Scholar] [PubMed]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment—RNA ligands to bacteriophage-t4 DNA-polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. Invitro selection of rna molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Raston, N.H.A.; Gu, M.B. Aptamer-based nanobiosensors. Biosens. Bioelectron. 2016, 76, 2–19. [Google Scholar] [PubMed]
- Chiu, T.C.; Huang, C.C. Aptamer-functionalized nano-biosensors. Sensors 2009, 9, 10356–10388. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.O.; So, H.M.; Jeon, E.K.; Chang, H.; Won, K.; Kim, Y.H. Aptamers as molecular recognition elements for electrical nanobiosensors. Anal. Bioanal. Chem. 2008, 390, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Farid, S.; Meshik, X.; Choi, M.; Mukherjee, S.; Lan, Y.; Parikh, D.; Poduri, S.; Baterdene, U.; Huang, C.E.; Wang, Y.Y.; et al. Detection of interferon gamma using graphene and aptamer based fet-like electrochemical biosensor. Biosens. Bioelectron. 2015, 71, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kim, K.S.; Kim, C.J.; Hahn, S.K.; Jo, M.H. Electrical detection of vegfs for cancer diagnoses using anti-vascular endotherial growth factor aptamer-modified si nanowire fets. Biosens. Bioelectron. 2009, 24, 1801–1805. [Google Scholar] [CrossRef] [PubMed]
- Maehashi, K.; Matsumoto, K.; Takamura, Y.; Tamiya, E. Aptamer-based label-free immunosensors using carbon nanotube field-effect transistors. Electroanal 2009, 21, 1285–1290. [Google Scholar] [CrossRef]
- Ohno, Y.; Maehashi, K.; Matsumoto, K. Label-free biosensors based on aptamer-modified graphene field-effect transistors. J. Am. Chem. Soc. 2010, 132, 18012–18013. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Na, W.; Jang, J. One-pot synthesis of multidimensional conducting polymer nanotubes for superior performance field-effect transistor-type carcinoembryonic antigen biosensors. RSC Adv. 2016, 6, 14335–14343. [Google Scholar] [CrossRef]
- So, H.M.; Won, K.; Kim, Y.H.; Kim, B.K.; Ryu, B.H.; Na, P.S.; Kim, H.; Lee, J.O. Single-walled carbon nanotube biosensors using aptamers as molecular recognition elements. J. Am. Chem. Soc. 2005, 127, 11906–11907. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Kim, J.; Zhu, Y.; Yang, J.; Lee, G.-H.; Lee, S.; Yu, J.; Pei, R.; Liu, G.; Nuckolls, C.; et al. An aptameric graphene nanosensor for label-free detection of small-molecule biomarkers. Biosens. Bioelectron. 2015, 71, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Pacios, M.; Martin-Fernandez, I.; Borrise, X.; del Valle, M.; Bartroli, J.; Lora-Tamayo, E.; Godignon, P.; Perez-Murano, F.; Esplandiu, M.J. Real time protein recognition in a liquid-gated carbon nanotube field-effect transistor modified with aptamers. Nanoscale 2012, 4, 5917–5923. [Google Scholar] [CrossRef] [PubMed]
- Grant, S.; Peter, W.; Tal, S.; Matthew, R.L.; Jenna, L.W.; Christopher, A.H.; Adeniyi, A.A.; Vincent, T.R.; Ethan, D.M. Scalable graphene field-effect sensors for specific protein detection. Nanotechnology 2013, 24, 355502. [Google Scholar]

| Target Analyte | Electroactive Nanomaterial | Range | Detection Limit | Reference |
|---|---|---|---|---|
| –H5N2 avian influenza virus | SiNW | 10−17–10−12 M | 10−17 M | [78] |
| Hepatitis B marker (HBsAg) and cancer marker α-fetoprotein (AFP) | SiNW | - | 10−14 M for HBsAg and 10−15 M for AFP | [79] |
| Human thyroid stimulating hormone | SiNW | 0.02–30 mIU·L−1 | 0.02 mIU·L−1 | [61] |
| Biomarkers of oral squamous cell carcinoma Interleukin-8 (IL-8) and tumor necrosis factor α (TNF-α) | SiNW | 1 fg·mL−1–1 ng·mL−1 | 10 fg·mL−1 in PBS and 100 fg·mL−1 in saliva | [55] |
| Cancer biomarkers cytokeratin 19 fragment (CYFRA21-1) and prostate specific antigen (PSA) | SiNW | 1 fg·mL−1–1 ng·mL−1 | 1 fg·mL−1 in buffer solution and 10 fg·mL−1 in undiluted human serums | [47] |
| Prostate cancer biomarker PSA | SiNW | 5 fg·mL−1–500 pg·mL−1 | 5 fg·mL−1 in buffer and desalted serum | [48] |
| AFP and carcinoembryonic antigen (CEA) primary hepatic carcinoma biomarkers | SiNW | 500 fg·mL−1–50 ng·mL−1 for AFP and 50 fg·mL−1–10 ng·mL−1 for CEA | 500 fg·mL−1 for AFP and 50 fg·mL−1 for CEA | [54] |
| Cardiac troponin I (cTnI) biomarker for acute myocardial infarction | SiNW | 0.092 ng·mL−1–46 ng·mL−1 | 0.092 ng·mL−1 | [60] |
| Cardiac disease biomarker cTnI | SiNW | 5 pg·mL−1–5 ng·mL−1 | 5 pg·mL−1 | [59] |
| C-reactive protein (CRP) inflammatory biomarker related with cardiovascular diseases | SWCNT | 10−4 to 102 µg·mL−1 | 10−4 µg·mL−1 | [58] |
| Prostate cancer biomarker osteopontin (OPN) | SWCNT | 1 pg·mL−1–1 µg·mL−1 | 0.3 pg·mL−1 | [80] |
| Pro-inflammatory cytokine and anti-inflammatory myokine interleukin-6 (IL-6) | SWCNT | 1 pg·mL−1–100 pg·mL−1 | 1.37 pg·mL−1 | [81] |
| Stress biomarker cortisol in saliva | SWCNT | 1 pg·mL−1–1000 ng·mL−1 | 1 pg·mL−1 | [82] |
| Lyme disease antigen | SWCNT | 1 ng·mL−1–3000 ng·mL−1 | 1 ng·mL−1 | [83] |
| Prostate cancer biomarker OPN | SWCNT | 1 pg·mL−1–1 μg·mL−1 | 1 pg·mL−1 or 30 fM | [84] |
| Alzheimer biomarker, amyloid-β | SWCNT | 10−12–10−9 g·mL−1 | 1 pg·mL−1 in human serum | [63] |
| Chondroitin sulfate proteoglycan 4, multiple cancer types biomarker | Graphene | 0.01 fM–10 pM | 0.01 fM | [85] |
| Pancreatic cancer biomarker, carbohydrate antigen 19-9 (CA 19-9) | Graphene | 0.01 unit·mL−1–1000 unit·mL−1 | 0.01 unit·mL−1 | [56] |
| Prostate specific antigen/α1-antichymotrypsin (PSA-ACT) complex | Graphene | 100 fg·mL−1–1 µg·mL−1 | 100 fg·mL−1 | [49] |
| Breast cancer biomarkers human epidermal growth factor receptor 2 (HER2) and epidermal growth factor receptor (EGFR) | SiO2/graphene | 100 pM–1 µM | 1 pM for HER2 and 100 pM for EGFR | [52] |
| Human immunodeficiency virus (HIV) | Graphene/CPPyNP | 1 pM–10 nM | 1 pM | [86] |
| Bladder cancer biomarker, urinary APOA2 protein | Graphene/SiNW | 19.5 pg·mL−1–1.95 mg·mL−1 | 6.7 pg·mL−1 | [57] |
| Prostate cancer biomarker PSA–ACT complex | Graphene/ZnO nanorods/TiO2 | 100 fg·mL−1–100 ng·mL−1 | 1 fM | [87] |
| D-Dimer, biomarker of venous thromboembolism | Graphene/TiO2 | 10 pg·mL−1–100 ng·mL−1 | 10 pg·mL−1 in buffer and 100 pg·mL−1 in serum sample | [62] |
| Prostate cancer biomarker PSA | MoS2 | 3.75 nM, 37.5 pM, and 375 fM | 375 fM | [51] |
| Prostate cancer biomarker PSA | MoS2 | 1 pg·mL−1–10 ng·mL−1 | 1 pg·mL−1 | [50] |
| Breast cancer biomarker EGFR | ZnO nanofilm | 10 fM–10 nM | 10 fM | [53] |
| Prostate cancer biomarker PSA | Si nanobelt | 50 fg·mL−1–500 pg·mL−1 | 5 pg·mL−1 | [88] |
| Index for diabetes, Hemoglobin-A1c | Au nanoparticles | 1.67 ng·mL−1–170.5 ng·mL−1 | in the order of ng·mL−1 | [65] |
| Diabetes related hormone, insulin | Si nanogratings | 1 fM–1 nM | 10 fM in buffer and diluted human serum | [64] |
| CRP inflammatory biomarker | P3HT polymer | 4 pM–2 μM | 2 pM | [89] |
| Target Analyte | Electroactive Nanomaterial | Range | Detection Limit | Reference |
|---|---|---|---|---|
| Food safety | ||||
| Salmonella bacteria | SWCNT | 103–108 CFU·mL−1 | 103 CFU·mL−1 | [66] |
| Escherichia coli foodborne pathogen | SWCNT | 102–105 CFU·mL−1 | 102 CFU·mL−1 | [71] |
| Escherichia coli O157:H7 and bacteriophage viruses | SWCNT | 103–107 CFU·mL−1 for E. coli and 102–107 PFU·mL−1 for bacteriophage | 105 CFU·mL−1 for E. coli and 103 PFU·mL−1 for bacteriophage | [69] |
| Cryptosporidium parvum intestinal parasitic protozoan | Graphene | 102–104 Cp. oocysts per 4 mL buffer | 25 Cp. oocysts per mL buffer | [73] |
| Escherichia coli bacteria | Graphene | 10–105 CFU·mL−1 | 10 CFU·mL−1 | [70] |
| Escherichia coli O157:H7 bacteria | Graphene | 10–104 CFU·mL−1 | 10 CFU·mL−1 | [68] |
| Food toxin aflatoxin B1 | Graphene | 10−4 ppt–1·ppt | 0.1 fg·mL−1 | [90] |
| Rotavirus | Graphene | 0–105 PFU·mL−1 | 102 PFU·mL−1 | [72] |
| Salmonella typhimurium bacteria | CeOx | 2–5 × 105 cells·mL−1 | 2–3 cells·mL−1 | [67] |
| Environmental monitoring | ||||
| Atrazine pesticide | SWCNT | 0.001–10 ng·mL−1 | 0.001 ng·mL−1 | [74] |
| 2,4-Dichlorophenoxyacetic acid herbicides | SWCNT | 5 fM–500 µM | 500 fM in soil sample and 50 pM in buffer | [75] |
| 2,4,6-Trinitrotoluene (TNT) contamination | SWCNT | 0.5 ppb–5000 ppb | 0.5 ppb | [76] |
| Microcystin-LR (cyanotoxin in surface waters) | SWCNT | 1–1000 ng·L−1 | 0.6 ng·L−1 | [91] |
| Citrus tristeza virus and Xylella fastidiosa phytopathogens | InP | 60–340 ng·mL−1 for Citrus tristeza virus and 34–250 ng·mL−1 for Xylella fastidiosa | 2 nM for both phytopathogens | [77] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Moraes, A.C.M.; Kubota, L.T. Recent Trends in Field-Effect Transistors-Based Immunosensors. Chemosensors 2016, 4, 20. https://doi.org/10.3390/chemosensors4040020
De Moraes ACM, Kubota LT. Recent Trends in Field-Effect Transistors-Based Immunosensors. Chemosensors. 2016; 4(4):20. https://doi.org/10.3390/chemosensors4040020
Chicago/Turabian StyleDe Moraes, Ana Carolina Mazarin, and Lauro Tatsuo Kubota. 2016. "Recent Trends in Field-Effect Transistors-Based Immunosensors" Chemosensors 4, no. 4: 20. https://doi.org/10.3390/chemosensors4040020
APA StyleDe Moraes, A. C. M., & Kubota, L. T. (2016). Recent Trends in Field-Effect Transistors-Based Immunosensors. Chemosensors, 4(4), 20. https://doi.org/10.3390/chemosensors4040020