Effect of Regular Taekwondo Self-Defense Training on Oxidative Stress and Inflammation Markers in Postmenopausal Women
Abstract
1. Introduction
2. Materials and Methods
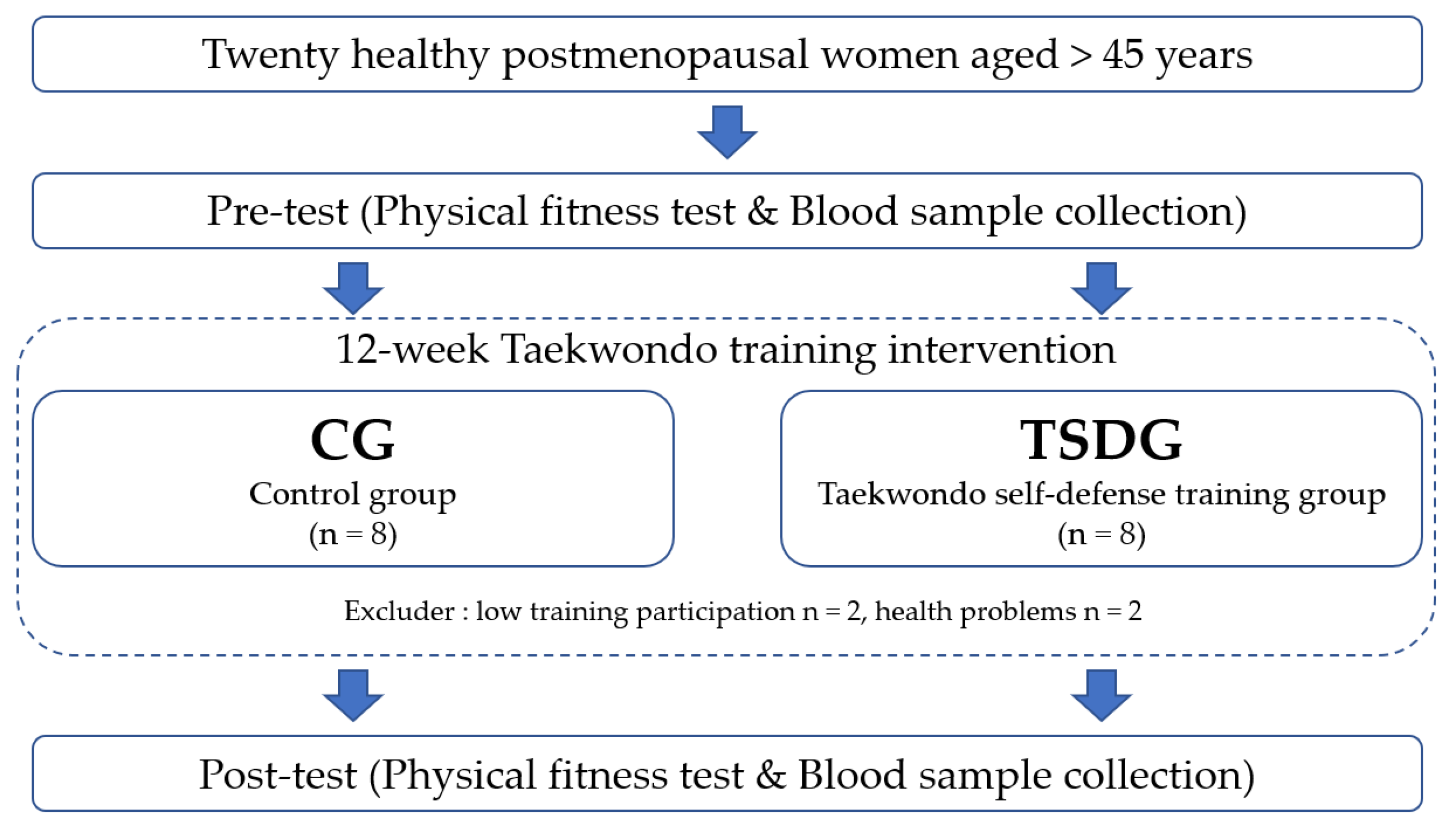
2.1. Subjects
2.2. Physical Fitness Test
2.3. Taekwondo Training
2.4. Blood Sample Analysis
2.5. Statistical Analyses
3. Results
3.1. Changes in Physical Fitness after 12 Weeks of Taekwondo Training Intervention
3.2. Changes in Oxidative Stress after 12 Weeks of Taekwondo Training Intervention
3.3. Changes in Chronic Inflammatory Factors after 12 Weeks of Taekwondo Training Intervention
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carr, M.C. The emergence of the metabolic syndrome with menopause. J. Clin. Endocrinol. Metab. 2003, 88, 2404–2411. [Google Scholar] [CrossRef] [PubMed]
- Lizcano, F.; Guzmán, G. Estrogen Deficiency and the Origin of Obesity during Menopause. Biomed. Res. Int. 2014, 2014, 757461. [Google Scholar] [CrossRef]
- Dai, Q.; Gao, Y.T.; Shu, X.O.; Yang, G.; Milne, G.; Cai, Q.; Wen, W.; Rothman, N.; Cai, H.; Li, H.; et al. Oxidative stress, obesity, and breast cancer risk: Results from the Shanghai Women’s Health Study. J. Clin. Oncol. 2009, 27, 2482–2488. [Google Scholar] [CrossRef] [PubMed]
- Naregal, G.V.; Devaranavadagi, B.B.; Patil, S.G.; Aski, B.S. Elevation of Oxidative Stress and Decline in Endogenous Antioxidant Defense in Elderly Individuals with Hypertension. J. Clin. Diagn. Res. 2017, 11, BC09–BC12. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, B. Teaching the basics of redox biology to medical and graduate students: Oxidants, antioxidants and disease mechanisms. Redox. Biol. 2013, 1, 244–257. [Google Scholar] [CrossRef]
- Poljsak, B.; Šuput, D.; Milisav, I. Achieving the balance between ROS and antioxidants: When to use the synthetic antioxidants. Oxid. Med. Cell Longev. 2013, 2013, 956792. [Google Scholar] [CrossRef] [PubMed]
- Magherini, F.; Fiaschi, T.; Marzocchini, R.; Mannelli, M.; Gamberi, T.; Modesti, P.A.; Modesti, A. Oxidative stress in exercise training: The involvement of inflammation and peripheral signals. Free Radic. Res. 2019, 53, 1155–1165. [Google Scholar] [CrossRef]
- Spirlandeli, A.L.; Deminice, R.; Jordao, A.A. Plasma malondialdehyde as biomarker of lipid peroxidation: Effects of acute exercise. Int. J. Sports Med. 2014, 35, 14–18. [Google Scholar] [CrossRef]
- Nocella, C.; Cammisotto, V.; Pigozzi, F.; Borrione, P.; Fossati, C.; D’Amico, A.; Cangemi, R.; Peruzzi, M.; Gobbi, G.; Ettorre, E.; et al. Impairment between oxidant and antioxidant systems: Short- and long-term implications for athletes’ health. Nutrients 2019, 11, 1353. [Google Scholar] [CrossRef]
- Yu, Y.; Gao, Q.; Xia, W.; Zhang, L.; Hu, Z.; Wu, X.; Jia, X. Association between Physical Exercise and Biomarkers of Oxidative Stress among Middle-Aged and Elderly Community Residents with Essential Hypertension in China. Biomed. Res. Int. 2018, 2018, 4135104. [Google Scholar] [CrossRef] [PubMed]
- World Taekwondo Academy. 2018 Taekwondo a White Paper on Education; Kukkuwon: Seoul, Korea, 2018. [Google Scholar]
- Shin, Y.S.; Yang, S.M.; Kim, M.Y.; Lee, L.K.; Park, B.S.; Lee, W.D.; Noh, J.W.; Kim, J.H.; Lee, J.U.; Kwak, T.Y.; et al. Differences in respirogram phase between taekwondo poomsae athletes and nonathletes. J. Phys. Ther. Sci. 2016, 28, 2556–2559. [Google Scholar] [CrossRef][Green Version]
- Pons van Dijk, G.; Lenssen, A.F.; Leffers, P.; Kingma, H.; Lodder, J. Taekwondo training improves balance in volunteers over 40. Front. Aging Neurosci. 2013, 5, 10. [Google Scholar] [CrossRef]
- Cho, S.Y.; Roh, H.T. Taekwondo Enhances Cognitive Function as a Result of Increased Neurotrophic Growth Factors in Elderly Women. Int. J. Environ. Res. Public Health 2019, 16, 962. [Google Scholar] [CrossRef]
- Lee, S.H.; Scott, S.D.; Pekas, E.J.; Lee, S.; Lee, S.H.; Park, S.Y. Taekwondo training reduces blood catecholamine levels and arterial stiffness in postmenopausal women with stage-2 hypertension: Randomized clinical trial. Clin. Exp. Hypertens. 2019, 41, 675–681. [Google Scholar] [CrossRef]
- Hollander, J.A. “I Can Take Care of Myself”: The Impact of Self-Defense Training on Women’s Lives. Violence Against Women 2004, 10, 205–235. [Google Scholar] [CrossRef]
- Bae, J.Y.; Jang, K.S.; Kang, S.; Han, D.H.; Yang, W.; Shin, K.O. Correlation between basic physical fitness and pulmonary function in Korean children and adolescents: A cross-sectional survey. J. Phys. Ther. Sci. 2015, 27, 2687–2692. [Google Scholar] [CrossRef] [PubMed]
- World Taekwondo Academy. The International Taekwondo Master Course (3rd Class) Textbook (Taekwondo Self-Defense Training); Kukkiwon: Seoul, Korea, 2018. [Google Scholar]
- Bae, J.Y.; Ok, D.P.; Park, J.S.; Choi, J.; Kim, J.K.; Kang, S. Brain function factors after high acceleration exposure in Korea Air Force cadets. Biomed. Res. 2018, 29, 11. [Google Scholar] [CrossRef]
- Valdés-Badilla, P.; Herrera-Valenzuela, T.; Ramirez-Campillo, R.; Aedo-Muñoz, E.; Báez-San Martín, E.; Ojeda-Aravena, A.; Branco, B.H.M. Effects of Olympic Combat Sports on Older Adults’ Health Status: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 7381. [Google Scholar] [CrossRef] [PubMed]
- Leenen, R.; van der Kooy, K.; Seidell, J.C.; Deurenberg, P.; Koppeschaar, H.P. Visceral fat accumulation in relation to sex hormones in obese men and women undergoing weight loss therapy. J. Clin. Endocrinol. Metab. 1994, 78, 1515–1520. [Google Scholar]
- Mesch, V.R.; Boero, L.E.; Siseles, N.O.; Royer, M.; Prada, M.; Sayegh, F.; Schreier, L.; Benencia, H.J.; Berg, G.A. Metabolic syndrome throughout the menopausal transition: Influence of age and menopausal status. Climacteric 2006, 9, 40–48. [Google Scholar] [CrossRef]
- Seo, D.I.; So, W.Y.; Ha, S.; Yoo, E.J.; Kim, D.; Singh, H.; Fahs, C.A.; Rossow, L.; Bemben, D.A.; Bemben, M.G.; et al. Effects of 12 weeks of combined exercise training on visfatin and metabolic syndrome factors in obese middle-aged women. J. Sports Sci. Med. 2011, 10, 222–226. [Google Scholar]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 8th ed.; Lippincott Williams & Wilkins: Baltimore, PA, USA, 2010. [Google Scholar]
- Dhaliwal, R.; Mikhail, M.; Usera, G.; Stolberg, A.; Islam, S.; Ragolia, L.; Aloia, J.F. The relationship of Physical performance and Osteoporosis prevention with vitamin D in older African Americans (PODA). Contemp. Clin. Trials 2018, 65, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Grindler, N.M.; Santoro, N.F. Menopause and exercise. Menopause 2015, 22, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Fong, S.S.; Ng, G.Y. Does Taekwondo training improve physical fitness? Phys. Ther. Sport. 2011, 12, 100–106. [Google Scholar] [CrossRef]
- Simioni, C.; Zauli, G.; Martelli, A.M.; Vitale, M.; Sacchetti, G.; Gonelli, A.; Neri, L.M. Oxidative stress: Role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 2018, 9, 17181–17198. [Google Scholar] [CrossRef]
- Incalza, M.A.; D’Oria, R.; Natalicchio, A.; Perrini, S.; Laviola, L.; Giorgino, F. Oxidative stress and reactive oxygen species in endothelial dysfunction associated with cardiovascular and metabolic diseases. Vascul. Pharmacol. 2018, 100, 1–19. [Google Scholar] [CrossRef]
- Wang, Y.; Branicky, R.; Noë, A.; Hekimi, S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J. Cell Biol. 2018, 217, 1915–1928. [Google Scholar] [CrossRef]
- He, F.; Li, J.; Liu, Z.; Chuang, C.C.; Yang, W.; Zuo, L. Redox Mechanism of Reactive Oxygen Species in Exercise. Front. Physiol. 2016, 7, 486. [Google Scholar] [CrossRef]
- Always, S.E.; McCrory, J.L.; Kearcher, K.; Vickers, A.; Frear, B.; Gilleland, D.L.; Bonner, D.E.; Thomas, J.M.; Donley, D.A.; Lively, M.W.; et al. Resveratrol Enhances Exercise-Induced Cellular and Functional Adaptations of Skeletal Muscle in Older Men and Women. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1595–1606. [Google Scholar] [CrossRef] [PubMed]
- Nan, S.N.; Kim, J.H.; Ji, M.C. Effect of Aerobic Exercise with Resistance Exercise Programs on Blood MDA and SOD, GPx Activities in Elderly Women. J. Korea Contents Assoc. 2009, 9, 391–398. [Google Scholar] [CrossRef]
- Palasuwan, A.; Suksom, D.; Margaritis, I.; Soogarun, S.; Rousseau, A.S. Effects of tai chi training on antioxidant capacity in pre- and postmenopausal women. J. Aging Res. 2011, 2011, 234696. [Google Scholar] [CrossRef] [PubMed]
- Roh, H.T.; Cho, S.Y.; So, W.Y. Effects of Regular Taekwondo Intervention on Oxidative Stress Biomarkers and Myokines in Overweight and Obese Adolescents. Int. J. Environ. Res. Public Health 2020, 17, 2505. [Google Scholar] [CrossRef]
- Ceci, R.; Duranti, G.; Di Filippo, E.S.; Bondi, D.; Verratti, V.; Doria, C.; Caporossi, D.; Sabatini, S.; Dimauro, I.; Pietrangelo, T. Endurance training improves plasma superoxide dismutase activity in healthy elderly. Mech. Ageing Dev. 2020, 185, 111190. [Google Scholar] [CrossRef] [PubMed]
- Popko, K.; Gorska, E.; Stelmaszczyk-Emmel, A.; Plywaczewski, R.; Stoklosa, A.; Gorecka, D.; Pyrzak, B.; Demkow, U. Proinflammatory cytokines Il-6 and TNF-α and the development of inflammation in obese subjects. Eur. J. Med. Res. 2010, 15, 120–122. [Google Scholar] [CrossRef] [PubMed]
- Arango Duque, G.; Descoteaux, A. Macrophage cytokines: Involvement in immunity and infectious diseases. Front. Immunol. 2014, 5, 491. [Google Scholar] [CrossRef] [PubMed]
- Bełtowski, J. Adiponectin and resistin--new hormones of white adipose tissue. Med. Sci. Monit. 2003, 9, RA55–RA61. [Google Scholar]
- Donges, C.E.; Duffield, R.; Drinkwater, E.J. Effects of resistance or aerobic exercise training on interleukin-6, C-reactive protein, and body composition. Med. Sci. Sports Exerc. 2010, 42, 304–313. [Google Scholar] [CrossRef]
- Mendham, A.E.; Donges, C.E.; Liberts, E.A.; Duffield, R. Effects of mode and intensity on the acute exercise-induced IL-6 and CRP responses in a sedentary, overweight population. Eur. J. Appl. Physiol. 2011, 111, 1035–1045. [Google Scholar] [CrossRef]
- Abd El-Kader, S.M.; Al-Jiffri, O.H. Impact of aerobic versus resisted exercise training on systemic inflammation biomarkers and quality of Life among obese post-menopausal women. Afr. Health Sci. 2019, 19, 2881–2891. [Google Scholar] [CrossRef]
| Variable | CG (n = 8) | TSDG (n = 8) | t | p |
|---|---|---|---|---|
| Age (year) | 50.25 ± 2.12 | 49.62 ± 4.03 | 0.388 | 0.706 |
| Height (cm) | 160.67 ± 6.12 | 158.98 ± 2.67 | 0.715 | 0.487 |
| Weight (kg) | 60.10 ± 6.70 | 58.92 ± 9.05 | 0.295 | 0.772 |
| BMI (kg/m2) | 23.05 ± 1.32 | 23.36 ± 3.89 | −0.215 | 0.835 |
| Fat mass (kg) | 19.46 ± 3.39 | 17.93 ± 5.90 | 0.633 | 0.537 |
| Percent fat (%) | 32.31 ± 4.02 | 29.76 ± 5.32 | 1.146 | 0.271 |
| LBM (kg) | 40.53 ± 4.67 | 40.98 ± 3.27 | −0.223 | 0.827 |
| Component | Manufacturer | Equipment |
|---|---|---|
| Body composition | InBody (Seoul, Korea) | InBody J05 |
| Lower back strength | Takei Scientific Instruments (Niigata, Japan) | T.K.K.5402 |
| Side-step | CASIO (Tokyo, Japan) | DPS2750 |
| Trunk flexion | Takei Scientific Instruments (Niigata, Japan) | T.K.K.5103 |
| Sargent jump | Takei Scientific Instruments (Niigata, Japan) | T.K.K.5406 |
| Taekwondo Self-Defense Training | ||
|---|---|---|
| Warm-up (10 min) | Static stretching | |
| Main exercise (40 min) | Taekwondo self-defense <Basic movement> five times each side, 1–2 min rest | (1) Chigi (Strike) (2) Jireugi (Punch) (3) Chagi (Kick) (4) Makgi (Block) |
| Taekwondo self-defense <Defense and counterattack with a kick> five times, 1–2 min rest | (1) Olgul Oreunbal Apdollyeo-Chagi (Right foot front spin kick to the face) (2) Olgul Oenbal Yeop-Chagi (Left foot side kick to the face) (3) Olgul Oreunbal Yeop-Chagi (Right foot side kick to the face) (4) Dwidola Yeop-Chagi (Turning side kick to the flank) (5) Apdollyeo-Chagi (Front foot spin kick to the face) (6) Ttwimyeo Yeop-Chagi(Jump and side kick to the flank) | |
| Taekwondo self-defense <one-step sparring> five times | (1~10) No.1~No.10 Gyeorugi (10 indicated movements) | |
| Cool-down (10 min) | Static stretching | |
| Variable | Baseline | 12 Weeks | Group × Times F p | ||
|---|---|---|---|---|---|
| Height (cm) | CG | 160.67 ± 6.12 | 160.95 ± 6.27 | 0.575 | 0.461 |
| TSDG | 158.98 ± 2.67 | 159.41 ± 2.74 | |||
| Weight (kg) | CG | 60.10 ± 6.70 | 60.26 ± 6.65 | 0.070 | 0.796 |
| TSDG | 58.92 ± 9.05 | 59.18 ± 9.19 | |||
| BMI (kg/m2) | CG | 23.05 ± 1.32 | 23.20 ± 1.34 | 0.068 | 0.798 |
| TSDG | 23.36 ± 3.89 | 23.34 ± 3.99 | |||
| Body fat (kg) | CG | 19.46 ± 3.39 | 19.55 ± 3.56 | 0.242 | 0.630 |
| TSDG | 17.93 ± 5.90 | 18.15 ± 6.03 | |||
| Percent fat (%) | CG | 32.31 ± 4.02 | 32.51 ± 4.36 | 0.230 | 0.639 |
| TSDG | 29.76 ± 5.32 | 28.38 ± 5.24 | |||
| LBM (kg) | CG | 40.53 ± 4.67 | 40.67 ± 4.81 | 0.083 | 0.778 |
| TSDG | 40.98 ± 3.27 | 41.03 ± 3.30 | |||
| Variable | Baseline | 12 Weeks | Group × Times F p | ||
|---|---|---|---|---|---|
| Lower back strength (kg) | CG | 44.35 ± 6.25 | 43.83 ± 5.94 | 0.864 | 0.368 |
| TSDG | 59.18 ± 22.09 | 62.06 ± 16.69 | |||
| Trunk flexion (cm) | CG | 19.70 ± 2.85 | 19.32 ± 2.83 | 0.020 | 0.890 |
| TSDG | 21.90 ± 5.30 | 21.47 ± 5.33 | |||
| Side-step (n) | CG | 5.37 ± 0.51 | 5.37 ± 0.51 | 9.333 | 0.009 * |
| TSDG | 7.37 ± 1.06 # | 8.37 ± 1.06 #,$ | |||
| Sargent jump (cm) | CG | 25.87 ± 6.19 | 24.62 ± 6.18 | 2.132 | 0.166 |
| TSDG | 26.75 ± 4.23 | 27.12 ± 4.32 | |||
| Variable | Baseline | 12 Weeks | Group × Times F p | ||
|---|---|---|---|---|---|
| MDA (nmol/mL) | CG | 2.21 ± 0.49 | 2.51 ± 0.87 | 18.222 | 0.001 * |
| TSDG | 2.60 ± 0.67 | 1.91 ± 0.34 $ | |||
| SOD (U/mL) | CG | 2.27 ± 0.81 | 2.04 ± 0.47 | 48.297 | 0.000 * |
| TSDG | 1.49 ± 0.34 | 3.42 ± 0.92 #,$ | |||
| GPx (μU/mL) | CG | 34.38 ± 11.70 | 33.86 ± 10.48 | 0.491 | 0.495 |
| TSDG | 36.35 ± 11.23 | 37.66 ± 10.86 | |||
| Variable | Baseline | 12 Weeks | Group × Times F p | ||
|---|---|---|---|---|---|
| TNF-α (pg/mL) | CG | 4.69 ± 0.58 | 4.79 ± 0.50 | 15.473 | 0.001 * |
| TSDG | 4.55 ± 0.65 | 4.09 ± 0.60 #,$ | |||
| IL-6 (pg/mL) | CG | 3.95 ± 0.76 | 4.10 ± 0.65 | 4.310 | 0.057 |
| TSDG | 4.02 ± 0.78 | 3.66 ± 0.42 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ku, B.-J.; Ko, K.; Shin, K.-O.; Bae, J.-Y. Effect of Regular Taekwondo Self-Defense Training on Oxidative Stress and Inflammation Markers in Postmenopausal Women. Healthcare 2021, 9, 985. https://doi.org/10.3390/healthcare9080985
Ku B-J, Ko K, Shin K-O, Bae J-Y. Effect of Regular Taekwondo Self-Defense Training on Oxidative Stress and Inflammation Markers in Postmenopausal Women. Healthcare. 2021; 9(8):985. https://doi.org/10.3390/healthcare9080985
Chicago/Turabian StyleKu, Beom-Jun, Kangeun Ko, Ki-Ok Shin, and Ju-Yong Bae. 2021. "Effect of Regular Taekwondo Self-Defense Training on Oxidative Stress and Inflammation Markers in Postmenopausal Women" Healthcare 9, no. 8: 985. https://doi.org/10.3390/healthcare9080985
APA StyleKu, B.-J., Ko, K., Shin, K.-O., & Bae, J.-Y. (2021). Effect of Regular Taekwondo Self-Defense Training on Oxidative Stress and Inflammation Markers in Postmenopausal Women. Healthcare, 9(8), 985. https://doi.org/10.3390/healthcare9080985






