Risk Factors of Lyme Disease: An Intersection of Environmental Ecology and Systems Science
Abstract
1. Introduction
2. Data
2.1. Vegetation Data
2.2. Tick-Rodent Data
2.3. Human Risk Data
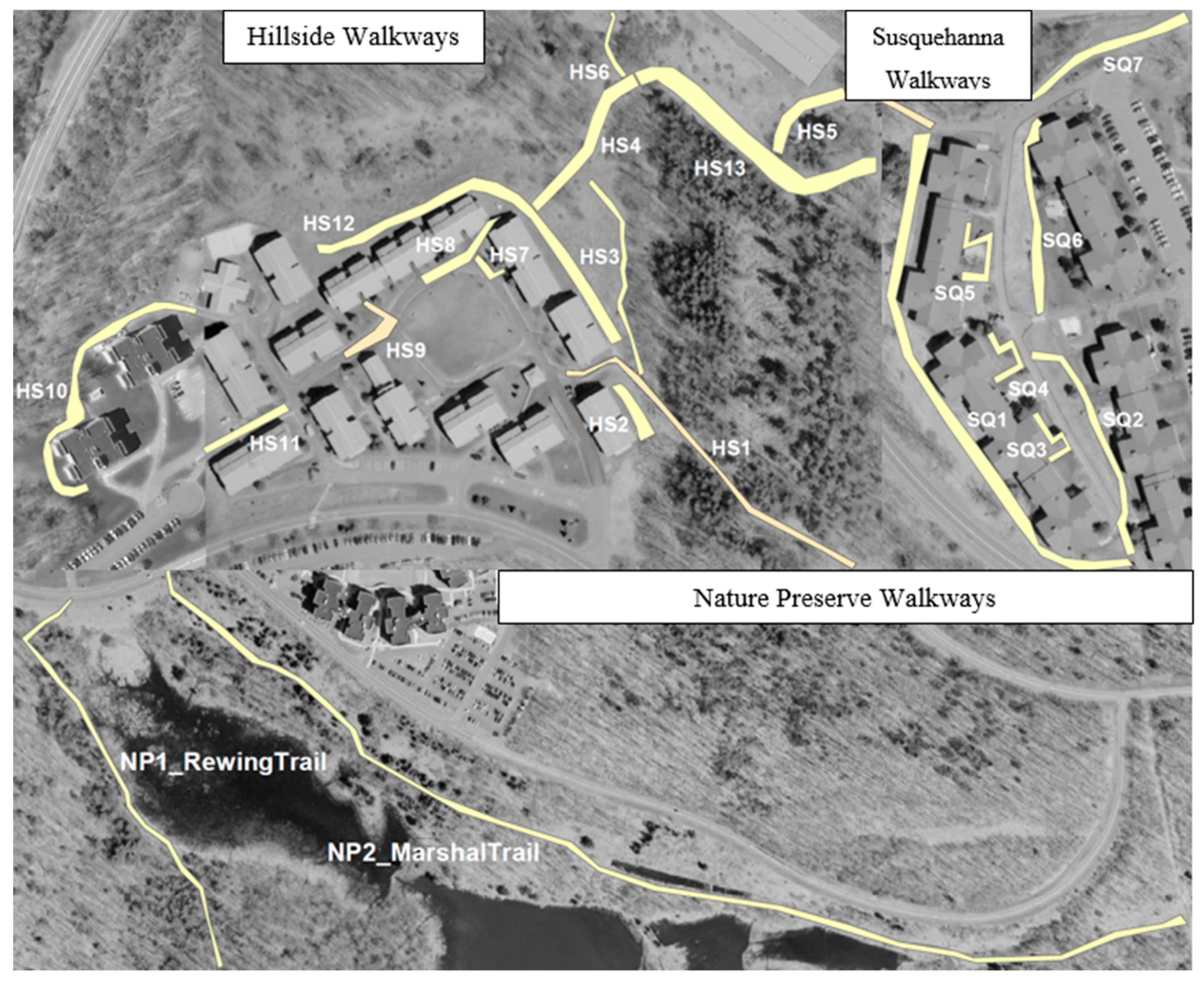
2.4. Walkway Data
3. Methods
Data Processing
4. Results and Discussion
Logistic Regression Model
5. Conclusions
6. Limitations
7. Software and/or Data Availability Section
8. Human Subject Research
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walter, K.S.; Carpi, G.; Caccone, A.; Diuk-Wasser, M.A. Genomic insights into the ancient spread of Lyme disease across North America. Nat. Ecol. Evol. 2017, 1, 1569. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, A.; Hauffe, H.C.; Carpi, G.; Vourc’h, G.; Neteler, M.; Rosa, R. Lyme borreliosis in Europe. Eurosurveillance 2011, 16, 19906. [Google Scholar] [PubMed]
- Hofhuis, A.; Harms, M.; van den Wijngaard, C.; Sprong, H.; van Pelt, W. Continuing increase of tick bites and Lyme disease between 1994 and 2009. Ticks Tick Borne Dis. 2015, 6, 69–74. [Google Scholar] [CrossRef]
- Masuzawa, T. Terrestrial distribution of the Lyme borreliosis agent Borrelia burgdorferi sensu lato in East Asia. JPN J. Infect. Dis. 2004, 57, 229–235. [Google Scholar] [PubMed]
- Robinson, S.J.; Neitzel, D.F.; Moen, R.A.; Craft, M.E.; Hamilton, K.E.; Johnson, L.B.; Mulla, D.J.; Munderloh, U.G.; Redig, P.T.; Smith, K.E.; et al. Disease risk in a dynamic environment: The spread of tick-borne pathogens in Minnesota, USA. EcoHealth 2015, 12, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.; Tran, L. Validating negative binomial lyme disease regression model with bootstrap resampling. Environ. Modelling Softw. 2016, 82, 121–127. [Google Scholar] [CrossRef]
- Adrion, E.R.; Aucott, J.; Lemke, K.W.; Weiner, J.P. Health care costs, utilization and patterns of care following Lyme disease. PloS ONE 2015, 10, e0116767. [Google Scholar] [CrossRef]
- JHSPH. Lyme Disease Costs Up to $1.3 Billion Per Year to Treat, Study Finds. Available online: https://www.jhsph.edu/news/news-releases/2015/lyme-disease-costs-more-than-one-billion-dollars-per-year-to-treat-study-finds.html (accessed on 20 April 2019).
- Gubler, D.J. Resurgent vector-borne diseases as a global health problem. Emerg. Infect. Dis. 1998, 4, 442. [Google Scholar] [CrossRef]
- Johnson, R.C.; Schmid, G.P.; Hyde, F.W.; Steigerwalt, A.; Brenner, D.J. Borrelia burgdorferi sp. nov.: Etiologic agent of Lyme disease. Int. J. Syst. Evol. Microbiol. 1984, 34, 496–497. [Google Scholar] [CrossRef]
- Spielman, A.; Levine, J.F.; Wilson, M. Vectorial capacity of North American Ixodes ticks. Yale j. Biol. Med. 1984, 57, 507–513. [Google Scholar]
- Wormser, G.P.; Dattwyler, R.J.; Shapiro, E.D.; Halperin, J.J.; Steere, A.C.; Klempner, M.S.; Krause, P.J.; Bakken, J.S.; Strle, F.; Bockenstedt, G.S.L.; et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: Clinical practice guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 2006, 43, 1089–1134. [Google Scholar] [CrossRef]
- Feder, H.M., Jr.; Johnson, B.J.; O’connell, S.; Shapiro, E.D.; Steere, A.C.; Wormser, G.P. A critical appraisal of “chronic Lyme disease”. N. Eng. J. Med. 2007, 357, 1422–1430. [Google Scholar] [CrossRef]
- Kugeler, K.J.; Farley, G.M.; Forrester, J.D.; Mead, P.S. Geographic distribution and expansion of human Lyme Disease, United States. Emerg. Infect. Dis. 2015, 21, 1455–1457. [Google Scholar] [CrossRef]
- Lantos, P.M.; Tsao, J.; Nigrovic, L.E.; Auwaerter, P.G.; Fowler, V.G.; Ruffin, F.; Foster, E.; Hickling, G. Geographic expansion of Lyme disease in Michigan, 2000–2014. In Open Forum Infectious Diseases; Oxford University Press: Oxford, UK, 2017. [Google Scholar]
- Seukep, S.E.; Kolivras, K.N.; Hong, Y.; Li, J.; Prisley, S.P.; Campbell, J.B.; Gaines, D.N.; Dymond, R.L. An examination of the demographic and environmental variables correlated with Lyme disease emergence in Virginia. Ecohealth 2015, 12, 634–644. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.A.; Marrotte, R.R.; Desrosiers, N.; Fiset, J.; Gaitan, J.; Gonzalez, A.; Koffi, J.K.; Lapointe, F.J.; Leighton, P.A.; Limdsay, L.R.; et al. Climate change and habitat fragmentation drive the occurrence of B orrelia burgdorferi, the agent of Lyme disease, at the northeastern limit of its distribution. Evol. Appl. 2014, 7, 750–764. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.; Malawista, S.; Snydman, D.; Shope, R.E.; Andiman, W.A.; Ross, M.R.; Steele, F.M. Lyme arthritis: An epidemic of oligoarticular arthritis in children and adults in three Connecticut communities. Arthritis Reum. 1977, 20, 7–17. [Google Scholar] [CrossRef]
- Wood, C.L.; Lafferty, K.D. Biodiversity and disease: A synthesis of ecological perspectives on Lyme disease transmission. Trends Ecol. Evol. 2013, 28, 239–247. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Ogden, N.H.; Beard, C.B. Linkages of weather and climate with Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae), enzootic transmission of Borrelia burgdorferi, and Lyme disease in North America. J. Med. Entomol. 2015, 53, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Hamer, S.A.; Hickling, G.J.; Sidge, J.L.; Walker, E.D.; Tsao, J.I. Synchronous phenology of juvenile Ixodes scapularis, vertebrate host relationships, and associated patterns of Borrelia burgdorferi ribotypes in the midwestern United States. Ticks Tick Borne Dis. 2012, 3, 65–74. [Google Scholar] [CrossRef]
- CDC. Life cycle of Hard Ticks that Spread Disease. Available online: https://www.cdc.gov/ticks/life_cycle_and_hosts.html (accessed on 20 April 2018).
- Brownstein, J.S.; Holford, T.R.; Fish, D. Effect of climate change on Lyme disease risk in North America. EcoHealth 2005, 2, 38–46. [Google Scholar] [CrossRef]
- Killilea, M.E.; Swei, A.; Lane, R.S.; Briggs, C.J.; Ostfeld, R.S. Spatial dynamics of Lyme disease: A review. EcoHealth 2008, 5, 167–195. [Google Scholar] [CrossRef]
- Li, S.; Gilbert, L.; Harrison, P.A.; Rounsevell, M.D. Modelling the seasonality of Lyme disease risk and the potential impacts of a warming climate within the heterogeneous landscapes of Scotland. J. R. Soc. Interface 2016, 13, 20160140. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.; Bjork, J.; Neitzel, D.; Dorr, F.; Schiffman, E.; Eisen, R. Habitat suitability model for the distribution of Ixodes scapularis (Acari: Ixodidae) in Minnesota. J. Med. Entomol. 2016, 53, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, A.M.; Dobson, A.D.; Levi, T.; Salkeld, D.J.; Swei, A.; Ginsberg, H.S.; Kjemtrup, A.; Padgett, K.A.; Jensen, P.M.; Fish, D.; et al. Lyme disease ecology in a changing world: consensus, uncertainty and critical gaps for improving control. Phil. Trans. R. Soc. B 2017, 372, 20160117. [Google Scholar] [CrossRef]
- McClure, M.; Diuk-Wasser, M. Reconciling the Entomological Hazard and Disease Risk in the Lyme Disease System. IJERPH 2018, 15, 1048. [Google Scholar] [CrossRef] [PubMed]
- Gabriele-Rivet, V.; Koffi, J.K.; Pelcat, Y.; Atsenault, J.; Cheng, A.; Lindsay, L.R.; Lysyk, T.J.; Rochon, K.; Ogden, N.H. A risk model for the Lyme disease vector Ixodes scapularis (Acari: Ixodidae) in the prairie provinces of Canada. J. Med. Entomol. 2017, 54, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Ripoche, M.; Lindsay, L.R.; Ludwig, A.; Ogden, N.H.; Thivierge, K.; Leighton, P.A. Multi-Scale Clustering of Lyme Disease Risk at the Expanding Leading Edge of the Range of Ixodes scapularis in Canada. IJERPH 2018, 15, 603. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.E.; Hilborn, E.D.; Thomas, J.C. Towards landscape design guidelines for reducing Lyme disease risk. Int. J. Epidemiol. 2006, 35, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; O’fallon, L.R.; Dearry, A. Creating healthy communities, healthy homes, healthy people: Initiating a research agenda on the built environment and public health. Am. J. Public Health 2003, 93, 1446–1450. [Google Scholar] [CrossRef]
- Sharareh, N.; Sabounchi, N.S.; Roome, A.; Spathis, R.; Garruto, R.M. Model-based risk assessment and public health analysis to prevent Lyme disease. R. Soc. Open Sci. 2017, 4, 170841. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, S. Peromyscus leucopus_white-footed mouse. Available online: http://animaldiversity.org/accounts/Peromyscus_leucopus/ (accessed on 20 April 2019).
- Böhning, D. Multinomial logistic regression algorithm. Ann. Inst. Stat. Math. 1992, 44, 197–200. [Google Scholar] [CrossRef]
- Lorenz, A.; Dhingra, R.; Chang, H.H.; Bisanzio, D.; Liu, Y.; Remais, J.V. Inter-model comparison of the landscape determinants of vector-borne disease: implications for epidemiological and entomological risk modeling. PloS ONE 2014, 9, e103163. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J. Using geographic information systems and decision support systems for the prediction, prevention, and control of vector-borne diseases. Ann. Rev. Entomol. 2011, 56, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Stoltzfus, J.C. Logistic regression: a brief primer. Acad. Emerg. Med. 2011, 18, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- StatisticsSolutions. Assumptions of Logistic Regression. Available online: https://www.statisticssolutions.com/assumptions-of-logistic-regression/ (accessed on 20 April 2019).
- Dishaw. To standardize data or not to standardize data—That is the question. Available online: https://communities.sas.com/t5/SAS-Communities-Library/To-standardize-data-or-not-to-standardize-data-that-is-the/ta-p/361726 (accessed on 20 April 2019).
- Roome, A.; Spathis, R.; Garruto, R.M. Density of Ticks in Household Backyards—A Case Study at Binghamton University. unpublished data. 2018. [Google Scholar]
- Roome, A.; Hill, L.; Al-feghali, V.; Murnock, C.G.; Goodsell, J.A.; Spathis, R.; Garruto, R.M. Impact of white-tailed deer on the spread of B orrelia burgdorferi. Med. Vet. Entomol. 2017, 31, 1–5. [Google Scholar] [CrossRef] [PubMed]
| Walkway ** | Simulated LD Cases in 2014 | Walkway Length (meter) | Walkway Area (meter2) | Canopy (%) | Understory (%) | Ground Cover (%) | Density of Larvae Ticks | Density of Nymph Ticks | Density of Adult Ticks | Density of Infected Ticks | Number of Rodents | Human Risk | Type of Walkway *** | Number of Passersby |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HS1 | 0.0023 | 251.2 | 7787.2 | 50 | 10 | 84.75 | 0.0 | 0 | 10.7 | 7.705 | 75 | 0.865 | NO | 1690 |
| HS2 | 6.2567 | 51.5 | 103 | 0 | 5 | 45 | 0.0 | 0 | 126.2 | 87.379 | 15 | 1 | O | 208 |
| HS3 | 1.6712 | 111.9 | 2014.2 | 26 | 5 | 100 | 0.0 | 0 | 11.4 | 6.454 | 34 | 0.991 | O | 50 |
| HS4 | 3.9596 | 73.5 | 441 | 40 | 50 | 67.5 | 0.0 | 0 | 4.5 | 2.268 | 22 | 0.992 | O | 119 |
| HS5 | 4.1989 | 44.2 | 699.8 | 27.5 | 6.25 | 70 | 0.0 | 0 | 1.4 | 1.429 | 13 | 1 | O | 123 |
| HS6 | 1.1354 | 44.8 | 627.2 | 55 | 87.5 | 15 | 4.8 | 41.5 | 3.2 | 14.349 | 13 | 1 | O | 26 |
| HS7 | 0.0060 | 24.7 | 148.2 | 35 | 0 | 100 | 0 | 0 | 6.7 | 6.748 | 7 | 0.678 | NO | 649 |
| HS8 | 0.0007 | 17.7 | 106.2 | 30 | 0 | 100 | 0 | 0 | 0 | 0 | 5 | 0.489 | NO | 574 |
| HS9 | 0.0005 | 42.7 | 256.2 | 20 | 0 | 100 | 0 | 0 | 0 | 0 | 13 | 0.400 | NO | 800 |
| HS10 | 0.00006 | 148.7 | 2676.6 | 7.5 | 12.5 | 55 | 0 | 0 | 2.2 | 0.374 | 45 | 0.562 | O | 126 |
| HS11 | 0.00007 | 60.35 | 724.2 | 14.75 | 0 | 30 | 0 | 0 | 0 | 0 | 18 | 0.757 | NO | 573 |
| HS12 | 0.00009 | 215.5 | 1939.5 | 2.5 | 5 | 97.5 | 0 | 0 | 2.1 | 0.516 | 65 | 0.983 | O | 118 |
| HS13 | 0.00006 | 154.5 | 4480.17 | 47.5 | 35 | 53.75 | 0 | 0.7 | 19.2 | 12.5 | 46 | 1 | O | 60 |
| NP-MT | 0 | 1134.8 | 26100.4 | 51.5 | 52.5 | 75 | 0 | 0 | 0.3 | 0.115 | 340 | 1 | O | 639 |
| NP-RW | 0.0004 | 548.6 | 3291.6 | 95 | 10 | 56 | 0 | 0 | 0.6 | 0.304 | 165 | 0.999 | O | 168 |
| SQ1 | 0.00003 | 296.08 | 2072.56 | 20 | 15 | 100 | 0 | 2.9 | 0 | 0.482 | 89 | 0.824 | O | 25 |
| SQ2 | 0.0002 | 120.31 | 721.86 | 5 | 6.75 | 97.5 | 0 | 0 | 2.8 | 0 | 36 | 0.646 | NO | 213 |
| SQ3 | 0.0006 | 34.58 | 69.16 | 30 | 5 | 100 | 0 | 0 | 0 | 0 | 10 | 0.729 | NO | 391 |
| SQ4 | 0.0007 | 41.47 | 82.94 | 5 | 0 | 95 | 0 | 0 | 0 | 0 | 12 | 0.697 | NO | 577 |
| SQ5 | 0.0007 | 53.18 | 106.36 | 20 | 0 | 80 | 0 | 0 | 0 | 0 | 16 | 0.755 | NO | 588 |
| SQ6 | 0.0003 | 38.5 | 462 | 7.5 | 17.5 | 80 | 0 | 0 | 6.5 | 0 | 12 | 0.527 | NO | 341 |
| SQ7 | 1.3754 | 45.03 | 495.33 | 3.75 | 35 | 65 | 0 | 0 | 22.2 | 8.075 | 14 | 0.573 | NO | 1295 |
| Walkway ** | Infected Tick Risk (Dependent Variable) | Location | Simulated LD Cases in 2014 | Canopy (%) | Understory (%) | Ground Cover (%) | Number of Rodents | Human Risk | Number of Passersby |
|---|---|---|---|---|---|---|---|---|---|
| HS1 | H | 1 | −0.488 | 1.001 | −0.279 | 0.358 | 0.355 | 0.353 | 2.967 |
| HS2 | H | 1 | 3.131 | −1.173 | −0.502 | −1.227 | −0.446 | 1.026 | −0.510 |
| HS3 | H | 1 | 0.477 | −0.042 | −0.502 | 0.966 | −0.192 | 0.981 | −0.880 |
| HS4 | H | 1 | 1.802 | 0.566 | 1.501 | −0.330 | −0.352 | 0.986 | −0.718 |
| HS5 | H | 1 | 1.94 | 0.023 | −0.446 | −0.230 | −0.472 | 1.026 | −0.709 |
| HS6 | H | 1 | 0.167 | 1.219 | 3.169 | −2.423 | −0.472 | 1.026 | −0.935 |
| HS7 | H | 1 | −0.486 | 0.349 | −0.724 | 0.966 | −0.552 | −0.576 | 0.525 |
| HS8 | L | 1 | −0.489 | 0.131 | −0.724 | 0.966 | −0.579 | −1.519 | 0.350 |
| HS9 | L | 1 | −0.489 | −0.303 | −0.724 | 0.966 | −0.472 | −1.960 | 0.879 |
| HS10 | L | 1 | −0.489 | −0.847 | −0.168 | −0.828 | −0.045 | −1.154 | −0.702 |
| HS11 | L | 1 | −0.489 | −0.532 | −0.724 | −1.825 | −0.406 | −0.183 | 0.347 |
| HS12 | H | 1 | −0.489 | −1.064 | −0.502 | 0.866 | 0.221 | 0.940 | −0.720 |
| HS13 | H | 1 | −0.489 | 0.892 | 0.833 | −0.878 | −0.032 | 1.026 | −0.858 |
| NP-MT | L | 2 | −0.489 | 1.066 | 1.612 | −0.031 | 3.888 | 1.026 | 0.502 |
| NP-RW | L | 2 | −0.489 | 2.958 | −0.279 | −0.788 | 1.555 | 1.019 | −0.603 |
| SQ1 | H | 3 | −0.489 | −0.303 | −0.057 | 0.966 | 0.541 | 0.148 | −0.939 |
| SQ2 | L | 3 | −0.489 | −0.956 | −0.424 | 0.866 | −0.165 | −0.737 | −0.497 |
| SQ3 | L | 3 | −0.489 | 0.131 | −0.502 | 0.966 | −0.512 | −0.323 | −0.081 |
| SQ4 | L | 3 | −0.489 | −0.956 | −0.724 | 0.767 | −0.486 | −0.482 | 0.355 |
| SQ5 | L | 3 | −0.489 | −0.303 | −0.724 | 0.169 | −0.432 | −0.193 | 0.381 |
| SQ6 | L | 3 | −0.489 | −0.847 | 0.055 | 0.169 | −0.486 | −1.329 | −0.197 |
| SQ7 | H | 3 | 0.306 | −1.010 | 0.833 | −0.429 | −0.459 | −1.098 | 2.042 |
| Significant Indicator (High-Risk Walkways) | Coefficient Estimates | Standard Errors | p-Value |
|---|---|---|---|
| % of Understory | 5.9 | 3.84 | 0.009 |
| % of Human Risk | 5.388 | 3 | 0.000 |
| Number of Rodents | −5.108 | 2.86 | 0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharareh, N.; Behler, R.P.; Roome, A.B.; Shepherd, J.; Garruto, R.M.; Sabounchi, N.S. Risk Factors of Lyme Disease: An Intersection of Environmental Ecology and Systems Science. Healthcare 2019, 7, 66. https://doi.org/10.3390/healthcare7020066
Sharareh N, Behler RP, Roome AB, Shepherd J, Garruto RM, Sabounchi NS. Risk Factors of Lyme Disease: An Intersection of Environmental Ecology and Systems Science. Healthcare. 2019; 7(2):66. https://doi.org/10.3390/healthcare7020066
Chicago/Turabian StyleSharareh, Nasser, Rachael P. Behler, Amanda B. Roome, Julian Shepherd, Ralph M. Garruto, and Nasim S. Sabounchi. 2019. "Risk Factors of Lyme Disease: An Intersection of Environmental Ecology and Systems Science" Healthcare 7, no. 2: 66. https://doi.org/10.3390/healthcare7020066
APA StyleSharareh, N., Behler, R. P., Roome, A. B., Shepherd, J., Garruto, R. M., & Sabounchi, N. S. (2019). Risk Factors of Lyme Disease: An Intersection of Environmental Ecology and Systems Science. Healthcare, 7(2), 66. https://doi.org/10.3390/healthcare7020066






