Impact of Previous Physical Activity Levels on Symptomatology, Functionality, and Strength during an Acute Exacerbation in COPD Patients
Abstract
1. Introduction
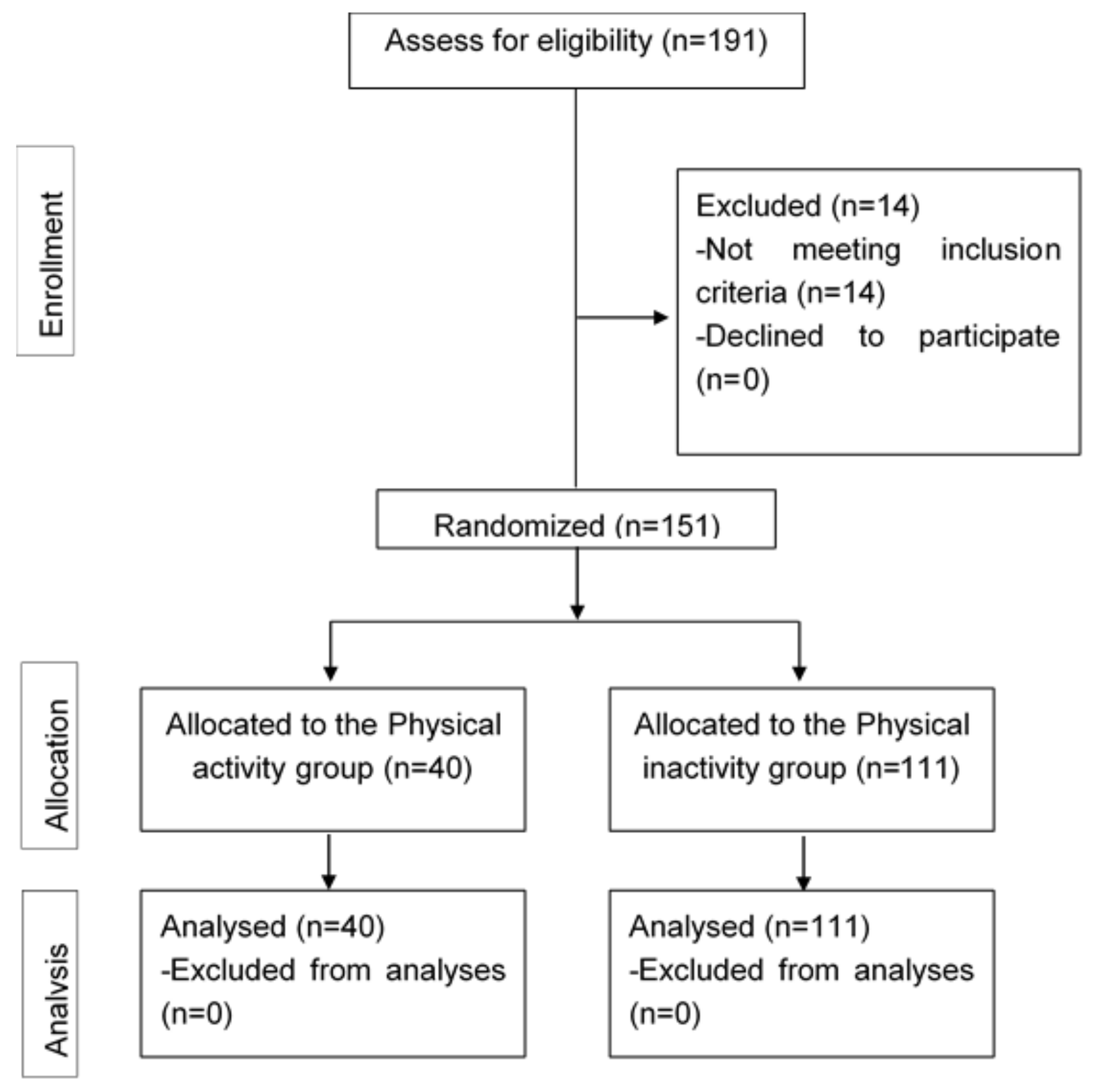
2. Materials and Methods
2.1. Participants
2.2. Measurements
2.3. Statistical Analysis
3. Results
4. Discussion
4.1. Pulmonary Symptoms
4.2. Extrapulmonary Symptoms
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Frith, P. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report. GOLD executive summary. Am. J. Respir. Crit. Care Med. 2017, 195, 557–582. [Google Scholar] [CrossRef] [PubMed]
- Wedzicha, J.A.; Brillm, S.E.; Allinson, J.P.; Donaldson, G.C. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.R.; Vestbo, J.; Anzueto, A.; Locantore, N.; Mullerova, H.; Tal-Singer, R.; Calverley, P. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N. Engl. J. Med. 2010, 363, 1128–1138. [Google Scholar] [CrossRef] [PubMed]
- Esteban, C.; Quintana, J.M.; Moraza, J.; Aburto, E.M.; Espana, P.P.; Perez-Izquierdo, J.; Aguirre, U.; Aizpiri, S.; Capelastegui, A. Impact of hospitalisations for exacerbations of COPD on health-related quality of life. Respir. Med. 2009, 103, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Celli, B.R.; MacNee, W. ATS/ERS Task Force. Standards for the diagnosis and treatment of patients with COPD: A summary of the ATS/ERS position paper. Eur. Respir. J. 2004, 23, 932–946. [Google Scholar] [CrossRef] [PubMed]
- Gea, J.; Casadevall, C.; Pascual, S.; Orozco-Levi, M.; Barreiro, E. Respiratory diseases and muscle dysfunction. Expert. Rev. Respir. Med. 2012, 6, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Schonhofer, B.; Ardes, P.; Geibel, M.; Kohler, D.; Jones, P.W. Evaluation of a movement detector to measure daily activity in patients with chronic lung disease. Eur. Respir. J. 1997, 10, 2814–2819. [Google Scholar] [CrossRef] [PubMed]
- Royo, M.M.; Císcar, C.P.; Villaescusa, C.G.; Fabra, M.J.B.; Benito, C.A.; Rodríguez, A.L.A.; Cataluña, J.J.S. Actividad física y su relación con el estado de salud en pacientes EPOC estables. Arch. Bronconeumol. 2011, 47, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Pleguezuelos, E.; Esquinas, C.; Moreno, E.; Guirao, L.; Ortiz, J.; Garcia-Alsina, J.; Miravitlles, M. Muscular dysfunction in COPD: Systemic effect or deconditioning? Lung 2016, 194, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Swallow, E.B.; Reyes, D.; Hopkinson, N.S.; Man, W.D.; Porcher, R.; Cetti, E.J.; Moore, A.J.; Moxham, J.; Polkey, M.I. Quadriceps strength predicts mortality in patients with moderate to severe chronic obstructive pulmonary disease. Thorax 2007, 62, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Nici, L.; Donner, C.; Wouters, E.; Zuwallack, R.; Ambrosino, N.; Bourbeau, J.; Garvey, C. American thoracic society/european respiratory society statement on pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2006, 173, 1390–1413. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aymerich, J.; Lange, P.; Benet, M.; Schnohr, P.; Antó, J.M. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: A population based cohort study. Thorax 2006, 61, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aymerich, J.; Lange, P.; Benet, M.; Schnohr, P.; Anto, J.M. Regular physical activity modifies smoking-related lung function decline and reduces risk of chronic obstructive pulmonary disease: A population-based cohort study. Am. J. Respir. Crit. Care Med. 2007, 175, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Vestbo, J.; Hurd, S.S.; Agustí, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Rodriguez-Roisin, R. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: Gold executive summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Qaseem, A.; Wilt, T.J.; Weinberger, S.E.; Hanania, N.A.; Criner, G.; Van der Molen, T.; MacDonald, R. Diagnosis and management of stable chronic obstructive pulmonary disease: A clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann. Intern. Med. 2011, 155, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Vilaró, J.; Gimeno, E.; Sánchez-Férez, N.; Hernando, C.; Díaz, I.; Ferrerc, M.; Roca, J.; Alonso, J. Daily living activity in chronic obstructive pulmonary disease: Validation of the Spanish version and comparative analysis of 2 questionnaires. Med. Clin. 2007, 129, 326–332. [Google Scholar] [CrossRef]
- Bestall, J.C.; Paul, E.A.; Garrod, R.; Garnham, R.; Jones, P.W.; Wedzicha, J.A. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax 1999, 54, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Berkhof, F.F.; Boom, L.N.; ten Hertog, N.E.; Uil, S.M.; Kerstjens, H.A.; van den Berg, J.W. The validity and precision of the Leicester Cough Questionnaire in COPD patients with chronic cough. Health Qual. Life Outcomes 2012, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Garrod, R.; Bestall, J.C.; Paul, E.A.; Wedzicha, J.A.; Jones, P.W. Development and validation of a standardized measure of activity of daily loving in patients with severe COPD: The London Chest Activity of Daily Living scale (LCADL). Respir. Med. 2000, 94, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Keith, R.A.; Granger, C.V.; Hamilton, B.B.; Sherwin, F.S. The functional Independence measure: A New tool for rehabilitation. Advances in clinical rehabilitation. Adv. Clin. Rehabil. 1987, 1, 6–18. [Google Scholar] [PubMed]
- Jones, C.J.; Rikli, R.E. Measuring functional fitness of older adults. J. Active Aging 2002, 24–30. [Google Scholar]
- Martin, H.J.; Yule, V.; Syddall, H.E.; Dennison, E.M.; Cooper, C.; Aihie–Sayer, A. Is Hand-Held dynamometry useful for the measurement of quadriceps strength in older people? A comparison with the gold standard biodex dynamometry. Gerontology 2006, 52, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Waschki, B.; Kirsten, A.; Holz, O.; Müller, K.C.; Meyer, T.; Watz, H.; Magnussen, H. Physical activity is the strongest predictor of all-cause mortality in patients with COPD: A Prospective cohort study. Chest 2011, 140, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rio, F.; Rojo, B.; Casitas, R.; Lores, V.; Madero, R.; Romero, D.; Galera, R.; Villasante, C. Prognostic value of the objective measurement of daily physical activity in patients with COPD. Chest 2012, 142, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Pitta, F.; Troosters, T.; Probst, V.S.; Spruit, M.A.; Decramer, M.; Gosselink, R. Physical activity and hospitalization for exacerbation of COPD. Chest 2006, 129, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aymerich, J.; Farrero, E.; Felez, M.A.; Izquierdo, J.; Marrades, R.M.; Anto, J.M. Risk factors of readmission to hospital for a COPD exacerbation: A prospective study. Thorax 2003, 58, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Patessio, A.; Casaburi, R.; Prefaut, C.; Folgering, H.; Donner, C. Exercise training in chronic lung disease: Exercise prescription. Eur. Respir. Rev. 1996, 2, 129–146. [Google Scholar]
- Van Buul, A.R.; Kasteleyn, M.J.; Chavannes, N.H.; Taube, C. The association between objectively measured physical activity and morning symptoms in COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 2831. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, M.; Okura, K.; Shibata, K.; Kawagoshi, A.; Sugawara, K.; Takahashi, H.; Shioya, T. Relationship between balance and physical activity measured by an activity monitor in elderly COPD patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 1505. [Google Scholar] [CrossRef] [PubMed]
| Variables | PA Group (n = 40) | PI Group (n = 111) | p-Value |
|---|---|---|---|
| Age (years) | 71.85 ± 7.65 | 71.52 ± 10.12 | 0.853 |
| BMI (kg/m2) | 31.73 ± 5.36 | 27.80 ± 5.13 | <0.001 ** |
| FVC predicted | 50.52 ± 18.34 | 48.82 ± 20.46 | 0.662 |
| FEV1 predicted | 40.43 ± 17.50 | 34.66 ± 17.52 | 0.093 |
| Hospital length of stay (days) | 8.88 ± 1.72 | 9.72 ± 4.26 | 0.355 |
| Charlson | 5.88 ± 1.88 | 4.99 ± 1.83 | 0.019 * |
| SGRQ | 51.40 ± 13.79 | 64.49 ± 12.39 | <0.001 ** |
| Variables | PA Group (n = 40) | PI Group (n = 111) | p-Value |
|---|---|---|---|
| Pulmonary symptoms | - | - | - |
| Dyspnea | - | - | - |
| mMRC | 2.75 ± 0.98 | 3.36 ± 0.92 | 0.010 * |
| Cough | - | - | - |
| LCQ | 16.47 ± 2.56 | 14.00 ± 3.71 | 0.002 * |
| Extrapulmonary symptoms | - | - | - |
| Functionality | - | - | - |
| LCADL | 18.69 ± 6.61 | 29.69 ± 15.06 | 0.013 * |
| FIM | 130.62 ± 5.91 | 111.63 ± 19.02 | 0.001 * |
| Exercise capacity | - | - | - |
| 2MSP | 43.33 ± 22.61 | 29.75 ± 23.44 | 0.016 * |
| Strength | - | - | - |
| Lower limb strength (Newton) | 156.21 ± 75.01 | 103.97 ± 34.79 | <0.001 ** |
| Upper limb strength (Newton) | 295.13 ± 68.01 | 227.74 ± 101.56 | <0.001 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-López, L.; Torres-Sánchez, I.; Romero-Fernández, R.; Granados-Santiago, M.; Rodríguez-Torres, J.; Valenza, M.C. Impact of Previous Physical Activity Levels on Symptomatology, Functionality, and Strength during an Acute Exacerbation in COPD Patients. Healthcare 2018, 6, 139. https://doi.org/10.3390/healthcare6040139
López-López L, Torres-Sánchez I, Romero-Fernández R, Granados-Santiago M, Rodríguez-Torres J, Valenza MC. Impact of Previous Physical Activity Levels on Symptomatology, Functionality, and Strength during an Acute Exacerbation in COPD Patients. Healthcare. 2018; 6(4):139. https://doi.org/10.3390/healthcare6040139
Chicago/Turabian StyleLópez-López, Laura, Irene Torres-Sánchez, Ramón Romero-Fernández, María Granados-Santiago, Janet Rodríguez-Torres, and Marie Carmen Valenza. 2018. "Impact of Previous Physical Activity Levels on Symptomatology, Functionality, and Strength during an Acute Exacerbation in COPD Patients" Healthcare 6, no. 4: 139. https://doi.org/10.3390/healthcare6040139
APA StyleLópez-López, L., Torres-Sánchez, I., Romero-Fernández, R., Granados-Santiago, M., Rodríguez-Torres, J., & Valenza, M. C. (2018). Impact of Previous Physical Activity Levels on Symptomatology, Functionality, and Strength during an Acute Exacerbation in COPD Patients. Healthcare, 6(4), 139. https://doi.org/10.3390/healthcare6040139






