Nonlocal Reaction–Diffusion Model of Viral Evolution: Emergence of Virus Strains
Abstract
1. Introduction
2. Bifurcations of Periodic Structures
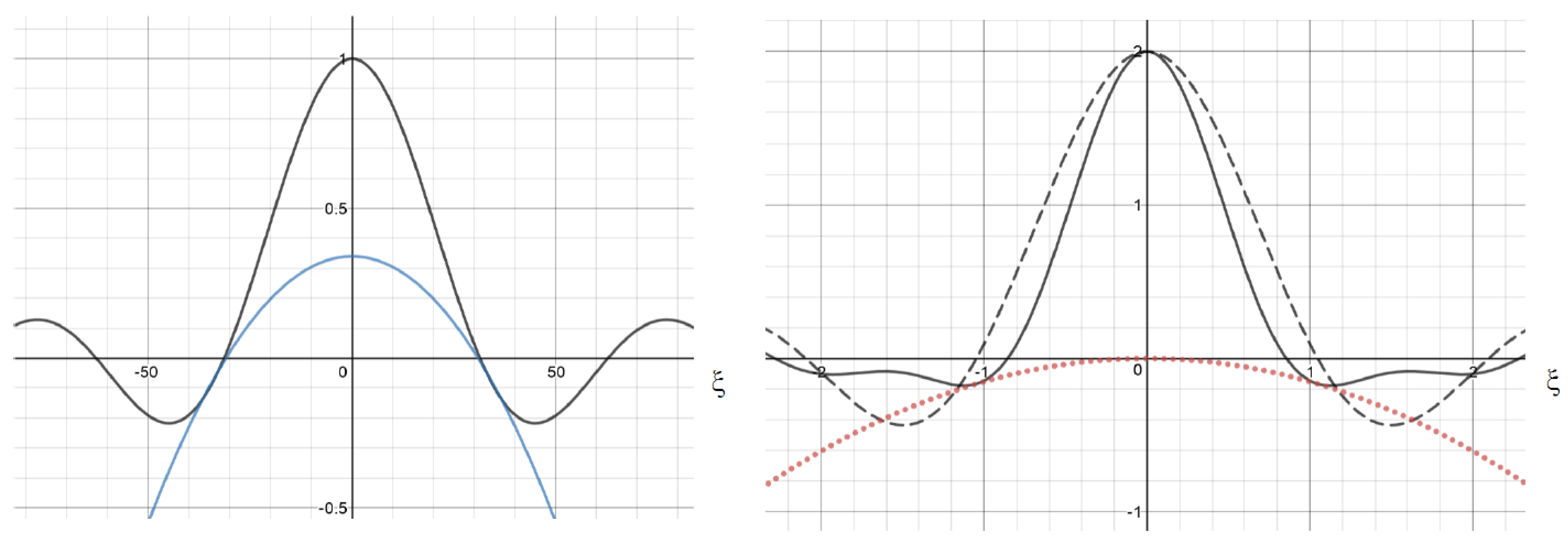
2.1. Single Nonlocal Term
2.2. Examples
2.3. Double Nonlocal Equation
2.4. Delay Equation
2.5. Nonlocal Delay Equation
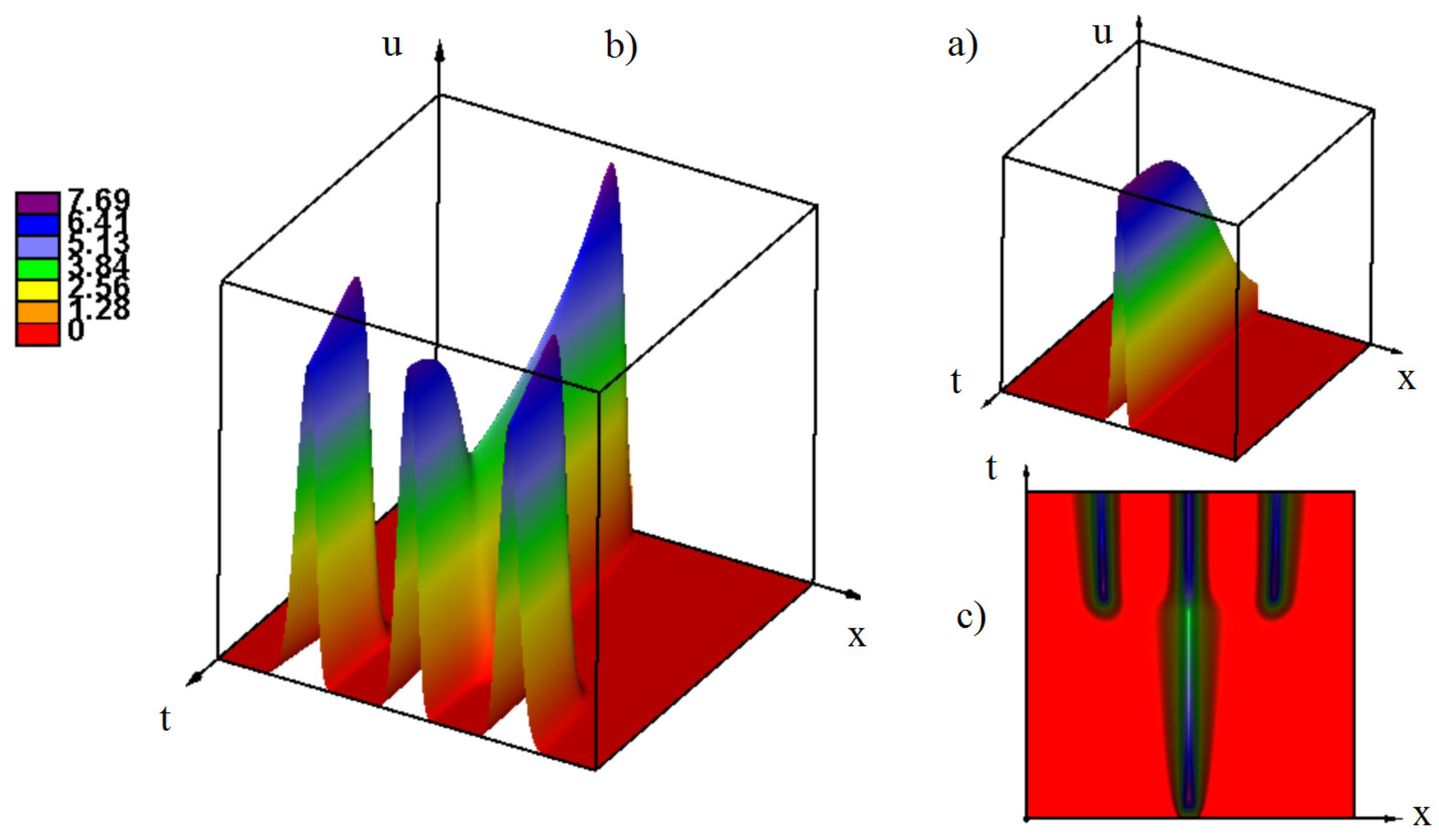
3. Emergence of Strains as Periodic Wave Propagation
3.1. Propagation Of Waves
3.1.1. Nonlocal Equation
3.1.2. Bifurcations of Waves and Pulses
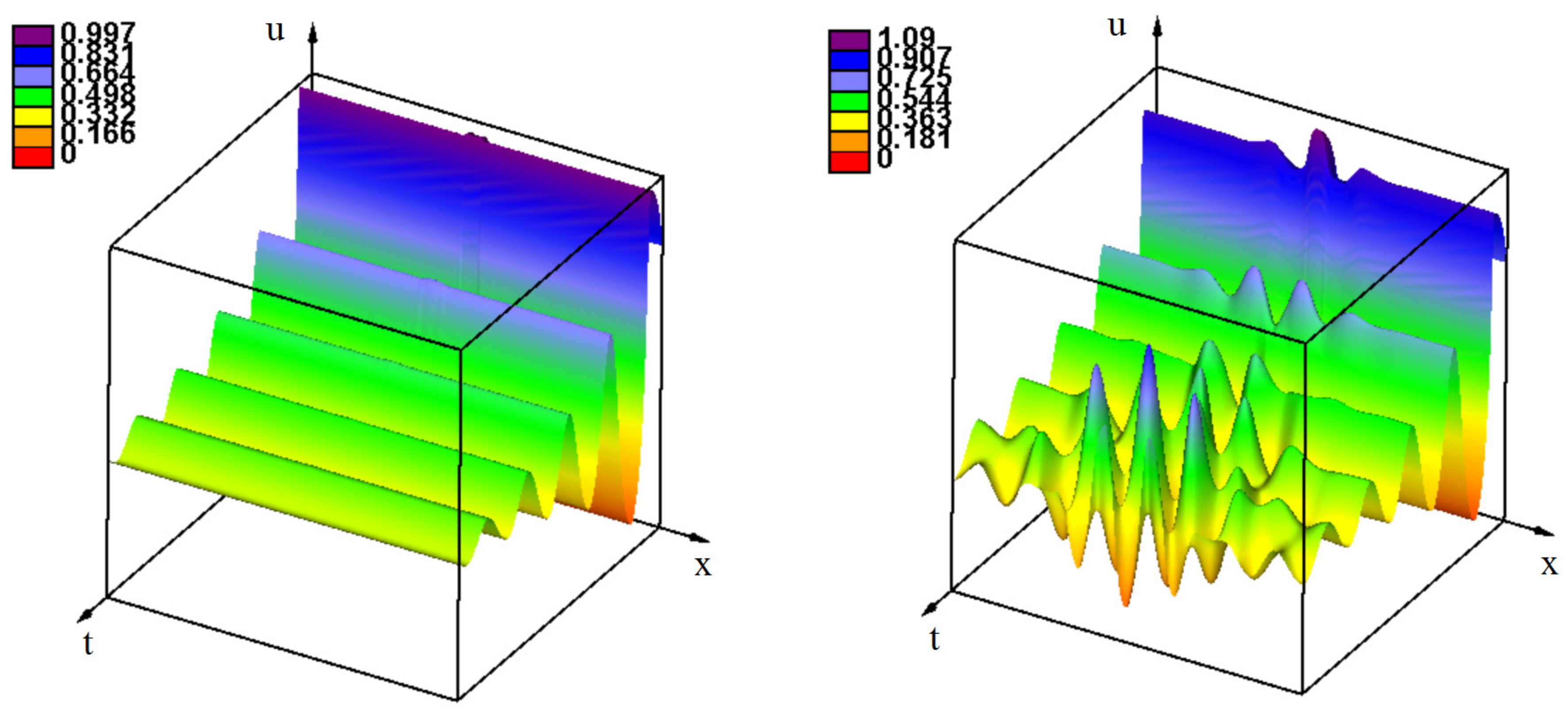
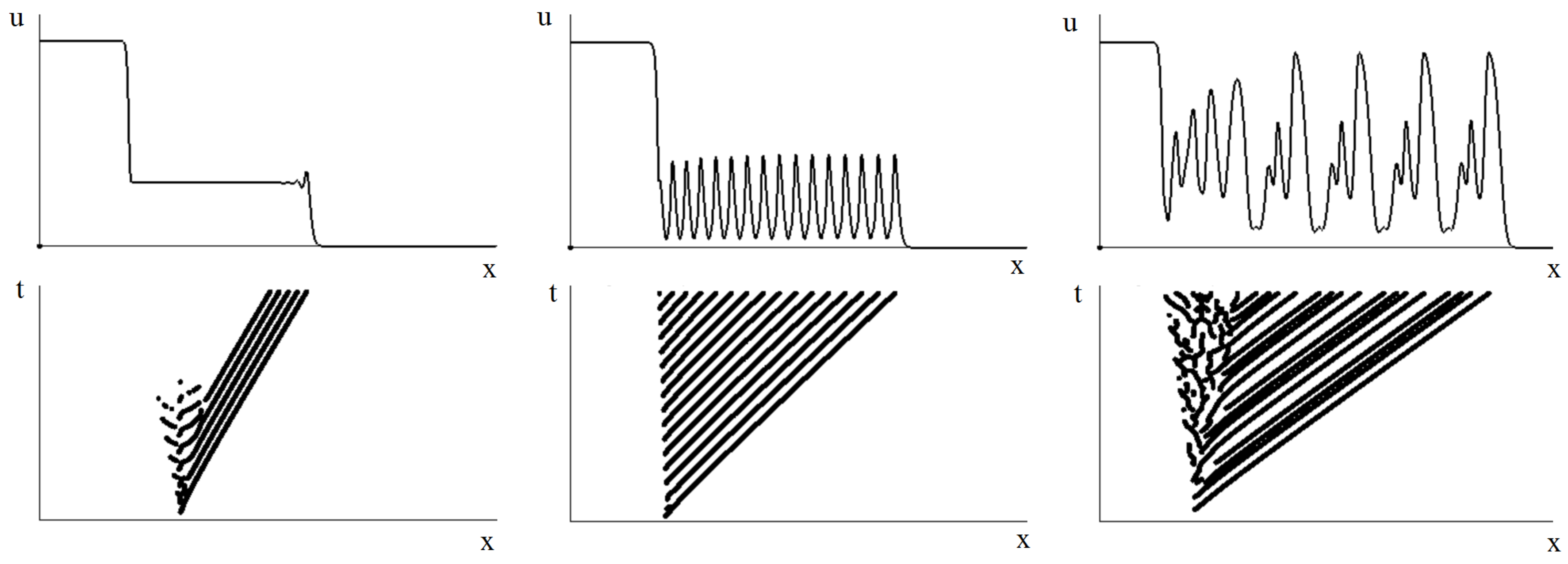
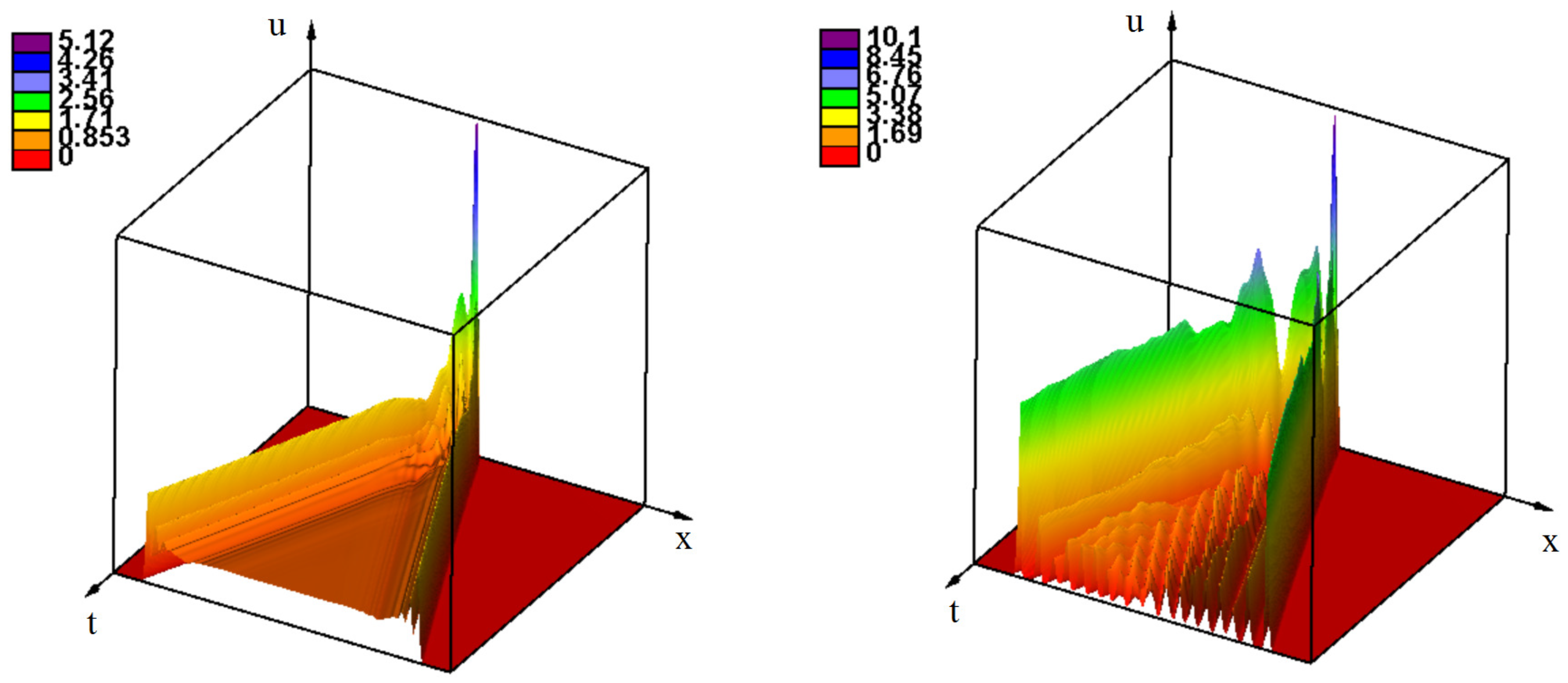
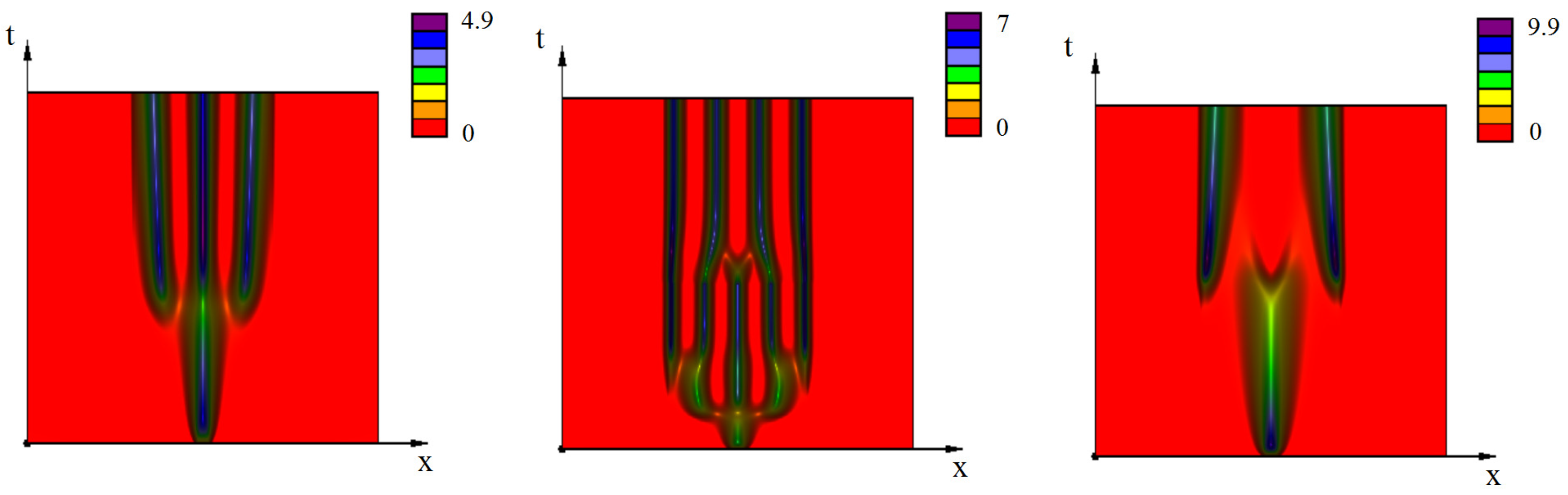
3.2. Emergence of Strains
3.2.1. Initiation of Periodic Waves
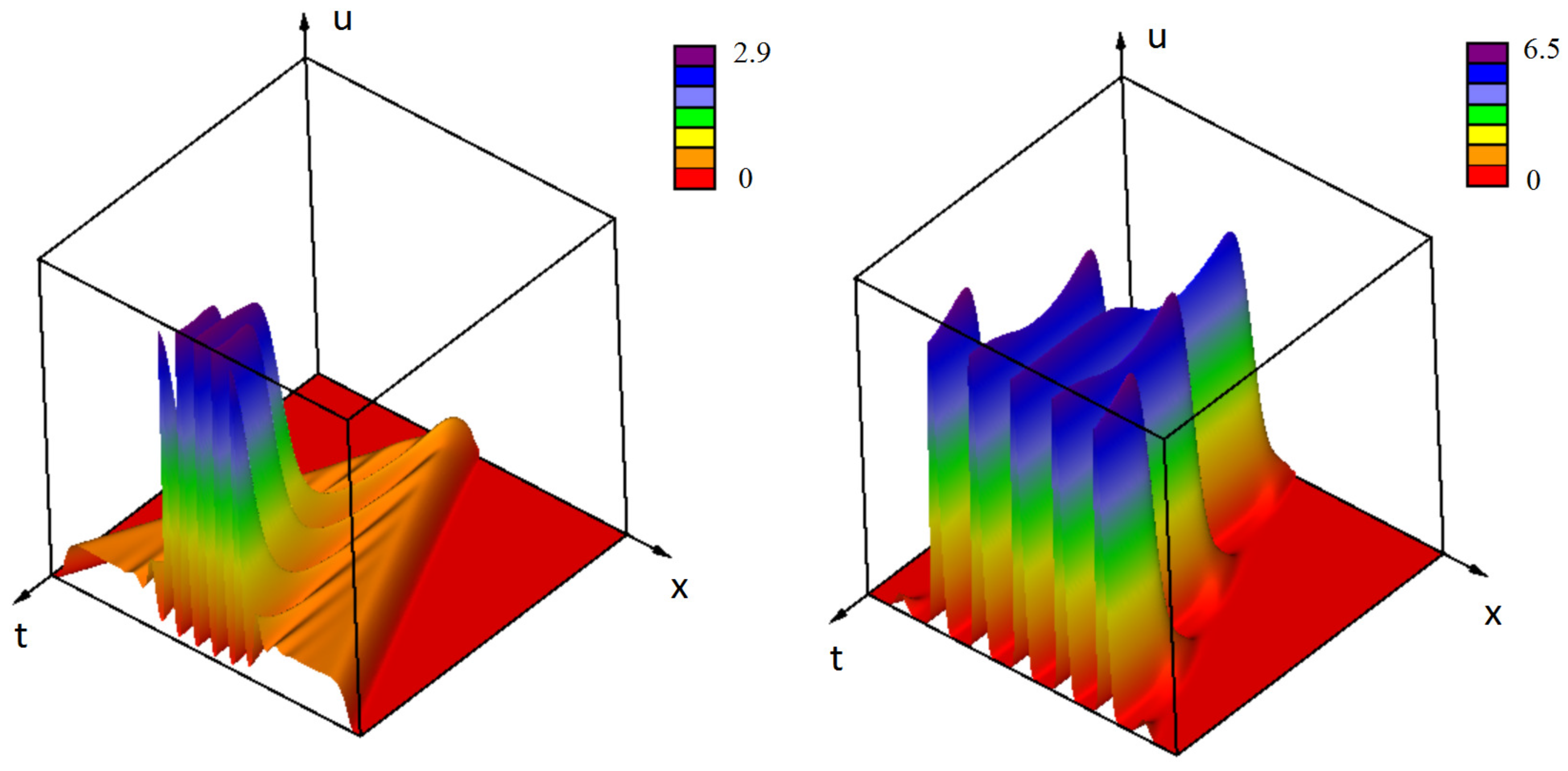
3.2.2. the Influence of Immune Response
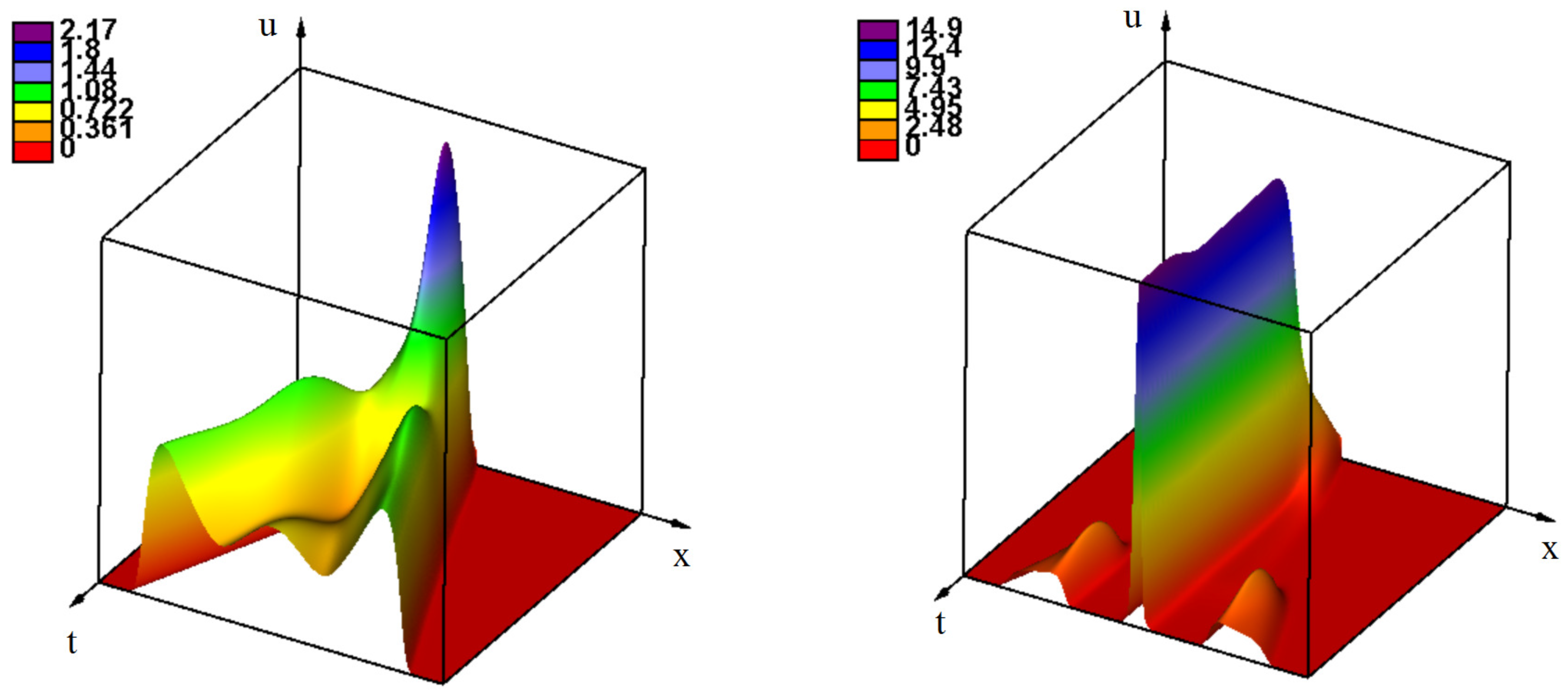
3.2.3. Effect of the Delay of the Antiviral Immune Response
3.2.4. the Influence of Genotype-Dependent Mortality
4. Discussion
4.1. Virus Quasi-Species
4.2. Emergence of New Quasi-Species: Summary of the Results
4.3. Biological Interpretations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
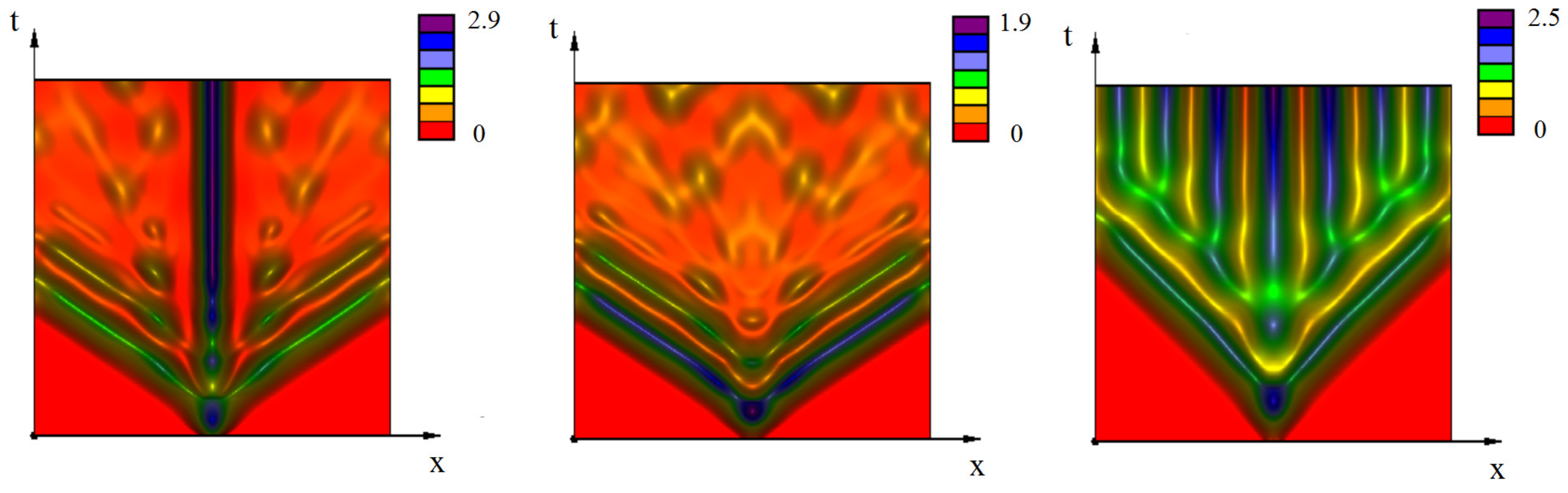
Appendix A. Additional Simulations
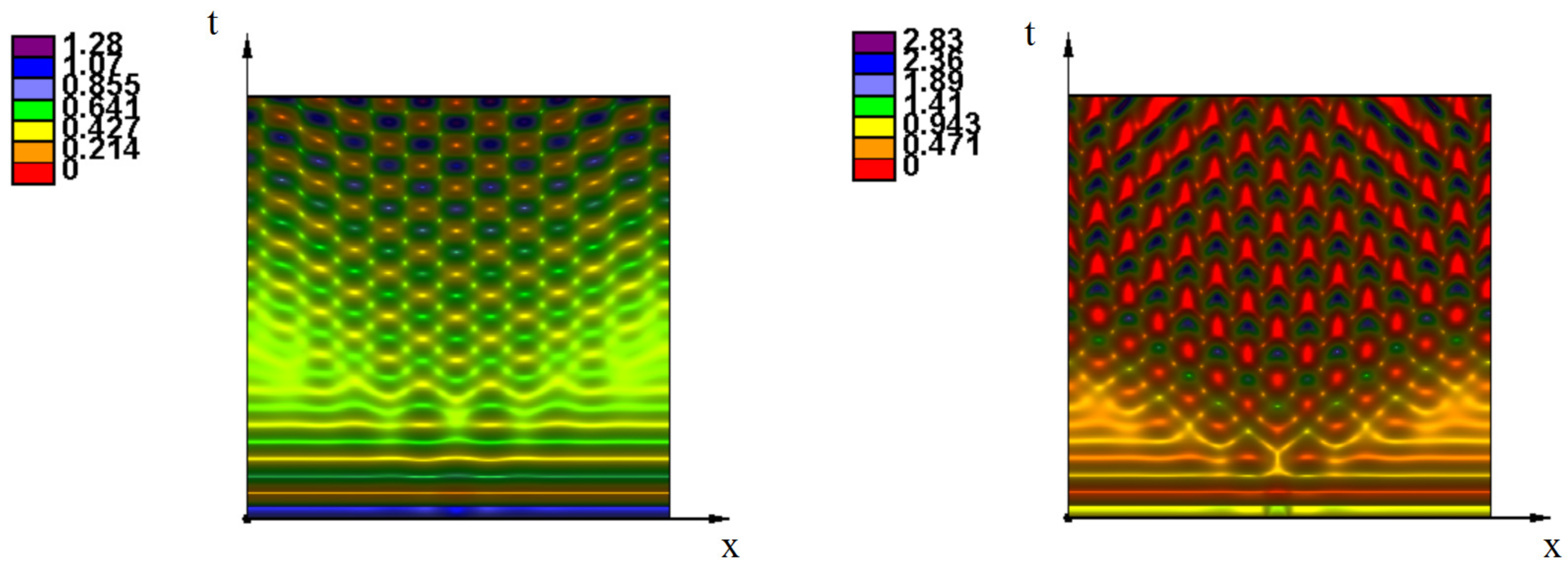
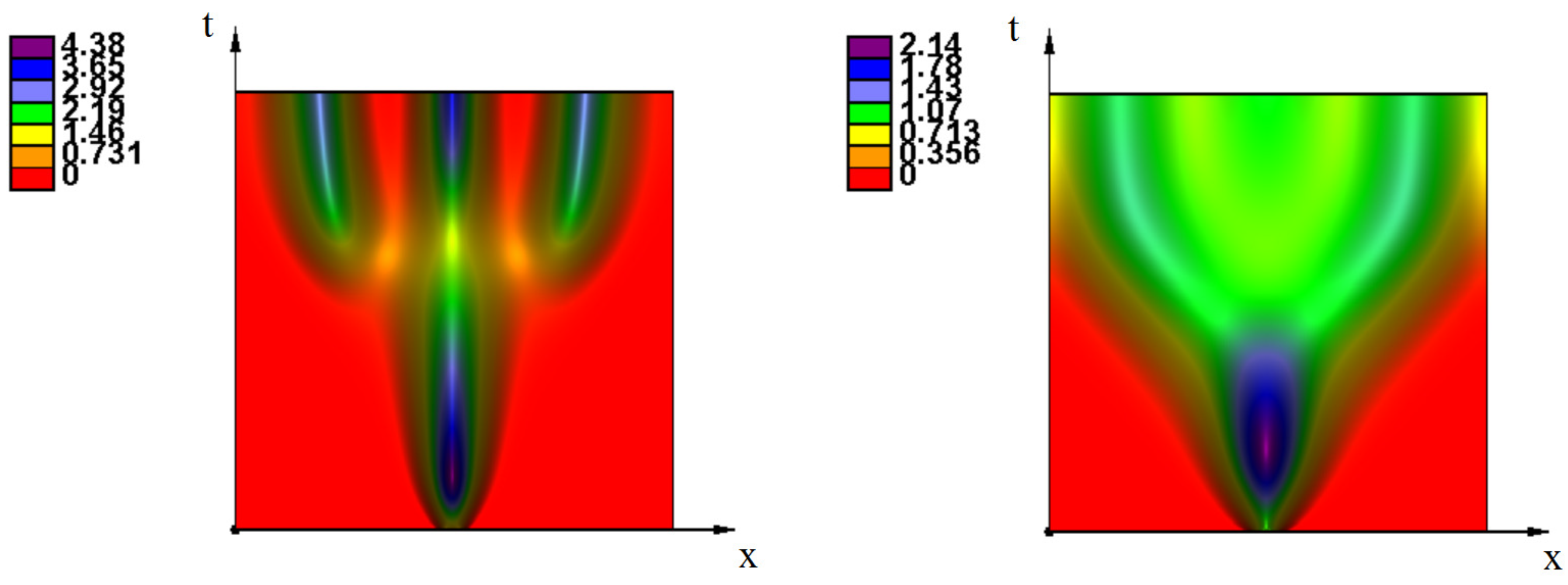
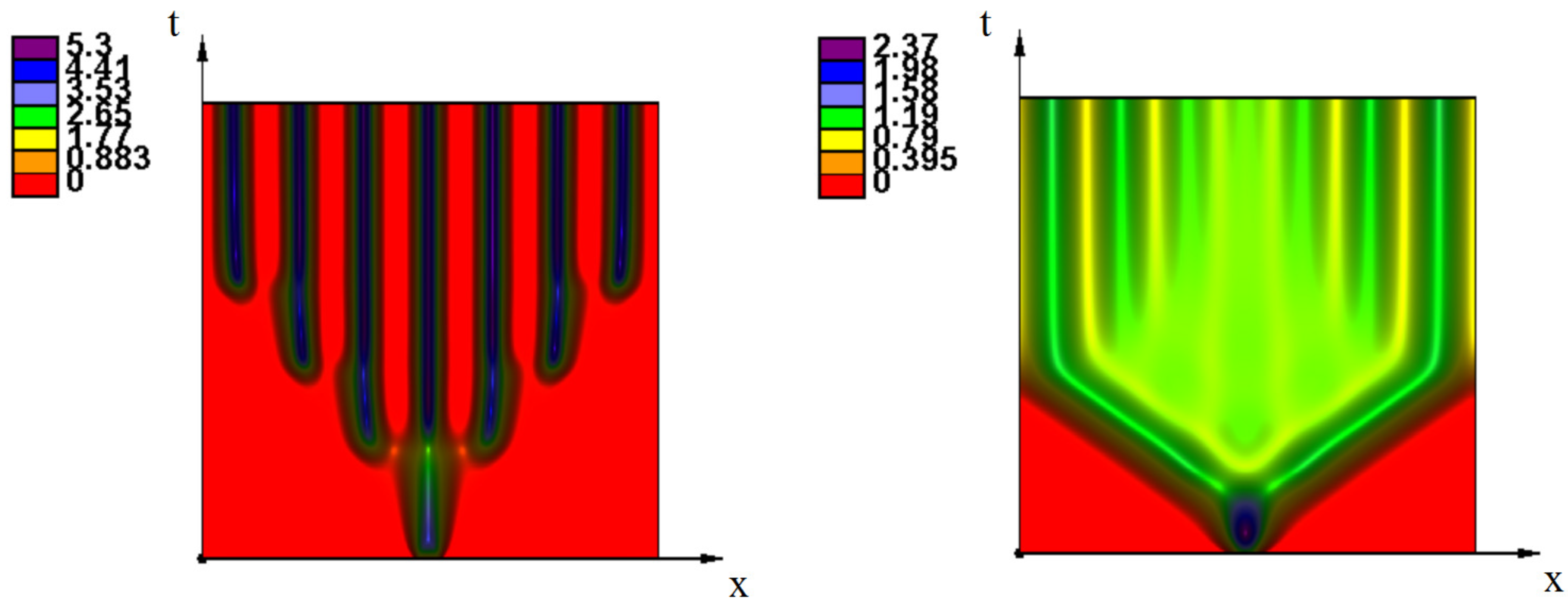
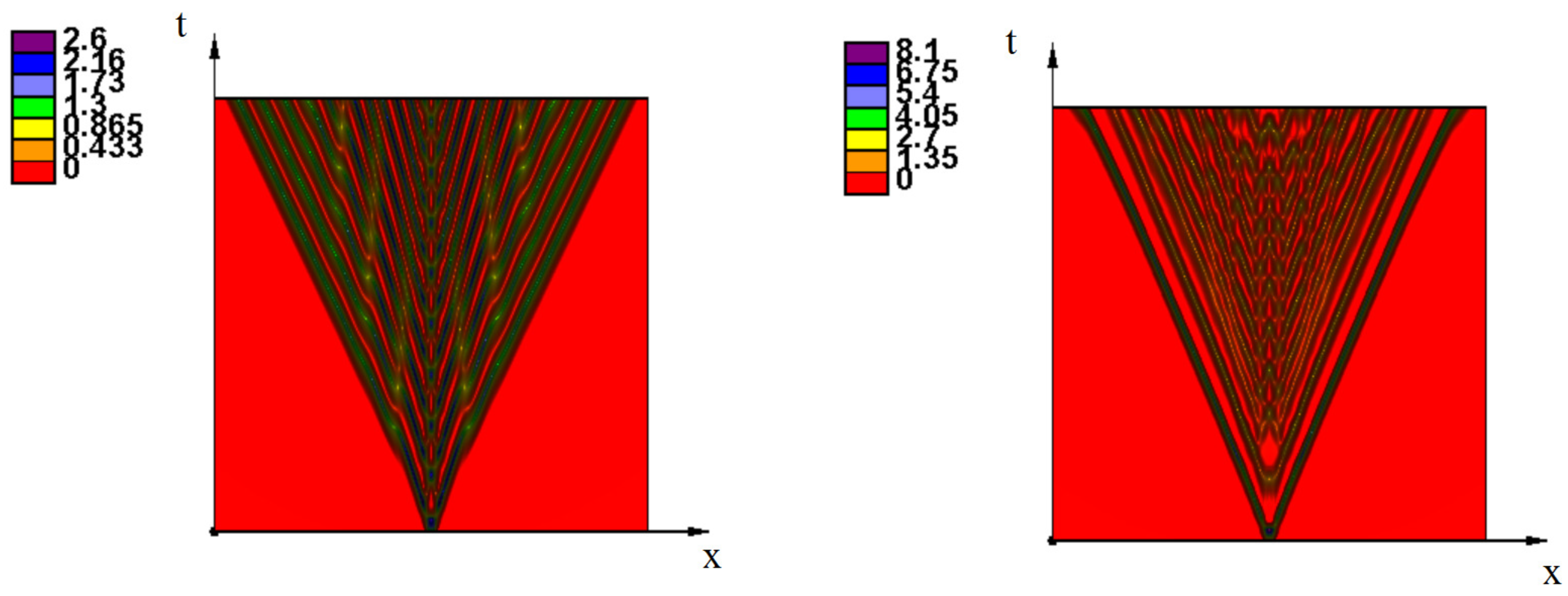
References
- Keele, B.H.; Giorgi, E.E.; Salazar-Gonzalez, J.F.; Decker, J.M.; Pham, K.T.; Salazar, M.G.; Sun, C.; Grayson, T.; Wang, S.; Li, H.; et al. Identication and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc. Natl. Acad. Sci. USA 2008, 105, 7552–7557. [Google Scholar] [CrossRef] [PubMed]
- Plikat, U.; Nieselt-Struwe, K.; Meyerhans, A. Genetic drift can dominate short-term human immunodeficiency virus type 1 nef quasispecies evolution in vivo. J. Virol. 1997, 71, 4233–4240. [Google Scholar] [CrossRef] [PubMed]
- Biebricher, C.K.; Eigen, M. What is a quasispecies? Curr. Top. Microbiol. Immunol. 2006, 299, 1–31. [Google Scholar] [PubMed]
- Domingo, E.; Perales, C. Viral quasispecies. PLoS Genet. 2019, 15, e1008271. [Google Scholar] [CrossRef] [PubMed]
- Goodenow, M.; Huet, T.; Saurin, W.; Kwok, S.; Sninsky, J.; Wain-Hobson, S. HIV-1 isolates are rapidly evolving quasispecies: Evidence for viral mixtures and preferred nucleotide substitutions. J. Acquir. Immune Defic. Syndr. 1989, 2, 344–352. [Google Scholar] [PubMed]
- Holland, J.J.; De La Torre, J.C.; Steinhauer, D.A. RNA virus populations as quasispecies. Curr. Top. Microbiol. Immunol. 1992, 176, 1–20. [Google Scholar]
- Meyerhans, A.; Cheynier, R.; Albert, J.; Seth, M.; Kwok, S.; Sninsky, J.; Morfeldt-Manson, L.; Asjo, B.; Wain-Hobson, S. Temporal fluctuations in HIV quasispecies in vivo are not reflected by sequential HIV isolations. Cell 1989, 58, 901–910. [Google Scholar] [CrossRef]
- Phillips, R.E.; Rowland-Jones, S.; Nixon, D.F.; Gotch, F.M.; Edwards, J.P.; Ogunlesi, A.O.; Elvin, J.G.; Rothbard, J.A.; Bangham, C.R.; Rizza, C.R.; et al. Human immunodeficiency virus genetic variation that can escape cytotoxic T cell recognition. Nature 1991, 354, 453–459. [Google Scholar] [CrossRef]
- Larder, B.A.; Kemp, S.D. Multiple mutations in HIV-1 reverse transcriptase confer high-level resistance to zidovudine (AZT). Science 1989, 246, 1155–1158. [Google Scholar] [CrossRef]
- Vignuzzi, M.; Stone, J.K.; Arnold, J.J.; Cameron, C.E.; Andino, R. Quasispecies diversity determines pathogenesis through cooperative interactions in a viral population. Nature 2006, 439, 344–348. [Google Scholar] [CrossRef]
- Collier, D.A.; Monit, C.; Gupta, R.K. The Impact of HIV-1 drug escape on the global treatment landscape. Cell Host Microbe 2019, 26, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Esposito, I.; Trinks, J.; Soriano, V. Hepatitis C virus resistance to the new direct-acting antivirals. Expert Opin. Drug Metab. Toxicol. 2016, 12, 1197–1209. [Google Scholar] [CrossRef] [PubMed]
- Kiepiela, P.; Leslie, A.J.; Honeyborne, I.; Ramduth, D.; Thobakgale, C.; Chetty, S.; Rathnavalu, P.; Moore, C.; Pfaerott, K.J.; Hilton, L. Dominant inuence of HLA-B in mediating the potential co-evolution of HIV and HLA. Nature 2004, 432, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Haas, G.; Plikat, U.; Debré, P.; Lucchiari, M.; Katlama, C.; Dudoit, Y.; Bonduelle, O.; Bauer, M.; Ihlenfeldt, H.G.; Jung, G.; et al. Dynamics of viral variants in HIV-1 Nef and specific cytotoxic T lymphocytes in vivo. J. Immunol. 1996, 157, 4212–4221. [Google Scholar] [PubMed]
- Ganusov, V.V.; Goonetilleke, N.; Liu, M.K.; Ferrari, G.; Shaw, G.M.; McMichael, A.J.; Borrow, P.; Korber, B.T.; Perelson, A.S. Fitness costs and diversity of the cytotoxic T lymphocyte (CTL) response determine the rate of CTL escape during acute and chronic phases of HIV infection. J. Virol. 2011, 85, 10518–10528. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, E.L.; Wong, M.; Wang, S.; Wei, X.; Jones, N.A.; Conrod, K.E.; Aldam, D.; Turner, J.; Pellegrino, P.; Keele, B.F.; et al. Kinetics of expansion of epitope-specific T cell responses during primary HIV-1 infection. J. Immunol. 2009, 182, 7131–7145. [Google Scholar] [CrossRef]
- Ganusov, V.V.; Neher, R.A.; Perelson, A.S. Mathematical modeling of escape of HIV from cytotoxic T lymphocyte responses. J. Stat. Mech. 2013, 2013, P01010. [Google Scholar] [CrossRef]
- Bocharov, G.A.; Ford, N.J.; Edwards, J.; Breinig, T.; Wain-Hobson, S.; Meyerhans, A. A genetic algorithm approach to simulating human immunodeficiency virus evolution reveals the strong impact of multiply infected cells and recombination. J. Gen. Virol. 2005, 86, 3109–3118. [Google Scholar] [CrossRef]
- Swanstrom, R.; Coffin, J. HIV-1 pathogenesis: The virus. Cold Spring Harb. Perspect. Med. 2012, 2, a007443. [Google Scholar] [CrossRef]
- Bessonov, N.; Reinberg, N.; Volpert, V. Mathematics of Darwin’s diagram. Math. Model. Nat. Phenom. 2014, 9, 5–25. [Google Scholar] [CrossRef]
- Bessonov, N.; Reinberg, N.; Banerjee, M.; Volpert, V. The origin of species by means of mathematical modelling. Acta Bioteoretica 2018, 66, 333–344. [Google Scholar]
- Genieys, S.; Volpert, V.; Auger, P. Adaptive dynamics: Modelling Darwin’s divergence principle. Comptes Rendus Biol. 2006, 329, 876–879. [Google Scholar] [CrossRef] [PubMed]
- Bessonov, N.; Bocharov, G.; Meyerhans, A.; Popov, V.; Volpert, V. Nonlocal reaction-diffusion model of viral evolution. Existence and dynamics of strains. Preprints 2019. [Google Scholar] [CrossRef]
- Volpert, V. Elliptic Partial Differential Equations. Volume 2. Reaction-Diffusion Equations; Birkhäuser: Basel, Switzerland, 2014. [Google Scholar]
- Bessonov, N.; Bocharov, G.; Meyerhans, A.; Volpert, V. Interplay between reaction and diffusion processes in governing the dynamics of virus infections. J. Theor. Biol. 2018, 457, 221–236. [Google Scholar]
- Bocharov, G.; Volpert, V.; Ludewig, B.; Meyerhans, A. Mathematical Immunology of Virus Infections; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Bocharov, G.; Meyerhans, A.; Bessonov, N.; Trofimchuk, S.; Volpert, V. Spatiotemporal dynamics of virus infection spreading in tissues. PLoS ONE 2006. [Google Scholar] [CrossRef]
- Bocharov, G.; Meyerhans, A.; Bessonov, N.; Trofimchuk, S.; Volpert, V. Modelling the dynamics of virus infection and immune response in space and time. Int. J. Parallel Emerg. Distrib. Syst. 2019, 34, 341–355. [Google Scholar] [CrossRef]
- Perthame, B.; Genieys, S. Concentration in the nonlocal Fisher equation: The Hamilton-Jacobi limit. Math. Model. Nat. Phenom. 2007, 4, 135–151. [Google Scholar] [CrossRef]
- Banerjee, M.; Volpert, V. Spatio-temporal pattern formation in Rosenzweig–Macarthur model: Effect of nonlocal interactions. Ecol. Complex. 2017, 30, 2–10. [Google Scholar] [CrossRef]
- Lorz, A.; Lorenzi, T.; Clairambault, J.; Escargueil, A.; Perthame, B. Modeling the effects of space structure and combination therapies on phenotypic heterogeneity and drug resistance in solid tumors. Bull. Math. Biol. 2015, 77, 1–22. [Google Scholar] [CrossRef]
- Gourley, S.A.; Chaplain, M.A.J.; Davidson, F.A. Spatio-temporal pattern formation in a nonlocal reaction-diffusion equation. Dyn. Syst. 2001, 16, 173–192. [Google Scholar] [CrossRef]
- Alfaro, M.; Ducrot, A.; Giletti, T. Travelling waves for a non-monotone bistable equation with delay: Existence and oscillations. Proc. Lond. Math. Soc. 2018, 116, 729–759. [Google Scholar] [CrossRef]
- Alfaro, M.; Coville, J.; Raoul, G. Bistable travelling waves for nonlocal reaction diffusion equations. Discret. Contin. Dyn. Syst. 2014, 34, 1775–1791. [Google Scholar] [CrossRef]
- Apreutesei, N.; Bessonov, N.; Volpert, V.; Vougalter, V. Spatial structures and generalized travelling waves for an integro-differential equation. Discret. Contin. Dyn. Syst. Ser. B 2010, 13, 537–557. [Google Scholar] [CrossRef]
- Nadin, G.; Rossi, L.; Ryzhik, L.; Perthame, B. Wave-like solutions for nonlocal reaction-diffusion equations: A toy model. Math. Model. Nat. Phenom. 2013, 8, 33–41. [Google Scholar] [CrossRef]
- Bessonov, N.; Bocharov, G.; Touaoula, T.M.; Trofimchuk, S.; Volpert, V. Delay reaction-diffusion equation for infection dynamics. Discret. Contin. Dyn. Syst. B 2019, 24, 2073–2091. [Google Scholar] [CrossRef]
- Volpert, V. Pulses and waves for a bistable nonlocal reaction-diffusion equation. Appl. Math. Lett. 2015, 44, 21–25. [Google Scholar] [CrossRef]
- Britton, N.F. Spatial structures and periodic travelling waves in an integro-differential reaction-diffusion population model. SIAM J. Appl. Math. 1990, 6, 1663–1688. [Google Scholar] [CrossRef]
- Volpert, V. Asymptotic behavior of solutions of a nonlinear diffusion equation with a source term of general form. Sib. Math. J. 1989, 30, 25–36. [Google Scholar] [CrossRef]
- Trofimchuk, S.; Volpert, V. Traveling waves for a bistable reaction-diffusion equation with delay. SIAM J. Math. Anal. 2018, 50, 1175–1199. [Google Scholar] [CrossRef]
- Trofimchuk, S.; Volpert, V. Global continuation of monotone waves for a unimodal bistable reaction-diffusion equation with delay. Nonlinearity 2019, 32, 2593. [Google Scholar] [CrossRef]
- Coyne, J.A.; Orr, H.A. Speciation; Sinauer Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Volpert, V. Branching and aggregation in self-reproducing systems. ESAIM Proc. Surv. 2014, 47, 116–129. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bessonov, N.; Bocharov, G.; Meyerhans, A.; Popov, V.; Volpert, V. Nonlocal Reaction–Diffusion Model of Viral Evolution: Emergence of Virus Strains. Mathematics 2020, 8, 117. https://doi.org/10.3390/math8010117
Bessonov N, Bocharov G, Meyerhans A, Popov V, Volpert V. Nonlocal Reaction–Diffusion Model of Viral Evolution: Emergence of Virus Strains. Mathematics. 2020; 8(1):117. https://doi.org/10.3390/math8010117
Chicago/Turabian StyleBessonov, Nikolai, Gennady Bocharov, Andreas Meyerhans, Vladimir Popov, and Vitaly Volpert. 2020. "Nonlocal Reaction–Diffusion Model of Viral Evolution: Emergence of Virus Strains" Mathematics 8, no. 1: 117. https://doi.org/10.3390/math8010117
APA StyleBessonov, N., Bocharov, G., Meyerhans, A., Popov, V., & Volpert, V. (2020). Nonlocal Reaction–Diffusion Model of Viral Evolution: Emergence of Virus Strains. Mathematics, 8(1), 117. https://doi.org/10.3390/math8010117





