Atherosclerotic Plaque Segmentation Based on Strain Gradients: A Theoretical Framework
Abstract
1. Introduction
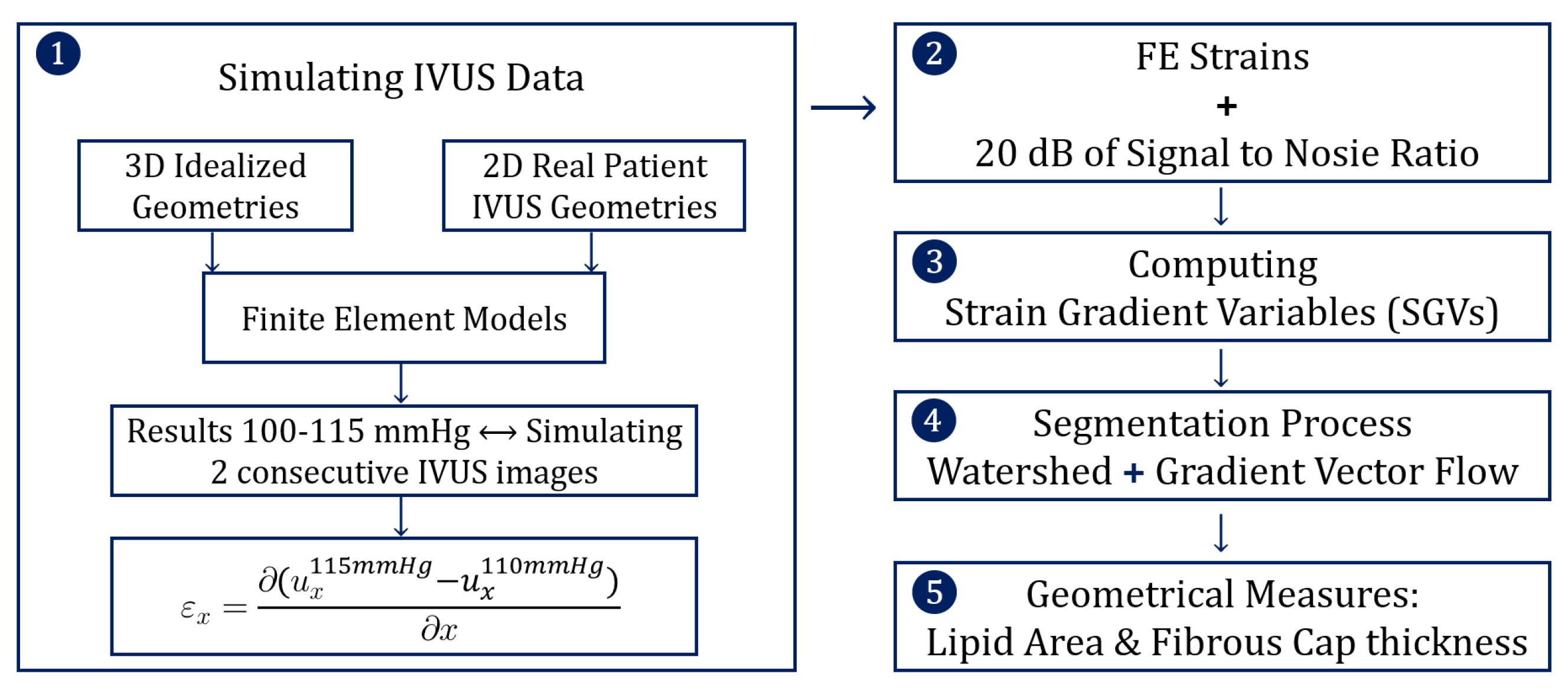
2. Materials and Methods
2.1. Simulating IVUS Data
2.1.1. Geometries
2.1.2. Modeling of Tissue Behavior
2.1.3. FE Models
2.1.4. Strain Variables
2.2. Adding Noise
2.3. Computing SGVs
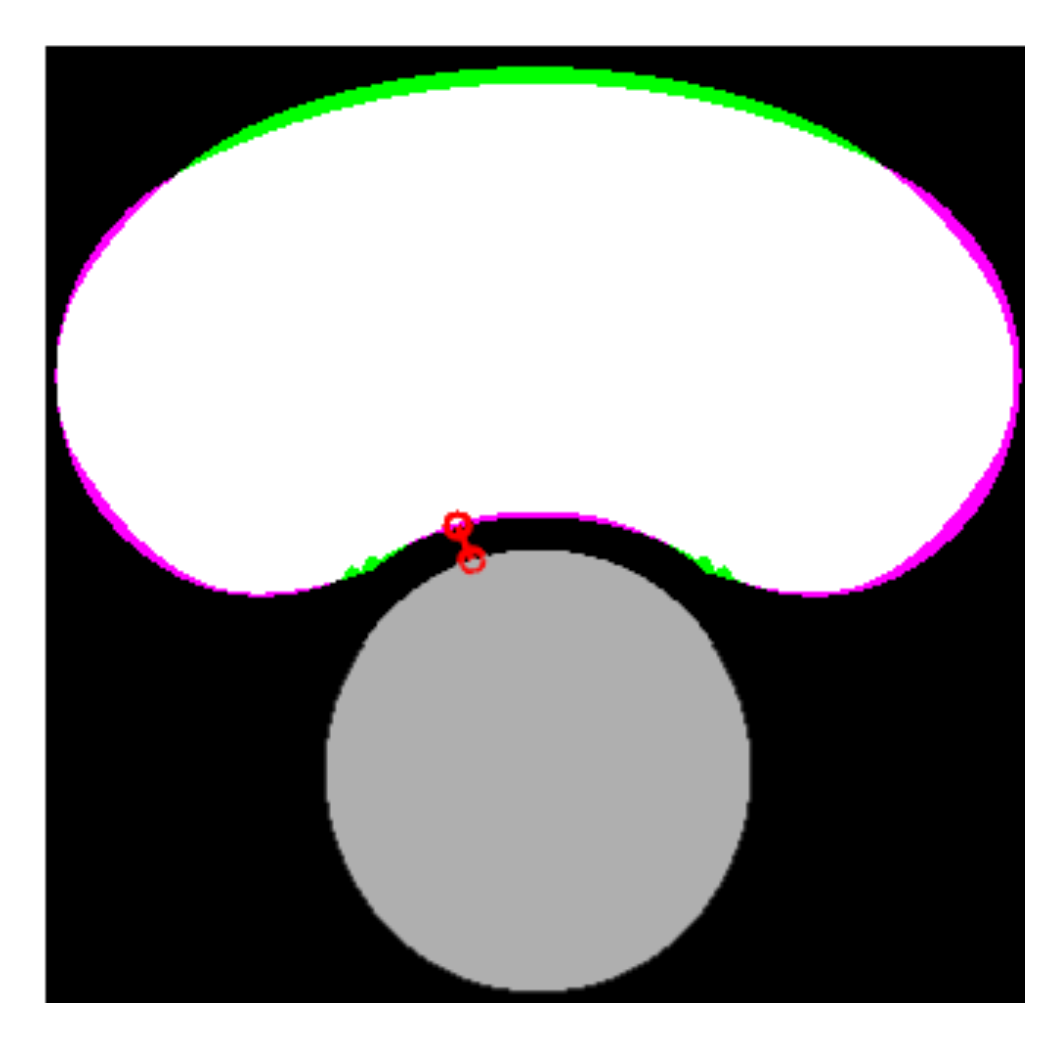
2.4. Segmentation Process
- Plaque-related variables: We analyzed the influence of considering the fibrotic tissue as fully incompressible or with different degrees of quasi-incompressibility. We have also considered four different fibrotic tissues (default, stiff, medium, and soft tissues). Furthermore, some inclusions were added to the FE model, mimicking the presence of micro calcifications. These inclusions were simplified as spheres with calcification properties presented in Table 1, and four diameters were studied (10, 50, 150, and 300 m).
- IVUS-related variables: The influence of the catheter position was studied by changing the origin and orientation of the coordinate system in the FE models. It was also important to check if the segmentation methodology was affected by the blood pressure. In addition, the pressure increment between both steps was also studied.
2.5. Geometrical Measures
3. Results
3.1. Idealized Geometries
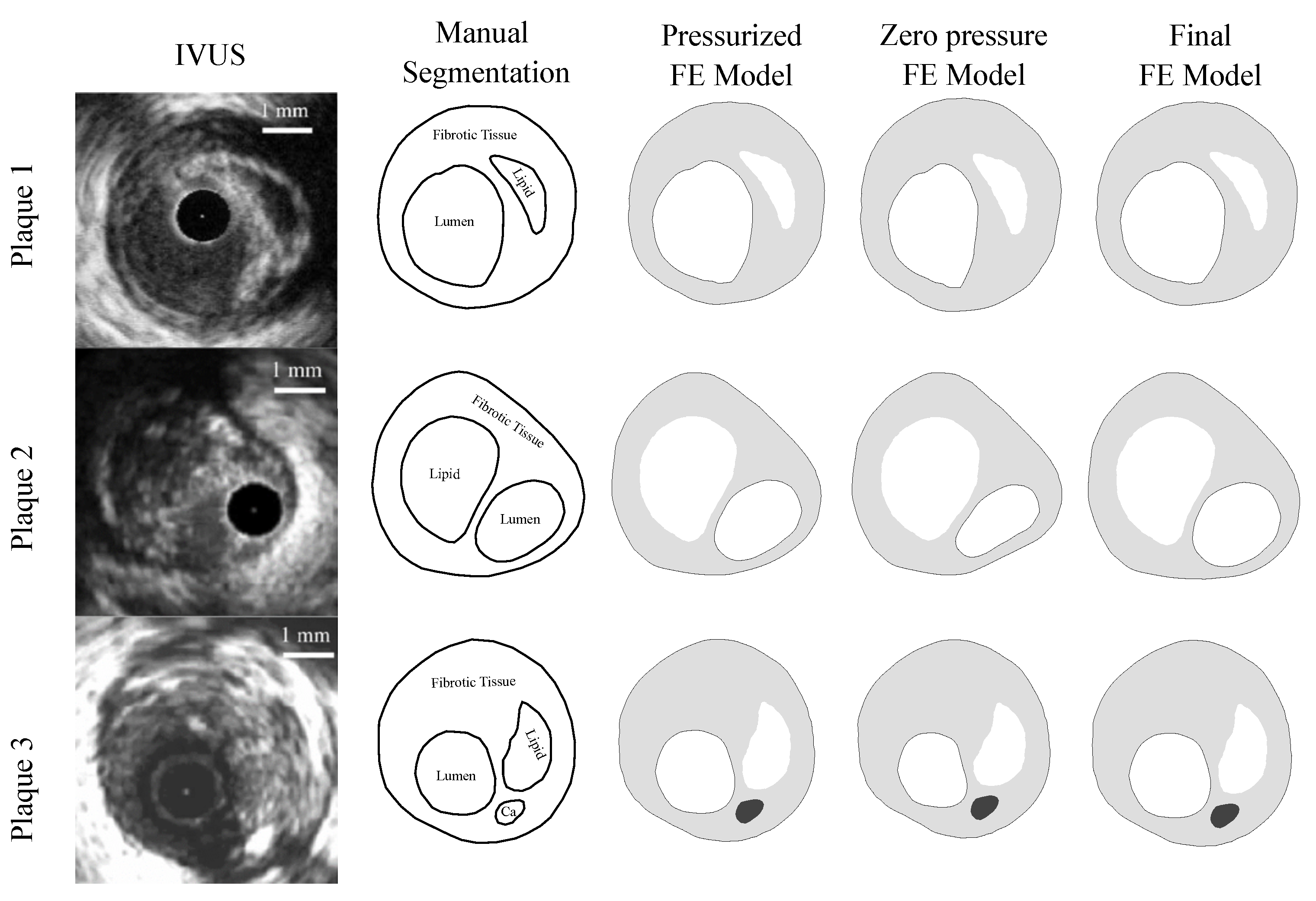
3.2. Real IVUS Geometries
3.3. SGV Candidates
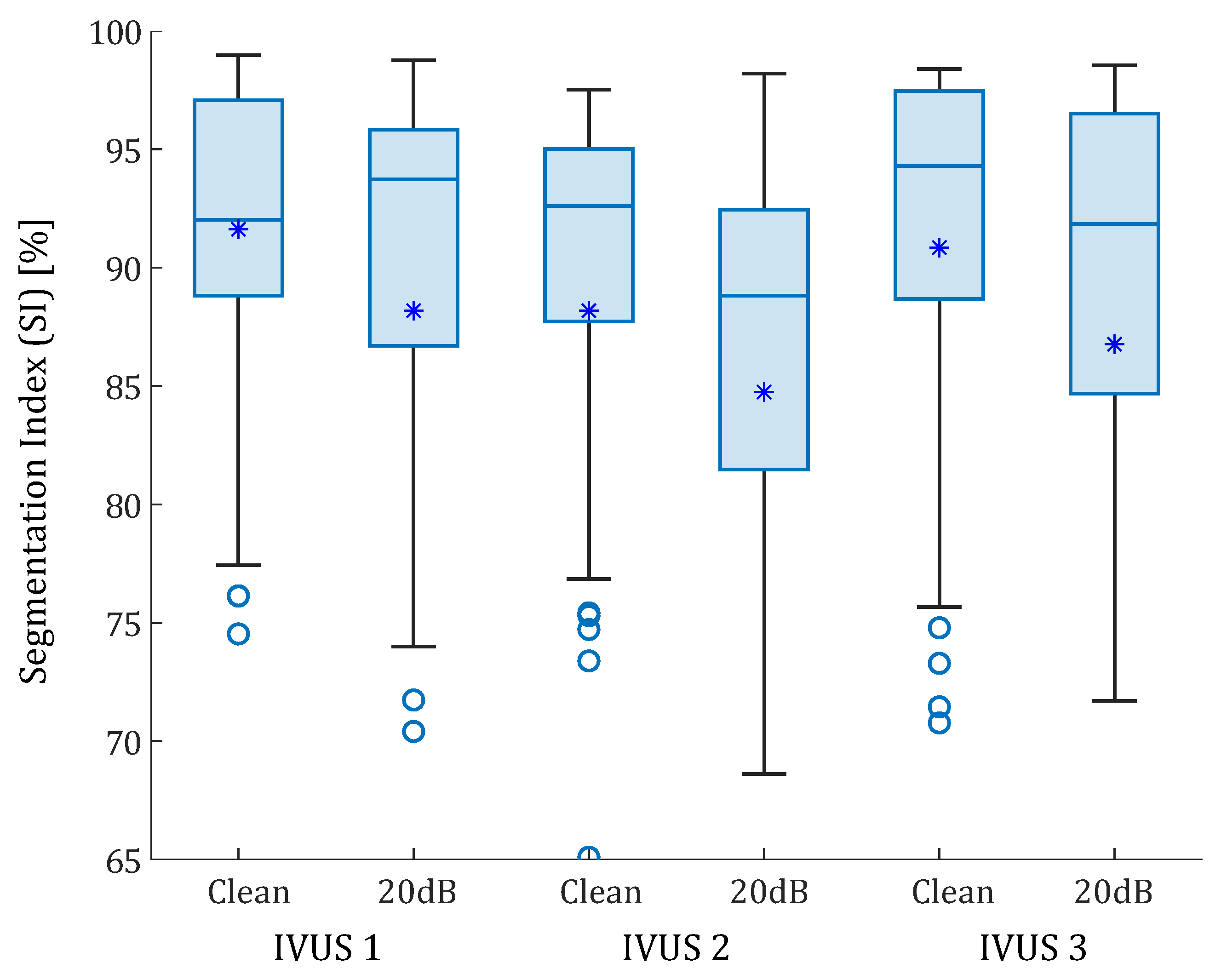
3.4. Sensitivity Analysis
4. Discussion
4.1. Segmentation Analysis
4.1.1. Idealized Geometries
4.1.2. IVUS Geometries
4.2. SGV Candidates
4.3. Sensitivity analysis
4.4. Relevance for Clinical Applications
- A segmentation process based on strain representation was presented to extract the different tissues of an atherosclerotic plaque. This methodology achieved high accuracy in measuring FCT and the lipid core area. These measurements play a key role in the vulnerability of the plaque.
- Unlike other segmentation processes, this method does not require a database to be trained or an optimization process, as it relies on image processing rather than machine learning or analysis of the mechanical properties of the tissues. In addition, it could be performed with many different strain variables instead of a single one [27,28,31,47]. Thus, there are different possibilities to obtain the segmentation using only one variable or combining different SGVs.
- The results show that the performance of the segmentation was linked to the plaque geometry and the selected SGVs. However, there were some SGVs with good results regardless of the geometry. The method also showed good robustness in sensitivity analysis, providing accurate results with different catheter positions, pressures, and noise addition.
4.5. Limitations
- Since this work was a theoretical framework, the methodology was only tested with computational models of in silico data. Therefore, the next step would be to prove the segmentation methodology with in vitro and in vivo IVUS data from patients with coronary atherosclerotic plaques. After analyzing the methodology with noise, which simulates the intrinsic noise of IVUS data, the results for segmentation are expected to be valid on real IVUS data.
- In the finite element analysis we only have considered the load of the blood pressure. We have disregarded the residual stress and the influence of heart motion. As the methodology is based on gradients and not on absolute strain/stress values, we could expect a minimum influence of the residual stress on this segmentation methodology.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IVUS | Intravascular Ultrasound |
| FCT | Fibrous Cap Thickness |
| VH | Virtual Histology |
| FE | Finite Element |
| CNN | Convolutional Neural Networks |
| SGV | Strain Gradient Variable |
| SNR | Signal-to-Noise Ratio |
| SI | Segmentation Index |
| W-GVF | Watershed-(Gradient Vector Flow) |
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Casscells, W.; Naghavi, M.; Willerson, J.T. Vulnerable atherosclerotic plaque: A multifocal disease. Circulation 2003, 107, 2072–2075. [Google Scholar] [CrossRef]
- Jebari-Benslaiman, S.; Galicia-García, U.; Larrea-Sebal, A.; Olaetxea, J.R.; Alloza, I.; Vandenbroeck, K.; Benito-Vicente, A.; Martín, C. Pathophysiology of Atherosclerosis. Int. J. Mol. Sci. 2022, 23, 3346. [Google Scholar] [CrossRef]
- Libby, P.; Theroux, P. Pathophysiology of coronary artery disease. Circulation 2005, 111, 3481–3488. [Google Scholar] [CrossRef]
- Cilla, M.; Peña, E.; Martínez, M.A. 3D computational parametric analysis of eccentric atheroma plaque influence of axial and circumferential residual stresses. Biomech. Model. Mechanobiol. 2012, 11, 1001–1013. [Google Scholar] [CrossRef] [PubMed]
- Finet, G.; Ohayon, J.; Rioufol, G. Biomechanical interaction between cap thickness, lipid core composition and blood pressure in vulnerable coronary plaque: Impact on stability or instability. Coron. Artery Dis. 2004, 15, 13–20. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, J.; Maehara, A.; Monoly, D.; Samady, H.; Wang, L.; Billiar, K.L.; Zheng, J.; Yang, C.; Mintz, G.S.; et al. Quantify patient-specific coronary material property and its impact on stress/strain calculations using in vivo IVUS data and 3D FSI models: A pilot study. Biomech. Model. Mechanobiol. 2017, 16, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Olender, M.L.; Athanasiou, L.S.; Michalis, L.K.; Fotiadis, D.I.; Edelman, E.R. A Domain Enriched Deep Learning Approach to Classify Atherosclerosis Using Intravascular Ultrasound Imaging. IEEE J. Sel. Top. Signal Process. 2020, 14, 1210–1220. [Google Scholar] [CrossRef]
- Gudigar, A.; Nayak, S.; Samanth, J.; Raghavendra, U.; Ashwal, A.J.; Barua, P.D.; Hasan, M.N.; Ciaccio, E.J.; Tan, R.S.; Rajendra Acharya, U. Recent Trends in Artificial Intelligence-Assisted Coronary Atherosclerotic Plaque Characterization. Int. J. Environ. Res. Public Health 2021, 18, 3. [Google Scholar] [CrossRef] [PubMed]
- Kubo, T.; Nakamura, N.; Matsuo, Y.; Okumoto, Y.; Wu, X.; Choi, S.Y.; Komukai, K.; Tanimoto, T.; Ino, Y.; Kitabata, H.; et al. Virtual Histology Intravascular Ultrasound Compared With Optical Coherence Tomography for Identification of Thin-Cap Fibroatheroma. Int. Heart J. 2011, 52, 175–179. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rezaei, Z.; Selamat, A.; Taki, A.; Mohd Rahim, M.S.; Abdul Kadir, M.R. Automatic plaque segmentation based on hybrid fuzzy clustering and k nearest neighborhood using virtual histology intravascular ultrasound images. Appl. Soft Comput. 2017, 53, 380–395. [Google Scholar] [CrossRef]
- Athanasiou, L.S.; Karvelis, P.S.; Tsakanikas, V.D.; Naka, K.K.; Michalis, L.K.; Bourantas, C.V.; Fotiadis, D.I. A Novel Semiautomated Atherosclerotic Plaque Characterization Method Using Grayscale Intravascular Ultrasound Images: Comparison with Virtual Histology. IEEE Trans. Inf. Technol. Biomed. 2012, 16, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Lee, J.; Hwang, Y. A novel intensity-based multi-level classification approach for coronary plaque characterization in intravascular ultrasound images. Biomed. Eng. Online 2018, 17, 1–15. [Google Scholar] [CrossRef]
- Selvathi, D.; Emimal, N.; Selvaraj, H. Automated Characterization of Atheromatous Plaque in Intravascular Ultrasound Images Using Neuro Fuzzy Classifier. Int. J. Electron. Telecommun. 2012, 58, 425–431. [Google Scholar] [CrossRef]
- Rezaei, Z.; Selamat, A.; Taki, A.; Mohd Rahim, M.S.; Abdul Kadir, M.R.; Penhaker, M.; Krejcar, O.; Kuca, K.; Herrera-Viedma, E.; Fujita, H. Thin Cap Fibroatheroma Detection in Virtual Histology Images Using Geometric and Texture Features. Appl. Sci. 2018, 8, 1632. [Google Scholar] [CrossRef]
- Sofian, H.; Than, J.C.M.; Mohammad, S.; Mohd Noor, N. Calcification Detection of Coronary Artery Disease in Intravascular Ultrasound Image: Deep Feature Learning Approach. Int. J. Integr. Eng. 2018, 10, 43–57. [Google Scholar] [CrossRef]
- Liu, S.; Neleman, T.; Hartman, E.M.; Ligthart, J.M.; Witberg, K.T.; van der Steen, A.F.; Wentzel, J.J.; Daemen, J.; van Soest, G. Automated Quantitative Assessment of Coronary Calcification Using Intravascular Ultrasound. Ultrasound Med. Biol. 2020, 46, 2801–2809. [Google Scholar] [CrossRef] [PubMed]
- Jun, T.; Kang, S.J.; Lee, J. Automated detection of vulnerable plaque in intravascular ultrasound images. Med. Biol. Eng. Comput. 2019, 57, 863–876. [Google Scholar] [CrossRef]
- Balocco, S.; Gonzalez, M.; Ñanculef, R.; Radeva, P.; Thomas, G. Calcified Plaque Detection in IVUS Sequences: Preliminary Results Using Convolutional Nets. In International Workshop on Artificial Intelligence and Pattern Recognition; Springer: Berlin/Heidelberg, Germany, 2018; pp. 34–42. [Google Scholar] [CrossRef]
- Sofian, H.; Ming, J.; Muhammad, S.; Noor, N. Calcification detection using convolutional neural network architectures in intravascular ultrasound images. Indones. J. Electr. Eng. Comput. Sci. 2019, 17, 1313–1321. [Google Scholar] [CrossRef]
- Du, H.; Ling, L.; Yu, W.; Wu, P.; Yang, Y.; Chu, M.; Yang, J.; Yang, W.; Tu, S. Convolutional networks for the segmentation of intravascular ultrasound images: Evaluation on a multicenter dataset. Comput. Methods Programs Biomed. 2022, 215, 1065–1099. [Google Scholar] [CrossRef]
- Balakrishna, C.; Dadashzadeh, S.; Soltaninejad, S. Automatic detection of lumen and media in the IVUS images using U-Net with VGG16 Encoder. arXiv 2018, arXiv:1806.07554. [Google Scholar]
- Li, Y.C.; Shen, T.Y.; Chen, C.C.; Chang, W.T.; Lee, P.Y.; Huang, C.C.J. Automatic Detection of Atherosclerotic Plaque and Calcification From Intravascular Ultrasound Images by Using Deep Convolutional Neural Networks. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 1762–1772. [Google Scholar] [CrossRef] [PubMed]
- Porée, J.; Garcia, D.; Chayer, B.; Ohayon, J.; Cloutier, G. Noninvasive vascular elastography with plane strain incompressibility assumption using ultrafast coherent compound plane wave imaging. IEEE Trans. Med. Imaging 2015, 34, 2618–2631. [Google Scholar] [CrossRef] [PubMed]
- Gómez, A.; Tacheau, A.; Finet, G.; Lagache, M.; Martiel, J.L.; Le Floc’h, S.; Yazdani, S.K.; Elias-Zuñiga, A.; Pettigrew, R.I.; Cloutier, G.; et al. Intraluminal ultrasonic palpation imaging technique revisited for anisotropic characterization of healthy and atherosclerotic coronary arteries: A feasibility study. Ultrasound Med. Biol. 2019, 45, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Porée, J.; Roy Cardinal, M.H.; Cloutier, G. Two-dimensional affine model-based estimators for principal strain vascular ultrasound elastography with compound plane wave and transverse oscillation beamforming. Ultrasonics 2019, 91, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Porée, J.; Chayer, B.; Cardinal, M.H.R.; Cloutier, G. Parameterized Strain Estimation for Vascular Ultrasound Elastography With Sparse Representation. IEEE Trans. Med. Imaging 2020, 39, 3788–3800. [Google Scholar] [CrossRef]
- Le Floc’h, S.; Ohayon, J.; Tracqui, P.; Finet, G.; Gharib, A.M.; Maurice, R.L.; Cloutier, G.; Pettigrew, R.I. Vulnerable atherosclerotic plaque elasticity reconstruction based on a segmentation-driven optimization procedure using strain measurements: Theoretical framework. IEEE Trans. Med. Imaging 2009, 28, 1126–1137. [Google Scholar] [CrossRef]
- Le Floc’h, S.; Cloutier, G.; Saijo, Y.; Finet, G.; Yazdani, S.K.; Deleaval, F.; Rioufol, G.; Pettigrew, R.I.; Ohayon, J. A four-criterion selection procedure for atherosclerotic plaque elasticity reconstruction based on in vivo coronary intravascular ultrasound radial strain sequences. Ultrasound Med. Biol. 2012, 38, 2084–2097. [Google Scholar] [CrossRef]
- Maurice, R.L.; Ohayon, J.; Finet, G.; Cloutier, G. Adapting the Lagrangian speckle model estimator for endovascular elastography: Theory and validation with simulated radio-frequency data. J. Acoust. Soc. Am. 2004, 116, 1276–1286. [Google Scholar] [CrossRef] [PubMed]
- Porée, J.; Chayer, B.; Soulez, G.; Ohayon, J.; Cloutier, G. Noninvasive vascular modulography method for imaging the local elasticity of atherosclerotic plaques: Simulation and in vitro vessel phantom study. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2017, 64, 1805–1817. [Google Scholar] [CrossRef]
- Bouvier, A.; Deleval, F.; Doyley, M.; Yazdani, S.; Finet, G.; Le Floc’h, S.; Cloutier, G.; Pettigrew, R.; Ohayon, J. A direct vulnerable atherosclerotic plaque elasticity reconstruction method based on an original material-finite element formulation: Theoretical framework. Phys. Med. Biol. 2013, 58, 8457–8476. [Google Scholar] [CrossRef]
- Peña, E.; Cilla, M.; Latorre, Á.T.; Martínez, M.A.; Gómez, A.; Pettigrew, R.I.; Finet, G.; Ohayon, J. Emergent biomechanical factors predicting vulnerable coronary atherosclerotic plaque rupture. In Biomechanics of Coronary Atherosclerotic Plaque; Academic Press: Cambridge, MA, USA, 2021; pp. 361–380. [Google Scholar] [CrossRef]
- Glagov, S.; Weisenberg, E.; Zarins, C.; Stankunavicius, R.; Kolettis, G. Compensatory enlargement of human atherosclerotic coronary arteries. N. Engl. J. Med. 1987, 316, 1371–1375. [Google Scholar] [CrossRef]
- Le Floc’h, S. Modulographie Vasculaire: Application à l’Identification In-Vivo du Module de Young local des Plaques d’Athérosclérose. Ph.D. Thesis, Université Joseph-Fourier-Grenoble I, Saint-Martin-de-Re, France, 2009. [Google Scholar]
- Gasser, T.C.; Ogden, R.W.; Holzapfel, G.A. Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. J. R. Soc. Interface 2006, 3, 15–35. [Google Scholar] [CrossRef] [PubMed]
- Versluis, A.; Bank, A.J.; Douglas, W.H. Fatigue and plaque rupture in myocardial infarction. J. Biomech. 2006, 39, 339–347. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Sommer, G.; Gasser, C.T.; Regitnig, P. Determination of layer-specific mechanical properties of human coronary arteries with nonatherosclerotic intimal thickening and related constitutive modeling. Am. J. Physiol.-Heart Circ. Physiol. 2005, 289, H2048–H2058. [Google Scholar] [CrossRef] [PubMed]
- Skacel, P. Hyperfit: Sotfware for Fitting of Hyperelastic Constitutive Models. 2018. Available online: http://www.hyperfit.wz.cz (accessed on 16 September 2020).
- Dassault Systèmes Simulia Corp. ABAQUS/Standard User’s Manual, Version 6.14. 2014. [Google Scholar]
- Ramzy, D. Definition of hypertension and pressure goals during treatment (ESC-ESH Guidelines 2018). Eur. Soc. Cardiol. J. 2019, 17. [Google Scholar]
- Raghavan, M.; Ma, B.; Fillinger, M. Non-invasive determination of zero-pressure geometry of arterial aneurysms. Ann. Biomed. Eng. 2006, 34, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- MATLAB. R2021b, The MathWorks Inc.: Natick, MA, USA, 2021.
- Soleimanifard, S.; Abd-Elmoniem, K.Z.; Agarwal, H.K.; Tomas, M.S.; Sasano, T.; Vonken, E.; Youssef, A.; Abraham, M.R.; Abraham, T.P.; Prince, J.L. Identification of myocardial infarction using three-dimensional strain tensor fractional anisotropy. In Proceedings of the 2010 IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Rotterdam, The Netherlands, 14–17 April 2010; pp. 468–471. [Google Scholar] [CrossRef]
- Wang, D.; Chayer, B.; Destrempes, F.; Gesnik, M.; Tournoux, F.; Cloutier, G. Deformability of ascending thoracic aorta aneurysms assessed using ultrafast ultrasound and a principal strain estimator: In vitro evaluation and in vivo feasibility. Med. Phys. 2022, 49, 1759–1775. [Google Scholar] [CrossRef]
- Sumi, C.; Nakayama, K. A robust numerical solution to reconstruct a globally relative shear modulus distribution from strain measurements. IEEE Trans. Med. Imaging 1998, 17, 419–428. [Google Scholar] [CrossRef]
- Tacheau, A.; Le Floc’h, S.; Finet, G.; Doyley, M.M.; Pettigrew, R.I.; Cloutier, G.; Ohayon, J. The imaging modulography technique revisited for high-definition intravascular ultrasound: Theoretical framework. Ultrasound Med. Biol. 2016, 42, 727–741. [Google Scholar] [CrossRef]






| Tissue | [kPa] | [kPa] | [-] | [-] | [°] |
|---|---|---|---|---|---|
| Adventitia | 4.22 | 547.67 | 568.01 | 0.26 | ±61.80 |
| Media | 0.7 | 206.16 | 58.55 | 0.29 | ±28.35 |
| Intima | 3.41 | 109.10 | 101.04 | 0.21 | ±52.72 |
| Fibrotic | 4.79 | 17,654.91 | 0.51 | 1/3 | - |
| Lipid Core | 0.025 | 956.76 | 70 | 1/3 | - |
| Calcification | 1875 | - | - | - | - |
| Segmentation Index (SI) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Idealized Geometry | Real IVUS Geometry | ||||||||
| 65 m | 150 m | 300 m | Plaque 1 | Plaque 2 | Plaque 3 | ||||
| One SGV | 95.20 | 97.13 | 94.31 | 96.98 | 92.06 | 92.50 | 94.70 | 90.62 | |
| 92.33 | 94.55 | 96.02 | 93.99 | 94.66 | 96.23 | 94.63 | 86.04 | ||
| 93.49 | 93.60 | 97.65 | 90.74 | 94.99 | 97.08 | 94.59 | 94.47 | ||
| 97.65 | 94.40 | 93.07 | 98.48 | 86.78 | 96.32 | 94.45 | 92.27 | ||
| 86.07 | 93.77 | 97.86 | 97.73 | 93.43 | 97.46 | 94.39 | 93.29 | ||
| Combination of two SGVs | 95.87 | 97.63 | 97.09 | 98.61 | 96.14 | 97.28 | 97.10 | 95.22 | |
| 95.74 | 97.93 | 94.79 | 98.53 | 97.51 | 96.76 | 96.88 | 95.68 | ||
| 95.74 | 98.21 | 94.23 | 98.53 | 95.55 | 98.28 | 96.75 | 94.28 | ||
| 96.97 | 96.32 | 96.09 | 97.36 | 95.98 | 97.43 | 96.69 | 93.67 | ||
| 97.73 | 94.08 | 98.58 | 97.76 | 92.33 | 98.03 | 96.42 | 92.88 | ||
| 97.85 | 93.01 | 96.17 | 97.46 | 95.24 | 97.47 | 96.20 | 94.76 | ||
| 95.17 | 93.28 | 96.87 | 97.43 | 95.98 | 98.37 | 96.18 | 95.10 | ||
| 92.81 | 97.49 | 97.95 | 98.94 | 93.13 | 96.43 | 96.13 | 88.68 | ||
| 93.30 | 96.25 | 95.81 | 97.34 | 96.12 | 97.77 | 96.10 | 93.67 | ||
| 93.06 | 97.64 | 95.17 | 98.41 | 93.55 | 97.94 | 95.96 | 93.09 | ||
| 92.87 | 94.88 | 96.53 | 97.51 | 95.96 | 97.49 | 95.87 | 92.88 | ||
| 97.53 | 94.46 | 92.74 | 96.34 | 95.40 | 97.96 | 95.74 | 93.65 | ||
| 93.68 | 97.40 | 95.04 | 98.56 | 90.99 | 97.53 | 95.53 | 92.69 | ||
| 95.93 | 95.79 | 95.51 | 92.44 | 95.29 | 98.23 | 95.53 | 92.03 | ||
| 94.32 | 94.21 | 94.84 | 96.49 | 95.12 | 97.16 | 95.36 | 93.04 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Latorre, Á.T.; Martínez, M.A.; Cilla, M.; Ohayon, J.; Peña, E. Atherosclerotic Plaque Segmentation Based on Strain Gradients: A Theoretical Framework. Mathematics 2022, 10, 4020. https://doi.org/10.3390/math10214020
Latorre ÁT, Martínez MA, Cilla M, Ohayon J, Peña E. Atherosclerotic Plaque Segmentation Based on Strain Gradients: A Theoretical Framework. Mathematics. 2022; 10(21):4020. https://doi.org/10.3390/math10214020
Chicago/Turabian StyleLatorre, Álvaro T., Miguel A. Martínez, Myriam Cilla, Jacques Ohayon, and Estefanía Peña. 2022. "Atherosclerotic Plaque Segmentation Based on Strain Gradients: A Theoretical Framework" Mathematics 10, no. 21: 4020. https://doi.org/10.3390/math10214020
APA StyleLatorre, Á. T., Martínez, M. A., Cilla, M., Ohayon, J., & Peña, E. (2022). Atherosclerotic Plaque Segmentation Based on Strain Gradients: A Theoretical Framework. Mathematics, 10(21), 4020. https://doi.org/10.3390/math10214020







