Preparation of TiO2-Poly(3-Chloro-2-Hydroxypropyl Methacrylate) Nanocomposite for Selective Adsorption and Degradation of Dyes
Abstract
:1. Introduction
2. E.xperimental Section
2.1. Materials
2.2. Measurements and Instrument
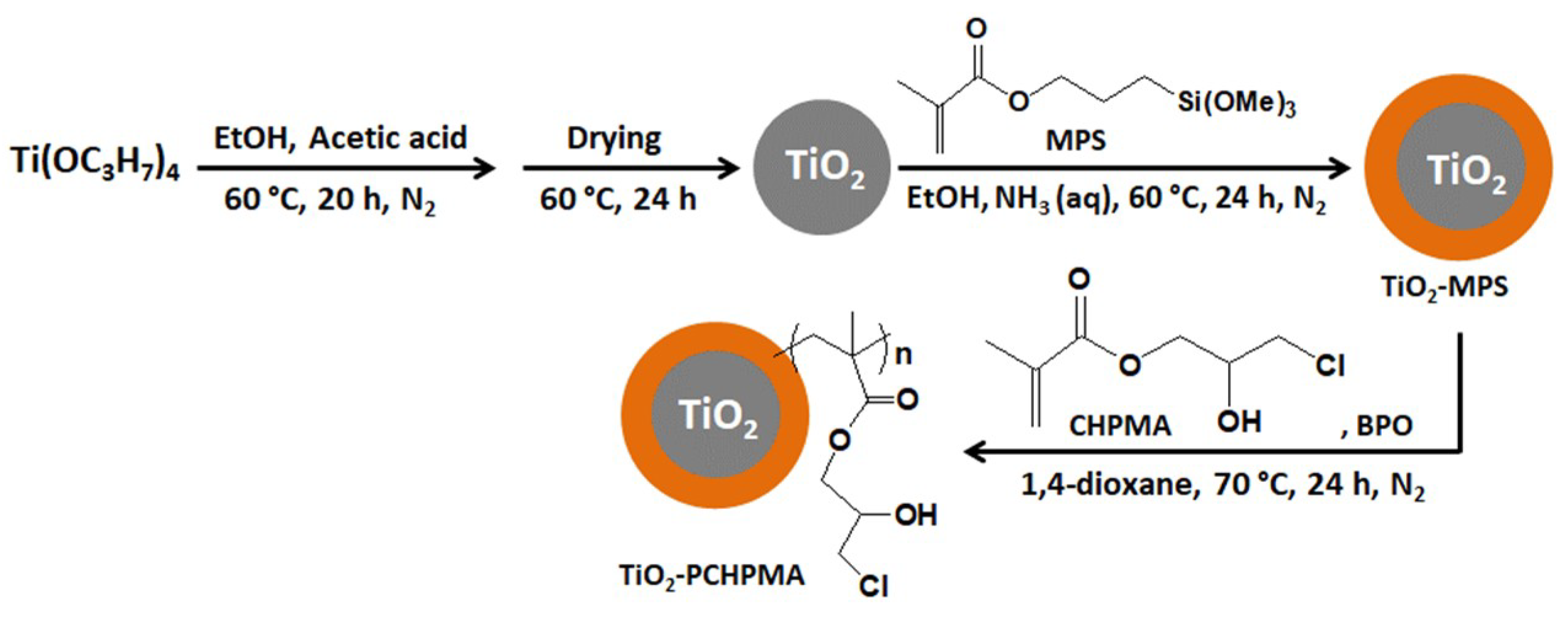
2.3. Preparation of TiO2 Nanoparticles from TIPOT
2.4. Preparation of TiO2-MPS
2.5. Preparation of TiO2-PCHPMA
2.6. Adsorption Experiment
2.7. Degradation Experiment
3. Results and Discussion
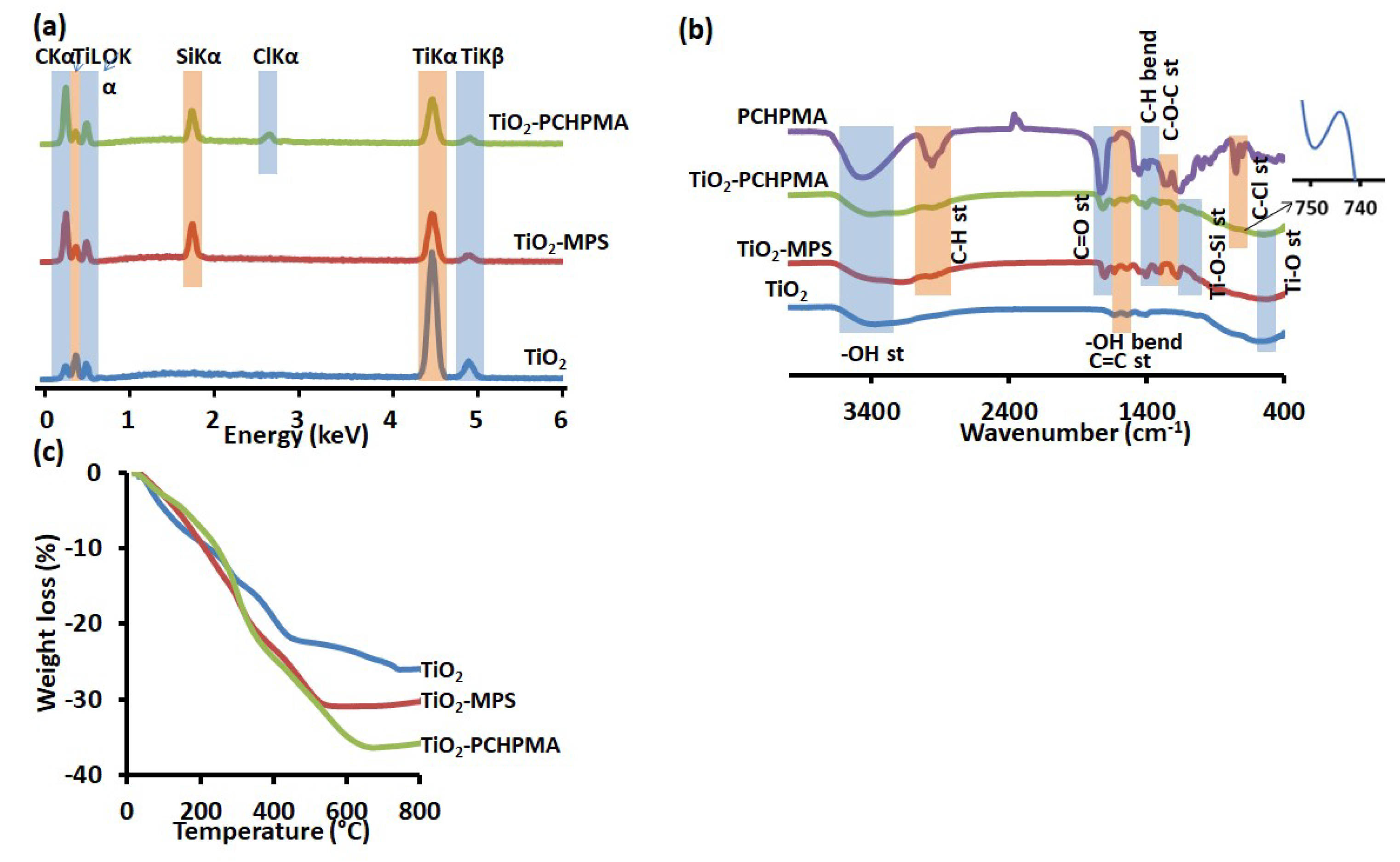
3.1. Characterization
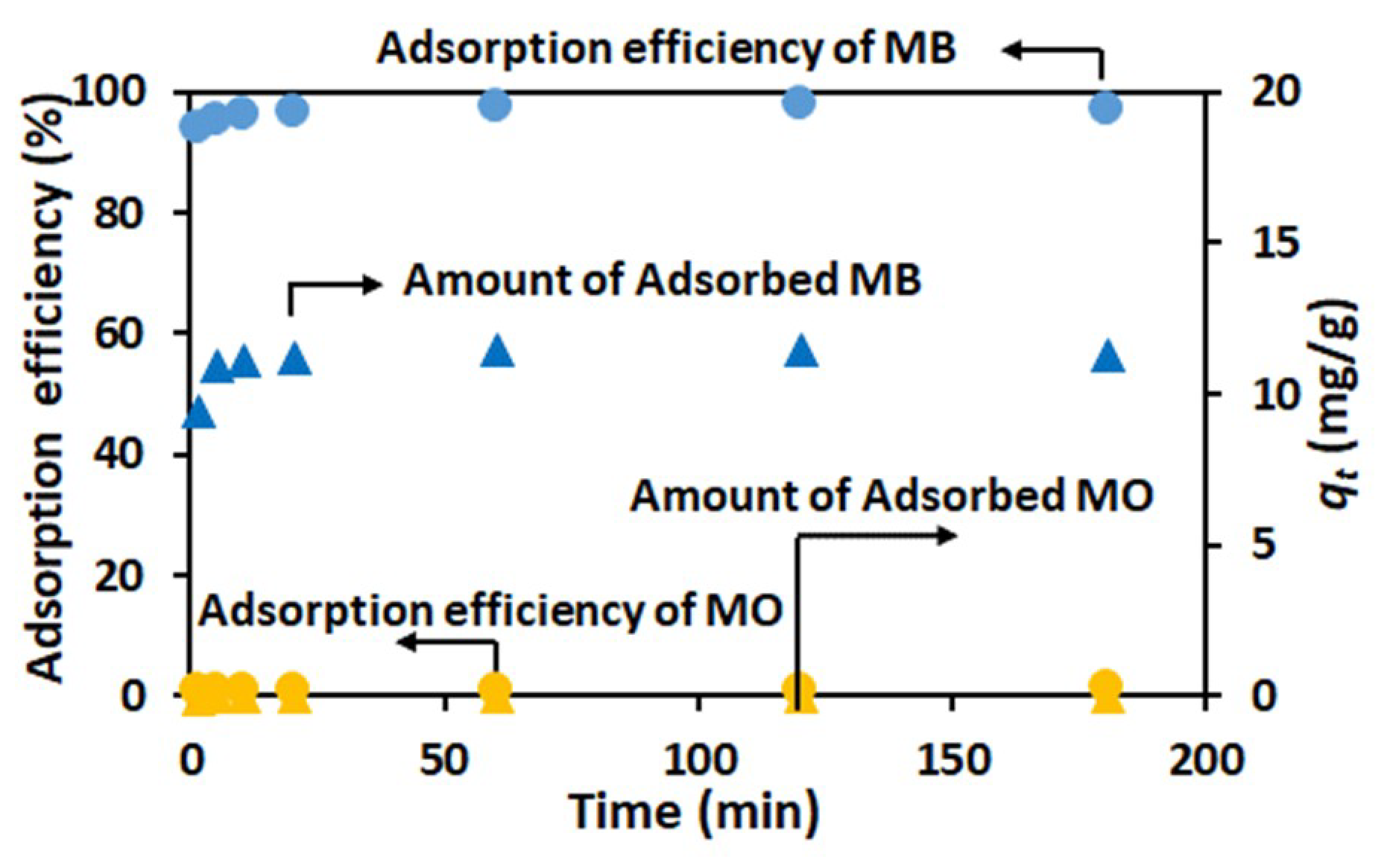
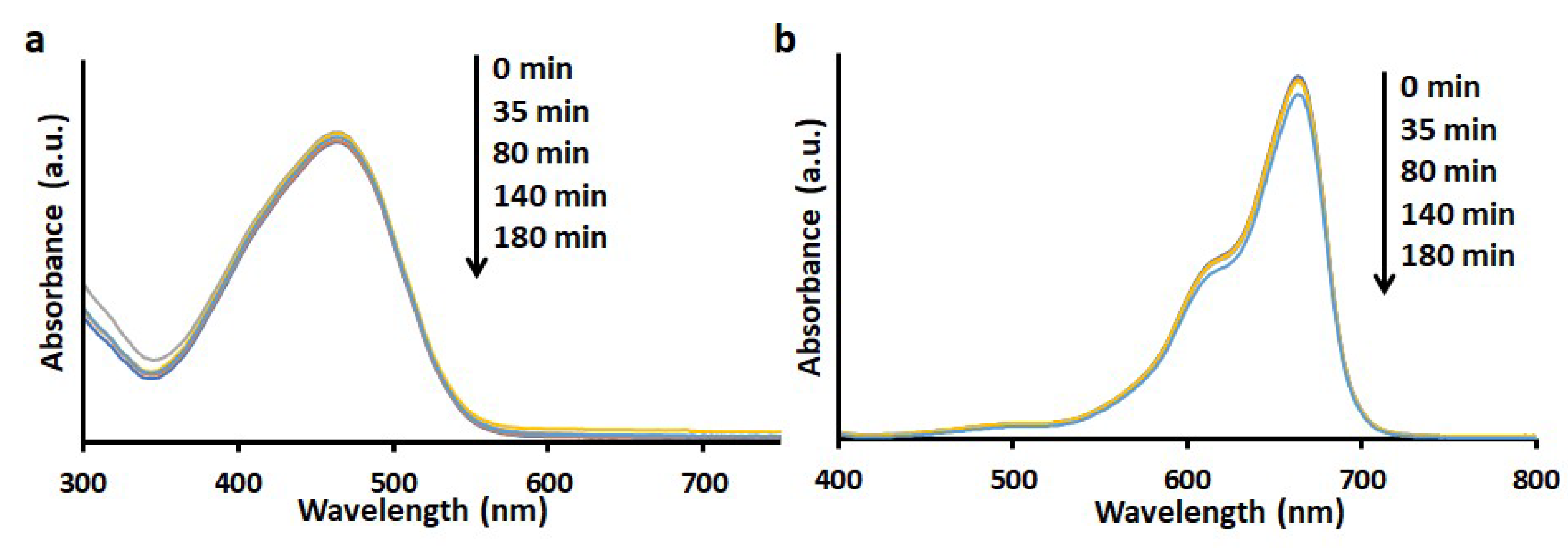
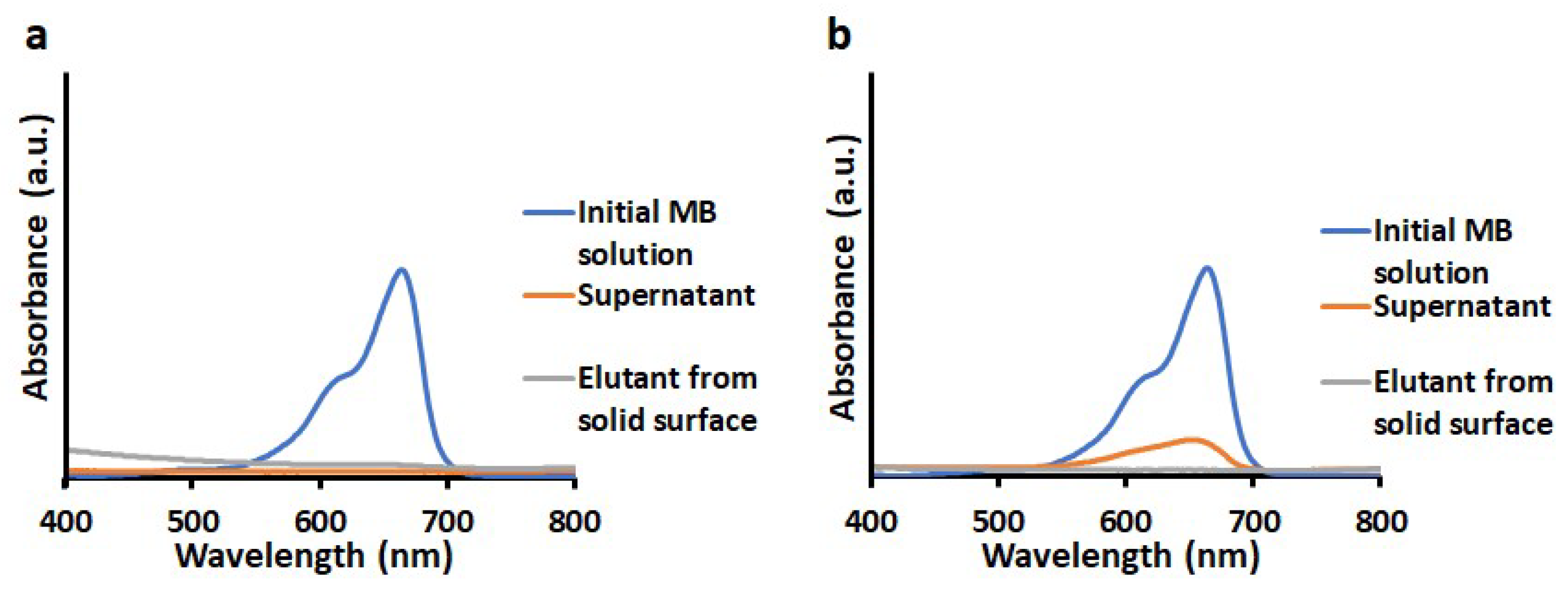
3.2. Adsorption of Dyes
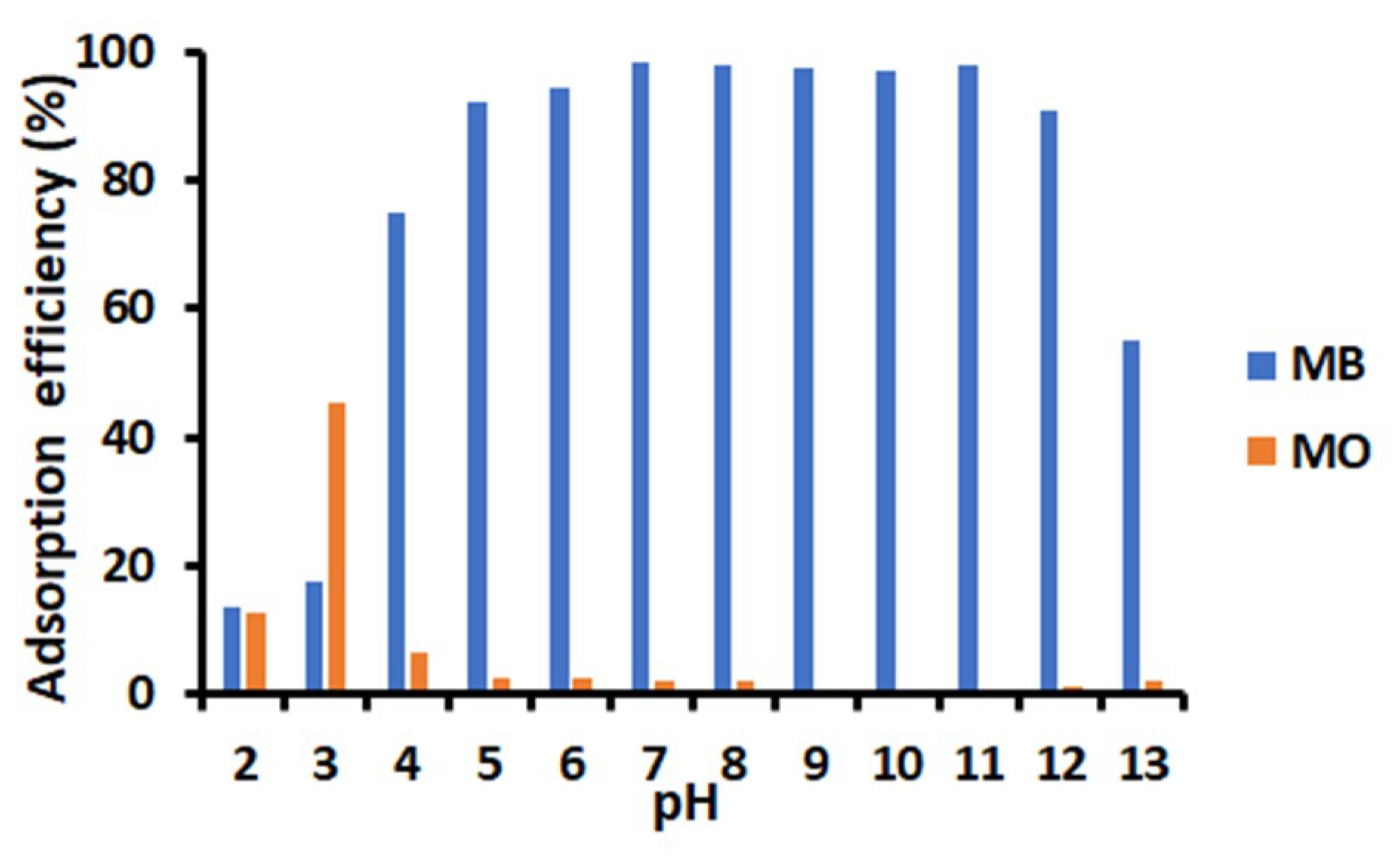
3.3. Effect of pH on the Adsorption of Dyes onto TiO2-PCHPMA
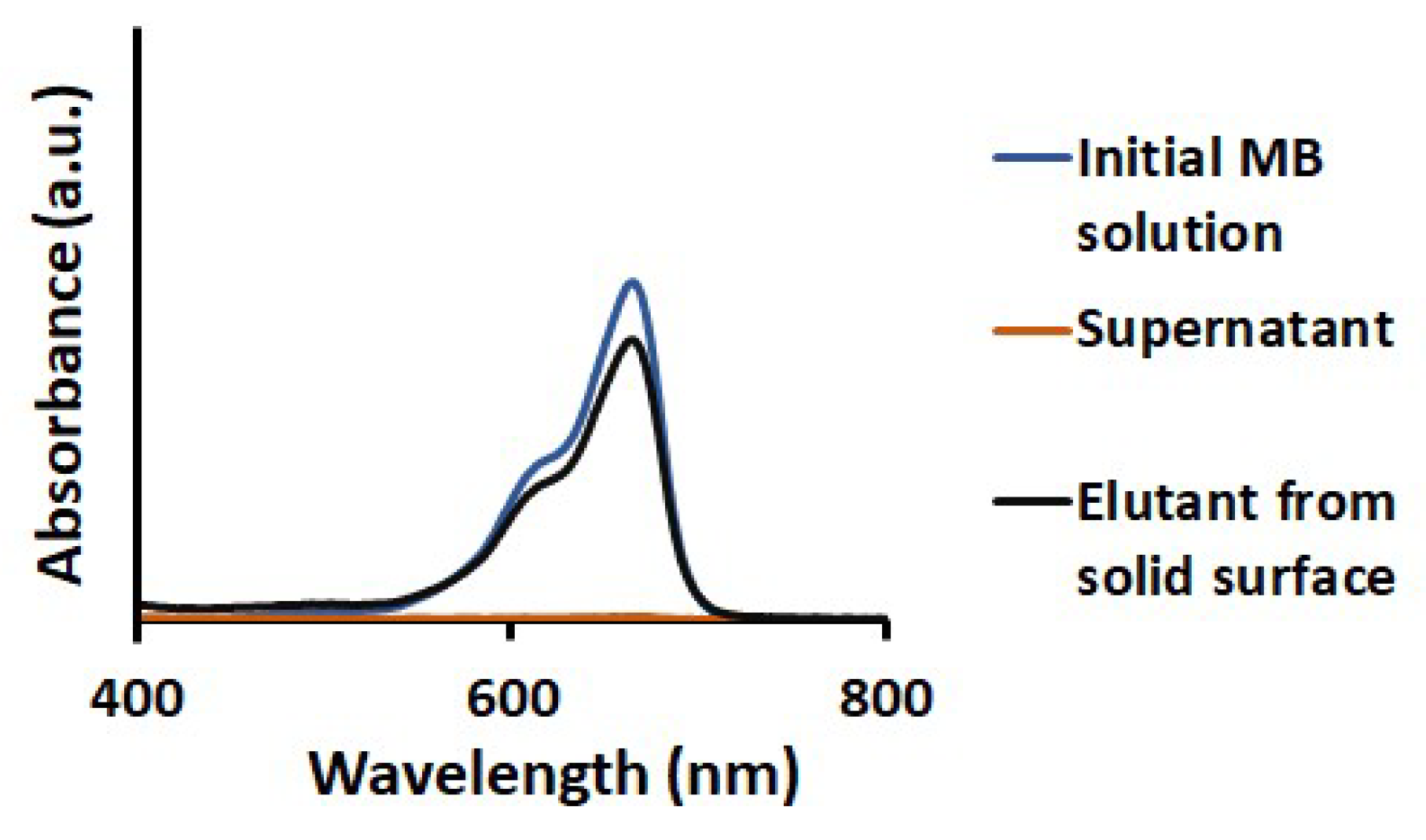
3.4. Desorption of MB
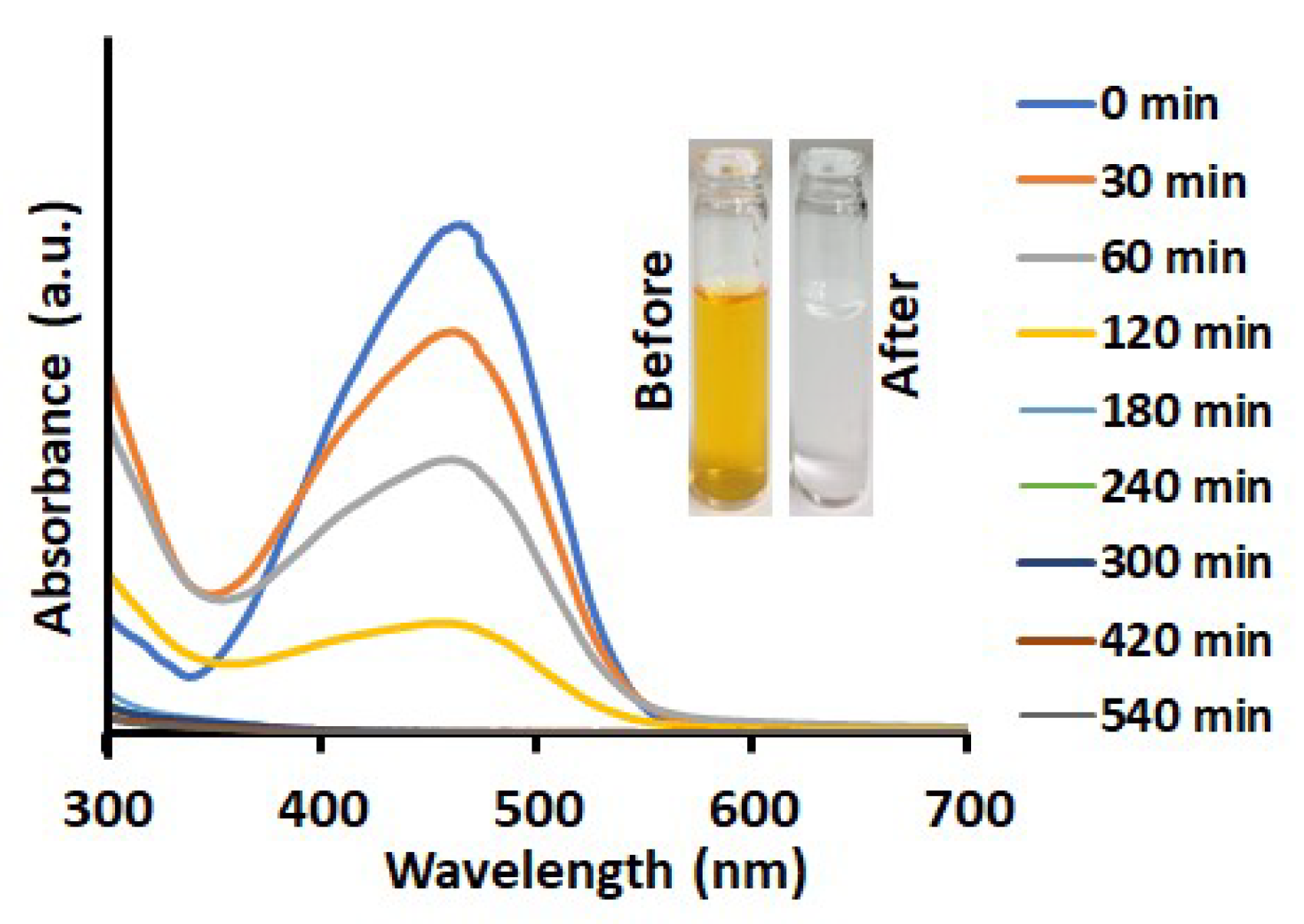
3.5. Degradation of Dyes
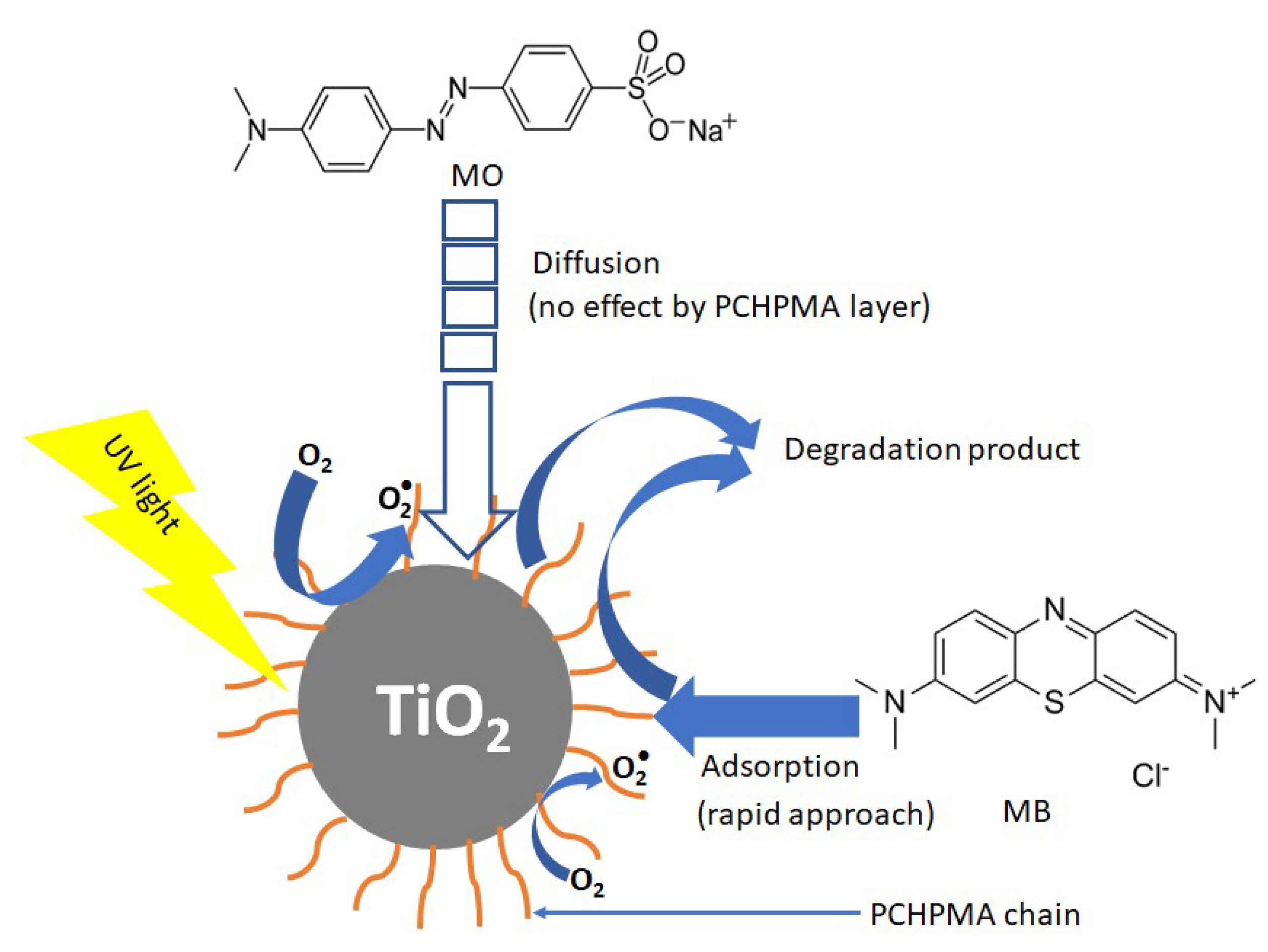
3.6. Mechanism of Photocatalysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robinson, T.; McMulan, G.; Marchant, R.; Nigam, P. Remediation of dyes in textile effluents: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef]
- Pearce, C.I.; Lioyd, J.R.; Guitrie, J.T. The removal of color from textile wastewater using whole bacterial cells: A review. Dyes Pigm. 2003, 58, 179–196. [Google Scholar] [CrossRef]
- Karan, C.K.; Bhattacharjee, M. Self-healing and moldable metallogels as the recyclable materials for selective dye adsorption and separation. ACS Appl. Mater. Interfaces 2016, 8, 5526–5535. [Google Scholar] [CrossRef] [PubMed]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C: Photochem. Rev. 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Li, H.; Bian, Z.; Zhu, J.; Huo, Y.; Li, H.; Lu, Y. Mesoporous Au/TiO2 nanocomposites with enhanced photocatalytic activity. J. Am. Chem. Soc. 2007, 129, 4538–4539. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Chen, C.; Wang, Y.; Ma, W.; Zhao, J.; Rajh, T.; Zang, L. Enhanced photocatalytic degradation of dye pollutants under visible irradiation on Al(III)-modified TiO2: Structure, interaction, and interfacial electron transfer. Environ. Sci. Technol. 2008, 42, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Naya, S.I.; Nikawa, T.; Kimura, K.; Tada, H. Rapid and complete removal of nonylphenol by gold nanoparticle/rutile titanium(IV) oxide plasmon photocatalyst. ACS Catal. 2013, 3, 903–907. [Google Scholar] [CrossRef]
- Sakthivel, S.; Kisch, H. Daylight photocatalysis by carbon-modified titanium dioxide. Angew. Chem. Int. Ed. 2003, 42, 4908–4911. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Ai, Z.; Jia, F.; Zhang, L.; Fan, X.; Zou, Z. Low temperature preparation and visible light photocatalytic activity of mesoporous carbon-doped crystalline TiO2. Appl. Catal. B 2007, 69, 138–144. [Google Scholar] [CrossRef]
- Dong, F.; Guo, S.; Wang, H.; Li, X.; Wu, Z. Enhancement of the visible light photocatalytic activity of c-doped TiO2 nanomaterials prepared by a green synthetic approach. J. Phys. Chem. C 2011, 115, 13285–13292. [Google Scholar] [CrossRef]
- Wang, W.; Serp, P.; Kalck, P.; Faria, J.L. Photocatalytic degradation of phenol on MWNT and titania composite catalysts prepared by a modified sol–gel method. Appl. Catal. B 2005, 56, 305–312. [Google Scholar] [CrossRef]
- Woan, B.K.; Pyrgiotakis, G.; Sigmund, W. Photocatalytic carbon-nanotube–TiO2 composites. Adv. Mater. 2009, 21, 2233–2239. [Google Scholar] [CrossRef]
- Vijayan, B.K.; Dimitrijevic, N.M.; Finkelstein-Shapiro, D.; Wu, J.; Gray, K.A. Coupling titania nanotubes and carbon nanotubes to create photocatalytic nanocomposites. ACS Catal. 2012, 2, 223–229. [Google Scholar] [CrossRef]
- Tryba, B.; Morawski, A.W.; Inagaki, M. Application of TiO2-mounted activated carbon to the removal of phenol from water. Appl. Catal. B 2003, 41, 427–433. [Google Scholar] [CrossRef]
- Asiltürk, M.; Şener, S. TiO2-activated carbon photocatalysts: Preparation, characterization and photocatalytic activities. Chem. Eng. J. 2012, 180, 354–363. [Google Scholar] [CrossRef]
- Alam, M.G.; Tawfik, A.; Ookawara, S. Enhancement of photocatalytic activity of TiO2 by immobilization on activated carbon for degradation of pharmaceuticals. J. Environ. Chem. Eng. 2016, 4, 1929–1937. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.R.; Fu, X.; Xu, Y.J. TiO2-graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2-graphene truly different from other TiO2-carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.T.; Vijayan, B.K.; Gray, K.A.; Hersam, M.C. Minimizing graphene defects enhances titania nanocomposite-based photocatalytic reduction of CO2 for improved solar fuel production. Nano Lett. 2011, 11, 2865–2870. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Sheng, G.; Chen, C.; Wang, X. Enhanced photocatalytic degradation of methylene blue under visible irradiation on graphene@TiO2 dyade structure. Appl. Catal. B 2012, 111–112, 302–308. [Google Scholar] [CrossRef]
- Liu, B.J.; Bai, H.; Wang, Y.; Liu, Z.; Zhang, X.; Sun, D.D. Self-assembling TiO2 nanorods on large graphene oxide sheets at a two-phase interface and their anti-recombination in photocatalytic applications. Adv. Funct. Mater. 2010, 20, 4175–4181. [Google Scholar] [CrossRef]
- Sang, Y.; Zhao, Z.; Tian, J.; Hao, P.; Jiang, H.; Liu, H.; Claverie, J.P. Enhanced photocatalytic property of reduced graphene oxide/TiO2 nanobelt surface heterostructures constructed by an in situ photochemical reduction method. Small 2014, 10, 3775–3782. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yu, L.; Lin, Z.; Yang, G. Reduced TiO2 graphene oxide heterostructure as broad spectrum-driven efficient water-splitting photocatalysts. ACS Appl. Mater. Interfaces 2016, 8, 8536–8545. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Dan, Y. Photocatalytic activity of poly(3-hexylthiophene)/titanium dioxide composites for degrading methyl orange. Sol Energy Mater Sol Cells 2010, 94, 1658–1664. [Google Scholar] [CrossRef]
- Zhang, H.; Zong, R.; Zhao, J.; Zhu, Y. Dramatic visible photocatalytic degradation performances due to synergetic effect of TiO2 with PANI. Environ. Sci. Technol. 2008, 42, 3803–3807. [Google Scholar] [CrossRef] [PubMed]
- Min, S.X.; Wang, F.; Feng, L.; Tong, Y.C.; Yang, Z.R. Synthesis and photocatalytic activity of TiO2/conjugated polymer complex nanoparticles. Chin. Chem. Lett. 2008, 19, 742–746. [Google Scholar] [CrossRef]
- Dimitrijevic, N.M.; Tepavcevic, S.; Liu, Y.; Rajh, T.; Silver, S.C.; Tiede, D.M. Nanostructured TiO2/polypyrrole for visible light photocatalysis. J. Phys. Chem. C 2013, 117, 15540–15544. [Google Scholar] [CrossRef]
- Gao, F.; Hou, X.; Wang, A.; Chu, G.; Wu, W.; Chen, J.; Zou, H. Preparation of polypyrrole/TiO2 nanocomposites with enhanced photocatalytic performance. Particuology 2016, 26, 73–78. [Google Scholar] [CrossRef]
- Liao, G.; Chen, S.; Quan, X.; Chen, H.; Zhang, Y. Photonic crystal coupled TiO2/polymer hybrid for efficient photocatalysis under visible light irradiation. Environ. Sci. Technol. 2010, 44, 3481–3485. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Li, X.; Li, X.; Wang, D.; An, J.; Li, X. Visible light photocatalytic activity of TiO2 nanoparticles modified by pre-oxidized polyacrylonitrile. Catal. Commun. 2012, 26, 239–243. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, J.; Luo, Q.; Li, X.; Duan, Y.; An, J. Characterization and photocatalytic activity of poly(3-hexylthiophene)-modified TiO2 for degradation of methyl orange under visible light. J. Hazard. Mater. 2009, 169, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Mahanta, D.; Manna, U.; Madras, G.; Patil, S. Multilayer self-assembly of TiO2 nanoparticles and polyaniline-grafted-chitosan copolymer (CPANI) for photocatalysis. ACS Appl. Mater. Inter. 2011, 3, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, D.; Cheng, G.; Luo, Q.; An, J.; Wang, Y. Preparation of polyaniline-modified TiO2 nanoparticles and their photocatalytic activity under visible light illumination. Appl. Catal. B 2008, 81, 267–273. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Y.; Li, X.; Luo, Q.; An, J.; Yue, J. Sunlight photocatalytic activity of polypyrrole–TiO2 nanocomposites prepared by ‘in situ’ method. Catal. Commun. 2008, 9, 1162–1166. [Google Scholar] [CrossRef]
- Tang, Q.; Lin, J.; Wu, Z.; Wu, J.; Huang, M.; Yang, Y. Preparation and photocatalytic degradability of TiO2/polyacrylamide composite. Eur. Polym. J. 2007, 43, 2214–2220. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, M.; Dong, W.; Cui, G.; Ren, Z.; Wang, W. Highly efficient photocatalytic degradation of methylene blue by PoPD/TiO2 nanocomposite. PLoS ONE 2017, 12, e0174104. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Min, S.; Han, Y.; Feng, L. Visible-light-induced photocatalytic degradation of methylene blue with polyaniline-sensitized TiO2 composite photocatalysts. Supperlatt. Microstruct. 2010, 48, 170–180. [Google Scholar] [CrossRef]
- Xu, S.; Gu, L.; Wu, K.; Yang, H.; Song, Y.; Jiang, L.; Dan, Y. The influence of the oxidation degree of poly(3-hexylthiophene) on the photocatalytic activity of poly(3-hexylthiophene)/TiO2 composites. Sol. Energy Mater. Sol. Cells. 2012, 96, 286–291. [Google Scholar] [CrossRef]
- Luo, Q.; Wang, X.; Wang, D.; An, J.; Li, X.; Yin, R. TiO2/cyclized polyacrylonitrile hybridized nanocomposite: An efficient visible-light photocatalyst prepared by a facile “in situ” approach. Mater. Sci. Eng. B 2015, 199, 96–104. [Google Scholar] [CrossRef]
- Muthirulan, P.; Nirmala, C.K.; Sundaram, M.M. Facile synthesis of novel hierarchical TiO2@Poly(o-phenylenediamine) core–shell structures with enhanced photocatalytic performance under solar light. J. Environ. Chem. Eng. 2013, 1, 620–627. [Google Scholar] [CrossRef]
- Kangwansupamonkon, W.; Jitbunpot, W.; Kiatkamjornwong, S. Photocatalytic efficiency of TiO2/poly[acrylamide-co-(acrylic acid)] composite for textile dye degradation. Polym. Degrad. Stabil. 2010, 95, 1894–1902. [Google Scholar] [CrossRef]
- Fukahori, S.; Ichiura, H.; Kitaoka, T.; Tanaka, H. Photocatalytic decomposition of bisphenol A in water using composite TiO2-zeolite sheets prepared by a papermaking technique. Environ. Sci. Technol. 2003, 37, 1048–1051. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Wen, L.; Nakata, K.; Zhao, X.; Liu, S.; Ochiai, T.; Murakami, T.; Fujishima, A. Polymeric adsorption of methylene blue in TiO2 colloids-highly sensitive thermochromism and selective photocatalysis. Chem. Eur. J. 2012, 18, 12705–12711. [Google Scholar] [CrossRef] [PubMed]
- Zang, G.; Choi, W.; Kim, S.H.; Hong, S.B. Selective photocatacatalytic degradation of aquatic pollutants by titania encapsulated into FAU-type zeolites. J. Hazard. Mater. 2011, 188, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Zhu, L.; Liu, G.; Yu, H.; Tang, H. Enhanced photocatalytic degradation and selective removal of nitrophenol by using surface molecular imprinted titania. Environ. Sci. Technol. 2008, 42, 1687–1692. [Google Scholar] [CrossRef] [PubMed]
- Inumaru, K.; Murashima, M.; Kasahara, T.; Yamanaka, S. Enhanced photocatalytic decomposition of 4-nonylphenol by surface-organografted TiO2: a combination of molecular selective adsorption and photocatalysis. Appl. Catal. B 2004, 52, 275–280. [Google Scholar] [CrossRef]
- Nakamura, K.J.; Ide, Y.; Ogawa, M. Molecular recognitive photocatalytic decomposition on mesoporous silica coated TiO2 particle. Mater. Lett. 2011, 65, 24–26. [Google Scholar] [CrossRef]
- Bao, L.; Meng, M.; Sun, K.; Li, W.; Zhao, D.; Li, H.; He, M. Selective adsorption and degradation of rhodamine B with modified titanium dioxide photocatalyst. J. Appl. Polym. Sci. 2014, 40890, 1–12. [Google Scholar] [CrossRef]
- Choi, H.; Kim, Y.J.; Varma, R.S.; Dionysiou, D.D. Thermally stable nanocrystalline TiO2 photocatalysts synthesized via sol-gel methods modified with ionic liquid and surfactant molecules. Chem. Mater. 2006, 18, 5377–5384. [Google Scholar] [CrossRef]
- Kumar, S.G.; Devi, L.G. Review on modified TiO2 photocatalysis under UV/visible light: selected results and related mechanisms on interfacial charge carrier transfer dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef] [PubMed]
- Lazar, M.A.; Walid, A.; Daoud, W.A. Achieving selectivity in TiO2-based photocatalysis. RSC Adv. 2013, 3, 4130–4140. [Google Scholar] [CrossRef]
- Robert, D.; Piscopo, A.; Weber, J.V. First approach of the selective treatment of water by heterogeneous photocatalysis. Environ. Chem. Lett. 2004, 2, 5–8. [Google Scholar] [CrossRef]
- Lazar, M.A.; Daoud, W.A. Selective adsorption and photocatalysis of low-temperature base-modified anatase nanocrystals. RSC Adv. 2012, 2, 447–452. [Google Scholar] [CrossRef]
- Genevrier, A.C.; Boissiere, C.; Nicole, L.; Grosso, D. Distance dependence of the photocatalytic efficiency of TiO2 revealed by in situ ellipsometry. J. Am. Chem. Soc. 2012, 134, 10761–10764. [Google Scholar] [CrossRef] [PubMed]
- Gokaltun, A.; Çelebi, B.; Tuncel, A. Octadecylamine-attached poly(3-chloro-2-hydroxypropyl methacrylate-co-ethylene dimethacrylate) microspheres as a new stationary phase for microbore reversed phase chromatography. Anal. Meth. 2014, 6, 5712–5719. [Google Scholar] [CrossRef]
- Molina, E.A.; Néstor, R.V.; Rodríguez, D.M.; Figueiras, C.C. Synthesis and characterization of TiO2 modified with polystyrene and poly(3-chloro-2hydroxypropyl methacrylate) as adsorbents for the solid phase extraction of organophosphorous pesticides. J. Chem. 2016, 2016, 1–11. [Google Scholar] [CrossRef]
- Cozzoli, P.D.; Kornowski, A.; Weller, H. Low-temperature synthesis of soluble and processable organic-capped anatase TiO2 nanorods. J. Am. Chem. Soc. 2003, 125, 14539–14548. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Mao, H.; Wang, C.; Fu, S. Dispersibility and hydrophobicity analysis of titanium dioxide nanoparticles grafted with silane coupling agent. J. Ind. Eng. Chem. Res. 2011, 50, 11930–11934. [Google Scholar] [CrossRef]
- Yuvaraj, H.; Kim, W.S.; Kim, J.T.; Kang, I.P.; Gal, Y.S.; Kim, S.W.; Lim, K.T. Synthesis of poly(methyl methacrylate) encapsulated TiO2 nanocomposite particles in supercritical CO2. Mol. Cryst. Liq. Cryst. 2009, 514, 355–365. [Google Scholar] [CrossRef]
- Wang, J.; Sun, W.; Zhang, Z.; Jiang, Z.; Wang, X.; Xu, R.; Li, R.; Zhang, X. Preparation of Fe-doped mixed crystal TiO2 catalyst and investigation of its sonocatalytic activity during degradation of azo fuchsine under ultrasonic irradiation. J. Colloid Interface Sci. 2008, 320, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Liu, H.; Dong, J.; Wei, J.; Zhang, Y. Preparation and Characterization of Ce/N-Codoped TiO2 Particles for Production of H2 by Photocatalytic Splitting Water Under Visible Light. Catal. Lett. 2010, 135, 219–225. [Google Scholar] [CrossRef]
- Ba-abbad, M.M.; Kadhum, A.A.H.; Mohamad, A.B.; Takriff, M.S.; Sopian, K. Synthesis and catalytic activity of TiO2 nanoparticles for photochemical oxidation of concentrated chlorophenols under direct solar radiation. Int. J. Electrochem. Sci. 2012, 7, 4871–4888. [Google Scholar]
- Wang, Y.Q.; Chen, S.G.; Tang, X.H.; Palchik, O.; Zaban, A.; Koltypin, Y.; Gedanken, A. Mesoporous titanium dioxide: sonochemical synthesis and application in dye-sensitized solar cells. J. Mater. Chem. 2001, 11, 521–526. [Google Scholar] [CrossRef]
- Yu, J.C.; Zhang, L.; Yu, J. Direct sonochemical preparation and characterization of highly active mesoporous TiO2 with a bicrystalline framework. Chem. Mater. 2002, 14, 4647–4653. [Google Scholar] [CrossRef]
- Pazokifard, S.; Mirabedini, S.M.; Esfandeh, M.; Mohseni, M.; Ranjbar, Z. Silane grafting of TiO2 nanoparticles: dispersibility and photoactivity in aqueous solutions. Surf. Interface Anal. 2012, 44, 41–47. [Google Scholar] [CrossRef]
- Siddiquey, I.A.; Ukaji, E.; Furusawa, T.; Sato, M.; Suzuki, N. The effects of organic surface treatment by methacryloxypropyltrimethoxysilane on the photostability of TiO2. Mater. Chem. Phys. 2007, 105, 162–168. [Google Scholar] [CrossRef]
- Pelouchova, H.; Janda, P.; Weber, J.; Kavan, L. Charge transfer reductive doping of single crystal TiO2 anatase. J. Electro. Chem. 2004, 566, 73–83. [Google Scholar] [CrossRef]
- Renault, C.; Nicole, L.; Sanchez, C.; Costentin, C.; Balland, V.; Limoges, B. Unraveling the charge transfer/electron transport in mesoporous semiconductive TiO2 films by voltabsorptometry. Phys. Chem. Chem. Phys. 2015, 17, 10592–10607. [Google Scholar] [CrossRef] [PubMed]

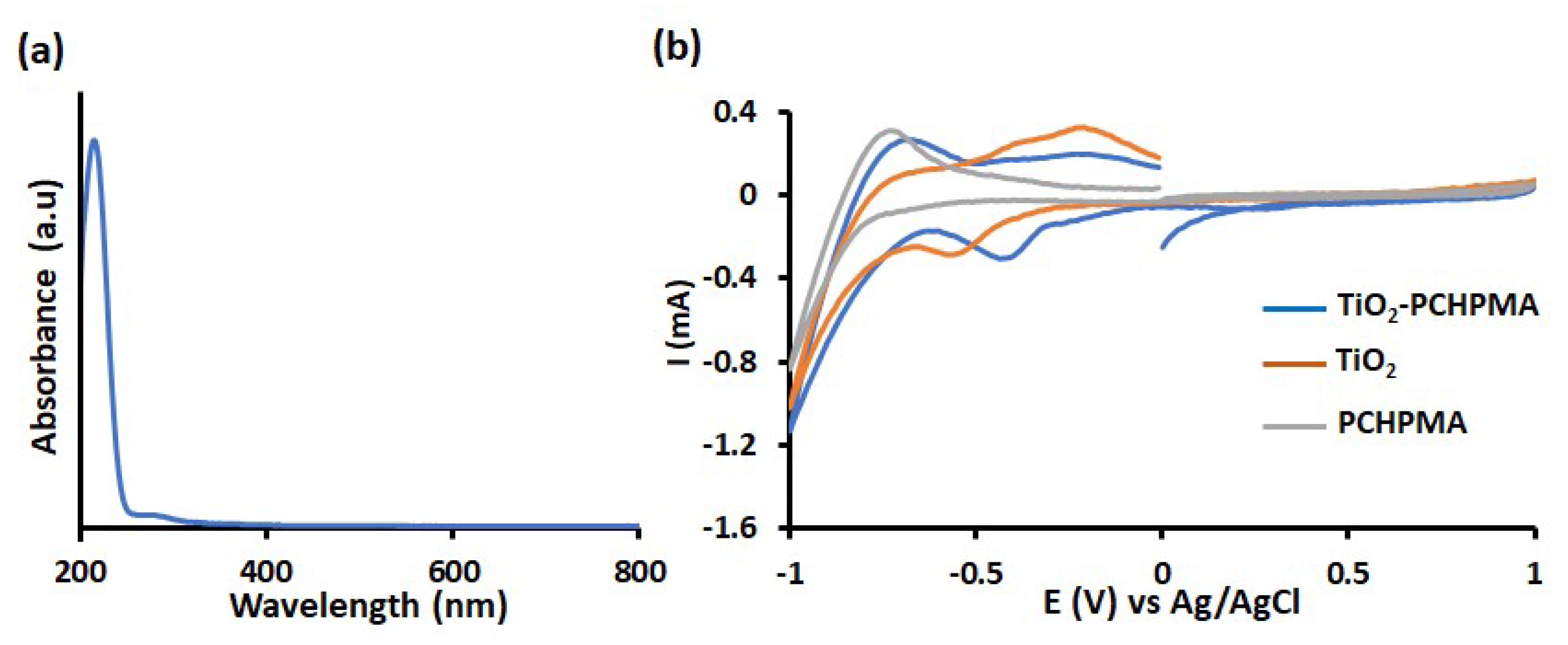
| Composites | TiO2 | TiO2-PCHPMA | ||
|---|---|---|---|---|
| Before Adsorption | After Adsorption | Before Adsorption | After Adsorption | |
| Zeta potential (mV) | −9 | −7 | −26 | −10 |
| Catalysts | Dye | Adsorption | Degradation | References | ||
|---|---|---|---|---|---|---|
| Conditions: concentration of dye (mg/L), composite (g/L) and adsorption time (min) | Efficiency (%) | Conditions: UV irradiation time (min) | Efficiency (%) | |||
| TiO2/poly[acrylamide-co-(acrylic acid)] | MB | 5, 0.2, and 15 | 87 | 40 | 91 | [40] |
| TiO2 treated with Na2CO3 | MO | 10, 1, and 30 | 23 | 120 | > 99 | [52] |
| TiO2 treated with NH4OH | MB | 10, 1, and 30 | 96 | 60 | > 99 | |
| TiO2-PCHPMA | MB | 10, 0.8, and 20 | 99 | 180 | 94 | This work |
| MO | 10, 0.8, and 20 | 1< | 180 | >99 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossan, M.S.; Ochiai, B. Preparation of TiO2-Poly(3-Chloro-2-Hydroxypropyl Methacrylate) Nanocomposite for Selective Adsorption and Degradation of Dyes. Technologies 2018, 6, 92. https://doi.org/10.3390/technologies6040092
Hossan MS, Ochiai B. Preparation of TiO2-Poly(3-Chloro-2-Hydroxypropyl Methacrylate) Nanocomposite for Selective Adsorption and Degradation of Dyes. Technologies. 2018; 6(4):92. https://doi.org/10.3390/technologies6040092
Chicago/Turabian StyleHossan, M. Shamim, and Bungo Ochiai. 2018. "Preparation of TiO2-Poly(3-Chloro-2-Hydroxypropyl Methacrylate) Nanocomposite for Selective Adsorption and Degradation of Dyes" Technologies 6, no. 4: 92. https://doi.org/10.3390/technologies6040092
APA StyleHossan, M. S., & Ochiai, B. (2018). Preparation of TiO2-Poly(3-Chloro-2-Hydroxypropyl Methacrylate) Nanocomposite for Selective Adsorption and Degradation of Dyes. Technologies, 6(4), 92. https://doi.org/10.3390/technologies6040092






