Advances in Magnesia–Dolomite Refractory Materials: Properties, Emerging Technologies, and Industrial Applications: A Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Criteria for Selection and Exclusion of Research
- Keyword Focus—Selected studies must explicitly address refractory systems based on magnesia, dolomite, or basic MgO–Dolomite compositions, combined with technical properties such as mechanical performance, thermal shock resistance, corrosion resistance, or recycling.
- Publication Date Range—Only publications between 2018 and 2025 were considered, ensuring a current perspective on recent advances.
- Subject Area Filtering—To maintain disciplinary focus, publications from non-engineering or non-materials domains such as dentistry, sociology, agriculture, mathematics, and business were excluded.
- Document Type—Only peer-reviewed journal articles in English were included. Non-article types such as patents, proceedings, book chapters, and technical reports were excluded to ensure consistency in the scientific rigor of the dataset.
- Thematic Relevance—Papers unrelated to refractory performance or focused on unrelated technologies (fuel cells, membranes, semiconductors, thin films, or medical applications) were excluded, as these do not align with the structural and thermomechanical objectives of this study.
- Final Search Code:
- The search string used was the following Listing 1:
| Listing 1. Scopus search string for magnesia—dolomite refractory materials |
| TITLE-ABS-KEY("magnesia refractory*" OR "dolomite refractory*" OR "basic refractory*" OR "MgO-CaO*") AND TITLE-ABS-KEY("mechanical properties" OR "thermal shock" OR "corrosion resistance" OR "slag resistance" OR "hydration resistance" OR "sustainability" OR "recycling") AND PUBYEAR > 2017 AND PUBYEAR < 2026 AND ( LIMIT-TO ( DOCTYPE,"ar" ) OR LIMIT-TO ( DOCTYPE,"English" ) OR EXCLUDE ( DOCTYPE,"p" ) OR EXCLUDE ( DOCTYPE,"k" ) OR EXCLUDE ( DOCTYPE,"d" ) OR EXCLUDE ( DOCTYPE,"Fuel Cells" ) OR EXCLUDE ( DOCTYPE,"Membranes" ) OR EXCLUDE ( DOCTYPE,"Semiconductors" ) OR EXCLUDE ( DOCTYPE,"Catalysts" ) OR EXCLUDE ( DOCTYPE,"Medical Applications" ) OR EXCLUDE ( DOCTYPE,"Thin Films" ) ) AND ( EXCLUDE ( SUBJAREA,"DENT" ) OR EXCLUDE ( SUBJAREA,"SOCI" ) OR EXCLUDE ( SUBJAREA,"AGRI" ) OR EXCLUDE ( SUBJAREA,"MATH" ) OR EXCLUDE ( SUBJAREA,"BUSI" ) ) |
2.2. Analysis Guide for Systemic Review of MgO–Dolomite Refractory Research
2.3. Bibliometric Trends in Magnesia–Dolomite Refractory Research
2.4. Comparative Analysis of Bibliometric Maps
2.5. Classification of Basic Refractories
2.5.1. Magnesia-Based Basic Refractories
2.5.2. Calcium Oxide-Based Refractories
2.5.3. Magnesia–Calcium Oxide Refractories
2.5.4. Magnesia–Carbon Refractories
2.5.5. Sintered Dolomite Refractories (Doloma)
2.6. Fundamental Physicochemical Properties
| Property | Description and Impact in Steelmaking | Ref. |
|---|---|---|
| Refractoriness | High melting point and thermal stability make dolomite suitable for lining furnaces and converters. | [71,72] |
| Bulk Density | Affects mechanical strength and resistance to slag penetration; influenced by calcination process. | [65,73] |
| Chemical Reactivity | Reactivity with slag and steel is determined by mineralogy, crystal size, and calcination degree. | [64,74] |
| Slag Resistance | Good resistance to basic slags (high CaO, MgO); calcined dolomite dissolves efficiently in slag. | [64,75] |
| Hydration Resistance | Calcined dolomite (Doloma) is prone to hydration; hydration resistance is lower than MgO bricks. | [72,76] |
2.7. Raw Materials: Characteristics, Availability, and Geopolitics
3. Processing and Synthesis Technologies
3.1. Conventional Processes
3.2. Innovative Processing Strategies
4. Modification with Nanostructured Additives
4.1. Assessment of Additives
4.2. Impact of Nanostructured Additives on Performance Parameters
- Hydration Resistance
- Mechanical Performance
- Thermal Behavior
Spinel Phase Engineering for Thermal Shock Resistance
4.3. Industrial Applications and In-Service Performance
4.3.1. Steelmaking
Refractories for Basic Oxygen Furnace (BOF) Units
Refractories for Electric Arc Furnace (EAF) Units
Refractories for Argon Oxygen Decarburization (AOD) Converters
Comparison with Magnesia–Carbon and Chromium-Based Refractories
4.3.2. Cement and Lime Industry
Thermal Spalling in Rotary Kilns and Preheaters
- Material selection: Refractories with moderate-to-high thermal expansion and controlled porosity, such as magnesia–doloma bricks enhanced with spinel-forming additives (MgAl2O4, hercynite), offer better thermal shock resistance [169].
- Microstructural design: The presence of fine, uniformly distributed pores in the refractory structure helps absorb thermal stresses and reduces crack propagation during thermal cycling [196].
- Operational controls: Controlled heating and cooling rates, along with regular kiln shell scanning, help reduce the severity of thermal gradients during operation [244].
4.3.3. Other Industrial Applications
Glass Industry
Technical Ceramics
Non-Ferrous Metallurgy
5. Technical and Competitive Strengths
6. Current Limitations and Technological Challenges
6.1. Hydration Susceptibility Without Additives
6.2. Cost and Availability of Nanoparticles
6.3. Industrial Scalability and Microstructural Uniformity
6.4. Recycling of Complex MgO–C and MgO–CaO–X Systems
7. Future Perspectives and Research Directions
8. Discussion
8.1. Research Evolution and Thematic Shifts in Magnesia–Dolomite Refractory Materials
8.2. Comparative Discussion with State-of-the-Art Reviews on Magnesia–Dolomite Refractories
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, S.; Xin, J.; Chen, R. Achieving high strength and low thermal conductivity: Additive manufacturing of mullite lightweight refractory. Ceram. Int. 2024, 50, 27880–27888. [Google Scholar] [CrossRef]
- Funch, C.V.; Proust, G. Laser-based additive manufacturing of refractory metals and their alloys: A review. Addit. Manuf. 2024, 94, 104464. [Google Scholar] [CrossRef]
- Ergashev, M. Exploring ceramic refractory materials: Classification and technological innovations. Int. J. Adv. Sci. Res. 2024, 4, 17–26. [Google Scholar] [CrossRef]
- Gilchrist, J.D. Classification of Refractories. In Fuels, Furnaces and Refractories; Elsevier: Amsterdam, The Netherlands, 1977; pp. 237–239. [Google Scholar] [CrossRef]
- Sengupta, P. Refractory: Characterization. In Refractories for the Chemical Industries; Springer: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Chandra, K.S.; Sarkar, D. Nanoscale reinforcement efficiency analysis in Al2O3–MgO–C refractory composites. Mater. Sci. Eng. A 2023, 865, 144613. [Google Scholar] [CrossRef]
- Sarkar, R. Refractory Technology: Fundamentals and Applications; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar] [CrossRef]
- Reitz, W. A Review of: “Refractories Handbook”. Mater. Manuf. Processes 2005, 20, 893–894. [Google Scholar] [CrossRef]
- Kusiorowski, R.; Wojsa, J.; Psiuk, B.; Wala, T. Influence of zirconia addition on the properties of magnesia refractories. Ceram. Int. 2016, 42, 11373–11386. [Google Scholar] [CrossRef]
- Hou, Q.; Luo, X.; Xie, Z.; Li, Y.Z.; An, D.; Li, J. Preparation and characterization of microporous magnesia-based refractory. Int. J. Appl. Ceram. Technol. 2020, 17, 2629–2637. [Google Scholar] [CrossRef]
- Fu, L.; Yue, J.; Liu, W.; Han, Z.; Bai, D.; Xu, G. Analysis and experiment of sintering and densification of magnesia particles. Chem. Eng. Sci. 2022, 268, 118396. [Google Scholar] [CrossRef]
- Sado, S.; Jastrzębska, I.; Zelik, W.; Szczerba, J. Self-organizing maps as a tool to assess possible substitution of fused by sintered MgO aggregates in MgO–C refractories. Ceram. Int. 2024, 50, 14996–15012. [Google Scholar] [CrossRef]
- Sadik, C.; Moudden, O.; Bouari, A.E.; Amrani, I.E. Review on the elaboration and characterization of ceramics refractories based on magnesite and dolomite. J. Asian Ceram. Soc. 2016, 4, 219–233. [Google Scholar] [CrossRef]
- Asabaev, D.K.h.; Badalov, F.A.; Normurodov, A.A. Dolomites, their formation conditions and features of territorial distribution in western uzbekistan. J. Geogr. Reg. Plan. Dev. 2024, 1, 1–3. [Google Scholar] [CrossRef]
- Dehsheikh, H.G.; Ghasemi-Kahrizsangi, S.; Karamian, E.; Shahmohammadian, F. Hydration resistance improvement of doloma particles using different nanoparticles. Ceram. Int. 2019, 45, 7390–7396. [Google Scholar] [CrossRef]
- Mohammadihooyeh, M.; Karamian, E.; Emadi, R. Effect of magnesium-aluminate spinel nano-particles on microstructure and properties behaviors of doloma-containing refractories. Ceram. Int. 2020, 46, 1662–1667. [Google Scholar] [CrossRef]
- Antonov, G.I.; Nedosvitii, V.P.; Kulik, A.S.; Semenenko, O. Stabilized Dolomite Refractories. Refract. Ind. Ceram. 2004, 45, 160–164. [Google Scholar] [CrossRef]
- Shahraki, A.; Ghasemi-Kahrizsangi, S.; Nemati, A. Performance improvement of MgO-CaO refractories by the addition of nano-sized Al2O3. Mater. Chem. Phys. 2017, 198, 354–359. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, J.; Li, Y.; Li, F.; Yan, W.; Shi, H. Effect of TiO2 addition on microstructures and properties of MgO–CaO refractory aggregates. J. Iron Steel Res. Int. 2024, 31, 1547–1554. [Google Scholar] [CrossRef]
- Kashaninia, F.; Sarpoolaky, H.; Naghizadeh, R.; Bagheri, A.; Zamanipour, M. Improving hydration resistance of magnesia-doloma refractories byiron oxide addition. Iran. J. Mater. Sci. Eng. 2011, 8, 34–40. [Google Scholar]
- Rabah, M.; Ewais, E. Multi-impregnating pitch-bonded Egyptian dolomite refractory brick for application in ladle furnaces. Ceram. Int. 2009, 35, 813–819. [Google Scholar] [CrossRef]
- Ghasemi-Kahrizsangi, S.; Karamian, E.; Dehsheikh, H.G.; Ghasemi-Kahrizsangi, A. A Review on Recent Advances on Magnesia-Doloma Refractories by Nano-Technology. J. Water Environ. Nanotechnol. 2017, 2, 206–222. [Google Scholar] [CrossRef]
- Obregón, Á.; Rodríguez-Galicia, J.L.; López-Cuevas, J.; Pena, P.; Baudín, C. MgO–CaZrO3-based refractories for cement kilns. J. Eur. Ceram. Soc. 2011, 31, 61–74. [Google Scholar] [CrossRef]
- Díaz-Tato, L.; López-Perales, J.F.; Contreras, J.M.; Banda-Muñoz, F.; Suárez-Suárez, D.; González-Carranza, Y.; Gómez-Rodríguez, C.; Rodríguez, E. Hydration resistance and mechano-physical properties improvement of a magnesia-dolomite dense refractory by hercynite spinel. Mater. Chem. Phys. 2022, 287, 126314. [Google Scholar] [CrossRef]
- Muñoz, I.; Soto, A.; Maza, D.; Bayón, F. Life cycle assessment of refractory waste management in a Spanish steel works. Waste Manag. 2020, 111, 1–9. [Google Scholar] [CrossRef]
- Klitzsch, M.; Geith, M. Setting New Standards for Circular Economy in the Cement Industry. In REWAS 2022: Developing Tomorrow’s Technical Cycles; Springer: Cham, Switzerland, 2022; Volume I. [Google Scholar] [CrossRef]
- Kara, S.; Erdem, S.; Lezcano, R. MgO-Based Cementitious Composites for Sustainable and Energy Efficient Building Design. Sustainability 2021, 13, 9188. [Google Scholar] [CrossRef]
- Sengupta, P. Manufacturing and Properties of Refractories. In Refractories for the Cement Industry; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Lee, W.E.; Vieira, W.; Zhang, S.; Ahari, K.G.; Sarpoolaky, H.; Parr, C. Castable refractory concretes. Int. Mater. Rev. 2001, 46, 145–167. [Google Scholar] [CrossRef]
- Burgos-Montes, O.; Álvarez, M.; Aza, A.H.; Pena, P.; Baudín, C. The main role of silica—Based cement free binders on the microstructural evolution and mechanical behaviour of high alumina castables. J. Eur. Ceram. Soc. 2018, 38, 4137–4148. [Google Scholar] [CrossRef]
- Tomsu, F.; Palco, S. Refractory Monolithics versus Shaped Refractory Products. Interceram—Int. Ceram. Rev. 2017, 66, 20–23. [Google Scholar] [CrossRef]
- Horckmans, L.; Nielsen, P.; Dierckx, P.; Ducastel, A. Recycling of refractory bricks used in basic steelmaking: A review. Resour. Conserv. Recycl. 2019, 140, 297–304. [Google Scholar] [CrossRef]
- Jassim, A.; Salmtori, S.A.; Jassam, J. Sustainable manufacturing process applied to produce magnesium oxide from sea water. IOP Conf. Ser. Mater. Sci. Eng. 2020, 757, 012021. [Google Scholar] [CrossRef]
- Wagri, N.K.; Carlborg, M.; Eriksson, M.; Ma, C.; Broström, M.; Andersson, B. High temperature interactions between coal ash and MgO-based refractories in lime kiln conditions. Fuel 2023, 342, 127711. [Google Scholar] [CrossRef]
- Chowdhury, A.; Panda, C.R. Preparation of high pure refractory grade magnesium oxide from east coast sea water. Indian J. Chem. Technol. 2023, 30, 614–622. [Google Scholar] [CrossRef]
- Hou, X.; Miao, Z.; Du, Y.; Chen, J.; Cao, Y.; Yan, W.; Xia, Y.; Wang, L.; Zhang, S.; Li, N. Fabrication Y2O3-doped MgO refractory raw materials based on magnesium hydroxide from salt-lake brine. Ceram. Inc. 2024, 50, 42729–42738. [Google Scholar] [CrossRef]
- Yang, M.; Yang, Y.; Xu, Y.; Zhao, J.; Zhao, W.; Sun, Z.; Yang, D.; Ren, L.; Zhao, X.; Yan, H.; et al. Oxidation and corrosion resistance of resin-bonded magnesia-based refractories reinforced by CaB6 addition for secondary refining. Ceram. Inc. 2024, 50, 40700–40712. [Google Scholar] [CrossRef]
- Park, S.H.; Jeon, S.M.; Jang, J. Characteristic Behavior of Hydration of Magnesium Oxide. In Geotechnics for Sustainable Infrastructure Development; Springer: Singapore, 2019; pp. 1275–1279. [Google Scholar] [CrossRef]
- Park, S.H.; Ma, J.; Yun, T.; Jeon, S.M.; Byeun, Y.K.; Kang, D.; Jang, J. Pore-scale swelling mechanism of magnesium oxide granules during hydration. Constr. Build. Mater. 2020, 251, 119101. [Google Scholar] [CrossRef]
- Fedoročková, A.; Raschman, P.; Sučik, G.; Švandová, M.; Doráková, A. Reactive, Sparingly Soluble Calcined Magnesia, Tailor-Made as the Reactive Material for Heavy Metal Removal from Contaminated Groundwater Using Permeable Reactive Barrier. Minerals 2021, 11, 1153. [Google Scholar] [CrossRef]
- Guo, Z.; Ma, Y.; Rigaud, M. Sinterability of macrocrystalline and cryptocrystalline magnesite to refractory magnesia. Int. J. Ceram. Eng. Sci. 2020, 2, 303–309. [Google Scholar] [CrossRef]
- Huang, W.; Yu, S.; Wu, X.; Zhang, Q. Effect of firing temperature on the densification and properties of dead burned magnesia. J. Eur. Ceram. Soc. 2018, 38, 4140–4146. [Google Scholar] [CrossRef]
- Bouchekrit, C.; Kolli, M.; Altıner, M.; Doufnoune, R. Synthesis of high purity magnesia MgO from Algerian dolomite ore. J. Min. Metall. Sect. B Metall. 2023, 59, 53–64. [Google Scholar] [CrossRef]
- Zhao, X.; You, J.; Yue, J.; Luo, X.; Ma, B. Microstructure and Mechanistic analysis of high-calcium fused magnesia molten heap. J. Aust. Ceram. Soc. 2024, 60, 1529–1539. [Google Scholar] [CrossRef]
- Bilge, A.; Yaman, C.; Sarıoğlu, N. Turkey’s Magnesite for Production of Fused Magnesia, Properties and Uses in Refractory Applications. In Proceedings of the Processing Technology, 60th Inter Colloquium on Refractories EUROGRESS, Aachen, Germany, 18–19 October 2017. [Google Scholar]
- Chen, C.; Ma, S.; Sun, M.; Wang, Y.; Cao, H.; Zhang, M.; Li, Y.; Jiang, Z. Influence of refractories on cleanliness in high-purity C96V saw wire steel. Can. Metall. Q. 2024, 64, 1633–1648. [Google Scholar] [CrossRef]
- Cui, K.; Fu, T.; Zhang, Y.; Wang, J.; Mao, H.; Tan, T. Microstructure and mechanical properties of CaAl12O19 reinforced Al2O3-Cr2O3 composites. J. Eur. Ceram. Soc. 2021, 41, 7935–7945. [Google Scholar] [CrossRef]
- He, J.; Wei, Y.; Wang, X.; Zhang, J.; You, D.; Wang, Y. Properties of CaO-MgO-SiO2 insulating refractories prepared from phosphorus tailings. Ceram. Inc. 2024, 50, 50867–50875. [Google Scholar] [CrossRef]
- Zhang, T.; Wei, Y.; Chen, J.; Li, N.; Han, B. Preparation of CaO-MgO-ZrO2 refractory and its desulfurization effect on Ni-based alloy in vacuum induction melting (VIM). J. Aust. Ceram. Soc. 2019, 56, 885–894. [Google Scholar] [CrossRef]
- Liu, N.; Gu, H.; Fu, L.; Huang, A.; Zhang, M. Improving corrosion resistance to CaO-Al2O3-SiO2 slag: Role of a novel dense calcium hexaluminate raw material. Constr. Build. Mater. 2025, 464, 140206. [Google Scholar] [CrossRef]
- Lin, C.; Sheng, N.; Fan, S.; Sun, S.; Hou, G.; Li, J.; Zhou, Y.; Sun, X. Interfacial Desulfurization Reaction between Binary Ni-Al/Ti/Ta Alloy Melt and CaO Ceramic Refractory. Surf. Interfaces 2023, 44, 103718. [Google Scholar] [CrossRef]
- Cheng, Y.; Duan, S.; Zhang, L. Comparison Study of the Effect of MgO, MgO-CaO, MgO-Al2O3-C, and MgO-C Refractories on Cleanliness of a SiMn-Killed Steel. Steel Res. Int. 2025, 96, 535–555. [Google Scholar] [CrossRef]
- Xu, T.; Su, Y.; Shi, T.; Zhang, X. Improving hydration resistance of MgO–CaO ceramics by in situ synthesized CaZrO3 coatings prepared using a non-hydrolytic sol. Ceram. Inc. 2021, 47, 2165–2171. [Google Scholar] [CrossRef]
- Zan, W.; Ma, B.; Liu, K.; Yu, C.; Liu, H.; Wang, Z.; Deng, C.; Zhu, Q. Effects of YSZ and CA6 additives on densification and thermal shock resistance of Al2O3-MgO-CaO-Y2O3 refractories. Mater. Sci. Eng. A 2024, 901, 146564. [Google Scholar] [CrossRef]
- Ren, X.; Ma, B.; Li, S.; Li, H.; Liu, G.; Yang, W.; Qian, F.; Zhao, S.; Yu, J. Comparison study of slag corrosion resistance of MgO–MgAl2O4, MgO–CaO and MgO–C refractories under electromagnetic field. J. Iron Steel Res. Int. 2020, 28, 38–45. [Google Scholar] [CrossRef]
- Kundu, R.; Sarkar, R. MgO-C Refractories: A Detailed Review of These Irreplaceable Refractories in Steelmaking. Interceram—Int. Ceram. Rev. 2021, 70, 46–55. [Google Scholar] [CrossRef]
- Bag, M.; Adak, S.; Sarkar, R. Study on low carbon containing MgO-C refractory: Use of nano carbon. Ceram. Inc. 2012, 38, 2339–2346. [Google Scholar] [CrossRef]
- Bavand-vandchali, M.; Naghizadeh, R. Characterization and post-mortem analysis of Al2O3-MgO-C refractories used in steelmaking ladle furnaces. Eng. Fail. Anal. 2020, 116, 104697. [Google Scholar] [CrossRef]
- Behera, S.; Sarkar, R. Formation of in-situ Ceramic Phase in N220 Nano Carbon Containing Low Carbon Mgo-C Refractory. World Acad. Sci. Eng. Technol. Int. J. Mater. Metall. Eng. 2015, 2. Available online: https://dspace.nitrkl.ac.in/dspace/handle/2080/2367 (accessed on 10 November 2025).
- Filkoski, V.R.; Petrovski, J.; Gjurchinovski, Z. Energy optimisation of vertical shaft kiln operation in the process of dolomite calcination. Therm. Sci. 2018, 22, 2123–2135. [Google Scholar] [CrossRef]
- Stein, V.; Aneziris, C.; Gueguen, E.; Hill, K. A Prospective Way to Reduce Emissions in Secondary Steel Making Metallurgy by Application of Functionalized Doloma Carbon Refractories. Int. J. Appl. Ceram. Technol. 2011, 9, 615–624. [Google Scholar] [CrossRef]
- Shen, Q.; Yu, Q.; Zhang, J.; Yao, X.; Yu, W. Numerical simulation of the dolomite in-situ desulfurization in molten iron. Mater. Res. Express 2023, 10, 016512. [Google Scholar] [CrossRef]
- Moorkah, H.I.; Abolarin, M.S. Investigation of the Properties of Locally Available Dolomite for Refractory Applications. Niger. J. Technol. 2005, 24, 79–86. [Google Scholar]
- Cheremisina, E.; Lesiak, S.; Rieger, J.; Schenk, J.; Firsbach, F.; Johnson, W.; Chopin, T.; Nispel, M. Assessment of the dissolution rate and behaviour of raw dolomite and limestone with different calcination degrees in primary steelmaking slags. Ironmak. Steelmak. 2022, 50, 379–391. [Google Scholar] [CrossRef]
- Dushevina, A. Study of the strength of caustic dolomite-based materials. Mech. Technol. 2024, 229–238. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Anbalagan, G. Thermal decomposition of natural dolomite. Bull. Mater. Sci. 2007, 30, 339–344. [Google Scholar] [CrossRef]
- Mcintosh, R.M.; Sharp, J.; Wilburn, F. The thermal decomposition of dolomite. Thermochim. Acta 1990, 165, 281–296. [Google Scholar] [CrossRef]
- Subagjo; Wulandari, W.; Adinata, P.M.; Fajrin, A. Thermal decomposition of dolomite under CO2-air atmosphere. AIP Conf. Proc. 2017, 1805, 040006. [Google Scholar] [CrossRef]
- Sandu, V.C.; Selejan, A.D.; Cormos, C.; Pop, A.; Cormos, A. High-temperature dolomite decomposition: An integrated experimental and computational fluid dynamics analysis for calcium looping and industrial applications. Appl. Therm. Eng. 2024, 253, 123742. [Google Scholar] [CrossRef]
- Valverde, J.; Perejón, A.; Medina-Carrasco, S.; Pérez-Maqueda, L. Thermal decomposition of dolomite under CO2: Insights from TGA and in situ XRD analysis. Phys. Chem. Chem. Phys. 2015, 17, 30162–30176. [Google Scholar] [CrossRef] [PubMed]
- Sivrikaya, O. A study on the physicochemical and thermal characterisation of dolomite and limestone samples for use in ironmaking and steelmaking. Ironmak. Steelmak. 2018, 45, 764–772. [Google Scholar] [CrossRef]
- Resio, L.C. Dolomite thermal behaviour: A short review. Phys. Chem. Miner. 2024, 51, 19. [Google Scholar] [CrossRef]
- Lan, Y.; Liu, Q.; Wu, G.; Yang, J.; Xu, M.; Ao, W.; Chen, Q. Recycling of Burned Dolomite Powder in Steelmaking. Metallurgist 2014, 57, 862–868. [Google Scholar] [CrossRef]
- Lesiak, S.; Cheremisina, E.; Rieger, J.; Schenk, J.; Firsbach, F.; Johnson, W.; Chopin, T.; Nispel, M. Calcination Condition of Dolomite-Based Materials Influencing Static Dissolution in Synthetic Electric Arc Furnace Slag. Steel Res. Int. 2022, 93, 2100675. [Google Scholar] [CrossRef]
- Feliciano, C.; Hernández, J.; Maldonado, J.; Cortes, K.; Schemmel, T.; Jansen, H. Use of MgO Briquettes in Electric Arc Furnaces: Principles and Industrial Experiences at Comsinac-Casimas. In Proceedings of the 6th Steel Industry Conference and Exposition (CONAC 2014), Monterrey, Mexico, 24 March 2014. [Google Scholar]
- Yu, Y.; Hwang, D.; Ahn, Y.; Cho, K.; Ahn, J.W.; Choi, J. A Comparative Study on the Calcination and Hydration of Dolomite Using Microwave and Electric Furnaces. J. Korean Soc. Miner. Energy Resour. Eng. 2021, 58, 107–118. [Google Scholar] [CrossRef]
- Sahu, N.; Biswas, A.; Kapure, G. Development of Refractory Material from Water Quenched Granulated Ferrochromium Slag. Miner. Process. Extr. Metall. Rev. 2016, 37, 255–263. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, H.; Tang, Y.; Xin, Y.; Yuan, H.; Ren, X.; Wan, Q. Synthesis mechanism and properties of lightweight mullite-corundum refractories obtained through high temperature liquid-assisted micrometer-scale Kirkendall effect. Ceram. Inc. 2020, 47, 9234–9244. [Google Scholar] [CrossRef]
- Guler, S.H.; Yakin, A.; Guler, O.; Chattopadhyay, A.K.; Şimşek, T. A critical review of the refractory high-entropy materials: RHEA Alloys, Composites, Ceramics, Additively Manufactured Alloys. Curr. Appl. Phys. 2024, 70, 87–124. [Google Scholar] [CrossRef]
- Miracle, D.; Tsai, M.; Senkov, O.; Soni, V.; Banerjee, R. Refractory high entropy superalloys (RSAs). Scr. Mater. 2020, 187, 445–452. [Google Scholar] [CrossRef]
- Tang, H.; Peng, Z.; Gu, F.; Yang, L.; Tian, W.; Zhong, Q.; Rao, M.; Li, G.; Jiang, T. Chromium-promoted preparation of forsterite refractory materials from ferronickel slag by microwave sintering. Ceram. Inc. 2021, 47, 10809–10818. [Google Scholar] [CrossRef]
- Nanda, S.; Choudhury, A.; Chandra, K.; Sarkar, D. Raw materials, Microstructure, and Properties of MgO–C refractories: Directions for Refractory Recipe Development. J. Eur. Ceram. Soc. 2022, 43, 14–36. [Google Scholar] [CrossRef]
- Chandra, K.S.; Sarkar, D. Refractories and Failures. In Ceramic Processing; CRC Press: Boca Raton, FL, USA, 2019; pp. 167–213. [Google Scholar] [CrossRef]
- Kullatham, S.; Sirisoam, T.; Lawanwadeekul, S.; Thiansem, S. Forsterite refractory brick produced by talc and magnesite from Thailand. Ceram. Inc. 2022, 48, 30272–30281. [Google Scholar] [CrossRef]
- Kizinievič, O.; Gencel, O.; Kizinievič, V.; Sutcu, M.; Skamat, J. Recycling of dolomite powder in clay bricks: Effects on characteristics and gas release. Constr. Build. Mater. 2023, 404, 133217. [Google Scholar] [CrossRef]
- Yeprem, H.A.; Hübner, H. Effect of Fe2O3 Additions on Sinterability of Konya Dolomite of Turkey. Key Eng. Mater. 2004, 264–268, 1819–1822. [Google Scholar] [CrossRef]
- Suvorov, S.A.; Nazmiev, M.I.; Baranov, A.P.; Dmitrienko, A.A. A High-Density Water-Resistant Magnesia-Lime Material Based on Dolomite. Refract. Ind. Ceram. 2005, 46, 217–219. [Google Scholar] [CrossRef]
- Wu, H.; Chen, Z.; Yan, W.; Schafföner, S.; Guiyuan, W.; Dai, Y.; Li, Y. A novel lightweight periclase-composite (Mg8−xFex+yAl16−yO32) spinel refractory material for cement rotary kilns. Ceram. Inc. 2021, 48, 615–623. [Google Scholar] [CrossRef]
- Yrjas, P.; Iisa, K.; Hupa, M. Limestone and dolomite as sulfur absorbents under pressurized gasification conditions. Fuel 1996, 75, 89–95. [Google Scholar] [CrossRef]
- Ay, Ş.; Atakül, H.; Sarioglan, A.; Akgün, F.; Isik-gulsac, I.; Çetin, Y.; Üresin, E.; Er, O.O.; Aksoy, P. Hot Gas Clean-Up with Dolomites: Effect of Gas Composition on Sulfur Removal Activity. Can. J. Chem. Eng. 2015, 93, 1643–1650. [Google Scholar] [CrossRef]
- Hycnar, E.; Ratajczak, T.; Sęk, M. Dolomites as SO2 Sorbents in Fluid Combustion Technology. Resources 2020, 9, 121. [Google Scholar] [CrossRef]
- Zhang, W.; Huang, A.; Zou, Y.; Gu, H.; Fu, L.; Li, G. Corrosion modeling of magnesia aggregates in contact with CaO–MgO–SiO2 slags. J. Am. Ceram. Soc. 2020, 103, 2128–2136. [Google Scholar] [CrossRef]
- Hou, Y.; Zhang, S.; Dang, J.; Guo, J.; Zhou, H.; Lü, X. Viscosity and structure relationship with equimolar substitution of CaO with MgO in the CaO–MgO–Al2O3–SiO2 slag melts. Int. J. Miner. Metall. Mater. 2025, 32, 70–79. [Google Scholar] [CrossRef]
- Kong, W.; Liu, J.; Yu, Y.; Hou, X.; He, Z. Effect of w(MgO)/w(Al2O3) ratio and basicity on microstructure and metallurgical properties of blast furnace slag. J. Iron Steel Res. Int. 2021, 28, 1223–1232. [Google Scholar] [CrossRef]
- Li, P.; Ning, X. Effects of MgO/Al2O3 Ratio and Basicity on the Viscosities of CaO-MgO-SiO2-Al2O3 Slags: Experiments and Modeling. Metall. Mater. Trans. B 2016, 47, 446–457. [Google Scholar] [CrossRef]
- Pang, Z.; Lv, X.; Jiang, Y.; Ling, J.; Yan, Z. Blast furnace ironmaking process with super-high TiO2 in the slag: Viscosity and melting properties of the slag. Metall. Mater. Trans. B 2020, 29, 1170–1178. [Google Scholar] [CrossRef]
- Zhan, X.; Wu, X.; Xing, Y.; Cui, X.; Wang, S.; Zhao, F.; Meng, W.; Ma, C.; Zhong, X. Improved hydration resistance of MgO–2CaO·SiO2–3CaO·SiO2 composite refractory using low-grade minerals. Mater. Res. Express 2020, 7, 085502. [Google Scholar] [CrossRef]
- Manzano, H.; Pellenq, R.; Ulm, F.; Buehler, M.; van Duin, A.V. Hydration of calcium oxide surface predicted by reactive force field molecular dynamics. Langmuir ACS J. Surf. Colloids 2012, 28, 4187–4197. [Google Scholar] [CrossRef]
- Fujimori, Y.; Zhao, X.; Shao, X.; Levchenko, S.; Nilius, N.; Sterrer, M.; Freund, H. Interaction of Water with the CaO(001) Surface. J. Phys. Chem. C 2016, 120, 5565–5576. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Cai, J.; Zhou, F.; Cheng, Z.; Zhang, J. Experimental Study on the Mechanical Properties and Microstructural Mechanisms of Coal Gangue-Based Cementitious Materials Synergistically Activated by Desulfurization Gypsum and Lime. Polymers 2025, 17, 932. [Google Scholar] [CrossRef]
- Barman, D.; Dash, S. Stabilization of expansive soils using chemical additives: A review. J. Rock Mech. Geotech. Eng. 2022, 14, 1319–1342. [Google Scholar] [CrossRef]
- Shahraki, A.; Keshavarz, M.; Malek Khachatourian, A.; Nemati, A.; Rezaei, B. Investigation of hydration, corrosion, and thermal shock resistance of MgO–CaO Ceramic Composites reinforced with electrospun carbon nanofibers. Mater. Res. Bull. 2025, 194, 113752. [Google Scholar] [CrossRef]
- Criado, Y.A.; Alonso, M.; Abanades, J.C. Enhancement of a CaO/Ca(OH)2 based material for thermochemical energy storage. Sol. Energy 2016, 135, 800–809. [Google Scholar] [CrossRef]
- Chen, Z.; Zhao, J.; Wu, C.; Chen, B.; Dai, M. Mechanism underlying effects of CaO-MgO combined expansive agent on hydration kinetics of cement-based systems. J. Sustain. Cem.-Based Mater. 2024, 13, 978–994. [Google Scholar] [CrossRef]
- Sun, Z.; Chi, H.; Fan, L. Physical and Chemical Mechanism for Increased Surface Area and Pore Volume of CaO in Water Hydration. Ind. Eng. Chem. Res. 2012, 51, 10793–10799. [Google Scholar] [CrossRef]
- Blamey, J.; Zhao, M.; Manović, V.; Anthony, E.; Dugwell, D.; Fennell, P. A shrinking core model for steam hydration of CaO-based sorbents cycled for CO2 capture. Chem. Eng. J. 2016, 291, 298–305. [Google Scholar] [CrossRef]
- Lee, J.K.; Choi, H.; Lee, S. Effect of Fe2O3 additions on the hydration resistance of CaO. J. Ceram. Process. Res. 2012, 13, 646–650. [Google Scholar] [CrossRef]
- Zhao, S.; Wang, W.; Xu, J. Review of Research Development of Hydration Resistance of Calcium Oxide Refractories. Mater. Rev. 2005, 39, 364–367. [Google Scholar]
- Yi, N.; Ma, Y.; Wang, Z.; Liu, H.; Wang, X.; Dong, Y.; Xia, Z.; Zhu, Y.; Deng, C. Microstructural regulation and properties enhancement of MgO-CaO ceramics by doping Y2O3. J. Rare Earths 2022, 41, 1771–1779. [Google Scholar] [CrossRef]
- Guo, R.; Funayama, S.; Kim, S.T.; Harada, T.; Takasu, H.; Kato, Y. Hydration reactivity enhancement of calcium oxide–based media for thermochemical energy storage. Energy Storage 2021, 3, e232. [Google Scholar] [CrossRef]
- Kang, S.H.; Kwon, M.; Kwon, Y.H.; Moon, J. Effects of polycarboxylate ether (PCE)-based superplasticizer on the dissolution and subsequent hydration of calcium oxide (CaO). Cem. Concr. Res. 2021, 146, 106467. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, J.; He, F. CO2 carbonation-induced improvement in strength and microstructure of reactive MgO-CaO-fly ash-solidified soils. Constr. Build. Mater. 2019, 229, 116914. [Google Scholar] [CrossRef]
- Chamhaydari, M.A.R.; Ahmadimoghadam, H.; Nilforoushan, M. Enhancing hydration resistance and mechanical properties of dolomite refractory through ZnO nanoparticle incorporation. Int. J. Appl. Ceram. Technol. 2024, 22, e14985. [Google Scholar] [CrossRef]
- Hadian, M.; Nazari, B. Influence of magnesia addition on hydration of iranian dolomite. Iran. J. Mater. Sci. Eng. 2010, 7, 51–56. [Google Scholar]
- Fan, Y. Production and Application Status of Magnesite-dolomite Refractories in China. Shandong Metall. 2006, 28, 28–30. [Google Scholar]
- Liu, J.; Chen, M.; Wang, N.; Sui, X. Effect of phase evolution and microstructure on thermal shock resistance and hydration resistance of low-carbon MgO-C refractories: Al-TiB2 hybrid addition. Constr. Build. Mater. 2025, 463, 140141. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, S.; Lee, W.E. Improving the hydration resistance of lime-based refractory materials. Int. Mater. Rev. 2008, 53, 1–20. [Google Scholar] [CrossRef]
- Liu, W.; Chen, X.; Liu, W.; Zhang, N.; Mao, Y.; Guo, Y. Impact and mechanism of bisphosphonate depressant 1-hydroxypropane-1,1-diphosphonic acid on flotation decalcification of dolomite-rich magnesite ore. Int. J. Min. Sci. Technol. 2024, 34, 1017–1032. [Google Scholar] [CrossRef]
- Yin, W.; Haoran, S.; Hong, J.; Hong, J.; Cao, S.; Bin, Y.; Won, C.; Song, M. Effect of Ca selective chelator BAPTA as depressant on flotation separation of magnesite from dolomite. Miner. Eng. 2019, 144, 106050. [Google Scholar] [CrossRef]
- Afify, A.M.; Sanz-Montero, M.; González-Acebrón, L. Dolomite–magnesite formation and polymetallic mineralization in a rift-sag basin on the western margin of the Red Sea: Paleoenvironmental, hydrothermal, and tectonic implications. J. Sediment. Res. 2022, 92, 144–165. [Google Scholar] [CrossRef]
- Yang, B.; Wang, D.; Cao, S.; Yin, W.; Xue, J.; Zhu, Z.; Fu, Y.; Yao, J. Selective adsorption of a high-performance depressant onto dolomite causing effective flotation separation of magnesite from dolomite. J. Colloid Interface Sci. 2020, 578, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Mateus, A.; Martins, L. Building a mineral-based value chain in Europe: The balance between social acceptance and secure supply. Miner. Econ. 2020, 34, 239–261. [Google Scholar] [CrossRef]
- Seaman, J. Critical Raw Materials, Economic Statecraft and Europe’s Dependence on China. Int. Spect. 2024, 60, 20–37. [Google Scholar] [CrossRef]
- Berthet, E.; Lavalley, J.; Anquetil-Deck, C.; Ballesteros, F.; Stadler, K.; Soytaş, U.; Hauschild, M.; Laurent, A. Assessing the social and environmental impacts of critical mineral supply chains for the energy transition in Europe. Glob. Environ. Change 2024, 86, 102841. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Lian, L.; Jia, X.; Qian, J. Recycling of waste magnesia refractory brick powder in preparing magnesium phosphate cement mortar: Hydration activity, mechanical properties and long-term performance. Constr. Build. Mater. 2023, 402, 133019. [Google Scholar] [CrossRef]
- Baumgart, C.; Weigelt, C.; Krüger, L.; Aneziris, C.G. Investigations on the sintering response of steel-ceramic composites. IOP Conf. Ser. Mater. Sci. Eng. 2016, 139, 012013. [Google Scholar] [CrossRef]
- Vlad, E.; Buzduga, R.; Buzduga, M.; Caloian, V.; Plopeanu, E.; Pandelescu, C.; Dobrescu, C.; Constantin, N. Experimental research on the effect of additives on the sintering process of alumina-based refractory materials. J. Phys. Conf. Ser. 2021, 1781, 012066. [Google Scholar] [CrossRef]
- Yu, T.; Zhao, Z.; Li, J. Sintering Strengthening Mechanism of MgO Sintering Additives Added in Fabricating Al2O3 Ceramics Using Binder Jetting. Adv. Eng. Mater. 2025, 27, 2402799. [Google Scholar] [CrossRef]
- Braulio, M.; Morbioli, G.G.; Medeiros, J.; Gallo, J.; Pandolfelli, V. Nano-Bonded Wide Temperature Range Designed Refractory Castables. J. Am. Ceram. Soc. 2012, 95, 1100–1104. [Google Scholar] [CrossRef]
- Tan, B.; Chen, K.; Huang, Z.; Fang, M.; Liu, Y.; Wu, X.W. Influence of the Binder Amount on the Properties of the Non-Sintering Ti(C,N)-Si3N4-SiC Composite Refractories. Adv. Mater. Res. 2013, 833, 221–224. [Google Scholar] [CrossRef]
- Apal’kova, G.D. The effect of nanodispersed iron oxide additives on the formation of the density of carbon refractories. Novye Ogneupor. 2018, 11, 50–52. [Google Scholar] [CrossRef]
- Gilmer, D.; Kim, S.; Goldsby, D.; Nandwana, P.; Elliott, A.; Saito, T. Predictive Binder Jet Additive Manufacturing enabled by Clean Burn-off Binder Design. Addit. Manuf. 2024, 80, 103955. [Google Scholar] [CrossRef]
- Bhandari, S.; Manière, C.; Sedona, F.; Bona, E.D.; Sglavo, V.; Colombo, P.; Fambri, L.; Biesuz, M.; Franchin, G. Ultra-rapid debinding and sintering of additively manufactured ceramics by ultrafast high-temperature sintering. J. Eur. Ceram. Soc. 2023, 44, 328–340. [Google Scholar] [CrossRef]
- Umar, A.M.; Wuritka, E.G.; Tokan, A.; Sadiq, Y.O. Production of dense zircon sand-alumina refractory brick from zircon sand of bakin ruwa for foundry application. Int. J. Appl. Adv. Eng. Res. 2025, 8, 44–54. [Google Scholar] [CrossRef]
- Reis, G.S.; Cazacliu, B.; Cothenet, A.; Poullain, P.; Wilhelm, M.; Sampaio, C.; Lima, E.; Ambrós, W.; Torrenti, J. Fabrication, microstructure, and properties of fired clay bricks using construction and demolition waste sludge as the main additive. J. Clean. Prod. 2020, 258, 120733. [Google Scholar] [CrossRef]
- Thuong, T.V.; Tashlykov, O.L.; Mahmoud, K. A unique Vietnam’s red clay-based brick reinforced with metallic wastes for γ-ray shielding purposes: Fabrication, characterization, and γ-ray attenuation properties. Nucl. Eng. Technol. 2024, 56, 1544–1551. [Google Scholar] [CrossRef]
- Odewale, I.O.; Aluma, C.C.; Idu, F.U.; Amaakaven, V.T.; Ogunkunle, D.; Olagunju, S. Consequence of Variations in Al2O3.2SiO2.2H2O and Grog Percentages on the Properties of Dense Refractory Bricks. Saudi J. Eng. Technol. 2021, 6, 451–462. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, Y.; Zhang, Z.; Mao, H.; Han, H.; Yang, J. Recycling reuse of municipal sewage sludge in sustainable structural materials: Preparation, properties, crystallization and microstructure analyses. Constr. Build. Mater. 2023, 398, 132507. [Google Scholar] [CrossRef]
- Yang, J.; Lu, S.; Wang, L. Fused magnesia manufacturing process: A survey. J. Intell. Manuf. 2020, 31, 327–350. [Google Scholar] [CrossRef]
- Niesyt, M.; Psiuk, B. Fused dolomite-magnesia co-clinker for fired dolomite refractories. Ceram. Inc. 2017, 43, 51–59. [Google Scholar] [CrossRef]
- Korczak, K.; Kochański, M.; Skoczkowski, T. Mitigation options for decarbonization of the non-metallic minerals industry and their impacts on costs, energy consumption and GHG emissions in the EU—Systematic literature review. J. Clean. Prod. 2022, 358, 132006. [Google Scholar] [CrossRef]
- Suzuki, Y.; Morgan, P.; Ohji, T. Pressureless-Sintering of CaZrO3/MgO In Situ Composites Derived from Natural Dolomite with Various Additives. In Design Manufacturing Composites; CRC Press: Boca Raton, FL, USA, 2021. [Google Scholar] [CrossRef]
- Qian, X.; Qin, Y.; Tao, Y.; Shen, P.; Hu, C.; Wang, F.; Hu, S. Development of highly reactive partially calcined dolomite precursor: Synergistic effect CaO and MgO. J. Am. Ceram. Soc. 2025, 108, e20530. [Google Scholar] [CrossRef]
- Kim, S.J.; Rocha, L.T.D.; Kim, S.; Jung, S. Effect of mill-scale and calcined dolomite on high Al2O3 sinter and its reduction behaviour. In Proceedings of the 12th International Conference of Molten Slags, Fluxes and Salts (MOLTEN 2024), Brisbane, Australia, 17–19 June 2024. [Google Scholar] [CrossRef]
- Liu, W.; Mao, Y.; Zheng, J.; Wang, Z.; Shang, C.; Liu, W.; Zhao, Q.; Zhao, S.; Shen, Y. Enhancing the flotation separation of magnesite and dolomite by introducing a phosphonic acid depressant during grinding. Sep. Purif. Technol. 2025, 361, 131412. [Google Scholar] [CrossRef]
- Tang, Y.; Xu, C.; Chen, Q.; Li, Q.; He, D.; Li, Z.; Fu, Y. Effect of a novel environmental-friendly chelating depressant DTPMP on the flotation separation of magnesite and dolomite. Appl. Surf. Sci. 2025, 690, 162650. [Google Scholar] [CrossRef]
- Ban, X.; Yao, J.; Yin, W.; Xie, Y.; Zhang, T.; Du, W.; Wang, Y. Selective adsorption of eco-friendly inhibitor sesbania gum on dolomite for efficient flotation separation of magnesite and dolomite. Process Saf. Environ. Prot. 2024, 194, 630–640. [Google Scholar] [CrossRef]
- Luo, N.; Shi, J.; Yan, B.; Wang, X. Flotation Separation of Magnesite from Dolomite Using Sodium Silicate Modified with Zinc Sulfate as a Selective Depressant. Minerals 2024, 14, 355. [Google Scholar] [CrossRef]
- Gong, X.; Yao, J.; Guo, J.; Yang, B.; Sun, H.; Yin, W.; Wang, Y.; Fu, Y. Role of tannin pretreatment in flotation separation of magnesite and dolomite. Int. J. Miner. Metall. Mater. 2024, 31, 452–461. [Google Scholar] [CrossRef]
- Wang, L.; Li, Z.; Zhang, H.; Huang, L.; Zhu, Y.; Li, F. Flotation separation of magnesite from dolomite with gellan gum as depressant and its depression mechanism. Miner. Eng. 2024, 212, 108718. [Google Scholar] [CrossRef]
- Wang, B.; Liu, C.; Fan, W.; Mao, Y.; Liu, W. An Environmentally Friendly Chelator for Improving the Flotation Separation of Magnesite and Dolomite: Flotation Behavior and Adsorption Mechanism. Minerals 2025, 15, 289. [Google Scholar] [CrossRef]
- Zhong, W.; Yin, W.; Wang, Y.; Yao, J. Selective flotation of magnesite from dolomite using α-chloro-oleate acid as collector. Powder Technol. 2020, 373, 147–151. [Google Scholar] [CrossRef]
- Sun, W.; Liu, W.; Liu, W.; Li, P.; Shen, Y.; Dai, S. Utilization of a novel bisphosphonic acid surfactant for reverse froth flotation of magnesite and dolomite. Miner. Eng. 2022, 185, 107668. [Google Scholar] [CrossRef]
- Cheng, G.; Ma, Y.; López-Valdivieso, A.; Yin, W. Selective depression of phenoxyacetyl chloride on magnesite: Implications for effective flotation separation of magnesite from dolomite. Miner. Eng. 2024, 218, 109017. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, P.; Hu, K.; Cao, Y.; Chai, W. Insights Into Flotation Separation of Dolomite From Magnesite with SDS Collector: Experiments and DFT Calculation. Asia-Pac. J. Chem. Eng. 2025, 20, e70028. [Google Scholar] [CrossRef]
- Yin, W.; Wang, Y.; Ma, Y.; Chen, K. Effects of ultrasonic treatment on the flotation behavior of magnesite and dolomite in a sodium oleate system. Green Smart Min. Eng. 2024, 1, 76–84. [Google Scholar] [CrossRef]
- Gong, X.; Yao, J.; Zhao, X.; Qi, Z.; Yang, B.; Yin, W.; Wang, Y. Effect of ultrasonic treatment on the surface roughness and floatability of magnesite and dolomite. J. Mol. Liq. 2024, 404, 125002. [Google Scholar] [CrossRef]
- Qi, G.; Shan, F.; Li, Q.; Yu, J. Energy Saving by Applying 3000kVA Electric Arc Furnace in Fused Magnesia Production. Mater. Sci. Forum 2013, 749, 299–302. [Google Scholar] [CrossRef]
- An, P.; Han, Z.; Wang, K.; Zhao, Z.; Situmorang, Y.A.; Rizkiana, J.; Abudula, A.; Guan, G. Energy-saving strategy for a transport bed flash calcination process applied to magnesite. Carbon Resour. Convers. 2021, 4, 122–131. [Google Scholar] [CrossRef]
- Zhao, L.; Feng, J.; Dong, H. Analysis of carbon footprint and reduction approach of magnesia production in China. J. Clean. Prod. 2021, 334, 130194. [Google Scholar] [CrossRef]
- An, J.; Li, Y.; Middleton, R. Reducing energy consumption and carbon emissions of magnesia refractory products: A life-cycle perspective. J. Clean. Prod. 2018, 182, 363–371. [Google Scholar] [CrossRef]
- Jiang, T.; Zhang, W.; Liu, S.K. Performance Evaluation of a Full-Scale Fused Magnesia Furnace for MgO Production Based on Energy and Exergy Analysis. Energies 2021, 15, 214. [Google Scholar] [CrossRef]
- Spyridakos, A.; Alexakis, D.E.; Vryzidis, I.; Tsotsolas, N.; Varelidis, G.; Kagiaras, E. Waste Classification of Spent Refractory Materials to Achieve Sustainable Development Goals Exploiting Multiple Criteria Decision Aiding Approach. Appl. Sci. 2022, 12, 3016. [Google Scholar] [CrossRef]
- Seifert, S.; Dittrich, S.; Bach, J. Recovery of Raw Materials from Ceramic Waste Materials for the Refractory Industry. Processes 2021, 9, 228. [Google Scholar] [CrossRef]
- Zou, Y.; Gu, H.; Huang, A.; Fu, L. Formation Mechanism of In Situ Intergranular CaZrO2 Phases in Sintered Magnesia Refractories. Metall. Mater. Trans. A 2020, 51, 5328–5338. [Google Scholar] [CrossRef]
- Jin, E.; Zou, C.; Ding, D.; Xiao, G.; Duan, F.; Jiang, B.; Han, S.; Zheng, K. Enhanced mechanical properties and thermal shock resistance of magnesia refractories via in situ formation of tetragonal zirconia. Ceram. Inc. 2024, 50, 14968–14979. [Google Scholar] [CrossRef]
- Otroj, S. Synthesis of Hercynite under Air Atmosphere using MgAl2O4 Spinel. Mater. Sci. 2015, 21, 288–292. [Google Scholar] [CrossRef][Green Version]
- Jastrzębska, I.; Stępień, J.; Żukrowski, J. Stabilization of hercynite structure at elevated temperatures by Mg substitution. Mater. Des. 2023, 235, 112449. [Google Scholar] [CrossRef]
- Zan, W.; Ma, B.; Cao, Y.; Tian, J.; Zhou, Z.; Wang, L.; Jiang, Z. Preparation and performance optimization of MgAl2O4 spinel materials by single-step reaction sintering. Ceram. Inc. 2023, 49, 23567–23578. [Google Scholar] [CrossRef]
- Xinming, R.; Beiyue, M.; Zhang, G.; Gaofeng, F.; Yu, J.; Liu, G. Preparation and properties of MgAl2O4 spinel ceramics by double-doped Sm2O3–(Y2O3, Nb2O5 and La2O3). Mater. Chem. Phys. 2020, 252, 123309. [Google Scholar] [CrossRef]
- Liang, Y.C.; Liu, W.; Wu, H.; Liu, Q.; Yao, L. Promoting effect of Si on MnOx catalysts for low-temperature NH2-SCR of NO: Enhanced N2 selectivity and SO2 resistance. Fuel 2023, 355, 129478. [Google Scholar] [CrossRef]
- Lei, Q.; Wang, S.; Wu, Q.; Cao, R.; Cai, Z.; Liu, C.; Ma, Y.; Song, G.; Yang, W.; Wen, C. In-situ synthesis of Mn2SiO4 and MnxSi dual phases through solid-state reaction to improve the initial Coulombic efficiency of SiO anode for Lithium-Ion batteries. J. Electroanal. Chem. 2024, 977, 118845. [Google Scholar] [CrossRef]
- Wang, T.; Chen, Z.; Chen, D.; Zhao, R. Hybrid MnO-SiOx@C Microspheres with a Hierarchical Mesoporous Structure for Advanced Lithium-Ion Battery Anodes. J. Alloys Compd. 2021, 899, 163251. [Google Scholar] [CrossRef]
- Woo, D.H.; Lee, H. Phase Equilibria of the MnO–SiO2–Al2O3–MnS System. J. Am. Ceram. Soc. 2010, 93, 2098–2106. [Google Scholar] [CrossRef]
- Wang, Z.; Sohn, I. Immobilizing chromium in stainless steel slags with MnO addition. J. Am. Ceram. Soc. 2021, 104, 697–705. [Google Scholar] [CrossRef]
- Wang, Z.; Sohn, I. Selective elemental concentration during the solidification of stainless steel slags for increased Cr recovery with MnO addition. J. Am. Ceram. Soc. 2020, 103, 6012–6024. [Google Scholar] [CrossRef]
- Kim, T.S.; Park, J. Viscosity-structure relationship of alkaline earth silicate melts containing manganese oxide and calcium fluoride. J. Am. Ceram. Soc. 2019, 102, 4943–4955. [Google Scholar] [CrossRef]
- Yuan, H.; Wang, Z.; Zhang, Y.; Wang, C. Roles of MnO and MgO on structural and thermophysical properties of SiO2-MnO-MgO-B2O3 welding Fluxes: A molecular dynamics study. J. Mol. Liq. 2023, 386, 122501. [Google Scholar] [CrossRef]
- Ma, H.B.; Yan, J.; Zhao, Y.H.; Liu, T.; Ren, Q.; Liao, Y.; Zuo, J.; Liu, G.; Yao, M. Oxidation behavior of Cr-coated zirconium alloy cladding in high-temperature steam above 1200 °C. npj Mater. Degrad. 2021, 5, 7. [Google Scholar] [CrossRef]
- Han, X.; Chen, C.; Tan, Y.; Feng, W.; Peng, S.; Zhang, H. A systematic study of the oxidation behavior of Cr coatings on Zry4 substrates in high temperature steam environment. Corros. Sci. 2020, 174, 108826. [Google Scholar] [CrossRef]
- Yang, R.; Lei, H.; Zhang, J. Preparation of SiO2@MnO2 composite abrasives and their performance in chemical-mechanical polishing of SiC substrates. Ceram. Inc. 2024, 50, 34796–34805. [Google Scholar] [CrossRef]
- Kaya, M.; Köksal, F.; Munir, M.; Kazmi, S.; Gencel, O.; Ozbakkaloglu, T. Effect of Natural and Artificial Silicon Additives on the Physicomechanical Performance of Dolomite-based Alkaline-Activated Mortar. Silicon 2023, 16, 215–230. [Google Scholar] [CrossRef]
- Zeng, X.; Yu, D.; Liu, F.; Fan, B.; Wen, C.; Yu, X.; Xu, M. Scavenging of refractory elements (Ca, Mg, Fe) by kaolin during low rank coal combustion. Fuel 2018, 223, 198–210. [Google Scholar] [CrossRef]
- Vakalova, T.V.; Reshetova, A.A.; Revva, I.B.; Rusinov, P.G.; Balamygin, D.I. Effect of thermochemical activation of clay raw materials on phase formation, microstructure and properties of aluminosilicate proppants. Appl. Clay Sci. 2019, 183, 105335. [Google Scholar] [CrossRef]
- Gao, J.; Su, W.; Hou, J.; Song, X.; Bai, Y.; Wang, J.; Lv, P.; Yu, G. Inhibition effects of wetting and corrosion behavior of high-alkali coal using typical additives under gasification conditions. Fuel 2024, 380, 133158. [Google Scholar] [CrossRef]
- Kahrizsangi, S.G.; Nemati, A.; Shahraki, A.; Farooghi, M. The effect of nano-additives on the hydration resistance of materials synthesized from the MgO-CaO system (research note). Int. J. Eng. Trans. A Basics 2016, 29, 539–545. [Google Scholar] [CrossRef]
- Li, B.; Wei, Y.; Wang, J.; Chen, J.; Li, N. Improved hydration resistance of CaO granules via sol-processed metal oxide protective layers. J. Am. Ceram. Soc. 2021, 104, 4878–4890. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Bingrong, L.; Li, M.; Li, N. Core-shell structured CaO aggregate prepared by granulating with Al chelating compound and its hydration resistance. Ceram. Inc. 2020, 46, 10788–10796. [Google Scholar] [CrossRef]
- Yao, L.; Gao, Y.; Li, Y.; Huang, Y.; Wang, Y.; Peng, X.; Liu, Q. Microstructure, mechanical properties, and strengthening mechanisms of nanostructural Y-Zr-O oxide dispersion-strengthened (ODS) Mo alloys. J. Alloys Compd. 2022, 921, 166155. [Google Scholar] [CrossRef]
- Xu, L.; Liu, Y.; Cai, X.; Ding, S.; Xin, S.; Sun, B.; Shen, T. Heterostructural nanolamellar oxide-dispersion-strengthened ferritic alloy with exceptional strength and ductility. Scr. Mater. 2024, 242, 115949. [Google Scholar] [CrossRef]
- Ovid’ko, I.; Valiev, R.; Zhu, Y. Review on superior strength and enhanced ductility of metallic nanomaterials. Prog. Mater. Sci. 2018, 94, 462–540. [Google Scholar] [CrossRef]
- Zhao, L.; Lee, T.; Zheng, S.; Zheng, W.; Ryu, S.; Zhang, D.; Guo, Q. Ultrastrong and Deformable Aluminum-Based Composite Nanolaminates with Transformable Binary Intergranular Films. Nano Lett. 2024, 24, 3843–3850. [Google Scholar] [CrossRef]
- Li, X.; Lu, L.; Li, J.; Zhang, X.; Gao, H. Mechanical properties and deformation mechanisms of gradient nanostructured metals and alloys. Nat. Rev. Mater. 2020, 5, 706–723. [Google Scholar] [CrossRef]
- Murashkin, M.; Sabirov, I.; Sauvage, X.; Valiev, R. Nanostructured Al and Cu alloys with superior strength and electrical conductivity. J. Mater. Sci. 2015, 51, 33–49. [Google Scholar] [CrossRef]
- Andrievski, R.; Glezer, A. Strength of nanostructures. Physics-Uspekhi 2009, 52, 315–334. [Google Scholar] [CrossRef]
- Zou, Y.; Gu, H.; Huang, A.; Huo, Y.; Fu, L.; Li, Y. Characterisation and properties of low-conductivity microporous magnesia based aggregates with in-situ intergranular spinel phases. Ceram. Inc. 2021, 47, 11063–11071. [Google Scholar] [CrossRef]
- Thakur, S.; Giri, A. Origin of Ultralow Thermal Conductivity in Metal Halide Perovskites. ACS Appl. Mater. Interfaces 2023, 15, 26755–26765. [Google Scholar] [CrossRef]
- Lou, Z.; Zhang, P.; Zhu, J.; Gong, L.; Xu, J.; Chen, Q.; Reece, M.; Yan, H.; Gao, F. A novel high-entropy perovskite ceramics Sr0.9La0.1(Zr0.25Sn0.25Ti0.25Hf0.25)O3 with low thermal conductivity and high Seebeck coefficient. J. Eur. Ceram. Soc. 2022, 42, 3480–3488. [Google Scholar] [CrossRef]
- Shi, Y.B.; Chen, Y.; Dong, H.; Wang, H.; Qian, P. Investigation of phase transition, mechanical behavior and lattice thermal conductivity of halogen perovskites using machine learning interatomic potentials. Phys. Chem. Chem. Phys. 2023, 25, 30644–30655. [Google Scholar] [CrossRef]
- Xuan, S.; Tian, Y.; Kong, X.; Hao, J.; Wang, X. Enhancement of thermal shock resistance of Al2O3–MgAl2O4 composites by controlling the content and distribution of spinel phase. Ceram. Inc. 2023, 49, 39908–39916. [Google Scholar] [CrossRef]
- Gruber, D.; Sistaninia, M.; Fasching, C.; Kolednik, O. Thermal shock resistance of magnesia spinel refractories—Investigation with the concept of configurational forces. J. Eur. Ceram. Soc. 2016, 36, 4301–4308. [Google Scholar] [CrossRef]
- Ma, S.H.; Shi, K.; Xia, Y.; Zhang, Y.; Han, X. Effect of modified MgO aggregates on mechanical properties of magnesium aluminate spinel refractories. Ironmak. Steelmak. 2020, 48, 292–298. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Z.; Yan, W.; Wang, J.; Ma, S.; Li, G. A comparative study on lightweight and dense periclase-magnesium aluminate spinel refractories from industrial preparation. J. Alloys Compd. 2023, 960, 170611. [Google Scholar] [CrossRef]
- Liu, Z.; Liang, X.; Luo, X.; Zhao, J.; Wu, F. Effect of in-situ generated MgAl2O4 spinel on thermal shock resistance of magnesia-zirconia refractories. Ceram. Inc. 2024, 50, 35936–35945. [Google Scholar] [CrossRef]
- Liu, Y.; Yan, W.; Yan, J.; Dai, Y.; Wang, X.; Wang, Q.; Li, N. Enhancement of strength and thermal shock resistance of MgO-MgAl2O4 ceramic filters with microporous MgO powder: Effect of α-Al2O3 micro-powder content. Int. J. Appl. Ceram. Technol. 2024, 22, e14921. [Google Scholar] [CrossRef]
- Gu, Q.; Zhao, F.; Liu, X.; Jia, Q. Preparation and thermal shock behavior of nanoscale MgAl2O4 spinel-toughened MgO-based refractory aggregates. Ceram. Inc. 2019, 45, 12093–12100. [Google Scholar] [CrossRef]
- Sokolov, A.; Deynega, G.I.; Kuzmina, N.; Kuzmina, I. Structural and mechanical properties of a composite material based on partially stabilized zirconium dioxide doped with magnesium aluminate spinel. Aviat. Mater. Technol. 2021, 66, 78–85. [Google Scholar] [CrossRef]
- Nguyen, M.; Sokolá, R. Formation and influence of magnesium-alumina spinel on the properties of refractory forsterite-spinel ceramics. Mater. Tehnol. 2020, 54, 135–141. [Google Scholar] [CrossRef]
- Nguyen, M.; Sokolář, R. Impact of Fly Ash as a Raw Material on the Properties of Refractory Forsterite–Spinel Ceramics. Minerals 2020, 10, 835. [Google Scholar] [CrossRef]
- Chen, Z.; Yan, W.; Li, G.; Hong, S.; Li, N. Enhanced mechanical properties of novel Al2O3-based ceramic filter by using microporous corundum-spinel and nano-Al2O3 powders. J. Eur. Ceram. Soc. 2023, 44, 1070–1080. [Google Scholar] [CrossRef]
- Ceylantekin, R.; Aksel, C. Improvements on the mechanical properties and thermal shock behaviours of MgO–spinel composite refractories by ZrO2 incorporation. Ceram. Inc. 2012, 38, 995–1002. [Google Scholar] [CrossRef]
- Moritz, K.; Aneziris, C.; Hesky, D.; Gerlach, N. Magnesium Aluminate Spinel Ceramics Containing Aluminum Titanate for Refractory Applications. J. Ceram. Sci. Technol. 2014, 5, 125–130. [Google Scholar]
- Zhao, L.; Yao, S. Modification effect of Mg(OH)2–2Al(OH)3 composite gels on Y-PSZ ceramics. Ceram. Inc. 2024, 50, 30061–30067. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, Z.; Liu, H.; Ma, Y.; Wang, X.; Yi, N.; Xia, Z.; Zhu, Y.; Deng, C.; Zhang, L. Selective sintering of magnesia–calcia materials by utilizing hot spots during induction sintering process. J. Iron Steel Res. Int. 2024, 31, 1914–1922. [Google Scholar] [CrossRef]
- Chandra, S.; Ramesh, K.V. Refractories in indian steel industry: Past and present scenario. Glob. J. Eng. Appl. Sci. 2011, 1, 50–52. [Google Scholar]
- Salomão, R.; Bittencourt, L.; Pandolfelli, V. A novel approach for magnesia hydration assessment in refractory castables. Ceram. Inc. 2007, 33, 803–810. [Google Scholar] [CrossRef]
- Cui, Y.; Qu, D.; Luo, X.; Li, G.; Tian, L.; Zheng, Y. Enhanced thermal shock resistance in magnesia refractories through in situ formation of CaO·2La2O3·3SiO2. Int. J. Appl. Ceram. Technol. 2025, 22, e15075. [Google Scholar] [CrossRef]
- Shchekina, T.I.; Gramenitskii, E.N.; Batanova, A.M.; Kurbyko, T.A.; Likhodievskii, A.V.; Grigor’ev, B.N.; Pyrikov, A.N. Use of magnesian-dolomite mixtures in steel-melting furnace hearths and the mechanism of their wear in service. 1. Study of Ankerharth refractories. Refract. Ind. Ceram. 2006, 47, 317–325. [Google Scholar] [CrossRef]
- Badapalli, P.K.; Kottala, R.B.; Sree, P.; Rajasekhar, M. Occurrence and structures of dolomites in North Eastern part of Anantapur district, and their use in engineering materials. Mater. Today Proc. 2021, 50, 1005–1010. [Google Scholar] [CrossRef]
- Park, J.; Suk, M.; Jung, I.; Guo, M.; Blanpain, B. Interfacial Reaction between Refractory Materials and Metallurgical Slags containing Fluoride. Steel Res. Int. 2010, 81, 860–868. [Google Scholar] [CrossRef]
- Ewais, E.; Ahmed, A.; Kasem, A.A.; El-SKERIF, A.R. Attack under load of tempered tar/pitch-bonded Egyptian dolomite by BOF slag. J. Ceram. Soc. Jpn. 2002, 110, 931–936. [Google Scholar] [CrossRef]
- Preisker, T.; Gehre, P.; Schmidt, G.; Aneziris, C.; Wöhrmeyer, C.; Parr, C. Kinetics of the formation of protective slag layers on MgO–MgAl2O4–C ladle bricks determined in laboratory. Ceram. Inc. 2020, 46, 452–459. [Google Scholar] [CrossRef]
- Rahou, J.; Rezqi, H.; Ouahabi, M.E.; Fagel, N. Characterization of Moroccan steel slag waste: The potential green resource for ceramic production. Constr. Build. Mater. 2022, 314, 125663. [Google Scholar] [CrossRef]
- Chen, H.; Liu, Y.; Cui, H.; Zhang, W.; Hu, L.; Mao, L. Effects of electric arc furnace slag on promoting quality and environmental safety of fired bricks incorporating municipal solid waste incineration fly ash. Constr. Build. Mater. 2022, 345, 128327. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, Z.; Wang, Z.; Wang, N.; Wang, X. Splattering Suppression for a Three-Phase AC Electric Arc Furnace in Fused Magnesia Production Based on Acoustic Signal. IEEE Trans. Ind. Electron. 2017, 64, 4772–4780. [Google Scholar] [CrossRef]
- Stein, V.; Aneziris, C.; Gueguen, E. New Approach for the Application of Functional Ceramic Material in Carbon Bonded Doloma Refractories to Reduce Emissions. Adv. Eng. Mater. 2011, 13, 1135–1141. [Google Scholar] [CrossRef]
- Kudrina, A.; Krivchenko, Y.S.; Gul’ev, G.F. The influence of physicochemical factors of basic refractories on the life of linings of oxygen converters. Refractories 1963, 4, 434–439. [Google Scholar] [CrossRef]
- Gómez-Rodríguez, C.; Antonio-Zárate, Y.; Revuelta-Acosta, J.; Verdeja, L.; Fernández-González, D.; López-Perales, J.F.; Rodríguez-Castellanos, E.A.; García-Quiñonez, L.; Castillo-Rodríguez, G.A. Research and Development of Novel Refractory of MgO Doped with ZrO2 Nanoparticles for Copper Slag Resistance. Materials 2021, 14, 2277. [Google Scholar] [CrossRef]
- Boenzi, F. Possible ecological advantages from use of carbonless magnesia refractory bricks in secondary steelmaking: A framework LCA perspective. Int. J. Environ. Sci. Technol. 2021, 19, 5877–5896. [Google Scholar] [CrossRef]
- Borges, O.; Aneziris, C.G.; Pandolfelli, V. Assessing and tailoring the dilatometric profile of novel chromium-free refractory raw materials. J. Am. Ceram. Soc. 2024, 108, e20271. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, M.; Huang, Z.; Zhao, J.; Zhao, W.; Yang, D.; Ren, L.; Zhao, X.; Yan, H.; Liu, L.; et al. Improved properties of in-situ MgAl2O4-TiO2 dense layer reinforced low-carbon MgO-based refractories. Ceram. Inc. 2023, 49, 33842–33850. [Google Scholar] [CrossRef]
- Kusiorowski, R. MgO-ZrO2 refractory ceramics based on recycled magnesia-carbon bricks. Constr. Build. Mater. 2020, 231, 117084. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhu, T.; Li, Y.; Sang, S. Microstructure and properties of MgO–C refractory with different carbon contents. Ceram. Inc. 2021, 47, 2538–2546. [Google Scholar] [CrossRef]
- Yu, C.; Dong, B.; Chen, Y.; Ma, B.; Ding, J.; Deng, C.; Zhu, H.; Di, J. Enhanced oxidation resistance of low-carbon MgO–C refractories with ternary carbides: A review. J. Iron Steel Res. Int. 2022, 29, 1052–1062. [Google Scholar] [CrossRef]
- Capelo-Avilés, S.; de Oliveira, R.T.; Stampino, I.I.G.; Gispert-Guirado, F.; Casals-Terré, A.; Giancola, S.; Galán-Mascarós, J. A thorough assessment of mineral carbonation of steel slag and refractory waste. J. CO2 Util. 2024, 82, 102770. [Google Scholar] [CrossRef]
- Mandal, S.; Hemrick, J.; Mahapatra, M. Chrome-free qandilite (Mg2TiO4) refractory aggregates: Role of titania source and evaluation of thermal expansion coefficient. J. Eur. Ceram. Soc. 2022, 42, 7343–7351. [Google Scholar] [CrossRef]
- Boenzi, F.; Meré, J.B.O.; Iavagnilio, R. Life Cycle Assessment Comparison of Two Refractory Brick Product Systems for Ladle Lining in Secondary Steelmaking. Sustainability 2019, 11, 1295. [Google Scholar] [CrossRef]
- Stoianov, O.; Niziaev, K.; Malii, K.; Kukhar, V. Application of refractory materials for steel ladles. Report. Priazovskyi State Tech. Univ. Sect. Tech. Sci. 2023, 46, 69–78. [Google Scholar] [CrossRef]
- Li, Z.; Hua, Y.; Chang, Z.; Yue, Y.; Qin, J.; Qian, J. Hydration, carbonation and strength development of reactive MgO cement blended with lime (CaO) under different curing conditions. J. Build. Eng. 2023, 76, 107082. [Google Scholar] [CrossRef]
- Burmistrova, E.; Abdrakhmanov, R.I.; Igonin, A. Refractory Materials for OAO MMK Degassers and main Areas for Improving their Operating Reliability1. Refract. Ind. Ceram. 2013, 54, 269–271. [Google Scholar] [CrossRef]
- Samanta, A.; Satpathy, S.; Tripathi, A.; Sengupta, S.; Tsuyuguchi, K.; Panda, P. Designing of low cement castables in Al2O3–SiO2–SiC system, having alkali and thermal spalling resistance and suitable for discharge zone of cement rotary kiln. Refract. Ind. Ceram. 2015, 43, 141–144. [Google Scholar]
- Blond, E.; Schmitt, N.; Arnould, O.; Hild, F.; Poirier, J.; Blumenfeld, P. Prevalent Material Parameters Governing Spalling of a Slag-Impregnated Refractory. Key Eng. Mater. 2004, 264–268, 1751–1754. [Google Scholar] [CrossRef]
- Jin, S.; Harmuth, H.; Gruber, D.; Buhr, A.; Sinnema, S.; Rebouillat, L. Thermomechanical modelling of a torpedo car by considering working lining spalling. Ironmak. Steelmak. 2018, 47, 145–149. [Google Scholar] [CrossRef]
- Gomez, R.S.; Porto, T.N.; Magalhães, H.L.F.; Moreira, G.; André, A.M.; Melo, R.B.F.; Lima, A.G.B. Natural Gas Intermittent Kiln for the Ceramic Industry: A Transient Thermal Analysis. Energies 2019, 12, 1568. [Google Scholar] [CrossRef]
- Zou, Y.; Gu, H.; Huang, A.; Fu, L.; Li, G. Fabrication and properties of in situ intergranular CaZrO3 modified microporous magnesia aggregates. Ceram. Int. 2020, 46, 16956–16965. [Google Scholar] [CrossRef]
- Booth, F.; Stábile, F.M.; Bruni, Y.; Gauna, M.; Rendtorff, N. Dolomite-zirconia reaction sintered bonded coarse magnesia ceramics: Effect of the bonding proportion. Cerâmica 2021, 67, 151–157. [Google Scholar] [CrossRef]
- Wei, C.; Ma, C.; Li, Y.; Bai, W.; Yang, J.; Cheng, Y.; Gao, L. Controllable preparation and slag corrosion resistance of novel MgO–MgAl2O4–ZrO2 refractory. Ceram. Int. 2024, 50, 21406–21416. [Google Scholar] [CrossRef]
- Jastrzębska, I.; Ludwig, M.; Przystaś, J. Cr-free Refractories for Copper Metallurgy: Raw Materials and Factsage Thermodynamic Simulations. Ceram. Inc. 2024, 51, 3778–3791. [Google Scholar] [CrossRef]
- Ludwig, M.; Śnieżek, E.; Jastrzębska, I.; Prorok, R.; Sułkowski, M.; Goławski, C.; Fischer, C.; Wojteczko, K.; Szczerba, J. Recycled magnesia-carbon aggregate as the component of new type of MgO-C refractories. Constr. Build. Mater. 2021, 272, 121912. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Ikeda, K.; Ito, H. Manufacture of the Special Dolomite Refractories (Dolomitic, Magnesia-rich Refractories) by Utilizing Waste Lime from Acetylene Generator to Sea Water. J. Ceram. Assoc. Jpn. 1956, 64, 37–42. [Google Scholar] [CrossRef]
- Hao, C.; Yu, L.; Chen, X.; Chen, Y.; Li, C.; Qi, P.; Sun, J.; Zhou, H. Research on the development of recycling technology for magnesium refractory materials. Mater. Res. Express 2024, 11, 115504. [Google Scholar] [CrossRef]
- Gómez-Rodríguez, C.; Fernández-González, D.; García-Quiñonez, L.; Castillo-Rodríguez, G.A.; Aguilar-Martínez, J.; Verdeja, L. MgO Refractory Doped with ZrO2 Nanoparticles: Influence of Cold Isostatic and Uniaxial Pressing and Sintering Temperature in the Physical and Chemical Properties. Metals 2019, 9, 1297. [Google Scholar] [CrossRef]
- Zou, Y.; Gu, H.; Huang, A.; Fu, L.; Li, G.; Wang, L.; Chen, D. Pore evolution of microporous magnesia aggregates with the introduction of nano-sized MgO. Ceram. Inc. 2022, 48, 18513–18521. [Google Scholar] [CrossRef]
- Rodríguez, J.L.; Rodríguez, M.A.; Aza, S.; Pena, P. Reaction sintering of zircon–dolomite mixtures. J. Eur. Ceram. Soc. 2001, 21, 343–354. [Google Scholar] [CrossRef]
- Berezhnoy, A.; Kordyuk, R.A. The CaO-MgO-ZrO2-SiO2 system and its significance in the production of refractories. Refractories 1962, 3, 69–73. [Google Scholar] [CrossRef]
- Song, Q.; Zha, X.; Gao, M.; Shi, J.; Ma, Y. Influence of ZrO2 on the phase composition and mechano-physical properties of MgO–ZrO2 refractories prepared by cold isostatic pressing. Ceram. Inc. 2024, 50, 30474–30482. [Google Scholar] [CrossRef]
- Park, J. Formation of CaZrO3 at the interface between CaO–SiO2–MgO–CaF2(–ZrO2) slags and magnesia refractories: Computational and experimental study. Calphad 2007, 31, 149–154. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, L.; Chen, C.; Li, J.; Li, X. Effect of a MgO–CaO–ZrO2-based refractory on the cleanliness of a K4169 Ni-based superalloy. Ceram. Inc. 2022, 49, 117–125. [Google Scholar] [CrossRef]
- Chen, M.; Lu, C.; Yu, J. Improvement in performance of MgO–CaO refractories by addition of nano-sized ZrO2. J. Eur. Ceram. Soc. 2007, 27, 4633–4638. [Google Scholar] [CrossRef]
- Chen, M.; Lu, C.; Yu, J. Sintering and Performance of MgO-CaO Materials with Nano-Sized ZrO2 Addition. Mater. Sci. Forum 2007, 561–565, 623–626. [Google Scholar] [CrossRef]
- Ghasemi-Kahrizsangi, S.; Sedeh, M.B.; Dehsheikh, H.G.; Shahraki, A.; Farooghi, M. Densification and properties of ZrO2 nanoparticles added magnesia-doloma refractories. Ceram. Inc. 2016, 42, 15658–15663. [Google Scholar] [CrossRef]
- Zheng, W.; Xiao, X.; Chang, C.; Dong, J.; Wen, J.; Huang, Q.; Zhou, Y.; Li, Y. Characterizing properties of magnesium oxychloride cement concrete pavement. J. Cent. South Univ. 2019, 26, 3410–3419. [Google Scholar] [CrossRef]
- Haigh, R. A Decade Review of Research Trends Using Waste Materials in the Building and Construction Industry: A Pathway towards a Circular Economy. Waste 2023, 1, 935–959. [Google Scholar] [CrossRef]
- Özkan, A.; Günkaya, Z.; Tok, G.; Karacasulu, L.; Metesoy, M.; Banar, M.; Kara, A. Life Cycle Assessment and Life Cycle Cost Analysis of Magnesia Spinel Brick Production. Sustainability 2016, 8, 662. [Google Scholar] [CrossRef]
- Mena, J.; Sabolsky, E.; Sierros, K.A.; Sabolsky, K.; Jones, M.; Stepp, C.; Winch, N.; Raughley, M. Smart Refractory Sensors Development for Corrosion and Erosion Monitoring in High Temperature Systems. ECS Meet. Abstr. 2023, 1475. [Google Scholar] [CrossRef]
- Sabolsky, E.; Sabolsky, K.; Yakaboylu, G.A.; Buzzo, B. Fabrication and Testing of Smart Refractory for Energy System Monitoring in Harsh-Environments. In Proceedings of the 2018 XV International Scientific Conference on Optoelectronic and Electronic Sensors (COE), Warsaw, Poland, 17–20 June 2018; pp. 1–4. [Google Scholar] [CrossRef]
- Ferreira, P.M.; Machado, M.; Carvalho, M.; Vidal, C. Embedded Sensors for Structural Health Monitoring: Methodologies and Applications Review. Sensors 2022, 22, 8320. [Google Scholar] [CrossRef]
- Huang, Q.; Bhattacharyya, D.; Pillai, R.C.; Sabolsky, K.; Sabolsky, E. Gasifier Health Monitoring using Smart Refractory Bricks. Struct. Health Monit. 2017, 16, 14160. [Google Scholar] [CrossRef]
- Bayram, Y.; Ruege, A.C.; Hagan, P.; Sperry, E.; Cetnar, D.P. Advanced Furnace Inspection and Monitoring Based on Radar Sensors. In Proceedings of the 76th Conference on Glass Problems, Columbus, OH, USA, 7–10 November 2016; pp. 117–132. [Google Scholar] [CrossRef]
- Bayram, Y.; Wechsel, J.; Sperry, E. New Industry Standard in Furnace Inspection. In 78th Conference on Glass Problems; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018. [Google Scholar] [CrossRef]
- Zanelli, C.; Dondi, M.; Raimondo, M.; Guarini, G. Phase composition of alumina–mullite–zirconia refractory materials. J. Eur. Ceram. Soc. 2010, 30, 29–35. [Google Scholar] [CrossRef]
- Betsis, K.; Kourtis, A.; Karalis, K.; Xenidis, A. Assessment of Magnesia Refractories Corrosion by Iron-Rich Slags. Mater. Proc. 2021, 5, 135. [Google Scholar] [CrossRef]
- Oliveira, R.; Rodrigues, J.; Pereira, J. Numerical simulations on refractory linings for steel casting vessels. Fire Saf. J. 2023, 138, 103794. [Google Scholar] [CrossRef]
- Mahmoud, S.M.A.S.; Faraji, G.; Baghani, M.; Hashemi, M.S.; Sheidaei, A.; Baniassadi, M. Design of Refractory Alloys for Desired Thermal Conductivity via AI-Assisted In-Silico Microstructure Realization. Materials 2023, 16, 1088. [Google Scholar] [CrossRef]
- Alhaddad, E.M.S.; Ahmed, H.; Alhaddad, M.S.; Ahmed, H.A. A review of magnesite mineral and its industrial application. Arab J. Sci. Publ. 2022, 2663, 5798. [Google Scholar]



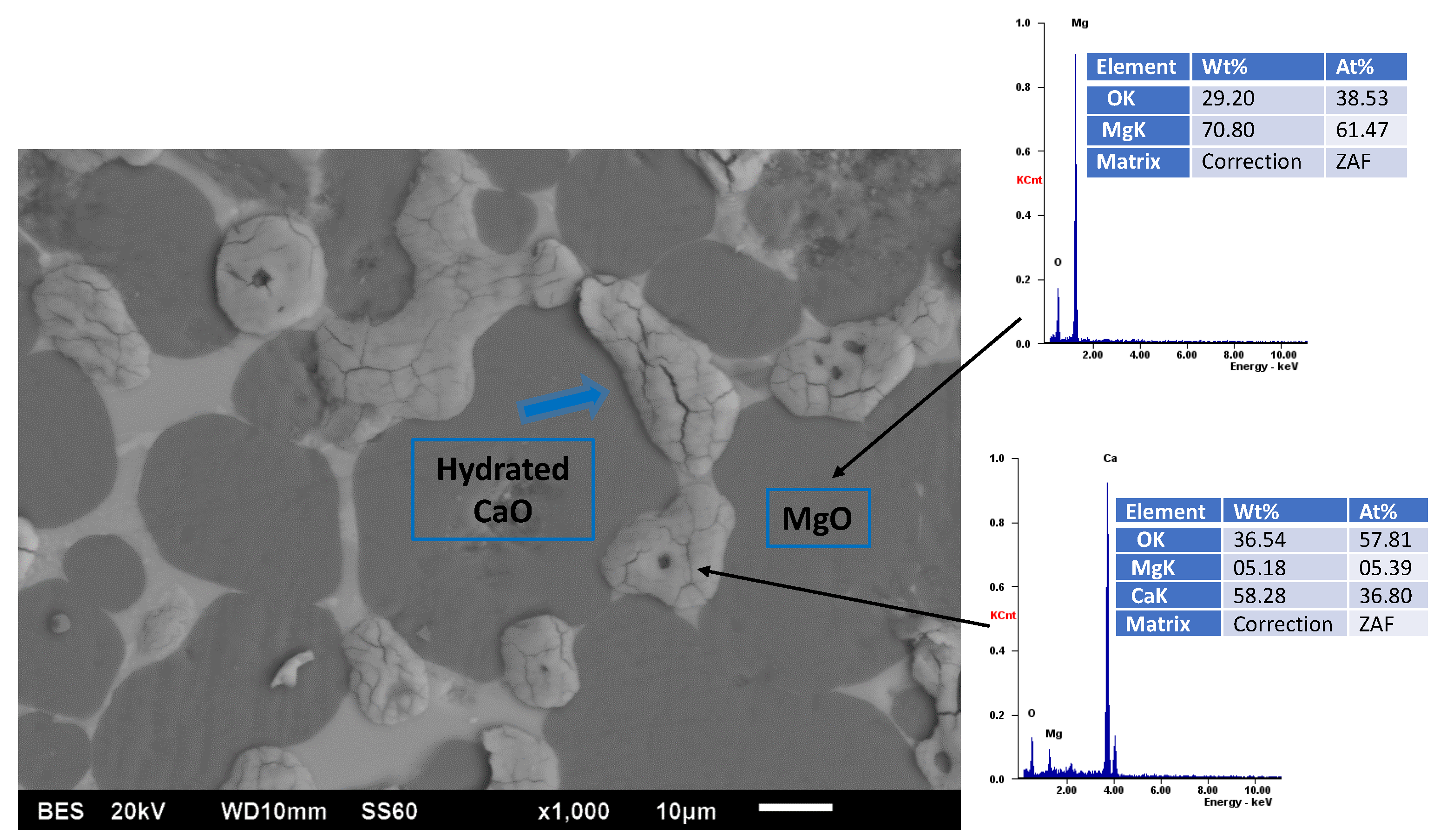
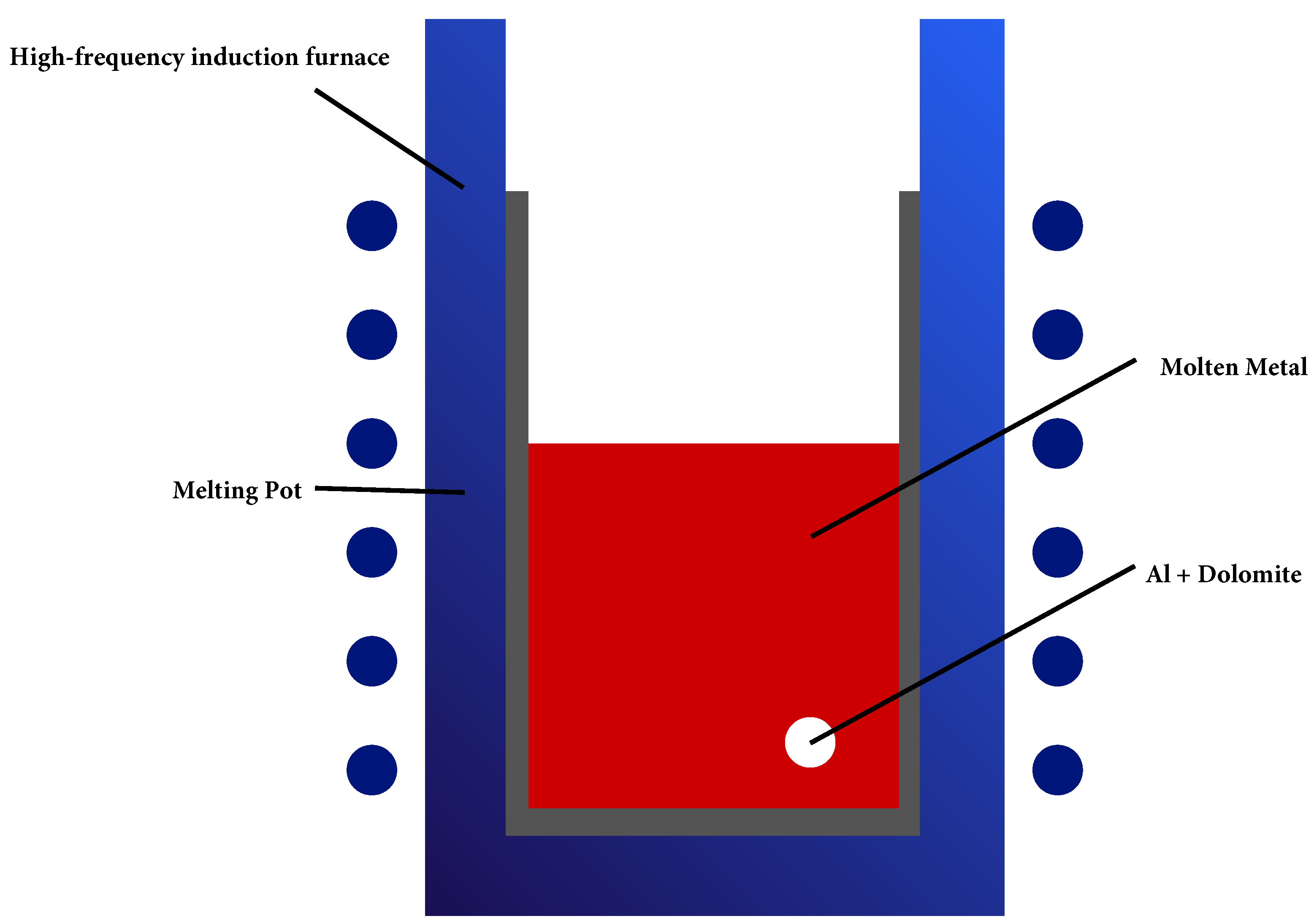


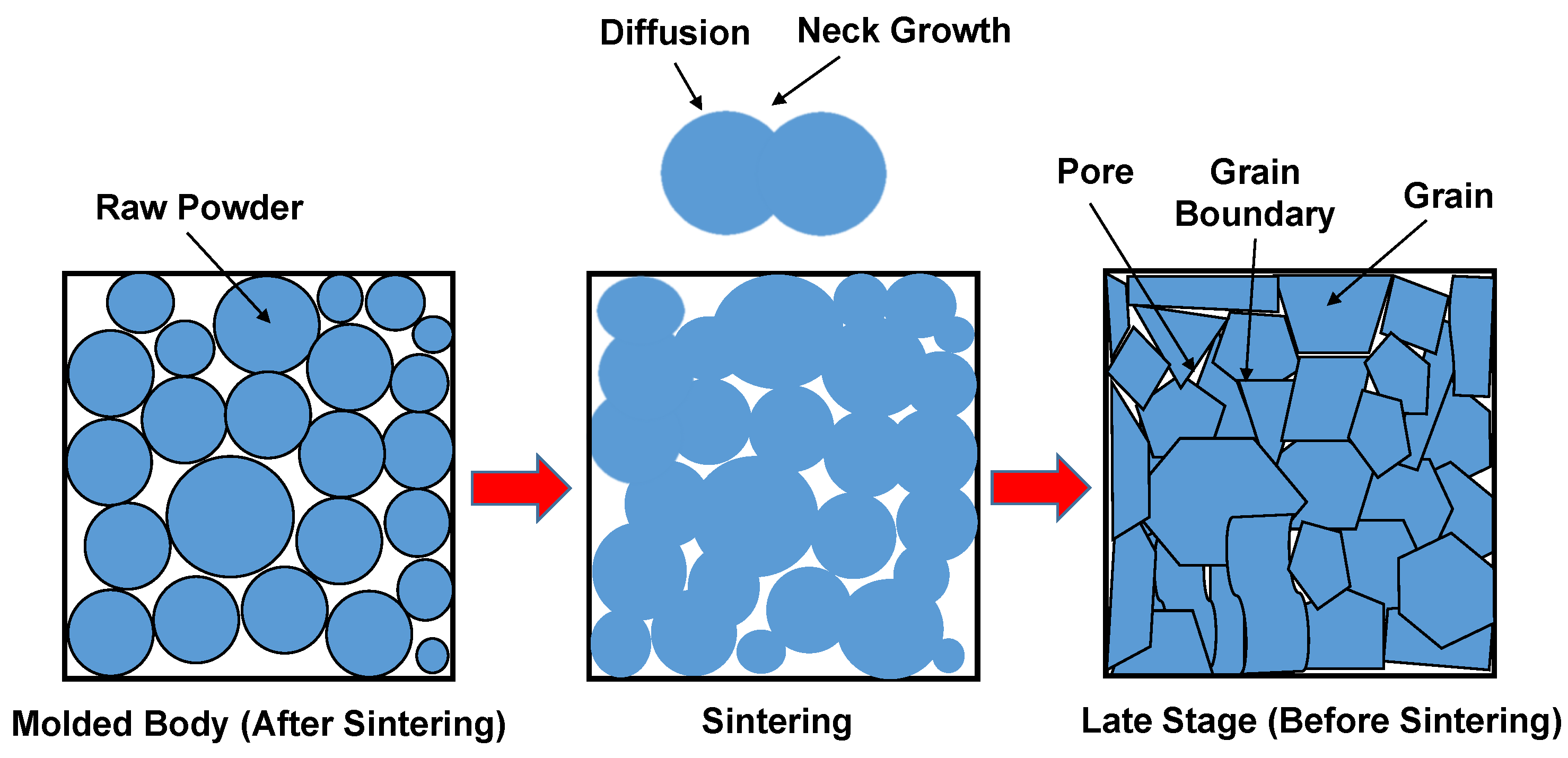









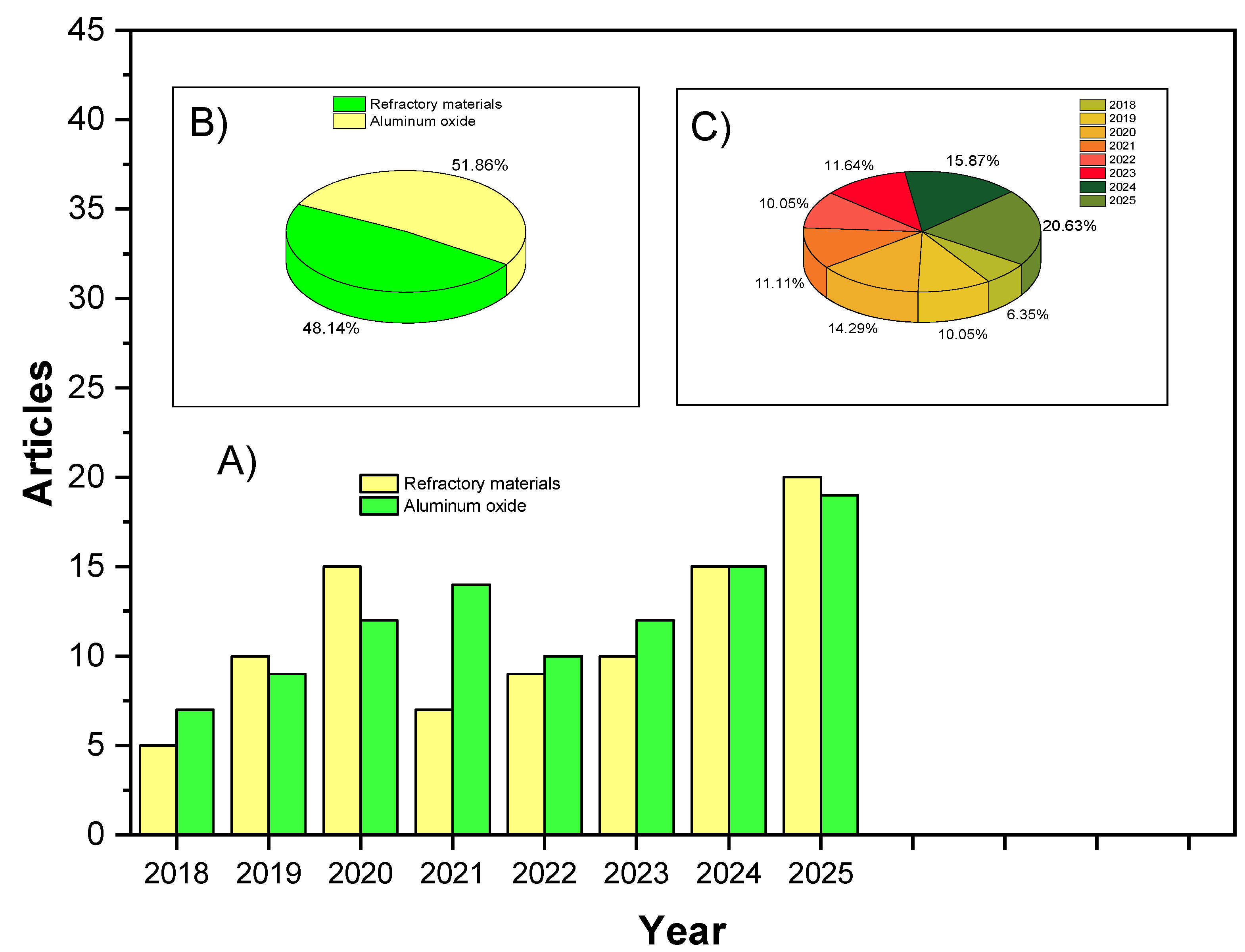

| Aspect | First Map (Figure 2) | Second Map (Figure 3) |
|---|---|---|
| Scope of keywords | Broader, multidisciplinary, including sustainability and advanced processing. | Narrow, technical focus on mechanical and chemical performance. |
| Representative terms | geopolymers, sustainable development, 3D printers, nano indentation, effluents. | fracture mechanics, slag corrosion, hydration resistance, oxidation resistance, fracture toughness. |
| Research orientation | Expands toward circular economy, recycling, and digital manufacturing. | Centers on durability, corrosion mechanisms, densification, and slag resistance. |
| Strengths | Captures interdisciplinary connections and emergent sustainable technologies. | Provides a clearer picture of core challenges for refractory performance in steelmaking and high-temperature service. |
| Limitations | Risk of including peripheral or loosely related domains. | May overlook innovative cross-disciplinary approaches. |
| Type | Production Method | Calcination Temperature | Main Properties and Applications |
|---|---|---|---|
| Caustic Calcined Magnesia (CCM) | From natural magnesite or brine, calcined at moderate temperatures | 800–1000 °C | Highly reactive; used as precursor in chemical processes or to produce other refractory compounds [40] |
| Dead Burned Magnesia (DBM) | High-temperature sintering of magnesite or seawater-derived Mg(OH)2 [41] | 1600–2100 °C | Low reactivity, high density; commonly used in refractory bricks [42] |
| Fused Magnesia (FM) | Electric arc fusion of magnesia | >3000 °C (fusion) | Highest purity (99.45%) [43] and grain sizes ≈ (938 μm) [44]; used in demanding applications such as EAFs and AOD converters [45] |
| Product Type | B. D. (g/cm3) | Notes | Ref. |
|---|---|---|---|
| Standard magnesia dolomite refractory | >3.00 | Apparent porosity < 8% | [86] |
| Dense magnesia lime (from dolomite) | 3.26–3.30 | Dead-burnt magnesia (DBM97) | [41] |
| High-density magnesia lime granules | 3.27–3.43 | 95–98% of theoretical; water-resistant | [87] |
| High-calcium fused magnesia (core region) | 3.57 | Very low porosity (1.05%) | [44] |
| Lightweight periclase composite spinel | 2.56 | Higher porosity; used in cement rotary kilns | [88] |
| Sintered dolomite refractory | >3.00 | Pure dolomite; high-temperature sintering | [86] |
| Region/Country | Resource Focus | Industrial Status/Challenges | Ref. |
|---|---|---|---|
| China | Magnesite, Dolomite | Leading producer, export controls, price setter | [118] |
| Uzbekistan | Dolomite | High-quality deposits, underdeveloped industry | [14] |
| Brazil, Turkey, US, EU | Magnesite, Dolomite | Significant reserves, less global influence | [119] |
| Factor | Effect/Description | Ref. |
|---|---|---|
| Particle size optimization and mixing | Fine and coarse particles improve packing density and final strength; sieve analysis ensures optimal blending | [134] |
| Use of grog or recycled materials | Grog increases porosity and reduces density; higher clay content improves compaction and mechanical strength | [135] |
| Compaction pressure (around 150 MPa) | Uniaxial or isostatic pressing minimizes open porosity and produces highly dense bricks | [136] |
| Hydraulic shaping and drying | Green body preparation before sintering via hydraulic pressing and oven drying | [137] |
| High-temperature sintering (1100–1400 °C) | Reduces porosity and enhances intergranular bonding; crucial for final microstructure | [138] |
| Final microstructure targets | Low open porosity (1–12%) and bulk density up to 2.26 g/cm3 achieved after optimized sintering | [137] |
| Ref. | Ceramic System | Spinel Approach | Main Results | Residual Strength After Shock |
|---|---|---|---|---|
| [169] | MgAl2O4 spinel ceramics | Co-doping with CeO2 and La2O3 | Higher density, lower porosity, improved thermal shock resistance | 89.15% |
| [196] | Microporous magnesia aggregates | In situ intergranular spinel formation | Improved thermal shock resistance, lower thermal conductivity | 65% after cycles |
| [204] | MgO–ZrO2–MgAl2O4 refractories | In situ spinel with micropores | >50% increase in residual flexural strength | Notable improvement |
| [205] | MgO–MgAl2O4 ceramic filters | Spinel neck formation via sintering | Enhanced mechanical and thermal shock resistance | 3.70 MPa post-shock |
| [213] | Y-PSZ modified ceramics | In situ spinel phase generation | Increased compressive and thermal shock resistance | 59.15% after 60 cycles |
| Ref. | Property | MgO-CaO | MgO–C | MgO–Cr2O3 |
|---|---|---|---|---|
| [233] | Thermal Shock Resistance | Moderate to High (enhanced with additives) | Excellent | High |
| [55] | Slag Resistance (Basic Slag) | Excellent | Excellent | Excellent |
| [234] | Oxidizing Atmosphere Stability | Good | Poor (Carbon Oxidation) | Good |
| [235] | Carbon Content/Emissions | Carbon-Free | High (Graphite) | Low |
| [236] | Chromium Toxicity (Cr6+ Risk) | No Risk | No Risk | High Risk (Cr6+) |
| [237] | Environmental Sustainability | High | Low | Low |
| [238] | Common Applications | BOF linings, AOD converters, EAF slag zones | EAF hot spots, ladles, slag lines | Secondary refining, BOF slag lines (declining use) |
| Works | Year | Thermal Behavior and Phase Evolution | High-Entropy or Advanced Refractory Systems | Comparative Analysis with MgO–C Refractories | Nanotechnology and Microstructural Design | Raw Materials and Magnesite Resources | Systematic PRISMA-Based Review |
|---|---|---|---|---|---|---|---|
| [72] | 2024 | ✓ | ✕ | ✕ | ✕ | ✕ | ✕ |
| [79] | 2024 | ✕ | ✓ | ✕ | ✕ | ✕ | ✕ |
| [56] | 2021 | ✕ | ✕ | ✓ | ✕ | ✕ | ✕ |
| [22] | 2017 | ✕ | ✕ | ✕ | ✓ | ✕ | ✕ |
| [275] | 2021 | ✕ | ✕ | ✕ | ✕ | ✓ | ✕ |
| This Work | 2025 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Tato, L.; Iturralde Carrera, L.A.; López-Perales, J.F.; Aviles, M.; Rodríguez-Castellanos, E.A.; Rodríguez-Resendiz, J. Advances in Magnesia–Dolomite Refractory Materials: Properties, Emerging Technologies, and Industrial Applications: A Review. Technologies 2025, 13, 523. https://doi.org/10.3390/technologies13110523
Díaz-Tato L, Iturralde Carrera LA, López-Perales JF, Aviles M, Rodríguez-Castellanos EA, Rodríguez-Resendiz J. Advances in Magnesia–Dolomite Refractory Materials: Properties, Emerging Technologies, and Industrial Applications: A Review. Technologies. 2025; 13(11):523. https://doi.org/10.3390/technologies13110523
Chicago/Turabian StyleDíaz-Tato, Leonel, Luis Angel Iturralde Carrera, Jesús Fernando López-Perales, Marcos Aviles, Edén Amaral Rodríguez-Castellanos, and Juvenal Rodríguez-Resendiz. 2025. "Advances in Magnesia–Dolomite Refractory Materials: Properties, Emerging Technologies, and Industrial Applications: A Review" Technologies 13, no. 11: 523. https://doi.org/10.3390/technologies13110523
APA StyleDíaz-Tato, L., Iturralde Carrera, L. A., López-Perales, J. F., Aviles, M., Rodríguez-Castellanos, E. A., & Rodríguez-Resendiz, J. (2025). Advances in Magnesia–Dolomite Refractory Materials: Properties, Emerging Technologies, and Industrial Applications: A Review. Technologies, 13(11), 523. https://doi.org/10.3390/technologies13110523











