Evaluation of Machine Learning Algorithms for Classification of EEG Signals
Abstract
1. Introduction
2. Materials and Methods
2.1. Hardware and Software
2.2. Machine Learning Algorithm Training
2.3. Input Data
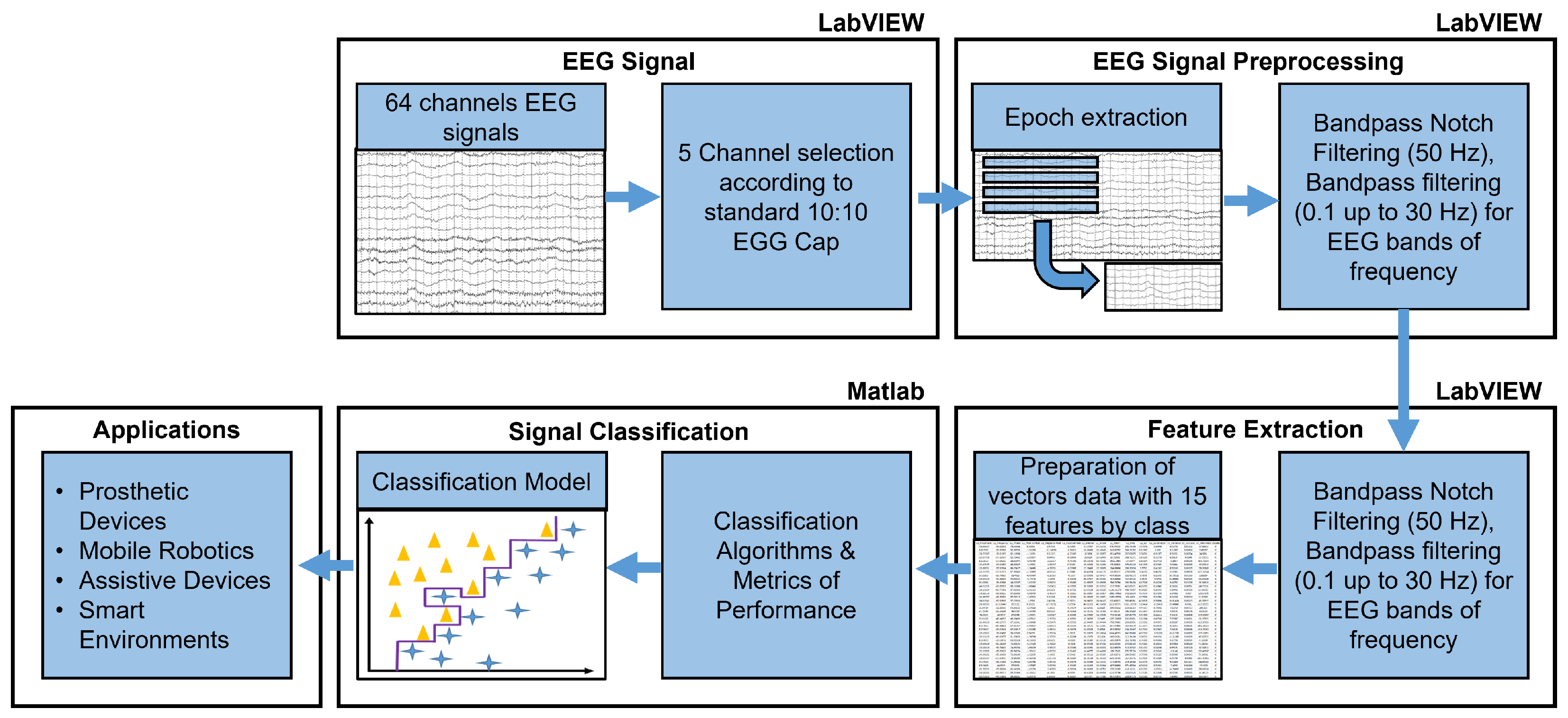
2.4. Proposed Method for EEG Signal Processing
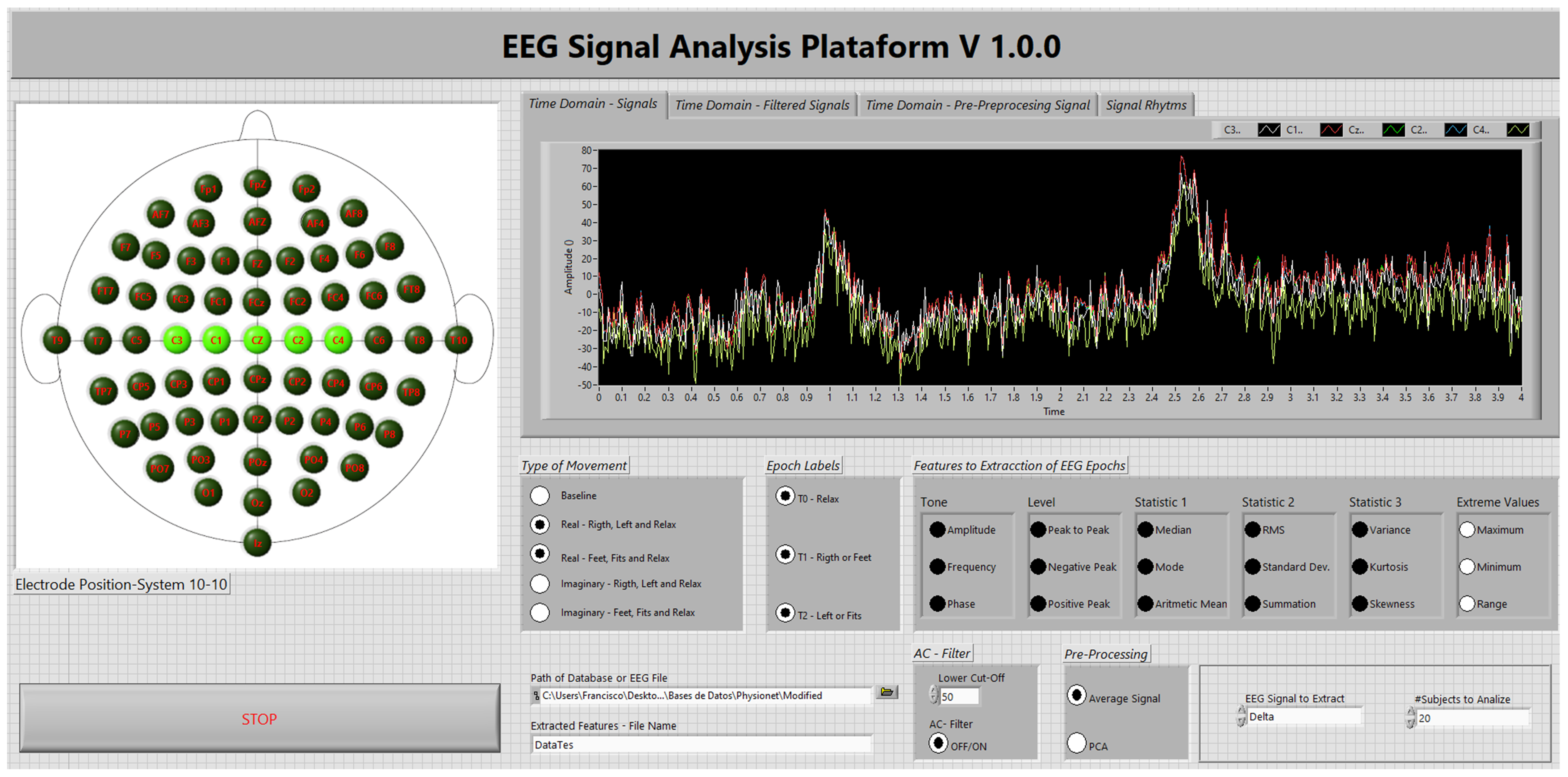
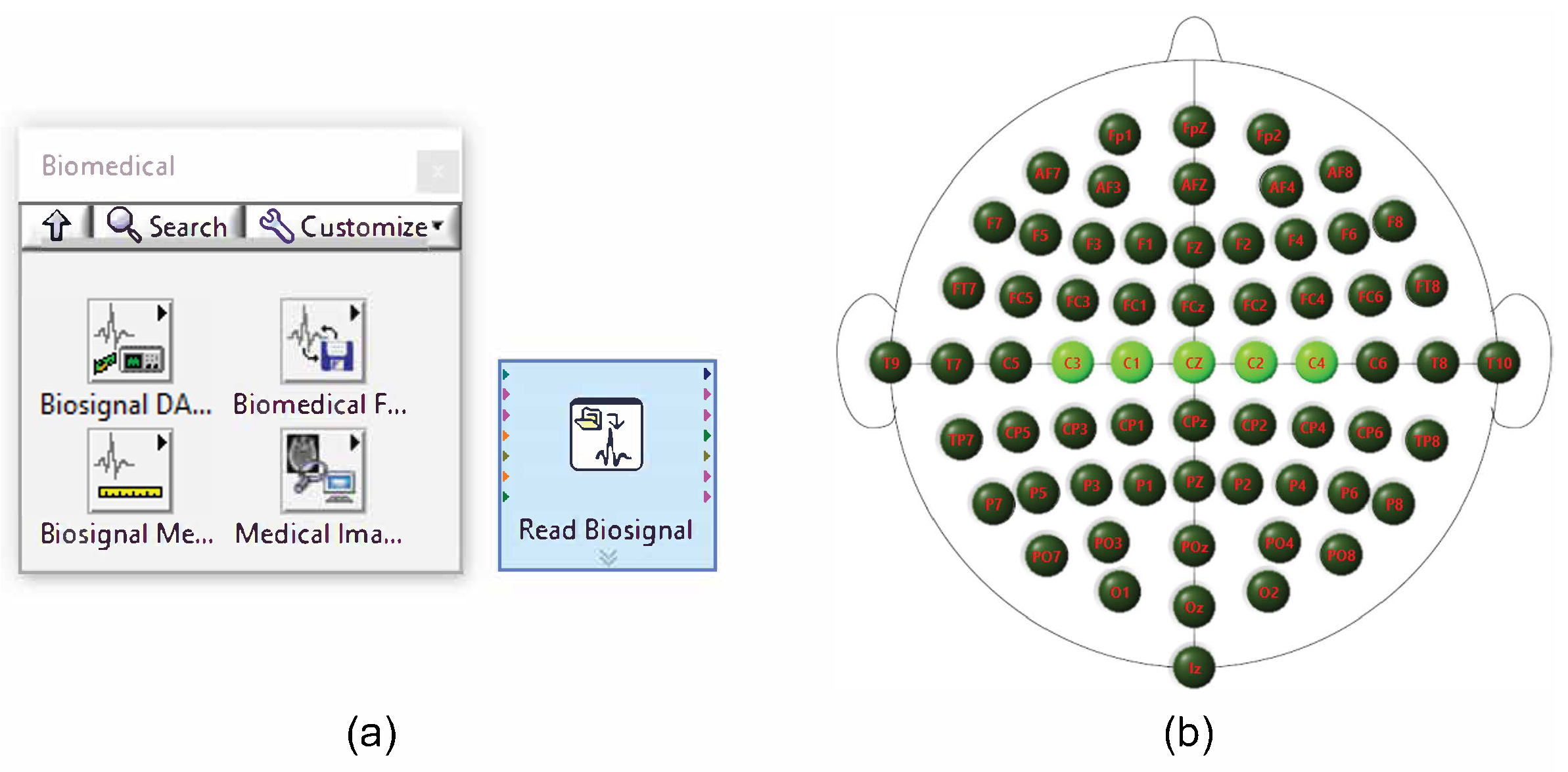
2.5. EEG Signal Acquisition and Channel Selection
2.6. Preprocessing
2.7. EEG Band Separation
2.8. Feature Extraction
Signal Analysis
- Tone measurements. The tone measurements carried out in the EEG signal epochs were the following: amplitude, frequency, and phase.
- Level measurements. The level measurements implemented in the EEG signal epochs were the following: peak-to-peak, negative peak, and positive peak.
- Statistical features. The statistical measurements applied to the different signal epochs were the following:
2.9. Dataset Preparation
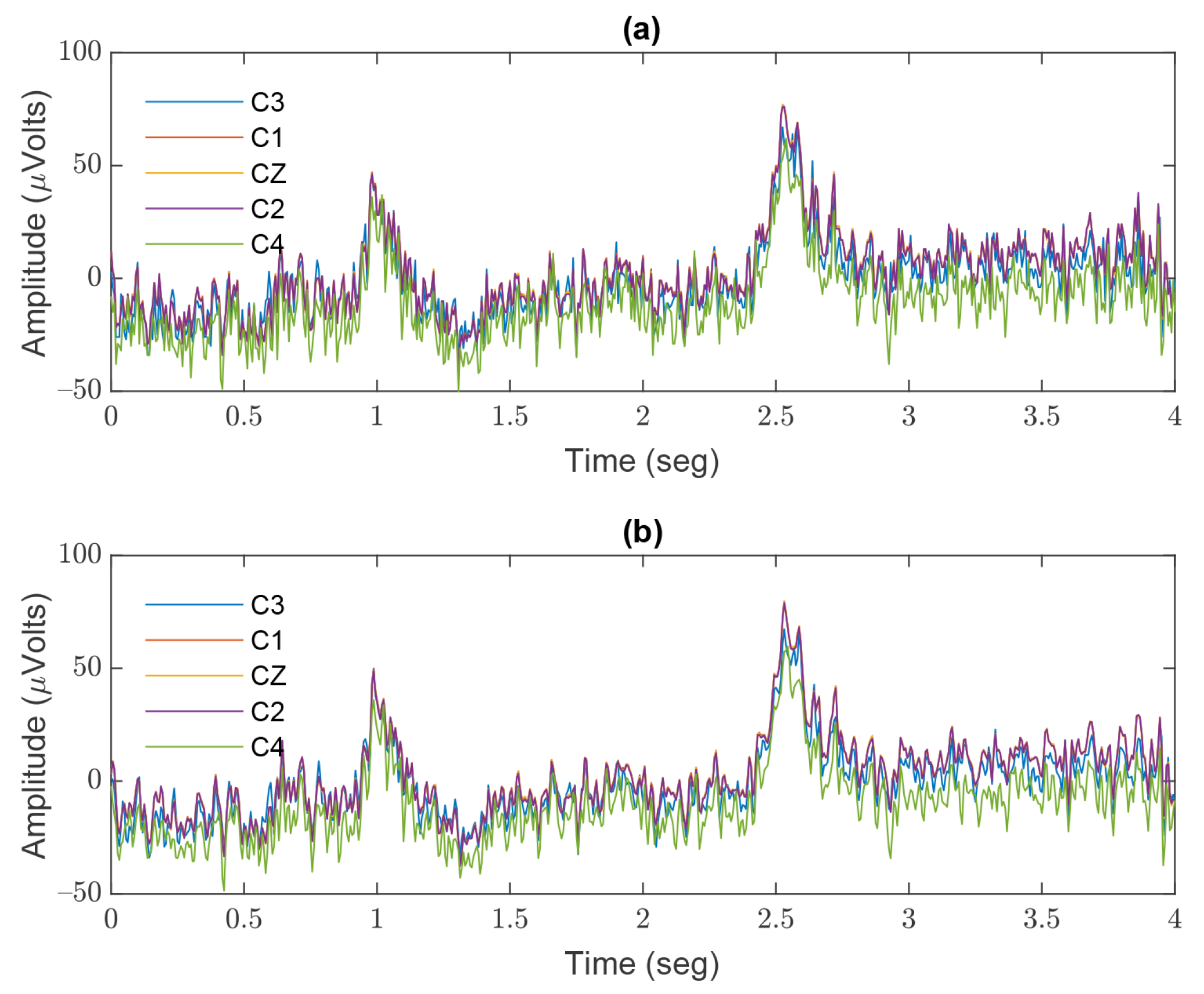
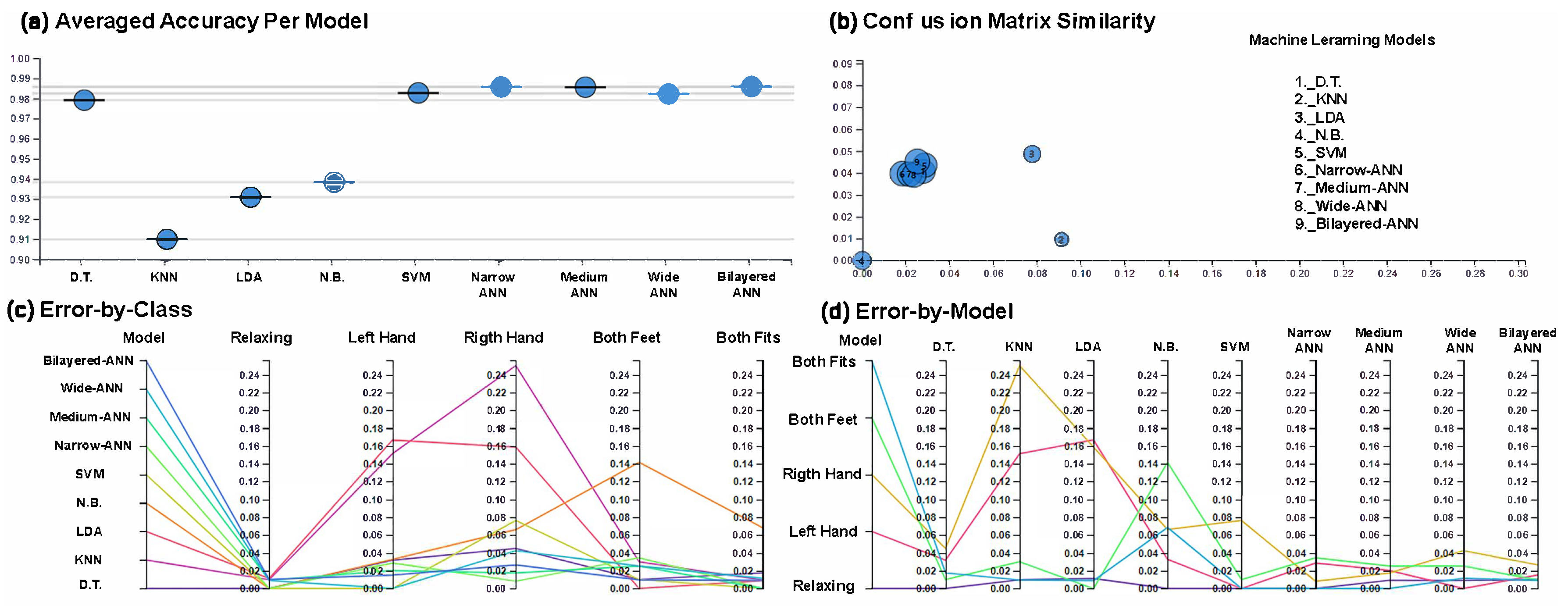
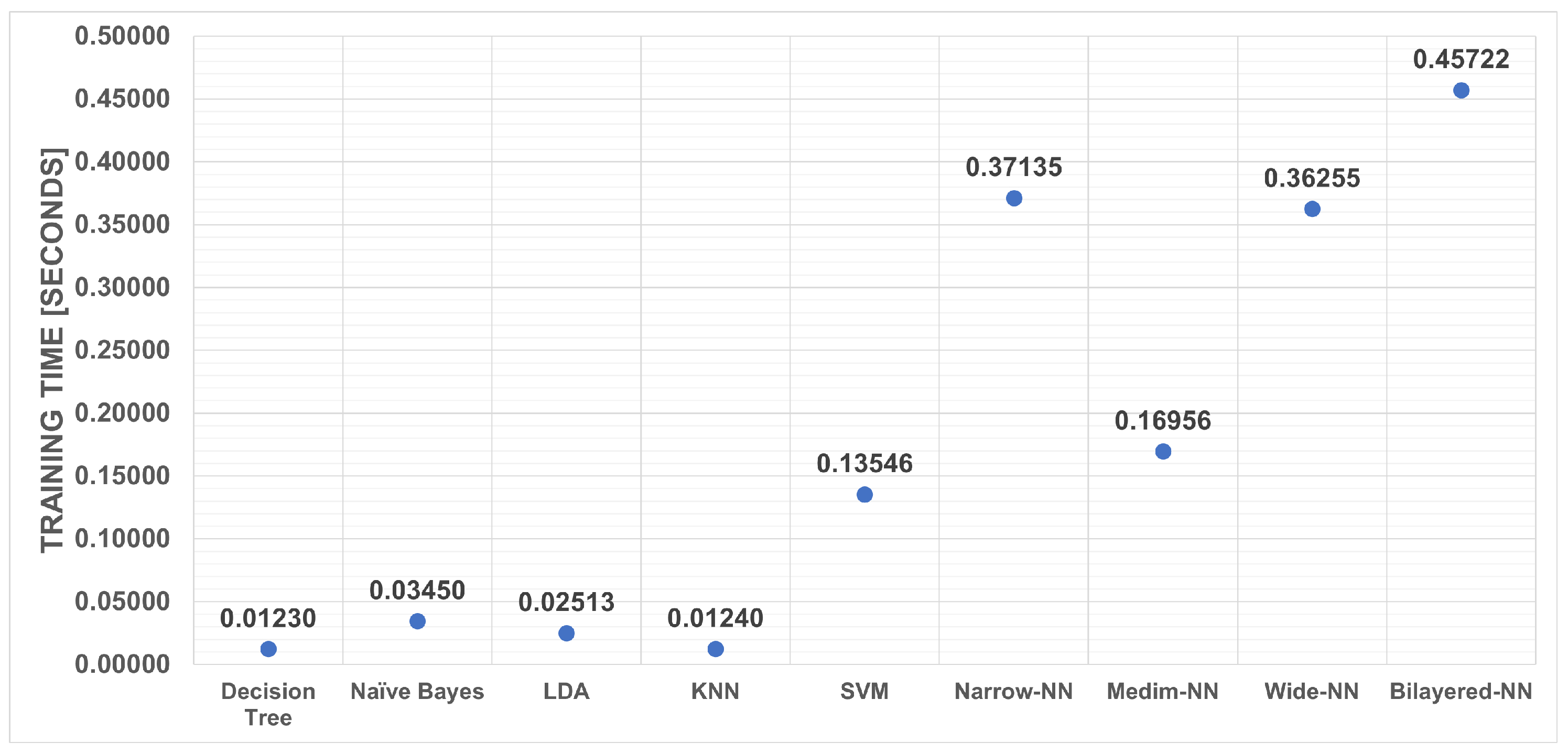
3. Results
4. Discussion
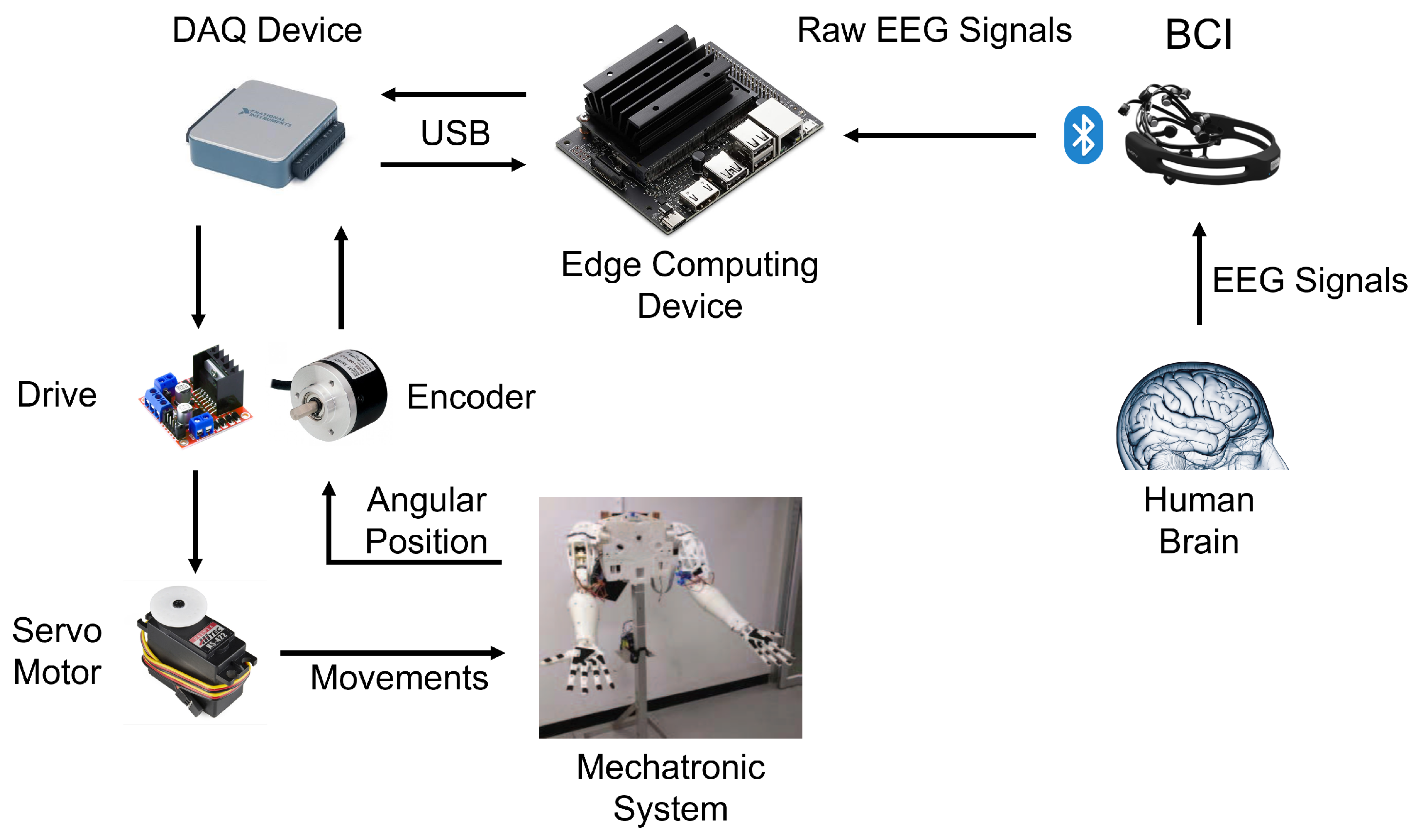
5. Proposed Usage Scenario
Limitations of the Study
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Fragment of the Dataset Created for This Study

Appendix B. Front Panel of Software (App) Developed for EEG Signal Analysis
References
- Savadkoohi, M.; Oladunni, T.; Thompson, L. A machine learning approach to epileptic seizure prediction using Electroencephalogram (EEG) Signal. Biocybern. Biomed. Eng. 2020, 40, 1328–1341. [Google Scholar] [CrossRef]
- Lent, R.; Azevedo, F.C.; Andrade-Moraes, C.; Pinto, A. How many neurons do you have? Some dogmas of quantitative neuroscience under revision. Eur. J. Neurosci. 2012, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Carter, R. The Human Brain Book: An Illustrated Guide to Its Structure, Function, and Disorders; Penguin: London, UK, 2019; Google-Books-ID: S8bhDwAAQBAJ. [Google Scholar]
- Gross, J. Magnetoencephalography in Cognitive Neuroscience: A Primer. Neuron 2019, 104, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Ince, N.F.; Goksu, F.; Tewfik, A.H. ECoG Based Brain Computer Interface with Subset Selection. In Biomedical Engineering Systems and Technologies; Fred, A., Filipe, J., Gamboa, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 357–374. [Google Scholar] [CrossRef]
- Son, J.; Ai, L.; Lim, R.; Xu, T.; Colcombe, S.; Franco, A.R.; Cloud, J.; LaConte, S.; Lisinski, J.; Klein, A.; et al. Evaluating fMRI-Based Estimation of Eye Gaze During Naturalistic Viewing. Cereb. Cortex 2020, 30, 1171–1184. [Google Scholar] [CrossRef] [PubMed]
- Coyle, S.; Ward, T.; Markham, C.; Mcdarby, G. On the suitability of near-infrared (NIR) systems for next-generation brain-computer interfaces. Physiol. Meas. 2004, 25, 815–822. [Google Scholar] [CrossRef]
- Ramadan, R.A.; Vasilakos, A.V. Brain computer interface: Control signals review. Neurocomputing 2017, 223, 26–44. [Google Scholar] [CrossRef]
- Fontanillo Lopez, C.A.; Li, G.; Zhang, D. Beyond Technologies of Electroencephalography-Based Brain-Computer Interfaces: A Systematic Review From Commercial and Ethical Aspects. Front. Neurosci. 2020, 14, 611130. [Google Scholar] [CrossRef]
- Esqueda-Elizondo, J.J.; Juárez-Ramírez, R.; López-Bonilla, O.R.; García-Guerrero, E.E.; Galindo-Aldana, G.M.; Jiménez-Beristáin, L.; Serrano-Trujillo, A.; Tlelo-Cuautle, E.; Inzunza-González, E. Attention Measurement of an Autism Spectrum Disorder User Using EEG Signals: A Case Study. Math. Comput. Appl. 2022, 27, 21. [Google Scholar] [CrossRef]
- Teplan, M. Fundamentals of Eeg Measurement. Meas. Sci. Rev. 2002, 2, 11. [Google Scholar]
- Tiwari, N.; Edla, D.R.; Dodia, S.; Bablani, A. Brain computer interface: A comprehensive survey. Biol. Inspired Cogn. Archit. 2018, 26, 118–129. [Google Scholar] [CrossRef]
- Luján, M.Á.; Jimeno, M.V.; Sotos, J.M.; Ricarte, J.J.; Borja, A.L. A Survey on EEG Signal Processing Techniques and Machine Learning: Applications to the Neurofeedback of Autobiographical Memory Deficits in Schizophrenia. Electronics 2021, 10, 3037. [Google Scholar] [CrossRef]
- Shoeibi, A.; Sadeghi, D.; Moridian, P.; Ghassemi, N.; Heras, J.; Alizadehsani, R.; Khadem, A.; Kong, Y.; Nahavandi, S.; Zhang, Y.D.; et al. Automatic Diagnosis of Schizophrenia in EEG Signals Using CNN-LSTM Models. Front. Neuroinform. 2021, 15, 777977. [Google Scholar] [CrossRef] [PubMed]
- Vrbancic, G.; Podgorelec, V. Automatic Classification of Motor Impairment Neural Disorders from EEG Signals Using Deep Convolutional Neural Networks. Electron. Electr. Eng. 2018, 24, 3–7. [Google Scholar] [CrossRef]
- Abdulkader, S.N.; Atia, A.; Mostafa, M.S.M. Brain computer interfacing: Applications and challenges. Egypt. Inform. J. 2015, 16, 213–230. [Google Scholar] [CrossRef]
- Neuper, C.; Müller, G.; Kübler, A.; Birbaumer, N.; Pfurtscheller, G. Clinical application of an EEG-based brain–computer interface: A case study in a patient with severe motor impairment. Clin. Neurophysiol. 2003, 114, 399. [Google Scholar] [CrossRef]
- Bartur, G.; Pratt, H.; Soroker, N. Changes in mu and beta amplitude of the EEG during upper limb movement correlate with motor impairment and structural damage in subacute stroke. Clin. Neurophysiol. 2019, 130, 1644–16451. [Google Scholar] [CrossRef]
- Sergeev, K.; Runnova, A.; Zhuravlev, M.; Kolokolov, O.; Akimova, N.; Kiselev, A.; Titova, A.; Slepnev, A.; Semenova, N.; Penzel, T. Wavelet skeletons in sleep EEG-monitoring as biomarkers of early diagnostics of mild cognitive impairment. Chaos 2021, 31, 073110. [Google Scholar] [CrossRef]
- Samuel, O.W.; Xiangxin, L.; Yanjuan, G.; Pang, F.; Shixiong, C.; Guanglin, L. Motor imagery classification of upper limb movements based on spectral domain features of EEG patterns. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju, Korea, 11–15 July 2017. [Google Scholar]
- Wang, H.; Song, Q.; Ma, T.; Cao, H.; Sun, Y. Study on Brain-Computer Interface Based on Mental Tasks. In Proceedings of the The 5th Annual IEEE International Conference on Cyber Technology in Automation, Control and Intelligent Systems, Shenyang, China, 8–12 June 2015; pp. 841–845. [Google Scholar] [CrossRef]
- Zander, T.O.; Kothe, C. Towards passive brain–computer interfaces: Applying brain–computer interface technology to human–machine systems in general. J. Neural Eng. 2011, 8, 025005. [Google Scholar] [CrossRef]
- Aggarwal, S.; Chugh, N. Signal processing techniques for motor imagery brain computer interface: A review. Array 2019, 1–2, 100003. [Google Scholar] [CrossRef]
- Mudgal, S.K.; Sharma, S.K.; Chaturvedi, J.; Sharma, A. Brain computer interface advancement in neurosciences: Applications and issues. Interdiscip. Neurosurg. 2020, 20, 100694. [Google Scholar] [CrossRef]
- Brunner, C.; Birbaumer, N.; Blankertz, B.; Guger, C.; Kübler, A.; Mattia, D.; Millán, J.d.R.; Miralles, F.; Nijholt, A.; Opisso, E.; et al. BNCI Horizon 2020: Towards a roadmap for the BCI community. Brain-Comput. Interfaces 2015, 2, 1–10. [Google Scholar] [CrossRef]
- Jurcak, V.; Tsuzuki, D.; Dan, I. 10/20, 10/10, and 10/5 systems revisited: Their validity as relative head-surface-based positioning systems. NeuroImage 2007, 34, 1600–1611. [Google Scholar] [CrossRef]
- Peng, H.; Hu, B.; Qi, Y.; Zhao, Q.; Ratcliffe, M. An improved EEG de-noising approach in electroencephalogram (EEG) for home care. In Proceedings of the 2011 5th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth) and Workshops, Dublin, Ireland, 23–26 May 2011; pp. 469–474. [Google Scholar] [CrossRef]
- Khalid, S.; Khalil, T.; Nasreen, S. A survey of feature selection and feature extraction techniques in machine learning. In Proceedings of the 2014 Science and Information Conference, Karlovy Vary, Czech Republic, 27–28 December 2014; pp. 372–378. [Google Scholar] [CrossRef]
- Saeidi, M.; Karwowski, W.; Farahani, F.V.; Fiok, K.; Taiar, R.; Hancock, P.A.; Al-Juaid, A. Neural Decoding of EEG Signals with Machine Learning: A Systematic Review. Brain Sci. 2021, 11, 1525. [Google Scholar] [CrossRef] [PubMed]
- Stancin, I.; Cifrek, M.; Jovic, A. A Review of EEG Signal Features and Their Application in Driver Drowsiness Detection Systems. Sensors 2021, 21, 3786. [Google Scholar] [CrossRef] [PubMed]
- Subasi, A.; Ismail Gursoy, M. EEG signal classification using PCA, ICA, LDA and support vector machines. Expert Syst. Appl. 2010, 37, 8659–8666. [Google Scholar] [CrossRef]
- Yazdani, A.; Ebrahimi, T.; Hoffmann, U. Classification of EEG signals using Dempster Shafer theory and a k-nearest neighbor classifier. In Proceedings of the 2009 4th International IEEE/EMBS Conference on Neural Engineering, Antalya, Turkey, 29 April–2 May 2009; pp. 327–330. [Google Scholar] [CrossRef]
- Edla, D.R.; Mangalorekar, K.; Dhavalikar, G.; Dodia, S. Classification of EEG data for human mental state analysis using Random Forest Classifier. Procedia Comput. Sci. 2018, 132, 1523–1532. [Google Scholar] [CrossRef]
- Saragih, A.S.; Pamungkas, A.; Zain, B.Y.; Ahmed, W. Electroencephalogram (EEG) Signal Classification Using Artificial Neural Network to Control Electric Artificial Hand Movement. IOP Conf. Ser. Mater. Sci. Eng. 2020, 938, 012005. [Google Scholar] [CrossRef]
- Han, Y.; Ma, Y.; Zhu, L.; Zhang, Y.; Li, L.; Zheng, W.; Guo, J.; Che, Y. Study on Mind Controlled Robotic Arms by Collecting and Analyzing Brain Alpha Waves; Atlantis Press: Amsterdam, The Netherlands, 2018; pp. 145–148. [Google Scholar] [CrossRef]
- Casey, A.; Azhar, H.; Grzes, M.; Sakel, M. BCI controlled robotic arm as assistance to the rehabilitation of neurologically disabled patients. Disabil. Rehabil. Assist. Technol. 2021, 16, 525–537. [Google Scholar] [CrossRef]
- Van Erp, J.; Lotte, F.; Tangermann, M. Brain-Computer Interfaces: Beyond Medical Applications. Computer 2012, 45, 26–34. [Google Scholar] [CrossRef]
- Navarro-Espinoza, A.; López-Bonilla, O.R.; García-Guerrero, E.E.; Tlelo-Cuautle, E.; López-Mancilla, D.; Hernández-Mejía, C.; Inzunza-González, E. Traffic Flow Prediction for Smart Traffic Lights Using Machine Learning Algorithms. Technologies 2022, 10, 5. [Google Scholar] [CrossRef]
- Cerrada, M.; Trujillo, L.; Hernández, D.E.; Correa Zevallos, H.A.; Macancela, J.C.; Cabrera, D.; Vinicio Sánchez, R. AutoML for Feature Selection and Model Tuning Applied to Fault Severity Diagnosis in Spur Gearboxes. Math. Comput. Appl. 2022, 27, 6. [Google Scholar] [CrossRef]
- Enríquez Zárate, J.; Gómez López, M.d.l.A.; Carmona Troyo, J.A.; Trujillo, L. Analysis and Detection of Erosion in Wind Turbine Blades. Math. Comput. Appl. 2022, 27, 5. [Google Scholar] [CrossRef]
- Janiesch, C.; Zschech, P.; Heinrich, K. Machine learning and deep learning. Electron. Mark. 2021, 31, 685–695. [Google Scholar] [CrossRef]
- Fong-Mata, M.B.; García-Guerrero, E.E.; Mejía-Medina, D.A.; López-Bonilla, O.R.; Villarreal-Gómez, L.J.; Zamora-Arellano, F.; López-Mancilla, D.; Inzunza-González, E. An Artificial Neural Network Approach and a Data Augmentation Algorithm to Systematize the Diagnosis of Deep-Vein Thrombosis by Using Wells’ Criteria. Electronics 2020, 9, 1810. [Google Scholar] [CrossRef]
- Cho, J.H.; Jeong, J.H.; Shim, K.H.; Kim, D.J.; Lee, S.W. Classification of Hand Motions within EEG Signals for Non-Invasive BCI-Based Robot Hand Control. In Proceedings of the 2018 IEEE International Conference on Systems, Man, and Cybernetics (SMC), Miyazaki, Japan, 7–10 October 2018; pp. 515–518. [Google Scholar] [CrossRef]
- Roy, G.; Bhoi, A.K.; Bhaumik, S. A Comparative Approach for MI-Based EEG Signals Classification Using Energy, Power and Entropy. IRBM 2021. [Google Scholar] [CrossRef]
- You, Y.; Chen, W.; Zhang, T. Motor imagery EEG classification based on flexible analytic wavelet transform. Biomed. Signal Process. Control 2020, 62, 102069. [Google Scholar] [CrossRef]
- Faiz, M.Z.A.; Al-Hamadani, A.A. Online Brain Computer Interface Based Five Classes EEG To Control Humanoid Robotic Hand. In Proceedings of the 2019 42nd International Conference on Telecommunications and Signal Processing (TSP), Budapest, Hungary, 1–3 July 2019; pp. 406–410. [Google Scholar] [CrossRef]
- LeCun, Y.; Bengio, Y.; Hinton, G. Deep learning. Nature 2015, 521, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ling, C. A strategy to apply machine learning to small datasets in materials science. NPJ Comput. Mater. 2018, 4, 25. [Google Scholar] [CrossRef]
- Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat. Mach. Intell. 2019, 1, 206–215. [Google Scholar] [CrossRef]
- Alomari, M.H.; Samaha, A.; AlKamha, K. Automated Classification of L/R Hand Movement EEG Signals using Advanced Feature Extraction and Machine Learning. arXiv 2013, arXiv:1312.2877. [Google Scholar] [CrossRef]
- Pinheiro, O.; Alves, L.; Souza, J. EEG Signals Classification: Motor Imagery for Driving an Intelligent Wheelchair. IEEE Lat. Am. Trans. 2018, 16, 254–259. [Google Scholar] [CrossRef]
- Bousseta, R.; El Ouakouak, I.; Gharbi, M.; Regragui, F. EEG Based Brain Computer Interface for Controlling a Robot Arm Movement Through Thought. IRBM 2018, 39, 129–135. [Google Scholar] [CrossRef]
- Tang, Z.; Sun, S.; Zhang, S.; Chen, Y.; Li, C.; Chen, S. A Brain-Machine Interface Based on ERD/ERS for an Upper-Limb Exoskeleton Control. Sensors 2016, 16, 2050. [Google Scholar] [CrossRef] [PubMed]
- Kant, P.; Laskar, S.H.; Hazarika, J.; Mahamune, R. CWT Based Transfer Learning for Motor Imagery Classification for Brain computer Interfaces. J. Neurosci. Methods 2020, 345, 108886. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Uslu, S.; Rittichier, K.J.; Durresi, A. Trustworthy Artificial Intelligence: A Review. ACM Comput. Surv. 2022, 55, 1–38. [Google Scholar] [CrossRef]
- Theissler, A.; Thomas, M.; Burch, M.; Gerschner, F. ConfusionVis: Comparative evaluation and selection of multi-class classifiers based on confusion matrices. Knowl.-Based Syst. 2022, 247, 108651. [Google Scholar] [CrossRef]
- Sabharwal, R.; Miah, S.J. An intelligent literature review: Adopting inductive approach to define machine learning applications in the clinical domain. J. Big Data 2022, 9, 53. [Google Scholar] [CrossRef]
- Haque, R.; Islam, N.; Islam, M.; Ahsan, M.M. A Comparative Analysis on Suicidal Ideation Detection Using NLP, Machine, and Deep Learning. Technologies 2022, 10, 57. [Google Scholar] [CrossRef]
- Contreras-Luján, E.E.; García-Guerrero, E.E.; López-Bonilla, O.R.; Tlelo-Cuautle, E.; López-Mancilla, D.; Inzunza-González, E. Evaluation of Machine Learning Algorithms for Early Diagnosis of Deep Venous Thrombosis. Math. Comput. Appl. 2022, 27, 24. [Google Scholar] [CrossRef]
- Aboneh, T.; Rorissa, A.; Srinivasagan, R. Stacking-Based Ensemble Learning Method for Multi-Spectral Image Classification. Technologies 2022, 10, 17. [Google Scholar] [CrossRef]
- Bi, L.; Fan, X.; Liu, Y. EEG-Based Brain-Controlled Mobile Robots: A Survey. IEEE Trans. Hum.-Mach. Syst. 2013, 43, 161–176. [Google Scholar] [CrossRef]
- Abbasi, B.; Goldenholz, D.M. Machine learning applications in epilepsy. Epilepsia 2019, 60, 2037–2047. [Google Scholar] [CrossRef] [PubMed]
- Majidov, I.; Whangbo, T. Efficient Classification of Motor Imagery Electroencephalography Signals Using Deep Learning Methods. Sensors 2019, 19, 1736. [Google Scholar] [CrossRef] [PubMed]
- Craik, A.; He, Y.; Contreras-Vidal, J.L. Deep learning for electroencephalogram (EEG) classification tasks: A review. J. Neural Eng. 2019, 16, 031001. [Google Scholar] [CrossRef] [PubMed]
- Padfield, N.; Zabalza, J.; Zhao, H.; Masero, V.; Ren, J. EEG-based brain-computer interfaces using motor-imagery: Techniques and challenges. Sensors 2019, 19, 1423. [Google Scholar] [CrossRef] [PubMed]
- Lawhern, V.J.; Solon, A.J.; Waytowich, N.R.; Gordon, S.M.; Hung, C.P.; Lance, B.J. EEGNet: A compact convolutional neural network for EEG-based brain-computer interfaces. J. Neural Eng. 2018, 15, 056013. [Google Scholar] [CrossRef]
- Lotte, F.; Bougrain, L.; Cichocki, A.; Clerc, M.; Congedo, M.; Rakotomamonjy, A.; Yger, F. A review of classification algorithms for EEG-based brain-computer interfaces: A 10 year update. J. Neural Eng. 2018, 15, 031005. [Google Scholar] [CrossRef]
- Mridha, M.F.; Das, S.C.; Kabir, M.M.; Lima, A.A.; Islam, M.R.; Watanobe, Y. Brain-Computer Interface: Advancement and Challenges. Sensors 2021, 21, 5746. [Google Scholar] [CrossRef]
- Samuel, O.W.; Geng, Y.; Li, X.; Li, G. Towards Efficient Decoding of Multiple Classes of Motor Imagery Limb Movements Based on EEG Spectral and Time Domain Descriptors. J. Med. Syst. 2017, 41, 194. [Google Scholar] [CrossRef]
- Goldberger, A.L.; Amaral, L.A.N.; Glass, L.; Hausdorff, J.M.; Ivanov, P.C.; Mark, R.G.; Mietus, J.E.; Moody, G.B.; Peng, C.K.; Stanley, H.E. PhysioBank, PhysioToolkit, and PhysioNet. Circulation 2000, 101, e215–e220. [Google Scholar] [CrossRef]
- Hosseini, M.P.; Hosseini, A.; Ahi, K. A Review on Machine Learning for EEG Signal Processing in Bioengineering. IEEE Rev. Biomed. Eng. 2021, 14, 204–218. [Google Scholar] [CrossRef] [PubMed]
- Schalk, G.; McFarland, D.J.; Hinterberger, T.; Birbaumer, N.; Wolpaw, J.R. BCI2000: A general-purpose brain-computer interface (BCI) system. IEEE Trans. Biomed. Eng. 2004, 51, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- Kemp, B.; Olivan, J. European data format ‘plus’ (EDF+), an EDF alike standard format for the exchange of physiological data. Clin. Neurophysiol. 2003, 114, 1755–1761. [Google Scholar] [CrossRef]
- Neuper, C.; Pfurtscheller, G. Evidence for distinct beta resonance frequencies in human EEG related to specific sensorimotor cortical areas. Clin. Neurophysiol. 2001, 112, 2084–2097. [Google Scholar] [CrossRef]
- Deecke, L.; Weinberg, H.; Brickett, P. Magnetic fields of the human brain accompanying voluntary movement: Bereitschaftsmagnetfeld. Exp. Brain Res. 1982, 48, 144–148. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Ushiba, J. EEG-based classification of imaginary left and right foot movements using beta rebound. Clin. Neurophysiol. 2013, 124, 2153–2160. [Google Scholar] [CrossRef]
- Sleight, J.; Pillai, P.J.; Mohan, S. Classification of Executed and Imagined Motor Movement EEG Signals. Comput. Sci. 2009. [Google Scholar]
- Lee, K.; Liu, D.; Perroud, L.; Chavarriaga, R.; Millán, J.d.R. A brain-controlled exoskeleton with cascaded event-related desynchronization classifiers. Spec. Issue New Res. Front. Intell. Auton. Syst. 2017, 90, 15–23. [Google Scholar] [CrossRef]
- Phinyomark, A.; Thongpanja, S.; Hu, H.; Phukpattaranont, P.; Limsakul, C. The Usefulness of Mean and Median Frequencies in Electromyography Analysis. In Computational Intelligence in Electromyography Analysis: A Perspective on Current Applications and Future Challenges; InTech: London, UK, 2012. [Google Scholar] [CrossRef]
- Zwillinger, D.; Kokoska, S. CRC Standard Probability and Statistics Tables and Formulae; CRC Press: Boca Raton, FL, USA, 1999; Google-Books-ID: TB3RVEZ0UIMC. [Google Scholar]
- Jatupaiboon, N.; Pan-ngum, S.; Israsena, P. Emotion classification using minimal EEG channels and frequency bands. In Proceedings of the 2013 10th International Joint Conference on Computer Science and Software Engineering (JCSSE), Lampang, Thailand, 2–30 June 2013; pp. 21–24. [Google Scholar] [CrossRef]
- Al-Ani, A.; Al-Sukker, A. Effect of Feature and Channel Selection on EEG Classification. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 30 August–3 September 2006; pp. 2171–2174. [Google Scholar] [CrossRef]
- Sadrawi, M.; Sun, W.Z.; Ma, M.M.; Yeh, Y.T.; Abbod, M.; Shieh, J.S. Ensemble Genetic Fuzzy Neuro Model Applied for the Emergency Medical Service via Unbalanced Data Evaluation. Symmetry 2018, 10, 71. [Google Scholar] [CrossRef]
- Guang-Hui, F.; Feng, X.; Bing-Yang, Z.; Lun-Zhao, Y. Stable variable selection of class-imbalanced data with precision-recall criterion. Chemom. Intell. Lab. Syst. 2017, 171, 241–250. [Google Scholar] [CrossRef]
- Castro, C.; Vargas-Viveros, E.; Sánchez, A.; Gutiérrez-López, E.; Flores, D. Parkinson’s Disease Classification Using Artificial Neural Networks. In Proceedings of the VIII Latin American Conference on Biomedical Engineering and XLII National Conference on Biomedical Engineering, CLAIB 2019, Cancun, Mexico, 2–5 October 2019; Volume 75. [Google Scholar] [CrossRef]
- Metrics for Multi-Class Classification: An Overview. arXiv 2020, arXiv:2008.05756.
- Vieira, S.M.; Kaymak, U.; Sousa, J.M.C. Cohen’s kappa coefficient as a performance measure for feature selection. In Proceedings of the International Conference on Fuzzy Systems, Yantai, China, 10–12 August 2010; pp. 1–8. [Google Scholar] [CrossRef]
- Matthews, B.W. Comparison of the predicted and observed secondary structure of T4 phage lysozyme. Biochim. Biophys. Acta (BBA)-Protein Struct. 1975, 405, 442–451. [Google Scholar] [CrossRef]
- Aguirre-Castro, O.; García-Guerrero, E.; López-Bonilla, O.; Tlelo-Cuautle, E.; López-Mancilla, D.; Cárdenas-Valdez, J.; Olguín-Tiznado, J.; Inzunza-González, E. Evaluation of underwater image enhancement algorithms based on Retinex and its implementation on embedded systems. Neurocomputing 2022, 494, 148–159. [Google Scholar] [CrossRef]


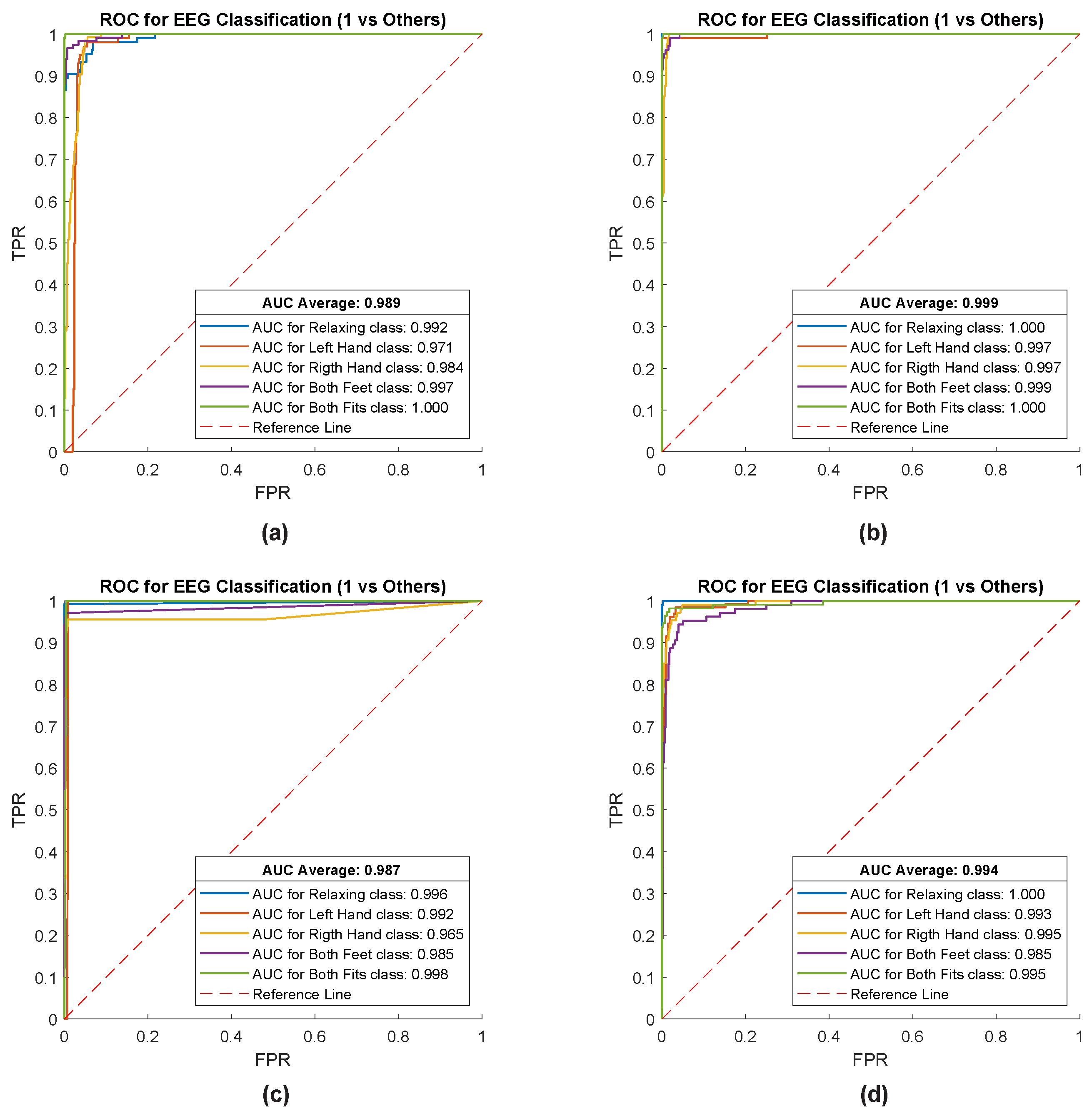

| Task | Real Movement | Imaginary Movement | To | T1 | T2 | Duration |
|---|---|---|---|---|---|---|
| 1 | Open Eyes | - | Relaxing | - | - | 1 min |
| 2 | Close Eyes | - | Relaxing | - | - | 1 min |
| 3 | Fist | - | Relaxing | Left | Right | 2 min |
| 4 | - | Fist | Relaxing | Left | Right | 2 min |
| 5 | Fist/Feet | - | Relaxing | Fist | Feet | 2 min |
| 6 | - | Fist/Feet | Relaxing | Fist | Feet | 2 min |
| 7 | Fist | - | Relaxing | Left | Right | 2 min |
| 8 | - | Fist | Relaxing | Left | Right | 2 min |
| 9 | Fist/Feet | - | Relaxing | Fist | Feet | 2 min |
| 10 | - | Fist/Feet | Relaxing | Fist | Feet | 2 min |
| 11 | Fist | - | Relaxing | Left | Right | 2 min |
| 12 | - | Fist | Relaxing | Left | Right | 2 min |
| 13 | Fist/Feet | - | Relaxing | Fist | Feet | 2 min |
| 14 | - | Fist/Feet | Relaxing | Fist | Feet | 2 min |
| Band of EEG Signal | Low Cut-Off Frequency | High Cut-Off Frequency |
|---|---|---|
| Delta | 0.1 Hz | 3.99 Hz |
| Theta | 4.0 Hz | 7.99 Hz |
| Alpha | 8.0 Hz | 11.99 Hz |
| Beta | 12.0 Hz | 29.99 Hz |
| Gamma | 30.0 Hz | 49.99 Hz |
| Features of the Channels for the Different Electrode Positions | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Band | C3 | C3 | C3 | C1 | C1 | C1 | Cz | Cz | Cz | C2 | C2 | C2 | C4 | C4 | C4 | Class |
| Delta | Amplitude | Frequency | Phase | Peak to Peak | Neg.Peak | Pos.Peak | Median | Mode | Mean | RMS | S.D. | Summation | Variance | Kurtosis | Skewness | Relaxing |
| Theta | Amplitude | Frequency | Phase | Peak to Peak | Neg.Peak | Pos.Peak | Median | Mode | Mean | RMS | S.D. | Summation | Variance | Kurtosis | Skewness | Left Hand |
| Alpha | Amplitude | Frequency | Phase | Peak to Peak | Neg.Peak | Pos.Peak | Median | Mode | Mean | RMS | S.D. | Summation | Variance | Kurtosis | Skewness | Right Hand |
| Beta | Amplitude | Frequency | Phase | Peak to Peak | Neg.Peak | Pos.Peak | Median | Mode | Mean | RMS | S.D. | Summation | Variance | Kurtosis | Skewness | Fist |
| Gamma | Amplitude | Frequency | Phase | Peak to Peak | Neg.Peak | Pos.Peak | Median | Mode | Mean | RMS | S.D. | Summation | Variance | Kurtosis | Skewness | Feet |
| Average Scoring Parameters | ||||||
|---|---|---|---|---|---|---|
| ML Algorithm | Accuracy | Error | Recall | Specificity | Precision | F1-Score |
| LDA | 0.9229 | 0.0771 | 0.9219 | 0.9807 | 0.9332 | 0.9228 |
| D.T. | 0.9803 | 0.0197 | 0.9777 | 0.9951 | 0.9792 | 0.9783 |
| KNN | 0.8996 | 0.1004 | 0.9037 | 0.9747 | 0.9099 | 0.9047 |
| N.B. | 0.9373 | 0.0627 | 0.9384 | 0.9844 | 0.9382 | 0.9378 |
| SVM | 0.9803 | 0.0197 | 0.9789 | 0.9950 | 0.9827 | 0.9803 |
| Narrow-ANN | 0.9857 | 0.0143 | 0.9863 | 0.9964 | 0.9857 | 0.9859 |
| Medium-ANN | 0.9857 | 0.0143 | 0.9854 | 0.9964 | 0.9856 | 0.9855 |
| Wide-ANN | 0.9821 | 0.0179 | 0.9834 | 0.9955 | 0.9824 | 0.9828 |
| Bilayered-ANN | 0.9857 | 0.0143 | 0.9854 | 0.9964 | 0.9859 | 0.9856 |
| Performance Metrics | ||||
|---|---|---|---|---|
| ML Algorithm | AUC Average | Cohen’s Kappa Coefficient | Matthews Correlation Coefficient | Loss |
| LDA | 0.9889 | 0.7592 | 0.9072 | 0.0787 |
| D.T. | 0.9873 | 0.9384 | 0.9736 | 0.0229 |
| KNN | 0.9392 | 0.6864 | 0.8810 | 0.0961 |
| N.B. | 0.9935 | 0.8040 | 0.9225 | 0.0616 |
| SVM | 0.9988 | 0.9384 | 0.9757 | 0.0217 |
| Narrow-ANN | 0.9982 | 0.9552 | 0.9824 | 0.0136 |
| Medium-ANN | 0.9998 | 0.9552 | 0.9819 | 0.0147 |
| Wide-ANN | 0.9984 | 0.9440 | 0.9783 | 0.0165 |
| Bilayered-ANN | 0.9988 | 0.9552 | 0.9820 | 0.0147 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Arias, F.J.; García-Guerrero, E.E.; Tlelo-Cuautle, E.; Colores-Vargas, J.M.; García-Canseco, E.; López-Bonilla, O.R.; Galindo-Aldana, G.M.; Inzunza-González, E. Evaluation of Machine Learning Algorithms for Classification of EEG Signals. Technologies 2022, 10, 79. https://doi.org/10.3390/technologies10040079
Ramírez-Arias FJ, García-Guerrero EE, Tlelo-Cuautle E, Colores-Vargas JM, García-Canseco E, López-Bonilla OR, Galindo-Aldana GM, Inzunza-González E. Evaluation of Machine Learning Algorithms for Classification of EEG Signals. Technologies. 2022; 10(4):79. https://doi.org/10.3390/technologies10040079
Chicago/Turabian StyleRamírez-Arias, Francisco Javier, Enrique Efren García-Guerrero, Esteban Tlelo-Cuautle, Juan Miguel Colores-Vargas, Eloisa García-Canseco, Oscar Roberto López-Bonilla, Gilberto Manuel Galindo-Aldana, and Everardo Inzunza-González. 2022. "Evaluation of Machine Learning Algorithms for Classification of EEG Signals" Technologies 10, no. 4: 79. https://doi.org/10.3390/technologies10040079
APA StyleRamírez-Arias, F. J., García-Guerrero, E. E., Tlelo-Cuautle, E., Colores-Vargas, J. M., García-Canseco, E., López-Bonilla, O. R., Galindo-Aldana, G. M., & Inzunza-González, E. (2022). Evaluation of Machine Learning Algorithms for Classification of EEG Signals. Technologies, 10(4), 79. https://doi.org/10.3390/technologies10040079










