Cystatin C: A Primer for Pharmacists
Abstract
1. Introduction
2. Overview of Measured Versus Estimated GFR
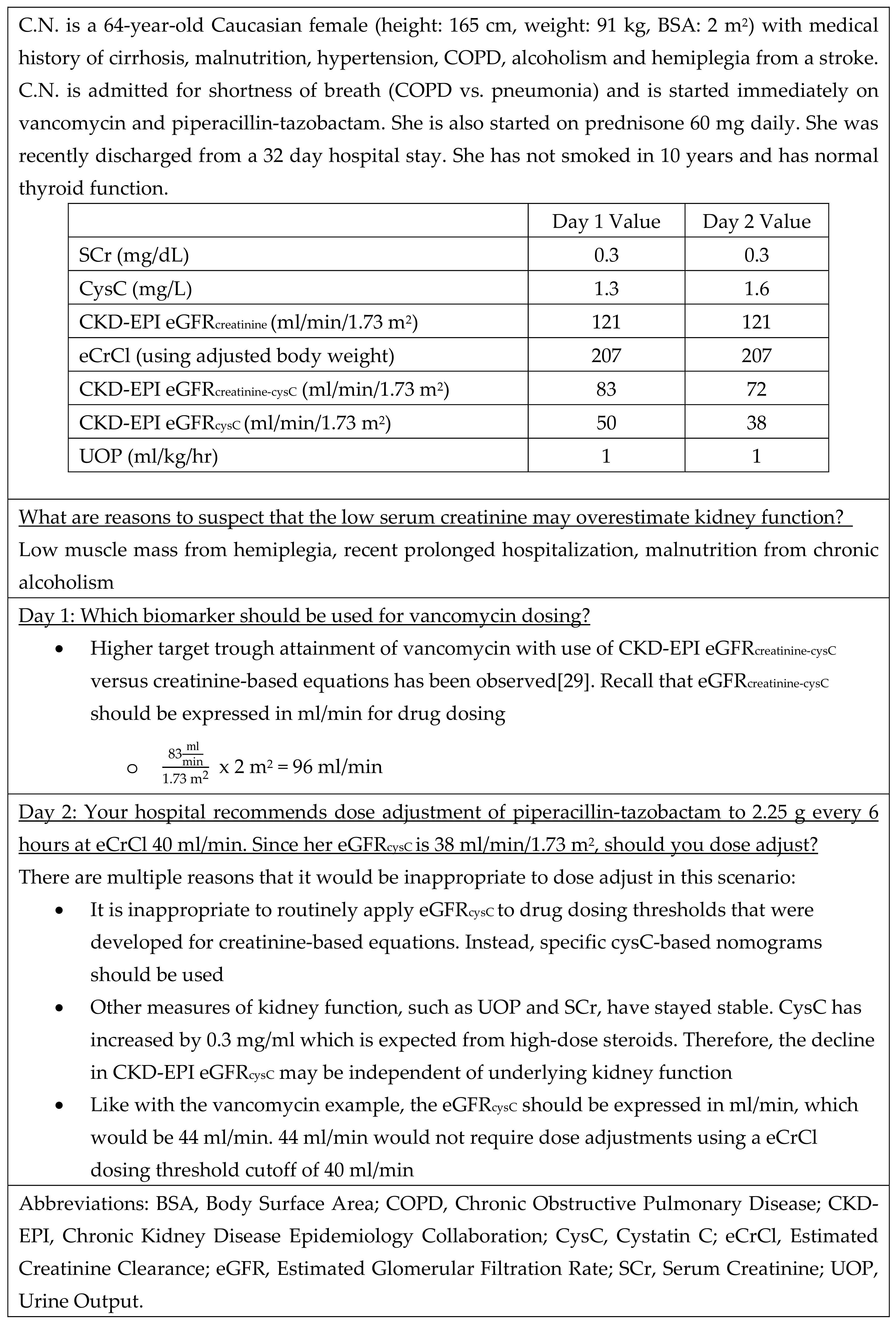
3. Medication Dosing Based on cysC
4. Special Populations with cysC
4.1. Obesity
4.2. Disorders of Compromised Muscle Mass or Cachexia
4.3. Malignancy
4.4. Corticosteroids
4.5. Transplant
4.5.1. Kidney Transplant
4.5.2. Non-Kidney Transplant
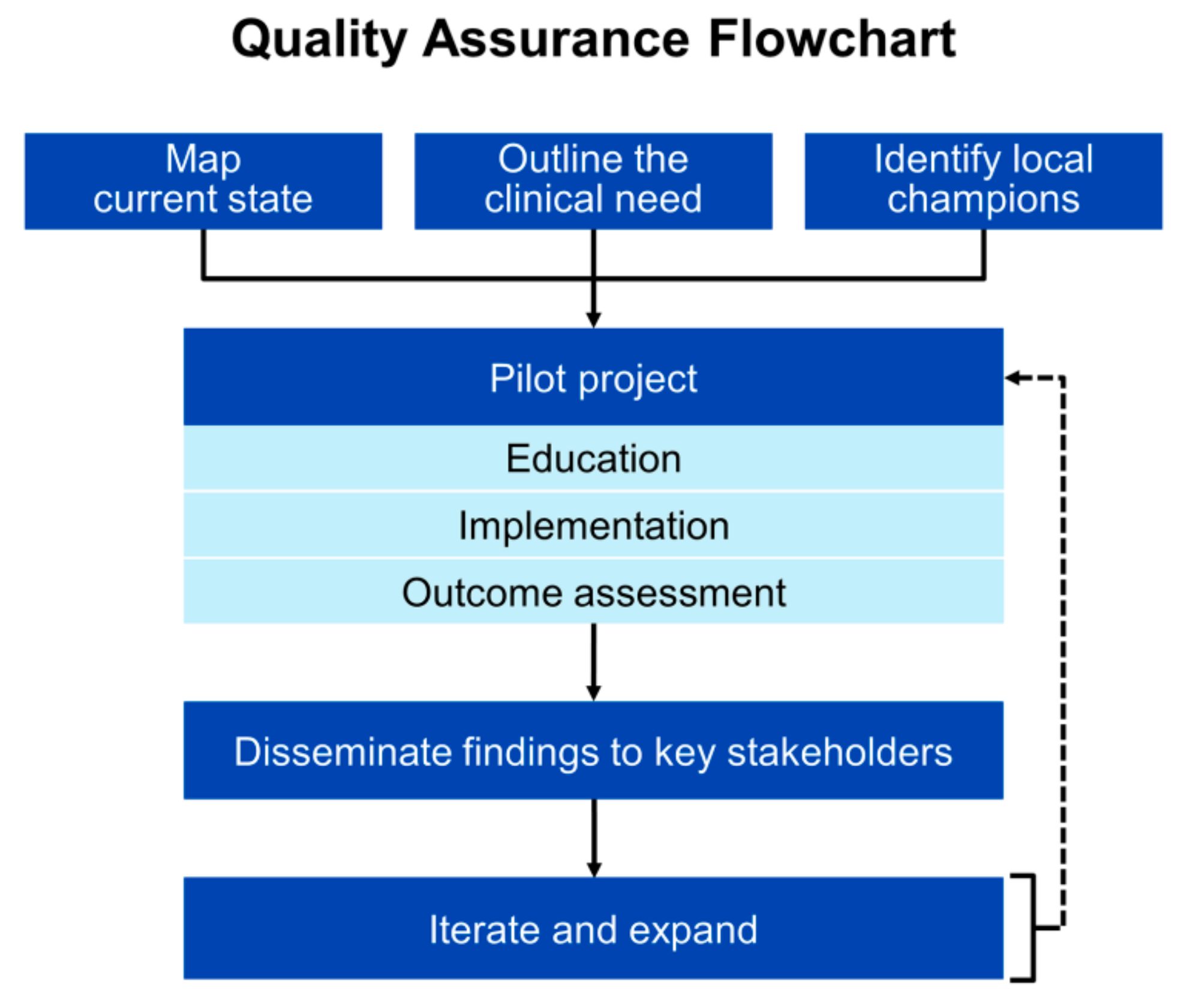
5. Implementation
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Seyffart, G. Seyffart’s Directory of Drug Dosage in Kidney Disease, 1st ed.; Dustri-Verlag Dr. Karl Feistle: Oberhaching, Germany, 2011; ISBN 387185400X. [Google Scholar]
- Elinder, C.G.; Bárány, P.; Heimbürger, O. The use of estimated glomerular filtration rate for dose adjustment of medications in the elderly. Drugs Aging 2014, 31, 493–499. [Google Scholar] [CrossRef]
- Taber, S.S.; Mueller, B.A. Drug-associated renal dysfunction. Crit. Care Clin. 2006, 22, 357–374. [Google Scholar] [CrossRef]
- Shlipak, M.G.; Mattes, M.D.; Peralta, C.A. Update on cystatin C: Incorporation into clinical practice. Am. J. Kidney Dis. 2013, 62, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Tawadrous, D.; Shariff, S.Z.; Haynes, R.B.; Iansavichus, A.V.; Jain, A.K.; Garg, A.X. Use of clinical decision support systems for kidney-related drug prescribing: A systematic review. Am. J. Kidney Dis. 2011, 58, 903–914. [Google Scholar] [CrossRef] [PubMed]
- Galanter, W.L.; Moja, J.; Lambert, B.L. Using computerized provider order entry and clinical decision support to improve prescribing in patients with decreased GFR. Am. J. Kidney Dis. 2010, 56, 809–812. [Google Scholar] [CrossRef]
- Levey, A.S.; Inker, L.A. Assessment of Glomerular Filtration Rate in Health and Disease: A State of the Art Review. Clin. Pharmacol. Ther. 2017, 102, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Barreto, E.F.; Rule, A.D.; Voils, S.A.; Kane-Gill, S.L. Innovative Use of Novel Biomarkers to Improve the Safety of Renally Eliminated and Nephrotoxic Medications. Pharmacotherapy 2018, 38, 794–803. [Google Scholar] [CrossRef]
- The Kidney Disease: Improving Global Outcomes. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. Suppl. 2013, 3, 1–150.
- Inker, L.A.; Schmid, C.H.; Tighiouart, H.; Eckfeldt, J.H.; Feldman, H.I.; Greene, T.; Kusek, J.W.; Manzi, J.; Van Lente, F.; Zhang, Y.L.; et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N. Engl. J. Med. 2012, 367, 20–29. [Google Scholar] [CrossRef]
- Stevens, L.A.; Schmid, C.H.; Greene, T.; Li, L.; Beck, G.J.; Joffe, M.M.; Froissart, M.; Kusek, J.W.; Zhang, Y.; Coresh, J.; et al. Factors other than glomerular filtration rate affect serum cystatin C levels. Kidney Int. 2009, 75, 652–660. [Google Scholar] [CrossRef]
- Ye, Y.; Gai, X.; Xie, H.; Jiao, L.; Zhang, S. Impact of Thyroid Function on Serum Cystatin C and Estimated Glomerular Filtration Rate: A Cross-Sectional Study. Endocr. Pract. 2013, 19, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Knight, E.L.; Verhave, J.C.; Spiegelman, D.; Hillege, H.L.; De Zeeuw, D.; Curhan, G.C.; De Jong, P.E. Factors influencing serum cystatin C levels other than renal function and the impact on renal function measurement. Kidney Int. 2004, 65, 1416–1421. [Google Scholar] [CrossRef] [PubMed]
- Luc, G.; Bard, J.-M.; Lesueur, C.; Arveiler, D.; Evans, A.; Amouyel, P.; Ferrieres, J.; Juhan-Vague, I.; Fruchart, J.-C.; Ducimetiere, P. Plasma cystatin-C and development of coronary heart disease: The PRIME Study. Atherosclerosis 2006, 185, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.Q.; Nolin, T.D. Pragmatic Use of Kidney Function Estimates for Drug Dosing: The Tide Is Turning. Adv. Chronic Kidney Dis. 2018, 25, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Cockcroft, D.W.; Gault, M.H. Prediction of creatinine clearance from serum creatinine. Nephron 1976, 16, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Modification of Diet in Renal Disease Study Group. The Modification of Diet in Renal Disease Study: Design, Methods, and Results From the Feasibility Study. Am. J. Kidney Dis. 1992, 20, 18–33. [Google Scholar] [CrossRef]
- Levey, A.S.; Coresh, J.; Greene, T.; Stevens, L.A.; Zhang, Y.L.; Hendriksen, S.; Kusek, J.W.; Van Lente, F. Chronic Kidney Disease Epidemiology Collaboration Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann. Intern. Med. 2006, 145, 247–254. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Iii, A.F.C.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Mathisen, U.D.; Melsom, T.; Ingebretsen, O.C.; Jenssen, T.; Njolstad, I.; Solbu, M.D.; Toft, I.; Eriksen, B.O. Estimated GFR associates with cardiovascular risk factors independently of measured GFR. J. Am. Soc. Nephrol. 2011, 22, 927–937. [Google Scholar] [CrossRef]
- Glassock, R.J.; Rule, A.D. Optimally predicting mortality with kidney function markers is not the same as optimally determining how kidney function predicts mortality. Nephrol. Dial. Transplant. 2017, 32, 585–587. [Google Scholar] [CrossRef]
- US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Draft Guidance for Industry: Pharmacokinetics in Patients with Impaired Renal Function—Study Design, Data Analysis, and Impact on Dosing and Labeling; CDER: Silver Springs, MD, USA, 2003; pp. 1–19. [Google Scholar]
- Stevens, L.A.; Nolin, T.D.; Richardson, M.M.; Feldman, H.I.; Lewis, J.B.; Rodby, R.; Townsend, R.; Okparavero, A.; Zhang, Y.; Schmid, C.H.; et al. Comparison of Drug Dosing Recommendations Based on Measured GFR and Kidney Function Estimating Equations. Am. J. Kidney Dis. 2009, 54, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Peters, B.J.; Rule, A.D.; Kashani, K.B.; Lieske, J.C.; Mara, K.C.; Dierkhising, R.A.; Barreto, E.F. Impact of Serum Cystatin C–Based Glomerular Filtration Rate Estimates on Drug Dose Selection in Hospitalized Patients. Pharmacotherapy 2018, 38, 1068–1073. [Google Scholar] [CrossRef] [PubMed]
- Barreto, E.F.; Rule, A.D.; Murad, M.H.; Kashani, K.B.; Lieske, J.C.; Erwin, P.J.; Steckelberg, J.M.; Gajic, O.; Reid, J.M.; Kane-Gill, S.L. Prediction of the Renal Elimination of Drugs With Cystatin C vs. Creatinine: A Systematic Review. Mayo Clin Proc. 2019, 94, 500–514. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Feng, J.F.; Li, B.; Zhang, L.; Yang, Y.W. Estimation of safe and effective dose of vancomycin in MRSA-infected patients using serum cystatin C concentrations. Int. J. Clin. Pharmacol. Ther. 2013, 51, 161–169. [Google Scholar] [CrossRef]
- Chung, J.-Y.Y.; Jin, S.-J.J.; Yoon, J.-H.H.; Song, Y.-G.G. Serum cystatin C is a major predictor of vancomycin clearance in a population pharmacokinetic analysis of patients with normal serum creatinine concentrations. J. Korean Med. Sci. 2013, 28, 48–54. [Google Scholar] [CrossRef]
- DeCarolis, D.D.; Thorson, J.G.; Marraffa, R.A.; Clairmont, M.A.; Kuskowski, M.A. Comparison of Equations With Estimate Renal Function to Predict Serum Vancomycin Concentration in Patients With Spinal Cord Injury—Does the Use of Cystatin C Improve Accuracy? Ther. Drug Monit. 2014, 36, 632–639. [Google Scholar] [CrossRef]
- Frazee, E.; Rule, A.D.; Lieske, J.C.; Kashani, K.B.; Barreto, J.N.; Virk, A.; Kuper, P.J.; Dierkhising, R.A.; Leung, N. Cystatin C–Guided Vancomycin Dosing in Critically Ill Patients: A Quality Improvement Project. Am. J. Kidney Dis. 2017, 69, 658–666. [Google Scholar] [CrossRef]
- Frazee, E.N.; Rule, A.D.; Hermann, S.M.; Kashani, K.B.; Leung, N.; Virk, A.; Voskoboev, N.; Lieske, J.C.; Herrmann, S.M.; Kashani, K.B.; et al. Serum cystatin C predicts vancomycin trough levels better than serum creatinine in hospitalized patients: A cohort study. Crit. Care 2014, 18, R110. [Google Scholar] [CrossRef]
- Hermida, J.; Tutor, J.C. Serum cystatin C for the prediction of glomerular filtration rate with regard to the dose adjustment of amikacin, gentamicin, tobramycin, and vancomycin. Ther. Drug Monit. 2006, 28, 326–331. [Google Scholar] [CrossRef]
- Kees, M.G.; Hilpert, J.W.; Gnewuch, C.; Kees, F.; Voegeler, S. Clearance of vancomycin during continuous infusion in Intensive Care Unit patients: Correlation with measured and estimated creatinine clearance and serum cystatin C. Int. J. Antimicrob. Agents 2010, 36, 545–548. [Google Scholar] [CrossRef]
- Okamoto, G.; Sakamoto, T.; Kimura, M.; Ukishima, Y.; Sonoda, A.; Mori, N.; Kato, Y.; Maeda, T.; Kagawa, Y. Serum cystatin C as a better marker of vancomycin clearance than serum creatinine in elderly patients. Clin. Biochem. 2007, 40, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Imanishi, Y.; Nakano, S.; Niwa, T.; Ohmori, T.; Shirai, K.; Yoshida, S.; Furuta, N.; Takemura, M.; Ito, H.; et al. Usefulness of serum cystatin C to determine the dose of vancomycin in critically ill patients. J. Pharm. Pharmacol. 2010, 62, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Suemaru, K.; Otsuka, T.; Ido, K.; Nishimiya, T.; Sakai, I.; Hasegawa, H.; Inoue, T.; Murase, M.; Yasukawa, M.; et al. Estimation of the initial dose setting of vancomycin therapy with use of cystatin C as a new marker of renal function. Ther. Drug Monit. 2007, 29, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Aiba, T.; Otsuka, T.; Suemaru, K.; Nishimiya, T.; Inoue, T.; Murase, M.; Kurosaki, Y.; Araki, H. Population pharmacokinetic analysis of vancomycin using serum cystatin C as a marker of renal function. Antimicrob. Agents Chemother. 2010, 54, 778–782. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-T.; Pang, H.-M.; Jing, L.; Wei, W.-X.; Qin, X.-L.; Guo, Q.; Lu, H.; Cheng, D.-H.; Jiang, W.-Z. A population pharmacokinetic model of vancomycin for dose individualization based on serum cystatin C as a marker of renal function. J. Pharm. Pharmacol. 2019, 71, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Kasai, H.; Tsuji, Y.; Hiraki, Y.; Tsuruyama, M.; To, H.; Yamamoto, Y. Population pharmacokinetics of teicoplanin in hospitalized elderly patients using cystatin C as an indicator of renal function. J. Infect. Chemother. 2018, 24, 284–291. [Google Scholar] [CrossRef]
- Jing, L.; Liu, T.; Guo, Q.; Chen, M.; Lu, J.; Lv, C. Development and comparison of population pharmacokinetic models of vancomycin in neurosurgical patients based on two different renal function markers. J. Clin. Pharm. Ther. 2020, 45, 88–96. [Google Scholar] [CrossRef]
- Pricker, M.; Wiesli, P.; Brändle, M.; Schwegler, B.; Schmid, C. Impact of thyroid dysfunction on serum cystatin C. Kidney Int. 2003, 63, 1944–1947. [Google Scholar] [CrossRef]
- Wiesli, P.; Schwegler, B.; Spinas, G.A.; Schmid, C. Serum cystatin C is sensitive to small changes in thyroid function. Clin. Chim. Acta 2003, 338, 87–90. [Google Scholar] [CrossRef]
- Pai, M.P. Estimating the Glomerular Filtration Rate in Obese Adult Patients for Drug Dosing. Adv. Chronic Kidney Dis. 2010, 17, e53–e62. [Google Scholar] [CrossRef]
- Chang, A.R.; Zafar, W.; Grams, M.E. Kidney Function in Obesity—Challenges in Indexing and Estimation. Adv. Chronic Kidney Dis. 2018, 25, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Prado, R.; Castillo-Rodriguez, E.; Velez-Arribas, F.J.; Gracia-Iguacel, C.; Ortiz, A. Creatinine Clearance Is Not Equal to Glomerular Filtration Rate and Cockcroft-Gault Equation Is Not Equal to CKD-EPI Collaboration Equation. Am. J. Med. 2016, 129, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, J.F.; Möller, E.; Van Slyke, D.D. Studies of urea excretion. III: The Influence of Body Size on Urea Output. J. Clin. Invest. 1928, 6, 467–483. [Google Scholar] [CrossRef] [PubMed]
- Dooley, M.J.; Poole, S.G. Poor correlation between body surface area and glomerular filtration rate. Cancer Chemother. Pharmacol. 2000, 46, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Redal-Baigorri, B.; Rasmussen, K.; Heaf, J.G. Indexing glomerular filtration rate to body surface area: Clinical consequences. J. Clin. Lab. Anal. 2014, 28, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Delanaye, P.; Mariat, C.; Cavalier, E.; Krzesinski, J.-M. Errors induced by indexing glomerular filtration rate for body surface area: Reductio ad absurdum. Nephrol. Dial. Transplant. 2009, 24, 3593–3596. [Google Scholar] [CrossRef]
- Geddes, C.C.; Woo, Y.M.; Brady, S. Glomerular filtration rate—What is the rationale and justification of normalizing GFR for body surface area? Nephrol. Dial. Transplant. 2008, 23, 4–6. [Google Scholar] [CrossRef]
- Kashani, K.B.; Frazee, E.N.; Kukrálová, L.; Sarvottam, K.; Herasevich, V.; Young, P.M.; Kashyap, R.; Lieske, J.C. Evaluating Muscle Mass by Using Markers of Kidney Function: Development of the Sarcopenia Index. Crit. Care Med. 2016, 45, 1–7. [Google Scholar] [CrossRef]
- Mindikoglu, A.L.; Opekun, A.R.; Mitch, W.E.; Magder, L.S.; Christenson, R.H.; Dowling, T.C.; Weir, M.R.; Seliger, S.L.; Howell, C.D.; Raufman, J.P.; et al. Cystatin C Is a Gender-Neutral Glomerular Filtration Rate Biomarker in Patients with Cirrhosis. Dig. Dis. Sci. 2018, 63, 665–675. [Google Scholar] [CrossRef]
- Tetsuka, S.; Morita, M.; Ikeguchi, K.; Nakano, I. Creatinine/cystatin C ratio as a surrogate marker of residual muscle mass in amyotrophic lateral sclerosis. Neurol. Clin. Neurosci. 2013, 1, 32–37. [Google Scholar] [CrossRef]
- Braat, E.; Hoste, L.; De Waele, L.; Gheysens, O.; Vermeersch, P.; Goffin, K.; Pottel, H.; Goemans, N.; Levtchenko, E. Renal function in children and adolescents with Duchenne muscular dystrophy. Neuromuscul. Disord. 2015, 25, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Beringer, P.M.; Hidayat, L.; Heed, A.; Zheng, L.; Owens, H.; Benitez, D.; Rao, A.P. GFR estimates using cystatin C are superior to serum creatinine in adult patients with cystic fibrosis. J. Cyst. Fibros. 2009, 8, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Erlandsen, E.J.; Hansen, R.M.; Randers, E.; Petersen, L.E.; Abrahamsen, J.; Johannesen, I.L. Estimating the glomerular filtration rate using serum cystatin C levels in patients with spinal cord injuries. Spinal Cord 2012, 50, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Thurlow, J.S.; Abbott, K.C.; Linberg, A.; Little, D.; Fenderson, J.; Olson, S.W. SCr and SCysC concentrations before and after traumatic amputation in male soldiers: A case-control study. Am. J. Kidney Dis. 2014, 63, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Nejat, M.; Pickering, J.W.; Walker, R.J.; Endre, Z.H. Rapid detection of acute kidney injury by plasma cystatin C in the intensive care unit. Nephrol. Dial. Transplant. 2010, 25, 3283–3289. [Google Scholar] [CrossRef] [PubMed]
- Aakjær, M.; Houlind, M.; Treldal, C.; Ankarfeldt, M.S.; Jensen, P.; Andersen, O.; Iversen, E.; Christrup, L.; Petersen, J. Differences in Kidney Function Estimates Based on Creatinine and/or Cystatin C in Non-Traumatic Amputation Patients and Their Impact on Drug Prescribing. J. Clin. Med. 2019, 8, 89. [Google Scholar] [CrossRef] [PubMed]
- Yamawaki, C.; Takahashi, M.; Takara, K.; Kume, M.; Hirai, M.; Yasui, H.; Nakamura, T. Effect of dexamethasone on extracellular secretion of cystatin C in cancer cell lines. Biomed. Rep. 2013, 1, 115–118. [Google Scholar] [CrossRef]
- Pianta, T.J.; Pickering, J.W.; Succar, L.; Chin, M.; Davidson, T.; Buckley, N.A.; Mohamed, F.; Endre, Z.H. Dexamethasone modifies cystatin c-based diagnosis of acute kidney injury during cisplatin-based chemotherapy. Kidney Blood Press. Res. 2017, 42, 62–75. [Google Scholar] [CrossRef]
- He, L.; Li, J.; Zhan, J.; Yi, F.; Fan, X.; Wei, Y.; Zhang, W. The value of serum cystatin C in early evaluation of renal insufficiency in patients undergoing chemotherapy: A systematic review and meta-analysis. Cancer Chemother. Pharmacol. 2019, 83, 561–571. [Google Scholar] [CrossRef]
- Hammouda, N.E.; Salah El-Din, M.A.; El-Shishtawy, M.M.; El-Gayar, A.M. Serum Cystatin C as a Biomarker in Diffuse Large B-Cell Lymphoma. Sci. Pharm. 2017, 85, 9. [Google Scholar] [CrossRef]
- Mulaomerović, A.; Halilbašić, A.; Čičkušić, E.; Zavašnik-Bergant, T.; Begić, L.; Kos, J. Cystatin C as a potential marker for relapse in patients with non-Hodgkin B-cell lymphoma. Cancer Lett. 2007, 248, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Christoulas, D.; Kastritis, E.; Katodritou, E.; Pouli, A.; Michalis, E.; Papassotiriou, I.; Dimopoulos, M.A. The chronic kidney disease epidemiology collaboration cystatin C (CKD-EPI-CysC) equation has an independent prognostic value for overall survival in newly diagnosed patients with symptomatic multiple myeloma; is it time to change from MDRD to CKD-EPI-CysC. Eur. J. Haematol. 2013, 91, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Kos, J.; Krasovec, M.; Cimerman, N.; Nielsen, H.J.; Christensen, I.J.; Brünner, N. Cysteine proteinase inhibitors stefin A, stefin B, and cystatin C in sera from patients with colorectal cancer: Relation to prognosis. Clin. Cancer Res. 2000, 6, 505–511. [Google Scholar] [PubMed]
- Kos, J.; Štabuc, B.; Schweiger, A.; Krašovec, M.; Cimerman, N.; Kopitar-Jerala, N.; Vrhovec, I. Cathepsins B, H, and L and their inhibitors stefin A and cystatin C in sera of melanoma patients. Clin. Cancer Res. 1997, 3, 1815–1822. [Google Scholar] [PubMed]
- Nückel, H.; Langer, C.; Herget-Rosenthal, S.; Wichert, M.; Assert, R.; Döhner, H.; Dührsen, U.; Liebisch, P. Prognostic significance of serum cystatin C in multiple myeloma. Int. J. Hematol. 2012, 95, 545–550. [Google Scholar] [CrossRef]
- Rajkumar, S.V. Multiple myeloma: 2018 update on diagnosis, risk-stratification, and management. Am. J. Hematol. 2018, 93, 981–1114. [Google Scholar] [CrossRef]
- Terpos, E.; Katodritou, E.; Tsiftsakis, E.; Kastritis, E.; Christoulas, D.; Pouli, A.; Michalis, E.; Verrou, E.; Anargyrou, K.; Tsionos, K.; et al. Cystatin-C is an independent prognostic factor for survival in multiple myeloma and is reduced by bortezomib administration. Haematologica 2009, 94, 372–379. [Google Scholar] [CrossRef]
- Thomas, F.; Seronie-Vivien, S.; Gladieff, L.; Dalenc, F.; Durrand, V.; Malard, L.; Lafont, T.; Poublanc, M.; Bugat, R.; Chatelut, E. Cystatin C as a new covariate to predict renal elimination of drugs: Application to carboplatin. Clin. Pharmacokinet. 2005, 44, 1305–1316. [Google Scholar] [CrossRef]
- Shibata, K.; Yasuda, Y.; Kobayashi, R.; Ando, Y.; Shimokata, T.; Kamiya, H.; Hayashi, M.; Maruyama, S.; Matsuo, S.; Nakao, M.; et al. Renal function evaluation in patients with cancer who were scheduled to receive carboplatin or S-1. Clin. Exp. Nephrol. 2015, 19, 1107–1113. [Google Scholar] [CrossRef]
- Cavalcanti, E.; Barchiesi, V.; Cerasuolo, D.; Di Paola, F.; Cantile, M.; Cecere, S.C.; Pignata, S.; Morabito, A.; Costanzo, R.; Di Maio, M.; et al. Correlation of Serum Cystatin C with Glomerular Filtration Rate in Patients Receiving Platinum-Based Chemotherapy. Anal. Cell. Pathol. 2016. [Google Scholar] [CrossRef]
- Bretagne, M.; Jouinot, A.; Durand, J.P.; Huillard, O.; Boudou Rouquette, P.; Tlemsani, C.; Arrondeau, J.; Sarfati, G.; Goldwasser, F.; Alexandre, J. Estimation of glomerular filtration rate in cancer patients with abnormal body composition and relation with carboplatin toxicity. Cancer Chemother. Pharmacol. 2017, 80, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Ylinen, E.; Jahnukainen, K.; Saarinen-Pihkala, U.M.; Jahnukainen, T. Assessment of renal function during high-dose methotrexate treatment in children with acute lymphoblastic leukemia. Pediatr. Blood Cancer 2014, 61, 2199–2202. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Seronie-Vivien, S.; Thomas, F.; Delord, J.P.; Malard, L.; Canal, P.; Chatelut, E.; Séronie-Vivien, S.; Thomas, F.; Delord, J.P.; et al. Serum cystatin C is a better marker of topotecan clearance than serum creatinine. Clin. Cancer Res. 2005, 11, 3038–3044. [Google Scholar] [CrossRef] [PubMed]
- Barreto, J.N.; McClanahan, A.L.; Rule, A.D.; Thompson, C.A.; Frazee, E. Incorporating Cystatin C to Predict Methotrexate Elimination in Patients with CNS Lymphoma and Suspicious Renal Function. Case Rep. Hematol. 2018. [Google Scholar] [CrossRef]
- Chew-Harris, J.S.C.; Florkowski, C.M.; George, P.M.; Endre, Z.H. Comparative performances of the new chronic kidney disease epidemiology equations incorporating cystatin C for use in cancer patients. Asia Pac. J. Clin. Oncol. 2015, 11, 142–151. [Google Scholar] [CrossRef]
- Zhu, X.-R.; Ge, N.; Wang, Y.; Zhai, J.-L.; Liu, C. Corticosteroids significantly increase cystatin C levels in the plasma by promoting cystatin C production in rats. Ren. Fail. 2019, 41, 698–703. [Google Scholar] [CrossRef]
- Bjarnadóttir, M.; Grubb, A.; Ólafsson, Í. Promoter-mediated, dexamethasone-induced increase in cystatin c production by hela cells. Scand. J. Clin. Lab. Invest. 1995, 55, 617–623. [Google Scholar] [CrossRef]
- Manetti, L.; Genovesi, M.; Pardini, E.; Grasso, L.; Lupi, I.; Morselli, L.L.; Pellegrini, G.; Martino, E. Early effects of methylprednisolone infusion on serum cystatin C in patients with severe Graves’ ophthalmopathy [2]. Clin. Chim. Acta 2005, 356, 227–228. [Google Scholar] [CrossRef]
- Cimerman, N.; Brguljan, P.M.; Krašovec, M.; Šuškovič, S.; Kos, J. Serum cystatin C, a potent inhibitor of cysteine proteinases, is elevated in asthmatic patients. Clin. Chim. Acta 2000, 300, 83–95. [Google Scholar] [CrossRef]
- Zhai, J.-L.L.; Ge, N.; Zhen, Y.; Zhao, Q.; Liu, C. Corticosteroids significantly increase serum cystatin c concentration without affecting renal function in symptomatic heart failure. Clin. Lab. 2016, 62, 203–207. [Google Scholar] [CrossRef]
- Foster, J.; Reisman, W.; Lepage, N.; Filler, G. Influence of commonly used drugs on the accuracy of cystatin C-derived glomerular filtration rate. Pediatr. Nephrol. 2006, 21, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Bardi, E.; Dobos, É.; Kappelmayer, J.; Kiss, C. Differential effect of corticosteroids on serum cystatin C in thrombocytopenic purpura and leukemia. Pathol. Oncol. Res. 2010, 16, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Bokenkamp, A.; Laarman, C.; Braam, K.; van Wijk, J.A.E.; Kors, W.A.; Kool, M.; de Valk, J.; Bouman, A.A.; Spreeuwenberg, M.; Stoffel-Wagner, B. Effect of Corticosteroid Therapy on Low-Molecular–Weight Protein Markers of Kidney Function. Clin. Chem. 2007, 53, 2219–2221. [Google Scholar] [CrossRef] [PubMed]
- Slort, P.R.; Ozden, N.; Pape, L.; Offner, G.; Tromp, W.F.; Wilhelm, A.J.; Bokenkamp, A. Comparing cystatin C and creatinine in the diagnosis of pediatric acute renal allograft dysfunction. Pediatr. Nephrol. 2012, 27, 843–849. [Google Scholar] [CrossRef]
- Nozawa, Y.; Sato, H.; Wakamatsu, A.; Kobayashi, D.; Nakatsue, T.; Wada, Y.; Kuroda, T.; Kazama, J.J.; Suzuki, Y.; Nakano, M.; et al. Utility of estimated glomerular filtration rate using cystatin C and its interpretation in patients with rheumatoid arthritis under glucocorticoid therapy. Clin. Chim. Acta 2018, 487, 299–305. [Google Scholar] [CrossRef]
- Chin, P.K.L.; Chew-Harris, J.S.C.; Florkowski, C.M.; Begg, E.J. The performance of contemporary cystatin C-based GFR equations in predicting gentamicin clearance. Br. J. Clin. Pharmacol. 2015, 79, 268–277. [Google Scholar] [CrossRef]
- Kashani, K.; Sarvottam, K.; Pereira, N.L.; Barreto, E.F.; Kennedy, C.C. The Sarcopenia Index: A Novel Measure of Muscle Mass in Lung Transplant Candidates. Clin. Transplant. 2018, 32, e13182. [Google Scholar] [CrossRef]
- Rule, A.D.; Bergstralh, E.J.; Slezak, J.M.; Bergert, J.; Larson, T.S. Glomerular filtration rate estimated by cystatin C among different clinical presentations. Kidney Int. 2006, 69, 399–405. [Google Scholar] [CrossRef]
- Pöge, U.; Gerhardt, T.; Bökenkamp, A.; Stoffel-Wagner, B.; Klehr, H.U.; Sauerbruch, T.; Woitas, R.P. Time course of low molecular weight proteins in the early kidney transplantation period—Influence of corticosteroids. Nephrol. Dial. Transplant. 2004, 19, 2858–2863. [Google Scholar] [CrossRef]
- Geramizadeh, B.; Azarpira, N.; Ayatollahi, M.; Rais-Jalali, G.-A.; Aghdai, M.; Yaghoobi, R.; Banihashemi, M.; Malekpour, Z.; Malek-Hosseini, S.A. Value of serum cystatin C as a marker of renal function in the early post kidney transplant period. Saudi J. Kidney Dis. Transpl. 2009, 20, 1015–1017. [Google Scholar]
- Lezaic, V.; Dajak, M.; Radivojevic, D.; Ristic, S.; Marinkovic, J. Cystatin C and serum creatinine as predictors of kidney graft outcome. Int. Urol. Nephrol. 2014, 46, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, E.; Ruiz, J.C.; Fernández-Fresnedo, G.; Fernández, M.D.; Piñera, C.; Palomar, R.; Monfá, E.; Gómez-Alamillo, C.; Arias, M. Cystatin C and albuminuria as predictors of long-term allograft outcomes in kidney transplant recipients. Clin. Transplant. 2013, 27, E177–E183. [Google Scholar] [CrossRef] [PubMed]
- Le Bricon, T.; Thervet, E.; Froissart, M.; Benlakehal, M.; Bousquet, B.; Legendre, C.; Erlich, D. Plasma cystatin C is superior to 24-h creatinine clearance and plasma creatinine for estimation of glomerular filtration rate 3 months after kidney transplantation [1]. Clin. Chem. 2000, 46, 1206–1207. [Google Scholar] [CrossRef] [PubMed]
- Kukla, A.; Issa, N.; Jackson, S.; Spong, R.; Foster, M.C.; Matas, A.J.; Mauer, M.S.; Eckfeldt, J.H.; Ibrahim, H.N. Cystatin C Enhances Glomerular Filtration Rate Estimating Equations in Kidney Transplant Recipients. Am. J. Nephrol. 2014, 39, 59–65. [Google Scholar] [CrossRef]
- Masson, I.; Maillard, N.; Cavalier, E.; Alamartine, E.; Mariat, C.; Delanaye, P. KDIGO Guidelines and Kidney Transplantation: Is the Cystatin-C Based Recommendation Relevant? Am. J. Transplant. 2015, 15, 2211–2214. [Google Scholar] [CrossRef]
- Harman, G.; Akbari, A.; Hiremath, S.; White, C.A.; Ramsay, T.; Kokolo, M.B.; Craig, J.; Knoll, G.A. Accuracy of cystatin C-based estimates of glomerular filtration rate in kidney transplant recipients: A systematic review. Nephrol. Dial. Transplant. 2013, 28, 741–757. [Google Scholar] [CrossRef]
- Malheiro, J.; Fonseca, I.; Martins, L.S.; Almeida, M.; Pedroso, S.; Dias, L.; Henriques, A.C.; Cabrita, A. A Comparison Between Serum Creatinine and Cystatin C-Based Equations for Estimation of Graft Function. Transplant. Proc. 2012, 44, 2352–2356. [Google Scholar] [CrossRef]
- Tang, Z.; Tao, J.; Sun, L.; Han, Z.; Chen, H.; Huang, Z.; Zhou, W.; Zhao, C.; Fei, S.; Que, H.; et al. Prospective Comparison of Equations Based on Creatinine and Cystatin C for the Glomerular Filtration Rate Estimation in Chinese Renal Transplant Recipients. Transplant. Proc. 2018, 50, 85–91. [Google Scholar] [CrossRef]
- White, C.; Akbari, A.; Hussain, N.; Dinh, L.; Filler, G.; Lepage, N.; Knoll, G.A. Estimating glomerular filtration rate in kidney transplantation: A comparison between serum creatinine and cystatin C-based methods. J. Am. Soc. Nephrol. 2005, 16, 3763–3770. [Google Scholar] [CrossRef]
- Tsujita, M.; Goto, N.; Yamamoto, T.; Hiramitsu, T.; Nanmoku, K.; Inaguma, D.; Takeda, A.; Kobayashi, T.; Tominaga, Y.; Morozumi, K.; et al. How to estimate kidney function in kidney transplant recipients with mild to moderate kidney impairment: Comparison of estimated glomerular filtration (eGFR) values between creatinine-based GFR equations and cystatin C-based GFR equations for Japanese pop. Clin. Exp. Nephrol. 2014, 18, 130–134. [Google Scholar] [CrossRef]
- Torre, A.; Aguirre-Valadez, J.M.; Arreola-Guerra, J.M.; García-Flores, O.R.; García-Juárez, I.; Cruz-Rivera, C.; Correa-Rotter, R.; Niño-Cruz, J.A. Creatinine Versus Cystatin C for Estimating GFR in Patients with Liver Cirrhosis. Am. J. Kidney Dis. 2016, 67, 342–344. [Google Scholar] [CrossRef] [PubMed]
- Krones, E.; Fickert, P.; Zitta, S.; Neunherz, S.; Artinger, K.; Reibnegger, G.; Durchschein, F.; Wagner, D.; Stojakovic, T.; Stadlbauer, V.; et al. The chronic kidney disease epidemiology collaboration equation combining creatinine and cystatin C accurately assesses renal function in patients with cirrhosis. BMC Nephrol. 2015, 16, 196. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wu, T.; Zhang, W.; Zhang, S.; Wang, W. Pretransplant renal function evaluated by serum cystatin C was associated with mortality after liver transplantation: A single-center experience. Ann. Transl. Med. 2019, 7, 243. [Google Scholar] [CrossRef] [PubMed]
- Bernal, V.; Pascual, I.; Piazuelo, E.; Esquivias, P.; Fernandez, C.; Garcia-Gil, F.A.; Simon, M.A. Cystatin C level as a predictor of death and cardiovascular events after liver transplantation. Transplant. Proc. 2011, 43, 732–734. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.M.; Moon, Y.J.; Jung, K.W.; Jun, I.G.; Song, J.G.; Hwang, G.S. Pretransplantation Cystatin C, but not Creatinine, Predicts 30-day Cardiovascular Events and Mortality in Liver Transplant Recipients With Normal Serum Creatinine Levels. Transplant. Proc. 2018, 50, 1094–1099. [Google Scholar] [CrossRef]
- Uguen, T.; Jezequel, C.; Ropert, M.; Houssel-Debry, P.; Latournerie, M.; Lainé, F.; Deugnier, Y.; Vigneau, C.; Boudjema, K.; Guyader, D.; et al. Pretransplant renal function according to CKD-EPI cystatin C equation is a prognostic factor of death after liver transplantation. Liver Int. 2016, 36, 547–554. [Google Scholar] [CrossRef]
- Hošková, L.; Franeková, J.; Málek, I.; Kautzner, J.; Szárszoi, O.; Jabor, A.; Pinďák, M.; Viklický, O.; Melenovský, V. Comparison of cystatin C and NGAL in early diagnosis of acute kidney injury after heart transplantation. Ann. Transplant. 2016, 21, 239–245. [Google Scholar] [CrossRef]
- Degen, D.A.; Janardan, J.; Barraclough, K.A.; Schneider, H.G.; Barber, T.; Barton, H.; Snell, G.; Levvey, B.; Walker, R.G. Predictive performance of different kidney function estimation equations in lung transplant patients. Clin. Biochem. 2017, 50, 385–393. [Google Scholar] [CrossRef]
- Manolio, T.A.; Chisholm, R.L.; Ozenberger, B.; Roden, D.M.; Williams, M.S.; Wilson, R.; Bick, D.; Bottinger, E.P.; Brilliant, M.H.; Eng, C.; et al. Implementing genomic medicine in the clinic: The future is here. Genet. Med. 2013, 15, 258–267. [Google Scholar] [CrossRef]
- Zhi, M.; Ding, E.L.; Theisen-Toupal, J.; Whelan, J.; Arnaout, R. The landscape of inappropriate laboratory testing: A 15-year meta-analysis. PLoS ONE 2013, 8, e78962. [Google Scholar] [CrossRef]
| Serum Cystatin C Concentration (mg/L) | |||||
|---|---|---|---|---|---|
| Description | 0.6 | 0.9 | 1.2 | 1.5 | 1.8 |
| eGFRcystatinC of a 50-year-old white male | 126 | 93 | 64 | 47 | 37 |
| eGFRcystatinC of a 80-year-old black female | 104 | 77 | 53 | 39 | 31 |
| All eGFR expressed in mL/min/1.73 m2 and calculated from the CKD-EPI eGFRcystatinC equation | |||||
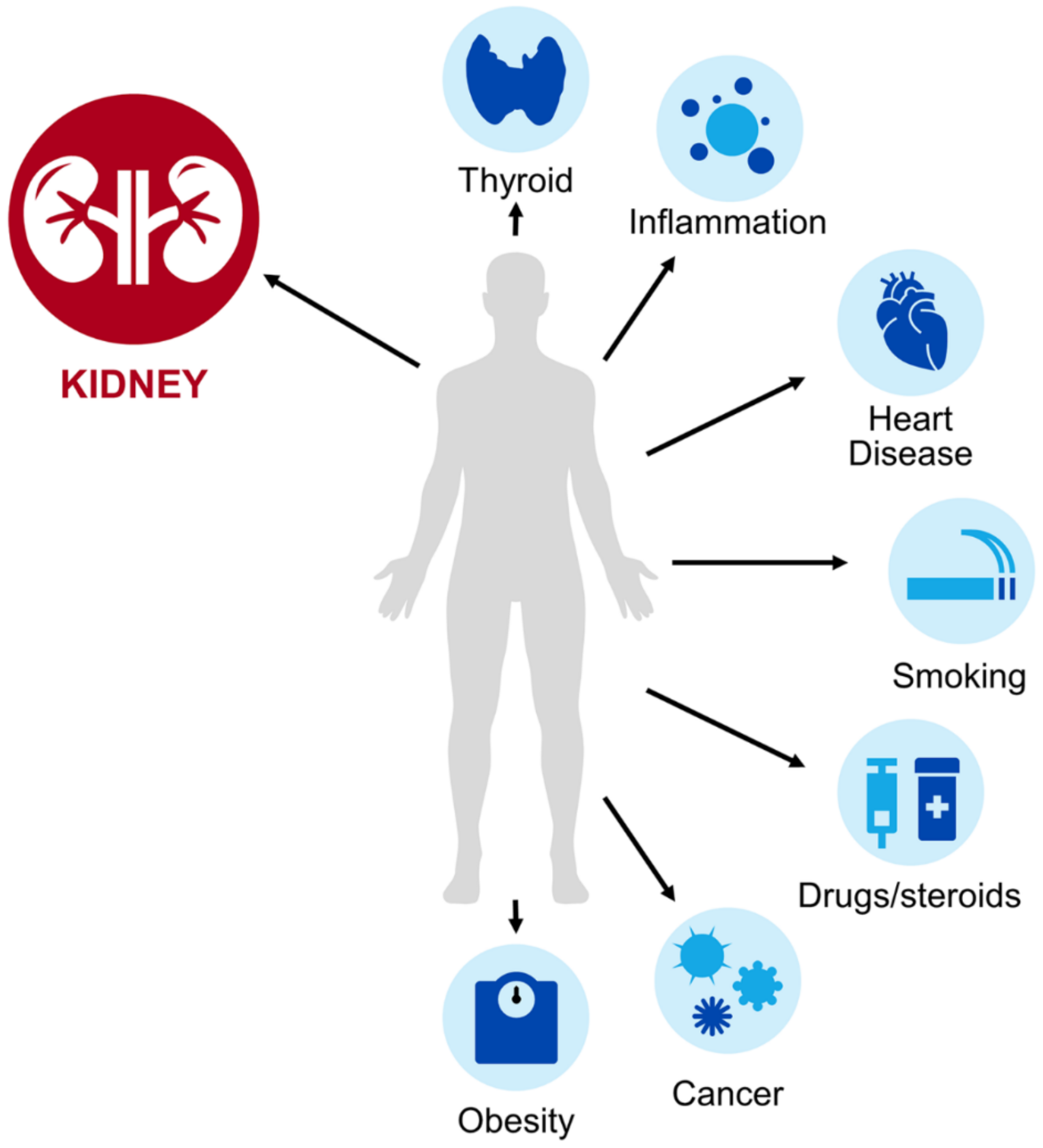
| Population | Effect on Estimation | Considerations |
|---|---|---|
| Obesity [11] | eGFRcysC could underestimate mGFR |
|
| Compromised muscle mass or cachexia [51,52,53,54,55,56,57,58] | eGFRcysC could more accurately predict mGFR |
|
| Malignancy [60,61,62,63,64,65] | eGFRcysC could underestimate mGFR |
|
| Corticosteroids [79,80,81,82,83,84,85,86,87,88] | eGFRcysC could underestimate mGFR |
|
| Solid-organ transplant recipients [92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111] | eGFRcysC could underestimate mGFR |
|
| Cardiovascular disease [11] | eGFRcysC could underestimate mGFR |
|
| Smoking [13] | eGFRcysC could underestimate mGFR |
|
| Thyroid disorder [12,40,41] | Hypothyroid eGFRcysC could overestimate mGFRHyperthyroid eGFRcysC could underestimate mGFR |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teaford, H.R.; Barreto, J.N.; Vollmer, K.J.; Rule, A.D.; Barreto, E.F. Cystatin C: A Primer for Pharmacists. Pharmacy 2020, 8, 35. https://doi.org/10.3390/pharmacy8010035
Teaford HR, Barreto JN, Vollmer KJ, Rule AD, Barreto EF. Cystatin C: A Primer for Pharmacists. Pharmacy. 2020; 8(1):35. https://doi.org/10.3390/pharmacy8010035
Chicago/Turabian StyleTeaford, Hilary R., Jason N. Barreto, Kathryn J. Vollmer, Andrew D. Rule, and Erin F. Barreto. 2020. "Cystatin C: A Primer for Pharmacists" Pharmacy 8, no. 1: 35. https://doi.org/10.3390/pharmacy8010035
APA StyleTeaford, H. R., Barreto, J. N., Vollmer, K. J., Rule, A. D., & Barreto, E. F. (2020). Cystatin C: A Primer for Pharmacists. Pharmacy, 8(1), 35. https://doi.org/10.3390/pharmacy8010035





